Manuscript accepted on :09-05-2025
Published online on: 18-07-2025
Plagiarism Check: Yes
Reviewed by: Dr. Hany Akeel
Second Review by: Dr. Pravinkumar Darji
Final Approval by: Dr. Prabhishek Singh
Rama Rao Nadendla1* , Srismitha Settipalli2
, Srismitha Settipalli2 , Sovan Bagchi3
, Sovan Bagchi3 , Gunamoni Das4
, Gunamoni Das4 , Tamalika Chakraborty5
, Tamalika Chakraborty5 , Tabinda Hasan6
, Tabinda Hasan6 , Kumaraswamy Dabburu7
, Kumaraswamy Dabburu7 and Suria Prabha Kannaiyan8
and Suria Prabha Kannaiyan8
1Department of Pharmaceutics, Chalapathi Institute of Pharmaceutical Sciences, Chalapathi Nagar, Lam, Guntur, Andhrapradesh, India.
2Department of Pathology, Sree Balaji Medical College and Hospital, Chennai, India.
3Department of Biomedical Sciences, Gulf Medical University, Ajman, UAE.
4Programme of Botany, Faculty of Science, Assam down town University, Sankar Madhab Path, Gandhi Nagar, Panikhaiti, Guwahati, Assam, India.
5Department of Life Science, Guru Nanak Institute of Pharmaceutical Science and Technology, Kolkata West Bengal, India.
6Department of Anatomy, Princess Nourah bint Abdur Rahman University, KSA.
7Department of Pharmacology, Bridgetown International University, Barbados.
8Department of Pharmaceutical Sciences, Chettinad School of Pharmaceutical Sciences, Chettinad Hospital and Research Institute, Chettinad Academy of Research and Education, Kelambakkam, Tamil Nadu, India.
Corresponding Author E-mail: bagchisovan60@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3213
Abstract
An excess of calories impairs visceral adipose tissue functioning, including heart adipose and perivascular-adipose-tissue (PAT). The secretory activity of adipose tissue (AT), which encompasses both pro-inflammatory and endocrine mediators, plays a pivotal role in inter-organ communication and contributes significantly to the pathogenesis of obesity. Excessive fat in visceral adipocytes hinders the endocrine and immunological response, resulting in pathological alterations and presenting significant threats to human health risk. Previous research has established that increased pro-inflammatory cytokines and decreased immune-modulatory disrupt the communication between vasculature, liver, and myocyte. Adiposopathy or dysfunctional AT contributes to the development of cardio-metabolic-risk factors (CMRF). These risk factors include metabolic resistance, hypertension, dyslipidemia, atherosclerosis, and a hypercoagulable state. These risk factors consecutively, unswervingly, and circuitously contribute to the growth of cardiometabolic disorders. The perivascular adipose tissue envelops most blood arteries throughout the body. Functional PAT has anticontractile and immune-modulatory properties. However, it has been proposed that impaired PAT activity may contribute to the association between CMRF and vascular dysfunction. Vascular oxidative stress is a critical contributor to the progression of cardiometabolic disorders associated with obesity, type 2 diabetes, and hypertension. Increasing evidence from both human and animal studies suggests that physical exercise supports the normalization of adipocyte size and promotes the browning of white adipose tissue (WAT). This review covers the existing studies concerning the impact of adiposopathy on cardiometabolic disorders and the significance of physical activity in reinstating adipocyte function. Additionally, it explores the processes that connect PAT to age-related vascular pathologies.
Keywords
Adipose Tissue; Age-Related Vascular Illnesses; Cardiometabolic; Cardio-Metabolic-Risk-Factors; Perivascular Adipose Tissue; White Adipose-Tissue
Download this article as:| Copy the following to cite this article: Nadendla R. R, Settipalli S, Bagchi S, Das G, Chakraborty T, Hasan T, Dabburu K, Kannaiyan S. P. Perivascular Adipose Tissue: Implications for Cardiometabolic Diseases. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Nadendla R. R, Settipalli S, Bagchi S, Das G, Chakraborty T, Hasan T, Dabburu K, Kannaiyan S. P. Perivascular Adipose Tissue: Implications for Cardiometabolic Diseases. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/46OUUzl |
Introduction
Hippocrates, the Greek physician from 460-377 BC, extensively documented the detrimental effects on overall health when individuals consume excessive food or calories without engaging in physical activity to counterbalance this surplus.1 According to the quote, when food is consumed and exercise is done, the body is filled and emptied. The ideal balance between these two activities results in the body remaining in its original perfect health condition. In modern decades, there has been a cumulative amount of research highlighting the same fact that the difference between the amount of energy consumed and the amount of energy expended is the cause of excessive body weight. Excessive use of energy and its storage above the body’s natural limit might disrupt the balance of fat accumulation in the body.2
The excessive buildup of fat and lack of physical activity contribute to the progress of several illnesses, including high cholesterol levels, obesity, heart disease, high blood pressure, and diabetes. The WAT plays a crucial function in maintaining energy balance.3 Triglycerides (TG) are stored as additional energy and free fatty acids are released when there is a lack of energy.4 A Previous study has identified two primary repositories of WAT: hypodermal AT and visceral AT.5 Visceral AT, comprising 21% of total body fat, is located in proximity to internal organs, including the Peritoneal fat, the perivisceral fat near the alimentary canal, retroperitoneal fat near the epicardial fat, kidney and perivascular fat encircling blood vessels.6 Genetically or environmentally sensitive people have functional impairment of adipocytes due to the excessive fat accretion in visceral adipocytes. In recent years, there has been a significant focus on adiposopathy, which refers to the pathological condition of adipose tissue similar to conditions such as encephalopathy, cardiomyopathy, enteropathy, retinopathy, neuropathy, and myopathy. Adipocytes that are unusually big have functional abnormalities that directly and indirectly contribute to the progress of cardiometabolic disorders. Interrelated local, hormonal, and neurological systems modulate vascular tone. The local control plays a dynamic role in regulating blood pressure and delivering nutrients to peripheral tissues. All layers of vascular cells actively participate in this process. PAT is the AT that surrounds various vascular beds, excluding capillaries and pulmonary and cerebral blood vessels.7 PAT is now emerging as a central content in vascular biology. In addition to providing anatomical support for blood vessels, PAT is now widely recognized for its paracrine/endocrine characteristics. It significantly influences vascular homeostasis and function, exerting beneficial and harmful effects on the blood vessels depending on the specific pathology. Healthy PAT positively impacts muscle contraction by releasing substances that promote relaxation, such as adiponectin, angiotensin 1.5-7, H2S, nitric oxide, and palmitic acid methyl esters.8 Furthermore, adipocytokines generated from PAT consist of adiponectin, leptin, visfatin, omentin, chemerin, and resistin. The primary interactive hazard factors for cardiovascular acute events, which elevate cardiometabolic risk, are an unnatural diet, tobacco use, physical slothfulness, and problematic alcohol use.9 Although it may be avoided, obesity is a widespread problem that increases the likelihood of emerging cardiometabolic problems such as insulin resistance, type 2 diabetes (T2D), high blood pressure and high cholesterol levels. CMRF has been shown to influence the functioning and restructuring of endothelium and smooth muscle cells, affecting vascular function. Furthermore, the PAT, which refers to the fat deposit that surrounds most blood arteries, has been identified as a regulator of vascular tone. This has significant implications for the understanding of CVD etiology. In healthy individuals, the PAT has an anticontractile role. However, in obese and atherosclerotic patients, the dysfunctional PAT is associated with an elevation in prooxidant and proinflammatory cytokines.10 An abnormal redox state in the PAT might indicate to the neighboring vascular wall and increase the likelihood of vascular harm. Oxidative stress is characterized by an imbalance favoring oxidants over antioxidants, which disrupts redox signaling. Thus, PAT oxidative stress is identified as a pathophysiological mechanism that causes vascular damage in response to CMRF.11 This study focuses on the processes that cause oxidative stress in PAT in response to CMRF and illnesses. The keywords used, without being restricted to, were ROS, alcohol, atherosclerosis, hypertension, obesity, diabetes, superoxide, and aging, all subsequently accompanied by the phrase PAT or perivascular fat. This review aims to examine the pathological characteristics of AT and the actions that elevate the peril of other metabolic disorders.
Perivascular adipose tissue (PAT)
The PAT is in direct contact with the blood arteries, without any fascial layer acting as a barrier between this fat deposit and the vascular wall. Furthermore, it is important to mention that there have been observations of adipocytes infiltrating the external area of the adventitia. PAT is similarly infiltrated by the vascular supply network, a system of very few blood vessels that nourish the conduit vessels. 12 Research has compared adipocytes in PAT to adipocytes in subcutaneous and visceral fat in both humans and animals. Research studies have consistently demonstrated that adipocytes in PAT are highly heterogeneous and distinct from those in hypodermal and visceral fat. Perivascular adipocytes (PAcs) in humans and animals are smaller and have irregular shapes associated with hypodermic and visceral adipocytes. There is ongoing debate over categorizing PAT as a storage site for either white or brown AT. While the levels of PRDM 16, peroxisome-proliferator-activated receptor co-activator 1, and uncoupling protein 1 (UCP-1)13 are higher in PAcs compared to subcutaneous and perineal adipocytes, the levels of UCP-1 in PAcs are around 1000 fold lower than in brown adipocytes. Additional research corroborates the notion that the white adipocyte-like phenotype is the usual characteristic in PAT. Therefore, the PAT of various blood arteries may possess many facets regarding physiology and function. The adipose tissue around the heart is often classified into four types: (i) epicardial AT, (ii) pericardial AT, (iii) paracardial AT, and (iv) PAT, as shown in Figure 1.
 |
Figure 1: Adipose tissue surrounding the heart. (Image credit: Konwerski et al. 14) |
The Nexus of Adiposopathy and Cardio-Metabolic Health
AT stores excess energy and acts as a glandular and lymphoid organ that can release various biologically active compounds intricate in glycemic regulation, cholesterol metabolism, hemostasis, blood pressure regulation, inflammation, and nourishing response (leptin)15 These inflammatory and hormonal variables play a significant role in inner-organ communication, which has been shown to impact the metabolism and function of tissues such as muscle, liver, and vasculature. Several studies have shown that an increase in pro-inflammatory adipokines and a decrease in anti-inflammatory adipokines in diseased AT play a role in developing cardio-metabolic disorders.16 One example is the pro-inflammatory cytokine IL-6, which directly promotes the production of adhesion molecules in endothelial cells and lymphocytes. Furthermore, it serves as a crucial factor in creating acute phase proteins. It can promote blood clotting in platelets and the buildup of lipids in macrophages. Several researchers have also recorded the athero-protective characteristics of IL-6 due to its ability to regulate lipids by increasing the expression of ATP-binding cassette transporter A1 (ABCA1), a crucial protein in removing lipids from macrophages.17 Further research is required to understand the opposing properties of IL-6 in its atherogenic and atheroprotective actions. TNF-α, a pro-inflammatory cytokine, is known to be excessively produced in dysfunctional AT and substantially impacts insulin resistance via modifying insulin signaling. Furthermore, it is important to mention that both animal and human research have provided evidence supporting the involvement of these pathways in endothelial dysfunction. In addition to these cytokines that promote inflammation, levels of resistance in the bloodstream are also elevated and are linked to the development of calcification in coronary arteries. In addition, PAI-1 is increased in dysfunctional AT and has a role in causing blood clotting issues in persons with unhealthy fat deposits, generally known as “sick fat.” PAT secretes many adipokines and chemicals, such as oxygen radicals, aliphatic acids, cytokines, apparatuses of the renin-angiotensin system, chemokines, and hormones.18 The epicardial AT, found among the heart and the cardiac membrane, has a virulent outline of adipokines in individuals diagnosed with cardiovascular disease.19
Physical Activity as a Regulator of Adipose Tissue Function
Recent epidemiological research has provided increasing evidence that physically inactive individuals have a greater likelihood of developing cardio-metabolic disorders.20 A sedentary lifestyle and a favorable calorie balance contribute to the development of harmful AT in individuals who are hereditarily and ecologically predisposed. Excessive fat accretion in adipocytes may cause a lack of oxygen, leading to the inhibition of the adipocyte differentiation process from pre-adipocytes. In addition, decreased oxygen supply to AT triggers the transformation of immune scavenger cells from M2 to M1 and disrupts the balance between anti-inflammatory and pro-inflammatory cytokines.21 The irregular secretion of adipokines results in metabolic dysfunction and increases the hazard of obesity and its linked metabolic syndrome. Published findings show that M1 macrophages generate immune-activating and promote chronic inflammation, whereas M2 macrophages yield anti-inflammatory cytokines and help mitigate this chronic inflammation. This suggests that the abnormal behavior of macrophages plays a crucial role in influencing the development of cardio-metabolic disorders. Physical exercise’s impact on regulating AT function has been extensively studied in both animals and humans. Exercise training reduces the infiltration of macrophages in AT and induces a shift in phenotype from M1 to M2. The precise molecular pathways accountable for these alterations remain unclear. Reported studies have shown that implementation preparation decreases lactate levels in the AT inside the abdomen while also increasing the presence of VEGF-A.22 VEGF-A is important for promoting the development of blood vessels and capillaries in the tissue. An oxidative implementation sequencer successfully lowers instinctual AT by more than 32 cm2 in females and more than 42 cm2 in men, as shown using computed tomography analysis.23 This reduction was achieved even without a hypocaloric diet. Regular exercise decreases the proportions of adipocytes and the amount of lipid stored inside them. Exercise training has been shown to improve malformed adipocytes and boost the manifestation of genes complicated in mitochondrial biogenesis and function.
Re-Browning WAT
In early life, human cardiac and PAT are classified as brown AT. However, as individuals age and accumulate extra fat, this tissue undergoes a process called whitening, which leads to malfunction. This dysfunction contributes to the development of atherosclerosis in the nearby blood arteries. Brown AT is well recognized for its abundant mitochondria and elevated appearance of UCP1,24 which is important for cellular thermogenesis. Multiple studies have proposed that the re-browning of WAT might decrease the risk of cardiovascular problems by reducing inflammation and hypoxia in the affected area. During physical activity, muscles contract and produce bioactive chemicals called myokines, which can change the metabolic characteristics of AT. These myokines also stimulate the process of browning and enhance the oxidation of fatty acids in AT.25 A myokine called Irisin can contribute to the process of browning AT. Research on animals has shown that physical activity increases the production of PPAR-γ co-activator 1-alpha (PGC-1α), which enhances the production of FNDC5, the gene responsible for producing Irisin. PGC-1α is well acknowledged as a regulatory co-activator of peroxisome proliferator-activated receptor gamma (PPAR-γ). PPAR-γ controls the production UCP-1 in mitochondria, affecting the intracellular metabolism of adipocytes.26 Exercise leads to an increase in PPAR-γ mediated signaling, which in turn improves the levels of different lipids in the blood. This includes greater HDL-C levels and lower TC, LDL-C,27, and M2 macrophage activation levels in AT.
AT in age-related vascular disease
Aged rats showed elevated leptin gene expression in the thoracic aortic PAT. They had high quantities of leptin in their bloodstream, indicating that PAT may be a potential cause of this hormone. Leptin has been shown to stimulate NADPH oxidase,28 indicating that this process may serve as a connection between aging and the production of superoxide in PAT. The morphology of the aorta PAT is altered by aging. In young mice, PAT is mostly formed of multi-locular brown adipocytes. Still, in aged mice, it comprises a combination of multi-locular brown adipocytes and uni-locular white adipocytes.29 PAT whitening increases the expression of inflammatory and pro-oxidant factors. Based on this, the thoracic PAT of aged rats exhibits higher levels of lipid accumulation and CD11c expression, a marker for pro-inflammatory macrophages. This indicates that inflammatory PAT may be involved in age-related vascular dysfunction. Bailey-Downs and co-authors examined the combined impact of aging and obesity on inflammation and oxidative stress in PAT,30, and the influence of PAT-secreted components on vascular function in aging. This increase was further intensified when aging was combined with obesity. The appliances accountable for oxidative strain in the PAT of obese elderly mice were a surge in NADPH oxidase commotion, a reduction in eNOS expression, and elevated levels of inflammatory markers.
The Impact of PAT Oxidative Stress on Cardiometabolic Disorders
The precise processes responsible for oxidative stress in PAT in obesity remain unknown. Increasing the expression of Mineralocorticoid Receptors (MR) leads to a surge in the manufacture of hydrogen peroxide (H2O2) in adipocytes.31 Blocking MR inhibits both the malfunction of mitochondria and the damage of the anticontractile function of PAT in stoutness. Obese patients have increased levels of the MR ligand aldosterone. This mineralocorticoid is recognized for its ability to activate NADPH oxidase and cause eNOS uncoupling. Thus, the activation of MR seems to be a potential mediator of oxidative stress in PAT in individuals with obesity. However, further research is required to understand this relationship fully. Research has shown that Type-1 diabetes (T1D) impacts the production of ROS in PAT.32 The production of H2O2 by PAT in skeletal muscle arteries is enhanced in rats with T1D caused by streptozotocin (STZ). This suggests that oxidative stress in PAT contributes to vascular dysfunction in the context of diabetes. Chronic ethanol treatment for 12 weeks in mice intensified vascular superoxide production and lipoperoxidation in the aorta due to increased PAT oxidative stress. The chronic use of alcohol resulted in increased oxidative stress in the blood vessels and PAT, which was accompanied by elevated stages of TNF-α and IL-6 in both the vascular wall and PAT. TNF-α receptor 1-deficient mice (TNFR1) were shielded from chronic ethanol-induced damage to the PAT and blood vessels. This protection was achieved by inhibiting the stimulation of NADPH oxidase and the development of superoxide in the vascular and perivascular tissue. Multiple studies have shown that PAT is impaired in conditions such as chronic alcohol intake, diabetes, hypertension, obesity, and atherosclerosis. However, in the early stages of obesity and atherosclerosis, PAT may have a protective effect. CMRF may affect the redox state of PAT and lead to oxidative stress, a pathological process contributing to vascular damage. Figure 2 shows the oxidative stress on cardiometabolic disorders.
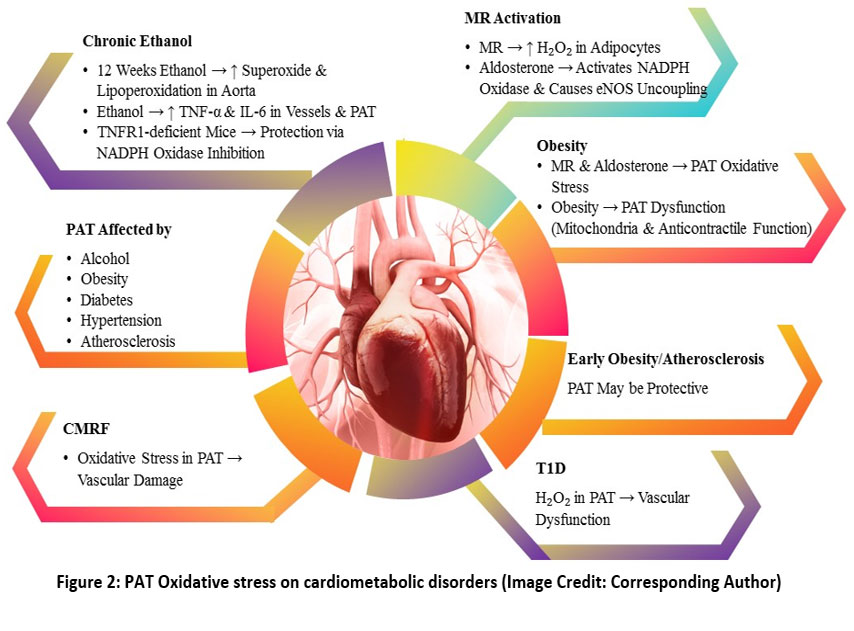 |
Figure 2: PAT Oxidative stress on cardiometabolic disorders (Image Credit: Corresponding Author) |
PAT in Atherosclerosis
The involvement of the protected system and tenderness in atherosclerosis has remained recognized for many years.33 Initial studies on the immunological processes of atherosclerosis take mostly absorbed on the neo-intima and atherosclerotic plaques. Current research indicates that perivascular inflammation plays a significant role in different phases of atherosclerosis. Inflammation around blood vessels occurs before the creation of atherosclerotic plaques and even the development of endothelial dysfunction and oxidative stress in apolipoprotein. Although most of the atherosclerosis research primarily examines adventitial inflammation, there are obvious connections to PAT in these investigations. Atherosclerotic mice exhibit elevated production of pro-inflammatory cytokines, namely IL-6 and IL-1, in the PAT. The molecular processes of PAT inflammation in atherosclerosis reveal many crucial objectives. Signal transducer and activator of transcription 4 (STAT4) is present in both adipocytes and immune cells and may play a role in the inflammation of PAT.34 Although macrophages and T cells regulate inflammation in atherosclerosis, current research has discovered the critical involvement of perivascular mast cells. Throughout the development of plaque, mast cells that have been triggered gather in the artery’s outer layer and stimulate the death of macrophages and the leaking of small blood vessels. The activation of mast cells surrounding blood vessels increases the adherence of monocytes by a mechanism that depends on the presence of CXCR-2 and vascular cell adhesion molecule 1 (VCAM1). Alternatively, emerging evidence suggests that not all immune responses exacerbate atherosclerosis. Certain regulatory immune pathways may exert protective effects during early lesion formation, contradicting the classical pro-inflammatory model.35 Moreover, the functional plasticity of immune cells such as macrophages and T cells under different microenvironmental conditions introduces complexity and controversy regarding their net effect in atherosclerosis progression. These findings underscore the context-dependent duality of immune mechanisms in vascular inflammation and plaque development.
PAT in High arterial pressure
High arterial pressure is linked to the stimulation of the renin-angiotensin-aldosterone system and increased oxidative stress in the blood vessels.36 Ang II and ROS are essential in starting and sustaining vascular inflammation. The main location of the early inflammation in hypertension is in the PAT and the boundary between PAT and the adventitia. PAT is a term used to describe the presence of complex machinery that includes ROS, NADPH, eNOS, and antioxidant enzymes. ROS produced from PAT may contribute to endothelial dysfunction. This dysfunction can occur either by PAT-derived ROS scavenging endothelial NO or modulating perivascular inflammation, subsequently impacting endothelial function. Throughout the development of high arterial pressure, immune cells mostly gather in the PAT surrounding both major and smaller blood vessels, such as the mesenteric and aorta arteries. Infections are prominent in PAT, whereas immune cell infiltration in non-perivascular visceral fat is less evident in hypertension generated by non-obesity. Long-term oxidative stress contributes to the development of vascular inflammation in individuals with hypertension.37 Mice without certain NADPH oxidase components, such as p47 phlox, NOX1, and NOX4, are shielded from hypertension. On the other hand, mice with excessive p22 phlox (a subunit of NADPH oxidase), specifically in smooth muscle cells, experience a surge in the manufacture of superoxide in blood vessels. This increase is related to a rise in the total number of leukocytes in PAT. Recent studies suggest that while the ROS-NADPH axis in PAT plays a crucial role in vascular inflammation during hypertension, alternative mechanisms such as mitochondrial dysfunction and gut microbiota-derived metabolites (e.g., trimethylamine N-oxide) may also contribute to endothelial impairment. Furthermore, the role of adaptive immunity, particularly T cell subtypes, in mediating hypertensive inflammation remains controversial and is under active investigation. These emerging insights challenge the traditional focus solely on oxidative stress pathways and suggest a more integrative model of vascular pathology in hypertension.38
PAT in Aneurysms
Abdominal aortic aneurysm (AAA) is an inflammatory condition characterized by significant alterations in the cellular makeup of the aorta wall and PAT. 39 Aneurysm development often occurs with atherosclerosis. Several types of inflammatory cells, including neutrophils, macrophages, T and B cells, and mast cells, have a role in the development of AAA. These immune cells are seen in both the PAT and the luminal thrombi. They are partly associated with advanced atherosclerotic plaques. However, they significantly enhance the likelihood of AAA formation. 40 A lack of TLR4 or myeloid differentiation factor 88 (MyD88) decreases perivascular inflammation and AAA development. 41 In addition to causing overall inflammation, leukocytes in the PAT may generate proteases, such as cathepsins, which facilitate the breakdown of cells in the aorta wall. While inflammation is a well-established contributor to AAA, emerging hypotheses suggest a pivotal role for vascular smooth muscle cell (VSMC) phenotypic switching and cellular senescence in aneurysm progression, independent of classical immune pathways. Recent studies highlight mitochondrial dysfunction and impaired autophagy in VSMCs as alternative mechanisms underlying aortic wall degradation. Moreover, controversy persists regarding whether perivascular adipose tissue (PAT) inflammation is a cause or consequence of AAA. PAT growth may initially serve a protective, compensatory role before becoming pathogenic, challenging the conventional unidirectional inflammatory model.42
Future implications
There are still unresolved inquiries that need more investigation, such as The underlying source of the deficiency in the adipocyte growth process and the conversion of superior fat into unhealthy fat. The causative factors for the shift of macrophages from an M2 to an M1 polarization state also need to be investigated. Further research is required to identify the optimal exercise period and concentration to stimulate, activate, and recruit brown AT. PAT has a significant role in controlling vascular function in both healthy and diseased states. Although there have been recent advancements in this area, the existing experimental models still have limitations that result in significant gaps in our understanding. The processes responsible for regulating the changes in phenotype and communication of PAcs with other cell types, such as inflammatory cells, nerve cells, and progenitor cells, and their effects on age-related vascular disease are poorly understood. Developing more and improved animal models is essential for addressing these inquiries in the future. There is a lack of research on the interactions between the immune system and PAT in aging and age-related disorders. It is essential to do more research in this area to better understand the underlying processes and discover new therapy targets. PAT is a prognosticator of vascular illness and a promising focus for treatment in aging blood arteries, warranting more research. Recent insights reveal that PAT influences vascular function via anti-inflammatory and vasoactive mediators. Therapeutic strategies like exercise, caloric restriction, and agents targeting AMPK or PPARγ pathways help restore PAT function. Inducing PAT browning also shows promise in mitigating vascular dysfunction and cardiometabolic risk.
Conclusion
Excessive buildup of AT may cause pathological alterations in adipocytes, which can contribute to obesity and the onset of cardiometabolic disorders. This review explores the multifaceted role of PAT in cardio-metabolic health and disease. PAT acts as a dynamic endocrine organ, linking adiposopathy to vascular dysfunction. Physical activity modulates adipose tissue homeostasis, promoting metabolic health and inducing the browning of WAT, which enhances thermogenic and anti-inflammatory functions. The review highlights how aging alters PAT, contributing to vascular stiffness and endothelial dysfunction. Oxidative stress in PAT is identified as a key driver of cardiometabolic disorders. Disease-specific sections delve into PAT’s role in atherosclerosis, hypertension, and aneurysm formation, influencing immune cell infiltration, vascular remodeling, and inflammation. Finally, the review underscores future directions targeting PAT-mediated mechanisms for therapeutic interventions, emphasizing the need for deeper insights into its molecular pathways to mitigate cardiovascular disease progression. The elevated adiponectin and nitric oxide levels produced by eNOS in PAT suggest a counter-regulatory mechanism. This mechanism may play a protective role by reducing the formation of ROS in blood vessels and slowing the progression of atherosclerosis.
Acknowledgement
The authors would like to thank Chalapathi Institute of Pharmaceutical Sciences, Chalapathi Nagar, Lam, Guntur, India and Department of Biomedical Sciences, Gulf Medical University, Sree Balaji Medical College and Hospital, Chromepet, Chennai, Bridgetown International University, Barbados, Princess Nourah bint Abdur Rahman University, SA, Guru Nanak Institute of Pharmaceutical Science and Technology, Kolkata West Bengal, Assam down town University, Sankar Madhab Path, Gandhi Nagar, Panikhaiti, Guwahati, Assam and other facilities to draft this review.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Rama Rao Nadendla, Kumaraswamy Dabburu, Suria Prabha Kannaiyan, Gunamoni Das: Concept, Designed;
- Sovan Bagchi, Kumaraswamy Dabburu, Tamalika Chakraborty, Tabinda Hasan and Srismitha Settipalli: Reviewed the manuscript;
- Sovan Bagchi, Kumaraswamy Dabburu, Gunamoni Das, Tamalika Chakraborty and Tabinda Hasan: Manuscript preparation, Editing and designed the manuscript;
- Kumaraswamy Dabburu, Srismitha Settipalli and Gunamoni Das All image works.
References
- SuX,ChangD.Role of Adiposopathy and PhysicalActivity in Cardio-MetabolicDisorderDiseases. Chim. Acta 2020, 511, 243–247.
CrossRef - BarA,Kieronska‐RudekA,ProniewskiB, et al.In VivoMagneticResonanceImaging‐BasedDetection of HeterogeneousEndothelialResponse in Thoracic and AbdominalAorta to Short‐TermHigh‐FatDietAscribed to Differences in PerivascularAdipose Tissue in Mice. Am. Heart Assoc. 2020, 9 (21), e016929.
CrossRef - LeiriaLO,TsengLipidomics of Brown and WhiteAdipose Tissue: Implications for EnergyMetabolism. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2020, 1865 (10), 158788.
CrossRef - IbarzabalA,BalibreaJM, Vidal JM, OrtegaE,de HollandaA. FABP4 Expression in Subcutaneous Adipose Tissue Is Independently Associated with Circulating Triglycerides in Obesity. Clin. Med. 2023, 12 (3):1013, FABP4. DOI: 10.3390/jcm12031013
CrossRef - Małodobra-MazurM, CierzniakA, PawełkaD, KaliszewskiK, RudnickiJ, DoboszT.Metabolic DifferencesBetweenSubcutaneous and VisceralAdipocytesDifferentiated with an Excess of Saturated and MonounsaturatedFattyAcids. Genes 2020, 11 (9), 1092.
CrossRef - ChaitA,Den HartighLJ.Adipose TissueDistribution, Inflammation and ItsMetabolicConsequences, IncludingDiabetes and CardiovascularDisease. Cardiovasc. Med. 2020, 7, 22.
CrossRef - BalakumarP, AlqahtaniA,KhanNA,AlqahtaniT,A T,JagadeeshG.The Physiologic and PhysiopathologicRoles of PerivascularAdipose Tissue and ItsInteractions with Blood Vessels and the Renin-AngiotensinSystem. Res. 2021, 173, 105890.
CrossRef - Liu CH, Hsu HJ, Tseng TL, et al.COMT-CatalyzedPalmitic AcidMethylEsterBiosynthesis in PerivascularAdipose Tissue and ItsPotentialRoleAgainstHypertension. Pharmacol. Exp. Ther. 2020, 373 (2), 175–183.
CrossRef - LeeH,KimH,JeonJS,NohH,LeeEJ,KwonSH.Association BetweenAbdominalPerivascularAdipose TissueQuantity and QualityAssessed by CT and CardiometabolicRisk. Nutr. 2023, 42 (6), 869–878.
CrossRef - AzulL,LeandroA,BoroumandP,KlipA,SeiçaR,SenaCM.Increased Inflammation, OxidativeStress, and a Reduction in AntioxidantDefenseEnzymes in PerivascularAdipose TissueContribute to VascularDysfunction in Type 2 Diabetes. Free Biol. Med. 2020, 146, 264–274.
CrossRef - MarchiniRedox and InflammatoryMechanismsLinkingAirPollutionParticulateMatter with CardiometabolicDerangements. Free Radic. Biol. Med. 2023, 209 (2), 320–341.
CrossRef - Hillock-WatlingC,GotliebAI.The Pathobiology of PerivascularAdipose Tissue (PVAT), the FourthLayer of the Blood VesselWall. Pathol. 2022, 61, 107459.
CrossRef - Huesca-GómezC,Torres-PazYE,Fuentevilla-ÁlvarezG,et al.Expressions of mRNA and EncodedProteins of MitochondrialUncoupling ProteinGenes (UCP1, UCP2, and UCP3) in Epicardial and MediastinalAdipose Tissue and Associations with CoronaryArteryDisease. Endocrinol. Metab. 2023, 67 (2), 214–223.
CrossRef - KonwerskiM,Gąsecka A,OpolskiG,GrabowskiM,MazurekT.Role of Epicardial Adipose Tissue in Cardiovascular Diseases: A Review. Biology 2022, 11 (3), 355.
CrossRef - BerneaEG,SuicaVI,UyyE, et al.Exosome ProteomicsReveals the Deregulation of Coagulation, Complement and LipidMetabolismProteins in GestationalDiabetes Mellitus. Molecules 2022, 27 (17), 5502.
CrossRef - VlioraM,RavelliC,GrilloE,CorsiniM,FlourisAD,MitolaS.The Impact of Adipokines on VascularNetworks in Adipose Tissue. Cytokine Growth Factor 2023, 69, 61–72.
CrossRef - HeP,GelissenIC,AmmitAJ.Regulation of ATP Binding CassetteTransporter A1 (ABCA1) Expression: Cholesterol-Dependent and–IndependentSignalingPathways with Relevance to InflammatoryLungDisease. Res. 2020, 21 (1), 250.
CrossRef - KalupahanaNS,GoonapienuwalaBL,Moustaid-MoussaN.Omega-3 Fatty Acids and Adipose Tissue: Inflammation and Browning. Rev. Nutr. 2020, 40 (1), 25–49.
CrossRef - Al-MansooriL,Al-JaberH,PrinceMS,ElrayessMA.Role of InflammatoryCytokines, Growth Factors and Adipokines in Adipogenesis and InsulinResistance. Inflammation 2022, 45 (1), 31–44.
CrossRef - SuX,PengD.Adipokines as NovelBiomarkers of Cardio-MetabolicDisorders. Chim. Acta 2020, 507, 31–38.
CrossRef - JamesG,ChenX,DiwanA,HodgesPW.Fat Infiltration in the MultifidusMuscleIsRelated to Inflammatory Cytokine Expression in the Muscle and EpiduralAdipose Tissue in Individuals Undergoing Surgery for Intervertebral DiscHerniation. Spine J. 2021, 30 (4), 837–845.
CrossRef - Wu M,Chen L,Qi Y,et al.Human Umbilical Cord Mesenchymal Stem Cell Promotes Angiogenesis via Integrin β1/ERK1/2/HIF-1α/VEGF-A Signaling Pathway for Off-the-Shelf Breast Tissue Engineering. Stem Cell Ther. 2022, 13 (1), 99.
CrossRef - Pimanov S, Bondarenko V, Makarenko E. Visceral Fat in Different Locations Assessed by Ultrasound: Correlation with Computed Tomography and Cut‐OffValues in Patients with Metabolic Syndrome. Obes. 2020, 10 (6), e12404.
CrossRef - BelJS,TaiTC,KhaperN,LeesSJ.Chronic Glucocorticoid Exposure Causes Brown Adipose Tissue Whitening, Alters Whole‐Body Glucose Metabolism and Increases Tissue Uncoupling Protein‐1. Rep. 2022, 10 (9), e15292.
CrossRef - JiangS,Bae JH, WangY,SongW.The PotentialRoles of Myokines in Adipose TissueMetabolism with Exercise and ColdExposure. J. Mol. Sci. 2022, 23 (19), 11523.
CrossRef - TangQ,LuM,XuB,et al.Electroacupuncture RegulatesInguinalWhiteAdipose TissueBrowning by Promoting Sirtuin-1-dependent PPAR γ Deacetylation and MitochondrialBiogenesis. Endocrinol. 2020, 11, 607113.
CrossRef - SchaltenbergN,JohnC,HeineM,et al.Endothelial LipaseIsInvolved in Cold-InducedHigh-Density Lipoprotein Turnover and Reverse Cholesterol Transport in Mice. Cardiovasc. Med. 2021, 8, 628235.
CrossRef - Tanaka S, Watanabe H, Nakano T, et al. Indoxyl Sulfate Contributes to Adipose Tissue InflammationThrough the Activation of NADPH Oxidase. Toxins 2020, 12 (8), 502.
CrossRef - Negron S G, Xu B, Lin Z. Isolating Brown Adipocytes from Murine Interscapular Brown Adipose Tissue for Gene and Protein ExpressionJ. Vis. Exp.; Jove 2021, (169), e62332.
CrossRef - Al-hussaniy HA, AL-Biati HA. The role of leptin hormone, neuropeptide Y, ghrelin and leptin/ghrelin ratio in Obesogenesis. Med Pharm J. 2022, 1(2), 52-63.
CrossRef - Armani A, Caprio M. Mineralocorticoid Receptor and Aldosterone: From Hydro-saline Metabolism to Metabolic Diseases. InInhydro Saline Metabolism: Epidemiology, Genetics, Pathophysiology, Diagnosis and Treatment; Springer International Publishing, 2023, pp 431–471.
CrossRef - ZobelEH,ChristensenRH,WintherSA, et al.Relation of CardiacAdipose Tissue to CoronaryCalcification and MyocardialMicrovascularFunction in Type 1 and Type 2 Diabetes. Diabetol. 2020, 19 (1), 16.
CrossRef - Ahmadieh S, Kim H W, Weintraub N L.Potential Role of Perivascular Adipose Tissue in Modulating Atherosclerosis. Sci. (Lond) 2020, 134 (1), 3–13.
CrossRef - Zierold S, Buschmann K, Gachkar S, et al.Brain‐Derived Neurotrophic Factor Expression and Signaling in Different Perivascular Adipose TissueDepots of Patients with Coronary Artery Disease. Am. Heart Assoc. 2021, 10 (6), e018322.
CrossRef - Libby P. Inflammation during the life cycle of the atherosclerotic plaque. Cardiovasc Res. 2021, 117(13), 2525-2536.
CrossRef - HaberkaM,SiniarskiA,GajosG, et al.PericardialFat and Glucagon-LikePeptide-1 and-2 ReceptorsExpression in StablePatients with MultivesselCoronaryArteryDisease: An Association with the Renin-Angiotensin-AldosteroneSystem. Arch. Med. Wewnętrznej= Polish Archives of Internal Medicine 2021, 131 (3).
CrossRef - WangD,LiJ,LuoG, et al.Nox4 as a NovelTherapeuticTarget for DiabeticVascularComplications. Redox 2023, 64, 102781.
CrossRef - Barhoumi T, Kassan M, Tabet F. Role of T cells in hypertension-induced vascular inflammation and remodeling. Hypertens Res. 2022, 45(5), 720-732
CrossRef - YeT,ZhangG,LiuH, et al.Relationships BetweenPerivascularAdipose Tissue and AbdominalAorticAneurysms. Endocrinol. 2021, 12, 704845.
CrossRef - ThanigaimaniS,GolledgeJ.Role of Adipokines and PerivascularAdipose Tissue in Abdominal Aortic Aneurysm: A Systematic Review and Meta-analysis of Animal and Human Observational Studies. Endocrinol. 2021, 12, 618434.
CrossRef - OwensIII A P, Rateri D L, Howatt D A, et al.MyD88 Deficiency Attenuates Angiotensin II-Induced Abdominal Aortic Aneurysm Formation Independent of Signaling ThroughToll-Like Receptors 2 and 4. Thromb. Vasc. Biol. 2011, 31 (12), 2813–2819.
CrossRef - Sun J, Wang J, Zhang J, et al. Perivascular adipose tissue: friend or foe in vascular pathologies? Front Cardiovasc Med. 2023, 10, 1169042
CrossRef
Abbreviations List
Adipose Tissue (AT)
Cardiometabolic Risk Factors (CMRF)
Cardiovascular Diseases (CVD)
Endothelial Nitric Oxide Synthase (eNOS)
Extracellular-Signal-Regulated Kinase (ESRK)
Nicotinamide Adenine Dinucleotide Phosphate (NADPH)
Perivascular Adipose Tissue (PAT)
Perivascular Adipocytes (PAcs)
Positive-Regulatory-Domain-Containing (PRDM)
Reactive Oxygen Species (ROS)
Triglycerides (TG)
Type 2 diabetes (T2D)
Type-1 diabetes (T1D)
Uncoupling Protein 1 (UCP1)
Vascular Endothelial Growth Factor A (VEGF-A)
White adipose tissue (WAT)
Abdominal aortic aneurysm (AAA)
vascular smooth muscle cell (VSMC)







