Manuscript accepted on :05-01-2026
Published online on: 28-01-2026
Plagiarism Check: Yes
Reviewed by: Dr. Victor Oti
Second Review by: Dr. Gowri Burle
Final Approval by: Dr. Patorn Piromchai
Alankar Shrivastav1* , Monika Singh2
, Monika Singh2 , Tribhuwan Kumar Vashishtha1
, Tribhuwan Kumar Vashishtha1 , Rajat Saxena1
, Rajat Saxena1 , Arun Kumar Mishra1
, Arun Kumar Mishra1 and Navneet Verma1
and Navneet Verma1
1Pharmacy Academy, Faculty of Pharmacy, IFTM University, Moradabad, Uttar Pradesh, India Moradabad, Uttar Pradesh, India
2Department of Pharmacy, Bhartiya College of Pharmacy, Kankhater, Brijghat, Gajraula, Uttar Pradesh, India
Corresponding Author E-mail: alankar1994.ss@gmail.com
Abstract
The Human metapneumovirus (HMPV) is an important human pathogen that predominantly causes acute respiratory tract infections (ARTIs) in infants, the elderly, and immunocompromised hosts. HMPV is also a member of the Paramyxoviridae family and shares several features with the respiratory syncytial virus, including main genetic similarity, seasonality, modes of human‐to‐human transmission, and immune evasion. The virus has a single-stranded, negative-sense RNA genome with nine open reading frames (ORFs) that encode key proteins essential for viral replication, immune evasion, and pathogenic mechanisms. HMPV is highly contagious and spreads through respiratory droplets and direct contact, with peak infections during late winter and early spring. The disease spectrum ranges from mild upper respiratory symptoms to bronchiolitis and pneumonia in the lower respiratory tract. The virus’s capacity for suppressing type I interferon responses accounts for frequent reinfections and more severe disease in high-risk groups. RT-PCR remains the assay of choice for highly sensitive and specific diagnosis of COVID-19. There is currently no approved vaccine or antiviral therapy, and management is supportive, including oxygen therapy and respiratory support in severe cases. Ongoing promising research is directed toward antiviral agents, monoclonal antibodies, and vaccines based on mRNA or protein subunits, as well as live-attenuated and viral vector vaccines. Further studies are important to reduce HMPV attributable morbidity and mortality.
Keywords
Antivirals; Diagnosis; Human Metapneumovirus (HMPV); Immune response; Pathogenesis; Respiratory Infections; Treatment; Vaccine Development
| Copy the following to cite this article: Shrivastav A, Singh M, Vashishtha T. K, Saxena R, Mishra A. K, Verma V. Human Metapneumovirus (HMPV): Epidemiology, Pathogenesis, Clinical Impact, and Advances in Diagnosis, Treatment, and Vaccine Development. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Shrivastav A, Singh M, Vashishtha T. K, Saxena R, Mishra A. K, Verma V. Human Metapneumovirus (HMPV): Epidemiology, Pathogenesis, Clinical Impact, and Advances in Diagnosis, Treatment, and Vaccine Development. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/3Z3Z7KW |
Introduction
Human metapneumovirus (HMPV) is a globally distributed respiratory pathogen that causes acute respiratory tract infections (ARTIs), particularly affecting risk groups such as infants, young children, the elderly, and immunocompromised patients. HMPV was first discovered in 2001, and retrospective analyses now confirm that it must have been circulating in humans for decades and shares genetic relatedness to avian metapneumoviruses. It belongs to the Paramyxoviridae family and the Pneumovirinae subfamily and is homologous to respiratory syncytial virus (RSV).1
HMPV is an enveloped, negative-sense, single-stranded RNA virus whose genome is around 13 kilobases long. The genome encodes nine essential proteins, including the virally encoded fusion (F) and attachment (G) glycoproteins, crucial for viral entry into host cells as well as the avoidance of host antibody-mediated neutralisation. The virus has been divided into two main genetic lineages (A and B) with subspecies (A1, A2, B1, and B2) implicated in its antigenic variation and seasonal resurgence.2 HMPV infection usually peaks during late winter to early spring and often co-circulates with RSV and influenza viruses, which can increase disease severity. The virus is mainly spread through respiratory droplets, direct contact between people, and contaminated surfaces (fomites). It initially settles on the respiratory epithelium after entry, causing inflammation, airway obstruction, and pulmonary injury.
HMPV has a clinical spectrum consisting of mild illness (cough, nasal congestion, fever) to severe lower respiratory tract disease (bronchiolitis, pneumonia), and exacerbation of asthma or chronic obstructive pulmonary disease.3 It is a leading cause of hospitalization for respiratory illness in children. There, it frequently leads to prolonged hospitalization and an increased healthcare burden.4 Molecular techniques, particularly reverse transcription polymerase chain reaction (RT-PCR), are commonly used to diagnose HMPV due to their high sensitivity and specificity. Other tests, including enzyme-linked immunosorbent assay (ELISA) and immunofluorescence assays, are performed less frequently due to their lower sensitivity. However, underdiagnosis is one of the main barriers to an accurate epidemiological picture of the neurological disease.5
Despite significant progress, critical gaps remain in our understanding of hMPV biology. In particular, the molecular basis of immune evasion strategies remains poorly characterised, with limited knowledge of how hMPV modulates interferon signalling and innate immune recognition. Similarly, the precise host–virus interactions that determine disease severity, persistence, and viral clearance are not fully elucidated. Comparative studies with respiratory syncytial virus (RSV) may provide valuable insights, but systematic analyses are lacking. Addressing these gaps is essential to advancing both therapeutic and vaccine development.
HMPV currently has no licensed antiviral agents or vaccines. The clinical management is supportive, providing oxygen supplementation in addition to IV fluids in more severe cases. Other experimental therapies, including ribavirin and fusion inhibitors, are being studied. A multitude of vaccine development platforms are under investigation, including live-attenuated, recombinant protein, virus-like particle (VLP), and mRNA-based platforms.6 Co-infection with RSV, influenza, or bacterial pathogens can worsen disease outcomes and complicate treatment. In-depth research will aid in strengthening knowledge on HMPV pathogenesis, immunity, and lasting immunity. Further awareness of HMPV, enhanced diagnostic capabilities, and faster development of potent vaccines and antiviral therapies are essential steps to mitigating the burden on global health that this pathogen poses.
Virology and Genomic Structure
Virology of HMPV
Human metapneumovirus (HMPV) is a negative-sense, single-stranded RNA virus that belongs to the Paramyxoviridae family and Pneumovirinae subfamily. Its genome is a non-segmented, negative-sense, single-stranded RNA. HMPV was first described in the Netherlands in 2001, but retrospective studies suggest it has been circulating in human populations for decades. The structure and clinical features of HMPV are very similar to those of the respiratory syncytial virus (RSV), sharing similarities in terms of morphology, replication strategies, and disease manifestation.7
HMPV is an enveloped virus, so its RNA genome is enclosed within a lipid bilayer derived from the host cell membrane. It mainly affects epithelial cells of the respiratory tract, leading to inflammation and immune activation as well as lung injury. It is spread primarily via respiratory droplets and through direct contact with contaminated secretions. After entering the host cell, HMPV binds to the host cell surface and fuses the cell membrane, mediated by the viral fusion (F) glycoprotein. This is necessary so that the viral genome can be released into the cytoplasm by allowing the viral envelope to fuse with the host plasma membrane. Subsequent pH-dependent fusion in endosomes further facilitates the release of viral RNA necessary for replication and transcription in the cytosol of the host cell.8
Host immune response to HMPV (innate and adaptive). Enhanced signalling from PRRs (including TLRs) TLRS recognising viral components after viral infection, can upregulate, activate type I or type II IFN and pro-inflammatory cytokines. In contrast, HMPV developed immune evasion strategies targeting some features of personal immunity, e.g., inhibition of IFN signalling, alteration of antigen presentation, and even the modulation of T-cell responses.9
Genomic Structure of HMPV
The genome of HMPV is about 13.3 kb in length and encodes eight major proteins involved in viral replication, immunoevasion, and host cell infection. The nucleoprotein (N) binds and encapsidates viral RNA to create a ribonucleoprotein (RNP) complex to protect and stabilise the genome and interacts with phosphoprotein (P) and large polymerase (L) protein to control RNA synthesis.10 The phosphoprotein (P) serves as a cofactor of the RNA-dependent RNA polymerase (L), stabilising the replication complex and ensuring proper transcription. The matrix protein (M) preserves virion structure and promotes viral assembly, budding, and genome encapsidation. The fusion protein (F) promotes viral entry via membrane fusion between the viral envelope and host cell membranes and is a main target for neutralising antibodies and vaccine development. Small hydrophobic protein SH encoded by the viral genome regulates the host immune response, possibly by blocking cytokine signalling and inflammation.11 Viral attachment is mediated by glycoprotein (G), which, however, is not required for infection, and its high variability can yield antigenic diversity necessary for neutralisation escape. Large polymerase protein (L): The large polymerase protein (L) acts as the RNA-dependent RNA polymerase, directing genome replication and mRNA transcription. The M2 gene codes for two proteins: M2-1 promotes transcription, and M2-2 regulates the transition from transcription to replication to ensure an appropriate level of viral gene expression. HMPV has a typical Paramyxoviridae genome structure yet does not encode for RSV’s NS1 and NS2 immune evasion proteins, hinting at different mechanisms to modulate host immunity.12 These proteins (F and G) are crucial for viral entry and antigenic variation and thus have been the target of vaccine and antiviral research efforts. Thus, these proteins represent promising targets for the development of therapeutic interventions against HMPV.
Genetic Variability and Evolution
HMPV is classified into two major genetic lineages (A and B), which can be further subdivided into two subgroups, A1, A2, B1, and B2, which have a wide range of genetic diversity (mainly in cellular and viral subgenomic G and F proteins) and can antigenically escape immune detection. Although this variability exists, HMPV does not exhibit the significant antigenic drift of the influenza viruses, and so, while reinfection is common, the general genetic stability results in milder disease owing to some level of pre-existing immunity over the lifetime. 13
Studies looking at the evolution of HMPV show that it is related through a common ancestor to avian metapneumoviruses (AMPVS), which infect birds. Molecular clock analyses indicate that HMPV diverged from AMPVS hundreds of years ago when HMPV mutations adapted to human hosts. Because the evolutionary history of these viruses is interconnected across species, this highlights the need to monitor the activity of closely related viruses in the wild.
Epidemiology
Human Metapneumovirus (HMPV) is a worldwide disseminated respiratory virus that predominantly affects youngsters, the elderly and immunocompromised individuals, and accounts for substantial morbidities and healthcare-related costs. Discovered in 2001, HMPV has been identified in all continents, with nearly universal exposure by the age of five. It accounts for 5–15% of viral respiratory infections and is among the most significant causes of pneumonia and bronchiolitis after RSV and influenza, with the highest burden in low-income regions or where healthcare access is restricted.14 HMPV has a distinct seasonal distribution with peaks during late winter and early spring in temperate zones and throughout the year in tropical zones. Infants, the elderly, immunocompromised and chronically ill patients are more likely to be severely affected and hospitalised. It is spread by respiratory droplets, direct contact, and fomites, with nosocomial outbreaks frequently occurring, as well as in long-term care facilities and schools. Co-infections with RSV (infection with another agent at the same time) and other respiratory pathogens can complicate the course of RSV disease, leading to a longer hospitalisation and higher hospitalisation rate.15 As there are no specific antiviral treatments or vaccines available, the COVID-19 pandemic emphasises the need for better surveillance, understanding of pathogenesis, and development of vaccines to lessen the burden on global health.
Recent epidemiological data (2022–2025) have reinforced hMPV as a significant contributor to acute respiratory tract infections worldwide. Global prevalence estimates suggest hMPV accounts for 5–15% of viral respiratory illnesses in children under five years and up to 10% of infections in elderly and immunocompromised populations. Regional variations are notable: higher prevalence rates are reported in South and Southeast Asia, whereas seasonal peaks are more prevalent in temperate regions of Europe and North America. Vulnerable populations, including infants, elderly adults, and patients with chronic cardiopulmonary conditions, continue to experience disproportionate morbidity and hospitalisation risks, underscoring the current disease burden.
Pathogenesis and Immune Response
HMPV is a single-stranded RNA virus that infects the respiratory tract through its F and G proteins: F proteins help enter the cell, while G proteins help attach the virus to the cell. After infection, the virus replicates in the airway epithelium, producing cytopathic effects, cellular damage, and inflammation, which result in airway obstruction and impaired gas exchange. HMPV provokes an immune response mediated by both innate and adaptive immunity, 16 with pattern recognition receptors (PRRs) (Toll-like receptors (TLRs)), which recognize viral RNA, triggering the release of interferons (IFNs) as well as pro-inflammatory cytokines (IL-6, IL-8, TNF-α) and chemokines that recruit neutrophils, macrophages, and dendritic cells to the infection site. Nevertheless, HMPV is equipped with capabilities to evade immune recognition, such as the inhibition of IFN responses and modulation of apoptosis pathways, leading to the increased viral persistence typical of immunocompromised patients. The adaptive immune response consists of CD4+ and CD8+ T cells for viral clearance, as well as the generation of neutralising antibodies, particularly against the F protein. However, no long-lasting immunity is generated following HMPV infections, so that reinfection occurs continuously throughout life, especially in children, the elderly, and immunosuppressed patients.
hMPV pathogenesis involves complex molecular and immunological interactions. The virus interferes with innate immune sensing by antagonising RIG-I/MDA5 and interferon regulatory pathways, thereby dampening antiviral responses. The dysregulated production of cytokines, such as IL-6, IL-8, and TNF-α, contributes to enhanced inflammation and airway damage. Additionally, impaired T-cell and B-cell responses may influence viral clearance and disease persistence. These mechanisms underscore the pivotal roles of immune evasion and cytokine imbalance in determining clinical outcomes, ranging from mild respiratory symptoms to severe pneumonia requiring intensive care.
In severe cases, this excessive immune response can damage the cells of the lungs, causing lung inflammation, increased mucus production, and airway remodelling, thereby worsening respiratory distress. In sum, the interplay between viral replication, immune evasion, and host immune activation likely determines disease severity and clinical outcomes.17
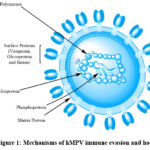 |
Figure 1: Mechanisms of hMPV immune evasion and hostClick here to view Figure |
Diagnosis of Human Metapneumovirus (HMPV)
The diagnosis of HMPV is challenging due to its clinical similarity to other respiratory viruses, such as respiratory syncytial virus, influenza, and rhinovirus. Accurate diagnosis relies on a combination of clinical assessment and laboratory testing, particularly in high-risk patients with severe symptoms.18
Table 1: Clinical severity of hMPV infections by age group and comorbidity
| Population Group | Common Comorbidities | Clinical Severity Indicators | Outcomes |
| Infants (<2 years) | Prematurity, congenital heart disease | Bronchiolitis, hypoxemia, ICU admission | High hospitalisation rates |
| Children (2–17 years) | Asthma, recurrent wheeze | Pneumonia, recurrent wheezing episodes | Moderate to severe illness |
| Adults (18–64 years) | Chronic lung disease, immunosuppression | Pneumonia, respiratory distress | Variable severity |
| Elderly (>65 years) | COPD, cardiovascular disease | Severe pneumonia, respiratory failure | Increased mortality risk |
| Immunocompromised hosts | Transplant, hematologic malignancy | Prolonged shedding, severe lower respiratory infection | High ICU/ventilation needs |
Clinical Diagnosis
HMPV is suspected in individuals with symptoms of acute respiratory tract infections (ARTIS), including fever, cough, nasal congestion, wheezing, dyspnea, and pneumonia, especially during seasonal outbreaks. However, clinical features alone are insufficient for a definitive diagnosis due to the overlapping symptoms with other viral infections.19
Laboratory Diagnosis
Molecular, antigen-based, and serological assays are the most commonly used laboratory tests for the diagnosis of Human Metapneumovirus (HMPV). The most sensitive and specific test currently in use is RT-PCR, which can detect SARS-CoV-2 ribonucleic acid (RNA) in specimens from the nasopharynx, throat, sputum, or bronchoalveolar lavage (BAL). Rapid Antigen Detection Tests (RADTs) are available for near-patient testing, providing fast results; however, their low sensitivity limits their use. 20 Viral culture, which is possible to perform in mammalian cell lines, including LLC-MK2, is rarely used in routine diagnostics due to the slow growth of viruses and low titers. Serological tests, including ELISA and immunofluorescence assays, which indicate the presence of specific IgM and IgG antibodies against HMPV, are primarily used for population-based epidemiological studies rather than for the acute diagnosis of HMPV. 21
Differential Diagnosis
Because HMPV has overlapping symptoms with RSV, influenza, adenovirus, and parainfluenza, multiplex RT-PCR panels that tested for multiple respiratory viruses simultaneously were often used in clinical settings to differentiate among pathogens. RT-PCR remains the most sensitive and commonly used diagnostic approach for HMPV, particularly in severe cases, among immunocompromised patients, and during outbreaks. Early and accurate diagnosis is crucial for infection control, effective patient management, and epidemiological surveillance. 22
Table 2: Comparison of diagnostic methods for hMPV.
| Diagnostic Method | Sensitivity | Specificity | Turnaround Time |
| RT-PCR | High | High | 4-6 hours |
| Antigen Test | Moderate | High | 30 minutes |
| Serology | Low | Moderate | Days to weeks |
| Viral Culture | High | High | 5-7 days |
Beyond conventional RT-PCR and antigen-based methods, next-generation sequencing (NGS) has emerged as a powerful tool for unbiased detection of pathogens and genomic surveillance of hMPV. Multiplex PCR platforms enable the simultaneous detection of hMPV alongside RSV, influenza, and SARS-CoV-2, thereby improving diagnostic efficiency in clinical settings. Furthermore, point-of-care (POC) molecular assays offer rapid turnaround times and decentralisation of testing, which is particularly valuable in resource-limited environments. However, high costs, technical expertise requirements, and limited global accessibility currently restrict the widespread use of NGS and multiplex PCR. At the same time, POC platforms still face challenges in terms of sensitivity compared to laboratory-based methods.
Treatment and Management of Human Metapneumovirus (HMPV)
Currently, there is no specific antiviral therapy or approved vaccine available for Human Metapneumovirus (HMPV). As a result, management is primarily supportive and symptomatic, focusing on controlling respiratory signs, preventing impairments, and managing severe episodes in high-risk patient groups, including young children, older individuals (≥65 years), and immunocompromised patients. Depending on the severity of symptoms, the approach to management is either supportive care at home for mild infections or hospitalisation with intensive care for severe cases.23
Supportive and Symptomatic Treatment
HMPV infection is typically self-limiting, and the primary management approach is supportive and symptomatic treatment. In moderate cases, they may need respiratory support, such as oxygen therapy delivered through a nasal cannula, face mask, or high-flow nasal oxygen in hypoxic patients. Non-invasive ventilatory support (NIPPV or CPAP) may help people with moderate-to-severe respiratory distress; however, critically ill patients, such as those who are immunocompromised, infants, or the elderly with underlying lung diseases, may require mechanical ventilatory support. For fever and pain management, antipyretics such as acetaminophen or ibuprofen can help lower fever-related metabolic stress in patients and improve their comfort.24 Staying hydrated and nourished is vital, particularly for infants and young children, who may sometimes require intravenous (IV) fluids in extreme conditions. Nasal suctioning and humidified air are examples of airway clearance strategies to relieve congestion and breathing difficulties. Bronchodilators, such as β2-agonists (e.g., albuterol/salbutamol), are not routinely recommended but may be beneficial in patients with asthma, bronchospasm, or reactive airway disease to address wheezing and airway constriction.25
Hospitalisation and Intensive Care Management
Most HMPV infections are mild and self-limiting; however, severe disease, particularly in infants, the elderly, and patients with underlying chronic diseases, may require hospitalisation and dedicated supportive therapy. Severe HMPV infections require hospitalisation and intensive care management, especially among high-risk groups, such as infants, elderly patients, and patients with other chronic conditions. You may be told to come to the hospital if you develop persistent hypoxia (SpO₂ < 92%) due to supplemental oxygen, severe respiratory distress with nasal flaring and intercostal retractions, tachypnea, or respiratory failure that requires ventilatory support.26 Other reasons for admission may include complications such as pneumonia, secondary bacterial infections, sepsis, dehydration, or failure to maintain adequate oral intake. Patients needing intensive care unit (ICU) admission usually consist of patients with worsening respiratory failure who need mechanical ventilation, with severe hypoxia, multi-organ dysfunction, or unstable hemodynamics. As discussed earlier, individuals taking precautions, such as those with immunocompromising conditions and a history of recurrent infections, may require close monitoring and vigilance.27
Experimental and Investigational Therapies
Experimental treatment options and investigational drugs are being examined for potential therapeutic benefit because no approved antiviral therapy for HMPV is available. As there are currently no approved antiviral therapies for HMPV, experimental and investigational therapies have been studied. Antiviral drugs like ribavirin have shown in vitro activity; however, they have also not been clinically efficacious and are toxic, making them available only for use in extreme cases, such as in immunocompromised individuals.28 Even so, a similar efficacy has yet to be demonstrated in HMPV infections in the laboratory setting with favipiravir and remdesivir, which were initially developed to treat influenza and COVID-19, respectively. Monoclonal antibodies (mAbs) directed against the HMPV fusion (F) protein are being developed and may reduce viral replication, similar to palivizumab for RSV, although clinical trials have yet to be conducted. Intravenous immunoglobulin (IVIG) has been used in some immunocompromised people, but it is not a standard treatment. Corticosteroids, such as dexamethasone and prednisone, have been used for their ability to inhibit inflammation and airway hyperresponsiveness; however, they may also impair viral clearance and prolong viral infection.29
Prevention and Infection Control
Since no HMPV vaccine exists, prevention strategies emphasise infection control measures, particularly in hospitals and long-term care facilities. Human Metapneumovirus (HMPV) has no licensed vaccine, and primary prevention and infection control measures focus on reducing transmission rates, particularly in healthcare and community settings.30 Despite the severity of the aforementioned illnesses, HMPV infections are largely preventable through basic infection control practices in healthcare environments, which include rigorous hand hygiene with soap and alcohol, as well as respiratory precautions, droplet and contact isolation of infected patients, and environmental disinfection of high-touch surfaces, given that HMPV can persist on surfaces for hours. Community precautions include maintaining distance from vulnerable populations, coughing into a tissue or elbow, and disposing of used tissues properly. Active research is ongoing on live-attenuated, protein subunit, and vector-based vaccines targeting the HMPV fusion (F) protein as a means to induce long-term immunity.31
Herbs Used in the Management of HMPV
Many medicinal herbs have traditionally been utilised to enhance respiratory function and boost the immune system, possibly aiding in the management of Human metapneumovirus (HMPV). Elderberry (Sambucus nigra) contains high levels of flavonoids and has antiviral properties that may alleviate the severity and duration of respiratory infections. Echinacea (Echinacea purpurea), known for boosting the immune system, might bolster the body’s defence against viral attacks. Known to soothe the throat, liquorice root (Glycyrrhiza glabra) also possesses antiviral and anti-inflammatory properties that can help reduce mucus buildup and lung irritation. Allium sativum (garlic), the active compound of which is allicin, has strong antiviral, antibacterial, and immune-boosting effects, and may be useful for preventing secondary infections.32 Ginger (Zingiber officinale) possesses anti-inflammatory and antiviral properties, which can help alleviate symptoms such as sore throat, congestion, and nausea. Turmeric (Curcuma longa), which contains curcumin, is a potent anti-inflammatory and antioxidant that may help alleviate lung inflammation associated with viral infections. It is used in traditional medicine to promote immune and antiviral activity for respiratory infections (Andrographis paniculata). Oregano (Origanum vulgare) also possesses bioactive compounds such as carvacrol and thymol, which would also be beneficial as those components have been shown to exhibit antimicrobial and immune-support properties, promoting the clearance of respiratory congestion.33 Astragalus (Astragalus membranaceus), a mainstay of traditional Chinese medicine, boosts the immune system and may protect against respiratory infections. Mullein (Verbascum thapsus): Helps soothe irritated respiratory tissues, calm a cough, and promote lung health. These herbs can be taken in various forms, including teas, tinctures, capsules, and syrups. Nonetheless, you must speak with a healthcare professional before taking any herbal treatment, especially if you are using it for your child, you are pregnant, or suffer from any pre-existing medical conditions, as certain herbs can clash with prescription drugs, or result in negative side effects when consumed in excess.34
Therapeutic updates with clinical trial status
Current therapeutic options for hMPV remain largely supportive, but promising candidates are under investigation. Fusion inhibitors targeting the hMPV F protein have shown efficacy in vitro and in animal models. Polymerase inhibitors are being evaluated for broad-spectrum anti-Pneumoviridae activity. Importantly, monoclonal antibodies (mAbs) directed against conserved epitopes of the F protein are advancing through preclinical and early clinical evaluation, with some demonstrating potent neutralizing activity. Ongoing Phase I/II clinical trials are assessing safety, pharmacokinetics, and protective efficacy, though high costs and potential viral escape mutations remain limitations.
 |
Table 3: Additional herbal plants, their active metabolites, parts used, types of extracts, modes of action (MOA) relevant to Human Metapneumovirus (HMPV)Click here to view Figure |
Vaccine Development and Future Perspectives
Human Metapneumovirus (HMPV) continues to be a common cause of acute respiratory tract infections (ARTIS) worldwide and has a special impact on infants, elderly individuals, and immunocompromised patients. HMPV has received relatively little attention, and no licensed vaccine is presently available for HMPV, despite posing a significant public health burden, primarily due to difficulties in evoking durable immune responses and the complexities of the immune responses to the virus. But there are vaccine candidates in development, with encouraging findings in preclinical and early clinical trials. Advancements in vaccine technology, novel antiviral therapies, and enhanced surveillance approaches will inform the future of HMPV prevention.52
Challenges in HMPV Vaccine Development
Developing an effective HMPV vaccine is challenging due to several factors. Natural infections provide only short-lived immunity, leading to recurrent infections throughout life. The virus employs immune evasion mechanisms, such as suppressing type I interferon responses and modulating apoptosis pathways, which limit effective immune activation. Additionally, high genetic variability, with two major lineages (A and B) and four subtypes (A1, A2, B1, B2), complicates vaccine design. Age-dependent immune responses further hinder vaccine development, as infants, the primary target group, have immature immune systems that struggle to generate strong and lasting immunity. Moreover, there is a risk of vaccine-associated enhanced respiratory disease (VAERD), similar to RSV, where inactivated whole-virus vaccines could exacerbate disease severity upon subsequent natural infection.53-54
Current Vaccine Strategies Under Development
The vaccine landscape for hMPV is rapidly evolving, with multiple innovative platforms under development. mRNA vaccines encoding stabilised hMPV fusion proteins have shown strong immunogenicity in preclinical models. Viral vector-based approaches (e.g., adenoviral vectors) are being explored for their ability to elicit robust cellular responses. Additionally, nanoparticle vaccines that mimic the native conformation of hMPV surface glycoproteins have demonstrated promise in eliciting broadly neutralising antibodies. Despite these advances, challenges persist in achieving durable immunity, cross-protection against viral genotypes, and effectiveness in high-risk groups.
Several approaches are being explored for HMPV vaccination, including live-attenuated, subunit, viral vector, and mRNA-based vaccines.
Live-Attenuated Vaccines
Live-attenuated HMPV vaccines are engineered by introducing mutations that reduce viral replication but maintain their immunogenic capacity. One common approach to creating these genetically engineered strains has been to alter or remove viral genes involved in immune evasion, such as SH (small hydrophobic) and G (glycoprotein). Intranasal administration of these vaccines is associated with robust mucosal immunity and sustained protection, as demonstrated in preclinical studies and animals.55 But safety remains a concern, especially for some types of immunocompromised individuals, since even an attenuated virus could cause disease.
Protein Subunit Vaccines
Protein subunit vaccines for HMPV promote an immune response using recombinant viral proteins, specifically the F (fusion) protein, without exposing the immune system to the live virus. The F protein in its stabilised prefusion conformation has emerged as a prime target for the generation of potent neutralising antibodies.56 Subunit vaccines are a safer option than live-attenuated vaccines but may need adjuvants to improve immunogenicity and booster immunisation for long-term protective immunity.57
Viral Vector-Based Vaccines
Viral vector-based vaccines utilise engineered viruses, such as adenoviruses or measles virus vectors, to transport HMPV antigens to the immune system. This does allow for the generation of humoral and cellular immunity, resulting in strong protective responses. It has shown promising effectiveness in pre-clinical studies, but must undergo further safety and efficacy trials.58
mRNA-Based Vaccines
The concept of mRNA-based vaccines for HMPV originated from the success of mRNA vaccines for COVID-19, where synthetic mRNA composed of translated viral proteins promotes an immune response. These vaccines have a short development time, excellent immunogenicity, and can be rapidly modified to respond to viral variants. Although preclinical studies are underway, additional research is necessary to confirm human safety, efficacy, and durability of protection.59
Future Research Priorities
To accelerate progress in hMPV prevention and treatment, several areas require urgent attention. First, the development of reliable animal models that accurately mimic human disease is essential for preclinical testing. Second, exploring pan-Pneumoviridae vaccine approaches could yield cross-protective immunity against both hMPV and RSV. Third, efforts must focus on improving diagnostic and treatment accessibility in low- and middle-income countries (LMICs), where the burden of viral respiratory infections is highest but resources are limited. Collectively, addressing these priorities will strengthen translational research and global preparedness for hMPV-related disease.
Conclusion
HMPV was first isolated in 2001 and has since been globally identified as a respiratory pathogen with a significant disease burden, mimicking RSV and influenza infection. Its genomic architecture is composed of a negative-sense single-stranded RNA genome that encodes 9 prominent proteins essential for viral replication, immune evasion, and infection of host cells. HMPV is endemic worldwide, with seasonal epidemics typically reported in late winter to early spring. The virus is transmitted by direct contact, respiratory droplets, or contact with contaminated surfaces, and infects people of all ages but disproportionately harms young children, older people, and those with preexisting respiratory conditions. HMPV appears clinically similar to other respiratory viruses, causing mild cold-like syndromes in the general population, but can also cause severe lower respiratory tract infections, including bronchiolitis and pneumonia, among vulnerable groups. The pathogenesis of HMPV is associated with the ability of the virus to downregulate host immune responses, including interferon production and adaptive immunity, which is linked to persistent viral replication and reinfections. Diagnosis is based mostly on molecular methods, including RT-PCR, which have high sensitivity and high specificity, as well as immunoassays and viral culture methods in research settings.
At present, HMPV therapy is limited to supportive care as no specific antiviral treatment or vaccine is currently available. The severe cases require oxygen therapy, hydration, bronchodilators and, if necessary, intensive care support, while ribavirin, monoclonal antibodies and immunomodulators are under consideration as experimental agents. With the aforementioned challenges, numerous promising vaccine candidates are currently in development, including live-attenuated, protein subunit, viral vector, and mRNA-based approaches. Increased experience with monoclonal antibody therapies, combination vaccines, and universal respiratory virus vaccines gives hope for future preventive strategies. Further research and increased global surveillance, as well as better public health initiatives, are needed to reduce the impact of HMPV infections. Strengthening infection control measures, increasing diagnostic approaches, and developing therapeutic strategies will be crucial in mitigating the morbidity and mortality of HMPV. As research into vaccines, antiviral drugs, and epidemiology progresses, there is hope that vaccines and treatments that are effective at decreasing morbidity from HMPV-related respiratory disease will be achieved shortly.
Acknowledgement
The authors are thankful to the Management of IFTM University, Moradabad.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author contributions
- Alankar Shrivastav: Manuscript Preparation
- Monika Singh: Designing the structures and Tables
- Tribhuwan Kumar Vashthista: Literature Survey
- Rajat Saxena: Data analysis and other things
- Arun Kumar Mishra: helping in data collection and management
- Navneet Verma: helping as a guide during paper preparation
References
- Jefferson T, Dooley L, Ferroni E, Al-Ansary L. A, van Driel M. L, Bawazeer G. A, et al. Physical interventions to interrupt or reduce the spread of respiratory viruses. Cochrane Database Syst. Rev. 2023;1(1): CD006207.
CrossRef - Cong S, Wang C, Wei T, Xie Z, Huang Y, Tan J, et al. Human metapneumovirus in hospitalized children with acute respiratory tract infections in Beijing, China. Infect. Genet. Evol. 2022;106: 105386.
CrossRef - Wang C, Wei T, Ma F, Wang H, Guo J, Chen A, et al. Epidemiology and genotypic diversity of human metapneumovirus in paediatric patients with acute respiratory infection in Beijing, China. Virol. J. 2021;18(1): 40.
CrossRef - Qian W, Huang J, Wang T, He X, Xu G, Li Y. Visual detection of human metapneumovirus using CRISPR-Cas12a diagnostics. Virus Res. 2021;305: 198568.
CrossRef - Zhao J, Ragupathy V, Liu J, Wang X, Vemula S. V, El Mubarak H. S, et al. Nanomicroarray and multiplex next-generation sequencing for simultaneous identification and characterization of influenza viruses. Emerg. Infect. Dis. 2015; 21(3): 400–408.
CrossRef - Luo K. M, Liang Y. W, Zhang S. S. Detection and analysis of pandemic virus in children with acute respiratory tract infection in Yangshan from 2012 to 2014. Chin. J. Health Lab. Tec. 2016;26: 3004–3006.
- Li J, Wang Z, Gonzalez R, Xiao Y, Zhou H, Zhang J, et al. Prevalence of human metapneumovirus in adults with acute respiratory tract infection in Beijing, China. J. Infect. 2012;64(1): 96–103.
CrossRef - Jin Y, Zhang R. F, Xie Z. P, Yan K. L, Gao H. C, Song J. R, et al. Newly identified respiratory viruses associated with acute lower respiratory tract infections in children in Lanzhou, China, from 2006 to 2009. Clin. Microbiol. Infect. 2012;18(1): 74–80.
CrossRef - Nagasawa M, Udagawa T, Kato T, Tanaka I, Yamamoto R, Sakaguchi H, Sekikawa Y. Observational study on the clinical reality of community-acquired respiratory virus infections in adults and older individuals. Pathogens. 2024; 13: 983.
CrossRef - Shafagati N, Williams J. Human metapneumovirus—What we know now. F1000. Research. 2018;7: 135.
CrossRef - Levi M. Platelets in critical illness. Semin. Thromb. Hemost. 2016;42: 252–257.
CrossRef - Du Y, Li W, Guo Y, Li L, Chen Q, He L, Shang S. Epidemiology and genetic characterization of human metapneumovirus in pediatric patients from Hangzhou, China. J. Med. Virol. 2022;94(11): 5401–5408.
CrossRef - Divarathna M. V. M, Rafeek R. A. M, Noordeen F. A review on epidemiology and impact of human metapneumovirus infections in children using TIAB search strategy on PubMed and PubMed Central articles. Rev. Med. Virol. 2020;30(1): e2090.
CrossRef - Ogonczyk Makowska D, Hamelin M. È, Boivin G. Engineering of live chimeric vaccines against human metapneumovirus. Pathogens. 2020;9(2): 135.
CrossRef - Tang X, Dai G, Wang T, Sun H, Jiang W, Chen Z, et al. Comparison of the clinical features of human bocavirus and metapneumovirus lower respiratory tract infections in hospitalized children in Suzhou, China. Front. Pediatr. 2023;10: 1074484.
CrossRef - Van den Hoogen B. G, van Doornum G. J, Fockens J. C, Cornelissen J. J, Beyer W. E, de Groot R, et al. Prevalence and clinical symptoms of human metapneumovirus infection in hospitalized patients. J. Infect. Dis. 2003;188: 1571–1577.
CrossRef - Brooks W. A, Erdman D, Terebuh P, et al. Human metapneumovirus infection among children, Bangladesh. Emerg. Infect. Dis. 2007;13(10): 1611–1613.
CrossRef - Panda S, Mohakud N. K, Pena L, Kumar S. Human metapneumovirus: review of an important respiratory pathogen. Int. J. Infect. Dis. 2014;25: 45–52.
CrossRef - Edwards K. M, Zhu Y, Griffin M. R, et al.; New Vaccine Surveillance Network. Burden of human metapneumovirus infection in young children. N. Engl. J. Med. 2013;368(7): 633–643.
CrossRef - Stockton J, Stephenson I, Fleming D, Zambon M. Human metapneumovirus as a cause of community-acquired respiratory illness. Emerg. Infect. Dis. 2002;8(9): 897–901.
CrossRef - Jin X, Ren J, Li R, Gao Y, Zhang H, et al. Global burden of upper respiratory infections in 204 countries and territories, from 1990 to 2019. eClinicalMedicine. 2021;37: 100986.
CrossRef - Kahn J. S. Epidemiology of human metapneumovirus. Clin. Microbiol. Rev. 2006;19: 546–557.
CrossRef - Walsh E. E, Peterson D. R, Falsey A. R. Human metapneumovirus infections in adults: another piece of the puzzle. Arch. Intern. Med. 2008;168: 2489–2496.
CrossRef - Bruno R, Marsico S, Minini C, Apostoli P, Fiorentini S, et al. Human metapneumovirus infection in a cohort of young asymptomatic subjects. J. Clin. Virol. 2009;32: 297–301.
- Pitoiset C, Darniot M, Huet F, Aho S. L, Pothier P, et al. Human metapneumovirus genotypes and severity of disease in young children (n = 100) during a 7-year study in Dijon hospital, France. J. Med. Virol. 2010;82: 1782–1789.
CrossRef - Ye H, Zhang S, Zhang K, Li Y, Chen D, Tan Y, et al. Epidemiology, genetic characteristics, and association with meteorological factors of human metapneumovirus infection in children in southern China: a 10-year retrospective study. Int. J. Infect. Dis. 2023;137: 40–47.
CrossRef - Yi L, Zou L, Peng J, Yu J, Song Y, Liang L, et al. Epidemiology, evolution and transmission of human metapneumovirus in Guangzhou, China, 2013–2017. Sci. Rep. 2019;9(1): 14022.
CrossRef - Tulloch R. L, Kok J, Carter I, Dwyer D. E, Eden J. S. An amplicon-based approach for the whole-genome sequencing of human metapneumovirus. Viruses. 2021;13(3): 499.
CrossRef - Ishiguro N, Akutsu Y, Azuma K, Yonekawa M, Sato D, Ishizaka A, et al. Evaluation of a novel immunochromatographic assay using monoclonal antibodies against the matrix protein of human metapneumovirus. Clin. Lab. 2021;67(10): 1.
CrossRef - Brooks D. G, McGavern D. B, Oldstone M. B. A. Reprogramming of antiviral T cell prevents inactivation and restores T cell activity during persistent viral infection. J. Clin. Invest. 2006;116(6): 1675–1685.
CrossRef - Wolfrum N, Greber U. F. Adenovirus signalling in entry. Cell Microbiol. 2013;15(1): 53–62.
CrossRef - Broadbent A. J, Boonnak K, Subbarao K. Respiratory virus vaccines. Mucosal Immunol. 2015.
CrossRef - Ghebremedhin B. Human adenovirus: viral pathogen with increasing importance. Eur. J. Microbiol. Immunol. 2014;4(1): 26–33.
CrossRef - Al-Halifa S, Gauthier L, Arpin D, Bourgault S, Archambault D. Nanoparticle-based vaccines against respiratory viruses. Front. Immunol. 2019;10: 1–11.
CrossRef - Huang L, Yang M, Yuan Y, Li X, Kuang E. Niclosamide inhibits lytic replication of Epstein-Barr virus by disrupting mTOR activation. Antiviral Res. 2017;138: 68–78.
CrossRef - Antonelli M, Donelli D, Maggini V, Firenzuoli F. Phytotherapic compounds against coronaviruses: Possible streams for future research. Phytother. Res. 2020;34(7): 1469.
CrossRef - Quintão N. L, Pastor M. V, Antonialli C. D, da Silva G. F, Rocha L. W, Berté T. E, de Souza M. M, Meyre-Silva C, Lucinda-Silva R. M, Bresolin T. M, Cechinel Filho V. Aleurites moluccanus and its main active constituent, the flavonoid 2″-O-rhamnosylswertisin, in experimental model of rheumatoid arthritis. J. Ethnopharmacol. 2019;235: 248–254.
CrossRef - Pichon M, Picard C, Simon B, Gaymard A, Renard C, Massenavette B, et al. Clinical management and viral genomic diversity analysis of a child’s influenza A (H1N1) pdm09 infection in the context of a severe combined immunodeficiency. Antiviral Res. 2018;160: 1–9.
CrossRef - Huang J, Ma K, Zhang J, Zhou J, Yi J, Qi W, Liao M. Pathogenicity and transmission of novel highly pathogenic H7N2 variants originating from H7N9 avian influenza viruses in chickens. Virology. 2024;597: 110121.
CrossRef - Toms L. M, Hearn L, Mueller J. F, Harden F. A. Assessing infant exposure to persistent organic pollutants via dietary intake in Australia. Food Chem. Toxicol. 2016;87: 166–171.
CrossRef - Lu M, Li G, Yang Y, Yu Y. A review on in-vitro oral bioaccessibility of organic pollutants and its application in human exposure assessment. Sci. Total Environ. 2021;752: 142001.
CrossRef - Sun D. Y, Cheng C, Moschke K, Huang J, Fang W. S. Extensive structure modification on luteolin-cinnamic acid conjugates leading to BACE1 inhibitors with optimal pharmacological properties. Mol. 2019;25(1): 102.
CrossRef - Li J, Zhao Y, Dai Y, Zhao J. Identification of γ-Fagarine as a novel antiviral agent against respiratory virus (hMPV) infection. Virus Res. 2023;336: 199223.
CrossRef - Pal D, Bareth K. Respiratory viral infections and the role of medicinal plants in prevention and treatment. In: Anti-Viral Metabolites from Medicinal Plants. Cham: Springer Int. Publ. 2023:397–427.
CrossRef - Pal D, Lal P. Plants showing anti-viral activity with emphasis on secondary metabolites and biological screening. In: Anti-Viral Metabolites from Medicinal Plants. Plants. 2023:29–95.
CrossRef - Taran T, Bharadwaj A, Majumdar T. A review on uses of ethno-medicinal plants for treatment of whooping cough: A highly contagious respiratory tract infection. Acta Biol. Slov. 2025;68(2).
CrossRef - Zhao T, Sun Q, Marques M, Witcher M. Anticancer properties of Phyllanthus emblica (Indian gooseberry). Oxid. Med. Cell. Longev. 2015;2015(1): 950890.
CrossRef - Dubey A, Kumar M, Tufail A, Dwivedi V. D. Unlocking antiviral potentials of traditional plants: A multi-method computational study against human metapneumovirus (hMPV). bioRxiv. 2025:2025–01.
CrossRef - Alrashedi H. S, Al-Ataie S. S, Banoon S. R, Fayed M. I. Potential role of medicinal plants for the treatment of respiratory viruses: A review. Egypt. J. Chem. 2021;64(12): 7495–7508.
- Padakanti A. P, Arjun S, Chella N. Role of herbal formulations in antiviral therapy—an overview. Adv. Antivir. Res. 2024;3: 211–253.
CrossRef - Duijker G, Bertsias A, Symvoulakis E. K, Moschandreas J, Malliaraki N, et al. Reporting effectiveness of an extract of three traditional Cretan herbs on upper respiratory tract infection: Results from a double-blind randomized controlled trial. J. Ethnopharmacol. 2015;163: 157–166.
CrossRef - Iyer S, Yadav R, Agarwal S, Tripathi S, Agarwal R. Bioengineering strategies for developing vaccines against respiratory viral diseases. Clin. Microbiol. Rev. 2022;35(1): 00123–221.
CrossRef - Bhat R, Almajhdi F. N. Induction of immune responses and immune evasion by human bocavirus. Int. Arch. Allergy Immunol. 2021;182(8): 728–735.
CrossRef - Vassal-Stermann E, Effantin G, Zubieta C, Burmeister W, Iseni F, Wang H, et al. CryoEM structure of adenovirus type 3 fibre with desmoglein 2 shows an unusual mode of receptor engagement. Nat. Commun. 2019;10(1): 1181.
CrossRef - Allander T, Tammi M. T, Eriksson M, Bjerkner A, Tiveljung-Lindell A, Andersson B. Cloning of a human parvovirus by molecular screening of respiratory tract samples. Proc. Natl. Acad. Sci. USA, 2005;102(36): 12891–12896.
CrossRef - Rikhotso M. C, Kabue J. P, Ledwaba S. E, Traoré A. N, Potgieter N. Prevalence of human bocavirus in Africa and other developing countries between 2005 and 2016: A potential emerging viral pathogen for diarrhea. J. Trop. Med. 2018;2018: 7875482.
CrossRef - Thammawat S, Sadlon T. A, Hallsworth P. G, Gordon D. L. Role of cellular glycosaminoglycans and charged regions of viral G protein in human metapneumovirus infection. J. Virol. 2008;82: 11767–11774.
CrossRef - Ali S. A, Williams J. V, Chen Q, Faouri S, Shehabi A, et al. Human metapneumovirus in hospitalized children in Amman, Jordan. J. Med. Virol. 2010;82: 1012–1016.
CrossRef - Piedimonte G, Perez M. K. Respiratory syncytial virus infection and bronchiolitis. Pediatr. Rev. 2014;35: 519–530.
CrossRef







