Manuscript accepted on :September 06, 2017
Published online on: --
Plagiarism Check: Yes
Md. Sarwar Hossain1, Md. Sahab Uddin1, Md. Tanvir Kabir2, Mst. Marium Begum3, Paras Koushal4, Oscar Herrera-Calderon5, Raushanara Akter2, Md. Asaduzzaman1 and Mohamed M. Abdel-Daim6
1Department of Pharmacy, Southeast University, Dhaka, Bangladesh.
2Department of Pharmacy, BRAC University, Dhaka, Bangladesh.
3Department of Pharmacy, East West University, Dhaka, Bangladesh.
4Department of Pharmacology, Maharishi Markendeshwar Institute of Medical Sciences, Haryana, India.
5Department of Pharmaceutical Sciences, Faculty of Pharmacy and Biochemistry, Universidad Nacional San Luis Gonzaga de Ica, Ica, Peru.
6Pharmacology Department, Faculty of Veterinary Medicine, Suez Canal University, Ismailia, Egypt.
Corresponding Author E-mail: msu-neuropharma@hotmail.com
DOI : https://dx.doi.org/10.13005/bpj/1229
Abstract
Searching for natural curative agents with a superior safety profile for the management of oxidative stress linked disorders are continuing owing to multiple unwanted effects linked with synthetic antioxidant. Medicinal plants are still considered as one of the imperative cradles of modern medicine. Therefore the drive of this study was to analyse the phytochemical constituents and antioxidant activities of the Syngonium podophyllum (S. podophyllum) L. leaves. The fresh leaves of S. podophyllum L. were extracted with methanol (CME) afterward fractionation using n-hexane (NHF), chloroform (CLF), ethyl acetate (EAF) and aqueous (AQF). The plant extract and its fractions were evaluated for phytochemical content by alkaloids, carbohydrates, saponins, tannins, resins, flavonoids and steroids tests. Total phenolic content (TPC) was also determined. Antioxidant activities were determined by total antioxidant activity (TAA), reducing power activity (RPA) and DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging activity tests. The phytochemical analysis of CME and its fractions of S. podophyllum L. leaves exposed the presence of alkaloids, tannins, resins, flavonoids and steroids. The CME of S. podophyllum L. leaves exhibited the highest amount (10.55 mg of GAE/g of dried extract) of TPC compared to remaining fractions. The highest absorbance, 3.791 nm was found in CME with respect to remaining fractions for TAA test. Among the crude extract and its different fractions, highest absorbance, 3.015 nm was reported for CME in RPA test. In the DPPH radical scavenging test, CME showed the highest scavenging activity (79.89%) with IC50 of 46.55 μg/mL. This study indorsed that CME has marked antioxidant activities. This plant extract might be a promising source of natural antioxidant which can be effective for the treatment of oxidative stress related disorders.
Keywords
Syngonium podophyllum; Antioxidant; Oxidative stress; Safety profile; Synthetic antioxidant
Download this article as:| Copy the following to cite this article: Hossain M. S, Uddin M. S, Kabir M. T, Begum M. M, Koushal P, Herrera-Calderon O, Akter R, Asaduzzaman M, Abdel-Daim M. M. In Vitro Screening for Phytochemicals and Antioxidant Activities of Syngonium Podophyllum L.: an Incredible Therapeutic Plant. Biomed Pharmacol J 2017;10(3). |
| Copy the following to cite this URL: Hossain M. S, Uddin M. S, Kabir M. T, Begum M. M, Koushal P, Herrera-Calderon O, Akter R, Asaduzzaman M, Abdel-Daim M. M. In Vitro Screening for Phytochemicals and Antioxidant Activities of Syngonium Podophyllum L.: an Incredible Therapeutic Plant. Biomed Pharmacol J 2017;10(3). Available from: http://biomedpharmajournal.org/?p=16606 |
Introduction
Traditionally, usage of different medicinal plants to treat a number of diseases is well established.1 Surprisingly, 80% of the total global population is primarily dependent on traditional medicines to treat various maladies.2 In India, since ancient times, it is well established that thousands of species have known medicinal values and different parts of these plants are used to alleviate specific illnesses.3 Medicinal plants are considered as a precious natural resource. Generally, medicinal plants are considered as much safer drugs and over the time their biological, antioxidant, hypoglycemic and antimicrobial effects have been tested and they are often found to play a vital role in the modern medicine.4,5 It is also often found that most of the synthetic drugs that we have today have their origin from medicinal plants.6 Currently scientific interest has been particularly flourished due to the improved efficiency of plant derived products and their comparatively lesser side effects than modern medicine. Free radicals including hydroxyl radical (•OH), hydrogen peroxide (H2O2), superoxide radical (O2-•) and singlet oxygen (1O2) when generated in great amount in human body, can exert toxic effects like oxidative stress.7-9 These free radicals are highly reactive at larger amount and during oxidative stress they are strong enough to damage body’s proteins, lipids and even deoxyribonucleic acid molecules that can ultimately lead to a range of disorders including anemia, diabetes, allergies, inflammation, ischemia, kidney failure, liver and cardiovascular diseases and various degenerative diseases of the nerve and even cancer.10-12 Nevertheless, at low level reactive oxygen species is essential to maintain various normal physiological and biochemical functions including cell signaling, apoptosis of defective cells and so on.10-12 On the other hand, compounds which are obtained from medicinal plant are found to prevent the harmful effects of oxidative stress. In addition, these compounds are cheaper to obtain and have lesser side effects than synthetic antioxidant agents.13-17 However, medicinal plants are also found to produce a great number of secondary metabolites that contain various physiological and biochemical effects.18 Other than their antioxidant effects, these secondary metabolites and their derivatives also contain significant pharmacological effects such as diuretic, anti-hepatotoxic, anti-allergic and anticancer effects.19-21 Strong antioxidant activity containing phytochemicals including tocopherols, ascorbates, phenols, carotenoids and tocotrienols have already well-established their imperative roles in health care system.22 Presently, there is an increased research interest in the discovery of new natural antioxidant agents from medicinal plant. Furthermore, the demand of these antioxidant agents is rising not only in pharmaceutical industry but also in food and cosmetic sector. Medicinal plant contains antioxidant compounds which show a protective role by averting the production of free radicals within the cell.23-25 The plant, Syngonium podophyllum (S. podophyllum) L. belongs to the family of Araceae, which is a simple yet elegant looking and commonly used as an ornamental plant.26 The species is mainly native to a diverse region of Latin America from Mexico to Bolivia. Furthermore, the species is naturalized in the Florida, Hawaii, Texas, West Indies and many other places.27 A decoction of the plant’s crushed and boiled leaf is used as a remedy to treat stomachache.28 In addition, milky-white sap from a broken stem of the plant is applied on skin as a topical agent for the bite of Paraponera ants.28 The sap is also applied into the cavity of an aching tooth in order to relieve toothache.28 The roots and bark have antibacterial and anti-inflammatory activity; hence they are used to treat superficial and deep wound, and several skin disorders.28 Moreover, extracts of the leaves and bark have significant anti-inflammatory activity and possess a dose-dependent effect upon treating edema.28 In the local folk medicine of Belize, traditionally leaves and bark of the plant have been found to be used due to their wound healing effects, while the tincture of the leaf is used to treat arthritis, rheumatism, swelling and pain.29 The literature is indigent about biochemical actions of S. podophyllum L. leaves. In our previous study we examined the analgesic and anti-inflammatory activities of this plant.30 Hence the present study was conducted to evaluate the phytochemical content and antioxidant activities of S. podophyllum L. leaves.
Materials and Methods
Chemicals
Folin-Ciocalteu (FC) reagent, ammonium molybdate, ascorbic acid (ACA), 2,2‐diphenyl‐1‐picrylhydrazyl (DPPH), 2-deoxy-D-ribose, EDTA (ethylenediaminetetraacetic acid), trichloro aceticacid (TCA), thiobarbituric acid (TBA), methanol and quercetin (QR) were purchased from Sigma-Aldrich, USA. Sulphuric acid and butylatedhydroxy toluene (BHT) were purchased from Merck, Germany. Gallic acid (GA) was purchased from Wako pure chemicals Ltd., Japan. Unless otherwise specified, all other chemicals were of analytical grade and purchased from Active Fine Chemicals Ltd., Bangladesh.
Collection and Identification of Plant Materials
In the month of January 2016, the leaves of S. podophyllum L. were entirely collected from Dhaka city of Bangladesh. For the plant identification purposes, help from an expert of Bangladesh National Herbarium, Mirpur, Dhaka, Bangladesh was taken. Additionally, for future references, the herbarium also preserved a voucher specimen (DACB-38722).
Drying and Grinding of Plant Materials
Freshly collected leaves of S. podophyllum L. were washed properly to eliminate any possible dirty materials. Washed leaves were then shade dried for several days with sporadic sun drying. Then the dried leaves were grinded to make powder and then labeled and hermetic for further use.
Extraction and Fractionation of Plant Materials
Powdered plant materials about 500 g were extracted with 1.5 L of 98% methanol in an amber colored glass bottle for 7 days and filtered by cotton and then through Whatman No.1 filter paper. The extracts were then subjected to rotary evaporator at 50°C temperature to evaporate methanol and give crude extract (10.45 g). This crude methanol extract (CME) was subjected to sequential fractionated, at first with n-hexane (NHF) after that chloroform (CLF) then ethyl acetate (EAF) and lastly by using water (AQF) consistent with the method of Kupchan with minor modification.31 The generated methanol extract and its fractions were re-filtered and then evaporated to remove extra solvent and obtained NHF, 2.95 g; CLF, 2.58 g; EAF, 1.09 g and AQF 3.38 g extract respectively.
Phytochemical Content
Phytochemical screening was carried out on CME and its four fractions (NHF, CLF, EAF, AQF) to detect the presence of secondary metabolites such as alkaloids, carbohydrates, saponins, tannins, resins, flavonoids and steroids employing standard phytochemical screening.32
Total Phenolic Content
The method as described by Singleton and Rossi with slight modification was used to determine the total phenolic content (TPC) of S. podophyllum L. leaves extract and its fraction(s)33. Although previously diluted 1000-fold using distilled water, 0.1 mL of plant extract/fraction(s) with a concentration of 1000 µg/mL was mixed with 0.75 mL of Folin-ciocalteu reagent. Followed by 5 min incubation period, 0.06% sodium carbonate solution was added; afterward again incubated for the period of 90 min at 22°C. A spectrophotometer (at 725 nm) was used to estimate the absorbance of the solution against a reagent blank that is a solution contained all the reagents excluding plant extract/fraction(s) or standard (GA). The results of dried sample were denoted as mg of gallic acid equivalents (GAE)/g.
Antioxidant Activity
In order to start antioxidant activity a solution of each obtained plant extract/fractions was made by using 98% methanol at 1 mg/mL concentration.
Total Antioxidant Activity
The prescribed method of Prieto et al., with minor adjustments was used to determine the total antioxidant activity (TAA) of different extractives of S. podophyllum L. leaves extract and its fraction(s)34. 0.5 mL of S. podophyllum L. extract/fraction(s) or standard [(+)-catechin, CTC] solution of different concentration was taken and mixed with 3 mL of reagent solution containing 0.6 M sulfuric acid, 28 mM sodium phosphate and 4 mM ammonium molybdate. The absorbance of the solution was determined by a spectrophotometer (at 695 nm) against a reagent blank that is a solution contained all reagents excluding plant extract/fraction(s) or standard and the whole spectrophotometric analysis was followed by an incubation period of 15 min at 95°C.
Reducing Power Activity
The reducing power activity (RPA) of S. podophyllum L. leaves extract and its fraction(s) was evaluated by the method of Oyaizu with minor modifications35. 0.2 mL of plant extract/fraction(s) or standard (ACA) solution of different concentration was taken and mixed with 0.5 mL of 200 mM/L trisodium phosphate buffer (pH 6.6) and 2.5 mL of potassium ferricyanide (1%). The mixture was incubated for 20 min at 50°C then 2.5 mL of trichloroacetic acid (10%) was added to the mixture, which was then centrifuged at 650 rpm for 10 min. From the upper layer of the solution, 0.5 ml was mixed with 0.5 mL distilled water and 0.1 mL of ferric chloride (0.1%). After that the absorbance of the solution was measured at 700 nm using a spectrophotometer against a reagent blank that is a solution contained all reagents except plant extract/fraction(s) or standard.
DPPH Radical Scavenging Activity
The method described by Choi et al., with minor modifications, was used to determine the DPPH radical scavenging activity of S. podophyllum L. leaves extract and its fraction(s). 2 mL of plant extract/fraction(s) or standard (BHT) solution of different concentration was added to 3 ml 0.02 % of methanol solution of DPPH. Before measuring the absorbance of the solution, it was incubation in a dark place for 30 min. Subsequently, absorbance of the solution was determined at 517 nm using a spectrophotometer against a reagent blank that is a solution contained all reagents except plant extract/fraction(s) or standard. The percentage of scavenging of the DPPH free radical was calculated using the following equation: DPPH radical scavenging (%) = [1– (A/Ao)] ×100 Where, Ao is the absorbance of control and A is the absorbance of extract/fraction(s) or standard.
Statistical Analysis
The experiments were carried out in triplicate and all the results were expressed as mean ± SD. The data in all the experiments were analyzed by Student’s t test. For the statistical and graphical evaluations SPSS 14.0 (Chicago, IL, USA) and Microsoft Excel 2010 (Roselle, IL, USA) were used. The level of significance was considered at P < 0.05.
Results Determination of Phytochemical Content
Phytochemical examination of S. podophyllum L. leaves extract and its fractions revealed the presence or absence of bioactive components such as alkaloids, carbohydrates, saponins, tannins, resins, flavonoids and steroids showed in Table 1. Saponin and carbohydrates are absent in crude extract and it’s all fractions but others are present at various concentration (i.e. largely, moderately, mildly).
Table 1: Phytochemical screening of S. podophyllum L. leaves extract and its fractions.
| Phytochemicals | CME | NHF | CLF | EAF | AQF |
| Alkaloids | ++ | − | + | ++ | ++ |
| Carbohydrates | − | − | − | − | − |
| Saponins | − | − | − | − | − |
| Tannins | + | + | + | + | − |
| Resins | +++ | + | +++ | − | − |
| Flavonoids (Ammonia test) | ++ | + | − | + | − |
| Flavonoids (Aluminium chloride solution test) | + | + | − | − | − |
| Steroids | ++ | ++ | ++ | − | − |
Based on intensity of the color reaction where, + = Present in mild amount, ++ = Present in moderate amount, +++ = Present in large amount, − = Not present. Where, CME: Crude methanol extract; NHF: n-hexane fraction; CLF: Chloroform fraction; EAF: Ethyl acetate fraction; AQF: Aqueous fraction.
Determination of TPC
The TPC of S. podophyllum L. leaves extract and its fractions was calculated on the basis of the standard curve for GA (y = 0.0144x + 0.0328; R2 = 0.9991) that is expressed in mg of GAE/g of dried sample shown in Figure 1. The CME of S. podophyllum L. leaves exhibited the highest amount (10.55±0.41 mg of GAE/g of dried extract) of total phenolics, followed by NHF, CLF, EAF and AQF, which were 7.56±0.51, 6.22 ±0.26, 5.31±0.28 and 2.55±0.38 mg of GAE/g of dried extract, respectively.
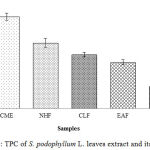 |
Figure 1: TPC of S. podophyllum L. leaves extract and its fractions. |
Values were reported as mean ± SD (n = 3). Where, CME: Crude methanol extract; NHF: n-hexane fraction; CLF: Chloroform fraction; EAF: Ethyl acetate fraction; AQF: Aqueous fraction.
Determination of TAA
In the TAA, at maximum concentration (600 µg/mL), the absorbance of CME and CAT were 3.916±0.006 and 3.988±0.003 nm respectively that is showed in Figure 2. Among the crude methanol extract and its different fractions, CME showed the highest TAA followed by CLF, EAF, AQF and NHF with absorbance’s which are 3.791±0.005, 2.788±0.009, 1.505±0.012 and 1.440±0.006 nm respectively at maximum concentration.
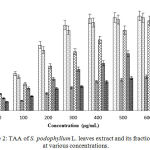 |
Figure 2: TAA of S. podophyllum L. leaves extract and its fractions at various concentrations. |
Values were reported as mean ± SD (n = 3). Where, CTC: (+)-catechin; CME: Crude methanol extract; NHF: n-hexane fraction; CLF: Chloroform fraction; EAF: Ethyl acetate fraction; AQF: Aqueous fraction.
Determination of RPA
The RPA of CME, NHF, CLF, EAF and AQF of leaves of S. podophyllum L. are showed in Figure 3. The extract/fractions increased the reducing activity with the increased concentration of the extract/fractions. Among the CME and its different fractions, CME showed the highest iron RPA with absorbance of 3.015±0.006 nm followed by EAF, CLF, AQF and NHF which are 2.477±0.004, 1.898±0.009, 1.569±0.007 and 0.929±0.006 nm respectively at maximum concentration (600 µg/mL).
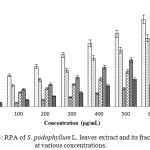 |
Figure 3: RPA of S. podophyllum L. leaves extract and its fractions at various concentrations. |
Values were reported as mean ± SD (n = 3). Where, ACA: Ascorbic acid; CME: Crude methanol extract; NHF: n-hexane fraction; CLF: Chloroform fraction; EAF: Ethyl acetate fraction; AQF: Aqueous fraction.
Determination of DPPH Radical Scavenging Activity
The DPPH radical scavenging activity of S. podophyllum L. leaves extract and its fractions is given in Figure 4. Among the crude extracts and its fractions the most potent activity was found in CME at highest concentration having IC50 value of 46.55±2.25 mg/mL (Figure 5). On the other hand, NHF, CLF, EAF and AQF showed DPPH radical scavenging activity with IC50 value of 394.01±4.27, 99.78±1.29, 96.58±2.88 and 98.31±2.74 mg/mL respectively.
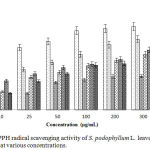 |
Figure 4: DPPH radical scavenging activity of S. podophyllum L. leaves extract and its fractions at various concentrations. |
Values were reported as mean ± SD (n = 3). Where, BHT: Butylatedhydroxy toluene; CME: Crude methanol extract; NHF: n-hexane fraction; CLF: Chloroform fraction; EAF: Ethyl acetate fraction; AQF: Aqueous fraction.
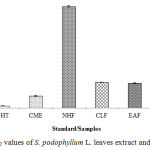 |
Figure 5: IC50 values of S. podophyllum L. leaves extract and its fractions. |
Values were reported as mean ± SD (n = 3). Where, BHT: Butylatedhydroxy toluene; CME: Crude methanol extract; NHF: n-hexane fraction; CLF: Chloroform fraction; EAF: Ethyl acetate fraction; AQF: Aqueous fraction.
Discussion
Over the past few decades, the significance of medicinal values of various plants has been widely accepted.37 Medicinal plants produce a wide range of secondary metabolites that are known to have antioxidant effects which are essential to block the pathogenesis of several diseases and the process of aging caused by the free radicals.38,39 In the present study, phytochemical content and antioxidant activities of S. podophyllum L. leaves were carried out by using various tests. In this study phytochemical analysis of crude extract and its fractions exposed a wide range of phytoconstituents such as alkaloids, tannins, resins, flavonoids and steroids. A previous study reported that plant phenols, flavonoids and tannins are accountable for antioxidant activity.40 Phenolic compounds are the most important and abundantly found antioxidant agents in various medicinal plants. Phenolic compounds contribute to numerous essential activities in plants including cementing material linking phenolic polymers to cell wall polysaccharides, defense against herbivores and pathogens, regulation of cell growth and cell division.41 In this study CME of S. podophyllum L. leaves reported highest TPC with respect to remaining fractions. The alteration in the phenolic contents may be owing to the occurrence of essential oils exist in the plant. Phenolic compounds plentiful in medicinal plants contribute considerably to the antioxidant properties.42 The combined effect of phenolic compounds, flavonoids and other reducing agents in the plant extracts are well represented by TAA.43 The phosphomolybdenum method is mainly based on the reduction of molybdenum, Mo (VI) to Mo (V) by the action of antioxidant compounds and the formation of a green phosphate, Mo (V) complex with a highest absorption at 695 nm.44 Here CME of S. podophyllum L. leaves reported highest total antioxidant activity. In the study of antioxidant activity of Torilis leptophylla L. Saeed et al., reported promising outcome.45 In case of reducing power assay, reducing power of the test specimen is depicted by the changes of the color of the test solution from yellow to green.46 The existence of the reducing agents in the solution causes the reduction of the ferric form (Fe3+) to the ferrous form (Fe2+). Hence, the ferrous form can be observed by absorbance measurement at 700 nm.47 In this study CME of S. podophyllum L. leaves reported highest iron reducing capacity. The higher absorbance of CME extracts indicates its possible strong reducing power potential. The reducing power of the extracts is probably due to the biologically active compounds in the extract which contain potent electron donating abilities. An earlier study in the study of antioxidant profile of Uvaria littoralis Blume. also reported auspicious action.48 DPPH radical purple-colored solution bleaching is often used to measure the electron donation capacity of natural products.49 The method is mainly based on the principle of scavenging of DPPH through the introduction of radical species or antioxidant that decolorizes the DPPH solution. In this method, the concentration and potency of the antioxidants is proportional to the extent of color change. Moreover, substantial free radical scavenging activity of the compound under test is indicated by a great decrease in the absorbance of the reaction mixture.50 The results of this study recommended the highest DPPH radical scavenging activity in CME of S. podophyllum L. leaves. In the study on antioxidant activity of Streblus asper leaves, Ibrahim et al., reported almost comparable findings.51 The replacement of synthetic with natural antioxidants may be advantageous. This study divulge a prominent in vitro antioxidant actions of S. podophyllum L. leaves, which may be a forceful natural source of healing agents for the controlling of oxidative stress related disorders.
Conclusion
The denouements obtained in the present study, assessed notable antioxidant activities of the S. podophyllum L. leaves. The CME of S. podophyllum L. leaves possessed highest antioxidant activity compared to remaining fractions. In this in vitro study a high association is reported between the TPC and the antioxidant activities. However, this is an initial study and further study needs to be carried out for the isolation of promising compound(s) and elucidate mechanism for the respective effects.
Abbreviations
Podophyllum: Syngonium podophyllum; CME: Crude methanol extract; NHF: n-hexane fraction; CLF: Chloroform fraction; EAF: Ethyl acetate fraction; AQF: Aqueous fraction; TPC: Total phenolic content; TAA: Total antioxidant activity; RPA: Reducing power activity; DPPH: 2,2-Diphenyl-1-picrylhydrazyl; EDTA: Ethylenediaminetetraacetic acid; TCA: Trichloro aceticacid; TBA: Thiobarbituric acid; QR: Quercetin; CTC: (+)-catechin; ACA: Ascorbic acid; BHT: Butylatedhydroxy toluene; GA: Gallic acid; GAE: Gallic acid equivalents.
Authors’ Contributions
This work was carried out in collaboration between all authors. Authors MSU and MA designed the study, wrote the protocol, managed the analyses of the study. Authors MSH, MSU, MTK and MMB performed the laboratory tests and prepared the draft of the manuscript. Authors PK, OHC and RA participated in literature review and statistical analysis. Author MMAD reviewed the scientific contents of the manuscript. All the authors read and approved the final manuscript.
Acknowledgements
The authors are grateful to the Department of Pharmacy, Southeast University, Dhaka, Bangladesh for providing research facilities. The authors wish to thank the anonymous reviewer(s)/editor(s) of this article for their constructive reviews.
Funding
This work was self-funded.
Competing Interests
The authors proclaim that they have no competing interests.
References
- Henrich M., Barnes J., Gibbons S., Williamson E. M. Fundamentals of pharmacognosy phytotherapy. Edinburgh: Cyrchill Livingstone. 2004.
- Uddin M. S., Mamun A. A., Khanum S., Begum Y., Alam M. S. Analysis of in vitro antioxidant activity of Caryota urens L. leaves: A traditional natural remedy. J Coast Life Med. 2016;4:483-489.
CrossRef - Parekh J., Darshana J., Sumitra C. Efficacy of aqueous and methanol extracts of some medicinal plants for potential antibacterial activity. J Biol. 2005;29:203-210.
- Hassawi D., Kharma A. Antimicrobial activity of medicinal plants against Candida albicans. J Biol Sci. 2006;6:104-109.
- Bhat S., Lobo S. M., Kumar K. V. C., Sukesh and Chandrashekar K. R. Antimicrobial spectrum and phytochemical study of Hopea parviflora Beddome saw dust extracts. J Phyt. 2009;1(6):469-474.
- Sofowara A. Medicinal plants and antimicrobial activity. J Ethanophar. 1982;100:80-84.
- Uddin M. S., Mamun A. A., Hossain M. S., Akter F., Iqbal M. A., Asaduzzaman M. Exploring the effect of Phyllanthus emblica L. on cognitive performance, brain antioxidant markers and acetylcholinesterase activity in rats: Promising natural gift for the mitigation of Alzheimer’s disease. Ann Neurosci. 2016;23(4):218-229.
CrossRef - Uddin M. S., Al Mamun A., Hossain M. S., Ashaduzzaman M., Noor M. A., Hossain M. S., Uddin M. J, Sarker J, Asaduzzaman M. Neuroprotective effect of Phyllanthus acidus L. on learning and memory impairment in a scopolamine-induced animal model of dementia and oxidative stress: Natural wonder for regulating the development and progression of Alzheimer’s disease. Adv Alzheimer Dis, 2016; 5(2): 53-72.
CrossRef - Uddin MS, Mamun AA, Iqbal MA, Islam A, Hossain MF, Khanum S, Rashid M. Analyzing nootropic effect of Phyllanthus reticulatus Poir. on cognitive functions, brain antioxidant enzymes and acetylcholinesterase activity against aluminium-induced Alzheimer’s model in rats: Applicable for controlling the risk factors of Alzheimer’s disease. Adv Alzheimer Dis, 2016; 5(3): 87-102.
CrossRef - Uddin MS, Asaduzzaman M, Mamun AA, Iqbal MA, Wahid F. Neuroprotective activity of Asparagus racemosus Linn. against ethanol-induced cognitive impairment and oxidative stress in rats brain: Auspicious for controlling the risk of Alzheimer’s disease. J Alzheimers Dis Parkinsonism, 2016; 6(4): 1-2.
CrossRef - Uddin MS, Haque A, Mamun AA, Iqbal MA, Kabir MT, Rony RK, Begum M. Searching the linkage between high fat diet and Alzheimer’s disease: A debatable proof stand for ketogenic diet to alleviate symptoms of Alzheimer’s patient with APOE ε4 allele. J Neurol Neurophysiol, 2016; 7(5): 1-2.
CrossRef - Uddin MS, Nasrullah M, Hossain MS, Rahman MM, Sarwar MS, Amran MS, et al. Evaluation of nootropic activity of Persicaria flaccida on cognitive performance, brain antioxidant markers and acetylcholinesterase activity in rats: Implication for the management of Alzheimer’s disease. American J Psychi Neuroscience, 2016; 4(2):26-37.
CrossRef - Harmanto N. Mahkota Dewa Obat Pusaka Para Dewa. Jakarta, Indonesia: Cetakan keempat, Agromedia Pustaka; 2002.
- Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun J. Cancer Statistics, 2007. CA Cancer J Clin, 2007; 57(1):43-66.
CrossRef - Cai Y, Luo Q, Sun M, Corke H. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sci, 2004; 74:2157-2184.
CrossRef - Halliwell B. Biochemistry of oxidative stress. Biochem Soc Trans, 2007; 35(5):1147-1150
CrossRef - Devasagayam T, Tilak J, Boloor K, Sane KS, Ghaskadbi SS, Lele R. Free radicals and antioxidants in human health: current status and future prospects. J Assoc Physicians India, 2004; 52:794-804.
- Hasan MF, Iqbal MA, Uddin MS. Antibacterial and antifungal activity of Litsea monopetala leaves on selected pathogenic strains. Eur J Med Plants, 2016; 12(4):1-8.
CrossRef - Williams RJ, Spenser JPE, Rice-Evans C. Flavonoids: antioxidants or signalling molecules? Free Radic Biol Med, 2004; 36:838-849.
CrossRef - Mulubagal V, Tsay H. Plant cell cultures–an alternative and efficient source for the production of biologically important secondary metabolites. Int J Appl Sci Eng Tech, 2004; 2:29-48.
- Borneo R, Leon EA, Aguirre A, et al. Antioxidant capacity of medicinal plants from the Province of Cordoba (Argentina) and their in vitro testing in model food system. Food Chem, 2008; 112:664-670.
CrossRef - Kumar S, Sandhir R, Ojha S. Evaluation of antioxidant activity and total phenol in different varieties of Lantana camara leaves. BMC Res Not, 2014; 7:560.
CrossRef - Couladis M, Tzakou O, Verykokidou E, Harvala C. Screening of some Greek aromatic plants for antioxidant activity. Phytother Res, 2003; 17(2):194-195.
CrossRef - Akinmoladun AC, Obuotor EM, Farombi EO. Evaluation of antioxidant and free radical scavenging capacities of some Nigerian indigenous medicinal plants. J Med Food, 2010; 13:444-451.
CrossRef - Özen T, Çöllü Z, Korkmaz H. Antioxidant Properties of Urtica pilulifera Root, Seed, Flower, and Leaf Extract. J Med Food, 2010; 13:1224-1231.
CrossRef - Hossain MS, Uddin MS, Moniruzzaman M, et al. Comparative study of cytotoxic potential and phytochemical screening of Xanthosoma sagittifolium rhizome and Syngonium podophylam leaf. J Med Plants, 2015; 3(3): 43-46.
- Hernandez J. In Hawaiian rainforests: Exotic aroid ecologies. Aroideana 2007; 30: 91-97.
- Tropical.theferns.info. Useful tropical plants. (Cited 2017 June 25): Available from: http://tropical.theferns.info/viewtropical.php?id=Syngonium+podophyllum.
- Zeraati F, Araghchian M, Esna-ashari F. Fazlian MM, Torabian S, Fallah N, Ghavimi M. Antinociceptive properties of ascorbic acid: evidence for the mechanism of action. Avicenna J Med Biochem, 2014; 2(1): e18572.
CrossRef - Hossain MS, Uddin MS, Kabir MT, Akhter S, Goswami S, Mamun AA, et al. In vivo screening for analgesic and anti-inflammatory activities of Syngonium podophyllum L.: A remarkable herbal medicine. Annu Res Rev Biol, 2017; 16(3): 1-12.
CrossRef - Camporese A, Balick MJ, Arvigo R, Esposito RG, Morsellinom N, De Simone F, Tubaro A. Screening of anti-bacterial activity of medicinal plants from Belize (Central America). J Ethnopharm, 2003; 87(1): 103-7.
CrossRef - Trease GE, Evans MC. Text book of Pharmacognosy. 12th edition. Balliere, Tindall, London; 1983, p. 343-383.
- Singleton VL, Rossi JA. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic, 1965; 16: 144-158.
- Prieto P, Pineda M, Aguilar M. Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex, specific application to the determination of vitamin E. Anal Biochem, 1999; 269: 337-41.
CrossRef - Oyaizu M. Studies on products of browning reaction antioxidative activities of products of browning reaction prepared from glucosamine. Japanese J Nutr, 1986; 44 (6): 307-315.
CrossRef - Choi HY, Jhun EJ, Lim BO. Application of flow injection-chemilumineacence to the study of radical scavenging activity in plants. Phytother Res, 2000; 14: 250-3.
CrossRef - Rahman MM, Uddin MS, Nejum MR, Din SMAA, Uddin GMS. Study on antibacterial activity of Cajanus cajan L. against coliforms isolated from industrial waste water in Bangladesh. Plant, 2017; 5(5-1): 13-18.
- Mamun AA, Uddin MS, Wahid F, Iqbal MA, Rahman MM. Neurodefensive effect of Olea europaea L. in alloxan-induced cognitive dysfunction and brain tissue oxidative stress in mice: Incredible natural nootropic. J Neuro Neurosci, 2016; 7(S3): 1-9.
- Mamun AA, Hossain M, Islam A, Zaman S, Uddin MS. Asparagus racemosus Linn. potentiate the hypolipidemic and hepatoprotective activity of fenofibrate in alloxan-induced diabetic rats. Plant, 2017; 5(5-1): 1-12.
- Uddin MS, Uddin GMS, Begum MM, Begum Y, Herrera-Calderon O, Islam MI, Abdel-Daim MM. Inspection of phytochemical content and in vitro antioxidant profile of Gnaphalium luteoalbum L.: An unexplored phytomedicine. J Pharm Nutr Sci, 2017; 7(3): 136-146.
CrossRef - Wallace G, Fry SC. Phenolic compounds of the plant cell. Int Rev Cytol 1994;151:229-267.
CrossRef - Binns AN, Chen RH, Wood HN, Lynn DG. Cell division promotion activity of naturally occurring dehydrodiconiferyl glucosides: Do cell wall components control cell division? Proc Natl Acad Sci USA, 1987; 84:980-984.
CrossRef - Sharififar F, Dehghn-Nudeh G, Mirtajaldini M. Major flavonoids with antioxidant activity from Teucrium polium L. Food Chem, 2009; 112:885-888.
CrossRef - Khan RA, Khan MR, Sahreen S. Assessment of flavonoids contents and in vitro antioxidant activity of Launaea procumbens. Chem Central J, 2012; 6:43.
CrossRef - Saeed N, Khan MR, Shabbir M. Antioxidant activity, total phenolic and total flavonoid contents of whole plant extracts Torilis leptophylla L. BMC Complement Altern Med, 2012; 12:221.
CrossRef - Ferreira ICFR, Baptista P, Vilas-Boas M, Barros L. Free-radical scavenging capacity and reducing power of wild edible mushrooms from northeast Portugal: Individual cap and stipe activity. Food Chemy, 2007; 100:1511-1516.
CrossRef - Gordon MH. Food antioxidants. 1st ed. London: Elsevier Applied Science; 1990.
- Rahman A, Haque A, Uddin MS, Mian MM, Sufian MA, Rahman MM, et al. In vitro screening for antioxidant and anticholinesterase effects of Uvaria littoralis Blume.: A nootropic phytotherapeutic remedy. J Intellect Disabl – Diagn Treat, 2017; 5(2): 50-60.
- Priya SH, Prakasan N, Purushothaman J. Antioxidant activity, phenolic-flavonoid content and high-performance liquid chromatography profiling of three different variants of Syzygium cumini seeds: A comparative study. J Interc Ethnopharma, 2017;6(1):107-114.
CrossRef - Krishnaiah D, Sarbatly R, Nithyanandam RR. A review of the antioxidant potential of medicinal plant species. Food Bioprod Process, 2011; 89:217-233.
CrossRef - Ibrahim NM, Mat I, Lim V, Ahmad R. Antioxidant activity and phenolic content of Streblus asper leaves from various drying methods. Antioxidants, 2013, 2, 156-166.
CrossRef







