Manuscript accepted on :06-10-2025
Published online on: 14-10-2025
Plagiarism Check: Yes
Reviewed by: Dr. Murali Krishna Prasad Vallabhaneni and Dr. Aditya Vikram Jain Parmar
Second Review by: Dr. Preeti Yadav
Final Approval by: Dr. Anton R Keslav
Lakshmana Rao Atmakuri1* , Satya Sree Bandaru2
, Satya Sree Bandaru2 , Vasudha Dadi3
, Vasudha Dadi3 , Suresh Babu Madda4
, Suresh Babu Madda4 , Vijaya Kumar Ghanta5
, Vijaya Kumar Ghanta5 and Ramesh Alluri6
and Ramesh Alluri6
1Department of Pharmaceutical Analysis, V. V. Institute of Pharmaceutical Sciences, Gudlavalleru, Andhra Pradesh, India.
2Department of Pharmaceutical Chemistry, V. V. Institute of Pharmaceutical Sciences, Gudlavalleru, Andhra Pradesh, India.
3Department of Pharmaceutical Chemistry, Vignan Institute of Pharmaceutical Technology, Duvvada, Andhra Pradesh, India.
4School of Pharmacy, GIET University, Gunupur, Odisha, India.
5Department of Pharmacy Practice, KVSR Siddhartha College of Pharmaceutical Sciences, Vijayawada, Andhra Pradesh, India.
6Department of Pharmacology, Vishnu Institute of Pharmaceutical Education and Research, Narsapur, Telangana, India.
Corresponding Author E-mail: dralrao@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3289
Abstract
A robust and repeatable RP-HPLC technique has been created for the concurrent measurement of Bupivacaine as well as Meloxicam in pharmaceutical formulation. The method underwent validation following applicable regulatory guidelines. The optimized chromatographic conditions included the use of a C18 Zorbax SB column (250x4.6 mm, 5 µ), an isocratic mobile phase comprising acetonitrile and a hexane sulfonic acid (2.5 pH adjusted with ortho-phosphoric acid) in a 60:40 volume ratio. The method operated at a rate of flow 1.0 ml/min, volume of 10 µl injection, UV detection set at 260 nm, as well as an ambient temperature of 25°C, with a total runtime of 5 min. Under these conditions, Bupivacaine eluted at 2.774 min and Meloxicam at 3.494 min. System suitability parameters, including, tailing factor, plate count as well as %RSD, were within acceptable limits. The method exhibited high precision, with %RSD values below 2%, and excellent linear correlation over a range of 50-300 µg/ml for Bupivacaine as well as 1.5-9 µg/ml for Meloxicam. Sensitivity analysis determined LOD and LOQ values of Bupivacaine 0.60 µg/ml and 2.00 µg/ml, for Meloxicam 0.018µg/ml and 0.060 µg/ml respectively. Forced degradation studies showed that the method could show stability under different stress conditions, with peroxide causing the highest degradation. The validated method was applied for assay of pharmaceutical formulation confirming compliance with label claim.
Keywords
Bupivacaine; Forced degradation studies; Meloxicam; Method validation; Pharmaceutical analysis; RP-HPLC; Stability-indicating method
Download this article as:| Copy the following to cite this article: Atmakuri L. R, Bandaru S. S, Dadi V, Madda S. B, Ghanta V. K, Alluri R. A Novel RP-HPLC Method for the Quantitative Determination of Bupivacaine and Meloxicam: Stability-Indicating Approach. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Atmakuri L. R, Bandaru S. S, Dadi V, Madda S. B, Ghanta V. K, Alluri R. A Novel RP-HPLC Method for the Quantitative Determination of Bupivacaine and Meloxicam: Stability-Indicating Approach. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/3LlOKi0 |
Introduction
Bupivacaine (BUP) is an amide local anaesthetic that blocks nerve impulse conduction by inhibiting voltage-gated sodium channels.1-3 It crosses the neuronal membrane to act on the intracellular portion of these channels. The blockade is increasing with repetitive depolarization.4-6 Chemically it is 1-butyl-N-(2,6-dimethylphenyl) piperidine-2-carboxamide (Figure 1). Meloxicam (MEL) is a anti-inflammatory non-steroidal drug that particularly inhibits COX-2 over COX-1.7-9 This reduces prostaglandin synthesis, leading to decreased inflammation leads to analgesic and inflammatory effects, minimize gastrointestinal side effects.9,10 Chemically it is 4-hydroxy-2-methyl-N-(5-methyl-1,3-thiazol-2-yl)-1,1-dioxo-1,2-benzothiazine-3-carboxamide (Figure 2).10-12 The combination of Bupivacaine and Meloxicam is used for enhanced postoperative pain control.13 Bupivacaine provides local anesthesia by blocking nerve conduction, while Meloxicam reduces inflammation and pain via COX-2 inhibition.14 Together, they offer synergistic, longer-lasting analgesia with reduced opioid need.15
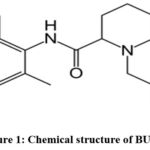 |
Figure 1: Chemical structure of BUP
|
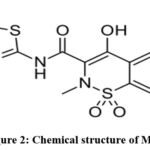 |
Figure 2: Chemical structure of MEL |
High-Performance Liquid Chromatography (HPLC) is widely used for the accurate and precise evaluation of drug concentrations in pharmaceutical formulations and biological samples.16-21 It allows for the separation, identification, and quantification of active drug components, ensuring quality control and regulatory compliance. Despite its utility, only a limited number of techniques have been reported for simultaneous RP-HPLC analysis of Bupivacaine as well as Meloxicam and these methods often fall short in terms of acceptance criteria.22,23 Prior approaches have demonstrated limitations such as inadequate specificity and sensitivity, along with prolonged retention times. Considering the growing demand for analytical procedures that are accurate, economical, and time-efficient, this study was undertaken to validate and develop a novel RP-HPLC method for the simultaneous estimation of Bupivacaine and Meloxicam as per ICH guidelines.24-26
Materials and Methods
Chemicals and reagents
BUP as well as MEL standard was provided as gift samples from Shree Icon Pharmaceuticals Laboratories, Vijayawada, India. HPLC grade acetonitrile (ACN), analytical grade ortho phosphoric acid (OPA) and hexane sulphonic acid (HSA) were obtained from Rankem Chemicals, Mumbai, India. All analytical procedures were performed using triple distilled HPLC grade water produced in-house.
Instrumentation
The analysis was conducted using a HPLC, Alliance model from Waters connected with 2998 PDA detector, e2695 model pump, as well as Empower software with 2.0version. Separations were attained on a C18 Zorbax SB column (250×4.6 mm, 5µ). For stress degradation studies, thermostatic water bath with digital controller was utilized. All weighing operations were conducted using a Sartorius analytical balance. Samples were ultrasonically processed using a Powersonic ultrasonicator.
Preparation of HSA buffer solution
Dissolve 1.8 gms of HSA in 1 litre of HPLC water and then it was adjusted to pH-2.5 with OPA then filter the mixture through membrane filter made up of nylon (0.45µ).
Preparation of mobile phase
ACN and HSA pH-2.5/OPA were combined in a 60:40 v/v ratio to develop mobile phase. In order to remove any contaminants that could impact the final chromatogram, it was filter through membrane filter made up of nylon (0.45µ).
Preparation of standard stock solution
In a 100 ml dry volumetric flask, weigh 200 mg of Bupivacaine as well as 6 mg of Meloxicam exactly. To prepare the stock solution, add the diluent to the mixture and sonicate until fully dissolved. After dissolution, bring the solution up to the 100 ml mark with the same diluent to prepare the stock solution. Finally, measure 5 ml of this prepared stock solution mentioned above into a 50 ml volumetric flask, subsequently dilute to volume with the same diluent to achieve the desired level to achieve the final concentration 200 µg/ml of Bupivacaine and 6 µg/ml of Meloxicam.
Preparation of stock solution
200 mg of Bupivacaine as well as 6 mg of Meloxicam standard should be carefully measured before being poured into volumetric flask of 100 ml that has been washed and dried. Use the same stock solution to rise up to the desired level after adding the diluent and sonicating it until it is completely dissolved.
Preparation of sample solution
Transfer 0.5 ml of the Bupivacaine and Meloxicam sample that has been precisely measured, into a dry, clean 100 ml volumetric flask. Add the designated diluent to sonicate the mixture for up to 30 minutes to dissolve it fully. Then, centrifuge the mixture for 30 minutes to ensure complete dissolution as well as adjust the volume with the same diluent. The solution was filter through a membrane filter made up of nylon (0.45µ) to obtain stock solution. From this stock solution, carefully measure 5 ml and transfer it into a 50 ml volumetric flask. Fill to the desired level with the designated to achieve the final concentration 200 μg/ml for Bupivacaine and 6 μg/ml for Meloxicam.
Method development
Using a PDA detector, the drug solution’s maximum absorption wavelength was scanned between 200 and 400 nm, with ACN and HSA pH-2.5 adjusted with OPA (60:40 v/v) acting as a blank. At 260 nm, the absorption curve exhibits an isobestic point. Hence a wavelength of 260 nm was selected. Out of 6 different trials, in the trial 6 achieved the best chromatographic separation (Figure 3&4) employing a HPLC quaternary gradient pump integrated with PDA detector, with a mobile phase of ACN: HSA pH-2.5/OPA (60:40 v/v), a Zorbax SB C18 (250×4.6 mm, 5 µ) column, in isocratic mode and UV wavelength of 260 nm. The runtime was 5 min with a rate of flow at 1 ml/min at room temperature (25°C). Under these conditions, BUP eluted at 2.774 min and a peak tailing value of 1.14, while MEL eluted at 3.494 min and a peak tailing value of 1.02, and a resolution of 3.82. This set of conditions was set optimal and had good peak resolution.
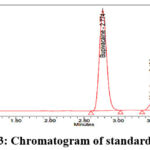 |
Figure 3: Chromatogram of standard solution |
 |
Figure 4: Chromatogram of blank |
Method validation
Linearity
Accurately weigh 200 mg of Bupivacaine and 6 mg of Meloxicam reference standards, now transfer them into a clean, dry 100 ml volumetric flask. Add the designated diluent and sonicate until fully dissolved. Once dissolved, dilute to desired volume with the same diluent to prepare the stock solution. Stock solutions containing 1.25 ml, 2.5 ml, 3.75 ml, 5 ml, 6.25 ml, as well as 7.25 ml were taken and diluted up to 50 ml in volumetric flasks to get concentration of 50 µg/ml, 100 µg/ml, 150 µg/ml, 200 µg/ml, 250 µg/ml, and 300 µg/ml, respectively. Measure the peak area after injecting each level into the chromatographic devices. The chromatograms corresponding to the linearity levels (25%–150%) are shown in Figures 5–10, while the calibration curves of Bupivacaine and Meloxicam are presented in Figures 11 and 12, respectively.
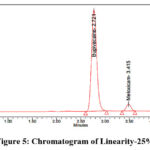 |
Figure 5: Chromatogram of Linearity-25% |
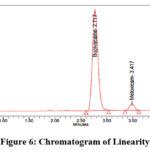 |
Figure 6: Chromatogram of Linearity-50% |
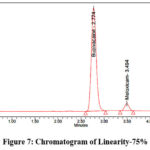 |
Figure 7: Chromatogram of Linearity-75%Click here to view Figure |
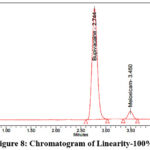 |
Figure 8: Chromatogram of Linearity-100% |
 |
Figure 9: Chromatogram of Linearity-125% |
 |
Figure 10: Chromatogram of Linearity-150% |
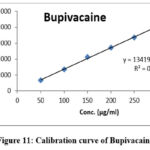 |
Figure 11: Calibration curve of Bupivacaine |
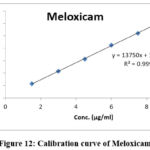 |
Figure 12: Calibration curve of Meloxicam |
Accuracy
Precisely measure and transfer 200 mg of Bupivacaine, 6 mg of Meloxicam reference standards into dry 100 ml volumetric flasks. Add an appropriate amount of diluent as well as sonicate the mixtue until fully dissolved. Dilute to volume using the same solvent to prepare the stock solution. From the stock solution, prepare solutions at 50%, 100%, and 150% concentration levels. The representative chromatograms for the accuracy studies at 50%, 100%, and 150% concentration levels are shown in Figures 13–15, respectively.
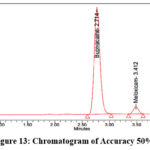 |
Figure 13: Chromatogram of Accuracy 50% |
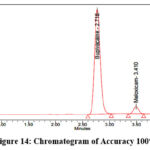 |
Figure 14: Chromatogram of Accuracy 100% |
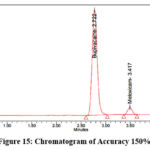 |
Figure 15: Chromatogram of Accuracy 150% |
Precision
Standard substances are used to confirm system precision and ensure the analytical system is functioning properly. It is necessary to figure out the percentage of the RSD and the peak area and drug percentage of six determinations. To evaluate method precision six replicate analyses of a homogeneous sample from a single batch must be conducted. Instrumental precision was assessed by performing six consecutive injections (n=6) of a preparation containing 200 µg/ml of Bupivacaine as well as 6 µg/ml of Meloxicam. The representative chromatogram for inter-day precision is shown in Figure 16.
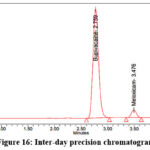 |
Figure 16: Inter-day precision chromatogram |
Robustness
It can be assessed by changing in the rate of flow from 0.9 ml/min to 1.1 ml/min a well as by modifying the ratio of the organic component within the mobile phase composition.
Ruggedness
Ruggedness was evaluated and the analyst was changed to carry out the inter-day variation of the suggested procedures at a concentration equivalent to the standard concentration.
Limit of detection (LOD) and limit of quantitation (LOQ)
Six repetitions of the standard curve preparation process were carried out using the standard serial dilutions. The average value of slope(s) and data variability of intercept were utilised to find out the limit of detection (Figure 17) and limit of quantitation (Figure 18).
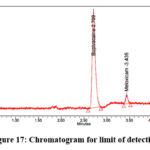 |
Figure 17: Chromatogram for limit of detection |
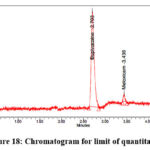 |
Figure 18: Chromatogram for limit of quantitation |
Forced degradation studies
As per the International Conference on Harmonization (ICH) Q1A(R2) guideline, Stability Testing of New Drug Substances and Products, stress testing is essential to characterize the intrinsic stability of active substances. Forced degradation studies of Bupropion (BUP) and Meloxicam (MEL) were therefore performed under acidic, alkaline, oxidative, thermal, photolytic, and hydrolytic conditions. For acidic degradation, drug solutions were treated with 0.1N HCl and maintained at 60 °C for 4 hours, while alkaline degradation was carried out using 0.1N NaOH under identical conditions. Oxidative stress was induced by exposing drug solutions to 3% hydrogen peroxide at room temperature for 4 hours, whereas thermal degradation was assessed by keeping solid drug samples in a hot air oven at 80 °C for 24 hours. Photolytic degradation was performed by exposing the drugs to UV (254 nm) and visible light according to ICH Q1B photostability guidelines, and hydrolytic degradation was studied by incubating drug solutions in distilled water at 60 °C for 24 hours. Following stress exposure, samples were neutralized where required, diluted to appropriate concentrations, and analyzed by a validated stability-indicating HPLC method. The untreated control samples exhibited 100% assay with no detectable degradation, confirming the initial stability of both compounds. Under acidic stress, BUP and MEL degraded by 14.5% and 11.1%, respectively (Figure 19), whereas alkaline conditions produced slightly lower degradation levels of 12.2% for BUP and 11.0% for MEL (Figure 20). Oxidative stress with hydrogen peroxide resulted in the highest degradation, with BUP showing 12.0% and MEL 13.8% degradation (Figure 20). In contrast, thermal degradation caused only 2.9% and 1.3% loss for BUP and MEL, respectively (Figure 21). Photolytic conditions also produced minimal degradation, with BUP at 4.8% and MEL at 1.7% (Figure 22). Hydrolytic stress had the least effect, yielding 1.2% degradation for BUP and 0.8% for MEL (Figure 23). Overall, these findings indicate that both drugs are more susceptible to acidic and oxidative conditions, while exhibiting considerable stability under thermal, photolytic, and hydrolytic stress. Such insights are valuable for developing stability-indicating analytical methods and predicting shelf life under ICH-recommended conditions.
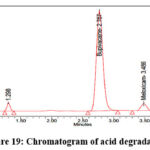 |
Figure 19: Chromatogram of acid degradation |
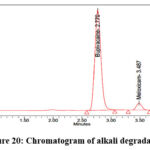 |
Figure 20: Chromatogram of alkali degradation |
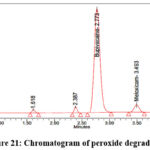 |
Figure 21: Chromatogram of peroxide degradation |
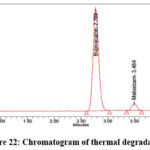 |
Figure 22: Chromatogram of thermal degradation |
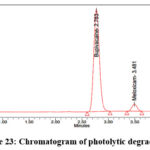 |
Figure 23: Chromatogram of photolytic degradation |
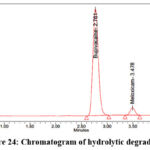 |
Figure 24: Chromatogram of hydrolytic degradation |
Assay procedure
Bupivacaine and Meloxicam peak areas are measured after injecting standard solution 10 µl into the chromatographic system. Use the relevant equations to calculate the assay percentage.
Results
Method validation
The analytical method validation for BUP and MEL was conducted in accordance with regulatory ICH guidelines. System suitability testing (Table 1) confirmed that all measured parameters fell within acceptable range, with retention times of 2.774 min for BUP as well as 3.494 min for MEL. Plate counts exceeding 2000, and %RSD values below 2%. Specificity testing demonstrated no interfering peaks at the analytes retention times, confirming the method capability to distinguish the analytes from other potential interferences. Linearity (Table 2) was formed within a range of 50-300 µg/ml for BUP and 1.5-9 µg/ml for MEL, with correlation coefficients above 0.999, confirming a proportional relationship between concentration and peak area. Precision studies were assessed with six replicate injections showing %RSD values below 2%, indicating high repeatability (Table 3). Accuracy was examined via recovery experiments at three concentration levels of 50%, 100%, and 150%, yielding mean recoveries of 100.01% for BUP (Table 4) and 100.34% for MEL (Table 5), demonstrating the method’s reliability in quantification. Robustness testing (Table 6&7) assessed the impact of variations in flow rate (±10%) and mobile phase composition (±3% organic phase), with results showing minor variations within acceptable limits, confirming the method’s robustness. Sensitivity analysis determined the LOD at 0.60 µg/ml for BUP as well as 0.018 µg/ml for MEL, while the LOQ was found to be 2.00 µg/mL for BUP and 0.060 µg/mL MEL respectively (Table 8), demonstrating the method’s capability for detecting low analyte concentration. The purity angle and purity threshold values confirmed that no major co-eluting impurities interfered with the peaks, demonstrating the stability of the proposed method (Table 9).
Table 1: System suitability study
| Name | RT (min.) | Peak area | Plate count | Tailing factor | Resolution |
| BUP | 2.731 | 11432014 | 2488 | 1.274 | — |
| MEL | 3.635 | 218116 | 6585 | 1.05 | 4.47 |
Table 2: Results of linearity for BUP&MEL
| S. No. | Bupivacaine | Meloxicam | ||
| Conc. (µg/ml) |
Area of peak | Conc. (µg/ml) |
Area of peak | |
| 1 | 50 | 669383 | 1.5 | 22624 |
| 2 | 100 | 1358638 | 3.0 | 42784 |
| 3 | 150 | 2124572 | 4.5 | 62647 |
| 4 | 200 | 2730368 | 6.0 | 85351 |
| 5 | 250 | 3350920 | 7.5 | 103646 |
| 6 | 300 | 4049679 | 9.0 | 125942 |
| Equation | y=13488.46x+17239.89 | y=13867.55x+880.89 | ||
| Slope | 13488.46 | 13867.55 | ||
| Intercept | 17239.89 | 880.89 | ||
| R2 | 0.99961 | 0.99978 | ||
Table 3: Precision results for BUP & MEL
| S. No. | Area of BUP | Area of MEL |
| 1 | 2727654 | 85123 |
| 2 | 2703216 | 85154 |
| 3 | 2746241 | 85854 |
| 4 | 2767548 | 85215 |
| 5 | 2758452 | 85974 |
| 6 | 2750543 | 85020 |
| Average | 2742275 | 85390 |
| SD | 23340.47 | 412.52 |
| %RSD | 0.85 | 0.48 |
Table 4: Accuracy results of BUP
| Concentration level | Area of peak | Amount added (µg/ml)) | Amount found(µg/ml) | %Recovery | Mean recovery |
| 50% | 1362882 | 100 | 99.58 | 99.58 | 100.01 |
| 100% | 2781021 | 200 | 203.2 | 101.6 | |
| 150% | 4059614 | 300 | 296.63 | 98.87 |
Table 5: Accuracy results for MEL
| Concentration level | Area of peak | Amount added(µg/ml) | Amount found(µg/ml) | %Recovery | Mean recovery |
| 50% | 43069 | 3 | 3.02 | 100.66 | 100.34 |
| 100% | 85564 | 6 | 6.01 | 100.16 | |
| 150% | 128465 | 9 | 9.02 | 100.22 |
Table 6: Robustness results of BUP
| Parameters | Condition | Retention time (min) | Plate count | Tailing factor | Peak area | %RSD |
| Flow rate change (ml/min) | Less flow (0.9 ml) | 3.047 | 11301 | 1.08 | 2562369 | 0.57 |
| Actual(1 ml) | 2.774 | 11324 | 1.14 | 2737401 | 0.38 | |
| More flow (1.1 ml) | 2.541 | 11394 | 1.16 | 2831487 | 0.49 | |
| Organic phase change | Less organic (54:46 v/v ) | 2.845 | 11292 | 1.04 | 2453640 | 0.47 |
| Actual (60:40 v/v) | 2.771 | 11359 | 1.17 | 2739513 | 0.32 | |
| More organic (66:34 v/v) | 2.967 | 11407 | 1.2 | 3054324 | 0.68 |
Table 7: Robustness results of MEL
| Parameters | Condition | Retentiontime(min) | Plate count | Tailing factor | Peak area | %RSD |
| Flow rate change(ml/min) | Less flow(0.9 ml) | 3.727 | 9323 | 0.98 | 83125 | 0.48 |
| Actual(1 ml) | 3.494 | 9367 | 1.02 | 85065 | 0.32 | |
| More flow (1.1 ml) | 3.323 | 9435 | 1.07 | 87462 | 0.55 | |
| Organic phase change | Less organic (54:46 v/v) | 3.631 | 9310 | 1.01 | 82742 | 0.24 |
| Actual(60:40 v/v) | 3.490 | 9361 | 1.04 | 85651 | 0.59 | |
| More organic (66:34 v/v) | 3.766 | 9458 | 1.11 | 89567 | 0.66 |
Table 8: Sensitivity parameters (LOD & LOQ)
| Drug | LOD (µg/ml) | LOQ (µg/ml) |
| Bupivacaine | 0.60 | 2.00 |
| Meloxicam | 0.018 | 0.060 |
Table 9: Stress Degradation results for BUP & MEL
| Degradation conditions | Bupivacaine | Meloxicam | ||
| Area | %Degradation | Area | %Degradation | |
| Acid | 2338219 | 14.5 | 75931 | 11.1 |
| Alkali | 2402440 | 12.2 | 75617 | 11 |
| Peroxide | 2324711 | 15.0 | 73652 | 13.8 |
| Thermal | 2654823 | 2.9 | 84373 | 1.3 |
| Photolytic | 2603047 | 4.8 | 84017 | 1.7 |
| Hydrolysis | 2701588 | 1.2 | 84754 | 8 |
Table 10: Assay results of BUP & MEL
| Brand | Drug | Average peak area (n=5) | Label amount (mg) | Amount found (mg) | %Assay |
| Zynrelef | Bupivacaine | 2741189 | 200 | 200.29 | 100.1 |
| Meloxicam | 85586 | 6 | 6.01 | 100.2 |
Assay of formulation
The assay of the formulation (Table 10), Zynrelef was performed to determine the drug content of BUP and MEL. The label claim of 200 mg for BUP and the amount found was 200.29 mg, corresponding to a %assay of 100.10%. Similarly, for MEL, the label claim was 6 mg and the amount found was 6.01 mg, yielding a %Assay of 100.20%. These results confirm that the formulation met the specified drug content requirements, ensuring its quality and consistency.
Discussion
The validated RP-HPLC method for BUP and MEL proved to be precise, accurate, specific, linear, robust, and sensitive, meeting regulatory standards. System suitability parameters were within limits, with %RSD values below 2%, ensuring repeatability. Linearity (R²>0.999) and mean recoveries (100.01% for BUP, 100.34% for MEL) confirmed method reliability. Robustness studies showed minor variations had no significant impact, and LOD and LOQ values demonstrated high sensitivity. Degradation studies confirmed the method’s stability, with the highest degradation in peroxide (15.0% BUP, 13.8% MEL), while photolytic conditions had minimal impact. Purity parameters ensured no interfering peaks. The assay of Zynrelef confirmed compliance with drug content requirements, with %Assay values of 100.14% for BUP and 100.16% for MEL.
Conclusion
An innovative, cost-effective and most stable reversed-phase HPLC method was developed as well as confirmed using notable calibrated chromatographic parameters than earlier reported techniques for simultaneous quantification of Bupivacaine as well as Meloxicam in tablets. Validation study delivers proof that the procedure is simple, robust, specific, reliable, rugged, accurate, precise & linear, meeting regulatory standards. Stability assessments indicated that the method maintained its integrity in the presence of degradation products for up to 24 hours. Therefore, the proposed HPLC method is well-suited for routine quality control of Bupivacaine as well as Meloxicam in its dosage form.
Acknowledgement
The authors are very grateful to V. V. Institute of Pharmaceutical Sciences, Gudlavalleru for proving necessary facilities for carryout the research work.
Funding Source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not applicable
Author’s Contribution
- Lakshmana Rao Atmakuri: Conceptualization, Methodology, Writing – Original Draft;
- Satya Sree Bandaru: Data Collection, Analysis;
- Vasudha Dadi: Project Administration, Writing – Review & Editing;
- Suresh Babu Madda: Funding Acquisition, Resources;
- Vijaya Kumar Ghanta: Supervision, Data Interpretation;
- Ramesh Alluri: Visualization, Final Draft Approval.
REFERENCES
- Babst CR, Gilling BN. Bupivacaine: A review. Anesth Prog. 1978;25(3):87-91.
- Juan P, Nawal K, Somayah M, Eduardo Q, Sergio B. Safety of liposome extended-release Bupivacaine for postoperative pain control. Front Pharmacol. 2014;5:90.
CrossRef - Hu D, Onel E, Singla N, Kramer WG, Hadzic A. Pharmacokinetic profile of liposome Bupivacaine injection following a single administration at the surgical site. Clin Drug Investig. 2013;33(2):109-115.
CrossRef - Blair HA. Bupivacaine/Meloxicam prolonged release: A review in postoperative pain. Drugs. 2021;81(10):1203-1211.
CrossRef - Bourn T, Serpa SM. Bupivacaine/Meloxicam ER: A new dual-acting extended-release local anesthetic for opioid-sparing postoperative pain management. Ann Pharmacother. 2022;57(1):71-85.
CrossRef - El-Malla SF, Hamza AA, Elagamy SH. Simultaneous determination of Meloxicam and Bupivacaine via novel chemometric and spectroscopic approaches. Sci Rep. 2024;14:1893.
CrossRef - Khalil NY, Aldosari KF. Meloxicam. Profiles Drug Subst Excip Relat Methodol. 2020;45:159-197.
CrossRef - Fleischmann R, Iqbal I, Slobodin G. Meloxicam. Expert Opin Pharmacother. 2002;3(10):1501-1512.
CrossRef - Turck D, Roth W, Busch U. A review of the clinical pharmacokinetics of Meloxicam. 1996;35(Suppl1):13-16.
CrossRef - Noble S, Balfour JA. Meloxicam. 1996;51(3):424-430.
CrossRef - Engelhardt G. Pharmacology of Meloxicam, a new non-steroidal anti-inflammatory drug with an improved safety profile through preferential inhibition of COX-2. Br J Rheumatol. 1996;35 Suppl 1:4-12.
CrossRef - Sternon J, Appelboom T. Meloxicam. Rev Med Brux. 1998;19(1):29-32.
- Blair HA. Bupivacaine/Meloxicam prolonged release: A review in postoperative pain. Drugs. 2021;81(10):1203-1211.
CrossRef - Bourn T, Serpa SM. Bupivacaine/Meloxicam ER: A new dual-acting extended-release local anesthetic for opioid-sparing postoperative pain management. Ann Pharmacother. 2022;57(1):71-85.
CrossRef - El-Malla SF, Hamza AA, Elagamy SH. Simultaneous determination of Meloxicam and Bupivacaine via a novel modified dual wavelength method and an advanced chemometric approach. Sci Rep.2024;14:
CrossRef - Raja T, Lakshmana Rao A. Development and validation of RP-HPLC method for the estimation of Abacavir, Lamivudine and Zidovudine in pharmaceutical dosage form. Int J PharmTech Res. 2011;3(2):852-857.
- Lakshmana Rao A, Sai Krishna K, Kiran Kumar Ch, Raja T. Simultaneous determination of Piperacillin and Tazobactum in bulk and pharmaceutical dosage forms by RP-HPLC. Int J Pharm Pharm Sci. 2011;3(2):134-136.
- Suneetha D, Lakshmana Rao A. A validated RP-HPLC method for the estimation of Quetiapine in bulk and pharmaceutical formulations. E-J Chem. 2010;7(S1):S261-S266.
CrossRef - Bhaskara Raju V, Lakshmana Rao A. Development, estimation and validation of Lisinopril in bulk and its pharmaceutical formulation by HPLC method. E-J Chem. 2012;9(1):340-344.
CrossRef - Lakshmana Rao A, Taraka Ramesh G, Rao JVLNS. Development and validation of RP-HPLC method for the estimation of Bicalutamide in pure and pharmaceutical dosage forms. Rasayan J Chem. 2009;2(2):512-515.
- Rajeswari KR, Sankar GG, Rao AL, Rao JVLNS. Development and validation of RP-HPLC method for the estimation of Aceclofenac in tablet dosage form. Indian Drugs. 2005;42(10):693-695.
- Stefi KS, Padmavathi S, Rahaman SA, Shabana SA, Grace SD, Sahana MA. Development and validation of stability indicating assay for simultaneous determination of Bupivacaine and Meloxicam in bulk and pharmaceutical formulations by using RP-HPLC method. Int J Life Sci Pharma Res. 2022;12(6):117-131.
CrossRef - Bahgat EA, Hashem H, Saleh H, Kamel EB, Eissa MS. Stability-indicating HPLC-DAD and TLC-densitometry methods for the quantification of Bupivacaine and Meloxicam in their co-formulated mixture. Microchem J. 2023;190:108683.
CrossRef - ICH Harmonised tripartite guideline. Validation of analytical procedures: text and methodology. Q2(R1), Geneva. 2005;1-13.
- ICH Harmonised tripartite guideline. Stability testing of new drug substances and products. Q1A(R2), Geneva. 2003;1-18.
- ICH Harmonised tripartite guideline. Photostability testing of new drug substances and products. Q1B, 1996;1-12.
Abbreviations
COX- Cyclooxygenase
HPLC-High Performance Liquid Chromatography
LOD-Limit of Detection
LOQ-Limit of Quantitation
PDA-Photodiode Array
RSD-Relative Standard Deviation
UV-Ultraviolet







