Manuscript accepted on :08-02-2026
Published online on: 09-03-2026
Plagiarism Check: Yes
Reviewed by: Dr. Farah Ramzi Noori
Second Review by: Dr. Audrey Jacob
Final Approval by: Dr. Patorn Piromchai
Yenny Kandarini1* and I Made Adi Narendranatha Komara2
and I Made Adi Narendranatha Komara2
1Department of Internal Medicine, Udayana University, Denpasar, Bali, Indonesia.
2Department of Internal Medicine Resident, Udayana University, Denpasar, Bali, Indonesia.
Corresponding Author E-mail: ykandarini@gmail.com
Abstract
Chronic kidney disease (CKD) is defined as kidney damage, structurally and/or functionally, that persists for more than three months. Continuous ambulatory peritoneal dialysis (CAPD) has several benefits compared to other renal replacement therapies (RRT), although the results are inconclusive. The objective of this review and meta-analysis was to evaluate the survival rate of CKD patients undergoing CAPD. A systematic search according to PRISMA 2020 guidelines was carried out through online databases for relevant studies evaluating the survival rate of CKD patients undergoing CAPD. The articles obtained were observational studies that had been adjusted to the inclusion and exclusion criteria. Newcastle Ottawa Scale (NOS) was used to assess the study quality. Analysis was carried out with Review Manager software v.5.4. 11 studies were obtained, most of which were retrospective cohort studies from China, with 3700 aged 7.6 to 74.1 years and follow-up durations ranging from 5 months to 18 months. Based on the analysis, the pooled survival rate in 1, 2, 3, 5, and 10 years was 91% (95% CI 82–96%), 73% (95% CI 60–83%), 63% (95% CI 44–78%), 63% (95% CI 40–82), and 73% (95% CI 38–92%) respectively. Heterogeneity was high across time points (I2 96%–99%). Egger's 2-year survival rate analysis test showed a significant publication bias risk (p=0.046). CKD patients who underwent CAPD had a survival rate of 91%, 73%, 63%, and 73% at 1, 2, 3, 5, and 10 years, respectively.
Keywords
Chronic Kidney Disease (CKD); Clinical Epidemiology; Continuous Ambulatory Peritoneal Dialysis (CAPD); Survival Rate
| Copy the following to cite this article: Kandarini Y, Komara I. M. A. N. Survival Rate of Chronic Kidney Disease (CKD) Patients Undergoing Continuous Ambulatory Peritoneal Dialysis (CAPD): A Meta-Analysis. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Kandarini Y, Komara I. M. A. N. Survival Rate of Chronic Kidney Disease (CKD) Patients Undergoing Continuous Ambulatory Peritoneal Dialysis (CAPD): A Meta-Analysis. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/4cUJWvN |
Introduction
Chronic kidney disease (CKD) is defined as kidney damage, structurally and/or functionally, that persists for more than three months. The functional disorder referred to is a decrease in the glomerular filtration rate (GFR) reaching <60 mL/minute/1.73 m2 or albuminuria reaching ≥ 30 mg in 24 hours.1 Based on its etiology, CKD can be caused by various conditions. In most cases, CKD itself is a complication that occurs due to other health problems. Some of these etiologies include diabetes mellitus (around 30%—50 % of all CKD cases are generally caused by DM), hypertension, kidney infections, congenital abnormalities, and others.2 However, the main mechanism of CKD can be explained in general as chronic dysfunction of microvascularization in the nephron, which ultimately causes kidney functional and structural disorders.1
When viewed from an epidemiological perspective, the exact incidence rate of CKD throughout the world is difficult to ascertain because the majority of CKD cases are asymptomatic, where the majority of patients who are finally diagnosed with CKD come for examination at an advanced stage.3 According to Centers for Disease Control and Prevention (CDC) data, the incidence of CKD elevated from 11.8% in 1988-1994 to 14.2% in 2015-2016.4 However, several recent studies have stated that the incidence of advanced-stage CKD has decreased. In a meta-analysis by Hill et al.,5 it was found that the prevalence of CKD was dominated by CKD stages 1, 2, and 3. On the other hand, the prevalence of CKD stages 4 and 5 only reached 0.4% and 0.1%, respectively.5
Renal replacement therapy (RRT) is one of the treatment choices for functional and structural damage to the kidney patients, both in acute and chronic conditions. This therapy substitutes for the function of impaired kidneys to carry out functions in fluid and electrolyte regulation, excretion of metabolic waste, and other harmful substances.6 This substitution can take the form of a kidney transplant (which is the most recommended type of RRT) or by using dialysis, either hemodialysis (HD) or peritoneal dialysis (PD).7
Peritoneal dialysis is one of the modalities used in dialysis and has several advantages compared to HD. Compared with HD, patients undergoing PD do not need to come regularly to the hospital for dialysis procedures because the procedures can be done at home.8 One of the developments in PD today is continuous ambulatory peritoneal dialysis (CAPD). CAPD is a development of PD, which is part of continuous renal replacement therapy (CRRT). Based on various studies, RRT using CAPD is known to have various advantages, such as having a risk of patients experiencing hypotension when compared to HD because CAPD has a slower filtration rate and has a greater collective filtration capacity when compared to HD, so it can provide efficacy in better elimination of toxins and metabolic waste.9
However, data regarding CAPD is still limited, both in clinical applications and research data regarding its efficacy. The literature states that only about 7% of patients receiving RRT in the United States undergo PD procedures.8 A study conducted by Gunawan and Sakti10 found a one-year survival rate of 80%. In addition, excellent results were found by Yi et al., 11 which found the one-year survival rate of patients undergoing CAPD at 99.6%. Based on other studies that discuss the efficacy and safety of CAPD, the results are inconclusive because there are inconsistent results ranges between studies. Apart from that, there are comorbid conditions and complications that play a role in the mortality rate of CKD patients undergoing CAPD, such as the incidence of peritonitis, comorbid hypertension, diabetes mellitus, and others. Thus, this systematic review and meta-analysis aims to assess the survival rate of patients undergoing CAPD.
Materials and Methods
All findings pertaining to the study topic were identified, evaluated, and interpreted in this meta-analysis study. To find all pertinent papers, the PICOS (Population, Intervention, Comparison, Outcome, Studies) approach was employed. The 2020 Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) standards became the basis of the systematic search technique.
Study selection
All of the study articles included in this review are studies that report and assess the survival rate of chronic renal disease patients receiving CAPD. All the studies were identified from PubMed, ScienceDirect, Google Scholar, and the Cochrane Library online databases. The survival rate referred to in this study is the percentage of patients in the research group who are still alive for a certain period after undergoing a procedure in the form of CAPD, expressed in the period 1 to 10 years after the procedure. The study’s population were CKD patients who had performed CAPD. Studies were excluded if (1) the populations underwent dialysis therapy other than using the CAPD method, (2) the follow-up time was less than one year, (3) no clear statement regarding the survival rate, (4) non-specific study designs.
Literature searching
To maximize search results, several keywords relevant to the PICOs were used to look for pertinent literature. Initially, the author used medical subject headings (MeSH) to determine keywords for the study search. Following MeSH keyword selection, Boolean operators (AND, OR, and NOT) were used to group the keywords in accordance with the research topic. Advanced search and bibliographic search were used to search for the studies. Survival rate AND chronic renal disease AND (peritoneal dialysis) OR (continuous ambulatory peritoneal dialysis) were the search keywords used.
Data extraction and study quality evaluation
This study evaluated the quality of papers to be using critical appraisal. Data extraction was done independently by each author. The New Castle and Ottawa Scale (NOS) for Cohort and Cross-Sectional Studies, which has three assessment components: findings, comparability, and selection, was utilized to assess the study quality. The article was deemed to fit the requirements and included in the inclusion criteria if the overall interpretation reached was rather good, and vice versa.
Data analysis
To analyse the data and draw conclusions, all data was systematically integrated and characterized. The information included population demographic characteristics, measuring techniques, measurement parameters, primary research findings, and research features (such as the last name of first author, the publication year, and the research site). To make analysis easier, data was supplied in tabular form (synthesis matrix). Review Manager software v.5.4 and single proportion analysis were used to quantitatively calculate overall patient survival rate. I-squared and t-squared tests were used to assess heterogeneity. Random effect model was indicated if there was significant heterogeneity value. A quantitative Egger’s test and a qualitative funnel plot method were utilized to assess the publication bias risk. A p-value of <0.05 was determined to be significant.
Results
Literature searching
3688 publications from four online databases (PubMed, ScienceDirect, Google Scholar, and Cochrane) were found during the study search process. Using computer software (Citation Manager), duplicates were eliminated, yielding 2408 papers. 37 publications that could be accessed and subsequently evaluated for eligibility were found through the title and abstract screening process. Additionally, 11 included papers were used for both qualitative (systematic review) and quantitative (meta-analysis) analysis after 26 articles were eliminated due to incomplete or irrelevant data. The following is a description of the PRISMA guideline-based study search flow (Figure 1).
 |
Figure 1: PRISMA Flowchart |
Study characteristics
A total of 11 studies were obtained in this review. A total of 9 studies were retrospective cohort studies, 1 study was a cross-sectional study, and 1 was case-control study. Most of the studies came from China with the total population involved in all inclusion studies being 3700 CKD patients undergoing CAPD therapy. The duration of study follow-up ranged from 5 months to 18 months, with patient averages ranging from 7.6 years to 74.1 years. Table 1 presents the study characteristics.
Table 1: Study characteristic
| Author, year | Study design | Country | Population | Duration of follow-up (Months) | Mean of age (year) |
| Ataei et al.12 | Cross-sectional | Iran | 182 | 18 | 7.69±4.72 |
| Gunawan and Sakti.10 | Retrospective cohort | Indonesia | 674 | 5 | N/A |
| Isla et al.13 | Retrospective cohort | South Africa | 152 | 5 | 36.8±11.4 |
| Joshi et al.14 | Retrospective cohort | China | 805 | 5 | 71.3±4.3 |
| Kit-Chung et al.15 | Case-control | China | 133 | 10 | 74.1+5.9 |
| Prasad et al.16 | Retrospective cohort | India | 66 | 12 | 12.3±3.91 |
| Sakaci et al.17 | Retrospective cohort | Turki | 58 | 13 | 71.8±4.9 |
| Vikrant18 | Retrospective cohort | India | 60 | 10 | 60.2±9.2 |
| Wen et al.19 | Retrospective cohort | China | 716 | 5 | 51±15 |
| Yi et al.11 | Retrospective cohort | China | 433 | 6 | 51.3±15.7 |
| Zhang et al.20 | Retrospective cohort | China | 421 | 10 | 57.9±14.8 |
Study outcome
Most studies report that patients with CAPD have a higher survival rate or lower mortality than patients who do not undergo CAPD, especially in end-stage renal disease (ESRD) patients. A number of studies also report that increasing age has an effect on survival rates regardless of the use of CAPD therapy. On the other hand, 9 studies had good study quality, and 2 others had moderate study quality, so all studies could be used in quantitative analysis. All study outcomes are described in Table 2.
Table 2: Study outcome
| Author, year | Study outcome | Quality of study (NOS) |
| Ataei et al.12 | The long-term survival rate in this study was higher than data in previous studies reported in Europe. | Good |
| Gunawan and Sakti.10 | End Stage Renal Disease (ESRD) patients who received CAPD in this study had good 5-year survival rates. | Good |
| Isla et al.13 | Management of comorbidities that accompany ESRD patients receiving CAPD can prevent morbidity and mortality in patients | Good |
| Joshi et al.14 | Elderly patients have lower survival rates than younger patients. | Moderate |
| Kit-Chung et al.15 | Patients who underwent Helper-Assisted CAPD had worse survival rates | Moderate |
| Prasad et al., 201916 | CAPD is a possible option for ESRD patients in pediatric patients in developing countries considering aspects of limited resources. | Good |
| Sakaci et al.17 | The mortality rate is higher in elderly patients. This is influenced by advanced age and the presence of comorbid conditions that worsen the patient’s condition. | Good |
| Vikrant,18 | The study results showed higher mortality in elderly patients, especially elderly people with a burden of comorbid conditions. | Good |
| Wen et al.19 | High peritoneal dialysate glucose concentrations are associated with mortality rates in patients receiving CAPD | Moderate |
| Yi et al.11 | Doctor-patient contact of no more than two months was independently associated with greater survival rates in patients undergoing CAPD. | Good |
| Zhang et al.20 | The survival rate of patients who received CAPD was found to be better | Good |
Quantitative analysis
To determine the overall survival rate from all included studies, a quantitative analysis was carried out. According to data obtained from the inclusion study, the analysis was carried out using a number of time durations, including 1, 2, 3, 5, and 10 years.
 |
Figure 2: One-year survival rate analysis |
The analysis of survival rates at 1 year included 10 studies, where the overall survival rate was 91% (random effect; 95% CI 82 – 96%; heterogeneity; 𝜏2 <0.01; I2 = 96%) (Figure 2).
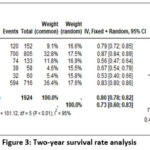 |
Figure 3: Two-year survival rate analysis |
6 studies were included for the survival rates at 2 years analysis, which resulted in 73% of overall survival rate (random effect; 95% CI 60 – 83%; heterogeneity; 𝜏2 <0.01; I2 = 95%) (Figure 3).
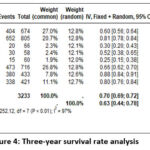 |
Figure 4: Three-year survival rate analysis |
Survival rates at 3 years analysis encompassed 8 studies and the overall survival rate result was 63% (random effect; 95% CI 44 – 78%; heterogeneity; 𝜏2 <0.01; I2 = 97%) (Figure 4).
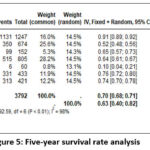 |
Figure 5: Five-year survival rate analysis |
7 studies were pooled and analyzed for the 5-year survival rates, which resulted in a 63% of overall survival rate (random effect; 95% CI 40 – 82%; heterogeneity; 𝜏2 <0.01; I2 = 98%) (Figure 5).
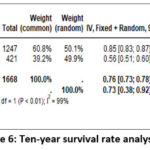 |
Figure 6: Ten-year survival rate analysis |
For the 10-year survival rates, 2 studies were analyzed, which resulted in an overall survival rate of 73% (random effect; 95% CI 38 – 92%; heterogeneity; 𝜏2 <0.01; I2 = 99%) (Figure 6).
Publication bias
Next, publication bias analysis was carried out using Egger’s test; quantitative analysis showed an insignificant p-value in the survival rate analysis for 1, 3, 5, and 10 years, reflecting a low risk of publication bias. Meanwhile, a p-value <0.05 in the 2-year survival rate analysis reflects a significant risk of publication bias. The Egger’s test analysis results are presented in Table 3.
Table 3: Analysis of publication bias with Egger’s test
| No | Survival rate | SE | p-value |
| 1 | 1-year | 57.96 | 0.251 |
| 2 | 2-years | 35.55 | 0.046 |
| 3 | 3-years | 60.26 | 0.167 |
| 4 | 5-years | 175.97 | 0.423 |
Discussion
This meta-analysis aimed to estimate survival rates among CKD patients undergoing continuous ambulatory peritoneal dialysis (CAPD) using 11 included observational studies. CAPD is a home-based dialysis modality that may reduce the need for frequent hospital visits and support quality of life compared with facility-based hemodialysis.
The pooled 1-year survival rate was 91% (95% CI 82–96%), consistent with several included cohorts reporting >90% 1-year survival among CAPD-treated patients, namely by Ataei et al.,12 Joshi et al.,14 Wen et al.,19 Yi et al.,11 and Zhang et al.20 who found high levels of Survival in ESRD patients undergoing CAPD reaches more than 90%. However, the study run by Prasad et al.,16 reported the lowest 1-year survival, which may relate to a high peritonitis burden in pediatric CAPD patients (45/66 cases) and potential loss to follow-up. Peritonitis is a major CAPD complication associated with mortality and technique failure, and outcomes may be worse with virulent or antibiotic-resistant organisms.21,22 Accordingly, prompt pathogen identification, timely antibiotic treatment, and prevention strategies (aseptic technique, early detection, and patient education) are central to improving outcomes.23
When viewed from the 2-year survival rate, the 2-year pooled survival rate was 73% (random effect; CI 95% 60 – 83%). The results of this analysis are consistent with the included studies in this meta-analysis, such as Isla et al.,13 Joshi et al.,14 and Wen et al.,19 which found a 2-year survival rate of more than 70%. However, if we look back at the included studies, those conducted by Vikrant, 201418 and Kit-Cheung et al.15 had the lowest survival rates (53% and 56%, respectively). Based on research conducted by Vikrant, 2014,18 similar results were obtained by Prasad et al.,16 where the percentage of peritonitis incidents reached more than 50% of the entire research sample. Furthermore, research by Kit-Cheung et al.15 found the involvement of other comorbid conditions, such as diabetes mellitus and hypertension. Furthermore, in the analysis of patient survival rates at 3, 5, and 10 years, each pooled survival rate was found to be 63% (95% CI 44 – 78%); 63% (95% CI 40 – 82%); and 73% (95% CI 38 – 92%).
A non-linear trend was observed across survival time points, with survival declining from 1 to 3 years, remaining similar at 5 years, and appearing higher at 10 years. This pattern should be interpreted cautiously, as the 10-year pooled estimate was derived from only two studies, reporting survival rates of 84.8% in the study performed by Ataei et al.12 and 55.7% in the study performed by Zhang et al.20 A non-linear trend was observed across survival time points, with survival declining from 1 to 3 years, remaining similar at 5 years, and appearing higher at 10 years. This pattern should be interpreted cautiously, as the 10-year pooled estimate was derived from only two studies, reporting survival rates, such as the study conducted by Prasad et al.,16 which in their study found a three-year survival rate of only 30%. which documented a 3-year survival of only 30%. Conversely, large-scale studies with moderate survival outcomes, including a study conducted by Gunawan and Sakti10 (3- and 5-year survival of 60% and 52% in 647 patients), exerted substantial weight on the pooled estimates.
Mortality in CKD patients undergoing CAPD is multifactorial and influenced by patient-related, treatment-related, and socioeconomic factors. Established predictors include advanced age, comorbidities such as diabetes and cardiovascular disease, nutritional status, and dialysis adequacy. In a study run by Kit-Cheung et al.,15 it was found that the average blood pressure of patients was more than 140mmHg. This study’s results align with other inclusion studies, such as Gunawan and Sakti10 which also found that CKD patients had comorbid hypertension. CKD is a chronic condition that can affect or be influenced by the function of other organ systems in the body, especially the cardiovascular system, or what is often referred to as cardio-renal syndrome (CRS). a bidirectional interaction in which dysfunction of the heart and kidneys mutually exacerbates disease progression.24 Hemodynamic impairment, neurohormonal activation (RAAS and sympathetic pathways), and endothelial dysfunction contribute to reduced renal perfusion, sodium retention, inflammation, and chronic renal ischemia, ultimately promoting renal fibrosis and progression of CKD.25,26
This meta-analysis has several limitations. This study only involved studies on the Asian and African continents, thereby limiting the generalization of data characteristics in the included studies. In addition, this study did not control for certain study characteristics that might play a role in the clinical outcomes of CAPD treatment because the number of studies discussing the survival rate of CAPD patients is quite rare. Importantly, heterogeneity was very high across all pooled time points (I2 > 90%), indicating that the summary survival estimates should be interpreted as approximate averages across markedly different study populations and settings, rather than precise universal benchmarks. Potential sources of heterogeneity include regional differences, patient age distributions, comorbidity burden, peritonitis incidence, dialysis adequacy, and variations in clinical protocols and follow-up duration. Given this heterogeneity and the limited number of studies contributing to long-term outcomes, conclusions regarding 10-year survival should remain conservative.
Conclusion
Based on the findings of this meta-analysis, patients with chronic kidney disease undergoing continuous ambulatory peritoneal dialysis demonstrate favourable short- to mid-term survival outcomes. However, substantial variability across included studies indicates that survival estimates should be interpreted cautiously. Differences in patient characteristics, comorbid conditions, and CAPD-related complications may significantly influence survival outcomes.
Although pooled long-term survival appeared acceptable, interpretation of 10-year survival rates should remain conservative due to the limited number of studies contributing long-term data and the presence of substantial heterogeneity. Further large-scale, well-designed studies with standardized follow-up and comprehensive adjustment for clinical confounders are needed to better characterize long-term survival among CAPD patients and to improve the generalizability of these findings across different populations and healthcare settings.
Acknowledgement
We deeply thank the Department of Internal Medicine, the Faculty of Medicine, and Udayana University for supporting this study.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Yenny Kandarini: Conceptualization, Methodology, Writing.
- I Made Adi Narendranatha Komara: Data Collection, Analysis, Writing.
Reference
- Wilson S, Mone P, Jankauskas SS, Gambardella J, Santulli G. Chronic kidney disease: Definition, updated epidemiology, staging, and mechanisms of increased cardiovascular risk. J Clin Hypertens. 2021;23(4):831-834. doi:10.1111/jch.14186
CrossRef - Webster AC, Nagler EV, Morton RL, Masson P. Chronic kidney disease. Lancet. 2017;389(10075):1238-1252. doi:10.1016/S0140-6736(16)32064-5
CrossRef - Vaidya SR, Aeddula NR. Chronic kidney disease. In: Statpearls. 2024. PMID:30571025
- Chronic kidney disease in the united states, 2021. Adv Surg Med Spec. 2023:167-182.
- Hill NR, Fatoba ST, Oke JL, et al. Global prevalence of chronic kidney disease – A systematic review and meta-analysis. PLoS One. 2016;11(7):e0158765. doi:10.1371/journal.pone.0158765
CrossRef - Phadke G, Khanna R. Renal replacement therapies. Mo Med. 2011;108(1):45-49. PMID:21462611
- Lameire N, Van Biesen W. Epidemiology of peritoneal dialysis: A story of believers and nonbelievers. Nat Rev Nephrol. 2010;6(2):75-82. doi:10.1038/nrneph.2009.210
CrossRef - Sachdeva B, Zulfiqar H, Aeddula NR. Peritoneal dialysis. In: StatPearls. 2023. PMID:30422574
- Bailie GR, Eisele G. Continuous ambulatory peritoneal dialysis: A review of its mechanics, advantages, complications, and areas of controversy. Ann Pharmacother. 1992;26(11):1409-1420. doi:10.1177/106002809202601115
CrossRef - Gunawan A, Sakti PT. Five-year survival rate of patients with end-stage renal disease on continuous ambulatory peritoneal dialysis (CAPD) at Malang CAPD Center, Indonesia. Acta Med Indones. 2023;55(1):4-9. PMID:36999264
- Yi C, Guo Q, Lin J, Li J, Yu X, Yang X. Patient-doctor contact interval and clinical outcomes in continuous ambulatory peritoneal dialysis patients. Am J Nephrol. 2017;45(4):346-352. doi:10.1159/000464258
CrossRef - Ataei N, Madani A, Esfahani ST, Otoukesh H. Chronic kidney disease in Iran: First report of the national registry in children and adolescences. Urol J. 2020; 18(1): 122- doi: 10.22037/uj.v16i7.5759
- Isla RAT, Mapiye D, Swanepoel CR, Rozumyk N, Hubahib JE, Okpechi IG. Continuous ambulatory peritoneal dialysis in Limpopo Province, South Africa: Predictors of patient and technique survival. Perit Dial Int. 2014;34(5):518-525. doi:10.3747/pdi.2013.00334
CrossRef - Joshi U, Guo Q, Yi C, et al. Clinical outcomes in elderly patients on chronic peritoneal dialysis: A retrospective study from a single center in China. Perit Dial Int. 2014;34(3):299-307. doi:10.3747/pdi.2012.00209
CrossRef - Kit-Cheung JN, Chan GCK, Chow KM, et al. Helper-assisted continuous ambulatory peritoneal dialysis: Does the choice of helper matter? Perit Dial Int. 2020;40(1):34-40. doi:10.1177/0896860819879873
CrossRef - Prasad N, Rangaswamy D, Patel M, et al. Long-term outcomes in children on chronic continuous ambulatory peritoneal dialysis: A retrospective cohort study from a developing country. Pediatr Nephrol. 2019;34(11):2389-2397. doi:10.1007/s00467-019-04311-w
CrossRef - Sakacı T, Ahbap E, Koc Y, et al. Clinical outcomes and mortality in elderly peritoneal dialysis patients. Clinics. 2015;70(5):363-368. doi:10.6061/clinics/2015(05)10
CrossRef - Vikrant S. Long-term clinical outcomes of peritoneal dialysis patients: 9-year experience of a single center from North India. Perit Dial Int. 2014;34(4):426-433. doi:10.3747/pdi.2013.00050
CrossRef - Wen Y, Guo Q, Yang X, et al. High glucose concentrations in peritoneal dialysate are associated with all-cause and cardiovascular disease mortality in continuous ambulatory peritoneal dialysis patients. Perit Dial Int. 2015;35(1):70-77. doi:10.3747/pdi.2013.00083
CrossRef - Zhang F, Liu H, Gong X, et al. Risk factors for mortality in Chinese patients on continuous ambulatory peritoneal dialysis. Perit Dial Int. 2015;35(2):199-205. doi:10.3747/pdi.2013.00164
CrossRef - Hsieh YP, Wang SC, Chang CC, Wen YK, Chiu PF, Yang Y. The negative impact of early peritonitis on continuous ambulatory peritoneal dialysis patients. Perit Dial Int. 2014;34(6):627-635. doi:10.3747/pdi.2013.00024
CrossRef - Li PKT, Szeto CC, Piraino B, et al. Peritoneal dialysis-related infections recommendations: 2010 update. Perit Dial Int. 2010;30(4):393-423. doi:10.3747/pdi.2010.00049
CrossRef - Ye H, Zhou Q, Fan L, et al. The impact of peritoneal dialysis-related peritonitis on mortality in peritoneal dialysis patients. BMC Nephrol. 2017;18(1):1-9. doi:10.1186/s12882-017-0588-4
CrossRef - Ajibowo AO, Okobi OE, Emore E, et al. Cardiorenal syndrome: A literature review. Cureus. 2023;15(7):e41252. doi:10.7759/cureus.41252
CrossRef - Ronco C, Haapio M, House AA, Anavekar N, Bellomo R. Cardiorenal syndrome. J Am Coll Cardiol. 2008;52(19):1527-1539. doi:10.1016/j.jacc.2008.07.051
CrossRef - Pokhrel N, Maharjan N, Dhakal B, Arora RR. Cardiorenal syndrome: A literature review. Exp Clin Cardiol. 2008;13(4):165-170. PMID: 19343160







