Manuscript accepted on :09-04-2025
Published online on: 01-05-2025
Plagiarism Check: Yes
Reviewed by: Dr. Rajan Singh
Second Review by: Dr. Wang Yue
Final Approval by: Dr. Mariia Shanaida
Shivaramakrishnan Balasubramanian2* , Moola Joghee Nanjan Chandrasekar1
, Moola Joghee Nanjan Chandrasekar1 , Praveen Thaggikuppe Krishnamurthy2
, Praveen Thaggikuppe Krishnamurthy2 and Balakrishnan Settu2
and Balakrishnan Settu2
1 Department of Pharmaceutical Chemistry, JSS College of Pharmacy, JSS Academy of Higher Education and Research, Ooty, The Nilgiris, Tamil Nadu, India
2Department of Pharmacology, JSS College of Pharmacy, JSS Academy of Higher Education and Research, Ooty, The Nilgiris, Tamil Nadu, India.
Corresponding Author E-mail:shivaram.krishna@jssuni.edu.in
DOI : https://dx.doi.org/10.13005/bpj/3170
Abstract
Tissue repair and regeneration depend on the synchronized phases of healing of wounds. Angiogenesis plays a vital role in this process. Cellular activities within the wound initiate angiogenesis from pre-existing vasculature, accelerating healing and repair. A delicate yet strategic balance between pro-angiogenic and anti-angiogenic biomolecules by circulating cells controls this process. Current studies suggest small molecules like drugs are under investigation for their potential to induce angiogenesis and promote healing. This study is aimed at investigating the efficacy of topical 5-amino salicylic acid (5-ASA), an anti-inflammatory drug for wound healing through angiogenesis. A more precise evaluation of the physiological processes underlying angiogenesis was achieved by employing chick chorioallantoic membrane assay, zebrafish embryogenesis, and the rat polyvinyl alcohol (PVA) sponge implantation model. Our findings showed that 5-ASA administration promoted angiogenesis across all three models. In the CAM assay, 5-ASA promoted blood vessel development without causing damage while, in the zebrafish model, embryos treated with 5-ASA sprouted more new blood vessels in intersegmental vessels. After these findings, we corroborated these results in PVA sponge implantation model; 5-ASA-treated groups exhibited enhanced cellular infiltration, collagen deposition, and neovascularization relative to vehicle-treated controls. These findings suggest that topical 5-ASA may offer a novel therapeutic approach to wound healing.
Keywords
5-amino Salicylic acid; Angiogenesis; Chick chorioallantoic membrane assay; Vascular endothelial growth factor receptor - 2; Wound healing; Zebrafish
Download this article as:| Copy the following to cite this article: Balasubramanian S, Chandrasekar M. J. N, Krishnamurthy P. T, Settu B. Assessment of Angiogenesis Potential of 5- Amino Salicylic Acid by In Vivo Studies. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Balasubramanian S, Chandrasekar M. J. N, Krishnamurthy P. T, Settu B. Assessment of Angiogenesis Potential of 5- Amino Salicylic Acid by In Vivo Studies. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/4lUqZeq |
Introduction
Deprived wound healing affects billions of individuals around the world regularly. A wound is a pathophysiological condition characterized by the loss of tissue structural integrity because of physical or chemical stress. Wound healing is a sequential process that comprises chemotaxis, followed by cellular infiltrations, and that propagates physiological consequences.2 The different phases of healing involve haemostasis, inflammation, cellular proliferation, and last, remodeling.3 Depending on the wound, the rate of healing varies. Hence, complete wound closure in this healing process may take 3 to 14 days. Spontaneous wound healing is influenced by factors such as nutrition, timely secretion of biological molecules, presence of comorbidities, and availability of oxygen at the wound site.4 Consumed antioxidants and vitamins may have a favourable effect in preventing free radicals and preventing additional tissue damage, demonstrating a tissue-protective function.5,6 However, co-morbid conditions, Smoking, alcohol consumption, unbalanced diet and age, can interfere with the healing process and are averse to healing.7 Reports suggest that therapy which include anti-inflammatory drugs, steroids and antibiotics may exhibit both beneficial and detrimental effects in the healing process.
Globally, a primary therapeutic challenge involves the management of acute and chronic wounds. For symptomatic alleviation in patients, the first two phases of haemostasis and inflammation are usually targeted. In the clinical setting, a split-thickness autograft is frequently used to heal persistent wounds. However, there are several limitations, including severe donor site morbidity, infection, and discomfort.8 We could deduce that the time to recover depends on the wound healing process’s sequential progression.9,10 The healing process is inappropriate if each phase takes longer than it should, which typically results in dysfunctional scar tissue. For wound healing to be effective, quantities of released chemical mediators are required. Excessive proliferation and maturation, resulting in hypertrophic scarring and faulty skin remodelling, can be caused by an inordinate number and irregular release of inflammatory mediators.11
Angiogenesis is a term that defined as a biological process of the formation of fresh blood vessels from existing vasculature and is vital in the wound healing. The term angiogenesis was coined in the year 1787 by Dr. John Hunter. Angiogenesis stimulation by small molecules has been studied for wound healing. This starts with the wound site’s underlying blood vessel cells proliferating.12 Following angiogenesis, depending on the type of biomolecules involved, rapidly dividing cells undergo cellular differentiation and maturation.13 This process is regulated with the initial action of pro-angiogenic factors released by various circulating cells like platelets, Macrophages, etc. followed by the release and action of anti-angiogenic factors to prevent further growth that might harm the body. An imbalance between these two factors can lead to a pathological condition such as cancer.14 After an injury, the level of pro-angiogenic factor rises until the maximum capillary content is reached, after which it falls to undetectable levels.
Growth factors are one of the potent mediators of intracellular communication that influence cellular proliferation, migration, differentiation, and maturation. For angiogenesis, several biomolecules often start this process. Vascular endothelial growth factor stands out among other molecules in its importance for angiogenesis via receptor interactions. Out of 3 subtypes, the VEGF-2 receptor (VEGFR-2) mediated action is recognized to be the most potent for cellular growth and differentiation. This receptor comprises one transmembrane region, seven Ig-like extracellular domains, and a tyrosine kinase sequence from the intracellular area by a kinase-insert domain. Through a PKC-γ dependent pathway, any activating ligand bound to the VEGFR-2 stimulates PLC-γ and starts Erk phosphorylation and proliferation. PLC-γ attaches to the phosphorylated Y 1175 in the C-terminal tail of the receptor and becomes phosphorylated, which increases the catalytic activity of PLC-γ. The second messenger molecules of inositol 1,4,5-trisphosphate (IP3) significantly increase the intracellular Ca2+ concentration and diacylglycerol (DAG) activates Protein kinase C downstream for angiogenesis. Indeed, a study reports that mice lacking PLC-γ cannot experience vasculogenesis and erythropoiesis in their embryos, which die on E9.0, a time when no vasculogenesis or erythropoiesis exists, underlining a role for PLC-γ in angiogenesis. PLC-γ is also essential for arterial development, according to a recent study of zebrafish mutants.15 PLC-γ defective zebrafish embryos do not respond to exogenous VEGF, showing that PLC-γ is involved in the VEGF function. Researchers are exploring repurposing the drugs such as cinnamaldehyde,16 simvastatin,17,18 and salvianolic acid.19,20 could promote angiogenesis by targeting VEGFR-2 via the MAPK/Erk pathway. The researchers report that 5-ASA down regulates antiangiogenic factors but were not clear on which factors will be responsible for the initiation of angiogenesis.21 With this in mind, we proposed the hypothesis that 5-ASA may influence wound healing through angiogenesis. This angiogenesis effect may be achieved by binding to the growth factor receptor, specifically VEGFR-2 receptor. Our previous studies have exhibited the wound healing potential of 5-ASA in animal models.22 In this study, we have explored the mechanism and pathway of 5-ASA to induce angiogenesis in the wound healing and repair.
Materials and Methods
Drugs and Chemicals
5-Aminosalicylic acid was purchased from Himedia, Pvt. Ltd. India. Fertilized eggs were acquired from the Poultry Research Station in Madhavaram, Chennai. Medical-grade polyvinyl alcohol (PVA) was supplied by Agarwal Enterprises of Dehradun, Uttarakhand.
Experimental animals
The in vivo study was evaluated using Wistar Rats and Transgenic Zebrafish. Wistar Rats was acquired from the Central Animal House facility, JSSCP Ooty and the Transgenic Zebrafish were acquired from NITTE NUCSER, Mangalore. Wistar albino rats weighing between 180-220 g were kept apart in standard cages (groups of six in a cage). Experimentation was done after a period of quarantine. The embryos of transgenic zebrafish of the strain Fli: GFP were used for angiogenesis study. Green fluorescent protein is denoted as GFP and Fli represents proto-oncogene, ETS transcription factor.23,24 Institutional Animal Ethics Committee number- JSSCP/OT/IAEC/50/2019-20 dated 29th February 2020 approved all the studies.
Chicken Egg Chorioallantoic Membrane Test for evaluating angiogenesis
Chorio allantoic membrane (CAM) of White Leghorn fertilized eggs is a widely used and recognized method for examining the angiogenesis and anti-angiogenic potential of small molecules. The CAM assay procedure is useful because of its versatile and rapid nature of formation of membrane within 4-5 days of embryonic development. Previous studies have used this protocol to study angiogenesis. After being collected, the eggs were incubated at 37 0 Celsius for 8 to 9 days. The air sac of each egg shell was then exposed through using sterilized scissors performing an incision on upper side of each egg shell. A small amount of light liquid paraffin was gently applied with cotton swabs to expose the chorioallantoic membrane, to make the vascular system of the egg more visible. Different concentrations of 5-ASA (20µl, 40µl, 60 µl, 80µl) were prepared using DMSO. The chorioallantoic membrane of chick embryos was treated with different concentrations of the drug solution. After application of drug solution, the eggs were observed at different time intervals. Image analysis was used to quantify the angiogenesis.25
Evaluation of angiogenic potential of topical 5-ASA using transgenic zebrafish model
Transgenic Zebrafish exhibiting fluorescence in blood vessel cells is a developing model to study angiogenesis in different pathological conditions.26,27 The transgenic zebrafish line Tg (fli1: EGFP) y1 is a valuable tool for studying angiogenesis, which exhibits green fluorescence in the endothelial cells. Two male and one female fish were used for each mating procedure. The embryos were collected and checked for cell division. The embryos whose cells did not proliferate were removed. Dechorionation was done after transferring healthy embryos to E3 medium. Later, the embryos were transferred to a 12 well plate, each containing 5 embryos in the E3 media. The embryos were dechorionated, and were exposed to the drugs concentrations selected and were incubated at 28.50 C. The sample size was 20 embryos per group (n=20). Stock solution of 5-ASA and its analogue in the concentration of 50,000 ppm was prepared in DMSO. At 24 hours post fertilization (hpf), 0.1ml of varying concentrations –20 ppm, 40 ppm, 60 ppm and 80 ppm were administered to the control group. Fluorescence imaging observation was performed for 72 hpf in the Intersegmental vessels (ISV). The visual screening was done with the help of Zoe’s fluorescent cell imager. The embryo was examined for viability, morphological defects, heart rate, and circulation. Using ImageJ, the vascular changes and the overall length of the ISV were determined.
Evaluation of angiogenic potential of 5-ASA using polyvinyl alcohol sponge implantation in Wistar rats
Polyvinyl alcohol (PVA) sponge implantation is a model used to study angiogenesis. The significance of this method is that it can evaluate the different cellular infiltrations during the wound healing process and can be used as an evaluating tool for angiogenesis by visualising the connective framework of the newer blood vessels formed over the sponge. This model has great alterability and gives reproducible results.28 Rats serve as a great model for studying skin wound recovery. A subcutaneous muscle called the panniculus carnosus exists in rats, and it helps the skin recover by contracting and producing collagen. Anaesthesia with ketamine at 60 mg per kg body weight and xylazine at 12 mg per kg body weight was administered intravenously. Following the removal of hair from the dorsal side of the body, the skin was washed with 70% (v/v) ethanol. The skin was incised for 1cm to show a subcutaneous pocket, which was then cut open with a surgical blade. The PVA sponge was subcutaneously placed in an air pocket, then sutured back with a sterile suturing needle and thread. Two similar sponges were implanted on both sides of the mid-dorsal line. Required postoperative care was provided until full recovery. Experimental rats (n=6) were divided into 4 groups comprises 6 animals each. The drug suspension was injected into the implanted sponge. Treatment Group I received vehicle Treatment Group II received 5-ASA 250 mg per kg body weight and Treatment Group III- received 5-ASA 500 mg per kg body weight for 14 days into the sponge. After the study period, the implants were taken out after euthanasia with Isoflurane. The tissue in the sponge was homogenized with 10 times cold 0.1 M PBS at neutral pH (10% homogenate) for 18 minutes at 550 rpm. Insoluble sponge was discarded. Residual tissue suspension sample was centrifuged for 20 minutes at 4˚C at 9,000 rpm. Aliquots of the supernatants were taken out and used. The total protein quantification was done using the sonicated sponge supernatant by the biuret process. Estimation of total protein, hydroxyproline and hexosamine is a strong indicator of collagen content and cellular proliferation.29 Tissues were hydrated, sectioned, and mounted with an alcohol-based fixative on a microscope slide. The slide was immersed in water for one minute before being agitated by hand. Then they were subjected to hematoxylin-eosin staining,30 Masson-Trichrome staining,31 and CD31 immune histological staining.32
Statistical Analysis
All numerical values in the tabulated data are presented as mean ± SEM. For the statistical analysis of various biochemical parameters, One-way analysis of variance (ANOVA) was done to compare the drug treatment groups vs Vehicle control with post-test module of Tukey’s multiple comparison test using Graph pad Prism, V8.01 (San Diego, CA, USA). P-value less than 0.05 was deemed statistically significant.
Results
Chicken Egg Chorioallantoic Membrane Test for evaluating angiogenesis
Fertilised egg’s CAM assay is a model to study the angiogenic potential of different chemical substances. The chorioallantoic membrane (CAM) is a highly vascularized structure that facilitates nutrient provision, gas exchange, and waste elimination of the developing embryo. This also can allow the delivery of drugs to determine the potential of its angiogenesis. The CAM assay revealed a dose-correlated pro-angiogenic effect of 5-ASA, characterized by significantly enhanced blood vessel growth when compared to the DMSO control group. Blood vessel destruction, a consequence of DMSO administration, allows for the assessment of treated egg’s relative angiogenic potential. Figure 1 demonstrates that blood vessel formation in chicken embryos increased after 5-ASA treatment at the concentrations between 60-80 μg/ml. These research findings validate the ability of 5-ASA to stimulate angiogenesis in this model.
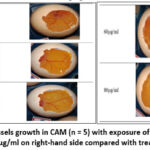 |
Figure 1: Images of blood vessels growth in CAM (n = 5) with exposure of 5-ASA at 10 μg/ml, 20 μg/ml, 40 μg/ml, 60 μg/ml and 80 μg/ml on right-hand side compared with treated DMSO on left hand sideClick here to view Figure |
Evaluation of angiogenic potential of 5-ASA using the transgenic zebrafish model
Transgenic zebrafish line Tg (fli1:EGFP)y1 is a valuable tool for studying angiogenesis. Driver gene fli1 promoter is an oncogene that drives the expression of enhanced green fluorescent protein (EGFP) in the endothelial cells of vasculature of the zebrafish, allowing for the visualization of blood vessels in live embryos. This action is mediated through VEGF-2 receptor. Therefore, the EGFP expression with the influence of drugs can be used to analyze the blood vessel growth. Angiogenesis was assessed by counting the number of complete intersegmental vessels (ISV) at 72 hours post fertilization (hpf) as shown in figure 2(A) & (B) and performing a vessel analysis using ImageJ plugin.The pro-angiogenic effect of 5-ASA treatment became most prominent in transgenic zebrafish embryos at an 80 μM dosage level, as it produced a maximum enhancement of intersegmental vessel formation. DMSO, as a negative control, causes the destruction of blood vessels and hence allows us to compare the angiogenic potential of treated groups. As shown in fluorescence imaging figure 2(C) and vessel analysis in figure 3 exhibit significantly higher ISV development after administering 5-ASA to transgenic embryos at significantly elevated in concentrations from 60-80 μM. A quantitative assessment supports 5-ASA’s angiogenic effect by VEGF-2 receptor pathway activation, which leads to elevated EGFP expression within transgenic embryos.
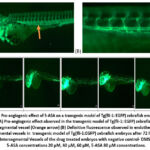 |
Figure 2: Pro-angiogenic effect of 5-ASA on a transgenic model of Tg(fli-1:EGFP) zebrafish embryos after 72 hrs. (A) Pro-angiogenic effect observed in the transgenic model of Tg(fli-1:EGFP) zebrafish embryos in Intersegmental vessel (Orange arrow).Click here to view Figure |
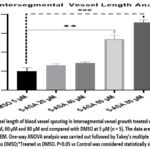 |
Figure 3: Vessel length of blood vessel spouting in Intersegmental vessel growth treated with 5-ASA at 20 μM, 40 μM, 60 μM and 80 μM and compared with DMSO at 5 μM (n = 5).Click here to view Figure |
Evaluation of angiogenic potential of 5-ASA using PVA sponge implantation model in Wistar rats
PVA sponge- a porous, artificial sponge made of inert, non-biodegradable material. Sponge is soft enough to be sectioned for histopathological examinations and easy for tissue homogenization. The model is used to analyze the effects of drugs on granulation tissue formation, collagen deposition, and wound repair. In this study, the PVA sponge was injected with different concentrations derived from previous studied acute dermal toxicity study. Figure 4 (A) & (B) shows PVA sponges exposed to 5-ASA treatment showed increased weight values besides elevated total protein content and elevated hydroxyproline and hexosamine level, along with enhanced neovascularization and collagen deposition patterns as revealed by histological observations. The PVA sponge implantation model shows that 5-ASA promotes substantial increases in cellular infiltration alongside collagen formation and neovascularization, whose effects depend on the medication dosage. Figure 5 shows the results from biochemical estimations and Figure 6 shows the histopathological examinations that confirm that 5-ASA produces pro-angiogenic effects in living tissues, which strengthens its case for wound healing usage.
 |
Figure 4: PVA sponges used (A) Sponges hydrated before Implantation (B) Excised sponges for analysisClick here to view Figure |
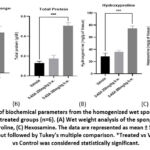 |
Figure 5: Estimation of biochemical parameters from the homogenized wet sponges of the treatment groups with vehicle treated groups (n=6). (A) Wet weight analysis of the sponge implants, (B) Total Protein, (C) Hydroxyproline, (C) HexosamineClick here to view Figure |
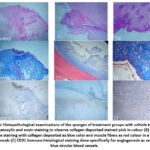 |
Figure 6: Histopathological examinations of the sponges of treatment groups with vehicle treated. (A) Hematoxylin and eosin staining to observe collagen deposited stained pink in colour.Click here to view Figure |
Discussion
Wound Care follows the principle of TIME, which is tissue viability, inflammation/infection extent, moisture imbalance and epithelial edge advancement. In contemporary medical practice, antiseptic, antibiotic, and anti-inflammatory agents are employed in the management of wounds. In addition, other strategies employed are growth factor delivery, surgical debridement with wound dressings, drug loaded scaffolds, self-forming hydrogels, stem cell therapy, hyperbaric oxygen therapy.
Wound healing is a multifaceted biological process linking interfaces between different cells, formation of extracellular matrix framework, and interchange of chemical mediators. Numerous treatments strategies have been employed. However, recent studies show that applying growth factors, stem cells externally, can enhance angiogenesis and speed up healing. Yet, their use is limited by expensive formulations, short half-lives of such protein-based preparations, and potential risk involved in off-target effects.33
Recently, research is intensive on repurposing drugs by topical application for treating acute and chronic wounds. In addition, small molecule induced Angiogenesis has taken a prominent stance in the wound healing process.34 Clinically used and FDA approved drugs such as simvastatin, cinnamaldehyde are used for treating wounds by angiogenesis.
Hence, topically induced angiogenesis with small molecules like 5-ASA may be beneficial for healing complex wounds.35 They offer several advantages over conventional formulations by their chemical nature, their site- specific action, fewer side effects because of lack of systemic action, may offer additional effects to counter co-morbid pathological conditions. Hence, by extensive literature search, we identified 5-ASA as a potential candidate for the hypothesis of our study. The chosen drug 5-ASA is an anti-inflammatory drug for treating colon diseases like Crohn’s disease, ulcerative colitis. The advantages of 5-ASA are that they are proven to treat UC locally without systemic effects, can be given topically at a much lesser dose, reported possessing surface-based action on growth factor receptors.
Among the several implicated targets in wound healing, we identified that growth factor receptors such as epidermal growth factor, platelet derived growth factor, fibroblast growth factor and vascular endothelial growth factor exhibit a vital role in the healing process of wounds. In the proliferative phase of wound healing, angiogenesis is a critical step that oversees scar less healing. Vascular endothelial growth factor and its receptors are one of the major factors of angiogenesis.36,37 In addition, early-stage angiogenesis is attained by binding on VEGF Receptor 2 (VEGFR-2). Within this receptor, ligands including vascular endothelial growth factor (VEGF) and angiogenin bind and interact with Ig-like sub-domains 2 and 3 (D2 and D3) of VEGFR-2. This triggers the activation and trans-autophosphorylation of the tyrosine kinase, which subsequently activates multifaceted intracellular signaling pathways. These events play a key role in regulating the proliferation, survival, migration, and permeability of vascular endothelial cells. The VEGFR-2 is a primary signaling receptor present on the endothelial cells, uses the PLC γ mediated activation of Protein Kinase C pathway for the development of newer blood vessels from existing vasculature.38 This system is an important target for pro-angiogenic therapy in treating chronic wounds such as diabetic foot ulcers, burn wounds.39
A previous study suggests that 5-ASA stimulates epithelial restitution and proliferation, enabling the quick repair of intestinal integrity by an action through growth factor receptors. In addition, 5-ASA being a BCS class IV drug, could only possess superficial action rather than systemic action. Therefore, we evaluated the potential of 5-ASA by different methods for determining probable molecular insights. By experimental models, the effect of 5-ASA applied in wound healing was studied for angiogenesis. All the biochemical estimations and histopathological examinations correlate with previous studies conducted.40,41 They show wound healing action by identifiable by biomarkers. However, the study further warranted an insight into the molecular aspect of healing, especially regarding whether the drug was possibly acting through VEGF-2 receptor mediated signaling. Hence, 5-ASA was studied on the transgenic zebrafish model and PVA sponge implantation models, which are specific tests to identify VEGF-2 receptor-based action.
Zebrafish (Danio rerio) is an evolving model organism to study angiogenesis because of its transparency during embryo and larval stages.42 Transgenic model of zebrafish used in this study is the strain Tg (fli1:EGFP). Green fluorescent protein is denoted as GFP and Fli represents the proto-oncogene, ETS transcription factor responsible for angiogenesis through VEGF-2 receptors. The results of 5-ASA induced angiogenic effect on the blood vessels using fluorescence imaging are comparable to a study.43 Similarly, a study conducted,44 showcases the angiogenic potential of curcumin nanoparticles in Zebrafish model and by comparing the results obtained in their study of vessel growth analysis, it indicates the angiogenesis potential of the test drug 5-ASA.
Lastly, a recent study,45,46 stated that the PVA sponge implantation model can be used to study granulation tissue formation, collagen deposition, and to analyze the effects of drugs on wound repair. Biochemical estimations of Hydroxy proline and Hexosamine levels, Masson trichrome Staining,47 (collagen deposition in blue and blood vessel formation in red colouration), presence of blood vessels growth observed in brown coloration in CD31 immunohisto staining,48,49 (staining specific for blood vessel growth) all corresponds to potential angiogenic property of 5-ASA.50 Results from our experimental models encompassing CAM, zebrafish, and PVA sponge implantation display identical pro-angiogenic responses, which confirm our hypothesis. CAM assay, while useful for visualization, lacks the complexity of mammalian wound healing environments. Zebrafish research model continues to serve as an important tool for VEGFR-2 mediated angiogenesis study while failing to maintain several characteristics of adult human tissue repair. PVA sponge model offers improved realness to wound healing condition, highlighting two potential experimental challenges between diseased animal responses and the implant’s impact on adjacent cell environments. The results show that 5-ASA enhances angiogenesis, although we need to examine additional mechanisms which might support these findings. It is possible to establish the mechanism by studying molecular dynamics of 5-ASA with VEGFR-2 and possible by studying the expression of eNOS in HUVEC cell lines in the future.
The pro-angiogenic effects of 5-ASA may be mediated by its antioxidant properties, thereby fostering an environment conducive to endothelial cell proliferation by mitigating oxidative damage at the injury site. The potential future applications of 5-ASA-based topical formulations such as ointments and creams encompass the treatment of diabetic ulcers, bone injuries, and other chronic wounds.
Conclusion
The significant clinical challenge of wound healing requires therapies that can orchestrate cellular migration, angiogenesis, and extracellular matrix remodelling for optimal tissue repair. Recent data states that among the drug induced signaling pathways of VEGFR-2, protein kinase C mediated new blood vessel growth was found to be more vital in the process of wound healing. Our initial findings indicate that 5-aminosalicylic acid exhibits angiogenesis activity, suggesting its potential therapeutic use in wound healing.
Acknowledgement
The authors acknowledge and thank the Department of Science and Technology – Fund for Improvement of Science and Technology Infrastructure in Universities and Higher Educational Institutions (DST-FIST), Govt. of India, New Delhi, for their infrastructure and support in our department. The authors acknowledge the generous research infrastructure and supports from JSS College of Pharmacy, JSS Academy of Higher Education & Research, Rocklands, Ooty, The Nilgiris, Tamil Nadu, India.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- S.B. (Shivaramakrishnan Balasubramanian): Conceptualization, Methodology, Writing – Original Draft
- M.J.N.C. (Moola Joghee Nanjan Chandrasekar): Visualization, Supervision, Project Administration
- P.T.K. (Praveen Thaggikuppe Krishnamurthy): Resources, Visualization, Supervision
- B.S. (Balakrishnan Settu): Data Collection, Analysis, Writing – Review & Editing
References
- Sen CK. Human Wound and Its Burden: Updated 2022 Compendium of Estimates. Adv Wound Care (New Rochelle). 2023;12(12):657-670. doi:10.1089/wound.2023.0150
CrossRef - Algermissen E. Wound healing: An overview. BDJ Student. 2023;30(2):24-26. doi:10.1038/s41406-023-0946-6
CrossRef - Kolimi P, Narala S, Nyavanandi D, Youssef AAA, Dudhipala N. Innovative Treatment Strategies to Accelerate Wound Healing: Trajectory and Recent Advancements. Cells. 2022;11(15):2439. doi:10.3390/cells11152439
CrossRef - Pang Q, Yang F, Jiang Z, Wu K, Hou R, Zhu Y. Smart wound dressing for advanced wound management: Real-time monitoring and on-demand treatment. Mater. & Des. 2023;229:111917. doi:10.1016/j.matdes.2023.111917
CrossRef - Potente M, Gerhardt H, Carmeliet P. Basic and Therapeutic Aspects of Angiogenesis. Cell. 2011;146(6):873-887. doi:10.1016/j.cell.2011.08.039
CrossRef - Chandimali N, Bak SG, Park EH, Lim HJ, Won YS, Kim EK, Park SI, Lee SJ. Free radicals and their impact on health and antioxidant defenses: a review. Cell Death Discov. 2025;11(1):19. doi:10.1038/s41420-024-02278-8
CrossRef - Xu B, Anderson DB, Park ES, Chen L, Lee JH. The influence of smoking and alcohol on bone healing: Systematic review and meta-analysis of non-pathological fractures. eClinicalMedicine. 2021;42:101179. doi:10.1016/j.eclinm.2021.101179
CrossRef - Eriksson E, Liu PY, Schultz GS, Martins-Green MM, Tanaka R, Weir D, Gould LJ, Armstrong DG, Gibbons GW, Wolcott R, Olutoye OO, Kirsner RS, Gurtner GC. Chronic wounds: Treatment consensus. Wound Repair Regen. 2022;30(2):156-171. doi:10.1111/wrr.12994
CrossRef - Anderson K, Hamm RL. Factors That Impair Wound Healing. JACCWS. 2012;4(4):84-91. doi:10.1016/J.JCCW.2014.03.001
CrossRef - Kuchar E, Karlikowska-Skwarnik M, Wawrzuta D. Anti-Inflammatory Therapy of Infections. In: Encyclopedia of Infection and Immunity. Elsevier; 2022:791-797. doi:10.1016/B978-0-12-818731-9.00181-6
CrossRef - Akbarian M, Bertassoni LE, Tayebi L. Biological aspects in controlling angiogenesis: current progress. Cell Mol Life Sci. 2022;79(7):349. doi:10.1007/s00018-022-04348-5
CrossRef - Ballmer-Hofer K. Vascular Endothelial Growth Factor, from Basic Research to Clinical Applications. IJMS. 2018;19(12):3750. doi:10.3390/ijms19123750
CrossRef - Gresham RCH, Bahney CS, Leach JK. Growth factor delivery using extracellular matrix-mimicking substrates for musculoskeletal tissue engineering and repair. Bioact. Mater. 2021;6(7):1945-1956. doi:10.1016/j.bioactmat.2020.12.012
CrossRef - Huang Z, Bao SD. Roles of main pro- and anti-angiogenic factors in tumor angiogenesis. World J. Gastroenterol. 2004;10(4):463. doi:10.3748/WJG.V10.I4.463
CrossRef - A single autophosphorylation site on KDR/Flk‐1 is essential for VEGF‐A‐dependent activation of PLC‐γ and DNA synthesis in vascular endothelial cells | The EMBO Journal. Accessed May 26, 2021. https://www.embopress.org/doi/full/10.1093/emboj/20.11.2768
- Tayyib NA. Wound healing applications of cinnamon. In: Cinnamon. Elsevier; 2025:511-525. doi:10.1016/B978-0-443-21820-0.00030-1
CrossRef - Asai J, Takenaka H, Hirakawa S, Sakabe J ichi, Hagura A, Kishimoto S, Maruyama K, Kajiya K, Kinoshita S, Tokura Y, Katoh N. Topical Simvastatin Accelerates Wound Healing in Diabetes by Enhancing Angiogenesis and Lymphangiogenesis. AJP. 2012;181(6):2217-2224. doi:10.1016/j.ajpath.2012.08.023
CrossRef - Tavakoli M, Al-Musawi MH, Kalali A, Shekarchizadeh A, Kaviani Y, Mansouri A, Nasiri-Harchegani S, Kharazi AZ, Sharifianjazi F, Sattar M, Varshosaz J, Mehrjoo M, Najafinezhad A, Mirhaj M. Platelet rich fibrin and simvastatin-loaded pectin-based 3D printed-electrospun bilayer scaffold for skin tissue regeneration. Int. J. Biol. Macromol. 2024;265:130954. doi:10.1016/j.ijbiomac.2024.130954
CrossRef - Szwedowicz U, Szewczyk A, Gołąb K, Choromańska A. Evaluation of Wound Healing Activity of Salvianolic Acid B on In Vitro Experimental Model. Int J Mol Sci. 2021;22(14):7728. doi:10.3390/ijms22147728
CrossRef - Qin M, Ou R, He W, Han H, Zhang Y, Huang Y, Chen Z, Pan X, Chi Y, He S, Gao L. Salvianolic acid B enhances tissue repair and regeneration by regulating immune cell migration and Caveolin-1-mediated blastema formation in zebrafish. Phytomedicine. 2024;130:155553. doi:10.1016/j.phymed.2024.155553
CrossRef - Deng X, Tolstanova G, Khomenko T, Chen L, Tarnawski A, Szabo S, Sandor Z. Mesalamine Restores Angiogenic Balance in Experimental Ulcerative Colitis by Reducing Expression of Endostatin and Angiostatin: Novel Molecular Mechanism for Therapeutic Action of Mesalamine. J. Pharmacol. Exp. Ther. 2009;331(3):1071-1078. doi:10.1124/jpet.109.158022
CrossRef - Chandrasekar MJN, Mouleeswaran R, Chenniah J, Balasubramanian S. A Comparative Topical Wound Healing Study of 5-Amino Salicylic Acid As Ointment And Gel By In- Vitro And In Vivo Models. IJSTR. 2019;8(12).
- ZFIN Gene: fli1. Accessed December 7, 2022. https://zfin.org/ZDB-GENE-980526-426
- Francescone R, Vendramini-Costa DB. In Vitro Models to Study Angiogenesis and Vasculature. In: Marques Dos Reis E, Berti F. Vasculogenic Mimicry. Vol 2514. Methods Mol. Biol. 2022:15-28. doi:10.1007/978-1-0716-2403-6_2
CrossRef - Ribatti D, Annese T, Tamma R. The use of the chick embryo CAM assay in the study of angiogenic activiy of biomaterials. Microvasc. Res. 2020;131:104026. doi:10.1016/j.mvr.2020.104026
CrossRef - Chávez MN, Aedo G, Fierro FA, Allende ML, Egaña JT. Zebrafish as an emerging model organism to study angiogenesis in development and regeneration. Front. Physiol. 2016;7(Mar):56. doi:10.3389/FPHYS.2016.00056/BIBTEX
CrossRef - Fernezelian D, Rondeau P, Gence L, Diotel N. Telencephalic stab wound injury induces regenerative angiogenesis and neurogenesis in zebrafish: unveiling the role of vascular endothelial growth factor signaling and microglia. Neural Regen. Res. 2025;20(10):2938-2954. doi:10.4103/NRR.NRR-D-23-01881
CrossRef - Wang P, Qian L, Liang H, Huang J, Jin J, Xie C, Xue B, Lai J, Zhang Y, Jiang L, Li L, Jiang Q. A Polyvinyl Alcohol/Acrylamide Hydrogel with Enhanced Mechanical Properties Promotes Full-Thickness Skin Defect Healing by Regulating Immunomodulation and Angiogenesis Through Paracrine Secretion. Engineering. 2024;37:138-151. doi:10.1016/j.eng.2024.02.005
CrossRef - Ghanbari M, Salkovskiy Y, Carlson MA. The rat as an animal model in chronic wound research: An update. Life Sci. 2024;351:122783. doi:10.1016/j.lfs.2024.122783
CrossRef - Kim J, Choi W, Yoo D, Kim M, Cho H, Sung HJ, Choi G, Uh J, Kim J, Go H, Choi KH. Solution-free and simplified H&E staining using a hydrogel-based stamping technology. Front Bioeng Biotechnol. 2023;11:1292785. doi:10.3389/fbioe.2023.1292785
CrossRef - Hernández-Morera P, Castaño-González I, Travieso-González CM, Mompeó-Corredera B, Ortega-Santana F. Quantification and Statistical Analysis Methods for Vessel Wall Components from Stained Images with Masson’s Trichrome. Cappello F, ed. PLoS ONE. 2016;11(1):e0146954. doi:10.1371/journal.pone.0146954
CrossRef - Mahmoud NN, Hamad K, Al Shibitini A, Juma S, Sharifi S, Gould L, Mahmoudi M. Investigating Inflammatory Markers in Wound Healing: Understanding Implications and Identifying Artifacts. ACS Pharmacol. Transl. Sci. 2024;7(1):18-27. doi:10.1021/acsptsci.3c00336
CrossRef - Mamun AA, Shao C, Geng P, Wang S, Xiao J. Recent advances in molecular mechanisms of skin wound healing and its treatments. Front Immunol. 2024;15:1395479. doi:10.3389/fimmu.2024.1395479
CrossRef - Haniff HS, Knerr L, Liu X, Crynen G, Boström J, Abegg D, Adibekian A, Lekah E, Wang KW, Cameron MD, Yildirim I, Lemurell M, Disney MD. Design of a small molecule that stimulates vascular endothelial growth factor A enabled by screening RNA fold–small molecule interactions. Nat Chem. 2020;12(10):952-961. doi:10.1038/s41557-020-0514-4
CrossRef - Sanapalli BKR, Deshpande A, Sanapalli V, Sigalapalli DK. Unveiling the Unexplored Multifactorial Potential of 5-Aminosalicylic Acid in Diabetic Wound Therapy. Diseases. 2024;12(8):172. doi:10.3390/diseases12080172
CrossRef - Sjöberg E, Melssen M, Richards M, Ding Y, Chanoca C, Chen D, Nwadozi E, Pal S, Love DT, Ninchoji T, Shibuya M, Simons M, Dimberg A, Claesson-Welsh L. Endothelial VEGFR2-PLCγ signaling regulates vascular permeability and antitumor immunity through eNOS/Src. J. Clin. Invest. 2023;133(20):e161366. doi:10.1172/JCI161366
CrossRef - Shibuya M. Vascular Endothelial Growth Factor (VEGF) and Its Receptor (VEGFR) Signaling in Angiogenesis: A Crucial Target for Anti- and Pro-Angiogenic Therapies. Genes & Cancer. 2011;2(12):1097-1105. doi:10.1177/1947601911423031
CrossRef - Wang X, Bove AM, Simone G, Ma B. Molecular Bases of VEGFR-2-Mediated Physiological Function and Pathological Role. Front Cell Dev Biol. 2020;8:599281. doi:10.3389/fcell.2020.599281
CrossRef - Canha F, Soares R. The use of innovative targeted angiogenic therapies for ischemic diabetic foot ulcer repair: From nanomedicine and microRNAs toward hyperbaric oxygen therapy. Porto Biomed. J. 2023;8(1):e187. doi:10.1097/j.pbj.0000000000000187
CrossRef - Wang C gui, Lou Y ting, Tong M ji, Zhang L lian, Zhang Z jie, Feng Y zeng, Li S, Xu H zi, Mao C. Asperosaponin VI promotes angiogenesis and accelerates wound healing in rats via up-regulating HIF-1α/VEGF signaling. Acta Pharmacol. Sin. 2018;39(3):393-404. doi:10.1038/aps.2017.161
CrossRef - Saleh MA, Shabaan AA, May M, Ali YM. Topical application of indigo-plant leaves extract enhances healing of skin lesion in an excision wound model in rats. J Appl Biomed. 2022;20(4):124-129. doi:10.32725/jab.2022.014
CrossRef - Huang HT, Huang CY, Lee CJ, Sun BJ, Jhang ZW, Wen CC, Wang YH, Li TS, Chern CY, Chen YH. The angiogenesis-modulating effects of coumarin-derivatives. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2024;278:109862. doi:10.1016/j.cbpc.2024.109862
CrossRef - Lawson ND, Weinstein BM. In Vivo Imaging of Embryonic Vascular Development Using Transgenic Zebrafish. Dev. Biol. 2002;248(2):307-318. doi:10.1006/dbio.2002.0711
CrossRef - Singha A, Harshitha M, Kalladka K, Chakraborty G, Maiti B, Satyaprasad AU, Chakraborty A, Sil SK. Exploring the potential of curcumin-loaded PLGA nanoparticles for angiogenesis and antioxidant proficiency in zebrafish embryo (Danio rerio). Futur J Pharm Sci. 2024;10(1):167. doi:10.1186/s43094-024-00727-w
CrossRef - Saeed S, Martins-Green M. Assessing Animal Models to Study Impaired and Chronic Wounds. IJMS. 2024;25(7):3837. doi:10.3390/ijms25073837
CrossRef - Deskins DL, Ardestani S, Young PP. The polyvinyl alcohol sponge model implantation. J Vis Exp. 2012;(62):3885. doi:10.3791/3885
CrossRef - Karimi A, Shahrooz R, Hobbenagh R, Mohammadi R, Delirezh N, Amani S, Garssen J, M Adcock I. Histological Evidence for Therapeutic Induction of Angiogenesis Using Mast Cells and Platelet-Rich Plasma within A Bioengineered Scaffold following Rat Hindlimb Ischemia. Cell J. 2019;21(4). doi:10.22074/cellj.2020.6287
- Kim H, Cho HJ, Kim SW, Liu B, Choi YJ, Lee J, Sohn YD, Lee MY, Houge MA, Yoon Y sup. CD31+ cells represent highly angiogenic and vasculogenic cells in bone marrow: novel role of nonendothelial CD31+ cells in neovascularization and their therapeutic effects on ischemic vascular disease. Circ Res. 2010;107(5):602-614. doi:10.1161/CIRCRESAHA.110.218396
CrossRef - Kondo T, Takahashi M, Yamasaki G, Sugimoto M, Kuse A, Morichika M, Nakagawa K, Sakurada M, Asano M, Ueno Y. Immunohistochemical analysis of CD31 expression in myocardial tissues from autopsies of patients with ischemic heart disease. Legal Medicine. 2022;59:102127. doi:10.1016/j.legalmed.2022.102127
CrossRef - Schlüter A, Weller P, Kanaan O, Nel I, Heusgen L, Höing B, Haßkamp P, Zander S, Mandapathil M, Dominas N, Arnolds J, Stuck BA, Lang S, Bankfalvi A, Brandau S. CD31 and VEGF are prognostic biomarkers in early-stage, but not in late-stage, laryngeal squamous cell carcinoma. BMC Cancer. 2018;18(1):272.
CrossRef







