Manuscript accepted on :12-05-2025
Published online on: 20-05-2025
Plagiarism Check: Yes
Reviewed by: Dr. Mehdi Loukhnati and Dr. Ayan Chatterjee
Second Review by: Dr. Durgeshranjan Kar
Final Approval by: Dr. Anton R Keslav
Anton Sumarpo1 , Catherine Keiko Gunawan2
, Catherine Keiko Gunawan2 , Vegy Supriadi1, Erica Valencia Imannuel1
, Vegy Supriadi1, Erica Valencia Imannuel1 , Budiono Raharjo3
, Budiono Raharjo3 , Stephani Linggawan4
, Stephani Linggawan4 , Wivina Riza Devi5
, Wivina Riza Devi5 , Een Hindarsih6, Diane Lukito Setiawan7
, Een Hindarsih6, Diane Lukito Setiawan7 and Siprianus Ugroseno Yudho Bintoro8
and Siprianus Ugroseno Yudho Bintoro8
1Department of Clinical Pathology, Faculty of Medicine, Maranatha Christian University, Bandung, Indonesia
2Medical Profession Study Program, Faculty of Medicine, Maranatha Christian University, Bandung, Indonesia
3Department of Clinical Pathology, Faculty of Medicine, Universitas Wijaya Kusuma Surabaya, Surabaya, Indonesia
4Department of Ear, Nose, and Throat, Faculty of Medicine, Universitas Wijaya Kusuma Surabaya, Surabaya, Indonesia
5Department of Clinical Pathology, Lambung Mangkurat University, Banjarmasin, Indonesia
6Department of Internal Medicine, Siloam Hospital, Surabaya
7Department of Clinical Pathology, Siloam Hospital, Surabaya
8Department of Internal Medicine, Universitas Airlangga-Dr. Soetomo General Hospital, Surabaya
Corresponding Author E-mail:anton.sumarpo@med.maranatha.edu
DOI : https://dx.doi.org/10.13005/bpj/3212
Abstract
Multiple myeloma (MM) is a hematological malignancy characterized by the clonal proliferation of plasma cells and the excessive accumulation of monoclonal protein. While primary manifestations such as bone pain, renal dysfunction, anemia are well-documented features, extra-hematological complications, including achalasia, are exceedingly rare. We present a 76-year-old male with 10-month history of persistent dysphagia. Laboratory test revealed anemia, leukocytosis, and neutrophilia, while serum protein electrophoresis indicated dysproteinemia. Bone marrow aspiration confirmed MM with 25% plasma cell infiltration. Imaging and endoscopic findings further identified a lytic cervical spine lesion and esophageal motility dysfunction consistent with achalasia. This case presents a rare presentation extra-hematological presentation of MM, emphasizing the need for a comprehensive diagnostic approach to facilitate timely recognition and appropriate management of MM.
Keywords
Achalasia; Clonal proliferation; Dysphagia; Extra-hematological presentation; Multiple myeloma
Download this article as:| Copy the following to cite this article: Sumarpo A, Gunawan C. K, Supriadi V, Immanuel E. V, Raharjo B, Linggawan S, Devi W. R, Hindarsih E, Setiawan D. L, Bintoro S. U. Y. Achalasia: A Rare Manifestation of Multiple Myeloma - A Case Report. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Sumarpo A, Gunawan C. K, Supriadi V, Immanuel E. V, Raharjo B, Linggawan S, Devi W. R, Hindarsih E, Setiawan D. L, Bintoro S. U. Y. Achalasia: A Rare Manifestation of Multiple Myeloma - A Case Report. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/43sL6bq |
Introduction
Multiple myeloma (MM) is a hematological malignancy characterized by the clonal proliferation of ≥10% plasma cells within the bone marrow, leading to the accumulation of monoclonal proteins in the serum and/or urine.1,2 Clinically, MM presents with a diverse spectrum of complications, including malignant bone lesions, renal dysfunction, anemia, hypercalcemia and pain-associated fractures.1,2 Despite its well-characterized pathological features, the rare extra-hematological complications of MM remain insufficiently understood.3,4 Achalasia, a rare eosophageal motility disorder, represents an exceptionally uncommon findings in MM.3,4 This disorder is characterized by the progressive degeneration of enteric neurons, resulting in the absence of peristalsis and impaired relaxation of the lower eosophageal sphincter (LES).5,6 Consequently, affected individuals experience dysphagia, regurgitation of undigested food, chest pain, weight loss and respiratory issues such as nocturnal cough, recurrent aspiration, and pneumonia. The underlying pathophysiology of achalasia is attributed to the selective loss of noncholinergic, noradrenergic inhibitory ganglion cells, while excitatory neurons remain functionally preserved.5,6 This imbalance results in a hypertensive eosophageal sphincter that fails to undergo proper relaxation.4–6 This case report presents a rare instance of the co-occurence of MM and achalasia in a 76-year-old male, potentially contributing to a broader understanting of the intricate interplay between hematological malignancies and rare neuromusculural disorders.
Case Report
A 76-year old male was admitted to our hospital in March 2024 with a 10-month history of progressive dysphagia, significantly impairing his ability to eat and swallow. The patient had no prior gastrointestinal complaints.
Laboratory examination revealed anemia (hemoglobin level of 7.7 g/dL; normal: 13.2-17.3 g/dL), leukocytosis (white blood cell count of 21.68×109/L; normal: 3.80-10.60 x 109/L), neutrohpilia (neutrophil count of 18.37 x 103/µL; normal: 2,000-7,000/µL), and a markedly increased erythrocyte sedimentation rate (115 mm; normal: 0-10 mm). Kidney function test indicated impaired renal function, evidenced by an elevated urea concentration (68.7 mg/dL, normal: 20-50 mg/dL), an increased creatinine level (1.19 mg/dL, normal: 0.67-1.17 mg/dL), and a reduced glomerular filtration rate (58.57 mL/min/1.73 m^2). Serum protein electrophoresis demonstrated elevated alpha-1, alpha-2, and gamma globulin fractions, suggestive of dysproteinemia (Table 1).
Table 1: Electrophoresis analysis demonstrated dysproteinemia
| Fraction | Measured (%) | Normal range (%) | Concentration (g/dL) | Normal range (g/dL) | Interpretation |
| Albumin | 32.8 | 55.8-66.1 | 2 | 4.0 – 4.8 | Decreased |
| Alpha-1 globulin | 9.2 | 2.9-4.9 | 0.6 | 0.2 – 0.4 | Elevated |
| Alpha-2 globulin | 12.2 | 7.1-11.8 | 0.7 | 0.5 – 0.9 | Elevated |
| Beta-1 globulin | 6.5 | 4.7-7.2 | 0.4 | 0.3 – 0.5 | Normal |
| Beta-2 globulin | 7.3 | 3.2-6.5 | 0.4 | 0.2-0.5 | Elevated |
| Gamma globulin | 32 | 11.1-18.8 | 1.9 | 0.8-1.4 | Significantly elevated |
The persistence of dysphagia necessitated an upper gastrointestinal endoscopy which revealed food retention, reduced peristaltic movement, and increased tone of the distal esophageal sphincter, resulting in contstriction at the eosophago-gastric junction. Further evaluation with magnetic resonance imagine (MRI) demonstrated diffuse disc bulging between the C3 and C7 vertebrae, which may have contributed to the dysphagia. Additionally, multi-sliced computed tomography (CT) imaging confirmed the diagnosis of primary achalasia, characterized by obstruction at the gastrophageal junction.
Bone marrow aspiration revealed hypercellularity with decreased erythropoiesis, increased granulopoiesis, and increased thrombopoiesis. Additionally, plasma cell infiltration was observed, comprising 25% of the total bone marrow cellularity (Figure 1).
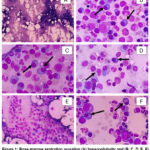 |
Figure 1: Bone marrow aspiration revealing (A) hypercellularity and (B, C, D, E, F) plasma cell infiltration, indicative of clonal proliferation (black arrow). |
Based on the laboratory examination, imaging studies, and bone marrow aspirations, the patient was diagnosed with multiple myeloma (MM) complicated by achalasia.
The patient received a blood transfusion and was initiated on a regimen of lenalidomide, administered once daily for a week, in conjunction with dexamethasone (20 mg daily divided into three dosage). Additionally, supportive therapy with esomeprazole, vitamin D, and celecoxib was provided for symptomatic management.
Discussion
This case highlights the diagnostic challenge presented by achalasia occurring in a patient with multiple myeloma (MM), an exceptionally rare association. Multiple myeloma (plasma cell myeloma, or Kähler’s disease) is the second most common hematological malignancy of hematopoietic origin, accounting for approximately 1% of all malignancies and predominantly affecting individual aged 65-74.7,8 Multiple myeloma (MM) is characterized by the neoplastic proliferation of a single clone of plasma cells derived from B-cells within the bone marrow, leading to the excessive production of an abnormal immunoglobulin known as monoclonal protein (M protein) and free light chains, known as either kappa or lambda.9 The diagnosis is established by the presence of ≥10% clonal plasma cells in the bone marrow aspirates, in conjunction with at least one multiple myeloma-defining event (MDE), including hypercalcemia, renal dysfunction, anemia, and/or lytic bone lesions. Additionally, MM may be identified through specific biomarkers, including clonal plasmacytosis in the bone marrow ≥ 60%, a free light chain ratio (involved/uninvolved) ≥100 (with involved FLC ≥100 mg/L), or multiple focal lesions observed on MRI.7 While primary (idiopathic) achalasia involves degeneration of esophageal inhibitory neurons, achalasia presenting in the context of malignancy is often considered secondary or ‘pseudoachalasia’. The precise mechanisms linking MM to esophageal dysmotility are not fully elucidated, but potential indirect pathways related to MM complications warrant consideration in this case.10
In this 76-year-old male, investigations revealed findings consistent with achalasia, including dysphagia, endoscopic evidence of reduced peristalsis and lower esophageal sphincter (LES) dysfunction, and CT confirmation of obstruction at the gastroesophageal junction. Several factors potentially link his MM to these findings. Firstly, MRI revealed significant cervical spine abnormalities, including diffuse disc bulging (C3-C7) causing anterior dural sac compression and bilateral uncovertebral spondylosis leading to neural foraminal stenosis. Such structural changes, potentially exacerbated by underlying MM bone involvement, could plausibly lead to extrinsic nerve compression or dysfunction affecting esophageal motility.11 Amyloid accumulation in the esophagus submucosal layer further leads to decreased motility, and increased muscle rigidity and affects the function of myenteric plexus and vagal nerve.6 Confirmation of achalasia was obtained through CT-scan, which revealed an obstruction at the gastroesophageal junction. Endoscopic evaluation provided further diagnostic support, demonstrating reduced peristalsis, elevated pressure in the distal esophageal sphincter, and narrowing of the esophagogastric junction. This aligns with the possibility that complications related to MM’s effects on bone and surrounding structures contribute to achalasia.12
Secondly, systemic amyloidosis (specifically AL type) is a known complication of MM and can infiltrate various organs, including the gastrointestinal tract.13 Amyloid deposition within the esophageal wall can directly impair neuromuscular function by affecting the myenteric plexus and vagal nerve, leading to reduced motility, increased muscle rigidity, and symptoms mimicking primary achalasia.14 Although amyloid deposition was not biopsy-proven in the esophagus in this patient, it remains a significant potential indirect mechanism contributing to the observed dysphagia and achalasia, given the established MM diagnosis.
A third, though less defined possibility, involves paraneoplastic syndromes, where autoimmune responses triggered by the malignancy affect the nervous system remotely. While specific paraneoplastic antibodies were not assessed here, such syndromes are known to occur with MM and can cause neurological dysfunction, potentially contributing to esophageal dysmotility in some cancer patients.15
Through the laboratory findings, we observe the presence of moderate anemia, leukocytosis and neutrophilia. While anemia is a well-documented hallmark of multiple myeloma (MM), the presence of leukocytosis and neutrophilia in this case contrast with previous findings.16 The development of anemia in MM is primarily linked to the clonal expansion of malignant plasma cells, which infiltrate the bone marrow and suppresses normal hematopoiesis.16 Conversely, leukopenia and neutropenia are more commonly associated with MM, while the underlying mechanism of leukocytosis and neutropenia in this case remain incompletely understood. However, existing literature suggest that these hematological abnormalities may result from an inflammatory response, potentially induced by the malignancy itself or its associated complications.17
The diagnostic approach encompasses not only laboratory assessments and imaging studies, but also serum protein electrophoresis, which plays a crucial role in identifying hematological abnormalities. In this case, electrophoresis revealed elevated in the alpha-1, alpha-2, and gamma fractions—findings frequently associated with multiple myeloma (MM). The increased levels of alpha-globulins are indicative of both malignancy and acute inflammation, driven by acute-phase reactants.18 Meanwhile, the elevated gamma fraction primarily reflects the excessive production of monoclonal immunoglobulin (M-protein) by malignant plasma cells. Wijinands et al 19 identified serum protein electrophoresis as the gold standard for detecting M-protein, which is characterized by a distinct, well-defined band consisting of a single heavy chain and a corresponding kappa or lambda light chain. Furthermore, as reported by Liyasova et al., 20 M-protein serves as a critical biomarker for monitoring disease progression in MM. Bone marrow aspiration findings in this case demonstrated hypercellularity with decreased erythropoiesis, increased granulopoiesis and thrombopoiesis, and plasma cell infiltration accounting for 25% of total cellularity—findings consistent with a diagnosis of multiple myeloma. On the other side, while a direct causal effect of MM plasma cells on esophageal neurons is uncertain and less likely, the achalasia observed in this patient is reasonably considered secondary to MM-related complications. This may involve a combination of structural nerve compression from spinal pathology and/or underlying systemic processes like amyloidosis. Differentiating primary achalasia from pseudoachalasia remains challenging but is crucial for appropriate patient management and prognostic assessment.21
Recent advancement in understanding the cellular and molecular mechanisms underlying multiple myeloma (MM) have facilitated the development of a more effective therapeutic strategies, which include chemotherapy, stem cell transplantation, and various pharmacological agents such as corticosteroid, anti-cancer drugs, proteasome inhibitors, and immunomodulatory drugs.22 The introduction of targeted therapies, particularly proteasome inhibitors and immunomodulatory drugs, in combination with high-dose melphalan and autologous stem cell transplantation, has significantly improved patient outcomes. In this case, the patient was prescribed lenalidomide, an immunomodulatory drug (IMiDs) known to regulate the immune response by altering T cell activity and cytokine production, while also inducing apoptosis through disruption of immune homeostasis.23 Additionally, dexamethasone, a corticosteroid commonly used in the induction phase of MM treatment before stem cell transplantation, was administered. The therapeutic efficacy of dexamethasone in MM is primarily attributed to its ability to promote apoptosis in malignant plasma cells. This mechanism is mediated through the suppression of pro-inflammatory cytokines via inhibition of nuclear factor-kappa B (NF-κB), a transcription factor essential for cytokine activation and the regulation of genes involved in cell survival and proliferation.24
Conclusion
Achalasia in the context of multiple myeloma is a rare clinical manifestation, posing considerable challenges to clinician in both accurately determining the diagnosis and implementing effective management implication to enhance life expectancy.
Acknowledgement
The authors extend their heartfelt thanks to Susi Efawati from the Clinical Pathology Laboratory at Mitra Keluarga Hospital in Surabaya, Indonesia, for her invaluable assistance in collecting the laboratory data that contributed to the development of this case report.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The present case report was approved by The Health Research Ethics Committee, Faculty of Medicine, Universitas Wijaya Kusuma Surabaya (approval no. 43/SLE/FK/UWKS/2025; Surabaya, Indonesia).
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to Reproduce Material from Other Sources
Not applicable
Author Contributions
- Anton Sumarpo: Conceptualization, formal analysis, data curation, supervision, writing original draft, review, and editing
- Catherine Keiko Gunawan: Conceptualization, formal analysis, data curation, supervision, writing original draft, review, and editing
- Vegy Supriadi: Conceptualization, formal analysis, data curation, supervision, writing original draft, review, and editing
- Erica Valencia Imannuel: Conceptualization, formal analysis, data curation, supervision, writing original draft, review, and editing
- Budiono Raharjo: Investigation, patient administration, software, visualization, validation, review, and editing
- Stephani Linggawan: Investigation, patient administration, software, visualization, validation, review, and editing
- Wivina Riza Devi: Investigation, patient administration, software, visualization, validation, review, and editing
- Een Hindarsih: Investigation, patient administration, software, visualization, validation, review, and editing
- Diane Lukito Setiawan: Investigation, patient administration, software, visualization, validation, review, and editing
- Siprianus Ugroseno Yudho Bintoro: Investigation, patient administration, software, visualization, validation, review, and editing
References
- Gandolfi S, Prada CP, Richardson PG. How I treat the young patient with multiple myeloma. Blood. 2018;132(11):1114-1124.
CrossRef - Abduh MS. An overview of multiple myeloma: A monoclonal plasma cell malignancy’s diagnosis, management, and treatment modalities. Saudi J Biol Sci. 2024;31(2):1-18.
CrossRef - Ameen Z. Alherabi, Ali M. Khan, et al. Abdulfattah. Multiple myeloma presenting as dysphagia. Saudi Med J. 2013;34(6):648-650
- Padala SA, Barsouk A, Barsouk A, et al. Epidemiology, staging, and management of multiple myeloma. Med Sci (Basel). 2021;9(1):3
CrossRef - Monteith BE, Sandhu I, Lee AS. Management of multiple myeloma: A review for general practitioners in oncology. Curr Oncol. 2023;30(5):4382-4401.
CrossRef - Cowan AJ, Green DJ, Kwok M, et al. Diagnosis and management of multiple myeloma: A review. JAMA. 2022;327(5):464-477
CrossRef - Guedes A, Becker RG, Teixeira LEM. Multiple myeloma (part 1) – Update on epidemiology, diagnostic criteria, systemic treatment and prognosis. Rev Bras Ortop. 2022;58(3):361-367.
CrossRef - Monteith BE, Sandhu I, Lee AS. Management of multiple myeloma: A review for general practitioners in oncology. Curr Oncol. 2023;30(5):4382-4401.
CrossRef - Hussain A, Almenfi HF, Almehdewi AM, et al. Laboratory features of newly diagnosed multiple myeloma patients. Cureus. 2019;11(5):e4716.
CrossRef - Touzeau C, Moreau P. How I treat extramedullary myeloma. Blood. 2016;127(8):971-975.
CrossRef - Ailawadi S, Cheema BK, Syed Salahuddin S, et al. A rare case of gastrointestinal amyloidosis presenting as dysphagia. Cureus. 2022;14(2):1-5.
CrossRef - Lazaraki G, Nakos A, Katodritou E, et al. A rare case of multiple myeloma initially presenting with pseudoachalasia. Dis Esophagus. 2009;22(6):E21-4. doi:10.1111/j.1442-2050.2008.00903.x
CrossRef - Bou Zerdan M, Nasr L, Khalid F, et al. Systemic AL amyloidosis: current approach and future direction. Oncotarget. 2023;14:384-394. doi:10.18632/oncotarget.28415
CrossRef - Ates F, Vaezi MF. The pathogenesis and management of achalasia: current status and future directions. Gut Liver. 2015;9(4):449-463. doi:10.5009/gnl14446
CrossRef - Darnell RB, Posner JB. Paraneoplastic syndromes affecting the nervous system. Semin Oncol. 2006;33(3):270-298. doi:10.1053/j.seminoncol.2006.03.008
CrossRef - Givi JP, Williams N, Hass R, et al. Educational case: multiple myeloma. Acad Pathol. 2024;11(2) :100117.
CrossRef - Chabot-Richards DS, George TI. Leukocytosis. Int J Lab Hematol. 2014;36(3):279-288.
CrossRef - Tripathy S. The role of serum protein electrophoresis in the detection of multiple myeloma: An experience of a corporate hospital. J Clin Diagn Res. 2012;6(9):1458-1461..
CrossRef - Wijnands C, Langerhorst P, Noori S, et al. M-protein diagnostics in multiple myeloma patients using ultra-sensitive targeted mass spectrometry and an off-the-shelf calibrator. Clin Chem Lab Med. 2023;62(3):540-550.
CrossRef - Liyasova M, McDonald Z, Taylor P, et al. A personalized mass spectrometry–based assay to monitor M-protein in patients with multiple myeloma (EasyM). Clin Cancer Res. 2021;27(18):5028-5037.
CrossRef - Vaezi MF, Pandolfino JE, Yadlapati RH, et al. ACG clinical guidelines: diagnosis and management of achalasia. Am J Gastroenterol. 2020;115(9):1393-1411. doi:10.14309/ajg.0000000000000731
CrossRef - Pinto V, Bergantim R, Caires HR, Seca H, Guimarães JE, Vasconcelos MH. Multiple Myeloma: Available Therapies and Causes of Drug Resistance. Cancers (Basel). 2020;12(2):407. doi:10.3390/CANCERS12020407
CrossRef - Abduh MS. An overview of multiple myeloma: A monoclonal plasma cell malignancy’s diagnosis, management, and treatment modalities. Saudi J Biol Sci. 2024;31(2). doi:10.1016/J.SJBS.2023.103920
CrossRef - Rosenberg AS. From mechanism to resistance – changes in the use of dexamethasone in the treatment of multiple myeloma. Leuk Lymphoma. 2023;64(2):283-291.
CrossRef







