Manuscript accepted on :11-09-2025
Published online on: 14-10-2025
Plagiarism Check: Yes
Reviewed by: Dr. Marwah Mohmmed
Second Review by: Dr. EXBRAYAT Jean-Marie
Final Approval by: Dr. Anton R Keslav
Raju Thenge* , Monali Ambuskar
, Monali Ambuskar , Nishant Chopade
, Nishant Chopade , Karan Gayakwad
, Karan Gayakwad , Aditya Bodade
, Aditya Bodade , Mohan Doijad
, Mohan Doijad
Department of Pharmaceutics, Dr. Rajendra Gode College of Pharmacy, Malkapur, Maharashtra, India.
Corresponding Author E-mail:rrthenge@drgcop.ac.in
DOI : https://dx.doi.org/10.13005/bpj/3277
Abstract
Human cancer therapy is a major issue in modern medical science. Lots of emerging materials are developing rapidly. Dendrimers, as a nanocarrier, are now widely used in the field biomedicine, pharmacy, and so on. As a super-branched macromolecule, dendrimers have a series of outstanding properties. Such as simple to functionalize, have nano & symmetric dimension and cavities for host-guest entrapment. It has made much progress in drug and gene delivery. Dendrimers have higher efficiency by perfecting the surface modification methods. The small molecules, DNAs, therapeutic agents, and more can be loaded into dendrimers. Also, the cytotoxicity is reducing gradually. dendrimers are promising drug delivery systems that can overcome the limitations associated with the currently used anticancer drugs. They have the capability to overcome drug resistance, reduce drug toxicity, improved the drug solubility and bioavailability. Different classes of anticancer drugs have been loaded into dendrimers, resulting in targeted drug delivery, sustained drug release mechanism, increased cellular uptake, reduced toxic side effects of the loaded drugs with enhanced anticancer activity in vitro and in vivo.
Keywords
Application; Characterization; Dendrimer; Evaluation; Nanocarriers
Download this article as:| Copy the following to cite this article: Thenge R, Ambuskar M, Chopade N, Gayakwad K, Bodade A, Doijad M. Dendrimer in Cancer Treatment: A Review. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Thenge R, Ambuskar M, Chopade N, Gayakwad K, Bodade A, Doijad M. Dendrimer in Cancer Treatment: A Review. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/3L9Fop9 |
Introduction
Cancer is a life-threatening disease characterized by abnormal and uncontrolled cell proliferation, which can invade other organs of the body.1 Approximately 90-95% of cancer cases are attributed to genetic mutation and 5-10% cases are caused by inherited genetic mutations.2 The World Health Organization (WHO) reported 9 million cancer-related deaths in 2018, which occurred mostly in Asia and Africa. Types of cancer are classified based on the body organ affected, and some examples of cancer types include breast, lung, liver, colorectal, skin, brain, stomach, and pancreatic cancer, etc.3-4 Nowadays, people are paying more and more attention to health issues. Cancer, which is considered one of the most serious health issues, has aroused people’s concerns. However, due to the complex medication environment and methods, those chemies for cancer therapy that ought to satisfy the stringent requirements for solubility, stability, and bioavailability urgently needed. Dendrimer, a type of highly ordered and branched polymeric molecules, just happened to satisfy such demands, especially in the process of gene delivery and drug delivery for cancer therapy.5
Human beings have fought against cancer for thousands of years since it was firstly recorded in ancient Egypt in 2500 BC. Nowadays, there are about 1,000,000 people die of cancer every year. As a result, people worldwide are trying to explore ways to fight cancer to prevent it from taking more lives. Although gene therapy and drug therapy can potentially treat cancer, the clinical implication is not effective because of the lack of an efficient delivery system.6
History of Dendrimer
Dendrimers are an attractive exclusive class of polymers with controlled structure. A dendrimer is both a covalently assemble molecule and also a distinct nanoparticle. The first dendrimers be completed by divergent synthesis advanced by Fritz Vogtle in 1978, R.G. Denkewalter at Allied Corporation in 1981, Donald Tomalia at Dow Chemical in 1983 and in 1985, and by George Newkome in 1985. In 1990 a convergent synthetic approach was introduce by Jean Fréchet. A lot of research has already been completed by studying the different properties and application of dendrimers but a lot of researchers still believe it to be in its initial stage.7-9
Dendrimers: Structure, Property
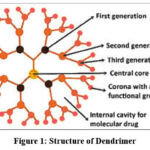 |
Figure 1: Structure of Dendrimer |
Dendrimers are built from a starting atom, such as nitrogen, to which carbon and other elements are added by a repeating series of chemical reactions that produce a spherical branching structure process repeats, successive layers are added and the sphere can be expanded to the size required by the investigator Dendrimers possess three distinguished architectural compounds, namely
An initial core.
Interior layers (generations) composed of repeating units, radically attached to the interior core.
Exterior (terminal functionality) attached to the outermost interior generations.
Dendrimers of lower generations (0, 1, and 2) have highly asymmetric shape and possess more open structures as compared to higher generation dendrimers. As the chain growing from the core molecule become longer and more branched (in 4 and higher generations) dendrimers adopt a globular structure. Dendrimers become densely packed as they extend out to the periphery, which forms a closed membrane-like structure. When a critical branched state is reached dendrimers cannot grow because of a lack of space. This is called the “starburst effect’’.10-11
Properties
Dendritic polymers that can be constructed with a well-defined molecular structure, ie. being mono- disperse, unlike to linear polymers.
Nanoscale sizes that have similar dimensions to important bio-building blocks, e.g., proteins, DNA.
When dendrimer surfaces modified with small functional groups or polyethylene glycol (PEG) show non or low-immunogenicity.
Ability to arrange excretion mode from body, function of nanoscale diameter.
An interior void space may be used to encapsulate small molecule drugs, metals, or imaging moieties. reduces the drug toxicity and facilitates controlled release.
Numbers of terminal surface groups suitable for bioconjugation of drugs, signaling groups, targeting moieties or biocompatibility groups.
Surfaces that may be designed with functional groups to resist trans-cellular, epithelial or vascular bio permeability.
Dendrimers are monodisperse macromolecules. Size and molecular mass of dendrimers can be specifically controlled during classical polymerization process.
When the molecular mass of increases, eases. their intrinsic viscosity goes through a maximum at the fourth generation and then begins to decline.
The presence of many chain-ends is responsible for high solubility and miscibilit and for high reactivity.
Dendrimer solubility is strongly influenced by the nature of surface groups.
The dendrimer should be: nontoxic, on immunogenic, able to cross bio barriers (biopermeable), able to stay in circulation for the time needed to have a clinical effect and able to target to specific structures.12-13
Methods of Synthesis of Dendrimers
The classical polymerization process which results in linear polymers is usually random in nature and produces molecules of different size, whereas size and molecular mass of dendrimers can be specifically controlled during synthesis.
Divergent Method
Convergent Method
Double Exponential and Mixed Method
Hypercores and Branched Monomers Growth
Divergent Method
The divergent growth approach was the first to be proposed, and it remains the most used today. This technique is based on Tomalia and Newkome’s pioneering work, as well as Vögtle’s branching model work.14 The creation of the dendrimer in the divergent process begins in the core and progresses to the periphery. To enhance the reaction with a new monomer, this approach involves two steps: I coupling of monomers and (ii) activation of the monomer end-group.15 The divergent growth approach consists of repeating the two preceding processes until the necessary dendrimer production is obtained. The initial generation of the dendrimer begins with the activation or alteration of the core and the coupling of the first monomer, resulting in divergent processing. The first generation (G1) is then deprotected or activated in order to react with additional branched monomers and create the second generation (G2), and so on. When a new layer of branching units is generated, a new generation is created, with the number of branched layers from the core corresponding to the generation number.16 To avoid deficiently created branches in the divergent method, it is critical that each phase of the reaction be fully finished before adding a new generation.17 In each stage of the synthesis, the surface of the dendrimer may be readily functionalized and changed, resulting in the desired pharmaceutical excipient at the end. Usually, the divergent approach leads to the synthesis of highly symmetric dendrimer molecules.18
Convergent Method
Another way for constructing finely controlled dendritic topologies is the convergent approach. The branching architecture in the convergent method begins from the molecular surface of the dendron and proceeds to a reactive focus point, culminating in the production of a single reactive Dendron.18 Its core operations, like the divergent method, include a coupling phase and an activation step. It allows for more structural control than the prior method. It features a modest number of simultaneous reactions at each development phase, resulting in a product with unrivalled purity and functional diversity. Because of the nanoscale steric problems encountered while connecting the dendrons to the molecular level core, the convergent technique is often confined to the synthesis of just lower generation dendrimers.19
Double exponential and mixed growth
In this method, dendrimers are synthesized by both divergent as well as convergent growth methods It is the most advanced method of dendrimer synthesis where both divergent and methods are used to form a triangle called ‘Dendrimer’. The triangle may be used to repeat the growth process.20
Hypercore and branched method
Pre-assembly of oligomers (a polymers whose molecules consist of relatively few repeating units) join together to produce the desired structure of dendrimer in few steps.
This is the appropriate method over convergent method because the fewer steps are required for synthesis of higher generations of dendrimer.21
Types of Dendrimers 20,21
Radially layered poly (amidoamineorganosilicon) dendrimers (PAMAMOS):
In 1990, Dr. Petar Dvornic and his colleagues at Michigan Molecular Institute discovered this unique first commercial silicon containing dendrimers. Consist of hydrophilic, nucleophilic polyamidosamine (PAMAM) interiors and hydrophobic organosilicon (OS) exteriors, Excellent its networks regularity and ability to complex and encapsulate various guest species offer unprecedented potentials for new applications innanolithography, lectronics, photonics, chemical catalysis etc. and useful precursors for the preparation of honeycomb like networks with nanoscopicPAMAM and OS domains
Poly (amidoamine) dendrimers (PAMAM)
Synthesized by the divergent method, starting from initiator core reagents like ammonia or ethylenediamine. When looking at the structure of the high generation in two-dimensions, star like pattem observed. They are commercially available as methanol solutions and ingeneration G 0-10 with 5 different core type and 10 functional surface groups.
Poly (Propylene Imine) dendrimers (PPI)
Poly (Propylene Imine) dendrimers (PPI) generallyhaving poly-alkyl amines as end groups, and numerous tertiary trispropylene amines present in interior portion.It commercially available up to G5, and wide applications in material science as well as in biology. PPI dendrimers are available as Astramol TM.
Chiral Dendrimers
The chirality in these dendrimers is based upon the construction of constitutionally different but chemically similar branches to chiral core. Their potential use chiral hosts for enantiomeric resolutions and as chiralcatalysts for asymmetric synthesis.
Liquid crystalline dendrimers
A highly-branched oligomer or polymer of dendritic structure containing mesogenic groups that can display mesophuse behaviour. They consist of mesogenic (lig. crystalline) monomers eg. mesogen functionalized carbosilane dendrimers,
Tectodendrimer
TectoDendrimer are composed of a core dendrimer, perform varied functions ranging from diseased cell recognition, diagnosis of disease state drug delivery, reporting location to reporting outcomes of therapy.
Hybrid dendrimers
Hybrid dendrimers are hybrids (block or graft polymers) of dendritic and linear polymers. Obtained by complete monofunctionalization of the peripheral amines of a “zero-generation “polyethyleneimine dendrimer, provide structurally diverse lamellar, columnar, and cubic selforganized lattices that are less readily available from other modified dendritic structures.
Multilingual Dendrimers
These types of dendrimers can be engineered with multiple functional groups, each capable of different types of interaction with cells, tissues, or biomolecules. This versatility can make them extremely useful for a variety of applications, including drug delivery, gene therapy, imaging, and diagnostics. Just as a multilingual person can switch between different languages based on the context, these dendrimers can switch or adapt to different biological interactions depending on the therapeutic needs.
Micellar Dendrimers
Micellar dendrimers represent an innovative class of drug delivery systems that combine the unique properties of both micelles (self-assembled structures) and dendrimers (highly branched macromolecules). These systems have shown great potential in improving the bioavailability, solubility, and controlled release of drugs, especially for poorly water-soluble or hydrophobic compounds.
Advantages
Direct administration of medication to a patient’s afflicted body area.
Drugs can also be released in a controlled and continuous manner.
Drugs may readily be made to stay inside the layers of skin and not enter the bloodstream.
Bypassing the gastrointestinal medium, and so avoiding variations caused by gastric secretions.
Reduced clearance of drug owing to changed distribution of the drug in organs at the site of localization and transportation due to regulated and prolonged release of the drug9
Drug loading is unusually high.
Drug activity is preserved because pharmaceuticals may be integrated into systems without causing a chemical reaction.
Other nanoparticles’ shortcomings are such as liposomes’ low encapsulation effectiveness, quick leakage of water-soluble drugs in the presence of blood components, and reduced storage stability.
Dendrimers are good for targeting solid tumours because of their high permeability and poor drainage in the tumour vasculature, which leads to macromolecule buildup in the tumour (enhanced permeation rate). Targeted delivery (attaching site-specific ligands at the surface or magnetic guiding) also reduces the quantity of medicine used, lowering systemic toxicity.21
Disadvantages
Divergent
Large excess of reagents
Difficulties in purification
Possible incomplete reactions with terminal groups
Incomplete reactions increases exponentially
Convergent
Does not allow the formation of high generation dendrimer because steric problems occure in the reactions of the dendrons and the core molecule.22
Evaluation of Dendrimers
Spectroscopy and spectrometry
Spectroscopy and spectrometry of dendrimer is qualitative or quantitative technique. It is used to determine both inorganic and organic compound. It can be done by IR. Raman spectroscopy, NMR, mass spectroscopy, fluorescence technique, x-ray photoelectron technique, atomic force microscopy, ultra-violet-visible (UV-vis). 23
Ultra-Violet-Visible (UV-Vis) Spectroscopy:
UV-Vis technique gives us the proof of synthesis due to specific highest absorption or shift in the value of lambda max. The range of the UV-Vis spectroscopy lies between 200 to 800nm. The functional moiety which is attached to the dendrimer molecules can be detected by UV spectroscopy, In UV spectroscopy characteristic curve tell us definite highest absorption peak at definite wavelengths.23
Infrared (IR)
At the surface of dendrimers this technique gives the information about routine analysis of chemical transformation. It is also used for the determination of synthesis, IR gives us the proof of synthesis progress by appearance- disappearance-reappearance of characteristics peaks. IR range lies between 0.8 to 1000 micrometer. Kolhe et al. confirmed that the disappearance of aldehydes all through the synthesis of phenoxy methyl (methyl hydrazono) (PMMH) dendrimers ruminates synthesis. 24
Raman Spectroscopy
This technique can be used for the study of vibrational, rotational and another frequency mode. It may be used in thedetermination of interaction of poly (amidoamine) dendrimer with a lipid layer.24
Nuclear Magnetic Resonance (NMR)
When we go for grade by grade synthesis of dendrimers (NMR) nuclear magnetic resonance analyses are mainly used. Because nuclear magnetic resonance (NMR) provides us the tips about analytical transmutation undergone by the last group. By the help of (REDOR) rotational echo double resonance, NMR solid-state spectroscopy complexed PAMAM dendrimers, and PAMAM dendrimers are evaluated. H¹ and C NMR are mostly used for an organic compound such as poly (ester ketone), Poly (propylene imine), polyphenylester.One-dimensional and two- dimensional NMR research used to probe the conformation of melamine dendrimer which bears specific NMR alerts from the middle to the periphery.25
Mass Spectroscopy
It is used to measure the mass to charge ratio of charged particles. The rapid and exact resolution of molar masses, impurities.etc. is realized with mass spectroscopy. On the idea of function fragmentation pattern, the completedinvestigation of faults dendrimer is suitable for (MALDI-TOF) matrix-assisted laser desorption/ionization time of flight. For the determination of fragmentation pattern of different dendrimers, mass spectroscopy is used.26
Scattering techniques
Scattering techniques can be done by (SAXS) small angle x- ray scattering. (SANS) small angle neutron. scattering. (LLS) laser light scattering.
(LLS) Laser Light Scattering
It can be used to determine the hydrodynamic radius of the dendrimer. We can also use this technique as a coupled detector to size exclusion chromatography instrument. For the detection of aggregates, dynamic laser light scattering is used. Laser light scattering (LLS) can be utilized in the direct analysis of the pattern of dendrimer in solution(LLS), we can evaluate the molecular weight of PPI dendrimers.27
(SAXS) Small Angle X-ray Scattering
This technique can be used for the evaluation of polymers. By applying this technique on the dendrimer, we can easily get information about their average (R) radius of gyration in solution. To afford the radius of gyration (R) values of Poly (amidoamine) (PAMAM) dendrimer and fluorinated dendrimers SAXS was used. 28
iii. (SANS) Small Angle Neutron Scattering: SANS is used to determine the area of completing organizations of the PPI dendrimers and PAMAM dendrimers. They are having labeled, and unlabeled end groups but in the prior case near the periphery end groups are concentrated. It gives us the correct data about the inner shape of the whole dendrimer than SAXS. SANS denoted us the molecular weight of Poly (amidoamine) (PAMAM) dendrimers, Poly (benzyl ether) (PBzE) dendrimers.28
Microscopy
Microscopy is a scientific instrument. It uses to observe objects on beam of an active electron utterly first-rate scale, merry Microscopy technique can be done by TEM transmission electron microscopy. SEM scanning electron microscopy.29
(SEM) Scanning Electron Microscopy
To study of the surface topography of dendrimers, SEM is usually applied. To attaining the deeper understanding and investigation of phenyl-OH terminated dendritmer and its surface properties, scanning electron microscopy was applied. At a few angstroms of sensitive cantilever arm with the sample, (SEM) scanning electron microscopy provide us an image by touch contact. To have a look at metallo-dendrimers which is having thodamine B at the focal point(NSOM) near-field, scanning optical microscopy has been used. Dadapeeret al. Implemented(SEM)Scanning Electron Microscopy in studying phenyl-OH terminated dendrimer that lets in you to get a deeper expertise of its surface properties. 29
(TEM) Transmission Electron Microscopy
It is a microscopic technique used to observing only aggregates of small arborols (ARB). In this approach, the beam of electrons is transmitted thru an extremely fine specimen. After transmitting thebeam of electrons thru the extremely fine specimen then they may be interacting with the specimen and as it is passed via it. Transmission electron microscopy was used to study the PAMAM dendrimer molecules if you want to get a deeper expertise of form, common length and length distribution for G10 to G5. 29
Electrical techniques
Electrical techniques are used for producing low detection limit. With the help of electrical technique, we can describe a lot of evaluation information electrochemically such as equilibrium constants for chemical reactions, the extent of adsorption and the rates, a rate of mass transfer, Stoichiometry and rate of interfacial charge transfer. It can be done by electrophoresis, electrochemistry. paramagnetic resonance (EPR) electron.30
Electrophoresis
This technique offers useful data about the purity and homogeneity of numerous water-soluble dendrimers. The purity of poly (amidoamine) (PAMAM) dendrimer was investigated by the use of mass spectroscopy, gel electrophoresis and C NMR spectroscopy (Ottaviani et, al). To the separation of biopolymers such as nucleic acid and proteins, gel electrophoresis is used. It is also used in biology for routine analysis”31
Electrochemistry
To concerning the structure of the dendrimer electrochemistry may afford generally three types of information. To measure the number of electroactive groups, exhaustive coulometry has been used. But in many studies, ferrocenes united to the surface of phenoxy methyl (methyl hydrazono) (PMMH) dendrimers, poly (propyl imine) (PPI) dendrimers etc. which is joined to the PAMAM dendrimers With the help of cyclic voltammetry, we can be detected the burying degree of the electroactive groups inside the dendrimers.32
(EPR) Electron Paramagnetic Resonance
To have look at the chemical species with one or more unpaired electron which includes inorganic complexes owning transition metallic or inorganic and natural free radicals, electron paramagnetic resonance (EPR) method is used. Ottaviani et al. Pronounced the usage of (EPR) electron paramagnetic resonance within the studies of the adsorption of dendrimers on activated alumina, homo-porous silica.32
Size Exclusion Chromatograph (SEC)
To the separation of molecules in step with size, size exclusion chromatography (SEC) is used. The detector inclusive of a differential refractive index is hooked up to the(SEC) size exclusion chromatography equipment for the determination of the polydispersity. There are many types of dendrimers evaluated by size exclusion chromatography (SEC), even self-assembled dendrimers are also evaluated by SEC. For polyether dendrimers, well-evaluated PAMAM dendrimer was used as a standard. Because of increasing the generation number of poly benzyl ether (PBzE) and Phenoxy methyl (methyl hydrazono) (PMMH) dendrimers. To change the monitor size of arborols (ARB) dendrimer with PH variance size exclusion chromatography (SEC) was used.33
Rheology, Physical Properties
Rheology, physical properties can be done by dielectric spectroscopy, intrinsic viscosity, (DSC) differential scanning calorimetry.
(DSC) Differential Scanning Calorimetry
To detect the glass transition temperature (Tg), (DSC) differential scanning calorimetry is used. Tg is predicated upon at the chain-end composition of polymers, entanglement and molecular weight. Tg is influenced by the molecular mass of poly benzyl ether (PBzE) dendrimers, correlates with n /M (n is the number of chain ends) and end group substitutions. To detect physical aging of phenoxy methyl (methyl hydrazono) (PMMH) dendrimers temperature modulated calorimetry (TMC) and DSC is used.34
Intrinsic viscosity: It is as unalytical probe of the morphological structure of deudrimers
Dielectric spectroscopy (DS): Gives complete information about molecular dynamic processes
X-ay Photoelection Spectroscopy (XPS): It provides detailed infoensation about chemical composition of deuttimers such as poly (aryl ether) dendrous or PAMAM dendrimers which was obtained using XPS. This technique is most generally used e the characterizatice of lovers
Sedimentation:Technique used for lactosylated PAMAM dendrizzers, measurements of dipole moments for PMMA dendrimer. 34
Titrimetry:is used for determining number of NH2 end groups of PAMAM dendrimers.
Table 1: List of API used in the formulation of Dendrimer as drug delivery system
| Drugs | Category | MOA | Uses | References |
| Methotrexate | Antimetabolite drug | adenosine metabolism disruption, purine nucleotide synthesis inhibition, and DNA synthesis inhibition. | Breast cancerHead and neck cancer
Lung cancer |
35,37,38,40 |
| Doxorubicin Hydrochloride | Cytotoxic Anthracyclin antibiotics | Activity of Topoisomerase ll inhibited by doxorubicin HCll | Ovarian cancerThyroid cancer
Wilms tumor |
41,42 |
| Paclitaxel | Platinum drug | Encourages tubilin formation and keep microtubules stable | Ovarian cancerBreast cancer
Lungs cancer |
40,43 |
| Campothecin | anticancer drug | anticancer medications causes DNA damage, and lead destruction | Colorectal CancerOverian cancer | ,40,45 |
| 5 Flurouracil | Pyrimidine antimetabolite,Chemotherapeutic drug | Prevent the production of erczyme which is necessary for DNA synthesis | Breast CancerColon cancer
Skin cancer |
37,40,44 |
| Docetaxal | Taxoid antineoplastic agent | DNA damage to cell,death of malignant cell | Metastatic breast cancer, and prostate cancer.andHead/neck cancer | 35,40 |
| Melphalan | Antineoplastic,Alkylating agent | Melphalan is an Alkylating substance that attaches itself to DNA causing damage to molecule and ultimately leading to the death of cancer cell | Overian cancer | 46 |
| Cisplatin | AntineoplasticAlkylating agent | By attaching itself to nuclear DNA and subsequently interfering with normal transcription orDNA medications produced it cytotoxic effect | Breast Cancer, lungs cancer,overian cancer | 39,40 |
| Cyclophosphamide or Cytoxan | Alkylating agent Anti-copianshe drug | Stop the production of DNA and cell division | Breast CancerOverian cancer | 47 |
Future Directions and Trends
Multifunctional and orthogonal chemistries
Researchers are exploring “click” and orthogonal chemistries to install drugs, targeting ligands, and imaging moieties in controlled ways without compromising surface properties, increasing payload, and reducing heterogeneity that limits translation.48
Biodegradable dendrimers
Development of biodegradable scaffolds like bis‑MPA, poly‑L‑lysine (PLL), PGLSA‑OH, or poly(phosphorhydrazone) aims to overcome accumulation and long-term toxicity seen with non‑degradable polymers.49
Gene therapy and theranostics
Continued preclinical progress for delivery of DNA/RNA, especially in cancer gene therapy; however, few have reached human trials so far Integrative platforms combining imaging (e.g. MRI agents) and therapy are also under development50 .
Improved pharmacokinetics
Surface modifications such as PEGylation and mixed-surface designs are helping reduce renal clearance, boost circulation time, and allow tumor targeting via ligands.51
Targeted clinical candidates
Companies such as Starpharma are leading with dendrimer drugs like SPL7013 (VivaGel) used topically for infections (approved in Japan for HIV/genital herpes prevention), and AZD0466, a poly‑lysine drug conjugate in early human studies for oncology52.
Translation to Clinical Use
Scarce clinical translation
Despite decades of study, relatively few dendrimer-based therapies have reached clinical trials. The gap stems from challenges in characterization, toxicity, reproducibility, and regulatory uncertainty.53
Validated clinical candidates
SPL7013 (Starpharma): a poly‑L‑lysine dendrimer in topical gel form with clinical trials showing safety and efficacy for bacterial vaginosis and STI prevention; commercialized in Japan as condom coating.
AZD0466: a dendrimer–drug conjugate based on a poly‑Lysine G5 core, currently in early trials for liver cancer; trials in small patient cohorts demonstrated safety and preliminary activity.54
Translational hurdles
Sterilization issues: Standard high‑heat sterilization (e.g. autoclaving at 134 °C) can destroy PAMAM dendrimers; sterile filtration may be needed, introducing risks of contamination or endotoxins.
Toxicology & biocompatibility: Cationic dendrimers (e.g. PAMAM) exhibit dose- and generation-dependent cytotoxicity, membrane disruption, hemolysis, neurotoxicity; surface modification (e.g. PEGylation or acetylation) is used to mitigate these effects.55
Critical-quality attributes (CQAs)
Dendrimer conjugates lie between small molecules and biologics; they require rigorous characterization of size, polydispersity, morphology, drug‑loading, drug release, stability, and interactions with plasma proteins-all of which must be defined and monitored in regulatory submissions.56
Regulatory and Manufacturing Considerations
Regulatory Pathways
Lack of specific guidance
While regulatory bodies like FDA and EMA have issued general nanomedicine guidelines (e.g. “Drug Products, Including Biological Products, that Contain Nanomaterials”), no dedicated guidance specifically addresses dendrimer‑drug conjugates. Regulatory pathways remain inconsistent and uncertain.57
Combination product classification
Many dendrimer therapeutics combine device-like architecture with drug payloads. They may be treated as combination products, requiring coordination between drug, biologics, and device division regulatory frameworks.
Need for new assays
Standard toxicology and PK/PD assays may not translate for dendrimers. There is strong need for new in vitro models that better predict in vivo outcomes, as well as bioinformatics tools to model interactions based on physico-chemical parameters.58
Manufacturing and Analytical Challenges
Complex multistep synthesis
Traditional divergent growth for high-generation dendrimers is slow, labor-intensive, and prone to structural defects, trailing generations, and impurities that are difficult to separate from the desired product.59.
Scale-up and reproducibility
Batch-to-batch consistency and large-scale GMP-compliant production are highly challenging. Purity, monodispersity, and defect removal become harder with higher generation and multifunctional scaffolds.60
Analytical toolbox
Regulatory approval demands an array of orthogonal characterization methods: e.g. DLS, SEC‑MALS, mass spectrometry, electron microscopy, to robustly measure size, morphology, composition, and drug release. Lack of validated assays restricts progress61.
Cost considerations
Manufacturing remains expensive due to high reagent usage, slow synthesis, and requirement for high-level analytical and process control. PEGylation or surface modifications add further cost.62
Table 2: Comparison of Dendrimers, Polymeric Micelles and Liposomes63-65
| Feature | Dendrimers | Polymeric Micelles | Liposomes |
| Size | ~1–20 nm, monodisperse | ~10–100 nm, size tunable | ~50–200 nm, less tunable |
| Drug loading | High; encapsulation, covalent binding | Hydrophobic core; limited capacity | Both hydrophilic and hydrophobic drugs |
| Stability | High (requires design) | Low below CMC; crosslinking needed | PEG improves stability; risk of clearance |
| Toxicity | Surface-charge dependent; modifiable | Low but polymer safety must be ensured | Generally biocompatible; concerns with PEG |
| Targeting | Multivalent ligand conjugation | Ligand-functionalizable; passive targeting | Ligand-targeting feasible but complex |
| Manufacturing | Complex, multi-step | More scalable, but polymer quality matters | Established but costly and variable |
| Clinical status | Preclinical | Early clinical trials | Multiple FDA-approved formulations |
Application of Dendrimers66-72
Drug Delivery: Encapsulation and Conjugation
Unimolecular micelles
Some dendrimers (e.g., with hydrophobic core and hydrophilic periphery) can encapsulate hydrophobic drugs within their interior cavities, enhancing solubility and stability (e.g. 9000‑fold increase for paclitaxel upon PAMAM conjugation).
Covalent drug conjugates
Drugs can be chemically linked to dendrimer periphery via cleavable linkers (pH-, redox-, ROS-sensitive). For instance, methotrexate half-life extended from 24 min to 24 h when conjugated to dendrimer; camptothecin‑CPT release via pH‑sensitive bonds downregulates tumor growth.
Surface modifications
PEGylated dendrimers improve circulation time and reduce toxicity. Conjugation with hyaluronic acid targets CD44 receptors in tumors, while mannose-functionalized dendrimers target macrophages via LXR ligands.
Targeting & Pharmacokinetics (PK/PD)
Brain targeting
Neutral hydroxyl-terminated PAMAM dendrimers preferentially accumulate in inflamed brain regions and activated microglia—shown in models of cerebral palsy, traumatic injury, and Rett syndrome. Dendrimer-N-acetylcysteine conjugates reversed pathology at much lower doses than free drug.
Gene Delivery and Nucleic Acid Transport
Non-viral gene vectors
Cationic dendrimers (e.g. PAMAM, PPI) efficiently bind DNA/RNA via electrostatic interaction, enhancing transfection while reducing toxicity when optimally balanced.
Advanced formats
dendrosomes—lipid-encapsulated dendrimer–DNA complexes favor delivery to antigen-presenting cells via intramuscular or mucosal injections. They’ve demonstrated siRNA delivery targeting oncogenes in cervical cancer models
Routes of Administration
Oral delivery
Dendrimers can enhance absorption by modulating tight junctions and efflux transporters. Conjugation with PEG or neutral ligands reduces GI toxicity and improves bioavailability with minimal degradation.
Transdermal delivery
PAMAM dendrimers significantly increase skin permeation of NSAIDs (e.g., indomethacin, resveratrol) by 2–4.5× over free drug, attributed to improved solubility and flux.
Ocular delivery:
In rabbit models, dendrimer-based eye formulations (e.g., microgels, hydrogels) show enhanced residence time and better intraocular pressure reduction than traditional polymers; DenTimol (dendrimer-timolol conjugate) outperformed standard eye drops in normotensive rats.
Intranasal delivery
PAMAM dendrimer formulations of haloperidol improved solubility and nose-to-brain transport, reaching higher brain concentrations via mucosal adhesion and charge-mediated interactions.
Other Biomedical and Non‑Biomedical Uses
Sensors and Diagnostics
Dendrimers functionalized with chromophores (e.g. coumarin, porphyrins) serve as light-harvesting antennae, energy transfer systems, and pH/metal-ion‑sensitive sensors for molecular detection.
Catalysis
Metallodendrimers (e.g. Ni‑, Pd‑, Ru‑based) act as heterogeneous catalysts in organic reactions like Heck and polymerization, and as electrocatalysts for small molecules at physiological pH.
Antimicrobial nano-antibiotics
Grafting PPI dendrimers onto mesoporous silica nanoparticles loaded with levofloxacin enhances bacterial membrane penetration and biofilm disruption against Gram-negative pathogens.
Conclusion
Dendrimers are highly branched, nanoscale macromolecules with precise molecular architecture, offering unique advantages in biomedical applications such as drug delivery, gene therapy, imaging, and diagnostics. Their well-defined structure allows for the encapsulation or conjugation of therapeutic agents, enhancing solubility, stability, and targeted delivery.Despite extensive research, the clinical translation of dendrimer-based therapies has been limited. Challenges include potential cytotoxicity, especially with cationic and higher-generation dendrimers, and the complexity of large-scale synthesis. Moreover, regulatory hurdles and the need for reproducible manufacturing processes have hindered their widespread clinical adoption.
Acknowledgement
Authors are thankful to the Dr. P. K. Deshmukh, Principal, Dr. Rajendra Gode College of Pharmacy, Malkapur and Management for proving the necessary library facilities to carry out this review work.
Funding Source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Raju Thenge : Concept, supervision
- Monali Ambuskar: Drafting of article
- Nishant Chopade: Supervision
- Karan Gayakwad: Data correction and rewriting
- Aditya Bodade: Data correction and writing
- Mohan Doijad: Literature survey and data collection
References
- Singh S, Sharma B, Kanwar SS, Kumar A. Lead phytochemicals for anticancer drug development. Front Plant Sci. 2016;7:1667. doi:10.3389/fpls.2016.01667.
CrossRef - Anand P, Kunnumakara AB, Sundaram C, et al. Cancer is a preventable disease that requires major lifestyle changes. Pharm Res. 2008;25(9):2097-2116. doi:10.1007/s11095-008-9661-9.
CrossRef - Okuhara T, Ishikawa H, Urakubo A, et al. Cancer information needs according to cancer type: a content analysis of data from Japan’s largest cancer information website. Prev Med Rep. 2018;12:245-252. doi:10.1016/j.pmedr.2018.01.014.
CrossRef - Peter S, Aderibigbe BA. Ferrocene-based compounds with antimalaria/anticancer activity. Molecules. 2019;24(20):3604. doi:10.3390/molecules24203604.
CrossRef - Chauhan A. Dendrimers for drug delivery. Molecules. 2018;23(4):938. doi:10.3390/molecules23040938.
CrossRef - Lu Z, Qiao P. Drug delivery in cancer therapy, quo vadis? Mol Pharmaceutics. 2018;15(9):3603-3616. doi:10.1021/acs.molpharmaceut.8b00634.
CrossRef - Tomalia D.A., Fréchet J.M. Discovery of Dendrimers and Dendritic Polymers: A Brief Historical Perspective. J. Polym. Sci. Part A: Polym. Chem. 2002;40:2719–2728. doi: 10.1002/pola.10301.
CrossRef - Gaudana R, Jwala J, Boddu SHS, Mitra AK. Recent perspectives in ocular drug delivery. Pharm Res. 2009;26(5):1197-1216. doi:10.1007/s11095-009-9922-7.
CrossRef - Kolhe P, Misra E, Kannan RM, Kannan S, Lieh-Laib M. Drug complexation, in vitro release and cellular entry of dendrimers and hyperbranched polymers. Int J Pharm. 2003;259(1):143-160. doi:10.1016/S0378-5173(03)00310-6.
CrossRef - Gondkar SB, Rasal SP, Saudagar RB. Dendrimer: a review. Asian J Pharm Res. 2016;6(3):188-192. doi:10.5958/2231-5691.2016.00027.7.
CrossRef - Gohel MC, Parikh RK. Dendrimer: an overview, targeted drug delivery systems. net. 2009;7(3):36-39.
- Wikipedia. Accessed June 14, 2025.
- Sakthivel T, Toth I, Florence AT. Synthesis and physicochemical properties of lipophilic polyamide dendrimers. Pharm Res. 1998;15(6):776-782. doi:10.1023/A:101195352.
CrossRef - Peeyushkumar KP, Meena P, Choudhary C, Thakur DS, Bajpayee P. Dendrimer: a novel polymer for drug delivery. J Ind Chem Soc. 2010;87:252-269.
- Grayson SM, Frechet JM. Convergent dendrons and dendrimers: from synthesis to applications. Chem Rev. 2001;101(12):3819-3868. doi:10.1021/cr990116h.
CrossRef - Boas U, Christensen JB, Heegaard PM. Dendrimers in medicine and biotechnology: new molecular tools. Cambridge, UK: Royal Society of Chemistry; 2006.
CrossRef - Caminade AM, Turrin CO, Laurent R, Ouali A, Delavaux Nicot B, eds. Dendrimers: towards catalytic, material and biomedical uses. Chichester, UK: John Wiley & Sons; 2011:1-33.
CrossRef - Mendes LP, Pan J, Torchilin VP. Dendrimers as nanocarriers for nucleic acid and drug delivery in cancer therapy. Molecules. 2017;22(9):1401. doi:10.3390/molecules22091401.
CrossRef - Sharma MS, Sheri PS. Dendrimers: a promising tool for drug delivery. J Pharm Sci Res. 2020;12(10):1309-1314.
- Akbarzadeh A, Khalilov R, Mostafavi E, et al. Role of dendrimers in advanced drug delivery and biomedical applications: a review. Exp Oncol. 2018;40(3):178-183. doi:10.31768/2312-8852.2018.40(3):178-183.
CrossRef - Petar R, Dvornic L, Douglas S, MichaelJ and Owen SP: Radially Layered Co poly(amid amine organ silicon) Dendrimers, United States Patent 1998; 5: 739.
- Singh J, Jain K, Mehra NK, Jain NK. Dendrimers in anticancer drug delivery: mechanism of interaction of drug and dendrimers. Artif Cells Nanomed Biotechnol. 2016;44(7):1626-1634. doi:10.3109/21691401.2015.1041912.
CrossRef - Dighe NS, Pattan SR, Musmade D, et al. Convergent synthesis: a strategy to synthesize compounds of biological interest. ChemInform. 2011;42(29):no. 29.
CrossRef - Thakur R, Gupta V. A review on evaluation of dendrimers. J Pharm Sci Res. 2012;4(1):1-10.
- Augustus EN, Allen ET, Nimibofa A, Donbebe W. A review of synthesis, characterization and applications of functionalized dendrimers. Am J Polym Sci. 2017;7(1):8-14. doi:10.5923/j.ajps.20170701.02.
- Tuija GR, Gowry DS, Shereesma M, Satyanarayana B. Spectrophotometric method for the determination of angiotensin II receptor antagonist in bulk and pharmaceutical dosage forms. Int J Pharm Sci. 2012;4(1):1-5.
- Furer VI, Vandyukov AE, Majoral JP, Caminade AM, Kovalenko VI. Fourier-transform infrared and Raman difference spectroscopy studies of the phosphorus-containing dendrimers. Acta Mol Biomol Spectrosc. 2011;60(12):1649-1657. doi:10.1016/j.saa.2011.06.019.
CrossRef - Victoria M, Guerra J, Aldrik H, Richard M. NMR characterization of fourth-generation PAMAM dendrimers in the presence and absence of palladium dendrimer-encapsulated nanoparticles. J Am Chem Soc. 2008;131(1):341-350. doi:10.1021/ja076907a.
CrossRef - Wang D, Imae T. Fluorescence emission from dendrimers and its pH dependence. J Am Chem Soc. 2004;126(42):13204-13205. doi:10.1021/ja047401m.
CrossRef - Dadapeer E, Babu HB, Reddy HC, Charmarthi RN. Synthesis, spectral characterization, electron microscopy and thermogravimetric analysis of a phosphorus-containing dendrimer with diphenylsilanediol as core unit. Beilstein J Org Chem. 2010;6:726-731. doi:10.3762/bjoc.6.85.
CrossRef - Jackson LC, Chanzu DH, Booy PF, Drake JB, Tomalia DA, Baur JB, Amis JE. Visualization of dendrimer molecules by transmission electron microscopy (TEM): staining methods and cryo-TEM of verified solutions. Macromolecules. 1998;31(18):6259-6265. doi:10.1021/ma980389p.
CrossRef - Ottaviani FM, Turro JN, Jockush JN, Tomalia DA. EPR investigation of the adsorption of dendrimers on porous surfaces. J Phys Chem B. 2003;107(9):341-350. doi:10.1021/jp0217269.
CrossRef - Gautam SP, Gupta AK, Agrawal S, Sureka S. Spectroscopic characterization of dendrimers. Int J Pharm Pharm Sci. 2012;4(2):77–80.
CrossRef - Touzani R. Dendrons, dendrimers: new materials for environmental and science applications. J Mater Environ Sci. 2011;2(3):201–214.
- Kaurav M, Ruhi S, Al-Goshae HA, et al. Dendrimer: An update on recent developments and future opportunities for the brain tumors diagnosis and treatment. Front Pharmacol. 2023;14:1159131.
CrossRef - Gajbhiye V, Jain NK. Dendrimers as therapeutic agents: a systematic review. J Pharm Pharmacol. 2009;61(8):989–1003.
CrossRef - Janiszewska J, Hegde M, Zhu X, Gorzkiewicz M. Recent advances in dendrimer-based drug delivery systems. J Control Release. 2020;320:1–14.
- Du J, Lu WL, Ying X, et al. Methotrexate-loaded polyether-copolyester dendrimers for the treatment of gliomas: enhanced efficacy and intra-tumoral transport capability. Mol Pharm. 2008;5(1):105–116.
CrossRef - Kesavan A, Ilaiyaraja P, Sofi Beaula W, et al. Tumor targeting using polyamidoamine dendrimer–cisplatin nanoparticles functionalized with diglycolamic acid and herceptin. Eur J Pharm Biopharm. 2015;96:255–263.
CrossRef - Sharma AK, Gothwal A, Kesharwani P, Alsaab H, Iyer AK, Gupta U. Dendrimer nanoarchitectures for cancer diagnosis and anticancer drug delivery. Drug Discov Today. 2017;22(2):314–326.
CrossRef - Lai PS, Lou PJ, Peng CL, et al. Doxorubicin delivery by polyamidoamine dendrimer conjugation and photochemical internalization for cancer therapy. J Control Release. 2007;122(1):39–46.
CrossRef - Xu X, Li J, Han S, et al. A novel doxorubicin-loaded folic acid conjugated PAMAM modified with borneol, a natural dual-functional product of reducing PAMAM toxicity and boosting BBB penetration. Eur J Pharm Sci. 2016;88:178–190.
CrossRef - Jain NK, Tare MS, Mishra V, Tripathi PK. The development, characterization and in vivo anti-ovarian cancer activity of poly(propylene imine) (PPI)-antibody conjugates containing encapsulated paclitaxel. Nanomedicine. 2015;11(1):207–218.
CrossRef - Jin Y, Ren X, Wang W, et al. A 5-fluorouracil-loaded pH-responsive dendrimer nanocarrier for tumor targeting. Int J Pharm. 2011;420(2):378–384.
CrossRef - Kong X, Yu K, Yu M, et al. A novel multifunctional poly(amidoamine) dendrimeric delivery system with superior encapsulation capacity for targeted delivery of the chemotherapy drug 10-hydroxycamptothecin. Int J Pharm. 2014;465(1–2):378–387.
CrossRef - Turner JG, Dawson J, Cubitt CL, Baz R, Sullivan DM. Inhibition of CRM1-dependent nuclear export sensitizes malignant cells to cytotoxic and targeted agents. Semin Cancer Biol. 2014;27:62–73.
CrossRef - Shilling D, Stillman RC, Chang AE, Goldberg NH, Seipp CA, Barofsky I, Rosenberg SA. A prospective evaluation of delta-9-tetrahydrocannabinol as an antiemetic in patients receiving adriamycin and cytoxan chemotherapy. Cancer. 1981;47(7):1746–1751.
CrossRef - Li J, Shen M, Shi X. Poly(amidoamine) dendrimer–gold nanohybrids in cancer gene therapy: a concise overview. ACS Appl Bio Mater. 2020;3(9):5590–605. doi:10.1021/acsabm.0c00863
CrossRef - Mignani S, Shi X, Rodrigues J, Roy R, Muñoz‑Fernández A, Ceña V, Majoral JP. Dendrimers toward translational nanotherapeutics: concise key step analysis. Bioconjug Chem. 2020;31(9):2060–71. doi:10.1021/acs.bioconjchem.0c00395
CrossRef - Gosika M, Mandal T, Maiti PK. Modulating interdendrimer interactions through surface adsorption. Langmuir. 2020;36(20):5492–501. doi:10.1021/acs.langmuir.0c00208
CrossRef - Apartsin EK. Dendrimers for drug delivery: where do we stand in 2023. 2023;15(12):2740. doi:10.3390/pharmaceutics15122740
CrossRef - Zenze M, Daniels A, Singh MD. Dendrimers as modifiers of inorganic nanoparticles for therapeutic delivery in cancer. 2023;15(2):398. doi:10.3390/pharmaceutics15020398
CrossRef - An H, Deng X, Wang F, Xu P, Wang N. Dendrimers as nanocarriers for the delivery of drugs obtained from natural products. Polymers. 2023;15 (10):2292. doi:10.3390/polym15102292
CrossRef - Dhull A, Yu C, Wilmoth AH, Chen M, Sharma A, Yiu S. Dendrimers in corneal drug delivery: recent developments and translational opportunities. 2023;15(6):1591. doi:10.3390/pharmaceutics15061591
CrossRef - Tian Y, Sun M, Song R, Yang Z, Zhang H. DNA dendrimer‑based nanocarriers for targeted co‑delivery and controlled release of multiple chemotherapeutic drugs. RSC Adv. 2025;15:2981–87. doi:10.1039/D4RA07839J
CrossRef - Khan MA, Peng R, Liu CL, Chen Z. Synthesis, dynamics and applications (cytotoxicity and biocompatibility) of dendrimers: a mini‑review. Eur Polym J. 2022;181:111708. doi:10.1016/j.eurpolymj.2022.111708
CrossRef - Dockery L, Zalesak‑Kravec S, Kane MA, Daniel MC. Modular and efficient synthesis of a poly(propylene imine) (PPI) dendron applied to acid‑sensitive doxorubicin conjugation. Tetrahedron. 2022;125:133044. doi:10.1016/j.tet.2022.133044
CrossRef - Choudhury H, Pandey M, Mohgan R, et al. Dendrimer‑based delivery of macromolecules for the treatment of brain tumor. Biomater Adv. 2022;141:213118. doi:10.1016/j.bioadv.2022.213118
CrossRef - González‑Méndez I, Loera‑Loera E, Sorroza‑Martínez K, et al. Synthesis of β‑cyclodextrin‑decorated dendritic compounds based on EDTA core: a new class of PAMAM dendrimer analogs. 2022;14 (11):2363. doi:10.3390/pharmaceutics14112363
CrossRef - Singh H, Tarannum S, Sahoo RK, et al. Dendritic polymer macromolecular carriers for drug delivery. In: Singh H, Tarannum S, Sahoo RK, editors. Therapeutic Concepts and Pharmaceutical Challenges. p.289–328. doi:10.1016/B978-0-323-91248-8.00006-4
CrossRef - Mejlsøe SL, Christensen JB. Dendrimers in drug delivery. In: Mejlsøe SL, Christensen JB, editors. Drug Delivery Advances. Elsevier; 2023. p.357–87. doi:10.1016/B978-0-323-91668-4.00005-8
CrossRef - Choi S, Yoon KY, Dong G. Modular synthetic platform for interior‑functionalized dendritic macromolecules enabled by Pd/Norbornene catalysis. J Am Chem 2024;146(28):18855–60. doi:10.1021/jacs.4c06090
CrossRef - Inayama S, Hayashi S. Iterative click‑addition process for urethane‑based 1st‑5th dendrons. ACS Appl Polym Mater. 2024;6(12):7329–37. doi:10.1021/acsapm.4c01618
CrossRef - Chen S, Ma C, Xu J, et al. Subnanometric Pt–W bimetallic clusters for efficient alkaline hydrogen evolution electrocatalysis. ACS Nano. 2024;18(49):33696–33705. doi:10.1021/acsnano.4c13743
CrossRef - Weng Z, Zaera F. Synthetic strategies for adding enantioselectivity to metal‑based solid catalysts using cinchonidine. J Phys Chem 2024;128(41):17446–62. doi:10.1021/acs.jpcc.4c05485
CrossRef - Chirag M, Gowda DV, Babu S, Roohi NK. A comprehensive review on dendrimers in current advanced drug delivery. Int J Res Pharm Sci. 2020;11(1):1055–66.
CrossRef - Alfei S, Catena S, Turrini F. Biodegradable and biocompatible spherical dendrimer nanoparticles with a gallic acid shell and antioxidant activity. Drug Deliv Transl Res. 2020;10:259–70.
CrossRef - Song C, Xiao Y, Ouyang Z, Shen M, Shi X. Co‑delivery of miR‑21 inhibitor and doxorubicin via core–shell tecto dendrimers formed via supramolecular host–guest assembly. J Mater Chem B. 2020;8:2768–74.
CrossRef - Desai PN, Yuan Q, Yang H. Photocurable polyamidoamine dendrimer hydrogels platform for tissue engineering and drug delivery. Biomacromolecules. 2010;11:666–73.
CrossRef - Oiberman U, Kambhampati SP, Wu T, et al. Subconjunctival injectable dendrimer‑dexamethasone gel for corneal inflammation. Biomaterials. 2017;125:38–53.
CrossRef - Wang G, Zhu D, Zhou Z, et al. A glutathione‑specific and intracellularly labile polymeric nanocarrier for efficient and safe cancer gene delivery. ACS Appl Mater Interfaces. 2020;12:14825–38.
CrossRef - Wang J, Cooper RC, Yang H. Polyamidoamine dendrimer grafted with an acid‑responsive charge‑reversal layer for improved gene delivery. Biomacromolecules. 2020;21:4008–16.
CrossRef







