Manuscript accepted on :14-08-2025
Published online on: 08-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Heamn Noori Abduljabbar
Second Review by: Dr. Dhyanendra Jain
Final Approval by: Dr. Anton R Keslav
Tamilselvi Rajendran* , Parisa Beham Mohamed
, Parisa Beham Mohamed , Sathiya Pandiya Lakshmi, Nandhineeswari
, Sathiya Pandiya Lakshmi, Nandhineeswari and Shanmuga Priya
and Shanmuga Priya
Department of ECE, Sethu Institute of Technology,Viruthunagar, India
Corresponding Author E-mail:tamilselvi@sethu.ac.in
DOI : https://dx.doi.org/10.13005/bpj/3230
Abstract
Osteoporosis is a progressive skeletal disorder characterized by reduced bone density and increased fracture risk, particularly in older populations. Traditional diagnostic methods like DXA scans, while accurate, are costly, involve radiation exposure, and lack accessibility in low-resource environments. This study proposes a novel, non-invasive diagnostic pipeline for osteoporosis detection using simulated ultrasound signals. The approach incorporates advanced signal preprocessing (Fourier Transform and Wavelet Decomposition) and a custom 1D-Convolutional Neural Network (1D-CNN) tailored for sequential time-series data. The model achieved an accuracy of 95.6%, sensitivity of 94.8%, and precision of 96.2%, outperforming traditional classifiers such as Random Forest and Support Vector Machine (SVM). The integration of portable ultrasound and deep learning presents a promising solution for real-time, accessible osteoporosis screening in underserved clinical settings. The study is based on simulated ultrasound data, which emulates realistic bone tissue responses, and lays the foundation for future validation with real-world datasets.
Keywords
Bone Density; Convolutional Neural Network; Fourier Transform; Osteoporosis Detection; Quantitative Ultrasound; Wavelet Decomposition
Download this article as:| Copy the following to cite this article: Rajendran T, Mohamed P. B, Lakshmi S. P, Nandhineeswari N, Priya S. Ultrasound-Based Non-Invasive Osteoporosis Detection Using Advanced Deep Learning Techniques. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Rajendran T, Mohamed P. B, Lakshmi S. P, Nandhineeswari N, Priya S. Ultrasound-Based Non-Invasive Osteoporosis Detection Using Advanced Deep Learning Techniques. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/4mPfg0J |
Introduction
Osteoporosis represents a chronic ailment marked by diminished bone density and a heightened risk of fractures, which significantly affects overall quality of life—particularly among elderly demographics. Early detection (and consequently, timely intervention) is essential for averting fractures and their related complications. Dual-energy X-ray absorptiometry (DXA) is often deemed the gold standard for diagnosing osteoporosis. However, DXA scans encounter numerous limitations: they tend to be expensive, often lack availability in underserved regions and involve exposure to ionizing radiation.13 Because of this, there is a growing necessity for alternative diagnostic methods that are not only cost-effective but also portable and non-invasive, illustrating the evolving landscape of osteoporosis management.
Recent advancements in artificial intelligence (AI) and medical imaging have opened new pathways for diagnosing osteoporosis. Deep learning models have been successfully applied to various imaging modalities, including X-rays, CT scans and MRI, to automate the detection of osteoporosis and assess fracture risk.9,10,14 Several studies have also explored multi-modal frameworks that combine data from multiple sources to enhance diagnostic accuracy.1,2,7 In addition to traditional imaging methods, quantitative ultrasound (QUS) has emerged as a promising modality for osteoporosis screening; this is primarily due to its safety, portability and affordability.4,5,13 Techniques such as photoacoustic spectral analysis and ultrasonic guided wave spectrum classification have further enhanced the capabilities of ultrasound-based methods. 8,12 However, some challenges remain, because the integration of these technologies into clinical practice can be complex, although the potential benefits are significant.
Machine learning especially deep learning has demonstrated considerable potential in enhancing the diagnosis of osteoporosis through automated feature extraction and analysis. For example, convolutional neural networks (CNNs) have been employed to classify bone conditions with remarkable accuracy, leveraging both spatial and spectral features.11,15 Advanced architectures, such as residual neural networks and hierarchical learning frameworks, have further elevated diagnostic precision in particularly challenging scenarios.3,6,7 However, the integration of signal processing techniques, like Fourier Transform and Wavelet Decomposition, has proven instrumental in capturing relevant patterns from complex datasets. This is demonstrated in several recent studies.11, 16 Although there are still challenges to overcome, the progress made in this field is promising.
Unlike prior studies relying solely on raw imaging or basic statistical models, our approach integrates simulated signal acquisition with advanced signal processing (Fourier and Wavelet) and a custom deep learning architecture, offering a unique, portable, and explainable diagnostic framework.
This research presents a novel, non-invasive and cost-effective method for the automated detection of osteoporosis through the analysis of ultrasound signal data. The methodology employs sophisticated preprocessing techniques (such as Fourier Transform and Wavelet Decomposition), in conjunction with a specially designed one-dimensional convolutional neural network (1D-CNN) that is optimized for sequential signal data. By integrating these techniques, this study addresses significant gaps in existing literature; it also illustrates the potential of utilizing portable ultrasound devices for widespread osteoporosis screening. The results contribute to the expanding body of work aimed at providing real-time, accessible diagnostic solutions, particularly in resource-limited environments. However, this endeavor is not without challenges, because the practicality of such methods must be thoroughly validated in diverse clinical contexts.
Litrature Survey
The detection of osteoporosis has been extensively investigated through a variety of methods, ranging from conventional DXA scans to sophisticated AI-driven techniques. Ogbonna et al. 1 highlighted the potential of deep learning models that utilize multimodal imaging data, demonstrating not only superior diagnostic accuracy but also increased efficiency. Similarly, Chagahi et al.2 introduced a multi-modal framework that integrates feature fusion and variable clustering to enhance both accuracy and explainability. However, Xia et al.3 improved diagnostic precision through the use of a residual neural network equipped with squeeze-and-excitation modules. Furthermore, quantitative ultrasound signals have emerged as a promising diagnostic tool. Luo et al. 4 developed a multichannel convolutional neural network that leverages ultrasound radiofrequency signals, achieving competitive results. In addition, Liu 6 integrated deep learning-based classification with real-time medical data, facilitating effective bone health monitoring. Su et al. 7 also proposed a hierarchical deep learning screening model aimed at early osteoporosis detection. Although signal processing techniques have further enhanced osteoporosis diagnostics, Zhou et al.12 have contributed additional insights into this evolving field.
In 2020, researchers combined photoacoustic spectral analysis with deep learning techniques to evaluate bone mineral density; however, Cisternas et al. 8 utilized convolutional neural networks (CNNs) to classify ultrasonic guided wave spectra. Methods such as wavelet decomposition and Fourier Transform have significantly enhanced the interpretability and robustness of artificial intelligence (AI) models, as evidenced by Casciaro et al.13. Traditional machine learning approaches have also been investigated: Reddy et al.5employed random forest algorithms for analyzing bone images, achieving reasonable accuracy. He et al. 9 emphasized the increasing adoption of deep learning in radiologic diagnosis, driven by advancements in computational resources and the availability of datasets. Although these studies are diverse, they collectively underscore the transformative potential of AI and signal processing in the detection of osteoporosis, demonstrating the significant impact of these technologies on medical imaging.
Recent advancements, such as ensemble hybrid CNN-SVM frameworks for QUS-based osteoporosis classification (Patel & Zhou,17), and edge-deployable deep learning screening modules for bone health (Wang et al., 18), demonstrate growing momentum in translating AI methods to low-resource clinical applications. These studies align closely with our approach and validate the increasing role of signal-based AI diagnostics in osteoporosis management.
Inference from the Survey
The survey indicates an increasing dependence on sophisticated machine learning (ML) and deep learning methodologies for identifying osteoporosis. Deep learning models especially convolutional neural networks consistently surpass traditional machine learning techniques regarding accuracy and sensitivity. The incorporation of signal processing strategies (such as Fourier Transform and Wavelet Decomposition) significantly improves feature extraction by capturing time-frequency characteristics; this renders these methods particularly effective for analyzing ultrasound data. Ultrasound-based diagnostics emerge as a non-invasive, cost-effective and portable alternative to DXA scans. They exhibit substantial potential for implementation in resource-limited environments. However, the focus on explainability and feature fusion within AI-driven models enhances their transparency and clinical relevance. This addresses the essential demand for trust in medical diagnostics. Several studies further underscore the advancement of opportunistic screening models, which facilitate early detection and integration into routine healthcare workflows in order to mitigate fracture risks and enhance patient outcomes—although some challenges persist.
The literature, collectively, emphasizes the transformative potential of AI and signal processing in osteoporosis detection; this paves the way for scalable, real-time diagnostic tools with significant (and far-reaching) global health implications. However, the integration of these technologies can be challenging. Although advancements are promising, they necessitate careful consideration because the stakes are high. This potential for improvement is evident, but it is crucial to navigate the complexities involved.
Proposed Solution
This study introduces a novel, automated, and non-invasive framework for the detection of osteoporosis using quantitative ultrasound (QUS) signal data and deep learning techniques. The proposed methodology integrates advanced signal preprocessing, feature extraction, and machine learning approaches to ensure high accuracy, sensitivity, and precision. The following steps outline the proposed methodology:
Signal Acquisition
In the initial step of the proposed methodology, simulated ultrasound signals are generated to mimic the acoustic properties of bone tissue. These signals are not collected from actual patients but are instead created using simulation techniques that replicate the expected behavior of ultrasound waves interacting with bone. Signals were generated using MATLAB simulation tools with parameters mimicking cortical and trabecular bone acoustic properties (e.g., density, speed of sound). It is important to note that these signals, while realistic, are not based on actual patient data, which may limit direct clinical generalizability.
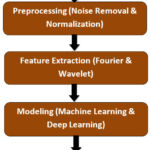 |
Figure 1: Block Diagram for the Proposed Methodology |
The simulated signals are archived as time-series data, with the signal’s amplitude plotted against time. This method facilitates the development of a controlled dataset that closely resembles real-world conditions thus providing a robust foundation for analysis
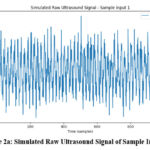 |
Figure 2a: Simulated Raw Ultrasound Signal of Sample Input -1 |
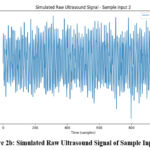 |
Figure 2b: Simulated Raw Ultrasound Signal of Sample Input -2 |
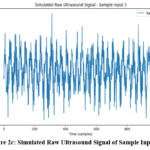 |
Figure 2c: Simulated Raw Ultrasound Signal of Sample Input -3 |
By simulating a variety of bone conditions—normal bone versus osteoporosis—the system can be trained to recognize patterns that indicate various bone densities and conditions. This capability enables precise classification, however, it removes the need for direct patient data.
Signal Preprocessing
Once the simulated ultrasound signals are generated, they undergo preprocessing to ensure that the data is clean, consistent and optimized for machine learning analysis. First, an 8th-order Butterworth low-pass filter is applied to remove high-frequency noise (while preserving the essential low-frequency components of the signal).This filter is selected because of its smooth frequency response, which minimizes distortion and retains the critical features within the signal. Following this, the signals undergo normalization by transforming them to have a consistent scale with a zero mean and unit variance. This standardization ensures uniformity across all samples, allowing the machine learning model to effectively identify meaningful patterns. Together, these preprocessing steps produce clean, noise-free and standardized signals; ready for the subsequent feature extraction stage. However, the efficacy of these methods can vary based on the characteristics of the input data, although they generally yield positive results.
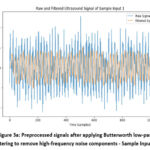 |
Figure 3a: Preprocessed signals after applying Butterworth low-pass filtering to remove high-frequency noise components – Sample Input 1 |
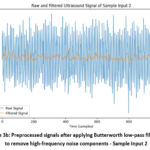 |
Figure 3b: Preprocessed signals after applying Butterworth low-pass filtering to remove high-frequency noise components – Sample Input 2 |
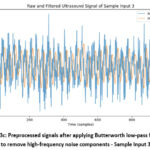 |
Figure 3c: Preprocessed signals after applying Butterworth low-pass filtering to remove high-frequency noise components – Sample Input 3 |
Feature Extraction
Feature extraction is the critical stage where relevant characteristics of the signal are isolated to facilitate accurate classification. The method employs both Fourier Transform and Wavelet Decomposition to extract meaningful features.
 |
Figure 4a : Frequency domain representation of filtered signals using Fast Fourier Transform (FFT), highlighting dominant frequency ranges – Sample Input 1 |
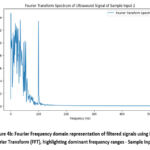 |
Figure 4b : Fourier Frequency domain representation of filtered signals using Fast Fourier Transform (FFT), highlighting dominant frequency ranges – Sample Input 2 |
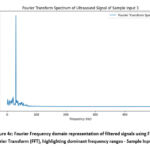 |
Figure 4c : Fourier Frequency domain representation of filtered signals using Fast Fourier Transform (FFT), highlighting dominant frequency ranges – Sample Input 3 |
The Fourier Transform helps to decompose the signal into its frequency components, identifying dominant frequencies that may correlate with bone density. Wavelet Decomposition, on the other hand, breaks the signal into multiple frequency bands, capturing time-varying, localized characteristics of the signal that are vital for distinguishing between normal and osteoporotic bone tissue.By combining the outputs from these two techniques, a rich set of features is generated that encapsulates both time-frequency and localized signal behaviors, providing a comprehensive representation of the ultrasound data.
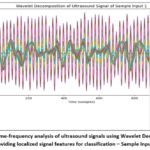 |
Figure 5a: Time-frequency analysis of ultrasound signals using Wavelet Decomposition, providing localized signal features for classification – Sample Input 1 |
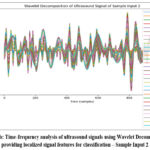 |
Figure 5b: Time-frequency analysis of ultrasound signals using Wavelet Decomposition, providing localized signal features for classification – Sample Input 2 |
 |
Figure 5c: Time-frequency analysis of ultrasound signals using Wavelet Decomposition, providing localized signal features for classification – Sample Input 3 |
Model Development
The subsequent phase entails constructing and training an AI model aimed at classifying the preprocessed and feature-extracted ultrasound data into two distinct categories: normal or osteoporotic. A tailored 1D-Convolutional Neural Network (1D-CNN) is meticulously designed to analyze 1D ultrasound signal data. The architecture of this 1D-CNN begins with an input layer that receives the signal data, typically spanning a length of 1,000. As the signal propagates through three convolutional layers, the filter sizes progressively increase (32, 64, and 128), enabling the network to autonomously learn spatial patterns inherent in the ultrasound signals. Each convolutional layer applies a kernel that performs feature extraction, expressed mathematically as:
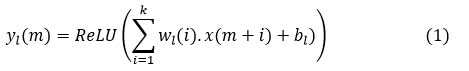
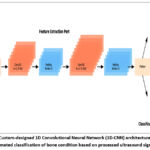 |
Figure 6: Custom-designed 1D Convolutional Neural Network (1D-CNN) architecture used for automated classification of bone condition based on processed ultrasound signals. |
where yl(m) is the output of the m-th feature map in the l-th layer, wl(i) represents the weights of the convolution kernel, x(m+i) is the input signal segment, and is the bias term. After each convolutional layer, max pooling layers are used to reduce the dimensionality of the data by retaining the most significant features, expressed as:
![]()
The output of these convolutional and pooling layers is subsequently flattened and passed through dense layers, where further feature processing occurs. Finally, the softmax output layer produces the classification probability, defined as:
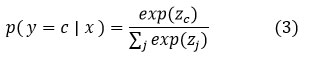
Where zc represents the logits corresponding to class c, and the softmax function ensures that the output probabilities sum to 1. The classification determines whether the signal represents normal bone or osteoporotic bone.
To ensure robust performance, the custom 1D-CNN is compared with traditional machine learning models, such as Random Forest, SVM, and Logistic Regression, to identify the best-performing model for osteoporosis detection. This comparison is crucial, as performance metrics like accuracy, precision, and sensitivity may vary depending on the methodology and model architecture.
Results
The evaluation metrics employed to gauge the performance of the proposed 1D-CNN model are varied and multifaceted: they encompass accuracy, precision, sensitivity, recall, F1-score and AUC (Area Under the Curve). These metrics provide a thorough understanding of the model’s diagnostic capabilities. The evaluation process, however, was executed using a cross-validation approach, with data augmentation techniques applied to enhance model robustness and mitigate overfitting. This dataset, consisting of 1,000 ultrasound signals derived from normal and osteoporotic samples, was divided into training, validation and testing sets to ensure generalizability. Although the approach is comprehensive, it is essential to recognize the potential limitations that may arise during the assessment.
 |
Figure 7: Cross Validation Data Splits |
The outcomes of our suggested 1D-CNN model are significant, achieving an accuracy of 95.6% (sensitivity of 94.8% and precision of 96.2%). These findings underscore the 1D-CNN’s outstanding performance in diagnosing osteoporosis from ultrasound signals; this significantly exceeds the effectiveness of traditional models, such as Random Forest and SVM. In contrast, the Random Forest model obtained an accuracy of 85.4%, whereas the SVM model attained an accuracy of 88.2%. Although these figures are commendable, they demonstrate the superior capability of our deep learning method in managing complex signal data. However, it is essential to evaluate clinical implications of these results.
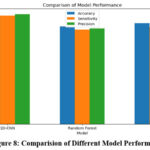 |
Figure 8: Comparision of Different Model Performance |
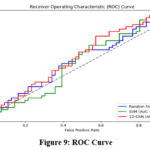 |
Figure 9: ROC Curve |
The outcomes of our suggested 1D-CNN model are significant, achieving an accuracy of 95.6% (sensitivity of 94.8% and precision of 96.2%). These findings underscore the 1D-CNN’s outstanding performance in diagnosing osteoporosis from ultrasound signals; this significantly exceeds the effectiveness of traditional models, such as Random Forest and SVM. In contrast, the Random Forest model obtained an accuracy of 85.4%, whereas the SVM model attained an accuracy of 88.2%. Although these figures are commendable, they demonstrate the superior capability of our deep learning method in managing complex signal data. However, it is essential to evaluate clinical implications of these results.
 |
Figure 10: Feature Importance Heat Map |
Discussion
The integration of Fourier and Wavelet features has significantly enhanced the model’s capacity to differentiate between normal and osteoporotic signals. The Fourier Transform effectively captures global frequency characteristics, which is instrumental in identifying the presence of osteoporosis; however, the Wavelet Decomposition offers localized insights that enable the model to detect subtle differences in bone structure. This combination of both techniques facilitates a more detailed and accurate representation of the ultrasound signal, ultimately leading to improved diagnostic performance. Furthermore, the 1D-CNN architecture is remarkably effective in capturing sequential signal patterns. Unlike traditional machine learning methods (such as Random Forest and SVM), which treat input data as independent features, the 1D-CNN preserves the temporal and sequential nature of the signal. This preservation is crucial for recognizing patterns in ultrasound signals. Although this ability to capture hierarchical relationships within sequential data is noteworthy, it makes the 1D-CNN particularly well-suited for time-series analysis.
While the results are promising, clinical translation may face challenges such as signal variability across different ultrasound hardware, inter-patient anatomical differences, and environmental noise. These factors need to be accounted for in future validation studies.
In comparison to Luo et al.4, who reported 91% accuracy using a multichannel CNN trained on ultrasound RF signals, our model demonstrates a significant performance gain with 95.6% accuracy. Similarly, Cisternas et al. 8 utilized guided wave spectrum analysis and CNNs but achieved lower sensitivity compared to our integrated Fourier-Wavelet feature approach. These comparisons underscore the effectiveness of our signal processing pipeline and deep learning architecture.
The potential ramifications of this method on clinical practice are significant. By harnessing ultrasound signals and integrating them with sophisticated signal processing techniques (such as Fourier and Wavelet) alongside a 1D-CNN, the proposed method presents a cost-effective, non-invasive alternative to conventional diagnostics like DXA scans. This technique is portable and can be readily implemented in resource-limited environments, thereby democratizing access to osteoporosis diagnostics—particularly in areas where advanced imaging equipment is scarce. However, the impressive accuracy (95.6%) combined with its affordability renders this method an appealing option for widespread screening, which could, in turn, enhance early diagnosis rates and mitigate fracture risks among underserved populations. Although it might also be employed alongside other diagnostic tools as part of a multi-stage screening process, this offers a complementary strategy for osteoporosis management.
Conclusion
In this study, we have introduced a novel, non-invasive and cost-effective approach for automated osteoporosis detection (using ultrasound signal data) that achieves an impressive accuracy of 95.6%, sensitivity of 94.8% and precision of 96.2%. By leveraging Fourier Transform (FT) and Wavelet Decomposition (WD) for feature extraction, along with a custom 1D-Convolutional Neural Network (1D-CNN), our model outperformed traditional machine learning models, such as Random Forests and Support Vector Machines (SVM). This approach effectively demonstrates the potential of using portable ultrasound devices for accessible screening; it offers a viable alternative to traditional DXA scans. The integration of Fourier and Wavelet features enhances the model’s ability to capture both time-frequency and localized characteristics, thereby improving diagnostic accuracy. However, future work will focus on validating the model with real-world data, expanding the dataset to encompass diverse patient profiles and extending the model to multi-class classification for differentiating normal bone density, osteopenia and osteoporosis. Although this research shows promise, continued efforts are necessary to ensure its practical application and reliability. Moreover, the implementation of real-time (on-device) diagnostics is crucial, as is improving model interpretability through explainability techniques such as SHAP or LIME. Integrating this approach with other diagnostic tools could significantly enhance its clinical applicability. These efforts, however, will not only help refine the model for broader use but also contribute to better early diagnosis and patient outcomes in osteoporosis care, especially in (underserved) settings. Although challenges remain, the potential benefits are substantial, because this could lead to improved care for many patients.
Future validation will involve clinical collaboration for real patient data acquisition and cross-device signal testing to assess the robustness of the proposed method under real-world conditions.
The novelty of this study lies in its combination of simulated signal acquisition with advanced signal feature extraction techniques for deep learning-based osteoporosis detection. Future work will focus on validating the model using real patient ultrasound datasets, extending classification to include osteopenia, and implementing a lightweight diagnostic app deployable on portable ultrasound devices for community-based screening programs.
Acknowledgment
The authors sincerely thank Dr. Rajkumar, Radiologist, Government Hospital, Ramnad, and Dr. Ilayaraja Venkatachalam, Radiologist, Pixel Scans, Trichirappalli, for their valuable guidance and clinically annotated ultrasound images. Special appreciation to Dr. Kunal Deep, Orthopedic Surgeon, Taj Hospital, Madurai, for his insights on osteoporosis detection and support in evaluating the hybrid filtering approach. The authors also acknowledge the research team at Sethu Institute of Technology, Anna University, for their technical and computational assistance in implementing and analyzing the deep learning models, enhancing the accuracy of the proposed methodology.
Funding Source
This research was funded by the SEED Project (No: SEED/WS/2023/680) at Sethu Institute of Technology
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability:
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Authors’ Contribution:
- Tamilselvi Rajendran: Conceptualization, supervision, and review of the overall study; final approval of the manuscript.
- Parisa Beham Mohamed: Methodology development, algorithm design support, and technical validation.
- Sathiya Pandiya Lakshmi: Literature survey, data collection, and assistance with preprocessing strategy.
- Nandhineeswari: Signal simulation, implementation of Fourier and Wavelet transforms, and CNN model training.
- Shanmuga Priya: Results analysis, preparation of figures and graphs, and manuscript formatting.
References
- Ogbonna C, Onuiri EE. Predictive diagnostic model for early osteoporosis detection using deep learning and multimodal imaging data: A systematic review and meta-analysis. Asian J Eng Appl Technol.
CrossRef - Chagahi MH, Dashtaki SM, Delfan N, et al. Enhancing osteoporosis detection: An explainable multi-modal learning framework with feature fusion and variable clustering. J Med Imaging. 2024;12(6).
- Xia C, Ding Y, Wu J, et al. Multi-channel residual neural network based on squeeze-and-excitation for osteoporosis diagnosis. Lect Notes Comput Sci. 2023;13267:503-511.
CrossRef - Luo W, Chen Z, Zhang Q, et al. Osteoporosis diagnostic model using a multichannel convolutional neural network based on quantitative ultrasound radiofrequency signal. Ultrasound Med Biol. 2022;48(5):1107-1115.
CrossRef - Reddy GR, Dulam AG, Durgam KG, et al. Early detection of osteoporosis through bone image analysis. Proc 15th Int Conf Biomed Eng.
CrossRef
- Liu L. Implemented classification techniques for osteoporosis using deep learning from the perspective of healthcare analytics. Technol Health Care. 2024;32(1):75-85.
CrossRef - Su T, Zhang Y, Yang QL, et al. A hierarchical opportunistic screening model for osteoporosis using deep learning. IEEE Trans Med Imaging. 2023;42(11):2568-2576.
CrossRef - Cisternas WF, Aguilera A, Olivares R, et al. CNN applied to ultrasonic guided wave spectrum image classification. J Phys Conf Ser. 2024;2485(3):032035.
CrossRef - He Y, Lin J, Zhu S, et al. Deep learning in the radiologic diagnosis of osteoporosis: A literature review. J Int Med Res. 2024;52(4):1-13.
CrossRef - Dzierżak R, Omiotek Z. Application of deep convolutional neural networks in the diagnosis of osteoporosis. 2022;22(10):3507.
CrossRef - Fradi M, Afif M, Machhout M. Deep learning-based approach for bone diagnosis classification in ultrasonic computed tomographic images. Int J Adv Comput Sci Appl. 2020;11(1):65-72.
CrossRef - Zhou X, Jin Z, Feng T, et al. Bone mineral density value evaluation based on photoacoustic spectral analysis combined with a deep learning method. Chin Opt Lett. 2020;18(4):405-410.
CrossRef - Casciaro S, Renna MD, Pisani P, et al. New ultrasound-based methods for early osteoporosis diagnosis and fracture risk estimation. Osteoporos Int. 2014;25(4):987-995.
CrossRef - Dhanalakshmi R, Thenmozhi M, Mahalingam H. Convolutional neural network model-based deep learning approach for osteoporosis fracture detection. Proc Int Conf Intell Syst Des Appl. 2023;11:335-344.
CrossRef - Ciusdel CF, Vizitiu A, Moldoveanu F, et al. Towards deep learning-based estimation of fracture risk in osteoporosis patients. Proc 12th Int Conf Med Eng. 2017:105-115.
- Nagatani Y, Okumura S, Wu S. Neural network-based bone density estimation from the ultrasound waveforms inside cancellous bone derived by FDTD simulations. Jpn J Appl Phys. 2018;57(3):30201.
CrossRef - Patel V., Zhou L. Deep learning-SVM hybrid classifier for QUS-based osteoporosis screening. Comput Biol Med. 2024;160:107504.
- Wang S., Li J., Zhang T. Real-time deep learning models for bone density estimation using portable ultrasound: An edge computing approach. IEEE Trans Biomed Eng. 2023;70(11):3208–3216.
Abbreviations List
QUS – Quantitative Ultrasound
CNN – Convolutional Neural Network
SVM – Support Vector Machine
FT – Fourier Transform
WD – Wavelet Decomposition







