Manuscript accepted on :12-09-2025
Published online on: 10-02-2026
Plagiarism Check: Yes
Reviewed by: Dr. Hina Imran
Second Review by: Dr. Samara Sameer Yonus
Final Approval by: Dr. Prabhishek Singh
Abdumalik Nigmatovich Aripov1 , Lola Lazizovna Akhunzhanova1
, Lola Lazizovna Akhunzhanova1 , Orifzhon Abdumalikovich Aripov1
, Orifzhon Abdumalikovich Aripov1 and Tolmas Tolibovich Khamroev1,2,3*
and Tolmas Tolibovich Khamroev1,2,3*
1Department of Clinical Laboratory diagnostics of Republican Specialized Scientific and Practical Medical Center of Pediatrics of the Ministry of Health of the Republic of Uzbekistan, Tashkent City, Uzbekistan
2Department of pharmacology and toxicology of The Institute of Chemistry of Plant Substances named after Academician S.Yu. Yunusov of the Academy of Sciences of the Republic of Uzbekistan, Tashkent City, Uzbekistan
3Department of pharmacology of Tashkent state medical university, Tashkent City, Uzbekistan
Corresponding Author E-mail: tolmastht@gmail.com
Abstract
Although only a few groups of drugs with hepatoprotective activity are recognized, the introduction of drugs with high liver protective activity, mainly based on natural compounds or plants, has become an urgent problem especially, over the last twenty years. A striking example of this is the fact that the hepatoprotective activity of existing phytotherapeutic drugs is not as high as that of synthetic drugs. However, secondary plant metabolites, including flavonoids, flavones, flavonols, and other naturally occurring biologically active compounds, are becoming a promising therapeutic alternative to traditional medicines in modern medicine. The priority purpose of this study is to introduce a new highly hepatoprotective and at the same time biologically active antidepressant of a new composition based on a combination of phospholipid, lycopene, glycerisic acid and yantacin obtained from plants. All of the research was done on rats, and the administration of heliothrin, a chemical that has a hepatotoxic effect, into the abdominal cavity in accordance with a rigorous plan resulted in significant chronic liver damage. Its hepatoprotector and antidepressant properties were compared to those of the well-known medication Phosphogliv and fluoxetine, which are both commonly used in medicine. The study's focus was a 1:1 mixture of heparin and yanatacin. As a result, the pharmacological composition that is being presented is completely safe, and research has shown that its constituents exhibit a markedly elevated activity not only in relation to the well-known medication phosphogliv, but also in relation to the ecdysteroid form of yantacin and hepalipin. Consequently, the pharmacological composition offered is entirely safe, and studies have demonstrated that its components have significantly higher activity not only when compared to the well-known drug phosphogliv, but also when compared to the ecdystenic form of yantacin and hepalipin.
Keywords
Fluoxetine; L. Steru; Noneecdystene form of hepalipin; Phosphogliv; R.Porsol; Yantacin
| Copy the following to cite this article: Aripov A. N, khunzhanova L. L, Aripov O. A, Khamroev T. T. Determination of the Simultaneous Hepatoprotective and Antidepressant Activity of the Composition Hepalipin and Yantacin. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Aripov A. N, khunzhanova L. L, Aripov O. A, Khamroev T. T. Determination of the Simultaneous Hepatoprotective and Antidepressant Activity of the Composition Hepalipin and Yantacin. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/3OfzsfW |
Introduction
The increase in the incidence of chronic hepatitis of various etiologies, mainly in men and people of working age, is the cause of not only medical, but also socio-economic burden. Among chronic liver diseases, the main share is accounted for by pathologies of viral etiology, with chronic hepatitis B and C. According to the World Health Organization, about 1.5 million new hepatitis C virus-related infections occur annually, and 3.2 million adolescents and children become infected with chronic hepatitis C infection. Currently, worldwide, chronic hepatitis C virus infection affects 58 million people, while hepatitis C virus is estimated to affect 350 million people.1, 2, 3 In 2015, 1.3 million deaths related to chronic hepatitis B virus was reported worldwide. and C, up from 2 million in 2019, accounting for almost 95.0 percent of all virus-related deaths. These pathologies pose not only a risk of death, but also damage to other organs, in addition to liver damage, in particular, patients with cryoglobulinemia, lymphoproliferative diseases, kidney diseases, insulin resistance, rheumatoid arthritis-polyarthritis, which negatively affects the quality of life and work.4, 5, 6 Cirrhosis of the liver and the development of hepatocellular Carcinomas resulting from chronic viral hepatitis or chronic liver diseases are the leading causes of death worldwide.7 Nowadays, when the frequency of metabolic risk factors for liver cirrhosis and hepatocellular carcinoma is increasing, chronic hepatitis B and C remain the most important risk factors for progressive liver diseases worldwide. Dangerous complications causing death and disability in the liver, including two-thirds of the global burden of cirrhosis of the liver alone, may be associated with hepatitis B and C viral infections, and 1 million deaths have been reported in patients, mainly due to grave complications of chronic liver diseases.4, 5, 8, 9
The goal of modern therapeutic measures for viral hepatitis is the formation of immunity in the form of a virological response in the blood serum six months after completion of the course of treatment or the absence of detection of viral antigens in pathogenic amounts. It is worth noting that over the past ten years, significant progress has been consistently made in the diagnosis and treatment of viral hepatitis in the early stages, which has changed the way viral hepatitis is treated. A decrease in frequency of occurrence associated with viral hepatitis C, combined with broader detection and treatment of the disease, may lead to the complete elimination of these viruses from the body in the future. Because no other viral hepatitis, such as chronic viral hepatitis C, has undergone significant changes in pharmacological treatment methods. Viral hepatitis B, on the other hand, is the main factor in virological treatment and, as a result, its eradication in combination with treatment, which includes a clear definition of important aspects or points of treatment, effective biomarkers and several measures aimed at preventing the mechanisms of viral replication. Achievements in the fight against hepatitis D virus infection are also growing in parallel, and, in contrast, additional measures are needed in the field of epidemiology and pharmacotherapy.11-13
As noted above, even in China, viral hepatitis is expected to be completely eradicated by 2030 as a result of the improvement of highly effective medicines and therapeutic measures that demonstrate a long-term negative result in blood serum even after stopping taking drugs against viral hepatitis. The possibility of timely detection of chronic viral hepatitis to date, significant progress has been made in the timely detection of chronic therapeutic measures, however, in the treatment of chronic liver damage, eliminating the etiological factor alone is not enough. This, in turn, requires a generalization of the latest advances in the treatment of antiviral drugs, which can significantly reduce the burden of the disease and the complications associated with the liver, as well as improve impaired liver function by strengthening the membranes of hepatocytes. As a result, however, all types of viral hepatitis can be recognized as a disease with definitive treatments due to the availability of safe and effective treatments for each patient. Thus, achieving high efficacy in the treatment of all cases of viral hepatitis, combined with timely identification of high-risk groups, screening, and the development of optimal vaccination methods, will also be important to eliminate liver changes caused by these infections.14-19
To date, there is no generally accepted classification of drugs with hepatoprotective activity, and they are conditionally divided into the following groups.20, 21
Drugs of animal origin. These are cyrepars, gepadifs, etc.
Contains natural and semi-synthetic flavanoids, preservative plant hepatoprotectors; milk thistle flavonoids. The active ingredient is silymarin. These are gepaben, sibectan, Silimar, Karsil and others.
Flavonoids of other medicinal plants. These are Liv-52 and others.
Essential phospholipids. These are Essentiale Forte, Essliver Forte, Phosphoglive and others.
Other hepatoprotectors are ursodeoxycholic acid, nonsteroidal anabolics and others.
Synthetic drugs;
Drugs belonging to different groups, in particular, amino acids and their derivatives. Ademethionine (Heptral), Gepamers, etc.
At the same time, although only a few groups of drugs with hepatoprotective activity have been recognized, the search for a highly active drug that protects the liver, mainly based on natural compounds or plants, became a serious problem more than a decade ago. Due to the fact that the hepatoprotective activity of existing herbal medicines is not as high as that of synthetic ones, secondary plant metabolites, including flavonoids, flavones, flavonols and other natural biologically active compounds, are becoming a promising therapeutic alternative to traditional medicines in modern medicine. These drugs are very effective and have minimal side effects, with the exception of plant-based compounds or secondary metabolites, i.e. naturally occurring phytochemicals present in sufficient quantities.23, 24
An important task of drug research in practice is to maximize the therapeutic potential of drugs, manage their clinical use and maximize their therapeutic value. Thus, the analysis of the effectiveness of pharmacotherapy in the treatment or prevention of liver diseases, as well as all diseases, establishes the basic prerequisite for further scientific research.
In this regard, the need for further work on the development of treatment methods capable of eliminating chronic liver damage and restoring impaired liver function remains urgent.19, 21, 22, 23, 24 It should also be noted that there are also factors that aggravate the development of malignant liver damage or chronic processes that reduce the effectiveness of treatment, and mental disorders, in particular depressive states, are directly considered as one of the main of these factors.
Depression is more common in cases where chronic liver damage occurs or grave complications, and it has been described that it negatively affects clinical outcomes, while a number of authors note that the causes of this relationship are complex and depend on many factors. Clinically significant anxiety, emotional arousal, anxiety states and depression are present in people with chronic liver damage as a result of these diseases or in the form of concomitant diseases that significantly reduce their quality of life. Because a long course of chronic liver damage can lead to a weakening of the patient’s body, and concomitant mental disorders can worsen the disease. With serious fatal complications of chronic liver damage of particularly grave liver pathologies, hepatic encephalopathy, as well as depression, are common, and morbidity and mortality significantly affect the number. Antidepressants are effective in chronic liver diseases, their complications, and even after orthotopic liver transplantation, but lower doses or reduced frequency of administration may be required to minimize side effects such as exacerbation of hepatic encephalopathy. In patients with diseases of the gastrointestinal system and liver biliary tract, it becomes necessary to choose a psychopharmacological measure as a psychotropic drug and individually depending on the degree of the underlying illness. When patients have liver disorders, it is preferable to use psychotropic drugs that prevent liver metabolism or have minimal effect on it. This, in turn, imposes on the attending physicians the need to be careful when prescribing any psychotropic drugs to any patient with a particularly serious liver disease. Thus, having a natural basis, simultaneous low-toxicity hepatoprotective and antidepressant drugs make it possible to avoid drug interactions and effects in each patient, and drugs that do not cause hepatotoxicity contribute to the successful pharmacological treatment of mental disorders in people with chronic and severe liver diseases. In this regard, it became necessary to study the pharmaco-toxicological properties of yantacin, a low-toxic preservative of proanthacyanidins, whose antidepressant activity was recognized by a number of authors.34-43 It is the combination of previously recognized hepatoprotective and antioxidant activity that can enable attending physicians to most effectively manage the pathological process in the liver and depression in these patients, justifying simultaneous hepatoprotective and antidepressant phallism.26, 33
Classical hepatoprotective and psychotropic antidepressants are not produced in Uzbekistan, that is, they are imported. Therefore, the development of new natural hepatoprotectors and drugs with high antidepressant activity is an urgent task. Based on the above-mentioned groups of drugs with hepatoprotective activity, the Republican Specialized Pediatric Scientific and Practical Medical Center (RSPSPMC) conducts comprehensive scientific studies of plant-based compounds with hepatoprotective activity belonging to various chemical groups. In this regard, researchers at the Laboratory of Lipid Chemistry at the Institute of Chemistry of Plant Substances of the Republic of Uzbekistan (ICPS RUz) conducted experimental studies of acute toxicity, hepatoprotective activity, and depression of combinations of yantacin based on the Alchaga pseudoalchaga hepalipin plant and its extension-free samples.25-33 As a result of this research, it will be possible to put into practice a remedy based on medicinal plants, which will allow it to be used to relieve chronic liver damage and depressive states related to the chemical and pharmaceutical industries, that is, lipid and falavonoid chemistry, pharmaceuticals and medicine, in particular, hepatology and pharmacology.
Experimental
Materials and Methods
Outbred white male rats weighing a certain amount of 170±10 grams and white mice with a body weight of 20±2 grams were used as study objects. The care and food conditions for each of the animals under study were the same.48
Chemical properties of the pharmacological composition obtained on the basis of plants.
It is worth noting that the components of the composition, the simultaneous hepatoprotective and antidepressant activity of which was studied for the first time, were isolated by researchers from the Laboratory of Lipid and Flavonoid Chemistry at the ICPS RUz.. Due to the fact that the chemical structure and properties of each substance included in the composition (Fig.1) are described in detail in previously obtained certificates of invention and published scientific publications, in our application for the invention we have focused and recognized only the activity of this composition. For a drug called hepalipin, which has hepatoprotective activity, obtained on the basis of the components of this composition, the state unitary enterprise “State Center for Expertise and Standardization of Medicines, Medical Devices and Medical Equipment” under the Ministry of Health of the Republic of Uzbekistan, together with “Radix” LLC, received permission to conduct clinical trials. However, a combination consisting of a non-ecdysterone form of hepalipin and yantacin was studied for the first time for concomitant hepatoprotective and antidepressant activity. The ecdystene contained in hepalipin is an agent with adaptogenic activity, and it is unlikely to affect the hormone testosterone in men, and this effect can only have a positive effect on adult athletes, causing hormonal imbalance in others. With this in mind, we hope that the new heptoprotector, whose activity does not cause discomfort when used in patients of all ages and genders, will also be a concomitant antidepressant.25-33
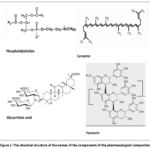 |
Figure 1: The chemical structure of the names of the components of the pharmacological composition. |
Thus, the task set in the manuscript is to create a new composition with hepatoprotective and antidepressant properties based on phospholipids, extracts of food and medicinal plants or secondary metabolites isolated from them with high biological activity. This problem is presented in the medicinal composition, which contains essential phospholipids, unrefined sunflower oil, tomato peel extract (Lycopersicon esculentum), glycyrrhizic acid from the root and rhizomes of licorice glabra, obtained in lower doses compared to the content of Phosphogliv and having a high antioxidant and antidepressant effect, a combination of falavonoids obtained on based on the plant alhagi pseudoalhagi, which has activity in combination with yantacin is solved through.
Initially, the chronic toxic form of hepatitis was caused by the administration of heliothrin to the studied animals in decreasing doses, starting with a lethal dose that caused death through Nina, which caused degeneration and stress in the abdominal cavity of experimental animals: according to the following regimen: three milligrams per kilogram of body weight, five milligrams per kilogram of body weight, seven milligrams per kilogram of body weight, and ten milligrams per hundred grammes of body weight. All observations and analyses in experimental animals were carried out at weekly intervals to assess chronic toxic liver damage. Hematological and biochemical confirmation of chronic toxic liver damage or chronic poisoning was obtained during the fifth week of the experiment.49 When determining liver function by secreting bile under anesthesia (sodium ethaminal 40 mg/kg), conjugates were placed in the bile ducts with open abdominal cavities and bile secretion was studied for 4 hours.50 The ratio of the amount of bile acids and bilirubin in blood plasma was also determined.58 Toxic liver damage, in compliance with the current guidelines for animal experimentation, the animals under study provided the materials needed for analysis.48 Experimental white rates with chronic toxic hepatitis were injected with Hepalipin in its ecdystene-free form and 50 mg/kg of yantacin, or a 1:1 ratio solution; 50 mg/kg of phosphogliv solution; and 10 mg/kg of fluoxetine taken orally for 28 days.
The psychopharmacological activity of the studied composition in the study is considered to be the main screening method. To this end, the psychomotor parameters of experimental animals in an unfamiliar and new calculated environment were studied using the elevated cruciform labyrinth (ECL) method.52
On the other hand, antidepressant activity is the main factor used in the determination of antidepressant methods proposed by L. Steru and studied it in white mice using the methods proposed by R.Porsolt.51, 52, 53
Results
General effects and acute toxicity of the pharmacological composition when administered orally to laboratory white mice and rats.
A sample of hepalipin without ecdystene and acute toxicity of yantacin were given orally to male white rats at a dose of 500 mg/kg to 2000 mg/kg after being dissolved to a state of 10–50% aqueous emulsion. In a vivarium, they were watched for one to two hours, then for a day, and finally for fourteen days. Relative motor activity decreased in the first few minutes at dosages of 3000 and 5000 mg/kg, but it returned to normal after four to five hours. According to GOST,46 the acute toxicity of a sample devoid of hepalipin ecdystene administered orally to rats in a single dose falls within the LD50 -5000 group and above, meaning it is essentially innocuous to class IV. Thus, the studied composition is low-toxic, which guarantees its wide therapeutic effect. A drug with hepatoprotective activity obtained as a comparative drug, the acute toxicity of phospholglyve is LD50 = 800 mg/kg when administered orally, while a drug with the antidepressant activity of fluoxetine is LD50 = 248 mg/kg.54, 55
Investigation of the effect of pharmacological composition affecting blood enzyme activity when chronic toxic hepatitis is present.
Administration of a combination consisting of the sum of an unstenized form of yantacin and hepalipin to the investigated animals show indicators similar to those of rats from the intact group, but their liver enzyme activity is lower than that of rats in the control group, which did not receive this chemical (Fig. 2). Alkaline phosphatase activity, however, stayed within the typical range.
Administration of quantities of yantacin and hepalipin had a normalizing impact on liver function, according to the above data on the dynamics of activity of incretor and excretory enzymes in the blood serum of rats treated with these substances under experimental settings.
Therefore, the pharmacological composition demonstrated similar or significantly higher hepatoprotective effect compared with Phosphogliv and stronger hepatoprotective activity in the model of chronic heliotrine hepatitis when compared to the control group.
Extensive preclinical studies have been conducted on the basis of the extension-free form of yantacin and hepalipin, which have a high reparative ability and exhibit high hepatoprotective properties in severe liver damage caused by chronic toxic damage to a number of plant derivatives.
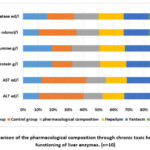 |
Figure 2: Comparison of the pharmacological composition through chronic toxic hepatitis and the functioning of liver enzymes. (n=10) |
The effect of the pharmacological composition on the amount of bile and bile acids in rats with chronic heliotrine hepatitis.
The pharmacological composition increased bile excretion at the study dose by 1.6 times, or up to 60.6%, in contrast to the control group, to 1.02 or 2.1% compared with hepalipin and 1.23 times, or up to 23%, compared with phosphogliv (Table 1). In proportion to this, it can be seen that it also has a positive effect on the level of bile acids and cholesterol in the blood plasma.
Therefore, in comparison to experimental groups receiving control, hepalipin, and phosphogliv, it was shown that the pharmacological composition greatly increased bile excretion against the backdrop of chronic toxic hepatitis. The pharmacological composition has strong choleretic and hepatoprotective properties that are either comparable to or noticeably better than those of phosphogliv and hepalipin.
Table 1: The impact of the pharmacological composition on the liver’s ability to secrete bile and bile acids. (n=10)
| № | Study groups | Doses in mg/kg | In 4 hoursTotal amount of separated grass (mg/100 g) | Bile acids in mmol/l | Total bilirubin in mkmol/l | Cholesterol in mmol/l |
| 1. | Intakt group (dis. water). | 1044,0±1,12 | 21,3±0,48 | 2,15±0,96 | 2,15±0,11 | |
| 2. | Control group (heliothrin | 570,7±2,24 | 11,6±0,55 | 5,12±0,96 | 4,14±0,12 | |
| 3. | Pharmacological composition | 50 + 50 | 916,8±1,12*,a | 18,7±0,89*,a | 3,1±,048*,a | 2,48±0,24*,a |
| 4. | Hepalipin | 100 | 897,6±3,96* | 18,4±0,72 | 3,3±0,72 | 3,2±0,11 |
| 5. | Yantacin | 50 | 796,7±1,12 | 16,2±0,84 | 3,45±1,1 | 3,15±0,19 |
| 6. | Phosphogliv | 50 | 745,2±2,24* | 15,2±0,6 | 3,4±0,96 | 3,05±0,17 |
Note: * – confidence in relation to the control group data-P<0.05
confidence in relation to phosphogliv-P<0.05
Determination of the anxiolytic activity of the pharmacological composition by the method of an elevated cruciform labyrinth (ECL).
In the conducted studies, a number of different results of the studied substances were obtained, which are pharmacological composition and fluoxetine preparation content in the studied doses in special areas of the ECL of experimental animals when observed for 5 minutes (Table 2). In particular, in the studies, the animals of the control group spent most of their time in closed dark handles, where the time spent in the central area was zero, and the latent period of access to the open handle was observed in 60% of the animals. This time was 24,5±1,2 seconds, while the number of transitions between handles was 1,6±0,24 times greater than K (ratio the time spent in the light handle compared to the time spent in the dark area) 0.22 it was found that. When exposed to the studied dose of the pharmacological composition, there was a significant increase in the time spent by experimental animals on light handles compared with the control group compared with the studied doses of Fluoxetine. In addition, the pharmacological composition reduced the latency period of access to the open handle in the study dose compared with the control and fluoxetine and increased the number of transitions. It was observed that the pharmacological composition increased up to 8.4 times in all studied doses compared with the control group by K, respectively, and up to 2.33 times compared with the studied dose of fluoxetine.
Table 2: Comparison of the pharmacological composition and the effect of fluoxetine on ECL. (n=10)
| № | Substances and groups | Time spent by experienced animals in ECL special zones | The K index (Tlight./ Tdark) | ||||
| latency period of access to the open handle in sec. | in closed dark handles (Tdark) in sec. | in the open handles (Tlight) in sec. | in the central region in sec. | the number of transitions between handles | |||
| 1. | Intakt group + Dis.water) | 24,5±1,2 | 207±7,71 | 93±2,41 | 12,8±0,36 | 1,9±0,48 | 0,45 |
| 2. | Control group + Heliotrin | 15,9±1,7 | 246±7,23 | 54±1,2 | 10,5±0,89 | 1,6±0,24 | 0,22 |
| 3. | Pharmacological composition 100 mg/kg | 68,3±3,13*,a | 105,2±2,65*,a | 194,8±5,78*,a | 44,1±1,45*,a | 13,44±1,2*,a | 1,85*,a |
| 4. | Yantacin 50 mg/kg | 44,7±2,41* | 135,5±3,61* | 164,5±2,89* | 35,6±3,86* | 8,95±1,93* | 1,21* |
| 5. | Fluopxetine 10 mg/kg | 40,6±2,65* | 167,7±3,13* | 132,3±0,24* | 27,2±0,48* | 6,37±1,69* | 0,79* |
Note: * – confidence in relation to the control group data-P<0.05
a- confidence in relation to fluopxetine -P<0.05
Pharmacological composition in conditions of chronic heliothrin hepatitis in white mice. Evaluation of antidepressant activity by the R. Porsolt method.
In the research that was done, it was observed that the pharmacological combination in the studied dose increased the period of forced swimming, that is, the period of mobility, of experimental animals by up to 81% in contrast to the control group and up to 26.7% compared with the comparable drug fluoxetine. The results showed that the studied combination showed significantly higher antidepressant activity compared to the control group than fluoxetine, a well-known drug widely used in medical practice (Table 3).
Table 3: Pharmacological composition Comparison of the antidepressant activity of the drug fluoxetine by the R. Porsolt method. (n=10)
| № | Substances and groups | Doses in mg/kg | The period of movement is min. | The rest period is min. | Efficiency in % compared to control | Effectiveness in % compared to fluoxetine |
| 1. | Control group | Dis.water | 148,2±1,12 | 211,8±0,89 | ||
| 2. | Pharmacological composition | 50+50 | 268,4±2,24* | 91,6±0,36* | +81 | +26,7 |
| 3. | Yantacin | 50 | 212,3±1,12 | 147,7±,44 | ||
| 4. | Fluopxetine | 10,0 | 211,8±0,44** | 148,2±0,72** | +43 |
Note: * – confidence in relation to the control group data-P<0.05
**- confidence in relation to fluopxetine -P<0.05
In conditions of chronic heliothrin hepatitis in white mice, the pharmacological composition of Evaluation of antidepressant activity by the L. Steru method.
In studies, it was observed that the pharmacological combination at the studied dose increased the activity period of white mice by up to 48% in contrast to the control group and up to 24% compared with the comparable drug fluoxetine, when experimental animals were left hanging from their tails in a special device and observed for 6 minutes. The results showed that the studied combination showed significantly higher antidepressant activity compared to the control group than fluoxetine, a well-known drug widely used in medical practice (Table 4).
Table 4: Pharmacological composition Comparison of the antidepressant activity of the drug fluoxetine by the L. Steru method. (n=10)
| № | Substances and groups | Doses in mg/kg | The period of movement is min. | The rest period is min. | Efficiency in % compared to control | Effectiveness in % compared to fluoxetine |
| 1. | Control group | Dis.water | 143,4±0,89 | 216,6±2,24 | ||
| 2. | Pharmacological composition | 50+50 | 212,2±2,24* | 147,8±1,12* | +48 | +24 |
| 3. | Yantacin | 50 | 181,4±0,89 | 178,6±0,36 | ||
| 4. | Fluopxetine | 10,0 | 171,2±0,36** | 188,8±0,44** | +20 |
Note: * – confidence in relation to the control group data-P<0.05
**- confidence in relation to fluopxetine -P<0.05
Discussion
Today, with promising advances in the treatment of viral hepatitis, it is an important task to eliminate liver damage caused by pathogenic viruses and normalize liver function.10 To this end, the issue of finding and implementing low-toxic, highly active natural compounds remains relevant. In this regard, the ICPS RUz, together with the RSPSPMC, has been conducting promising research for many years based on these studies. The essence of the presented work is to determine the main pharmacological properties inherent in this class of hepatoprotective drugs in this pharmacological combination., identification of detailed informative indicators on liver dysfunction and restoration of hepatocyte membranes in comparison with the previously studied substance hepalipin and the drug Phosphogliv with known hepatoprotective activity.45,46,47 As well as identifying the main psychopharmacological properties of this composition inherent in antidepressant activity, determining the severity of its antidepressant effect compared to the well-known antidepressant fluoxetine, identifying properties that distinguish it from fluoxetine as an antidepressant by studying the properties involved in specific effects. In this regard, it consists in predicting the advantages of this composition compared to the simultaneous use of Phosphogliv and fluoxetine.25, 26, 49, 56, 57
The result obtained on the basis of the conducted studies showed that the combination of the ecdysten form of hepalipin and yantatacin in a 1:1 ratio is in practice a low-toxic substance, more than 6.25 times less toxic in acute toxicity than a drug with known hepatoprotective activity Phosphogliv, and up to 20.2 times less toxic than the antidepressant drug fluoxetine. This, in turn, not only dramatically reduces the likelihood of side effects when using this formulation, but also provides a high breadth of its therapeutic effect. It should also be said that this composition has the same indicator in terms of the severity of toxicity and the breadth of therapeutic effect as the previously developed and obtained patent for the invention of hepalipin.33-37
It was noted that the presented pharmacological composition has a clear advantage over the comparable substance hepalipin and the well-known drug phosphogliv in terms of hepatoprotective activity. Based on the restoration of enzymatic endocrine and excretory functions of the liver, it was shown that bile secretion, bile acids and cholesterol also improve in proportion to the enzymatic activity of the liver and have a clear advantage over hepalipin and phosphogliv in this activity.38-44
Along with the high hepatoprotective activity, under the influence of this composition, there was a tendency to improve the psycho-emotional state of experimental animals with improve the adaptation of experimental animals to an unfamiliar and new environment. Also, when studying the antidepressant activity of biologically active substances, the main methods of treating depression are used. In particular, L. Steru and R. Porsolt demonstrated a clear high activity in their proposed models. Regarding antidepressant activity, medicine has demonstrated significant antidepressant activity, especially with fluoxetine, a well-known drug widely used in neurology and psychiatric practice.
Thus, studies have confirmed that the presented pharmacological composition has a pronounced increased hepatoprotective activity not only in relation to the well-known drug phosphogliv, but also in relation to the ecdystene form of hepalipin and yantacin. At the same time, it was found that its antidepressant activity is clearly superior not only to the comparable drug fluoxetine, but also to yantacin. The presented composition makes it possible to present it in medical practice as a promising hepatoprotective and antidepressant agent due to its simultaneous high hepatoprotective and antidepressant activity and low toxicity.
Conclusions
Thus, the presented pharmacological composition is absolutely harmless, and studies have confirmed that, according to the studied hepatoprotective activity, the components of the composition have a pronounced increased activity not only in relation to the well-known drug phosphogliv, but also in relation to the ecdysteroid form of hepalipin and yantacin.
In addition, it was found that its antidepressant activity is clearly superior not only to the comparable drug fluoxetine, but also to yantacin.
As a conclusion, we can say that the presented composition makes it possible to present it in medical practice as a promising hepatoprotective and antidepressant agent due to its simultaneous high hepatoprotective and antidepressant activity and low toxicity.
Acknowledgment
The authors’ acknowledgements go to the Institute of Chemistry of Plant Substances named after Academician S.Yu. Yunusov of the Academy of Sciences of the Republic of Uzbekistan and the Republican Specialized Center for Applied Medicine of Pediatric Science for supporting this work.
Funding Sources
The work financial supported by the Ministry of Higher Education, Science and Innovation of Republic of Uzbekistan (project No. AL-412105140 on the topic ”Development of a new biologically active agent for the treatment of liver fibrosis in severe chronic hepatitis”).
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
All research work in this article was carried out on the basis of the permission of the Ethics Committee under the Ministry of Health of the Republic of Uzbekistan No. 1/1-1628.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
\This research does not involve any clinical trials
Author Contributions
- A.A.Aripov: Conceptualization, Methodology, Writing – Original Draft.
- L.L.Akhunzhanova: Data Collection, Analysis, Writing – Review & Editing.
- O.A.Aripov: Funding Acquisition, Resources, Supervision.
- T.Khamroev – Conceptualization, Methodology, Writing – Original Draft. Pharmacological and toxicological experiments, statictical Analysis
References.
- Juliana R. de Carvalho, Cristiane A. et al. Burden of Chronic Viral Hepatitis and Liver Cirrhosis in Brazil – the Brazilian Global Burden of Disease Study. Ann Hepatol.2017. 16. (6). 893-900.
CrossRef - World Health Organization. Hepatitis C 2023. https://www.who.int/news-room/fact-sheets/detail/hepatitis-c
- GBD 2013 Mortality and Causes of Death Collaborators. Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet, 385 (2014), 117-171.
CrossRef - World Health Organization. Global hepatitis report 2017. Geneva, Switzerland. Available from: https://www.who.int/publications/i/item/9789241565455.
- Alberts CJ, Clifford GM, Georges D, et al. Worldwide prevalence of hepatitis B virus and hepatitis C virus among patients with cirrhosis at country, region, and global levels: a systematic review. Lancet Gastroenterol Hepatol. 2022;7:724-735.
CrossRef - Asrani S.K., Devarbhavi H., Eaton J., Kamath P.S. Burden of liver diseases in the world. J. Hepatol. 2019;70:151–171.
CrossRef - Kim BK, Revill PA, Ahn SH. HBV genotypes: relevance to natural history, pathogenesis and treatment of chronic hepatitis B. Antivir Ther. 2011;16(8):1169-86.
CrossRef - Coelho MPP, de Castro PASV, de Vries TP, et al. Sarcopenia in chronic viral hepatitis: From concept to clinical relevance. World J Hepatol 2023; 15(5): 649-665.
CrossRef - Parikh P, Ryan JD, Tsochatzis EA. Fibrosis assessment in patients with chronic hepatitis B virus (HBV) infection. Ann Transl Med 2017;5(3):40. 1-10.
CrossRef - Cowan ML, Thomas HC, Foster GR. Therapy for chronic viral hepatitis: current indications, optimal therapies and delivery of care. Clin Med (Lond). 2011 ;11(2):184-9.
CrossRef - Pol S, Parlati L. Treatment of hepatitis C: the use of the new pangenotypic direct-acting antivirals in “special populations”. Liver Int. 2018 ;38 (1):28-33.
CrossRef - Papaluca T, McDonald L, Craigie A, et al. Outcomes of treatment for hepatitis C in prisoners using a nurse-led, statewide model of care. J Hepatol. 2019 ;70(5):839-846.
CrossRef - Do A, Reau NS. Chronic Viral Hepatitis: Current Management and Future Directions. Hepatol Commun. 2020; 20;4(3):329-341.
CrossRef - You H, Wang F, Li T, et al. Guidelines for the Prevention and Treatment of Chronic Hepatitis B (version 2022). J Clin Transl Hepatol. 2023;11(6):1425-1442.
CrossRef - Holmberg M., Aass H.C.D., Dalgard O. et al. Treatment cessation in HBeAg-negative chronic hepatitis B: clinical response is associated with increase in specific proinflammatory cytokines. Sci Rep 2023;13, 22590. 1-13.
CrossRef - WHO. Hepatitis B Fact sheet 2020, https://www.who.int/news-room/fact-sheets/detail/hepatitis-b (2020).
- Wong G. L. H., Gane E., and Lok A. S. F. How to achieve functional cure of HBV: Stopping NUCs, adding interferon or new drug development? J. Hepatol.2022; 76, 1249–1262.
CrossRef - Jeng W. J., Chen Y. C., Chien R. N., Sheen I. S. & Liaw Y. F. Incidence and predictors of hepatitis B surface antigen seroclearance after cessation of nucleos(t)ide analogue therapy in hepatitis B e antigen-negative chronic hepatitis B. Hepatology 2018; 68, 425–434.
CrossRef - Hall SA.L. Burns GS, Anagnostou D, et al. Stopping nucleot(s)ide analogues in non-cirrhotic HBeAg-negative chronic hepatitis B patients: HBsAg loss at 96 weeks is associated with low baseline HBsAg levels. Aliment. Pharmacol. Ther.2022; 56, 310–320.
CrossRef - Li M, Luo Q, Tao Y, Sun X, Liu C. Pharmacotherapies for Drug-Induced Liver Injury: A Current Literature Review. Front Pharmacol. 2022 5;(12):806249.
CrossRef - Vinniskaya Ye.V. «Ogromnoe schaste dlya praktikuyushego vracha videt, kak tyajely patsient vozvrashaetsya k jizni» Medforum Effektivnaya farmakoterapiya. Gastroenterologiya.2020; 16. (1). 20-28.
- Sonal Datta, Diwakar Aggarwal, Nirmala Sehrawat, et al. Hepatoprotective effects of natural drugs: Current trends, scope, relevance and future perspectives, Phytomedicine,, 2023, 121; 155100,
CrossRef - Norhashima Abd Rashid, Syarifah Aisyah Syed Abd Halim, Seong Lin Teoh, et al. The role of natural antioxidants in cisplatin-induced hepatotoxicity, Biomedicine & Pharmacotherapy, 2021, 144, 112328,
CrossRef - Shuai-Nan Zhang, Qi Liu, Xu-Zhao Li, Wu-De Yang, Ying Zhou. Liver protein and metabolite biolabels reveal hepatoprotective effects and active compounds of Eucommiae folium: Exploration of new application of herb based on multi-omics and bioinformatics. Journal of Pharmaceutical and Biomedical Analysis, 2024, 239, 115870,
CrossRef - Aripov A. N, Akhunzhanova L. L, Nabiev A. U, Aripov A. O, Khamroev T. T.. Antifibrotic Efficacy of a New Phytocomposition of Essential Phospholipids with Glycyrrhizic Acid, Ecdysterone, Lycopene and Proanthacyanidin in Experimental Severe Chronic Hepatitis Compared with Phosphogliv. Biomed Pharmacol J 2023;16 (3). 1815-1825.
CrossRef - Sagdullaev Sh. Sh., Tursunova N. V., Gusakova S. D., et al, UZ Pat. IAP 05701; byull., № 12 (2018).
- Syrov V.N., Gusakova S.D., Khushbaktova Z.A. et al. Hepatoprotective Efficacy of a New Phytocomposition of Essential Phospholipids with Glycyrrhizic Acid, Ecdysterone, and Lycopene in Experimental Chronic Hepatitis Compared to Phosphogliv. Pharm Chem J (2023). 56, 1433–1438
CrossRef - Syrov V.N., Gusakova S.D., Khushbaktova Z.A. et al. Gepatozashitnaya effektivnost novoy fitokompozitsii iz essensialnyx fosfolipidovs glitsirrizinovoy kislotoy, ekdisteronom I likopinom pri eksperimentalnom xronicheskom gepatite sravnitelno s fosfoglivom. Ximiko farmasevticheskiy jurnal. (2022). Tom 56, ¹ 11 3-8
- Nishanbaev S.Z., Shamyanov I.D., Bobakulov X.M., Sagdullaev Sh.Sh. Ximicheskiy sostav I biologicheskaya aktivnost metabolitov rasteniy roda alhagi (obzor). Ximiya rastitelnogo syrya. 2019. ¹4. Ñ. 5–28.
CrossRef - Mamatkulova, N.M., Alimova, D.F., Nishanbaev, S.Z. et al. Neutral substances from Alhagi pseudalhagi. Chem Nat Compd 2012. 48, 908–909
CrossRef - Syrov V. N., Sagdullaev Sh. Sh., Gusakova S. D., et al. Kombinirovannyy preparat Gepalipin kak effektivnoe sredstvo ustraneniya toksicheskogo vliyaniya alkogolya na metabolicheski-funksionalnoe sostoyanie pecheni. Jurnal teoriticheskoy i klinicheskoy meditsiny, № 4, 2020, st.23-25.
- Yuldashev Sh. U., Ulchenko N. T., Khidoyatova Sh. K., Gazizov F. Yu., Gusakova S. D. and Sagdullaev Sh. Sh. New constituents of cuticular lipids from Lycopersicon esculentum fruit skin. Chemistry of Natural Compounds, 2016. 52. (1),35-37.
CrossRef - Abduraxmanov E.R., Surov V.N., Azimova Sh.S. Ekstrakt iz rasteniya alhagi pseudalhagi, farmasevticheskaya i kosmeticheskaya kompozitsiya na yego osnove, sposobы snijeniya soderjaniya xolesterina i triglitseridov v krovi, snijeniya urovnya produktov okisleniya lipidov, uluchsheniya mikrosirkulyatsii, antioksidantnyx svoystv, uluchsheniya sostoyaniya kojnyx pokrovov, stimulirovaniya obrazovaniya kollagena, ranozajivleniya i snijeniya vospaleniya. RU № 2402345:
- Izumi T, Terauchi M. The Diverse Efficacy of Food-Derived Proanthocyanidins for Middle-Aged and Elderly Women. Nutrients. 2020 15;(12):3833.
CrossRef - Jiachang Xiao, Maolin He, Peiran Chen, et al. Proanthocyanidins delay the senescence of young asparagus stems by regulating antioxidant capacity and synthesis of phytochemicals, Food Chemistry: X, 2024, 101222,
CrossRef - Kabra A, Garg R, Brimson J, et al. Mechanistic insights into the role of plant polyphenols and their nanoformulations in the management of depression. Front. Pharmacol. 2022; 13:1046599.
CrossRef - Cires MJ, Wong X, Carrasco-Pozo C and Gotteland M. The Gastrointestinal Tract as a Key Target Organ for the Health-Promoting Effects of Dietary Proanthocyanidins. Front. Nutr.2017; 3:57.
CrossRef - Mullish BH, Kabir MS, Thursz MR, Dhar A. Review article: depression and the use of antidepressants in patients with chronic liver disease or liver transplantation. Aliment Pharmacol Ther. 2014 ;40(8):880-92.
CrossRef - Menon V, Ransing R, Praharaj SK. Management of Psychiatric Disorders in Patients with Hepatic and Gastrointestinal Diseases. Indian J Psychiatry. 2022;64(Suppl 2):S379-S393.
CrossRef - Victoria Tatiana Kronsten, Debbie Lindsay Shawcross, Hepatic encephalopathy and depression in chronic liver disease: is the common link systemic inflammation?, Analytical Biochemistry, 2022, 636, 114437,
CrossRef - Huang X, Liu X and Yu Y. Depression and Chronic Liver Diseases: Are There Shared Underlying Mechanisms? Front. Mol. Neurosci. 2017, 10:134.
CrossRef - Banerjee A.J., Amlan K., Praharaj S.K. et al Depression and Anxiety in Patients with Chronic Liver Disease and their Relationship with Quality of Life. Annals of Indian Psychiatry 2020. 4(1): 28-32,
CrossRef - Cron D.C., Friedman J.F., Winder G.S. et al. Depression and Frailty in Patients With End-Stage Liver Disease Referred for Transplant Evaluation, American Journal of Transplantation, 2016, 16(6) 1805-1811,
CrossRef - Lisitsin A.N., Alymova T.B., Proxorova L.T., Grigoreva V.N., Gorshkova E.I. Nekotorye faktory, opredelyayushie stabilnost rastitelnyx masel k okisleniyu. – Maslojirovaya promyshl., 2005.- 3. 1-15.
- Nguyen MX., Schwartz S.J. Lycopene: chemical and biological properties. – Food tech. – 1999. 53.38-45.
- Gerster H. The potential role of lycopene for human health //J. Am. College of Nutrition. V. 199716. (2). 109-126.
CrossRef - Jarov S.N. Primenenie glitsirrizinovoy kislotы v terapii bolnyx xronicheskim gepatitom S // Farmateka. – 2006. 1. 1-3.
- National Research Council. 2011. Guide for the Care and Use of Laboratory Animals: Eighth Edition. Washington, DC: The National Academies Press. 2011; (8): 356.
- Aripov A.N, Akhunjanova L.L, Khamroev T.T, Aripov Abdumalik Nigmatovich, Akhunjanova Lola Lazizovna, & Khamroev Tolmas Tolibovich. Differential Analysis of Chronic Toxic Hepatitis Caused by The Introduction of Heliotrin Solution in Various Ways. Texas Journal of Medical Science,2022; 4, 58–62.
- Mayboroda A.A., Kalyagin A.N., Zobnin Yu.V., Sherbatyx A.V. Sovremennye podxody k podgotovke originalnoy stati v jurnal mediko biologicheskoy napravlennosti v svete konsepsii«dokazatelnoymeditsiny» // Sibirskiy meditsinskiy jurnal (Irkutsk). – 2008.;. 76. (1). .5-8.
- Porsolt R., Bertin A., Jalfre M. / Behavioral despair in mice: a primary screening test for antidepressants. // Archives Internationales de Pharmacodynamie et de Thérapie. 1977, 229 (2): 327–336.
- Mironov A. N. Rukovodstvo po provedeniyu doklinicheskix issledovaniy lekarstvennыx sredstv / Pod red.— M. : Grif i K, 2012; (1): 944. ISBN 978-5-.
- Steru L., Chermat R., Thierry B., Simon P. The tail suspension test: a new method for screening antidepressants in mice. Psychopharmacology 1985. 85 367–370.
CrossRef - Reichen J, Egger B, Ohara N, Zeltner TB, Zysset T, Zimmermann A. Determinants of hepatic function in liver cirrhosis in the rat. Multivariate analysis. J Clin Invest. 1988 ;82(6):2069-76.
CrossRef - Letalnye dozy veshestv. St.40. http://chemister.ru/Database/Tables/ld.php?pageNum=40
- Rakhimova S.K., Kurbanov U.K., Mezhlumyan L.G. et al. Proteins from the Aerial Part of Delphinium leptocarpum and Their Biological Activity. Chem Nat Compd 2023;(59): 750–753.
CrossRef - Aripov A. N, Akhunzhanova L. L, Nabiev A. U, Aripov O. A, Khamroev T. T. Evaluation of Antifibrotic Activity of a Combination of a New Phytocomposition and Proanthocyanidins in Rats.Biomed Pharmacol J 2024;17(4).2739-2749.
CrossRef - Simonova E.V., Sultanova K.T., Faustova N.M., Romanenko A.Yu., Makarova M.N. Content of total bile acids in blood and bile of laboratory animals. Laboratory Animals for Science. 2023; 4. 87–94.







