Manuscript accepted on :09-05-2025
Published online on: 20-05-2025
Plagiarism Check: Yes
Reviewed by: Dr. Swati Jogdand
Second Review by: Dr. Ankit Chowdhury
Final Approval by: Dr. Gul Ozcan
Retno Susilowati* , Maharani Retna Duhita
, Maharani Retna Duhita , Fitria Nungky Harjanti
, Fitria Nungky Harjanti , Isnaeni Hartiningsih
, Isnaeni Hartiningsih and Sherly Oktariyanti
and Sherly Oktariyanti
Department of Biology, Faculty of Science and Technology, Universitas Islam Negeri Maulana Malik Ibrahim Malang, Malang, Indonesia
Corresponding Author E-mail: retno.susilowati@bio.uin-malang.ac.id
DOI : https://dx.doi.org/10.13005/bpj/3174
Abstract
Type 2 diabetes mellitus (DM) is a chronic metabolic disorder characterized by hyperglycemia due to insulin resistance in peripheral tissues. This disease is difficult to cure but can be successfully managed with proper treatment adherence. Previous studies showed that the alcohol extract of Syzygium cumini (S. cumini) leaves inhibited α-glucosidase. In addition, the water extract of Lagerstroemia speciosa (L. speciosa) leaves enhances glucose transport in hepatocytes, and the corosolic acid component of the leaves promotes insulin receptor phosphorylation. Curcuma xanthorrhiza (C. xanthorrhiza) rhizomes are known for hepatoprotective properties, potentially mitigating diabetes-related complications. Therefore, this study aimed to evaluate the combination of the three herbal extracts as a safe antidiabetic treatment. The evaluation experiment was conducted on diabetic Wistar rats. Five treatment groups established were normal control, diabetic, metformin, and a combination of the extract at doses of 200 and 300 mg/kg BW (body weight). Parameters assessed were decreased glucose levels, insulin levels, insulin resistance, pancreatic histopathology, superoxide dismutase (SOD) and malondialdehyde (MDA), serum glutamic pyruvic transaminase (SGPT) and serum glutamic oxaloacetic transaminase (SGOT) levels. The results showed that the herbal extract combination significantly reduced blood glucose, insulin resistance, oxidative stress, and inhibited pancreatic damage in diabetic rats while maintaining liver safety. A dose of 300 mg/kg BW effectively reduced SGPT levels, signifying the potential of the combination as a safe and effective antidiabetic treatment.
Keywords
Curcuma xanthorrhiza; Diabetes; Lagerstroemia speciosa; Serum Glutamic Pyruvic Transaminase (SGPT); Syzygium cumini
Download this article as:| Copy the following to cite this article: Susilowati R, Duhita M. R, Harjanti F. N, Hartiningsih I, Oktariyanti S. Antidiabetic Ethanol Extract of Syzygium cumini L. Leaves, Lagerstroemia speciosa Leaves, and Curcuma xanthorrhiza Roxb. Rhizome in Diabetic Wistar Rats. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Susilowati R, Duhita M. R, Harjanti F. N, Hartiningsih I, Oktariyanti S. Antidiabetic Ethanol Extract of Syzygium cumini L. Leaves, Lagerstroemia speciosa Leaves, and Curcuma xanthorrhiza Roxb. Rhizome in Diabetic Wistar Rats. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/3SbQJW8 |
Introduction
Diabetes mellitus (DM) is a metabolic disease characterized by elevated blood glucose levels or hyperglycemia originating from impaired insulin secretion by pancreas (type 1 DM) or insulin resistance in peripheral tissues (type 2 DM). Type 2 DM is the most common form, accounting for approximately 90% of all diabetes cases, and the increasing global prevalence of this disease is a significant concern. According to International Diabetes Federation (IDF), in 2021, the global number of affected individuals was 536.6 million, with a projection to rise to 783.7 million by 2045. Indonesia reported 19.451 million cases in 2021, with an estimated increase to 28.569 million by 2045. Diabetes-related complications are a leading cause of mortality, contributing to approximately 59.5% of deaths in the country.1
Metformin is currently the first-line treatment for managing type 2 DM, which works by inhibiting gluconeogenesis in the liver, enhancing muscle glucose uptake, and suppressing glucagon activity.2 Prolonged use of metformin has risks, including adverse effects on kidney function, potentially leading to chronic kidney disease, as well as an increased vulnerability to lactic acidosis because of impaired circulation and tissue hypoxia.3 Consequently, safer alternative treatments, such as herbal medicines, are being explored.
Herbal medicines offer diverse mechanisms for improving diabetes symptoms. A literature review identified six primary mechanisms underlying herbal treatment effects, namely enhancing insulin secretion, improving insulin sensitivity, facilitating glucose uptake in muscles and adipose tissue, reducing intestinal glucose absorption, inhibiting gluconeogenesis in the liver, and exerting anti-inflammatory effects.4 These mechanisms signify the potential of herbal medicines in managing blood glucose levels while mitigating diabetes-related complications.
A promising herbal ingredient is S. cumini leaf extract, which has shown significant hypoglycemic effects in diabetic rats induced by streptozotocin.5 Ethanol extract from S. cumini leaves inhibits the α-glucosidase enzyme by approximately 97% in vitro and produces 81% antioxidant activity.6 These properties help regulate postprandial glucose levels by preventing sudden increases. Moreover, acute toxicity tests show that S. cumini seeds are safe at doses reaching 200 mg/kg BW (body weight).7
Another key herbal ingredient is L. speciosa because studies show that water and methanol extract from the leaves enhance insulin-mediated glucose transport in hepatocytes.8 Corosolic acid, a prominent active compound in the leaves, promotes insulin receptor phosphorylation, accelerating glucose uptake by muscle cells without affecting insulin saturation levels.9 L. speciosa leaves consist of a natural “phytoinsulin” property supporting endogenous insulin function.
The third herbal component is C. xanthorrhiza recognized for the hepatoprotective properties. Investigations have found that C. xanthorrhiza extract can normalize insulin levels, reduce blood glucose, and lower free fatty acid and triglyceride levels in obese rats.10 This plant reduces SGPT and SGOT levels to protect the liver from toxin-induced damage, such as the type caused by paracetamol.11
Combining the three herbs S. cumini, L. speciosa leaves, and C. xanthorrhiza provides a synergistic effect in diabetes management. S. cumini inhibits glucose absorption and enhances pancreatic function, L. speciosa leaves act as a phytoinsulin to support insulin activity, and C. xanthorrhiza protects against liver damage often associated with diabetes. These herbs provide antioxidant and anti-inflammatory benefits, which are crucial for preventing more complications. However, challenges remain in using the herbs, particularly the limited availability of S. cumini seeds, as the plant produces fruit seasonally and is considered rare.12 To address the scarcity of seeds, the use of S. cumini leaves is expected to facilitate the provision of this anti-diabetes herbal ingredient.
Materials and Methods
Ethical approval
The maintenance and handling of subjects adhered to the Guide for the Care and Use of Laboratory Animals 8th edition.13 The study animals were provided with a standard chow diet and water ad libitum. These included rats procured from the Malang Murine Farm Animal for an Experiment in Malang, Indonesia. The Ethics Commission of the Faculty of Science and Technology, UIN Maulana Malik Ibrahim Malang, approved all experimental protocols featuring animals under number 04/EC/KEP-FST/2024.
Preparation of plant extract
S. cumini leaves, L. speciosa leaves, and C. xanthorrhiza extract was obtained from UPT Materia Medica, located at Lahore Street No. 87, Pesanggrahan, Batu, East Java, Indonesia. The plant materials were dried at 60°C. The herbs ground into a fine powder until it could pass through an 80-mesh filter. A total of 500 g of plant powder was macerated in 96% ethanol at a ratio of 1:10 for 24 hours. The filtrat solution was filtered and concentrated using a rotary evaporator at a temperature of <60°C with a rotation speed of 80 rpm. Crude extract was stored in a refrigerator at 2-4°C.
Diabetes Induction
Diabetes induction was performed using a high-fat and fructose diet (HFFD) combined with propylthiouracil (PTU) and alloxan injection (HFFDUA). After an acclimatization period, rats (excluding the normal group) were fed a high-fat diet (HFD) and given a fructose solution for 2 weeks. The HFD formulation which consisted of standard feed, duck egg yolk, and lard14 was prepared by mixing 50 mL of lard, 50 mL of duck egg yolk, and 5 mL of PTU (0.001%).15 This was administered through a gastric tube, while a 15% fructose solution was provided as drinking water, and 40 g of normal feed was supplied daily. For diabetes induction, test animals, excluding normal controls, received an intraperitoneal injection of 170 mg of alloxan per 200 g of BW.16,17 The injected rats were given a 5% sucrose solution as a precaution against hypoglycemia, fasted for 8 hours after the 2-week HFFD regimen, and assessed to determine blood sugar levels. Rats with blood sugar levels exceeding 200 mg/dL post alloxan injection were classified as diabetic.
Design of animal experiment
A total of 50 Wistar rats of the same age (8 weeks) and average weight (150 g) were selected for this study. The rats were acclimated for 7 days at a 25–27°C temperature under a 12-hour light-dark cycle. During acclimatization, feeding was carried out with a standard chow diet and there was ad libitum access to water. Post acclimatization, 7 rats were maintained on a normal diet (control group), while 43 were fed HFD and provided 15% fructose solution as drinking water for 2 weeks, followed by alloxan injection. Among the subjects, 30 diabetic rats with blood glucose levels > 200 mg/dL were selected and divided into groups of five treatments. The groups included control or DM-H0 (Diabetic rats without treatment), DM-H200 (Diabetic rats treated with a herbal combination at 200 mg/kg BW), DM-H300 (Diabetic rats treated with a herbal combination at 300 mg/kg BW), and DM-Metf (Diabetic rats treated with metformin at a 45 mg/kg BW dose). BW and fasting blood sugar levels were measured before diabetes induction, 3 days after alloxan injection, and at the completion of this study. Treatments were administered daily for 2 weeks before euthanizing the rats to isolate blood serum, pancreas, and liver tissues for further analysis.
Serum, pancreas, and liver isolation and preparation
The animals given 14 days of herbal treatment were sacrificed, and blood and organ tissues were immediately collected. Blood was drawn from the ventricles of all rats after fasting for 8–10 hours. The blood samples were allowed to clot at room temperature for 10–20 minutes, then centrifuged at 2000–3000 RPM for 20 minutes. The supernatant was carefully collected, avoiding any sediment, and serum samples intended for use in 5 days were stored at 2 to 8°C.
SOD (superoxide dismutase) and MDA (malondialdehyde) levels were measured by thoroughly insing tissues of rats in ice-cold PBS (pH 7.4) to remove excess blood and weighed prior to homogenization. The tissues were minced and homogenized in PBS at a ratio of 1 g tissue: 9 mL PBS using a glass homogenizer on ice. The suspension was sonicated with an ultrasonic cell disrupter or subjected to freeze-thaw cycles to break down cells further. The homogenates were centrifuged at 12,000 RPM for 15 minutes at 4°C, the resulting supernatant was collected, and samples intended for use in 5 days were stored at 2 to 8°C.
A portion of pancreas was fixed in 10% formalin and processed into paraffin-embedded histological sections. The prepared sections were stained with hematoxylin and eosin (HE) for microscopic examination.
Glucose, SOD, MDA, insulin, SGOT, and SGPT levels measurement
Fasting glucose levels during the treatment period were measured with the Accu-Chek Instant (Roche), while fasting glucose levels in serum at the completion of the study were analyzed using a spectrophotometer. Insulin and MDA levels were determined using the procedures provided in the Rat IGF-1 and GRIN2B ELISA Kits (BT Lab), respectively.
SOD levels in the blood samples were assessed with the Mouse Cu-Zn SOD1 ELISA Kit (BT Lab). SGPT and SGOT levels were measured with the IUBIO iChem spectrophotometer following the IFCC-kinetic method, using reagents and protocols specified in the CliniQuant FSR-SGPT Reagent-21012014 and CliniQuant FSR-SGOT Reagent-21012014, respectively.
Histopathological observation
Pancreatic histological observations were performed with an Olympus CX23 microscope at 400x magnification.
Statistical analysis
All experimental data collected in this study were presented as mean ± standard deviation. Parametric data analysis was conducted using analysis of variance (ANOVA) followed by Duncan’s multiple range test for post hoc comparisons. Moreover, the statistical analyses were performed using SPSS version 21 for Windows (SPSS, Inc., Chicago, IL, USA).
Results
Effect of extract combination on blood glucose levels
The normal rats in this study had an average glucose levels of 114.6 ± 12.48 mg/dl. In comparison, the fasting glucose levels had a minimum value of 200 mg/dl with an average of 327.19 ± 108.45 mg/dl after HFFDUA (High Fat, Fructose Diet with PTU addition, and alloxan injection) induction. Thé results showed that precentage decreased blood glucose met the parametric rules, namely following a normal distribution with a homogeneous variance. The One-Way ANOVA test of negative data transformation found that the treatment had a very significant effect (p <0.01). DMRT test showed a decrease in blood sugar levels along with the increasing treatment dose. In the 2-week treatment, herbal dose of 200 or 300 mg/kg BW had the same effect as metformin administration but did not return to normal (Table 1).
Table 1: Insulin levels, insulin resistance, and percentage decreased glucose levels in blood serum (Mean ± SD) after the study
| Treatment | Insulin level (µIU/ml) | Insulin resistance | Glucose decreased (%) |
| Normal | 14.02±2.53 a | 4.0±0.94a | -7.4±4.21a |
| DM-H0 | 57.60±4.52 d | 24.93±5.34d | 33.2±4.56b |
| DM-H200 | 31.42±5.34 c | 11.33±3.99b | 49.84±15.01bc |
| DM-H300 | 22.37±3.19 b | 7.52±2.08ab | 53.49±12.80c |
| DM-Metf | 21.96±0.61 b | 6.26±0.52a | 73.47±0.14d |
Note: Average values followed by the different letter notations in the same column are significantly different from other treatments (p<0.05).
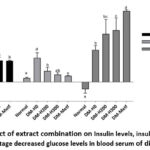 |
Figure 1: Effect of extract combination on Insulin levels, insulin resistance, and percentage decreased glucose levels in blood serum of diabetic rats. |
Effect of extract combination on insulin levels and insulin resistance
ANOVA test results showed a very significant effect of treatment on insulin levels and resistance (p<0.01). Meanwhile, DMRT test found that all diabetic rats experienced hyperinsulinemia, specifically DM-H0 group without treatment. The combination of herbal treatment significantly reduced hyperinsulinemia and insulin resistance, particularly the 300 mg/kg BW dose which decreased insulin levels the best. Both high dose extract and metformin administration produced this similar effect but did not return insulin to normal levels, the effect of a high dose herbal combination is able to reduce insulin resistance very well, the same as the effect of treatment using metformin until it returns to normal (Table 1, Figure 1).
Effect of extract combination on the histological structure of the islets of Langerhans in pancreas
Histological examinations (Image 1) were performed to assess both the endocrine tissue known as islets of langerhans and exocrine tissue composed of acinar cells in pancreas using qualitative and quantitative methods. HE staining of pancreatic sections showed that acinar gland cells participating in digestive enzyme production appeared darker than the lighter-colored islets of langerhans responsible for hormone secretion. In the normal treatment group, the cells in the islets of langerhans were compactly arranged, with a clear boundary between the endocrine and exocrine regions, suggesting healthy insulin production.
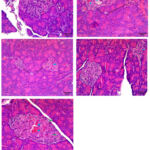 |
Image 1: Morphological and histological description of pancreas after combination treatment of L. speciosa L. leaf extract, S. cumini L. leaf extract, and C. xanthorrhiza R. rhizome extract. |
PI= Pancreatic Islet, A= Acinus cells, blue arrow= angiectasis, and green arrow= necrosis. Normal: Pancreatic Islets are circular, cells are arranged compactly, the nucleus is visible, no degeneration, and cells can be clearly distinguished from exocrine tissue. DM-H0: Pancreatic Islets are irregular in shape, cells are arranged non-compactly, nucleus appears brighter, and numerous angiectasis are observed. DM-H200: Pancreatic Islets are irregular in shape, cells are arranged less compactly, the nucleus appears pale, and small amount of angiectasis is observed. DM-H300: Pancreatic Islets are arranged compactly, the cells can be clearly distinguished from exocrine tissue, and there is small amount of angiectasis. DM-Metf: Pancreatic Islets are arranged less compactly, relatively large in size, while angiectasis and necrosis are observed. HE Staining at 400X. Normal= Normal diet, DM-H0= Diabetes mellitus without treatment, DM-H200= DM treated with combination of L. speciosa L. leaf extract, S. cumini L., and C. xanthorrhiza rhizome R. at 200 mg/kg BW, DM-H300= Diabetes mellitus treated with the combination of L. speciosa L. leaf extract, S. cumini L., and C. xanthorrhiza rhizome R. at 300 mg/kg BW, DM-Metf= Diabetes mellitus treated with Metformin.
Effect of extract combination on SOD and MDA levels of diabetic rats
The One-Way ANOVA test produced a p-value of <0.01, representing a significant effect on serum MDA, and Duncan’s test further confirmed the presence of two treatment groups with significantly different levels. The groups with low MDA levels were DM-H300 (2.197±0.81 U/ml) and DM-Metf (1.86±0.36 U/ml), while high MDA levels were found in the Normal (3.75±0.49 U/ml), DM-H0 (4.49±0.82 U/ml), and DM-H200 (4.68±0.56 U/ml) groups. This suggested that HFFD-alloxan induction induced type 2 diabetes in rats over the 2-week study period without reducing oxidative stress levels. However, the administration of the 300 mg/kg BW extract (DM-H300) significantly ameliorated oxidative stress by lowering MDA levels, showing similar effects to metformin treatment.
SOD and MDA hepatic data obtained from the One-Way ANOVA test showed a p-value >0.05. This suggested the absence of a significant treatment effect on SOD and MDA levels in the liver of experimental rats (Table 2, Figure 2).
Table 2: Stress oxidation and transaminase enzyme levels after treatment
| Treatment | Serum | Heparic lysate | Serum | |||
| SOD (U/ml) | MDA (U/ml) | SOD (U/ml) | MDA (U/ml) | SGPT (U/L) | SGOT (U/L) | |
| Normal | 9.37 ± 1.62 | 3.75 ± 0.49b | 4.19 ± 0.08 | 3.86 ± 0.24 | 21.92 ± 0.04a | 66.0± 0.10 |
| DM-H0 | 6.39 ± 1.03 | 4.49 ± 0.82b | 5.35 ± 0.89 | 1.31 ± 0.33 | 40.84 ± 1.41b | 96.8 ± 1.69 |
| DM-H200 | 6.02 ± 1.44 | 4.68 ± 0.56b | 5.43 ± 1.12 | 1.87 ± 1.11 | 43.43 ± 0.12b | 66.35 ± 0.25 |
| DM-H300 | 6.68 ± 3.21 | 2.20 ± 0.81a | 9.38 ± 2.10 | 2.28 ± 1.11 | 25.87 ± 0.73ab | 76.35 ± 0.11 |
| DM-Metf | 9.43 ± 0.66 | 1.86 ± 0.36a | 8.04 ± 2.24 | 0.82 ± 0.08 | 37.74 ± 2.20b | 65.3 ± 4.00 |
Note: Averages followed by different letter notations in the same column are significantly different from other treatments (p<0.05).
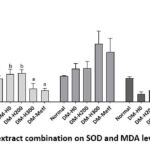 |
Figure 2: Effect of extract combination on SOD and MDA levels of diabetic rats |
Effect of extract combination on SGOT and SGPT levels
The results of the Kolmogorov-Smirnov normality test for SGOT levels showed normal data distribution with homogeneous variance, and ANOVA test found that the treatment had no significant effect (p>0.05). The Kolmogorov-Smirnov normality test for the LN transformation of SGPT data showed normal distribution (p>0.05), and the Kruskal-Wallis test produced homogeneous results (p>0.05). However, the One-Way ANOVA showed a p-value <0.05, suggesting a significant effect of the treatment on SGPT levels (Table 2, Figure 3).
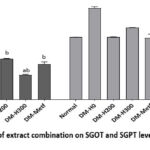 |
Figure 3: Effect of extract combination on SGOT and SGPT levels of Diabetic Rats |
Discussion
This study found that lower insulin resistance significantly decreased percentage blood glucose levels. The combination of herbs at a dose of 300 mg/kg BW administered for 2 weeks successfully improved insulin sensitivity, as evidenced by a substantial reduction in resistance levels (Table 1). The insulin resistance index (RI) in DM-H0 group was 24.93 ± 5.34, significantly decreasing to 7.52 ± 2.08 in DM-H300 treatment group. These results corresponded with the report by Sharma et al.18 that S. cumini effectively normalized insulin resistance, showing the potential as an antidiabetic agent. This effect is connected to the active compounds in S. cumini and L. speciosa leaf extract. Previous studies found that L. speciosa leaf extract at 25% and 50% concentrations provided a hypoglycemic effect comparable to insulin in alloxan-induced diabetic rats. The active compound corosolic acid in L. speciosa leaves enhances glucose transport, mimics insulin activity through GLUT4 activation, as well as inhibits α-amylase and α-glucosidase enzymes.19 These mechanisms were consistent with the results by Hibi et al.,20 showing that corosolic acid administration to diabetic patients for 2 weeks improved glucose tolerance by reducing glucose and insulin levels.
Resistance improvement in the experimental rats suggests that corosolic acid enhances insulin receptor phosphorylation and promotes glucose uptake into muscle cells, specifically at physiological insulin concentrations. These results show the potential of corosolic acid to normalize insulin signal transduction pathways, offering a novel approach to addressing insulin resistance. The induction of diabetes in this study uses alloxan, which selectively targets pancreatic beta cells through the GLUT2 glucose transporter. Alloxan generates reactive oxygen species (ROS), leading to beta-cell necrosis and reduced islet populations.21
Histological observations showed that angiectasis was observed in the islets of Langerhans in DM-H0 group, a condition characterized by the dilation of blood vessels and identified with irregularly shaped, enlarged spaces containing erythrocytes. Similar results of angiectasis in pancreatic tissue of alloxan-induced diabetic rats were reported by Ossai et al.22 In DM-Metf group, there were identifications of necrotic cells, characterized by vacuolated or empty cells. Histological observations found significant beta-cell damage in the untreated diabetic group, while the treatment groups showed protective effects. Specifically, the extract combination at 300 mg/kg BW showed the most significant treatment effect, promoting beta-cell regeneration in the islets of Langerhans. The examined cells showed compactly arranged pancreatic Islets, which were clearly distinguished from exocrine tissue, and small amount of angiectasis was identified. This observed effect may be supported by quercetin, a compound in S. cumini leaves known for playing a role in beta-cell regeneration and insulin production.
Hyperglycemia-induced oxidative stress was evident due to observations of elevated MDA levels, while MDA levels decreased in high-dose herbal treatment and metformin administration. These results corresponded with reports of decreased serum MDA levels followed by increased catalase activity.23 MDA is considered a reliable biomarker, particularly in comparison to SOD.24 Oxidative stress increases lipid peroxidation and tends to be complicated by environmental or physiological factors. The administration of the herbal extract combination reduced oxidative stress, as evidenced by decreased MDA levels. This improvement might be due to antioxidant properties of the extract, which contributed to decreased blood glucose levels. The results showed that improvements in oxidative stress were not directly related to SOD levels, suggesting the engagement of other enzymatic antioxidants such as glutathione peroxidase and catalase. Additionally, Keap1-Nrf2 signaling tended to play a role in mitigating oxidative damage. Inhibitors such as curcumin and corosolic acid stabilize Nrf2, facilitating nuclear translocation and activating antioxidant response elements (ARE). This process enhances the expression of antioxidant enzymes, including SOD and catalase,25 which help neutralize free radicals and protect cells from oxidative damage.
Liver function was a focal point of this study, considering the susceptibility to oxidative stress and diabetic complications. Based on the test results, the two parameters of liver physiological function showed different outcomes. There was no significant difference in the treatment effect on SGOT levels, but a significant effect was observed on SGPT levels. This is because the enzyme Aspartate Aminotransferase (AST) or SGOT is found in hepatocytes as well as produced in large quantities by myocardial muscle, skeletal muscle, the brain, and kidneys. Therefore, increased SGOT levels serve as a nonspecific marker of hepatocellular damage. In the case of the second enzyme, Alanine Aminotransferase (ALT) or SGPT is primarily found in the liver.26
Elevated SGPT levels in untreated diabetic rats suggested the presence of liver dysfunction. Administering the herbal extract combination at 300 mg/kg BW restored liver function, as evidenced by normalized enzyme levels. The result corresponds with studies showing the hepatoprotective effect of C. xanthorrhiza in reducing SGPT levels. 11 This hepatoprotective effect is attributed to phenolic compounds in the extract, which possess antioxidant and anti-inflammatory properties.27 Moreover, the ability of the phenolic compound xanthorrhizol to regulate DNA-binding activities of transcription factors, NF-κB and AP-1, can be a potential mechanism to clarify the preventive effect on hepatotoxicity. 28
This study confirms the safety and efficacy of the herbal treatment combination for managing diabetes and the complications. By restoring blood glucose levels, improving liver function, and mitigating oxidative stress, the extract combination offers a comprehensive method to address diabetes mellitus and the associated metabolic dysfunctions. Future studies should explore the long-term effect of this method and underlying molecular mechanisms to validate the clinical potential.
Conclusion
In conclusion, the results showed that the combination of extract from S. cumini L. leaves, L. speciosa leaves, and C. xanthorrhiza R. rhizomes had antidiabetic potential. A dose of 300 mg/kgbw is the best treatment in reducing blood glucose levels proportionally in diabetic rats, improving insulin resistance, improving the histological structure of the islets of Langerhans in the pancreas. This dose is also good in maintaining the status of oxidative stress in serum and liver and reducing SGPT enzyme levels which generally increase in diabetic conditions.
Acknowledgement
The authors are grateful to the Indonesian Ministry of Religion for providing study assistance funds in 2024. The authors are also grateful to the Medical Materials Center of Batu Malang City, East Java, for helping to prepare herbal simplicia.
Funding Sources
This study was supported by the Indonesian Ministry of Religion with the Decree number 4259 of 2024.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article
Ethics Statement
The Ethics Commission of the Faculty of Science and Technology, UIN Maulana Malik Ibrahim Malang, approved all experimental protocols featuring animals under number 04/EC/KEP-FST/2024.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Retno Susilowati: Conceptualization, Methodology, supervised the project, Writing – Original Draft, review &editing
- Maharani Retna Duhita: Conceptualization, Methodology, Analysis, Writing – Review & Editing.
- Fitria Nungky Harjanti:Data Collection, Analysis, Project Administration.
- Isnaeni Hartiningsih: Data Collection, Analysis & Visualization
- Sherly Oktariyanti: Data Collection, Analysis & Visualization
References
- Internasional Diabetes Federation. IDF Diabetes Atlas 10th edition, International Diabetes Federation., 2021. https://diabetesatlas.org/resources/idf-diabetes-atlas-2025/
- LaMoia TE, & Shulman GI. Cellular and molecular mechanisms of metformin action: Review. Endocrine Reviews, 2021;42(1):77–96. DOI: 1210/endrev/bnaa023
CrossRef - Rahman F, & Tuba S. Lactic acidosis associated with metformin in patients with diabetic kidney disease: Review. Med Arch. 2022;76(4):297–300. DOI: 5455/medarh.2022.76.297-300
CrossRef - Alam S, Sarker MMR, Sultana TN, Chowdhury MNR, Rashid MA, Chaity NI. Antidiabetic phytochemicals from medicinal plants: Prospective candidates for new drug discovery and development: Review. Frontiers in Endocrinology. 2022;3(800714):1–35. DOI: 3389/fendo.2022.800714
CrossRef - Fadhilah Z, Elya B, Setiawan H, Nugroho GA. Antidiabetic Activity and Phytochemical Constituents of Syzygium cumini Leave in Kadipaten, Central Java Indonesia, Indonesia. Pharmacogn J. 2021; 13(6): 1502-1508. DOI:5530/pj.2021.13.191
CrossRef - Artanti N, Maryani F, Dewi RT, Handayani S. In vitro antidiabetic, antioxidant, and cytotoxic activities of Syzygium cumini fractions from leaves ethanol extract. Indonesian Journal of Pharmacy.2019;10(1):24-29. https://ijcc.chemoprev.org/index.php/ijcc/article/view/262/178
CrossRef - Amudha S, Manna PK, Jeganathan NS. Evaluation of antidiabetic activity of Syzygium cumini extract and its phytosome formulation against streptozotocin-induced diabetic rats. The Pharma Innovation Journal, 2018;7(6): 603–608. https://www.thepharmajournal.com/archives/2018/vol7issue6/PartI/7-6-69-550.pdf
- Liu F, Kim J, Li Y, Li J, Chen X. An Extract of Lagerstroemia speciosa L. Has Insulin-Like Glucose Uptake–Stimulatory and Adipocyte Differentiation–Inhibitory Activities in 3T3-L1 Cells1. American Society for Nutritional Sciences: 2001;131(9):2442-2447. https://doi.org/10.1093/jn/131.9.2242
CrossRef - Shi L, Zhang W, Zhou Y, et al. Corosolic acid stimulates glucose uptake via enhancing insulin receptor phosphorylation. European Journal of Pharmacology, 2007;584(1):21–29. DOI: 1016/j.ejphar.2008.01.020
CrossRef - Kim M, Kim C, Song Y, Hwang J, Antihyperglycemic and anti-inflammatory effects of standardized Curcuma xanthorrhiza extract and its active compound xanthorrhizol in high-fat diet-induced obese mice. Evidence-Based Complementary and Alternative Medicine, 2014;2014(6):1–10. DOI: 10.1155/2014/205915
CrossRef - Pramono S, Arifah FH, Pribadi FH, Nugroho AE. Hepatoprotective activity of Curcuma xanthorrhiza on paracetamol-induced liver damage in rats and correlation. Thai Journal of Pharmaceutical Sciences. 2018;42(4):188-195. DOI:10.56808/3027-7922.2366
CrossRef - Yayasan Wanadri. Pohon jamblang (Syzygium cumini). West Java Conservation. 2022. https://conservation.id/2022/09/17/pohon-jamblang-syzygium-cumini/
- National Research Council of the National Academies. Guide for The Care and Use of Laboratory Animals 8th edition.. The National Academu Press, Washington DC. 2011. https://grants.nih.gov/grants/olaw/guide-for-the-care-and-use-of-laboratory-animals.pdf
- Alaydrus S, Pagal FR PA, Dermiati, Ervianingsih. Uji Efektivitas Ekstrak Etanol Biji Alpukat (Persea americana Mill.) terhadap Penurunan Kadar Kolesterol Total Tikus Putih Jantan (Rattus norvegicus) Model Hiperkolesterolemia Diabetes. Sains Dan Kesehatan, 2020;2(4): 405–411. https://jsk.ff.unmul.ac.id/index.php/JSK/article/view/225/224
- Adhitama S, Kuswanti N, & Khaleyla F. Pengaruh Ekstrak Daun Kedondong terhadap Penurunan Kadar Kolesterol Total dan Berat Badan Mencit Diabetes Melitus Tipe II Effect of Ambarella Leaf Extract in Reducing Total Cholesterol Levels and Body Weight of Mice with Diabetes Mellitus Type II. Lentera Bio. 2023;12: 354–362. https://doi.org/10.26740/lenterabio.v12n3.p354-362
CrossRef - Prambudi DTA, Meles DK, Widiyatno TV. Aktivitas Antihiperglikemia Fraksi Etil Asetat Daun Salam (Syzygium polyanthum) terhadap Tikus Putih yang Diinduksi Aloksan Monohidrat. Jurnal Kajian Veteriner, 2022;10(1):20–28. DOI: https://doi.org/10.35508/jkv.v10i1.6469
CrossRef - Sharma AK, Bharti S, Kumar R, et al. Syzygium cumini Ameliorates Insulin Resistance and β-Cell Dysfunction via Modulation of PPARγ, Dyslipidemia, Oxidative Stress, and TNF-α in Type 2 Diabetic Rats, J Pharmacol Sci. 2012;119:205 – 213. https://doi.org/10.1254/jphs.11184FP
CrossRef - Kim JM. Induction of diabetes mellitus using alloxan in Sprague Dawley rats. 2024; 16(6);1-8. DOI: 10.7759/cureus.63359
CrossRef - Chan EWC, Tan LN, Wong SK. Phytochemistry and pharmacology of Lagerstroemia speciosa: A natural remedy for diabetes: Review. International Journal of Herbal Medicine, 2014;2(1):81–87. : https://www.researchgate.net/publication/282183436
- Hibi M, Matsui Y, Niwa S, et al. Corosolic acid improves glucose and insulin responses in middle-aged men with impaired fasting glucose: A randomized, double-blinded, placebo-controlled crossover trial. Journal of Functional Foods 97. 2022;105256: 1-8. DOI: 1016/j.jff.2022.105256
CrossRef - Lenzen S. The mechanisms of alloxan- and streptozotocin-induced diabetes: Review. Diabetologia, 2008;51(2):216–222. DOI: 1007/s00125-007-0886-7
CrossRef - Ossai EC, Madueke AC, Amadi BE, et al. Potential enhancement of metformin hydrochloride in lipid vesicles targeting therapeutic efficacy in diabetic treatment. International Journal of Molecular Sciences, 2021;22(6):1-16. DOI: 3390/ijms22062852
CrossRef - Rita RS, Sy E. Syzygium Cumini Leaves Extract from West Sumatra Indonesia Alleviate Oxidative Stress by Decreasing Malondialdehyde Level and Enhancing Catalase Activity in Rat Induced by Lead Acetate. Pharmacogn J. 2021;13(6):1408-1412. DOI:5530/pj.2021.13.178
CrossRef - Camkurt MA, Fındıklı E, İzci F, Kurutaş B, Tuman TC. Evaluation of malondialdehyde, superoxide dismutase and catalase activity and their diagnostic value in drug naïve, first episode, non-smoker major depression patients and healthy controls. Psychitry Research. 2016; (238): 81-85. DOI: 1016/j.psychres.2016.01.075
CrossRef - Huang Y, Li W, Su Z, Kong AT. The Complexity of the Nrf2 Pathway: Beyond the Antioxidant Response: Review. Journal of Nutritional Biochemistry, 2015;26(2015):1401–1413. DOI: 1016/j.jnutbio.2015.08.001
CrossRef - De Vos A, De Troyer R, & Stove C. Biomarkers of alcohol misuse. Neuroscience of Alcohol. 2019:557–565. DOI:1016/B978-0-12-813125-1.00057-X
CrossRef - Devaraj S, Ismail S, Ramanathan S, Yam MF. Investigation of Antioxidant and Hepatoprotective Activity of Standardized Curcuma xanthorrhiza Rhizome in Carbon Tetrachloride-Induced Hepatic Damaged Rats. Scientific World Journal. 2014;2014(7):1-8 DOI: 1155/2014/353128
CrossRef - Kim SH, Hong KO, Chung WY, Hwang JK, Park KK. Abrogation of cisplatin-induced hepatotoxicity in mice by xanthorrhizol is related to its effect on the regulateion of gene transcription. 2004; 196(3):346-355. DOI: 1016/j.taap.2004.01.002
CrossRef







