Manuscript accepted on :11-06-2025
Published online on: 24-06-2025
Plagiarism Check: Yes
Reviewed by: Dr. Naveen Kumar and Dr. Aybike T
Second Review by: Dr. Moumita Hazra
Final Approval by: Dr. Patorn Piromchai
Yousef Abdel-Kareem Abusamra* , Mohammad Rasool Shomali
, Mohammad Rasool Shomali , Hani Hamad
, Hani Hamad and Abdel Naser Dakkah
and Abdel Naser Dakkah
1Department of Pharmaceutical Sciences, Faculty of Pharmacy, Philadelphia University, Amman, Jordan.
2Department of Clinical Nutrition and Dietetics, Faculty of Allied Medical Sciences, Philadelphia University, Amman, Jordan
Corresponding Author E-mail: yabusamra@philadelphia.edu.jo
DOI : https://dx.doi.org/10.13005/bpj/3184
Abstract
Achillea santolina is an important plant in traditional medicine in Jordan. To utilize the herb throughout the year, the storage process seems to be inevitable. This study investigates the effect of aging for one year on the main active ingredients of the volatile oil and some predominant polar compounds in a methanolic extract of the aerial parts of the plant. Fresh and old samples were evaluated and compared for their antioxidant activity and content of the phenolic and flavonoid compounds. The antioxidant activity of A. santolina was weaker than ascorbic acid (IC50 values for the old, fresh samples, and ascorbic acid: 185.6, 171.4, and 61.3, respectively). Plant storage did not affect the antioxidant capacity and the total phenolic and flavonoid content. Semiquantitative analysis using GC-MS showed that the storage process significantly affected the volatile oil content. Several monoterpenoid and sesquiterpenoid compounds significantly diminished or were even absent in the old sample. Conversely, the content of the polar compounds was not affected by the storage process. The polar compounds derived from azulene, naphthalene, and anthracene dominated the fresh and old samples. The azulene compound achillicin was noticeably abundant in both samples. In conclusion, the volatile oil content seemed to play a pivotal role in the activity of A. santolina, and consequently, storage for one year adversely affected its activity.
Keywords
Achillea santolina; Essential oil; Methanolic extract; Phenolics; Storage; Terpenoids
Download this article as:| Copy the following to cite this article: Abusamra Y. A. K, Shomali M. R, Hamad H, Dakkah A. N. Effect of Aging on the Main Active Ingredients in the Volatile Oil and Crude Methanolic Extract from Achillea santolina. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Abusamra Y. A. K, Shomali M. R, Hamad H, Dakkah A. N. Effect of Aging on the Main Active Ingredients in the Volatile Oil and Crude Methanolic Extract from Achillea santolina. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/3T6g1FB |
Introduction
Achillea (common name yarrow), a genus belonging to the plant family Compositae, comprises about 130 flowering plants that are widely distributed throughout the world, especially in the Mediterranean region. 1,2 These plants are extensively used in traditional medicine for numerous ailments and diseases.3,4 Achillea santolina (AS), a member of this genus, is indigenous to Jordan, where it is considered one of the important plants in traditional medicine. After being extracted by infusion or decoction, the herb is utilized by the users to calm down the stomach, accelerate wound healing, help in lowering the burden of the toxic substances by hastening their elimination, and as a hypoglycemic and antispasmodic.5
Patients utilizing AS mainly as a remedy to aid in controlling diabetes think that the herb is quite helpful when it is used fresh; that is, within several months from the collection. On the other hand, they noticed that AS loses a lot of its activity upon storage for one year or longer due to the low volatile oil content, which is proved by the evanescence of the herb’s aromatic odor. Accordingly, this research investigates the putative changes in the levels of the main ingredients, namely terpenes in the volatile oil and some major polar components present in a methanolic extract of the plant aerial portions. The investigation process mainly comprised a comparison between two samples: fresh and old (aged for one year) in terms of the antioxidant activity, the total phenolic content (TPC), and the total flavonoid content (TFC). Furthermore, the volatile oil from the aerial parts of the fresh and aged herb was sampled by the Clevenger method and analyzed by gas chromatography-mass spectrometry (GC-MS). The crude methanolic extracts from both the fresh and aged herb were sampled with the solid-phase extraction (SPE) technique, treated with n-hexane to eliminate the non-polar compounds, and the remaining material was treated again with methanol:water mixture (1:1) to concentrate the polar compounds. This latter extract was analyzed by GC-MS.
Materials and Methods
Chemicals and reagents
All the chemicals and reagents used in the experiments were of analytical grade. Folin-Ciocalteau reagent was purchased from Carlo Erba (Milano, Italy), Both gallic acid and quercetin were purchased from Santa Cruz Biotechnology (Dallas, USA), Methanol was purchased from Carbon Chemicals Group (Cork, Irland), DPPH was purchased from Merk (Darmstadt, Germany), aluminum chloride and potassium acetate were purchased from Rasayan Laboratories (Gujarat, India), sodium acetate anhydrous was purchased from Leonid chemicals (Pune, India) and ascorbic acid was purchased from B.B.C Chemicals (Bergamo, Italy).
Plant authentication and collection
AS grows in Jordan from north to south, particularly in the regions with white soil. The plant can reach a height of 30 cm, producing multi-leaf complexes. It blooms by late March, giving white flowers that turn yellow at maturity. The herb is recorded in the plant list site,6 and was authenticated by Dr. Khaled Abulaila, Ph.D. (a taxonomist at the National Agricultural Research Center (NARC), Al-Baqah, Amman, Jordan).
The herb was collected early in May when the active ingredients, namely, flavonoids and terpenes from the essential oil, are expected to be at their maximum levels.7 The herb grows extensively in the northern regions of the capital, Amman, particularly in Shafa Badran. The area of Shafa Badran is mountainous and ranges in height from nearly 880 to about 980 meters above sea level (Elevation Map Net). Jordan’s climate is typically Mediterranean; winter is moderately humid, whilst summer is hot and dry. Noteworthy, it is infrequent that the temperature rises above 40 C° in summer and most often remains above 0 C° in winter. About 90 % of Jordan is arid and semi-arid areas.8 November through March is the rainy period, and Western Heights, where the plant was collected, receives the highest average rainfall.
Plant extraction
Kinetic maceration with absolute methanol
After collection, the plant was dried in the shade for two weeks and stored in plastic bags until it was used. To simulate the conditions of how the plant is utilized in traditional medicine, it is deemed fresh when it is used within a few months, not exceeding four months, after collection. Notably, the plant is a perennial herb that loses its aerial parts, including the flowers, within a few months.9 These parts constitute the crude drug used to get the therapeutic effects sought from the herb. Accordingly, storing the dried herb for months and even years is an inevitable task to guarantee the crude drug supply throughout the year. The aged samples were taken from the plant that was stored for one year. The plant samples included the whole herb, excluding the roots. The plant material was then ground using a mill, and the extraction procedure was performed using methanol (absolute) at a ratio of 1:10 (w:v). Methanol was selected for extraction as it was found to be very effective in extracting the polar and non-polar principal ingredients, especially the phenolic compounds, namely flavonoids.10 The method of extraction employed was the kinetic maceration for two days at room temperature. The solvent with the extractives was filtered using a Whatman filter paper. The extract was subsequently subjected to a concentration process by removing the solvent using an evaporator at a temperature of 40 Cº and stored in the freezer till its analysis. The obtained extract was used in all experiments except in the GC-MS analysis of the volatile oil, where Clevenger distillation was used to obtain the volatile oil samples.
Volatile oil extraction by Clevenger method
The Clevenger apparatus with the hydrodistillation process was used to obtain the volatile oil from the aerial portions of the herb.11
Antioxidant evaluation using the DPPH free radical-scavenging activity method
The antioxidant activity of the crude extracts from both fresh and aged herbs was determined using the DPPH (2,2-diphenyl-1-picrylhydrazyl) free radical-scavenging technique.12 The antioxidant effects of both extracts at various concentrations (400, 200, 100, 50, 25, and 12.5 μg/ml) were evaluated in relation to ascorbic acid. To prepare the DPPH solution (0.004 % w/v), 4 mg of DPPH was dissolved in 100 ml of methanol (95 %). It is important to conduct experiments with DPPH in dark environments, as DPPH can be easily decomposed by light, leading to significant inaccuracies in the results due to the light impact on the absorption capacity of DPPH (Ozcelik et al., 2003). A standard solution of ascorbic acid at a concentration of 800 μg/ml was prepared by dissolving 2 mg of ascorbic acid in 2.5 ml of distilled water. This solution was utilized to create a calibration curve by performing serial dilutions to obtain various concentrations of 400, 200, 100, 50, and 25 μg/ml. 4 mg of the extract was dissolved in 10 ml of methanol to create a stock solution (400 μg/ml) of the crude extracts from the fresh and aged plants. Likewise, serial dilutions were performed to get the same concentrations above to plot the calibration curves. The calibration curves of ascorbic acid and the fresh and aged plant crude extracts were used to compare the degree of inhibition of the oxidizing effect of DPPH. DPPH solution was considered as a negative control, whilst methanol was deemed as the blank. After the preparation of the solutions to be used in this experiment, the test tubes were filled with 2 milliliters of the plant extracts’ methanolic solution in varying concentrations. 3 ml of the DPPH methanolic solution was added to each test tube, and the tubes were then allowed to sit at room temperature for half an hour in the dark. This will allow for a chemical reaction between the antioxidants in the extract and DPPH, which will be reduced. The higher the antioxidant effect, the less intense the color of the DPPH solution will be. At 517 nm, the absorbance of the solutions was measured using a UV spectrophotometer (Jenway spectrophotometer, model: 7305, manufactured by Bibby Scientific Ltd, UK) in comparison to the blank. The formula {% I = [(A0 – A1) / A0] X 100} was used to calculate the percentage of inhibition (% I) activity, where A0 represents the absorbance of the control and A1 represents the absorbance of the plant extract or the standard. The half-inhibitory concentration (IC50) values were obtained using the curve equations after the % I values were plotted against the concentrations. The experiment was repeated three times.
Determination of the total phenol content
The total phenol content (TPC) of the fresh and aged plant methanolic crude extracts was determined using the Folin-Ciocalteau method.13 Folin-Ciocalteau reduction by the phenolic compounds of the plant extracts yields a blue color that is measured spectrophotometrically. The intensity of this color is proportional to the phenolic compound content in the extracts. In a beaker, the Folin-Ciocalteau reagent was diluted 10 times by taking 2 ml and adding 18 ml of distilled water. In a 100-ml volumetric flask, 7.5 g of sodium carbonate (Na₂CO₃) was dissolved in distilled water, and then enough distilled water was added to reach the desired limit. Since gallic acid is a common building block of phenolic compounds in plants, it was chosen as a standard. 10 mg of gallic acid was mixed with 10 ml of distilled water to create a stock solution of gallic acid (1 mg/1 ml). From the stock solution, a calibration curve is obtained.was generated by mixing 5 ml of Folin-Ciocalteau with 1 ml of methanol and 1 ml of sodium carbonate. Individually, 10 mg of the crude extracts of both the fresh and aged plants were dissolved in 10 ml of distilled water. In a test tube, 1 ml of this solution was mixed with 2.5 and 3.5 ml of the Folin-Ciocalteau reagent and 7.5% sodium carbonate solutions, respectively. The absorbance at 760 nm was measured against the blank after the test tube was incubated for 20 minutes at room temperature to finish the reaction. The TPC of the fresh and aged samples was calculated using the gallic acid calibration curve and expressed as milligrams of gallic acid equivalent per gram of extract. The experiment was repeated three times.
Determination of the total flavonoid content
The most abundant form of phenolic compounds in plants is flavonoids. Of these, quercetin is regarded as the most common.14 Accordingly, quercetin was used as a standard to measure the total flavonoid content (TFC) in the fresh and aged samples from AS. The TFC was calculated using the aluminum chloride colorimetric technique (Jiao and Wang, 2000). Aluminum chloride forms colored complexes with the hydroxyl groups of the phenolic compounds (mainly flavonoids) that have a maximum absorbance of 420 nm. In a 100-ml volumetric flask, a 10% aluminum chloride solution was prepared by dissolving 10 g of aluminum chloride in distilled water. Likewise, the potassium acetate solution (1 M) was prepared by dissolving 9.8 g in distilled water. The standard solution of quercetin was generated by dissolving 1 g in distilled water to plot a calibration curve, where methanol served as the blank. A stock solution (1000 μg/ml) of the plant extracts of the fresh and aged crude drugs of AS was prepared. Then, the experimental procedure continued as follows: 1.0 ml of each of the plant extract solutions (fresh and old), and the different concentrations of the standard were mixed with 3 ml of methanol and 200 μl of the 10 % potassium chloride solution. Then, 200 μl of the 1 M potassium acetate solution was added to all test tubes. Finally, 5.6 ml of distilled water was added to the tubes, which were left in the dark for 30 minutes at room temperature to allow the reaction to be completed. For all the solutions, the measurement of the absorbance at 420 nm was taken. The TFC, expressed in mg quercetin per weight (g) plant extract, was calculated using the equation {TFC = (C×V) / m}, where C is the quercetin concentration (mg/ml) calculated from the calibration curve, V is the extract volume (ml), and m is the weight (g) of the plant extracts.
Gas chromatography-mass spectrometry analysis of the volatile oil
The GC-MS analysis conditions were as follows: a Shimadzu GC-MS – QP 2010 system was used. The used column was of low polarity (with dimensions 30 m X 0.25 mm, and with a film thickness of 0.25 µm), and manufactured by Macherey Nagel, Duren, Germany. The employed carrier gas was helium, operated at a flow rate of 1 ml/min, achieving a linear velocity of 36.3 cm/ sec. The syringe was rinsed with the solvent once and twice with the sample before injection. The plunger speed in the suction and injection modes was set at high and middle velocities, respectively. The syringe injection mode was normal, with the insertion speed set at high mode. The injection port dwell time was 0.3 sec, and the number of pumping times was 5. The injection was set at the splitless mode, and the sampling time was 1.00 min. The sample injection volume was 2 µL. The starting oven temperature was set at 50 °C for 2 min and then allowed to rise at a rate of 5 Cº/min to reach a maximum temperature (the injection temperature) of 280 °C, at which the system was held for 10 min. The MS detector started operating in the ionization mode at 70 eV at 4 min and ended up at 58 min. For identification and quantification, the full scan mode of the total ion chromatogram (TIC) and selective ion monitoring (SIMs) were used, respectively. The comparison between the peaks in the chromatograms of both samples was semi-quantitative and used the area% values of the compounds and their retention times. The MS system employs the library from the National Institute of Standards and Technologies in the identification of the peaks.
Solid-phase extraction–gas chromatography-mass spectrometry analysis of the major polar compounds in the methanolic extract of the fresh and old samples
Solid-phase extraction was used as a sampling technique coupled with GC-MS to qualitatively determine some chief polar compounds in the methanolic crude extracts from both AS samples. The procedure, illustrated below, aimed to exclude the non-polar compounds and highlight the main polar compounds, including some phenolic compounds. The resultant chromatograms appeared devoid of peak crowding. The SPE cartridges employed included a material with a medium polarity [(LC-Diol, 500 mg, 3 ml), Supelclean, USA]. Preparing the cartridges is of great importance to get high reproducibility-level results. This process, known as preconditioning, was carried out by passing 6 ml of methanol and then 6 ml of n-hexane through an elution machine that operates with a vacuum (Thermo Scientific Manifold). 6 ml of n-hexane was used to dissolve each oil sample, and the resulting solution was then put onto the SPE cartridge that had already been conditioned. For washing the cartridge, a solution composed of 6 ml of n-hexane and 4 ml of n-hexane:ethyl acetate, in a ratio of 85:15, was used. 10 mL of methanol was forced to pass through the cartridge to separate the polar compounds. At 25°C, the separated compounds were dried using nitrogen. The produced dry residue was redissolved in 500 µl of a methanol:water solution in a ratio of 1:1, stirred for one minute, and then filtered through a 0.45-µm syringe filter into a vial. For the determination of the phytochemical profile of both oil samples, the Shimadzu GC–MS–QP 2010 apparatus, mentioned above, was used. The same MS detector with the same specifications was used. In this experiment, the temperatures of the injector and oven were set at 250 Cº and 60 Cº, respectively, for 2 minutes. Afterward, the temperature was at a rate of 3 C/min to a maximum of 280 Cº. The total running time was 70 minutes.
Statistical analysis
Using Microsoft Excel, 2021 (Analysis ToolPak, add-on software), the t-test (one-tail with the assumption of unequal variances) was employed to verify the existence of any significant differences (α = 0.05) between the means of two sets of data; between the results of the fresh and old samples in the DPPH antioxidant activity, TPC and TFC experiments, between the results of the antioxidant activity of the standard (ascorbic acid) and fresh sample, ascorbic acid and the old sample. All experiments were repeated three times.
Results
Antioxidant activity using the DPPH free radical-scavenging activity method
Figure 1 shows the percentage of inhibition-concentration curves of ascorbic acid in the fresh and old samples of AS using the DPPH method.
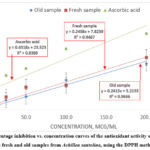 |
Figure 1: Percentage inhibition vs. concentration curves of the antioxidant activity of ascorbic acid, the fresh and old samples from Achillea santolina, using the DPPH method.Click here to view Figure |
Determination of the total phenol content
Figure 2 displays the gallic acid standard curve from which the TPC was determined
for both the fresh and old samples from AS. The TPC is calculated as mg gallic acid in 1 g plant extract.
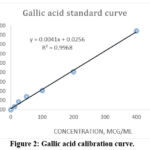 |
Figure 2: Gallic acid calibration curve.Click here to view Figure |
Determination of the total flavonoid content
Figure 3 demonstrates the quercetin standard curve that was used to calculate the TFC in both AS samples. TFC is calculated as the weight of quercetin in mg present in 1 g of the plant extract.
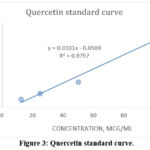 |
Figure 3: Quercetin standard curve.Click here to view Figure |
Gas chromatography-mass spectrometry analysis of the volatile oil of Achillea santolina
Figure 4 (A and B) shows the GC chromatograms of the volatile oil of the fresh and old samples from AS, respectively. The extraction was carried out with the Clevenger distillation technique. The numbered peaks refer to the major compounds which are listed in Table 1.
 |
Figures 4: (A and B). C chromatograms of volatile oil from the fresh and old samples of Achillea santolina, respectively.Click here to view Figure |
Table 1: The major compounds present in the volatile oil from the fresh and old samples of Achillea santolina, their weights, and the existence change in both samples.
| Chromatogram A: GC-MS of the volatile oil from the fresh sample of Achillea santolina | |||
| Peak number | Peak identity | Area % | The mode of change in the two samples |
| 1 | (+)-4-Carene | 4.26 | Noticeably decreased in the old sample |
| 2 | o-Cymene | 4.01 | Noticeably decreased in the old sample |
| 3 | Eucalyptol | 5.27 | Noticeably decreased in the old sample |
| 4 | γ-Terpinene | 9.43 | Noticeably decreased in the old sample |
| 5 | (+)-β-Thujone | 5.95 | Absent in the old sample |
| 6 | L-Camphor | 15.02 | Noticeably decreased in the old sample |
| 7 | Terpinene-4-ol | 8.04 | Noticeably decreased in the old sample |
| 8 | Fragranyl isobutyrate | 9.62 | Noticeably decreased in the old sample |
| 9 | Caryophyllene | 3.13 | Noticeably decreased in the old sample |
| 10 | D-Limonene | 2.88 | Noticeably decreased in the old sample |
| 11 | Spathulenol | 3.76 | Decreased in the old sample |
| 12 | Caryophyllene oxide | 3.27 | Noticeably decreased in the old sample |
| 13 | Oxacyclotetradecan-2-one | 3.27 | Noticeably decreased in the old sample |
| Total | 77.91 | ||
|
Chromatogram B: GC-MS of the volatile oil from the old sample of Achillea santolina |
|||
| 1 | α-Terpinene | 1.86 | Noticeably increased in the fresh sample |
| 2 | 1-Indanone | 2.64 | Absent in the fresh sample |
| 3 | 1-Indanone | 3.18 | Absent in the fresh sample |
| 4 | Eucalyptol | 2.92 | Noticeably increased in the fresh sample |
| 5 | γ-Terpinene | 2.37 | Noticeably increased in the fresh sample |
| 6 | Linalool | 6.23 | Noticeably decreased in the fresh sample |
| 7 | 2-Cyclohexen-1-ol, 1-methyl-4-(1-methylethyl) | 13.09 | Absent in the fresh sample |
| 8 | 2-Cyclohexen-1-ol, 3-methyl-6-(1-methylethyl) | 3.63 | Absent in the fresh sample |
| 9 | Carvone oxide | 4.50 | Noticeably decreased in the fresh sample |
| 10 | Carvomenthone | 2.45 | Noticeably decreased in the fresh sample |
| 11 | Shisool acetate | 2.93 | Absent in the fresh sample |
| 12 | Spathulenol | 2.64 | Increased in the fresh sample |
| 13 | Caryophyllene oxide | 2.67 | Decreased in the fresh sample |
| 14 | Caryophylla-4(12),8(13)-dien-5α-ol | 6.55 | Decreased in the fresh sample |
| 15 | 2-Naphthalenemethanol, decahydro-α,α,4a-trimethyl-8-methylene-, [2R-(2α,4aα,8aβ)] | 5.20 | Absent in the fresh sample |
| Total | 62.86 | ||
| Chromatogram C: SPE-GC-MS: some chief polar compounds from the fresh sample of Achillea santolina | |||
| 1 | Furanone 5-butyldihydro-4-methyl- cis- | 3.44 | Decreased in the old sample |
| 2 | Pyrazine 2-ethyl-3-methyl- | 1.88 | Absent in the old sample |
| 3 | 1-tert-butyl-4-methylpiperidin-3-ol | 1.11 | Absent in the old sample |
| 4 | 1(2H)-Naphthalenone, 3,4-dihydro-4,5,8-trimethyl- | 9.40 | Increased in the old sample, change in the substitution pattern of the methyl groups |
| 5 | Naphthalene, 1,2,3,5,6,7,8,8a-octahydro-1,8a-dimethyl-7-(1-methylethenyl)-, [1R-(1α,7β,8aα)]- | 1.52 | Increased in the fresh sample |
| 6 | 1-methyl-1,2,3,4,5,6,7,8-octahydroanthracene | 3.12 | Increased in the old sample; decreased in the fresh sample |
| 7 | 2-Methyl-1,2,3,4,5,6,7,8-octahydroanthracene | 1.86 | Increased in the old sample; decreased in the fresh sample |
| 8 | Achillicin | 63.24 | Decreased in the old sample |
| 9 | Xanthyletin | 1.76 | Decreased in the old sample |
| Total | 87.33 | ||
|
Chromatogram D: SPE-GC-MS: some chief polar compounds from the old sample of Achillea santolina |
|||
| 1 | 2,6-Dimethyl-3,7-octadiene-2,6-diol | 3.36 | Decreased in the fresh sample |
| 2 | 2,6-Dimethyl-1,7-octadiene-3,6-diol | 5.70 | Decreased in the fresh sample |
| 3 | 1(2H)-Naphthalenone, 3,4-dihydro-4,5,8-trimethyl- | 13.62 | Decreased in the fresh sample, change in the substitution pattern of the methyl groups |
| 4 | Naphthalene, 1,2,3,5,6,7,8,8a-octahydro-1,8a-dimethyl-7-(1-methylethenyl)-, [1R-(1α,7β,8aα)]- | 3.01 | Decreased in the fresh sample |
| 5 | 1-Methyl-1,2,3,4,5,6,7,8-octahydroanthracene | 5.14 | Decreased in the fresh sample |
| 6 | 2-Methyl-1,2,3,4,5,6,7,8-octahydroanthracene | 2.46 | Decreased in the fresh sample |
| 7 | Achillicin | 54.99 | Increased in the fresh sample, but still exists in considerable amounts in the old sample |
| 8 | Xanthyletin | 1.52 | Increased in the fresh sample |
| Total | 89.8 | ||
Solid-phase extraction–gas chromatography-mass spectrometry analysis of the major polar compounds in the methanolic extract of the fresh and old samples
Figure 5 (A and B) demonstrates the GC chromatograms of methanolic crude extracts of the fresh and old samples from Achillea santolina. SPE was used as a sampling technique. The numbered peaks refer to the chief compounds listed in Table 1.
Table 2 summarizes the statistical evaluation of the data obtained from the DPPH antioxidant activity, TPC, and TFC determination.
 |
Figure 5: (A and B). GC chromatograms of methanolic extracts of the fresh and old samples from Achillea santolina, respectively.Click here to view Figure |
Table 2: The results of the statistical evaluation of the data obtained from the DPPH antioxidant activity, TPC, and TFC determination experiments.
| Antioxidant activity –DPPH radical-scavenging method | ||||
| IC50 | Standard deviation | |||
| Ascorbic acid | 61.31 | 0.2227 | ||
| Fresh sample | 171.4 | 0.4637 | ||
| Old sample | 185.63 | 0.8162 | ||
| Statistical test,α –value | p-value | Result | ||
| Ascorbic acid vs. fresh sample | t-test,0.05 | 0.004 | Significant | |
| Ascorbic acid vs. old sample | t-test,0.05 | 0.003 | Significant | |
| Fresh sample vs. old sample | t-test,0.05 | 0.111 | Insignificant | |
| Total phenolic content | ||||
| Average (mg gallic acid / g plant) | Standard deviation | |||
| Fresh sample | 37.80 | 8.41 | ||
| Old sample | 36.88 | 0.94 | ||
| Statistical test,α -value | p-value | Result | ||
| Fresh sample vs. old sample | t-test,0.05 | 0.44 | Insignificant | |
| Total flavonoid content | ||||
| Average (mg quercetin / g plant) | Standard deviation | |||
| Fresh sample | 14.87 | 5.062 | ||
| Old sample | 6.378 | 4.324 | ||
| Statistical test,α -value | p-value | Result | ||
| Fresh sample vs. old sample | t-test,0.05 | 0.054 | Insignificant | |
Discussion
Achillea santolina is one of the commonly used plants in traditional medicine in Jordan. Drying and storing the plant is an indispensable process. The plant appears to lose much of its effectiveness when stored. As a result, this study investigates how storage and aging affect the main components and polar chemicals in the methanolic extract and volatile oil, respectively. It is believed that the targeted active components are essential to the herb’s overall therapeutic benefits.15 The methodology included comparing the antioxidant activity of two samples: fresh and aged, determination of their TPC and TFC, and semiquantitative and qualitative analysis of the main active ingredients present in the volatile oil and some chief constituents found in the methanolic extract of both samples using GC-MS. Table 2 summarises the statistical results obtained from the antioxidant activity, TPC, and TFC determination experiments. Both samples demonstrated weak antioxidant activity compared to vitamin C as evaluated from the t-test (significance level = 95 %). Vitamin C appeared approximately 3 times more potent than either sample. When comparing the antioxidant activity of the two samples as related to each other, the fresh sample slightly and insignificantly surpassed the old one. Accordingly, storing the herb for one year did not appear to weaken the antioxidant effect. The antioxidant activity of plants is mainly ascribed to the phytochemical content of the phenolic compounds, namely flavonoids.16,17 The results of the TPC and TFC experiments agreed with the results regarding antioxidant activity. Likewise, aging the herb by storing it for one year did not seem to affect the phenolic and flavonoid contents in both samples (Table 2). Both samples contained almost the same content of phenolic compounds, but regarding flavonoids, the fresh sample contained almost twice the quantity in the old sample; nevertheless, the difference is still insignificant (p < α, α = 0.05). The flavonoid content seemed to be more noticeably affected by the herb aging than did the other phenolic compounds, yet this effect is not significant. Figure 4 (A and B) shows the GC chromatograms of the volatile oil of the fresh and old samples, respectively. Clevenger distillation was used as a sampling method. The principal peaks in the two chromatograms were numbered and listed in Table 1. The selected peaks account for 77.9% and 62.9% of all the peak weights in chromatograms A and B, respectively. The peaks in chromatogram A are fewer and well resolved, and they seem to represent terpene compounds that are commonly found in the volatile oils of aromatic plants. On the other hand, chromatogram B is noticeably serried and compact with many overlapping peaks. Chromatogram B is dominated by many peaks representing hydrocarbons like cyclohexane, which forms the skeleton of many terpenes. It seems that the main peaks in chromatogram A represent compounds that have been oxidized during the storage process, such as caryophyllene and carvone, and many have undergone decomposition to the hydrocarbon precursors that appear as overlapping peaks in chromatogram B. Most of these peaks were not numbered or listed in Table 1 because of their minute weights in the volatile oil. We estimate that one-year storage massively affected the volatile oil terpene content by depleting the levels of the major terpenes (monoterpenes and sesquiterpenes). The following compounds were found to compose most of the fresh AS volatile oil: (+)-4-carene, o-cymene, eucalyptol, γ-terpinene, (+)-β-thujone, L-camphor, terpinene-4-ol, fragranyl isobutyrate, caryophyllene, D-limonene, spathulenol, caryophyllene oxide, and oxacyclotetradecan-2-one. These compounds noticeably decreased in the old sample or were even absent. On the other hand, 1-indanone, linalool, 2-cyclohexen-1-ol, 1-methyl-4-(1-methylethyl), carvone oxide, carvomenthone, shisool acetate, caryophylla-4(12),8(13)-dien-5α-ol and 2-naphthalenemethanol, decahydro-α,α,4a-trimethyl-8-methylene-, [2R-(2α,4aα,8aβ)] noticeably prevailed in the volatile oil of the aged sample. It is believed that these compounds are the products of the decomposition and oxidation of the classical terpene compounds that occurred upon storage for one year. These ingredients in the fresh sample can be sorted out according to their abundance, depending on the percentage area of the peaks in the chromatogram (from the highest to the lowest) as follows: L-camphor, fragranyl isobutyrate, γ-terpinene, terpinene-4-ol, (+)-β-thujone, eucalyptol, (+)-4-carene, o-cymene, spathulenol, caryophyllene oxide, oxacyclotetradecan-2-one, caryophyllene, and D-limonene. For the old sample, 2-cyclohexen-1-ol, 1-methyl-4-(1-methylethyl) accounted for the largest abundance followed by caryophylla-4(12),8(13)-dien-5α-ol, linalool, 2-naphthalenemethanol, decahydro-α,α,4a-trimethyl-8-methylene-, [2R-(2α,4aα,8aβ)] and carvone oxide. A short review of the main active ingredients in the fresh sample of AS and some of their documented biological effects is included in Table 3. Camphor was documented to have a variety of medicinal properties. This monoterpene possesses a good antifungal effect,18 neuroprotective effects,19 analgesic and anti-inflammatory effects.20 Fragranyl isobutyrate, the second most abundant in the fresh sample, is almost absent in the old sample. This non-terpene (C14H24O2) compound and its analogs
fragranyl acetate, are important components in volatile oils from aromatic plants, including Achillea species.21,22 As far as we are aware, these esterified compounds were not studied for any putative pharmacological effects. On the other hand, they may contribute to the toxic effects that appear with the use of some Achillea species.23 As a result, we recommend that these compounds be examined for any putative pharmacological or toxicological effects. γ-Terpinene is the third most abundant terpene in the fresh sample volatile oil. With terpinene-4-ol and α-terpinene, γ-terpinene was found to possess an antiprotozoal effect against Trypanosoma,24 and antibacterial activity against Listeria monocytogenes25 when combined with α-terpinene and eugenol. In addition, it was found to possess the potential to modulate the acute inflammatory response in mice.26 α and β-Thujone caused apoptosis in choriocarcinoma cells by interfering with a variety of metabolic processes.27 A huge number of effects have been confirmed for eucalyptol, including anticandidal,18 anti-inflammatory, and antinociceptive effects.28 Carene enhances sleep by targeting GABAA receptors.29 o-Cymene is an aromatic hydrocarbon (10 carbon atoms) that was not studied individually for any putative pharmacological or toxicological effects. However, combined with other terpenoidal components, it was related to a variety of biological effects, such as antibacterial activity.30 Spathulenol is a tricyclic sesquiterpene that exists in both samples. It possesses antimycobacterial activity, especially against resistant isolates,31 and mitigates 6-hydroxydopamine-mediated neurotoxicity in SH-SY5Y neuroblastoma cells.32 Caryophyllene, a bicyclic sesquiterpene, and its metabolic by-product caryophyllene oxide are thought to have anti-cancer and analgesic effects.33 Oxacyclotetradecan-2-one is a major ingredient in the volatile oil of a variety of plants and mushrooms, particularly of the genus Dictyophora. Oxacyclotetradecan-2-one and other components, such as 9,12-octadecadienoic acid and hexadecanoic acid, make up approximately 97.76 % of the quantity of the volatile oil of Dictyophora echinovalvata. The volatile oil of this mushroom interestingly had antibacterial and antifungal activities.34 D-limonene was almost absent in the old sample. It is one of the most extensively studied compounds with a noticeable increasing use owing to its multifunctional properties.35 This short review above solely includes a few pharmacological effects of the main active ingredients in the fresh sample volatile oil. Because these compounds are either absent or exist in lower amounts in the old sample, a more significant superiority and medicinal value of the fresh sample volatile oil can be assumed over that of the old sample. As mentioned earlier, these compounds seemed to have undergone decomposition and oxidation upon storage, resulting in the prevailing compounds in the old sample [Figure 4 (A and B)]. Figure 5 (A and B) demonstrates chromatograms of some polar compounds in the two samples. The two chromatograms are interestingly similar, supporting the belief that herb aging and storage seem to have no significant effect on the main polar compounds. Furthermore, this finding agrees with the results of the antioxidant activity, TPC, and TFC. Noteworthy, this method of extraction aimed to highlight some phenolic compounds, other than flavonoids, which are expected to contribute to the pharmacological activities of AS. The predominant peaks in both chromatograms, i.e., in the two samples, are derivatives of aromatic hydrocarbons such as naphthalene, anthracene, and azulene (a naphthalene isomer) (Table 1). Peaks 8 and 7 in chromatograms A and B in Figure 5, respectively, refer to the most predominant compound, achillicin (8-acetoxyartabsin), which is a derivative of sesquiterpene. Consequently, achillicin is an azulene derivative and significantly prevails in the Achillea genus.36 It was noticed that achillicin abundance in both samples was not significantly affected by the herb aging (Table 1). Despite its noticeable abundance, achillicin was not studied as a pure compound for any probable biological activities. However, achillinin A and ainsfragolide were shown to possess cytotoxic effects in two and five cell lines, respectively (Li et al).37 Both compounds are sesquiterpenoids with a guaianolide skeleton.38 Both samples contained xanthyletin as one of the major components. Xanthyletin, a pyranocoumarin, and its derivatives are widely distributed in nature. It was documented to possess an antifungal activity, particularly against Pythium insidiosum.39 Xanthyletin appears to be a promising compound, therefore, we recommend that more studies be devoted to this compound to evaluate any biological effects. It is noteworthy that peak identification in all chromatograms was implemented using the retention times, mass spectra, and comparison with the data available in the library NIST17s.lib. As previously mentioned, the shift in the levels of the principal peaks in both samples was used to illustrate how storage affected the plant’s phytochemical composition. The old sample even lost several peaks that indicated a medicinally proved benefit or value. However, in the old sample, many peaks pertaining to marginal chemicals predominated in terms of crowdedness in the chromatogram.
Table 3: Some of the main active ingredients in the volatile oil of the fresh sample from Achillea santolina and some of their documented biological activities.
| Active ingredient | Biological effect | Reference |
| Camphor | Antifungal effectNeuroprotective
Analgesic and anti-inflammatory |
[Ivanov et al 14][Salama et al19] [dos Santos et al20] |
| Fragranyl isobutyrate | Has not been studied for putative biological effects | – |
| γ-terpinene:With terpinene-4-ol and α-terpinene
With α-terpinene and eugenol |
Antiprotozoal effect against Trypanosoma
Antibacterial activity against Listeria monocytogenes Modulation of the acute inflammatory response in mice |
[Baldissera et al24] [ Oyedemi et al25] [Ramalho et al26] |
| β-thujone | Causing apoptosis in choriocarcinoma cells by interfering with a variety of metabolic processes | [ Lee et al27] |
| Eucalyptol | AnticandidalAnti-inflammatory and antinociceptive | [Ivanov et al18][Yin et al28] |
| Carene | Enhancement of sleep by targeting GABAA receptors | [Woo et al29] |
| o-cymenewith other terpenoidal components | Not studied by itselfAntibacterial | [ Esquivel-Ferriño et al30] |
| Spathulenol | Antimycobacterial, especially against resistant isolatesAlleviation of 6-hydroxydopamine-caused neurotoxicity in SH-SY5Y neuroblastoma cells | [Dzul-Beh et al31][ Manjima et al32] |
| CaryophylleneWith caryophyllene oxide | Anti-cancer and analgesic | [Fidyt et al33] |
| Oxacyclotetradecan-2-oneWith other compounds such as 9,12-octadecadienoic acid and hexadecanoic acid | Antibacterial and antifungal | [Tan et al.34] |
| D-limonene | Multifunctional properties | [Anandakumar et al35] |
Conclusion
Achillea santolina is indigenous to Jordan, where it is commonly used in traditional medicine. The herb is mostly dried and stored before use. AS is much weaker as an antioxidant compared to ascorbic acid. Storing the plant for one year did not seem to significantly affect its antioxidant properties, total phenolic, or total flavonoid contents. Plant storage had a noticeable effect on the AS volatile oil monoterpene and sesquiterpene content. Several ingredients in the fresh sample significantly diminished or were absent in the old sample, such as (+)-4-carene, o-cymene, eucalyptol, γ-terpinene, (+)-β-thujone, L-camphor, terpinene-4-ol, fragranyl isobutyrate, caryophyllene, D-limonene, spathulenol, caryophyllene oxide, and oxacyclotetradecan-2-one. The terpenoidal volatile oil content plays a very pivotal role in the overall biological activities of AS. On the contrary, plant aging did not seem to significantly affect the major polar compounds, mostly derived from naphthalene, anthracene, and azulene. One of these chief compounds is achillicin, which was remarkably abundant in both samples. As a result, we estimate that Achillea santolina loses a lot of its biological activities when stored for one year, presumably due to the effect on the terpene content of its volatile oil. To attain the maximum benefits, the fresh plant should be used.
Acknowledgement
The authors are also profoundly grateful to Dr. Khaled Abulaila, Ph.D. (a taxonomist at the National Agricultural Research Center (NARC), Al-Baqah, Amman, Jordan for his efforts in authenticating the study plant.
Funding Sources
The authors would like to thank Philadelphia University, Amman, Jordan, for the grant to cover the research costs and for securing a convenient environment for performing the experiments.
The project was funded by the Deanship of Scientific Research and Graduate Studies, Philadelphia University, Amman, Jordan.
Project number: 370-34/100 ب ع
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors contributions
- Yousef Abdel-Kareem Abusamra: the main author, designed the research, supervised the experimental work, analyzed the results, and wrote the manuscript.
- Mohammad Rasool Shomali: participated in performing the experiments in the lab.
- Abdel Naser Dakkah: participated in performing the experiments in the lab.
- Hani Hamad: did the statistical analysis and aided in writing the manuscript.
References
- Nemeth E, Bernath J. Biological activities of yarrow species (Achillea spp.). Curr Pharm Des. 2008;14(29):3151-67. doi: 10.2174/138161208786404281.
CrossRef - Saeidnia S, Gohari AR, Mokhber-Dezfuli N, Kiuchi F. A review on phytochemistry and medicinal properties of the genus Achillea. DARU, Journal of Pharmaceutical Sciences. 2011; 19:173–86.
CrossRef - Strzępek-Gomółka M, Gaweł-Bęben K, Kukula-Koch W. Achillea species as sources of active phytochemicals for dermatological and cosmetic applications. Oxidative Medicine and Cellular Longevity. 2021; Preprint at https://doi.org/10.1155/2021/6643827.
CrossRef - Mohammadhosseini M, Sarker SD, Akbarzadeh A. Chemical composition of the essential oils and extracts of Achillea species and their biological activities: A review. J Ethnopharmacol. 2017;199:257-315. Preprint at https://doi.org/10.1016/j.jep.2017.02.010.
CrossRef - Hudaib M, Mohammad M, Bustanji Y, Tayyem R, Yousef M, Abuirjeie M, Aburjai T. Ethnopharmacological survey of medicinal plants in Jordan, Mujib Nature Reserve and surrounding area. J Ethnopharmacol. 2008;120: 63–71.
CrossRef - Kalwij JM. Review of ‘The Plant List, a working list of all plant species’. Journal of Vegetation Science. 2012;23(5) Preprint at https://doi.org/10.1111/j.1654-1103.2012.01407.x.
CrossRef - Azizi M, Chizzola R, Ghani A, Oroojalian F. Composition at different development stages of the essential oil of four Achillea species grown in Iran. Nat Prod Commun. 2010; 5(2):283-90 doi:10.1177/1934578×1000500224.
CrossRef - Tadros, M. J., El-Shatnawi, M. K. J. & Jaradat, R. Q. Growth, persistence and quality of Trigonella arabica Del. and Trigonella caelesyriaca Boiss. grown in the semi-arid rangeland north of Jordan. J Food Agric Environ. 2011; 9(1):389-93.
CrossRef - Ahmed, W., Aburjai, T., Hudaib, M. & Al-Karablieh, N. Chemical Composition of Essential Oils Hydrodistilled from Aerial Parts of Achillea fragrantissima (Forssk.) Sch. Bip. and Achillea santolina L. (Asteraceae) Growing in Jordan. Journal of Essential Oil-Bearing Plants. 2020;23(1):15-25. doi:10.1080/0972060X.2020.1723442.
CrossRef - Truong H, Nguyen DH, Anh Ta NT, Bui, AV, Do TH, Nguyen HC. Evaluation of the use of different solvents for phytochemical constituents, antioxidants, and in vitro anti-inflammatory activities of Severinia buxifolia. J Food Qual. 2019;1, doi:10.1155/2019/8178294.
CrossRef - Jaradat NA, Zaid AN, Abuzant A, Shawahna R. Investigation of the efficiency of various methods of volatile oil extraction from Trichodesma africanum and their impact on the antioxidant and antimicrobial activities. J Intercult Ethnopharmacol. 2016; 5(3);250-6. doi: 10.5455/jice.20160421065949.
CrossRef - Braca A, De Tommasi N, Di Bari L, Pizza C, Politi M, Morelli I. Antioxidant principles from Bauhinia tarapotensis. J Nat Prod. 2001;64(7):892-5. doi: 10.1021/np0100845IF.
CrossRef - Uddin MN, Afrin R, Uddin MJ, Uddin MJ, Alam AH, Rahman AA, Sadik G. Vanda roxburghii chloroform extract as a potential source of polyphenols with antioxidant and cholinesterase inhibitory activities: identification of a strong phenolic antioxidant. BMC Complement Altern Med. 2015;15(195):1-9. doi: 10.1186/s12906-015-0728-y.
CrossRef - Duarte J, Pérez-Palencia R, Vargas F, Ocete MA, Pérez-Vizcaino F, Zarzuelo A, Tamargo J. Antihypertensive effects of the flavonoid quercetin in spontaneously hypertensive rats. Br J Pharmacol. 2001;133(1):117-24. doi: 10.1038/sj.bjp.0704064IF.
CrossRef - Al-Snafi AE. Chemical constituents and pharmacological activities of milfoil (Achillea santolina). A review. Int J Pharmtech Res. 2013;5(3): 1373-7.
CrossRef - Rahaman ST, Mondal S. Flavonoids: A vital resource in healthcare and medicine. Pharm Pharmacol Int J. 2020; 8(2):91-104 doi:10.15406/ppij.2020.08.00285.
CrossRef - Cosme P, Rodríguez AB, Espino J, Garrido M. Plant Phenolics: Bioavailability as a Key Determinant of Their Potential Health-Promoting Applications. Antioxidants (Basel). 2020;9(12):1263. doi: 10.3390/antiox9121263.
CrossRef - Ivanov M, Kannan A, Stojković DS, Glamočlija J, Calhelha RC, Ferreira ICFR, Sanglard D, Soković M. Camphor and Eucalyptol-Anticandidal Spectrum, Antivirulence Effect, Efflux Pumps Interference and Cytotoxicity. Int J Mol Sci. 2021;22(2):483. doi: 10.3390/ijms22020483IF.
CrossRef - Salama A, Mahmoud HA, Kandeil MA, Khalaf MM. Neuroprotective role of camphor against ciprofloxacin-induced depression in rats: modulation of Nrf-2 and TLR4. Immunopharmacol Immunotoxicol. 2021;43(3):309-18. doi: 10.1080/08923973.2021.1905658.
CrossRef - Dos Santos E, Leitão MM, Aguero Ito CN, Silva-Filho SE, Arena AC, Silva-Comar FMS, Nakamura Cuman RK, Oliveira RJ, Nazari Formagio AS, Leite Kassuya CA. Analgesic and anti-inflammatory articular effects of essential oil and camphor isolated from Ocimum kilimandscharicum Gürke leaves. J Ethnopharmacol. 2021;269:113697. doi: 10.1016/j.jep.2020.113697.
CrossRef - Motavalizadehkakhky A, Branch N, Shafaghat A, Akhlaghi H, Mohammadhosseini M. Compositions and the in vitro antimicrobial activities of the essential oils and extracts of two Achillea species from Iran. Journal of Medicinal Plants Research. 2013;7(19):1280-92. DOI: 10.5897/JMPR12.300.
CrossRef - Bader A, AlQathama A, Cioni PL, Ceccarini L, Abdelhady MIS, Al-Shareef W, Ascrizzi R, Flamini G. Essential Oil Biodiversity of Achillea ligustica All. Obtained from Mainland and Island Populations. Plants (Basel). 2022;11(8):1054. doi: 10.3390/plants11081054IF.
CrossRef - Radulović NS, Dekić MS, Ranđelović PJ, Stojanović NM, Zarubica AR, Stojanović-Radić ZZ. Toxic essential oils: anxiolytic, antinociceptive and antimicrobial properties of the yarrow Achillea umbellata Sibth. et Sm. (Asteraceae) volatiles. Food Chem Toxicol. 2012;50(6):2016-26. doi: 10.1016/j.fct.2012.03.047IF.
CrossRef - Baldissera MD, Grando TH, Souza CF, Gressler LT, Stefani LM, da Silva AS, Monteiro SG. In vitro and in vivo action of terpinen-4-ol, γ-terpinene, and α-terpinene against Trypanosoma evansi. Exp Parasitol. 2016;162:43-8. doi: 10.1016/j.exppara.2016.01.
CrossRef - Oyedemi SO, Okoh AI, Mabinya LV, Pirochenva G, Afolayan AJ. The proposed mechanism of bactericidal action of eugenol, α-terpineol and γ-terpinene against Listeria monocytogenes, Streptococcus pyogenes, Proteus vulgaris and Escherichia coli. Afr J Biotechnol. 2009;1263. doi:10.3390/antiox9121263
CrossRef - Ramalho TR, Oliveira MT, Lima AL, Bezerra-Santos CR, Piuvezam MR. Gamma-Terpinene Modulates Acute Inflammatory Response in Mice. Planta Med. 2015;81(14):1248-54. doi: 10.1055/s-0035-1546169IF.
CrossRef - Lee JY, Park H, Lim W, Song G. α,β-Thujone suppresses human placental choriocarcinoma cells via metabolic disruption. Reproduction. 2020;159(6):745-56. doi: 10.1530/REP-20-0018IF.
CrossRef - Yin C, Liu B, Wang P, Li X, Li Y, Zheng X, Tai Y, Wang C, Liu B. Eucalyptol alleviates inflammation and pain responses in a mouse model of gout arthritis. Br J Pharmacol. 2020;177(9):2042-57. doi: 10.1111/bph.14967IF.
CrossRef - Woo J, Yang H, Yoon M, Gadhe CG, Pae AN, Cho S, Lee CJ. 3-Carene, a Phytoncide from Pine Tree Has a Sleep-enhancing Effect by Targeting the GABAA-benzodiazepine Receptors. Exp Neurobiol. 2019;28(5):593-601. doi: 10.5607/en.2019.28.5.593.
CrossRef - Esquivel-Ferriño PC, Favela-Hernández JM, Garza-González E, Waksman N, Ríos MY, del Rayo Camacho-Corona M. Antimycobacterial activity of constituents from Foeniculum vulgare var. dulce grown in Mexico. Molecules. 2012;17(7):8471-82. doi: 10.3390/molecules17078471. PMID:
CrossRef - Dzul-Beh AD, García-Sosa K, Uc-Cachón AH, Bórquez J, Loyola LA, Barrios-García HB, Peña-Rodríguez LM, Molina-Salinas GM. In vitro growth inhibition and bactericidal activity of spathulenol against drug-resistant clinical isolates of Mycobacterium tuberculosis. Revista Brasileira de Farmacognosia. 2020;29(6):798-800.
CrossRef - Manjima RB, Ramya S, Kavithaa K, Paulpandi M, Saranya T, Winster SB, Balachandar V, Arul N. Spathulenol attenuates 6-hydroxydopamine induced neurotoxicity in SH-SY5Y neuroblastoma cells. Gene Reports. 2021;25:101396.
CrossRef - Fidyt K, Fiedorowicz A, Strządała L, Szumny A. β-caryophyllene and β-caryophyllene oxide-natural compounds of anticancer and analgesic properties. Cancer Med. 2016;5(10):3007-17. doi: 10.1002/cam4.816IF.
CrossRef - Tan D, Wu R, Liang M, Lin Y. Studies on chemical compositions and antimicrobial activity of volatile oil of Dictyophora echinovolvata. Jun wu xi Tong = Mycosystema. 2002;21(2):228-33.
CrossRef - Anandakumar P, Kamaraj S, Vanitha MK. D-limonene: A multifunctional compound with potent therapeutic effects. J Food Biochem. 2021;45(1):e13566. doi: 10.1111/jfbc.13566IF.
CrossRef - Glasl S, Kastner U, Jurenitsch J, Kubelka W. Qualitative and quantitative determination of sesquiterpenoids in Achillea species by reversed-phase high-performance liquid chromatography, mass-spectrometry and thin-layer chromatography. J Chromatogr B Biomed Sci Appl. 1999;729(1-2):361-8. doi: 10.1016/s0378-4347(99)00134-6.
CrossRef - Li Y, Zhang ML, Cong B, Wang SM, Dong M, Sauriol F, Huo CH, Shi QW, Gu YC, Kiyota H. Achillinin A, a cytotoxic guaianolide from the flower of Yarrow, Achillea millefolium. Biosci Biotechnol Biochem. 2011;75(8):1554-6. doi: 10.1271/bbb.110234IF.
CrossRef - Wittayapipath K, Yenjai C, Prariyachatigul C, Hamal P. Evaluation of antifungal effect and toxicity of xanthyletin and two bacterial metabolites against Thai isolates of Pythium insidiosum. Sci Rep. 2020;10(1):4495. doi: 10.1038/s41598-020-61271-0IF.
CrossRef
List of abbreviations
% I: Percentage of inhibition
AS: Achillea santolina
DPPH: 2,2-diphenyl-1-picrylhydrazyl
GC-MS: Chromatography-mass spectrometry
IC50: Half-maximal inhibitory concentration
SPE: Solid-phase extraction
TFC: Total flavonoid content
TPC: Total phenolic content







