Manuscript accepted on :04-11-2025
Published online on: 18-02-2026
Plagiarism Check: Yes
Reviewed by: Dr. Rakam Gopi Krishna
Second Review by: Dr. Priya Bhardwaj
Final Approval by: Dr. Anton R Keslav
Ravinesh Mishra , Manju Jakhar*
, Manju Jakhar* , Rajeev Dhiman
, Rajeev Dhiman , Laraib Khan
, Laraib Khan , Aditya Raj
, Aditya Raj , Vandana Devi
, Vandana Devi , Anjali Kumari
, Anjali Kumari  and Ajay Kumar
and Ajay Kumar
School of Pharmacy and Emerging Sciences, Baddi University of Emerging Sciences and Technology, Baddi, Dist. Solan, Himachal Pradesh, India.
Corresponding Author: Manjujhakhar0002@gmail.com
Abstract
Type 2 diabetes mellitus (T2DM), obesity, dyslipidaemia, and metabolic syndrome (MS) are metabolically driven conditions that are intimately related to one another and are rapidly becoming important global public health concerns. Insulin resistance, systemic low-grade inflammation, and long-term disruptions in glucose and lipid metabolism are characteristics of these disorders, which significantly raise the risk of hepatic and cardiovascular diseases as well as premature death. The pharmacotherapies that are now on the market are bad, often need polypharmacy, and have adverse effects that make it difficult for patients to take them as prescribed. Consequently, new drugs that can address the intricate pathophysiology of metabolic disorders are crucial. Due to the involvement of key metabolic sensors, such as AMP-activated protein kinase (AMPK), sirtuins (specifically SIRT1), and peroxisome proliferator-activated receptors (PPARs), a novel medication with multifunctional properties has been developed. These pathways further augment insulin sensitivity, repress hepatic gluconeogenesis, ameliorate mitochondrial function, normalise lipid profiles, and exert strong anti-inflammatory and antioxidant effects. Preclinical research indicates positive effects such as glycaemic control, weight loss, improvements of lipid metabolism or reduction of hepatic steatosis and fibrosis, as well as gut microbiota modulation. Preliminary human studies demonstrate enhanced metabolic and inflammatory biomarkers and excellent safety and compliance. The agent is a positive breakthrough in personalised, multi-targeted treatment of metabolic disease and needs to be validated by further large-scale clinical trials. This review explores recent advancements in drug discovery for metabolic disorders, focusing on novel therapeutic targets in insulin signalling, lipid metabolism, inflammation, and disease-specific complications. It highlights new therapies, challenges to drug development and future research directions. In addition to informing the reader about the possibility of breaking new ground in pharmacological management, this review is intended as a detailed, comprehensive picture of the most recent advances in the treatment of metabolic disorders
Keywords
Dyslipidemia; Gene Therapy; Gut Microbiome; Inflammation; Insulin Resistance; Metabolic Disorders; MP-Activated Protein Kinase (AMPK); Non-alcoholic Fatty Liver Disease (NAFLD); Peroxisome Proliferator-Activated Receptors (PPARs)
| Copy the following to cite this article: Mishra R, Jakhar M, Dhiman R, Khan L, Raj A, Devi V, Kumari A, Kumar A. The Therapeutic Potential of Novel Drug Targets in Metabolic Disorders- A Review. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Mishra R, Jakhar M, Dhiman R, Khan L, Raj A, Devi V, Kumari A, Kumar A. The Therapeutic Potential of Novel Drug Targets in Metabolic Disorders- A Review. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/46eI8JJ |
Introduction
The metabolic disorders are varied and have devastating effects on the ability to manage and regulate basic biochemical functions (e.g lipid and carbohydrate metabolism). These diseases (i.e. diabetes, obesity and dyslipidaemia) are a high threat to global health due to their high rates of morbidity and mortality.1,2
Metabolic disorders, including obesity, metabolic syndrome, non-alcoholic fatty liver disease (NAFLD), and type 2 diabetes mellitus (T2DM), are a major and rapidly expanding global health concern. The disruption of regular metabolic functions, including insulin resistance, chronic low-grade inflammation, and impaired glucose and lipid metabolism, is characteristic of these conditions. Although many pharmacological treatments have been developed, many patients still do not achieve optimal outcomes, suggesting a need for more effective treatment strategies. Therefore, the targeting of new molecular pathways that have a role in the intricate, multifactorial metabolic diseases has become hopeful in this context. The last decades have revealed novel drug targets owing to the development of genomics, proteomics and metabolomics. These are the adipokines, gut microbiome metabolites, regulators of mitochondrial function, as well as enzymes involved in glucose and lipid metabolism. FGF21, AMPK, and PPARs are also thought to be therapeutic targets because of their involvement in energy homeostasis and metabolic regulation. These targets have also evolved with favourable pressure of preclinical and early clinical data in the enhancement of insulin sensitivity, lowering of hepatic steatosis and facilitation of weight loss.
Metabolic Disorders and Their Worldwide Burden in on Health
Energy homeostasis imbalance is a characteristic of metabolic disorders, a leading cause of chronic illnesses worldwide. Due to the rising prevalence of NAFLD, type 2 diabetes, and obesity, innovative treatment methods are needed. These diseases reduce quality of life and are too expensive for healthcare systems.1-3
Lifestyle, environmental, and genetic variables increase the risk of metabolic disorders. Insufficient energy causes these illnesses to spread. Chronic metabolic illnesses are caused by energy imbalances, which are common worldwide. As obesity, type 2 diabetes (T2DM), and NAFLD become increasingly frequent, we need novel therapies immediately.3,4
Rising Prevalence of Diseases Like Diabetes, Obesity, and Dyslipidemia
Epidemiological studies indicate a very sizeable increase in metabolic disorders within the last several decades. According to the WHO, diabetes now affects 400 million people worldwide, and obesity has increased. Dyslipidaemia is one of the risk factors of cardiovascular disease and is associated with urbanisation. 08Over 650 million people around the globe are suffering from obesity (Table 1).5,6 The situation is even worse as obesity during childhood increases the incidence of metabolic diseases. Type 2 Diabetes: The Disease of Younger People. Many are now being diagnosed with Type 2 diabetes at a much younger age, hence the increased importance of early intervention.5-8
Table 1: Classification and Prevalence of Major Metabolic Disorders 18
| Disorder | Global Prevalence | Key Characteristics | Major Risk Factors |
| Type 2 Diabetes Mellitus (T2DM) | 400+ million | Hyperglycemia, Insulin Resistance | Obesity, Sedentary lifestyle |
| Obesity | 650+ million | BMI ≥30, Visceral Fat | High-calorie diet, Genetics |
| Dyslipidemia | Rising globally | ↑ LDL, ↓ HDL, ↑ TG | Poor diet, Smoking |
| NAFLD | 25–30% worldwide | Hepatic steatosis | Insulin resistance, Obesity |
Limitations of Current Treatments and the Need for Novel Drug Targets
Pharmacological interventions such as statins, insulin therapy, and weight-loss drugs have demonstrated efficacy, but they often have adverse effects and limited durability. Novel treatment targets that can change pathophysiological pathways will be needed to address this range of metabolic diseases.9,10
Metabolic disease management requires lifestyle changes, but adherence is challenging. A major drawback of diabetic medications such as metformin, sulfonylureas, and DPP-4 inhibitors and cholesterol medications such as statins, is drug resistance, side effects, and failure to halt disease progression. This emphasises the necessity for new therapeutic targets that modify the course of illness at the cellular level.11,12
Pathophysiology of Metabolic Disorders
Energy homeostasis, glucose regulation, and lipid metabolism are all out of balance in metabolic disorders, a category of diseases that interfere with regular metabolic functions. T2DM, obesity, metabolic syndrome, and dyslipidaemia are some of such conditions. Hormonal imbalances, environmental factors, genetic susceptibility, and chronic inflammation are all part of the complex pathophysiology of metabolic diseases. Understanding the physiology and the pathogenesis of these diseases is a crucial step in developing efficacious therapies.13-15 The pathogenesis involves a complex interplay between insulin resistance, inflammation, and mitochondrial dysfunction (Figure 1).16,17
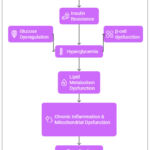 |
Figure 1: Pathophysiology of Metabolic Disorders |
Central Metabolism Pathways Implicated in Disease Development
Disturbance of fundamental metabolic pathways, such as fat and carbohydrate metabolism pathways, causes metabolic diseases.10 Many important pathways are involved in disease progression:
Dysregulated glucose metabolism
Pancreatic beta cells release insulin, which controls glucose metabolism.17,18 In healthy people, insulin increases muscle and adipose glucose absorption, decreases hepatic glucose production, and synthesises glycogen.11 Insulin resistance in metabolic diseases like type 2 diabetes impairs glucose absorption and increases hepatic glucose synthesis, causing hyperglycaemia. Glucotoxicity, beta-cell malfunction, and insulin resistance are linked to metabolic disorders.19 Insulin resistance causes high blood sugar because cells can’t use the hormone. Pancreatic beta-cells produce more insulin to compensate, but they overload and fail. The beta-cells’ progressive inability to fulfil the body’s expanding insulin requirements causes decreased insulin output and hyperglycaemia. Chronic hyperglycaemia, also known as glucotoxicity, worsens metabolism by reducing insulin production and its effects, creating a vicious cycle.21,22
Dysfunction of lipid metabolism
Disruptions in lipid metabolism, which is necessary for energy balance, contribute to metabolic disorders. Lipid disorders, including high FFA, low HDL, and high TG, amplify infrared radiation.23
Cardiovascular disease is strongly linked to dyslipidaemia. This disease is characterised by increased triglycerides, reduced HDL, and higher LDL. Ectopic lipid buildup in non-adipose organs, including the liver (hepatic steatosis) and skeletal muscle, causes inflammation and metabolic dysfunction.24
Ectopic lipid accumulation occurs in non-adipose tissues
Adiponectin, a hormone that protects against insulin sensitivity and cholesterol control, is generated less and pro-inflammatory cytokines are released more when adipose tissue malfunctions, especially when visceral fat develops. This is because adiponectin regulates lipids and insulin sensitivity.25
Energy Homeostasis and Mitochondrial Dysfunction
Homeostatic regulation regulates energy intake and expenditure. Metabolic illness disrupts this homeostasis for several reasons. Dysregulated mitochondrial oxidative phosphorylation (OXPHOS) reduces ATP production and increases oxidative stress.26 Deregulation of hunger and metabolism hormones, notably leptin and ghrelin, causes obesity and metabolic syndrome.27 Controlled insulin resistance and dyslipidaemia better than diabetic rats, although chronic low-grade inflammation disrupted metabolism due to elevated levels of pro-inflammatory cytokines (IL-6, TNF-α).28
Role of insulin resistance, glucose dysfunction and impaired lipid metabolism
Insulin Resistance: Root of All Metabolic Evil
Development of Insulin Resistance: Mechanisms
Insulin resistance is the central basis of metabolic dysfunction, the driver of numerous interconnected processes. The principal mechanism has been chronic inflammation, where pro-inflammatory cytokines (TNF-α, IL-6) impair the insulin receptor substrate (IRS) signalling pathway by directly antagonising the link between the insulin receptor and downstream signalling pathways. ER stress due to overnutrition further exacerbates this to compromise both beta-cell function and endothelium.17-19 Furthermore, under chronic conditions of high circulating levels of free fatty acids, lipotoxic intermediates, especially ceramides and diacylglycerols, accumulate, directly disrupting insulin signalling cascades and further worsening glucose dysregulation.20,21
Glucose-Lipid Metabolism Interconnections
By allowing the cycle of insulin resistance and hyperglycemia to continue, we thus worsen metabolic derangement down the cascade. So continued excess hyperglycemia promotes the generation of advanced glycation end products (AGEs), which leads to vascular damage or diabetic complications such as retinopathy and nephropathy. At the same time, increased glucagon secretion results in increased gluconeogenesis, which sustains fasting hyperglycemia, but also serves to produce reactive oxygen species (ROS), which further sustains oxidative stress and promotes cellular injury.22,23
These effects are exacerbated by lipid metabolism defects, which provoke enhanced lipolysis and raise blood levels of free fatty acids that further aggravate insulin resistance. This induces a vicious cycle of dyslipidaemia (high triglycerides, low HDL and high LDL), inducing both insulin resistance and atherogenesis, markedly increasing cardiovascular disease risk.24,30
Major Clinical Complications
This interconnected metabolic derangement presents with three major complication patterns:
Diabetic neuropathy: peripheral nerve injury due to oxidative stress, microvascular dysfunction, and inflammation-mediated nerve injury from chronic hyperglycemia leads to progressive neuropathic pain and sensory loss.31
NAFLD: Insulin resistance stimulates hepatic lipogenesis but inhibits lipolysis, resulting in hepatic steatosis. This is compounded by mitochondrial dysfunction, which leads to further decreased ATP production and amplified hepatic oxidative stress and inflammation. More advanced forms of NAFLD have the potential to progress to non-alcoholic steatohepatitis (NASH), the stages of liver fibrosis, and finally the sequela of cirrhosis.32
Cardiovascular Disease
Multiple pathways drive an increased risk of CVD with metabolic states: hypertension from insulin resistance, from sodium retention and endothelial dysfunction, accelerated atherosclerosis from dyslipidaemia and chronic inflammation, and cardiac metabolism dysregulation causing heart failure.31,32
The interlinking pathways inflammation, insulin resistance, glucose dysregulation, lipid abnormalities, and mitochondrial dysfunction are pivotal contributors to the pathogenesis of obesity, type 2 diabetes, NAFLD and cardiovascular disease, underscoring the necessity of multi-targeted therapeutic strategies.
Novel Drug Targets in Metabolic Disorders
AMPK Activators: The Ultimate Metabolic Modulators
Because AMP-activated protein kinase (AMPK) functions as a cellular energy sensor, it is a promising target for the treatment of metabolic diseases. Through multiple downstream pathways, the activation of AMPK promotes glucose uptake, stimulates lipid oxidation, and enhances insulin sensitivity.25,26
Metformin was found to be the most widely used antidiabetic drug, which primarily acts to activate AMPK. Apart from Metformin, direct activators of AMPK, such as AICAR and other synthetic compounds, have been reported to improve conditions in preclinical studies. These agents promote metabolism by enhancing glucose uptake in peripheral tissues, suppressing hepatic gluconeogenesis, and enhancing fatty acid oxidation.27
Significance: By targeting both glucose and lipid metabolism, AMPK seems ideally positioned for both therapeutic and clinical applications in patients with metabolic diseases that influence both pathways.
 |
Figure 2: Mechanism of Action of AMPK Activators |
 |
Figure 3: Key Molecular Targets and Their Therapeutic Roles.28 |
The SGLT2 Blockers: Reducing glucose and protecting the heart
Sodium-glucose cotransporter-2 (SGLT2) inhibitors represent a novel mechanism to lower blood glucose by increasing urinary glucose excretion independently of insulin action. Medicines such as empagliflozin and canagliflozin have benefits beyond blood sugar lowering, protecting the heart and kidneys.
They reduce the risk of cardiovascular death, slow the progression of chronic renal disease, and lead to some weight loss. This mechanism of action—blocking glucose reabsorption in the kidney opens up new potential applications of these agents in the treatment of heart failure in patients without diabetes.27,28
Promising clinical data: Heart- and kidney-protective actions of SGLT2 inhibitors: implications for joint metabolic disorders control.29
GLP-1 Receptor Agonists: Treating weight and glucose together
Liraglutide and semaglutide are two examples of multi-pathway targeting agents, glucagon-like peptide-1 (GLP-1) receptor agonists acting on multiple metabolic pathways simultaneously. They stimulate glucose-dependent insulin secretion, delay gastric emptying, increase satiety and decrease hepatic glucose production.
In clinical practice, it has recently been shown that these agents can induce considerable weight loss (10–15% of body weight) and control of blood glucose, in obese individuals with type 2 diabetes. 29,30 The latest formulations provide better efficacy and are easier to take.30
Clinical promise: Dual actions on glucose and body weight treat two key components of metabolic syndrome with a single intervention.
PPAR Modulators: Full Control of Lipids and Glucose
PPARs, or peroxisome proliferator-activated receptors, control genes that affect inflammation, lipid metabolism, and glucose homeostasis. PPAR-α agonists (fibrates) help dyslipidemia by breaking down fatty acids and lowering triglycerides. PPAR-γ agonists (thiazolidinediones) make insulin more sensitive.31-33
Dual PPAR-α/γ agonists that target both glucose and lipid metabolism at the same time are the subject of current research. These may have better therapeutic effects than single-target methods. 33 These drugs seem to work especially well for those with metabolic syndrome who need help with both glucose and cholesterol levels.33
New ways of treating diseases
A few more prospects seem promising, but they need further work. Using beta-3 adrenergic receptor agonists and thermogenic targets to activate brown adipose tissue is a new way to help people with obesity burn more calories.34 Hepatic lipogenesis inhibitors, such as ACC and DGAT inhibitors, work against the causes of NAFLD by lowering the production of triglycerides.35 Cytokine modulators (IL-1β inhibitors, TNF-α blockers) are examples of anti-inflammatory treatments that target chronic low-grade inflammation, which is a prevalent cause of metabolic illnesses. These treatments may help reduce metabolic dysfunction caused by inflammation.36-39 Gut microbiota-based treatments, such as probiotics and faecal microbiota transplantation, are another new area of research since the microbiome affects metabolism and inflammation.39 For some problems, targeted treatments include nerve growth factor (NGF) modulators and oxidative stress inhibitors for diabetic neuropathy37, FXR agonists and mitochondrial function enhancers for NAFLD/NASH and PCSK9 inhibitors with SIRT1 activators for heart problems.38,39
Advances in Drug Discovery and Development
AI-Driven Drug Discovery in Metabolic Disorders
Methods such as machine learning, deep learning, and bioinformatics are disrupting all steps in drug development for metabolic diseases, from target identification to drug discovery to even clinical practice.40,41 For example, AI technologies like AlphaFold and Atomwise are enabling us to identify targets and design drugs in unprecedented ways. They are specifically valuable for predicting protein structures, for virtual screening and drug repurposing (Table 4).42,43
Table 2: AI Tools in Metabolic Disorder Drug Discovery
| AI Tool | Application | Advantages |
| AlphaFold | Protein structure prediction | Speeds up target validation |
| DeepChem | Virtual screening | Accurate ligand docking |
| Atomwise | Drug repurposing | AI-driven predictions |
| GANs & RNNs | De novo drug design | Explores novel chemical space |
They say machine learning algorithms analyse large databases of proteins, metabolites and genes for therapeutic targets, drug mechanisms of action, toxicity prediction and pharmacokinetics. Deep learning approaches, such as convolutional and recurrent neural networks, have been shown to effectively process clinical data and molecular structures to discover complex biological relationships. AI-based virtual screening systems can screen millions of compounds in a few hours to identify potential drug-like compounds for metabolic disease targets. Using generative adversarial networks (GANs) and reinforcement learning, we can explore unexplored regions of chemical space to discover novel drugs.43-46
Drug repurposing is greatly aided by AI techniques that explore biological datasets, as well as drug interaction networks, to discover new therapeutic uses for known drugs. 47 This approach reduces drug development time and costs dramatically, as evidenced by the use of Metformin for more than just diabetes.47,48
Biomarkers and Personalized Medicine
The emergence of novel omics technologies has enabled the identification of clinically relevant biomarkers for early diagnosis, longitudinal tracking, and therapy-related management of metabolic diseases.48,49 Genetic predispositions to metabolic disorders has been established through genome-wide association studies (GWAS). Examples include PNPLA3 mutations that predispose the individual to NAFLD, and TCF7L2 polymorphisms that predispose the individual to diabetes.50-52
After mass spectrometry and NMR spectroscopy, circulating biomarkers such as adiponectin, leptin, fetuin-A and branched-chain amino acids (BCAAs) related to energy balance and insulin sensitivity were found to be associated with metabolic disorders. Specifically, non-invasive diagnostic approaches, and in particular liquid biopsy-based ones, which focus on cell-free DNA and microRNAs derived from exosomes in the circulation, permit continuous monitoring and an immediate response when required.52,53
Strategies for Repurposing Drugs
pharmacological repurposing is a rapid and cost-effective therapeutic exposure created by extensively screening existing pharmacological libraries. The expansion of the clinical use of SGLT2 inhibitors beyond diabetes, the anti-inflammatory effects of statins that also extend beyond lipid control, and the possible use of thiazolidinediones in NAFLD92 are some of the relevant milestones.54
Abstract: Definitions: Network pharmacology and systems biology try to predict if a drug will work on several targets using protein-protein interaction networks and for drug repurposing using metabolic pathway research. 55 On the other hand, issues in regulations remain major trial pits that require clearing. Each new indication requires complete clinical trials to establish efficacy, safety, and optimum dosing.56
Future Outlook
A better plan for treatment with fewer side effects using a personalised medicine approach integrating genomic, proteomic, and metabolomic data.55 Understanding the mechanisms of metabolic adaptation and compensatory responses is critical for creating combination medicines that are more effective and overcome drug resistance over time. Most new approaches, such as CRISPR-based gene editing, RNA treatments and nanotechnology, could treat metabolic issues at their root, possibly leading to cures.57
Translational Challenges and Future Perspectives
Overcoming Drug Resistance and Metabolic Adaptation
Metabolic diseases are challenging to treat, as adaptive metabolic pathways can lead to reduced effectiveness of drugs over time. As a result of this flexibility in metabolism, it can use different energy systems and new therapies need to arise to treat these people.58,59
Key mechanisms of resistance include receptor desensitisation from prolonged drug exposure, epigenetic modulation of gene expression and counter-regulatory hormonal responses that oppose medical therapy.60,61
To counter resistance, combination medicines that hit multiple pathways simultaneously, tailored dosing schedules according to genotype, and epigenetic modulators to enhance therapeutic efficacy are all valid strategies.62
Realising Personalised Medicine
A wealth of pharmacogenomic research identified key genetic variations that modulate the action of several important drugs, including polymorphisms in TCF7L2 affecting the response to sulfonylureas, variations in PPARG affecting the action of PPAR-γ agonists and polymorphisms in CYP2C9/CYP2C19 affecting the metabolism of statins.63-66
This has led to the gut microbiome being recognized as an indispensable player influencing metabolic health and also drug responses. Microbes living in our gut might change the metabolism of glucose and the sensitivity to insulin. Targeted delivery of therapies to the microbiota is a novel approach to disease treatment.67
New Therapeutic Innovations
Nanoparticle-based targeted delivery, implanted controlled-release devices for chronic illnesses, and improved formulations with better bioavailability-based drug delivery systems are making treatment more effective. 68,69 There are regenerative medicine techniques that have a glimmer of hope. Adipose stem cell therapy might alter metabolism, CRISPR-Cas9 gene-editing might correct metabolic disease-causing mutations, and pancreatic beta-cell transplant might provide a means for diabetes treatment. AI is revolutionising metabolic disorder management by accelerating the identification of drug targets, devising patient-specific treatment plans, developing predictive models of disease progression with machine learning approaches from patient-level data.70,71
Legal and Moral Issues
Drug development faces numerous regulatory challenges, from the protracted durations of clinical trial phases, exorbitant costs of development, to inconsistencies in regulatory regimes between countries, which can further hinder access to drugs in the world.72
Personalised medicine demands ethical dilemmas like protecting genetic data confidentiality, ensuring equal access to advanced medications, and obtaining adequate informed consent for genetic testing and treatment recommendations.73-75
Discussion
Clinical Implications of Novel Targets
The identification of novel pharmacological targets represents a shift towards mechanism-based therapy for metabolic diseases rather than symptomatic treatment. AMPK activators, SGLT2 inhibitors, GLP-1 receptor agonists, and PPAR modulators offer immediate metabolic improvement that is superior to conventional approaches and acts simultaneously on multiple pathophysiological pathways. These drugs improve glycaemic control and lipid levels and provide cardiovascular and renal protection, significantly reducing long-term complications (e.g., neuropathy, nephropathy, and cardiovascular events).76-78
It goes beyond single-patient outcomes and has greater significance for the healthcare system as a whole. The improved specificity of these medicines leads to greater adherence by patients as a result of fewer side effects and simplified dosing schedules. Importantly, they offer potential disease-modifying effects, rather than simply treating symptoms, and they can modify the underlying course of metabolic disease and reduce the advances of polypharmacy approaches (characteristic of current therapeutic strategies).78
Challenges in Translation to Practice
While there have been pioneering preclinical and early clinical data suggesting some of these targets may have benefit, the potential translatability of new targets into the clinic is limited by several barriers. Metabolic adaptation and drug resistance are important problems because the compensatory mechanisms of the body often diminish the long-term effect of therapy. 79,80 Metabolic diseases are multifactorial and intricate, and therefore, they require combinatorial strategies; however, the challenge continues to be the selection of medication combinations and their sequence.81
Regulatory barriers considerably prolong and raise the costs of developing drugs, requiring extensive safety and effectiveness data from a diversity of patients. 82 Metabolic diseases are also highly diverse, with their therapeutic response necessitating individualised treatment approaches, but the current healthcare system lacks the infrastructure to enable identifying target metabolic disorders with routine pharmacogenomic testing and appropriate dosage algorithms. The increased cost of new drugs raises concerns about access to health care and how treatment is distributed.82
Future Research Priorities
Key research objectives include predictive biomarkers for therapy response to create truly patient-specific medical approaches.83,84 Understanding the metabolic plasticity and fitness compensatory mechanisms will inform the design of combination medicines that maintain efficacy in the long term.85 This provides a new direction in research that combines traditional medication with gut microbiota modification.86
Further development of advanced delivery technologies, including those based on nano-enabled targeted or sustained-release formulations, is required to improve the pharmacological performance of therapies delivered to the ocular surface.68 First, the clinical efficacy of regenerative medicine strategies, particularly stem cell therapy and gene editing technology, should be validated. 70,71 In addition, applications of artificial intelligence for drug discovery and for use in patient care must progress from current proof-of-concept studies to established clinical use.87
Constraints and Considerations
Data supporting many of the new targets exists only in preclinical research or small clinical trials, and larger, longer studies will be needed to clarify the safety and efficacy profiles of these drugs.88 Therapeutic trials have focused primarily on Western populations, limiting transferability to ethnic and geographical groups with differing genetic and environmental determinants. Economic barriers are significant, but research costs for novel medicinal agents can be high, and these new medicines may be unaffordable within low-resource settings where metabolic diseases are accelerating.89 Such complexity of personalised medicine requires substantial changes to the healthcare delivery system and clinician education, as well as re-engineering of clinical decision-support technologies. As personalised medicine evolves, the ethical challenges of genetic testing, data privacy, and equal access to cutting-edge drugs should be carefully examined. Continuous monitoring is required to assess the long-term effects of novel therapeutic approaches, most of which alter the microbiome or introduce genetic alterations, as these are poorly characterised and understood.90
Conclusion
This review highlights several new drug targets that target disruptive pathophysiological mechanisms of metabolic disorders rather than treating symptoms, thus advancing mechanism-based therapeutics beyond symptomatic management. This is particularly because the identification and validation of these putative therapeutic targets has been hastened through an increasing usage of artificial intelligence in drug discovery and precision medicine approaches. This newly identified opportunity opens up exciting and unprecedented avenues for overcoming a global epidemic of diabetes, obesity, dyslipidaemia and metabolic syndrome that afflicts more than 1 billion people worldwide.
Among the therapeutic targets assessed, AMPK activators, SGLT2 inhibitors, GLP-1 receptor agonists, and PPAR modulators show the highest translational potential linked to multiple beneficial effects across diabetes pathophysiological pathways and overall safety. These agents simultaneously target insulin resistance, derangement in glucose homeostasis, disturbance in lipid metabolism, as well as chronic inflammation, and are the main pathophysiological contributors of metabolic disease. By targeting multiple mechanisms through single therapeutic approaches, they present a sizable improvement over the current polypharmacy strategy.
The possibilities for positive clinical impact reach far beyond just better metabolic control, with possibilities for significant decreases in cardiovascular and renal complications, improved patient adherence via easier treatment regimens, as well as disease modification. These therapies provide the hope of changing the course of disease in addition to better managing clinical symptoms, which may ultimately lead to lower long-term healthcare utilisation and costs in addition to improved quality of life for millions of patients worldwide. Because of the personalised medicine trend, this method has the potential to ensure better treatment results while decreasing undesirable reactions.
Acknowledgement
The author acknowledges the School of Pharmacy and Emerging Sciences, Baddi University of Emerging Sciences and Technology, Baddi, Dist. Solan, Himachal Pradesh, India (Pin Code: 173205), and Prof. Ravinesh Mishra, Dean of the School of Pharmacy and Emerging Sciences, for their support, literature, and guidance during the preparation of this review article.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest
Data Availability Statement
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors’ Contribution
- Ravinesh Mishra: Supervision
- Manju Jakhar: Supervision
- Rajeev Dhiman: Conceptualisation, Methodology, Writing – Original Draft
- Laraib Khan: Data Collection, Analysis, Writing – Review & Editing
- Aditya Raj: Visualisation- Literature survey
- Vandana Devi: Project Administration, Writing – Review & Editing
- Anjali Kumari: Data Collection, Writing – Review & Editing
- Ajay Kumar: Resources, Literature survey
References
- Natesan V, Kim SJ. Lipid metabolism, disorders, and therapeutic drugs—Review. Biomol Ther. 2021;29(6):596. doi:10.4062/biomolther.2021.074.
CrossRef - Mitrovic B, Gluvic ZM, Obradovic M, et al. Non-alcoholic fatty liver disease, metabolic syndrome, and type 2 diabetes mellitus: Where do we stand today? Arch Med Sci. 2022;19(4):884. doi:10.5114/aoms.2022.115846.
CrossRef - Ferguson D, Finck BN. Emerging therapeutic approaches for the treatment of NAFLD and type 2 diabetes mellitus. Nat Rev Endocrinol. 2021;17(8):484-495. doi:10.1038/s41574-021-00552-0.
CrossRef - Ordovas JM, Shen J. Gene–environment interactions and susceptibility to metabolic syndrome and other chronic diseases. J Periodontol. 2008;79:1508-1513. doi:10.1902/jop.2008.080082.
CrossRef - World Health Organization. Global report on hypertension: The race against a silent killer. World Health Organization. Published September 19, 2023.
- Stefan N, Cusi K. A global view of the interplay between non-alcoholic fatty liver disease and diabetes. Lancet Diabetes Endocrinol. 2022;10(4):284-296. doi:10.1016/S2213-8587(22)00036-4.
CrossRef - Golden SH, Robinson KA, Saldanha I, et al. Prevalence and incidence of endocrine and metabolic disorders in the United States: A comprehensive review. J Clin Endocrinol Metab. 2009;94(6):1853-1878. doi:10.1210/jc.2008-1987.
CrossRef - Faienza MF, Chiarito M, Molina-Molina E, et al. Childhood obesity, cardiovascular, and liver health: A growing epidemic with age. World J Pediatr. 2020;16:438-445. doi:10.1007/s12519-020-00324-5.
CrossRef - Pereira SS, Alvarez-Leite JI. Low-grade inflammation, obesity, and diabetes. Curr Obes Rep. 2014;3:422-431. doi:10.1007/s13679-014-0127-4.
CrossRef - Cusi K. Treatment of patients with type 2 diabetes and non-alcoholic fatty liver disease: Current approaches and future directions. Diabetologia. 2016;59:1112-1120. doi:10.1007/s00125-016-3971-4.
CrossRef - Lim S, Eckel RH. Pharmacological treatment and therapeutic perspectives of metabolic syndrome. Rev Endocr Metab Disord. 2014;15:329-341. doi:10.1007/s11154-014-9294-0.
CrossRef - Xie X, Wu C, Hao Y, et al. Benefits and risks of drug combination therapy for diabetes mellitus and its complications: A comprehensive review. Front Endocrinol. 2023;14:1301093. doi:10.3389/fendo.2023. 1301093.
CrossRef - Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006;444(7121):860-867. doi:10.1038/nature05485.
CrossRef - Galicia-Garcia U, Benito-Vicente A, Jebari S, et al. Pathophysiology of type 2 diabetes mellitus. Int J Mol Sci. 2020;21(17):6275. doi:10.3390/ijms21176275.
CrossRef - Samuel VT, Shulman GI. Mechanisms for insulin resistance: Common threads and missing links. Cell. 2012;148(5):852-871. doi:10.1016/j.cell.2012.02.017.
CrossRef - Norton L, Shannon C, Gastaldelli A, DeFronzo RA. Insulin: The master regulator of glucose metabolism. Metabolism. 2022;129:155142. doi:10.1016/j.metabol.2022.155142.
CrossRef - Rahman MS, Hossain KS, Das S, et al. Role of insulin in health and disease: An update. Int J Mol Sci. 2021;22(12):6403. doi:10.3390/ijms22126403.
CrossRef - Blüher M. Adipose tissue dysfunction contributes to obesity-related metabolic diseases. Best Pract Res Clin Endocrinol Metab. 2013;27(2):163-177. doi:10.1016/j.beem.2012.12.003.
CrossRef - Zong Y, Li H, Liao P, et al. Mitochondrial dysfunction: Mechanisms and advances in therapy. Signal Transduct Target Ther. 2024;9(1):124. doi:10.1038/s41392-024-00484-1.
CrossRef - Zhao X, An X, Yang C, et al. The crucial role and mechanism of insulin resistance in metabolic disease. Front Endocrinol. 2023;14:1149239. doi:10.3389/fendo.2023.1149239.
CrossRef - Khan S, Wang CH. ER stress in adipocytes and insulin resistance: Mechanisms and significance. Mol Med Rep. 2014;10(5):2234-2240. doi:10.3892/mmr.2014.2634.
CrossRef - Capurso C, Capurso A. From excess adiposity to insulin resistance: The role of free fatty acids. Vasc Pharmacol. 2012;57(2-4):91-97. doi:10.1016/j.vph.2012.03.007.
CrossRef - Al-Mrabeh A. β-Cell dysfunction, hepatic lipid metabolism, and cardiovascular health in type 2 diabetes: New directions of research and novel therapeutic strategies. Biomedicines. 2021;9(2):226. doi:10.3390/biomedicines9020226.
CrossRef - Drews G, Krippeit-Drews P, Düfer M. Oxidative stress and beta-cell dysfunction. Pflugers Arch Eur J Physiol. 2010;460:703-718. doi:10.1007/s00424-010-0840-3.
CrossRef - Xu L, Yang Q, Zhou J. Mechanisms of abnormal lipid metabolism in the pathogenesis of disease. Int J Mol Sci. 2024;25(15):8465. doi:10.3390/ijms25158465.
CrossRef - Eckel RH, Grundy SM, Zimmet PZ. The metabolic syndrome. Lancet. 2005;365(9468):1415-1428. doi:10.1016/S0140-6736(05)66378-7.
CrossRef - Kahn SE, Hull RL, Utzschneider KM. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature. 2006;444(7121):840-846. doi:10.1038/nature05486.
CrossRef - Saltiel AR, Olefsky JM. Inflammatory mechanisms linking obesity and metabolic disorders. J Clin Invest. 2017;127(1):1-4. doi:10.1172/JCI94008.
CrossRef - Glass CK, Olefsky JM. Macrophages, inflammation, and insulin resistance. Annu Rev Physiol. 2012;74:131-155. doi:10.1146/annurev-physiol-020911-153204.
- Landry Y, Gies JP. Drugs and their molecular targets: An updated overview. Fundam Clin Pharmacol. 2008;22(1):1-8. doi:10.1111/j.1472-8206.2007.00588.x.
CrossRef - Nagini S. Breast cancer: Current molecular therapeutic targets and new players. Anti-Cancer Agents Med Chem. 2017;17(2):152-163. doi:10.2174/1871520617666170119114400.
CrossRef - Lean MEJ, Leslie WS, Barnes AC, et al. Primary care-led weight management for type 2 diabetes. Lancet. 2018;391(10120):541-551. doi:10.1016/S0140-6736(17)32439-3.
CrossRef - Holman RR, Paul SK, Bethel MA, et al. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359(15):1577-1589. doi:10.1056/NEJMoa0806470.
CrossRef - Chinetti G, Fruchart JC, Staels B. Peroxisome proliferator-activated receptors (PPARs): Nuclear receptors at the crossroads between lipid metabolism and inflammation. Inflamm Res. 2000;49:497-505. doi:10.1007/s000110050684.
CrossRef - Tyagi S, Gupta P, Saini AS, Kaushal C, Sharma S. The peroxisome proliferator-activated receptor: A family of nuclear receptors role in various diseases. J Adv Pharm Technol Res. 2011;2(4):236-240. doi:10.4103/2231-4040.90928.
CrossRef - Kadowaki T, Yamauchi T. Adiponectin and adiponectin receptors. Endocr Rev. 2005;26(3):439-451. doi:10.1210/er.2005-0003.
CrossRef - Weiss R, Dziura J, Burgert TS, et al. Obesity and the metabolic syndrome in children and adolescents. N Engl J Med. 2004;350(23):2362-2374. doi:10.1056/NEJMoa031018.
CrossRef - Shoelson SE, Herrero L, Naaz A. Obesity, inflammation, and insulin resistance. Gastroenterology. 2007;132(6):2169-2180. doi:10.1053/j.gastro.2007.03.059.
CrossRef - Gallwitz B. Clinical use of DPP-4 inhibitors. Front Endocrinol. 2019;10:389. doi:10.3389/fendo.2019.00389.
CrossRef - Verma AK, Sharma A, Subramaniyam N, Gandhi CR. Augmenter of liver regeneration: Mitochondrial function and steatohepatitis. J Hepatol. 2022;77(5):1410-1421. doi:10.1016/j.jhep.2022.07.040.
CrossRef - Carr RM, Reid AE. FXR agonists as therapeutic agents for non-alcoholic fatty liver disease. Curr Atheroscler Rep. 2015;17:1-4. doi:10.1007/s11883-015-0510-4.
CrossRef - Xu H, Barnes GT, Yang Q, et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J Clin Invest. 2003;112(12):1821-1830. doi:10.1172/JCI200319510.
CrossRef - Gupta R, Srivastava D, Sahu M, et al. Artificial intelligence to deep learning: Machine intelligence approach for drug discovery. Mol Divers. 2021;25:1315-1360. doi:10.1007/s11030-021-10243-7.
CrossRef - Gupta U, Pranav A, Kohli A, et al. The contribution of artificial intelligence to drug discovery: Current progress and prospects for the future. In: Microbial Data Intelligence and Computational Techniques for Sustainable Computing. Springer; 2024:1-23.
CrossRef - Ozaybi MQ, Madkhali AN, Alhazmi MA, et al. The role of artificial intelligence in drug discovery and development. Egypt J Chem. 2024;67(13):1541-1547. doi:10.21608/ejchem.2024.32459.5183.
CrossRef - Perakakis N, Yazdani A, Karniadakis GE, Mantzoros C. Omics, big data and machine learning as tools to propel understanding of biological mechanisms and to discover novel diagnostics and therapeutics. Metabolism. 2018;87:A1-A9. doi:10.1016/j.metabol.2018.07.001.
CrossRef - Banerjee I, Ling Y, Chen MC, et al. Comparative effectiveness of convolutional neural network (CNN) and recurrent neural network (RNN) architectures for radiology text report classification. Artif Intell Med. 2019;97:79-88. doi:10.1016/j.artmed.2019.03.003.
CrossRef - Chandra A, Goyal N, Behera A, Samanta J, Sharma T. Artificial intelligence-based protein structure prediction and systems biology-guided smart drug screening. In: Biochemical and Molecular Pharmacology in Drug Discovery. Elsevier; 2024:323-352.
CrossRef - Martinelli DD. Generative machine learning for de novo drug discovery: A systematic review. Comput Biol Med. 2022;145:105403. doi:10.1016/j.compbiomed.2022.105403.
CrossRef - Zhou Y, Wang F, Tang J, Nussinov R, Cheng F. Artificial intelligence in COVID-19 drug repurposing. Lancet Digit Health. 2020;2(12):e667-e676. doi:10.1016/S2589-7500(20)30291-8.
CrossRef - Dar MA, Arafah A, Bhat KA, et al. Multiomics technologies: role in disease biomarker discoveries and therapeutics. Brief Funct Genomics. 2023;22(2):76-96. doi:10.1093/bfgp/elac010.
CrossRef - Fiatal S, Adany R. Application of single-nucleotide polymorphism-related risk estimates in identification of increased genetic susceptibility to cardiovascular diseases: A literature review. Front Public Health. 2018;5:358. doi:10.3389/fpubh.2017.00358.
CrossRef - Meroni M, Longo M, Tria G, Dongiovanni P. Genetics is of the essence to face NAFLD. Biomedicines. 2021;9(10):1359. doi:10.3390/biomedicines9101359.
CrossRef - Stančáková A, Laakso M. Genetics of metabolic syndrome. Rev Endocr Metab Disord. 2014;15:243-252. doi:10.1007/s11154-014-9284-2.
CrossRef - Chen ZZ, Gerszten RE. Metabolomics and proteomics in type 2 diabetes. Circ Res. 2020;126(11):1613-1627. doi:10.1161/CIRCRESAHA.120.316699.
CrossRef - Letertre MP, Giraudeau P, De Tullio P. Nuclear magnetic resonance spectroscopy in clinical metabolomics and personalized medicine: Current challenges and perspectives. Front Mol Biosci. 2021;8:698337. doi:10.3389/fmolb.2021.698337.
CrossRef - Mishra AS, Vasanthan M, Malliappan SP. Drug repurposing: A leading strategy for new threats and targets. ACS Pharmacol Transl Sci. 2024;7(4):915-932. doi:10.1021/acsptsci.4b00018.
CrossRef - Leung EL, Cao ZW, Jiang ZH, Zhou H, Liu L. Network-based drug discovery by integrating systems biology and computational technologies. Brief Bioinform. 2013;14(4):491-505. doi:10.1093/bib/bbs071.
CrossRef - Alberti KG, Zimmet P, Shaw J. The metabolic syndrome—a new worldwide definition. Lancet. 2005;366(9491):1059-1062. doi:10.1016/S0140-6736(05)67402-8.
CrossRef - Piché ME, Tchernof A, Després JP. Obesity phenotypes, diabetes, and cardiovascular diseases. Circ Res. 2020;126(11):1477-1500. doi:10.1161/CIRCRESAHA.120.315443.
CrossRef - Guerra JV, Dias MM, Brilhante AJ, et al. Multifactorial basis and therapeutic strategies in metabolism-related diseases. Nutrients. 2021;13(8):2830. doi:10.3390/nu13082830.
CrossRef - Scotti L, Monteiro AF, de Oliveira Viana J, et al. Multi-target drugs against metabolic disorders. Endocr Metab Immune Disord Drug Targets. 2019;19(4):402-418. doi:10.2174/1871530319666190128154157.
CrossRef - Pabreja K, Mohd MA, Koole C, Wootten D, Furness SG. Molecular mechanisms underlying physiological and receptor pleiotropic effects mediated by GLP-1R activation. Br J Pharmacol. 2014;171(5):1114-1128. doi:10.1111/bph.12486.
CrossRef - Keating ST, El-Osta A. Epigenetics and metabolism. Circ Res. 2015;116(4):715-736. doi:10.1161/CIRCRESAHA.116.303858.
CrossRef - Delou JM, Souza AS, Souza LC, Borges HL. Highlights in resistance mechanism pathways for combination therapy. Cells. 2019;8(9):1013. doi:10.3390/cells8091013.
CrossRef - Ilan Y. Next-generation personalized medicine: Implementation of variability patterns for overcoming drug resistance in chronic diseases. J Pers Med. 2022;12(8):1303. doi:10.3390/jpm12081303.
CrossRef - Wang N, Ma T, Yu B. Targeting epigenetic regulators to overcome drug resistance in cancers. Signal Transduct Target Ther. 2023;8(1):69. doi:10.1038/s41392-023-01092-6.
CrossRef - Karkhaneh L, Tabatabaei-Malazy O, Bandarian F, Mohseni S, Larijani B. Pharmacogenomics of sulfonylureas in type 2 diabetes mellitus: A systematic review. J Diabetes Metab Disord. 2022;21(1):863-879. doi:10.1007/s40200-022-00989-3.
CrossRef - Polyzos SA, Bugianesi E, Kountouras J, Mantzoros CS. Nonalcoholic fatty liver disease: Updates on associations with the metabolic syndrome and lipid profile and effects of treatment with PPAR-γ agonists. Metabolism. 2017;66:64-68. doi:10.1016/j.metabol.2016.08.008.
CrossRef - Wellen KE, Hotamisligil GS. Inflammation, stress, and diabetes. J Clin Invest. 2005;115(5):1111-1119. doi:10.1172/JCI25102.
CrossRef - Park H, Otte A, Park K. Evolution of drug delivery systems: From 1950 to 2020 and beyond. J Control Release. 2022;342:53-65. doi:10.1016/j.jconrel.2022.01.023.
CrossRef - Lopez-Yus M, Hörndler C, Borlan S, Bernal-Monterde V, Arbones-Mainar JM. Unraveling adipose tissue dysfunction: Molecular mechanisms, novel biomarkers, and therapeutic targets for liver fat deposition. Cells. 2024;13(5):380. doi:10.3390/cells13050380.
CrossRef - Arnaoutova I, Zhang LD, Chen HD, et al. Correction of metabolic abnormalities in a mouse model of glycogen storage disease type Ia by CRISPR/Cas9-based gene editing. Mol Ther. 2021;29(4):1602-1610. doi:10.1016/j.ymthe.2021.02.005.
CrossRef - Kaur G, Arora J, Sodhi AS, Bhatia S, Batra N. Nanotechnology and CRISPR/Cas-mediated gene therapy strategies: Potential role for treating genetic disorders. Mol Biotechnol. 2024:1-23. doi:10.1007/s12033-024-00380-5.
CrossRef - Heal DJ, Gosden J, Smith SL. Regulatory challenges for new drugs to treat obesity and comorbid metabolic disorders. Br J Clin Pharmacol. 2009;68(6):861-874. doi:10.1111/j.1365-2125.2009.03517.x.
CrossRef - Cordeiro JV. Ethical and legal challenges of personalized medicine: Paradigmatic examples of research, prevention, diagnosis and treatment. Rev Port Saúde Pública. 2014;32(2):164-180. doi:10.1016/j.rpsp.2014.04.001.
CrossRef - Francisco KK, Apuhin AE, Tan MJ, et al. Can personalized medicine coexist with health equity? Examining the cost barrier and ethical implications. arXiv preprint arXiv:2411.02307. Published November 4, 2024. doi:10.48550/arXiv.2411.02307.
- Islam Z, Aslam M, Khan AU, ur Rehman M. Genetic testing and its role in precision medicine: Understand disease risk and tailoring treatments. Indus J Biosci Res. 2025;3(1):461-470. doi:10.5281/zenodo.5844705.
CrossRef - Brown MS, Goldstein JL. Selective insulin resistance in the metabolic syndrome. Cell Metab. 2009;9(2):85-87. doi:10.1016/j.cmet.2008.12.009.
CrossRef - O’Rahilly S, Farooqi IS. Human obesity and insulin resistance. Curr Opin Genet Dev. 2008;18(4):346-352. doi:10.1016/j.gde.2008.06.001.
CrossRef - Grundy SM. Metabolic syndrome pandemic. Arterioscler Thromb Vasc Biol. 2008;28(4):629-636. doi:10.1161/ATVBAHA.107.151223.
CrossRef - DeFronzo RA, Tripathy D. Skeletal muscle insulin resistance is the primary defect in type 2 diabetes. Diabetes Care. 2009;32(Suppl 2):S157. doi:10.2337/dc09-S302.
CrossRef - Olefsky JM, Glass CK. Macrophages, inflammation, and insulin resistance. Annu Rev Physiol. 2010;72(1):219-246. doi:10.1146/annurev-physiol-021909-135846.
CrossRef - Rask-Madsen C, Kahn CR. Tissue-specific insulin signaling, metabolic syndrome, and cardiovascular disease. Arterioscler Thromb Vasc Biol. 2012;32(9):2052-2059. doi:10.1161/ATVBAHA.112.249944.
CrossRef - Blüher M. Obesity: Global epidemiology and pathogenesis. Nat Rev Endocrinol. 2019;15(5):288-298. doi:10.1038/s41574-019-0176-8.
CrossRef - Shulman GI. Cellular mechanisms of insulin resistance. J Clin Invest. 2000;106(2):171-176. doi:10.1172/JCI10542.
CrossRef - Ginsberg HN. Insulin resistance and cardiovascular disease. J Clin Invest. 2000;106(4):453-458. doi:10.1172/JCI10724.
CrossRef - Marchesini G, Brizi M, Bianchi G, et al. Nonalcoholic fatty liver disease: A feature of the metabolic syndrome. Diabetes. 2001;50(8):1844-1850. doi:10.2337/diabetes.50.8.1844.
CrossRef - Grundy SM. Obesity, metabolic syndrome, and cardiovascular disease. J Clin Endocrinol Metab. 2004;89(6):2595-2600. doi:10.1210/jc.2003-031370.
CrossRef - Hill JO, Wyatt HR, Peters JC. Energy balance and obesity. Circulation. 2012;126(1):126-132. doi:10.1161/CIRCULATIONAHA.111.087213.
CrossRef - Cusi K. Role of insulin resistance and lipotoxicity in non-alcoholic steatohepatitis. Clin Liver Dis. 2009;13(4):545-563. doi:10.1016/j.cld.2009.08.001.
CrossRef - Kim JY, Van De Wall E, Laplante M, et al. Obesity-associated improvements in metabolic profile through expansion of adipose tissue. J Clin Invest. 2007;117(9):2621-2637. doi:10.1172/JCI32030.
CrossRef - Weisberg SP, McCann D, Desai M, et al. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112(12):1796-1808. doi:10.1172/JCI200319629.
CrossRef







