Manuscript accepted on :11-03-2025
Published online on: 07-05-2025
Plagiarism Check: Yes
Reviewed by: Dr Zakir Hussain
Second Review by: Dr Jayanti Mukherjee
Final Approval by: Dr. Jihan Seid Hussein
Sunidhi Lohan* and Meenakshi Bhatia
and Meenakshi Bhatia
Department of Pharmaceutical Sciences, Guru Jambheshwar University of Science and Technology, Hisar, India
Corresponding Author E-mail:sunidhi.lohan1995@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3193
Abstract
Amphotericin B (BCS Class IV), an effective treatment for cutaneous leishmaniasis, has limited topical application due to poor skin penetration and stability. To overcome these challenges, this study aimed to formulate Amphotericin B-loaded nanostructured lipid carrier (NLC) gel to enhance drug encapsulation, stability, and controlled release. Amphotericin B-loaded NLCs were synthesized using a microwave-assisted method, which improved encapsulation efficiency and controlled particle size. The NLC dispersion was then incorporated into Carbopol 934 to form a gel. Characterization included particle size, zeta potential, morphology, spreadability, extrudability, in vitro drug release, ex-vivo skin permeation, and histopathological evaluation. The NLCs had a particle size of 180 nm, a polydispersity index of 0.32, and a zeta potential of –28.8 mV, ensuring stability. The Amphotericin B-NLC gel exhibited favorable physicochemical properties, including a smooth texture, a pH of 6.3, extrudability (9.5 ± 0.12 gm/cm²), and spreadability (6.4 cm). In vitro release studies demonstrated sustained release, with 48.05% drug release after 8 hours. The release kinetics followed the Higuchi model, indicating Fickian diffusion as the release mechanism. Ex-vivo permeation studies showed significantly enhanced skin permeation with the NLC and NLC gel formulations compared to the aqueous dispersion. Histopathological analysis indicated no signs of toxicity, with preserved skin integrity. The Amphotericin B-NLC gel formulation may significantly improve drug delivery for treating cutaneous leishmaniasis by enhancing skin penetration, providing controlled release, and demonstrating excellent safety.
Keywords
Amphotericin B; Histopathology; Microwave; Nanostructured Lipid Carriers; Topical Drug Delivery
Download this article as:| Copy the following to cite this article: Lohan S, Bhatia M. Amphotericin B-Loaded Nanostructured Lipid Carrier Gel: Preparation and Evaluation for Enhanced Skin Permeation. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Lohan S, Bhatia M. Amphotericin B-Loaded Nanostructured Lipid Carrier Gel: Preparation and Evaluation for Enhanced Skin Permeation. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/3Z4d8s8 |
Introduction
Parasitic skin infections represent a distinct category of dermatological conditions characterized by the invasion of parasites, leading to a spectrum of symptoms that can range from mild irritation to severe complications. A notable example is cutaneous leishmaniasis, which is caused by protozoan parasites of the genus Leishmania and primarily transmitted through the bites of infected sandflies.1 This disease presents a complex array of clinical manifestations, ranging from small, raised nodules to painful ulcers significantly affecting a patient’s quality of life. Lesions typically heal slowly, often leaving scars that can lead to psychological distress and social stigma.2,3 At present, 18 different Leishmania species are known to infect humans. Although these species have very similar morphologies, the primary clinical manifestations of leishmaniasis are visceral leishmaniasis and cutaneous leishmaniasis (CL).4CL is often described as a “cluster of diseases” due to the broad range of clinical manifestations it presents. These manifestations range from localized cutaneous leishmaniasis with ulcerative skin lesions to more extensive forms like diffused cutaneous leishmaniasis, which involves widespread lesions, and mucocutaneous leishmaniasis, which causes mucosal damage.5The first-line treatment for cutaneous leishmaniasis often involves systemic antimony compounds, such as sodium stibogluconate or meglumine antimoniate, which are effective but can cause severe side effects like liver and kidney toxicity, and are costly and require prolonged administration.6 Alternatively, oral azoles like ketoconazole or itraconazole may be used, offering an option with potentially fewer immediate side effects but posing risks of liver toxicity and drug interactions, and their efficacy can vary.7,8 Topical treatments such as paromomycin are used for localized lesions but are less effective for larger or deeper infections and can cause local irritation.9 Local therapies, including cryotherapy and thermotherapy, are effective for specific cases but can be painful, may lead to scarring, and often require multiple sessions.10 Amphotericin B is critical in treating cutaneous leishmaniasis due to its potent action against the parasites. It binds to ergosterol in the cell membranes, forming pores that disrupt membrane integrity, leading to cell lysis. While Amphotericin B is effective, its clinical use is hindered by significant nephrotoxicity and hematologic toxicity. Additionally, Amphotericin B’s poor oral bioavailability necessitates formulation strategies like nanostructured lipid carriers (NLCs) to improve therapeutic outcomes in cutaneous leishmaniasis treatment.11,12 Current treatments for leishmaniasis have significant limitations, highlighting the need for advanced drug delivery systems. Topical formulations are often favored for the treatment of dermatological disorders as they allow for direct delivery of the active pharmaceutical ingredient (API) to the target site, minimizing systemic exposure and associated side effects. Various formulations, including creams, ointments, gels, lotions, and patches, exhibit distinct physicochemical characteristics that make them suitable for specific therapeutic applications. These differences in texture, absorption kinetics, and delivery mechanisms enable the optimization of treatment strategies. For instance, the emulsion nature of creams and lotions facilitates effective hydration by retaining moisture, while the occlusive properties of ointments enhance the skin’s barrier function. Gels provide rapid absorption and a cooling effect, making them particularly beneficial for inflammatory conditions.13,14 Despite these advantages, conventional topical formulations face significant challenges in overcoming the stratum corneum, a major barrier to drug permeation that limits the therapeutic efficacy of treatments, especially for lipophilic drugs with poor solubility.15 To address this limitation, lipid-based nanocarriers have been widely investigated as a promising strategy for improving the topical delivery of poorly soluble drugs. These lipids based nanocarriers enhance the drug solubility within the matrix and facilitate its penetration through the skin layers, thereby improving the overall efficacy of the treatment. To improve the topical application of Amphotericin B for treating cutaneous leishmaniasis, we formulated a gel using NLCs. In our previous study, we investigated the efficacy of Amphotericin B-loaded Nanostructured Lipid Carriers against Leishmania donovani infection in THP1 cells.4 The results showed that Amphotericin B-NLC exhibited greater inhibition of L. donovani-infected cells compared to free Amphotericin B at the same concentrations, with IC50 values of 0.272 µg/ml and 0.414 µg/ml, respectively. This confirmed that the NLC formulation significantly enhanced the therapeutic efficacy of Amphotericin B. Building upon these findings, the current study focuses on the formulation of a topical Amphotericin B-NLC gel, aiming to leverage the previously demonstrated benefits of NLCs for improved drug delivery. The enhanced skin penetration and sustained release properties of the NLC gel may further amplify the therapeutic potential of Amphotericin B, particularly in the treatment of dermatological conditions, such as cutaneous leishmaniasis. The improved efficacy of Amphotericin B-NLC in previous research suggests that encapsulating Amphotericin B in NLCs can enhance its biological activity. Given the success in intracellular delivery and inhibition of L. donovani cells, it is hypothesized that the same NLC technology, when applied topically in gel form, will not only enhance skin penetration but also maintain sustained drug release at the application site, resulting in better treatment outcomes for dermatological conditions.
Materials and Methods
Materials
Tween 80 was obtained from Loba Chemie Private Limited, while Amphotericin B was sourced from MP Biomedical. Beeswax and oleic acid acquired from Sisco Research Laboratories Private Limited, and Carbopol 934 was purchased from HiMedia Laboratories Private Limited.
Microwave assisted synthesis of Amphotericin B brimmed NLC and NLC Gel
Amphotericin B-brimmed nanostructured lipid carriers were prepared employing a microwave-assisted single-pot method, as described in our previous research.4 Briefly, a specific amount of Amphotericin B was combined with a mixture of beeswax and oleic acid (7:3; 2% w/w), along with surfactant (Tween 80; 4% w/v), and heated at 80°C with continuous stirring (500 rpm) for 15 minutes using a microwave synthesizer with a power output not exceeding 18 W. This formulation was chosen based on our previous study,4 which optimized the composition for nanostructured lipid carrier (NLC) preparation. The resulting nanoemulsion was then dispersed in water by sonication in an ultrasonic bath for 30 minutes, yielding an Amphotericin B-loaded NLC dispersion. The NLCs were characterized for particle size (PS), polydispersity index (PdI) and zeta potential using dynamic light scattering (DLS), and encapsulation efficiency was also determined. After successfully preparing and characterizing the Amphotericin B-NLCs, a gel formulation was prepared. Specifically, a measured amount of Carbopol 934 was dispersed into the Amphotericin B-NLC dispersion and mixed thoroughly using a stirrer at 100 rpm until the gelling agent was uniformly distributed. The mixture was then neutralized with triethanolamine to achieve a gel with the desired viscosity. Three different concentrations of Carbopol (0.50%, 0.75%, & 1% w/v) were tested and evaluated to determine the optimal formulation. Additionally, an Amphotericin B aqueous dispersion was prepared at the same Amphotericin B concentration as the NLC gel for comparison.
Characterization of Amphotericin B-NLC and Amphotericin B-NLC Gel
The Amphotericin B-loaded Nanostructured Lipid Carriers were characterized using DLS, High-Resolution Transmission Electron Microscopy (HR-TEM), and Fourier Transform Infrared Spectroscopy (FT-IR). The physicochemical properties of the Amphotericin B-NLC gel were systematically evaluated, including color, odor, clarity, pH, and stability. Additionally, the Amphotericin B-NLC gel was assessed for attributes such as physical appearance, extrudability, spreadability & consistency. Further investigations included ex vivo permeation, in vitro release and histopathological analysis.
Dynamic Light Scattering (DLS)
DLS was utilized to assess the average PS and zeta potential of Amphotericin B-brimmed NLCs. This technique evaluates the PS distribution in aqueous suspension by monitoring the variations in light scattered from the particles as a result of Brownian motion.16 The PS and zeta potential measurements were carried out in triplicate using a Nano ZS90 Zetasizer (Malvern Instruments, UK) in automatic mode at a 90° scattering angle, with a 120-second equilibration time.
High-Resolution Transmission Electron Microscopy
The morphology of the Amphotericin B-NLC formulation was analyzed using HR-TEM. The sample was prepared by diluting the Amphotericin B-NLC dispersion 1:10 (v/v) with deionized water. A small amount of the diluted dispersion was applied to carbon-coated HR-TEM grids, and the excess liquid was removed by blotting. The grids were then left to dry at room temperature before being examined under a transmission electron microscope (Zeiss EM 900).17,18
Fourier Transform Infrared Spectroscopy
FT-IR spectroscopy was employed to analyze the interaction between Amphotericin B and the excipients of the formulation. The FT-IR spectra of both lyophilized Amphotericin B-NLC and pure Amphotericin B were recorded. To prepare the sample, a small amount of the lyophilized Amphotericin B-NLC was mixed with potassium bromide and then compressed into a pellet utilizing an IR hydraulic press (CAP-15 T, PCI Analytics, Mumbai, India). The FT-IR spectra were recorded over a range of 400–4000 cm−1 with a resolution of 1 cm−1 .20
Physical appearance, uniformity and pH
The physical characteristics, uniformity, and pH of the Amphotericin B-NLC gel were systematically evaluated for clarity, consistency, color, and the presence of any particulate matter .21 To detect potential undesirable particles, the preparation was examined against both white and black backgrounds.22 Given the critical impact of pH on the gel’s stability, solubility, and therapeutic efficacy, precise measurement was performed. For this, 1 gram of the Amphotericin B-NLC gel was dissolved in 35 mL of distilled water and stirred for 15 minutes to ensure a uniform dispersion. The pH was then measured at room temperature using a pH meter (Delta 320, Mettler Toledo, Switzerland).22 This procedure was repeated three times to ensure reproducibility, and the average pH value was calculated.
Extrudability test
The extrudability of the Amphotericin B-NLC gel was quantitatively assessed using a standardized extrusion test or tube test. In this method, the weight (gm) required to extrude a 0.5 cm ribbon of gel from an aluminum collapsible tube within 10 seconds was determined.23,24 To ensure accuracy, the test was conducted in triplicate, and the average weight required for extrusion was calculated. The extrudability of the formulation was then determined using the following formula:
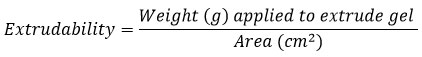
Spreadability
The spreadability of the Amphotericin B-NLC gel was quantitatively assessed to determine its ability to spread and uniformly distribute on the skin surface.25 In this test, 0.5 grams of the Amphotericin B-NLC gel was carefully placed within a pre-drawn circle with a 2 cm diameter on a 10 × 10 cm glass plate. A second glass plate of identical dimensions was then positioned on top and a 500 g weight was applied for 5 minutes.26,27 The extent of spread was evaluated by measuring the increase in the diameter of the gel, recorded as the final diameter (cm) of the expanded gel area.
In-vitro drug release studies
In vitro release of Amphotericin B from aqueous dispersion, Amphotericin B-NLC, and Amphotericin B-NLC gel were carried out using a dissolution apparatus (USP-II, Electrolab Dissolution Tester, 2,109,048, India). The Amphotericin B-NLC gel was placed in a pre-activated dialysis membrane & immersed in 300 ml of phosphate buffer (pH 6.8), stirred at 100 rpm. At specified intervals over an 8 h period, 5 ml samples were withdrawn & replenished with an equal volume of fresh release medium. The samples were then analyzed using a UV-Visible spectrophotometer at a wavelength of 407 nm (λmax). The experiment was repeated three times to ensure consistency. Additionally, the release behavior of Amphotericin B from both the Amphotericin B aqueous dispersion and Amphotericin B-NLC was evaluated. The release data were analyzed by fitting them to various kinetic models to understand the drug release mechanism.
Ex-vivo permeation
Ex-vivo permeation studies are crucial for evaluating the skin penetration and release profiles of topical formulations, providing valuable insights into their interaction with and diffusion through skin tissue outside of a living organism.28 In this research work, the transdermal permeation of Amphotericin B from aqueous dispersion of Amphotericin B, Amphotericin B-NLC and Amphotericin B-NLC gel was assessed using a Franz diffusion cell, with a receptor volume of 15 mL and an effective diffusion area of 3.14 cm². Goat skin was chosen for this study due to its permeability and structural characteristics, which have been shown to be similar to those of human skin, making it a suitable model for evaluating skin permeation.29,30 Additionally, goat skin is often sourced as a byproduct from slaughterhouses, making it an accessible and ethical alternative to human skin for ex-vivo research.31 Briefly, fresh goat ear skin, sourced from a slaughterhouse, was meticulously prepared by cleaning, depilating, and trimming it to fit the Franz diffusion cell. The goat ear skin was placed between the donor & receptor compartments. The receptor chamber was filled with 15 ml of phosphate-buffered saline (pH 6.8).4 A precise amount of Amphotericin B-NLC gel was added to the donor compartment, and the system was stirred at 100 rpm for 8 hours while maintaining a constant temperature of 37 ± 0.5°C throughout the experiment. At specific intervals, 1 ml aliquots were taken from the receptor compartment and replenished with an equal volume of fresh buffer to maintain consistent volume. The aliquots were analyzed by UV-visible spectrophotometer at wavelength of 407 nm to quantify the amount of Amphotericin B that permeated the skin. The experiment was repeated three times to ensure statistical reliability and reproducibility. A similar procedure was followed for the Amphotericin B aqueous dispersion and Amphotericin B NLC to enable direct comparison of the permeation profiles between the formulations.
Histopathology
A histochemical analysis was performed to examine the impact of the formulation on tissue structure and integrity. The freshly excised goat ear skin was thoroughly washed with normal saline to remove any contaminants. A measured amount of Amphotericin B-NLC gel was applied, and the skin was immediately preserved in a 10% (w/w) formalin solution for tissue fixation. Following fixation, the samples were dehydrated through an alcohol gradient and embedded in paraffin wax. The paraffin-embedded blocks were sectioned into 5 µm thick slices. The tissue sections were subsequently stained with hematoxylin & eosin and examined under a light microscope for structural evaluation.32
Results
Amphotericin B brimmed NLCs were successfully prepared using a microwave-assisted method and characterized for critical parameters, including PS, zeta potential, and entrapment efficiency. Following this, gel formulation was developed by incorporating the NLC dispersion into Carbopol 934 (0.5%). Detailed physicochemical characterization and evaluation of these formulations are presented in the following sections.
Dynamic Light Scattering (DLS), HRTEM and Entrapment efficiency of Amphotericin B NLC
Dynamic Light Scattering analysis of the Amphotericin B-loaded NLCs showed a PS of 180 nm, polydispersity index (PdI) of 0.32 & a zeta potential of – 28.8 mV (Fig. 1). The small particle size, and polydispersity index indicates a narrow size distribution, which is ideal for ensuring a uniform and consistent application in topical formulations.33-35
 |
Figure 1: Zeta potential graph of Amphotericin B NLC |
The negative zeta potential, resulting from the anionic nature of the lipid components, suggests strong electrostatic repulsion between the particles. This repulsion prevents particle aggregation, ensuring that the NLCs remain stable and well-dispersed, which is crucial for the effective and even application of the formulation.36 HRTEM analysis of the Amphotericin B-NLC formulation (Fig. 2) revealed spherical-shaped particles, with 200 nm size. The entrapment efficiency of the formulation was found to be 93.4%.
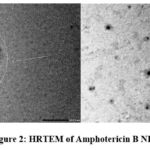 |
Figure 2: HRTEM of Amphotericin B NLC |
Fourier Transform Infrared Spectroscopy
The FT-IR spectrum of pure Amphotericin B (Fig. 3 a) revealed several distinct absorption peaks that correspond to specific molecular vibrations. A strong and broad peak at 3415 cm−1 was observed, corresponding to the O-H stretching vibration, indicative of a hydroxyl group involved in strong hydrogen bonding. This is further supported by the presence of a peak at 2923 cm−1, which is attributed to the C-H stretching vibration of aliphatic chains. Additionally, the band at 1686 cm−1 is consistent with the C=O stretching vibration, characteristic of the amide functional group. A sharp band at 1067 cm−1 was also noted, corresponding to the asymmetric stretching of the C=O bond, typical of ester or amide linkages. Upon analyzing the FTIR spectrum of the Amphotericin B-NLC formulation (Fig. 3 b), no significant shifts or new peaks were observed compared to the spectra of pure Amphotericin B and the excipients used in the formulation. This suggests the absence of significant chemical interactions or incompatibilities between the drug and the excipients, implying that the molecular integrity of Amphotericin B remains largely unaltered during the preparation of the NLC formulation.37
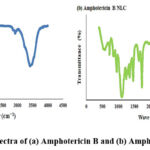 |
Figure 3: FTIR spectra of (a) Amphotericin B and (b) Amphotericin B NLC |
Physical appearance, pH and uniformity of Amphotericin B NLC gel
The Amphotericin B-NLCs gel displayed a yellowish color and was characterized by its odorless, smooth, stable, homogeneous, and translucent properties. With a pH of 6.3 ± 0.2, the gel is well-suited for skin application, aligning with the natural pH of skin.
Extrudability
Extrudability is an important property of gels, as it affects how easily they can be pushed out of a tube or container. Gels that are too thick can be hard to squeeze, leading to uneven application, while gels that are too thin may flow too quickly, causing waste.38 The Amphotericin B-NLC gel had an extrudability of 9.5 ± 0.12 gm/cm², indicating it has good consistency for smooth and controlled dispensing.
Spreadability
The Amphotericin B-NLC gel showed a spreadability diameter of 6.4 cm, indicating it can cover a larger area with minimal effort.
The in-vitro release profile
The in-vitro release profile of Amphotericin B from the Amphotericin B-NLC, Amphotericin B NLC gel and Amphotericin B aqueous dispersion showed a release of 54.18 ± 0.02%, 48.05± 0.03% and 16.12 %± 0.02% respectively, after 8 hours (Fig.4 a).The release kinetics of both Amphotericin B-NLC and Amphotericin B NLC gel were best described by the Higuchi model, exhibiting R² values of 0.997 and 0.994, respectively, with an n value of less than 0.5, indicating a release mechanism dominated by Fickian diffusion (n < 0.5).
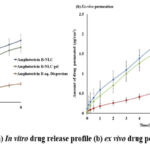 |
Figure 4: (a) In vitro drug release profile (b) ex vivo drug permeation |
Ex-vivo skin permeation study
Ex-vivo skin permeation studies were conducted to assess the permeability of Amphotericin B from an Amphotericin B-NLC formulation, aqueous dispersion of Amphotericin B, and Amphotericin B-NLC gel (Fig.4 b). The results showed significantly higher skin penetration of Amphotericin B from the Amphotericin B-NLC formulation compared to the aqueous dispersion.
Histopathology
A histological evaluation was performed on goat ear skin to evaluate the potential toxicological effects of the Amphotericin B-NLC gel. As demonstrated in the image (Fig. 5), treatment with the Amphotericin B-NLC gel did not induce any noticeable alterations in the skin histological structure. The integrity of both the epidermal and dermal layers was preserved, with no evidence of inflammation, cellular degeneration, or structural damage. These results indicate that the Amphotericin B-NLC gel does not adversely affect the normal histological features of goat ear skin, supporting its biocompatibility and safety for topical application.
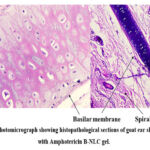 |
Figure 5: Photomicrograph showing histopathological sections of goat ear skin treated with Amphotericin B-NLC gel. |
Discussion
Amphotericin B-NLCs were developed using a binary mixture of solid and liquid lipids, exploiting the unique properties of this composition to enhance drug entrapment and facilitate controlled release profiles. The presence of the liquid lipid within the solid lipid matrix induces structural imperfections, which improve the encapsulation efficiency of Amphotericin B and contribute to the stability and sustained release characteristics of the NLCs, thereby optimizing their therapeutic performance.39 The results of the DLS analysis indicate that the Amphotericin B-loaded NLCs have a PS of 180 nm, which is ideal for topical formulations. The relatively low polydispersity index (0.32) suggests that the NLCs have a narrow size distribution, contributing to the consistency of the formulation. A narrow size distribution is crucial for achieving uniform application and optimal drug delivery.40,41 Additionally, the zeta potential of −28.8 mV indicates good stability of the NLCs. The negative zeta potential, due to the anionic characteristics of the lipid components, indicates significant electrostatic repulsion between the particles. This repulsion helps prevent aggregation, ensuring the stability and uniform dispersion of the NLCs, which is essential for the consistent and effective application of the formulation.42 The absence of significant shifts or new peaks in the FT-IR spectra of the Amphotericin B-NLC formulation suggests that there are no major chemical interactions or incompatibilities between the drug and the excipients. This indicates that the molecular integrity of Amphotericin B is largely preserved during the preparation of the NLC formulation, ensuring the stability of drug within the system. The yellowish color, smooth texture, and homogeneous, translucent appearance of the Amphotericin B-NLC gel indicate its aesthetic suitability for topical use. Additionally, with a pH of 6.3 ± 0.2, which closely matches the natural pH of the skin, the gel is well-suited for safe and effective use on the skin.43 The extrudability of the Amphotericin B-NLC gel indicates that the formulation has an ideal consistency, allowing for smooth and controlled dispensing. This is important for ensuring both effective application and minimizing waste during use. Spreadability is a crucial property for topical formulations as it influences the ease of application. A formulation with good spreadability ensures smooth and even coverage, enhancing both convenience and effectiveness.44 The spreadability of the Amphotericin B-NLC gel suggests it can cover a larger area with minimal effort, making it particularly beneficial for skin application. This attribute is especially important when applying gels to ulcerated skin, where efficient and even coverage is essential for optimal treatment. In vitro release data demonstrate that both the Amphotericin B-NLC formulation and NLC gel exhibited significantly higher drug release compared to the Amphotericin B aqueous dispersion after 8 hours, indicating a more controlled and sustained release profile. The enhanced skin permeation of Amphotericin B from the Amphotericin B-NLC formulation can be attributed to several factors, including the reduced PS of the NLC, which facilitates deeper skin penetration, and the use of Tween 80 as a surfactant to improve drug solubilization and dermal delivery, and the lipid matrix encapsulating Amphotericin B, which protects the drug and enables sustained release. The Amphotericin B-NLC gel exhibited comparable permeability to the Amphotericin B-NLC formulation but with a slower release profile, likely due to the increased viscosity of the gel, which reduces drug diffusion. This gel structure prolongs formulation retention on the skin, providing controlled release and extending drug contact time. The combination of smaller PS and Tween 80 in the NLC and gel formulations enhances dermal delivery and solubilization, offering a more sustained and controlled release compared to conventional formulations. This controlled release is advantageous for local treatment, maintaining therapeutic drug concentrations at the application site over time.45,46,47 The absence of any histological changes in goat ear skin following treatment with the Amphotericin B-NLC gel suggests that the formulation does not cause adverse effects on the skin. The integrity of the epidermal and dermal layers was preserved, indicating good biocompatibility and supporting the safety of the gel for topical application.
Future Prospects
Future work should focus on optimizing the Amphotericin B-NLC gel for enhanced stability, release control, and skin penetration. Clinical trials will be essential to validate its effectiveness in treating dermatological infections. Additionally, exploring combination therapies, scalable production, and long-term safety will be key for broader clinical application.
Conclusion
This study successfully developed an Amphotericin B-loaded Nanostructured Lipid Carrier gel with enhanced topical drug delivery properties. The Amphotericin B NLC gel demonstrated optimal stability, favorable physicochemical characteristics, good entrapment efficiency (93.4%) and a sustained release profile, with 48.05 % of Amphotericin B released over 8 hours. Ex-vivo permeation studies confirmed improved skin penetration. FT-IR analysis demonstrated that Amphotericin B retains its structural integrity during the NLC formulation process, with no significant shifts in peaks observed. This indicates the absence of undesirable interactions between the drug and excipients, confirming the stability of the formulation. Building on previous findings where AmpB-NLCs showed superior efficacy against Leishmania donovani compared to free Amphotericin B,4 the current gel formulation supports enhanced therapeutic potential for skin infections. Histopathological analysis confirmed its safety, suggesting the Amphotericin B-NLC gel as a promising candidate for effective and safe topical treatments.
Acknowledgement
The authors express their gratitude to the Dr. APJ Abdul Kalam Central Instrumentation Laboratory, GJUST, Hisar, India, for providing the FT-IR analysis facilities; the Department of Pharmaceutical Sciences, GJUST, Hisar, India, for offering Dynamic Light Scattering facilities; and the Sophisticated Analytical Instrumentation Facility, Panjab University Chandigarh, India for HR-TEM analysis.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest
Data Availability
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Sunidhi Lohan: Conceptualization, Investigation, Writing-original drafts preparation.
- Meenakshi Bhatia: Conceptualization, Supervision, Reviewing and Editing.
References
- Gurel MS, Tekin B, Uzun S. Cutaneous leishmaniasis: A great imitator. Clin Dermatol. 2020;38(2):140-151.
CrossRef - Zhuang L, Su J, Tu P. Cutaneous leishmaniasis presenting with painless ulcer on the right forearm: A case report. World J Clin Cases. 2022;10(7):2301.
CrossRef - Hepburn NC. Cutaneous leishmaniasis. Clin Exp Dermatol. 2000;25(5):363-370.
CrossRef - Lohan S, Bhatia M. Characterization and Evaluation of Microwave-Synthesized Nanostructured Lipid Carriers for Enhanced Amphotericin B Efficacy Against Leishmania donovani: A Novel Therapeutic Paradigm. BioNanoScience. 2024;3:2782-2800.
CrossRef - Riaz A, Ahmed N, Khan MI, Haq IU, ur Rehman A, Khan GM. Formulation of topical NLCs to target macrophages for cutaneous leishmaniasis. J Drug Deliv Sci Technol. 2019;54:101232.
CrossRef - Brito NC, Rabello A, Cota GF. Efficacy of pentavalent antimoniate intralesional infiltration therapy for cutaneous leishmaniasis: A systematic review. PLoS One. 2017;12(9):e0184777.
CrossRef - Prates FV, Dourado ME, Silva SC, Schriefer A, Guimarães LH, Brito MD, Almeida J, Carvalho EM, Machado PR. Fluconazole in the treatment of cutaneous leishmaniasis caused by Leishmania braziliensis: A randomized controlled trial. Clin Infect Dis. 2016;ciw662.
CrossRef - Saenz RE, Paz H, Berman JD. Efficacy of ketoconazole against Leishmania braziliensis panamensis cutaneous leishmaniasis. Am J Med. 1990;89(2):147-155.
CrossRef - Krause G, Kroeger A. Topical treatment of American cutaneous leishmaniasis with paramomycin and methylbenzethonium chloride: A clinical study under field conditions in Ecuador. Trans R Soc Trop Med Hyg. 1994;88(1):92-94.
CrossRef - Madusanka RK, Silva H, Karunaweera ND. Treatment of cutaneous leishmaniasis and insights into species-specific responses: A narrative review. Infect Dis Ther. 2022;11(2):695-711.
CrossRef - Abu Ammar A, Nasereddin A, Ereqat S, Dan-Goor M, Jaffe CL, Zussman E, Abdeen Z. Amphotericin B-loaded nanoparticles for local treatment of cutaneous leishmaniasis. Drug Deliv Transl Res. 2019;9:76-84.
CrossRef - Shirzadi MR. Liposomal amphotericin B: A review of its properties, function, and use for treatment of cutaneous leishmaniasis. Res Rep Trop Med. 2019:1-8.
- Buhse L, Kolinski R, Westenberger B, Wokovich A, Spencer J, Chen CW, Turujman S, Gautam-Basak M, Kang GJ, Kibbe A, Heintzelman B. Topical drug classification. Int J Pharm. 2005;295(1-2):101-112.
CrossRef - Barnes TM, Mijaljica D, Townley JP, Spada F, Harrison IP. Vehicles for drug delivery and cosmetic moisturizers: Review and comparison. Pharmaceutics. 2021;13(12):2012.
CrossRef - Hoffman MB, Hill D, Feldman SR. Current challenges and emerging drug delivery strategies for the treatment of psoriasis. Expert Opin Drug Deliv. 2016;13(10):1461-1473.
CrossRef - Sambhakar S, Saharan R, Narwal S, Malik R, Gahlot V, Khalid A, Najmi A, Zoghebi K, Halawi MA, Albratty M, Mohan S. Exploring LIPIDs for their potential to improve bioavailability of lipophilic drug candidates: A review. Saudi Pharm J. 2023;31(12):101870.
CrossRef - Gumbiowski N, Barthel J, Loza K, Heggen M, Epple M. Simulated HRTEM images of nanoparticles to train a neural network to classify nanoparticles for crystallinity. Nanoscale Adv. 2024;6(16):4196-4206.
CrossRef - Zheng M, Falkeborg M, Zheng Y, Yang T, Xu X. Formulation and characterization of nanostructured lipid carriers containing a mixed lipids core. Colloids Surf A Physicochem Eng Asp. 2013;430:76-84.
CrossRef - Karim A, Rehman A, Jafari SM, Miao S, Dabbour M, Ashraf W, Rasheed HA, Assadpour E, Hussain A, Suleria HA, Lianfu Z. Fabrication and characterization of sonicated peach gum-sodium caseinate nanocomplexes: Physicochemical, spectroscopic, morphological, and correlation analyses. Food Bioprocess Technol. 2024:1-20.
CrossRef - Kumbhar PS, Manjappa AS, Shah RR, Nadaf SJ, Disouza JI. Nanostructured lipid carrier–based gel for repurposing simvastatin in localized treatment of breast cancer: Formulation design, development, and in vitro and in vivo characterization. AAPS PharmSciTech. 2023;24(5):106.
CrossRef - Toler MR, Nema S. Visual inspection. In: Parenteral Medications, 4th ed. CRC Press; 2019:863-878.
CrossRef - Khot C, Kolekar K, Dabhole S, Mohite A, Nadaf S, Kumbhar PS, Disouza J. Optimized albendazole-loaded nanostructured lipid carrier gel: A redefined approach for localized skin cancer treatment. RSC Pharmaceutics. 2024.
CrossRef - Sareen R, Kumar S, Gupta G. Meloxicam carbopol-based gels: Characterization and evaluation. Curr Drug Deliv. 2011;8(4):407-415.
CrossRef - Ilievska B, Loftsson T, Hjalmarsdottir MA, Asgrimsdottir GM. Topical formulation comprising fatty acid extract from cod liver oil: Development, evaluation, and stability studies. Mar Drugs. 2016;14(6):105.
CrossRef - Malavi S, Kumbhar P, Manjappa A, Disouza J, Dwivedi J. Emulgel for improved topical delivery of Tretinoin: Formulation design and characterization. Ann Pharm Fr. 2022;80(2):157-168.
CrossRef - Bachhav YG, Patravale VB. Microemulsion-based vaginal gel of fluconazole: Formulation, in vitro and in vivo evaluation. Int J Pharm. 2009;365(1-2):175-179.
CrossRef - Ghareeb MM. Design and in vitro characterization of a topical nanoemulsion-enriched hydrogel of econazole nitrate. J Appl Pharm Sci. 2019;9(1):51-57.
CrossRef - Kocsis D, Klang V, Schweiger EM, Varga‐Medveczky Z, Mihály A, Pongor C, Révész Z, Somogyi Z, Erdő F. Characterization and ex vivo evaluation of excised skin samples as substitutes for human dermal barrier in pharmaceutical and dermatological studies. Skin Res Technol. 2022;28(5):664-676.
CrossRef - Ahmad N, Ahmad R, Buheazaha TM, AlHomoud HS, Al-Nasif HA, Sarafroz M. A comparative ex vivo permeation evaluation of a novel 5-Fluorocuracil nanoemulsion-gel by topically applied in the different excised rat, goat, and cow skin. Saudi J Biol Sci. 2020;27(4):1024-1040.
CrossRef - Jangdey MS, Gupta A, Saraf S. Fabrication, in-vitro characterization, and enhanced in-vivo evaluation of carbopol-based nanoemulsion gel of apigenin for UV-induced skin carcinoma. Drug Deliv. 2017;24(1):1026-1036.
CrossRef - Gupta V, Trivedi P. Ex vivo localization and permeation of cisplatin from novel topical formulations through excised pig, goat, and mice skin and in vitro characterization for effective management of skin-cited malignancies. Artif Cells Nanomed Biotechnol. 2015;43(6):373-382.
CrossRef - Slaoui M, Fiette L. Histopathology procedures: From tissue sampling to histopathological evaluation. In: Drug Safety Evaluation: Methods and Protocols. Springer; 2011:69-82.
CrossRef - Verma DD, Verma S, Blume G, Fahr A. Particle size of liposomes influences dermal delivery of substances into skin. Int J Pharm. 2003;258(1-2):141-151.
CrossRef - Danaei MR, Dehghankhold M, Ataei S, Hasanzadeh Davarani F, Javanmard R, Dokhani A, Khorasani S, Mozafari MR. Impact of particle size and polydispersity index on the clinical applications of lipidic nanocarrier systems. Pharmaceutics. 2018;10(2):57.
CrossRef - Liu X, Shen B, Shen C, Zhong R, Wang X, Yuan H. Nanoparticle-loaded gels for topical delivery of nitrofurazone: Effect of particle size on skin permeation and retention. J Drug Deliv Sci Technol. 2018;45:367-372.
CrossRef - Honary S, Zahir F. Effect of zeta potential on the properties of nano-drug delivery systems—a review (Part 2). Trop J Pharm Res. 2013;12(2):265-273.
CrossRef - Teng Z, Yu M, Ding Y, Zhang H, Shen Y, Jiang M, Liu P, Opoku-Damoah Y, Webster TJ, Zhou J. Preparation and characterization of nimodipine-loaded nanostructured lipid systems for enhanced solubility and bioavailability. Int J Nanomedicine. 2019;119-133.
CrossRef - Mahendra AG, Rasika DB. Formulation and evaluation of topical anti-inflammatory herbal gel. Asian J Pharm Clin Res. 2019;12(7):252-255.
CrossRef - Ashfaq R, Rasul A, Asghar S, Kovács A, Berkó S, Budai-Szűcs M. Lipid nanoparticles: An effective tool to improve the bioavailability of nutraceuticals. Int J Mol Sci. 2023;24(21):15764.
CrossRef - Unnisa A, Chettupalli AK, Alazragi RS, Alelwani W, Bannunah AM, Barnawi J, Amarachinta PR, Jandrajupalli SB, Elamine BA, Mohamed OA, Hussain T. Nanostructured lipid carriers to enhance the bioavailability and solubility of ranolazine: Statistical optimization and pharmacological evaluations. Pharmaceuticals. 2023;16(8):1151.
CrossRef - Ghadiri M, Fatemi S, Vatanara A, Doroud D, Najafabadi AR, Darabi M, Rahimi AA. Loading hydrophilic drug in solid lipid media as nanoparticles: Statistical modeling of entrapment efficiency and particle size. Int J Pharm. 2012;424(1-2):128-137.
CrossRef - Mehta M, Bui TA, Yang X, Aksoy Y, Goldys EM, Deng W. Lipid-based nanoparticles for drug/gene delivery: An overview of the production techniques and difficulties encountered in their industrial development. ACS Mater Au. 2023;3(6):600-619.
CrossRef - Ganeshpurkar A, Vaishya P, Jain S, Pandey V, Bansal D, Dubey N. Delivery of amphotericin B for effective treatment of Candida albicans induced dermal mycosis in rats via emulgel system: Formulation and evaluation. Indian J Dermatol. 2014;59(4):369-374.
CrossRef - Kulawik-Pióro A, Miastkowska M. Polymeric gels and their application in the treatment of psoriasis vulgaris: A review. Int J Mol Sci. 2021;22(10):5124.
CrossRef - Ahad A, Al-Saleh AA, Al-Mohizea AM, Al-Jenoobi FI, Raish M, Yassin AE, Alam MA. Formulation and characterization of Phospholipon 90 G and tween 80 based transfersomes for transdermal delivery of eprosartan mesylate. Pharm Dev Technol. 2018;23(8):787-793.
CrossRef - Algahtani MS, Ahmad MZ, Nourein IH, Albarqi HA, Alyami HS, Alyami MH, Alqahtani AA, Alasiri A, Algahtani TS, Mohammed AA, Ahmad J. Preparation and characterization of curcumin nanoemulgel utilizing ultrasonication technique for wound healing: In vitro, ex vivo, and in vivo evaluation. Gels. 2021;7(4):213.
CrossRef - Rancan F, Jurisch J, Günday C, Türeli E, Blume-Peytavi U, Vogt A, Schaudinn C, Günday-Türeli N. Screening of surfactants for improved delivery of antimicrobials and poly-lactic-co-glycolic acid particles in wound tissue. Pharmaceutics. 2021;13(7):1093.
CrossRef







