Manuscript accepted on :02-09-2025
Published online on: 25-09-2025
Plagiarism Check: Yes
Reviewed by: Dr Shwetha
Second Review by: Dr. Deepak Kumar
Final Approval by: Dr. Patorn Piromchai
Essenhaji Sanaa1,2* , Anaibar Fatima Ezzahra3
, Anaibar Fatima Ezzahra3 , Jarir Jamal2, Mohammadi Hicham2
, Jarir Jamal2, Mohammadi Hicham2 , Habbal Rachida4
, Habbal Rachida4 , Houari Chaymaa4, Chgoury Fatima2
, Houari Chaymaa4, Chgoury Fatima2 , Belhouari Abderrahmane5, Ghalim Noreddine2 and Kabine Mostafa1
, Belhouari Abderrahmane5, Ghalim Noreddine2 and Kabine Mostafa1
1Department of Biology; Laboratory of health and environment, Hassan II University, Faculty of Sciences, km 8 Road El Jadida BP5366, Casablanca, Morocco.
2Department of Medical biology; Laboratory of Biochemistry, Pasteur Institute of Morocco, Casablanca, Morocco.
3Department of Biology; Laboratory of Anthropogenetics, Biotechnology and Health, Chouaïb Doukkali University, El Jadida, Morocco.
4Department of Cardiology Service, Ibn Rochd University Hospital Center, Casablanca, Morocco.
5Department of Biology Faculty of Sciences Ben M'Sick ; Sidi Othmane Casablanca, Morocco.
Corresponding Author E-mail : sanaessenhaji@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3269
Abstract
The prevalence of cardiovascular diseases (CVDs) is steadily rising in Morocco and across North Africa, largely linked to dyslipidemia and persistent inflammatory states. This study was conducted to evaluate lipid profiles alongside High-Sensitivity Troponin I (hs-cTnI) and C-Reactive Protein (CRP) levels. A cohort of 240 healthy individuals served as controls, compared with 351 patients diagnosed with acute CVD. Patients exhibited significantly higher levels of LDL-C (1.48 ± 0.45 g/L vs. 1.14 ± 0.24 g/L), hs-cTnI (10.2 ± 5.3 ng/mL vs. 3.1 ± 1.8 ng/mL), and CRP (9.8 ± 7.9 mg/L vs. 2.4 ± 1.6 mg/L), with all differences reaching statistical significance (p < 0.001). Multivariate logistic regression analysis identified LDL-C, hs-cTnI, and CRP as independent predictors of acute CVD, with odds ratios (OR) of 1.82 (95% CI: 1.45-2.29), 1.06 (95\% CI: 1.03-1.09), and 1.12 (95\% CI: 1.04-1.21), respectively Receiver Operating Characteristic (ROC) curve analysis showed that hs-cTnI had moderate discriminative ability (AUC = 0.70), whereas CRP displayed limited predictive performance (AUC = 0.62). These findings suggest that hs-cTnI, and to a lesser extent CRP, may enhance risk stratification for acute CVD in Moroccan populations, highlighting the need for biomarker-guided protocols that are both effective and resource-conscious in clinical practice.
Keywords
Cardiovascular Diseases; C-Reactive Protein; Hypercholesterolemia; Morocco; North Africa; Troponin I.
Download this article as:| Copy the following to cite this article: Sanaa E, Ezzahra A. F, Jamal J, Hicham M, Rachida H, Chaymaa H, Fatima C, Abderrahmane B, Noreddine G, Mostafa K. High-Sensitivity Troponin I, C - reactive protein, and Hypercholesterolemia as Predictors of Acute Cardiovascular Events in Morocco. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Sanaa E, Ezzahra A. F, Jamal J, Hicham M, Rachida H, Chaymaa H, Fatima C, Abderrahmane B, Noreddine G, Mostafa K. High-Sensitivity Troponin I, C - reactive protein, and Hypercholesterolemia as Predictors of Acute Cardiovascular Events in Morocco. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/3KdTcih |
Introduction
Cardiovascular diseases (CVDs) are a leading global health threat, causing approximately 18 million annual deaths—accounting for 32% of all mortality worldwide—with 75% of these deaths occurring in low- and middle-income countries.1 This burden is starkly evident in Morocco, where CVDs are the predominant cause of death, responsible for 34% of annual mortality.1Nearly most cardiovascular-related mortality, about three out of four, happen in low- and middle-income regions, highlighting the unequal burden these regions face. 1.2
Morocco exemplifies this trend, having experienced a 42% increase in CVD prevalence since 2005. Currently, these diseases are responsible for 34% of national mortality.1.3This growing health crisis is closely linked to rapid urbanization, aging demographics, and a surge in modifiable risk factors such as hypertension (25.3%), tobacco use (45–50%), and sedentary lifestyles (21.1%).4
From a pathophysiological perspective, elevated LDL-C is involved in the disruption of endothelial integrity and the progression of atherogenesis.5 while systemic inflammation, often reflected by height levels of (hs-CRP), promotes plaque instability.6 Additionally, troponin I (hs-cTnI) serves as a good marker of myocardial damage and is widely employed in detecting acute coronary syndromes.7Despite well-established roles for these biomarkers in cardiovascular risk assessment, data from North African populations, including Morocco, remain limited.8
The primary objective of this study was to assess the prognostic value of LDL cholesterol (LDL-C), high-sensitivity cardiac troponin I (hs-cTnI), and high-sensitivity C-reactive protein (hs-CRP) in Moroccan patients presenting with acute cardiovascular events. Specifically, the research aimed to establish population-specific cutoff values for these biomarkers, investigate potential sex-related variations in their levels, and contribute to the development of diagnostic strategies tailored to the Moroccan healthcare setting.
Materials and Methods
Study Design and Participants
This prospective case–control study included 351 consecutive patients admitted with acute cardiovascular events—such as myocardial infarction, heart failure, or ischemic stroke—at Ibn Rochd University Hospital, Casablanca, Morocco. A control group of 240 apparently healthy volunteers was recruited from the Pasteur Institute of Morocco. Controls were matched to cases by sex and age (±5 years). All participants signed written informed consent prior to enrolment. The study was approved by the institutional ethics committee.
Sample Size and Sampling Technique
A formal sample size calculation was not performed in advance. Instead, all eligible patients meeting the inclusion criteria during the study period were consecutively enrolled to maximize statistical power and ensure a representative sample. Control participants were selected through simple random sampling and matched to patients based on age and sex.
Data and Sample Collection
Demographic and clinical information—including age, sex, body mass index (BMI), blood pressure, smoking habits, physical activity, and medical history—was collected using standardized questionnaires. After overnight fasting, venous blood samples were drawn into serum-separator tubes, centrifuged at 2,500 rpm for 15 minutes, and analyzed within two hours of collection.
Laboratory Analyses
All assays were performed at the accredited laboratories of the Pasteur Institute of Morocco.
Lipid profile: Total cholesterol, triglycerides, and HDL-C were quantified enzymatically using the VITROS® 5600 platform (Ortho Clinical Diagnostics). LDL-C was calculated using the Friedewald formula.
High-sensitivity C-reactive protein (hs-CRP): Determined via particle-enhanced immunoturbidimetry on the BN ProSpec® analyzer (Siemens).
High-sensitivity cardiac troponin I (hs-cTnI): Measured using a chemiluminescent immunoassay with a detection limit of 1.2 ng/mL.
Apolipoproteins (ApoA1 and ApoB) were measured in a subset of participants due to limited sample availability and logistical constraints. Missing measurements were not replaced or statistically imputed.
Inclusion and Exclusion Criteria
Cases: Patients hospitalized with acute CVD, diagnosed according to the 2020 European Society of Cardiology STEMI/NSTEMI guidelines. Patients with malignancy or autoimmune disease were excluded.
Controls: Healthy individuals without a history of CVD, diabetes, or hypertension, and with normal lipid profiles (total cholesterol <2 g/L; LDL-C <1 g/L).
General exclusion criteria: Pregnancy, incomplete clinical data, or refusal to provide informed consent.
Statistical Analysis
Continuous variables were reported as mean ± standard deviation (SD), and categorical variables as frequencies or percentages. Data normality was tested using the Shapiro–Wilk method; non-normally distributed variables (e.g., hs-cTnI) were log-transformed before analysis. Between-group comparisons used unpaired t-tests for continuous variables and Chi-square tests for categorical variables.
Multivariate logistic regression was performed to identify independent predictors of acute CVD, including covariates with p < 0.05 in univariate analysis (age, sex, diabetes, smoking, physical activity, lipid parameters, hs-cTnI, and hs-CRP). Multicollinearity was assessed using variance inflation factors (VIF <5).
Receiver operating characteristic (ROC) curves were used to determine optimal biomarker cutoffs, reporting the area under the curve (AUC), sensitivity, and specificity. Bonferroni correction was applied for multiple comparisons. All statistical analyses were conducted using SPSS v26 (IBM Corp.) and Python, with a significance threshold of p < 0.05.
Results
Participant Characteristics
A total of 351 patients with acute cardiovascular disease (CVD) and 240 age- and sex-matched controls were included in the analysis. As summarized in Table 1, significant differences were observed in all baseline characteristics. Patients were, on average, older than controls and had a higher proportion of males. Moreover, patients presented with significantly higher systolic and diastolic blood pressure levels, as well as elevated fasting glucose levels compared to the control group (all *p*-values < 0.001).
Table 1: Baseline demographic and clinical characteristics of study participants.
| Characteristic | Patients (n = 351) | Controls (n = 240) | *p*-value |
| Age, years | 59.5 ± 10.4 | 54.8 ± 6.2 | <0.001 |
| Sex, male | 212 (60.4%) | 116 (48.3%) | 0.003 |
| Systolic BP, mm Hg | 131.3 ± 19.9 | 121.1 ± 14.5 | <0.001 |
| Diastolic BP, mm Hg | 75.5 ± 6.8 | 72.4 ± 6.8 | <0.001 |
| Fasting glucose, g/L | 1.50 ± 0.11 | 0.90 ± 0.14 | <0.001 |
Data are presented as mean ± standard deviation or n (%).
BP, blood pressure.
*p*-values were derived from independent samples t-test for continuous variables and Chi-square test for categorical variables (sex).
Lipid Profile
As detailed in Table 2, the lipid profile of patients was markedly atherogenic compared to controls. Patients exhibited significantly higher levels of total cholesterol, LDL-C, and triglycerides, along with significantly lower levels of HDL-C (all *p* < 0.001). In a subgroup analysis, ApoA1 levels were also significantly reduced in patients (*p* < 0.001), while ApoB levels did not differ significantly between the groups (*p* = 0.078).
Table 2: Comparison of lipid profiles between patients and controls.
| Parameter | Patients (n = 351) | Controls (n = 240) | *p*-value |
| Total cholesterol, g/L | 1.86 ± 0.43 | 1.56 ± 0.42 | <0.001 |
| LDL-C, g/L | 1.48 ± 0.45 | 1.14 ± 0.24 | <0.001 |
| HDL-C, g/L | 0.40 ± 0.14 | 0.49 ± 0.10 | <0.001 |
| Triglycerides, g/L | 1.80 ± 0.77 | 0.90 ± 0.34 | <0.001 |
| ApoA1, g/L a | 1.20 ± 0.30 (n=190) | 1.45 ± 0.19 (n=140) | <0.001 |
| ApoB, g/L a | 0.95 ± 0.22 (n=204) | 0.90 ± 0.10 (n=140) | 0.078 |
a ApoA1 and ApoB analyses were performed in a subset of participants due to sample availability.
Data are presented as mean ± standard deviation. p-values were derived from independent samples t-test.
*LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; ApoA1, apolipoprotein A1; ApoB, apolipoprotein B.*
ApoA1 and ApoB analyses were performed in a subset of participants (ApoA1: 190 patients, 140 controls; ApoB: 204 patients, 140 controls) due to sample availability.
Clinical Risk Factors
As shown in Table 3, the prevalence of modifiable risk factors was significantly higher in patients than in controls. Smoking demonstrated the strongest association with acute CVD (OR = 14.55, 95% CI: 7.22–29.36, *p* < 0.001), followed by physical inactivity (OR = 2.88, 95% CI: 2.03–4.09, *p* < 0.001). A significant association was also observed for alcohol consumption; however, the odds ratio was extremely high and statistically unstable due to the absence of exposed individuals in the control group (OR = 53.95, 95% CI: 3.29–882.90, *p* = 0.005), and this result should be interpreted with extreme caution.
Table 3: Association between modifiable risk factors and acute cardiovascular disease.
| Risk Factor | Patients (n=351) n (%) | Controls (n=240) n (%) | Odds Ratio (OR) | 95% CI | *p*-value |
| Smoking | 127 (36.2) | 9 (3.8) | 14.55 | 7.22 – 29.36 | <0.001 |
| Physical inactivity | 266 (75.8) | 125 (52.1) | 2.88 | 2.03 – 4.09 | <0.001 |
| Alcohol consumption ⁺ | 35 (10.0) | 0 (0.0) | 53.95 | 3.29 – 882.90 | 0.005 |
a Haldane correction applied due to zero cell count in controls.
Biomarker Distribution
The analysis revealed distinct sex-specific and age-specific patterns in biomarker levels.
For high-sensitivity cardiac troponin I (hs-cTnI), the distribution was significantly influenced by sex. Women exhibited notably lower median values compared to men, although with considerable overlap in the interquartile ranges between the two groups (Figure 1).
For high-sensitivity C-reactive protein (hs-CRP), a pronounced age and sex-specific pattern was observed. Analysis of age-stratified medians revealed that men experienced a relatively stable, gradual increase in hs-CRP levels with advancing age. In stark contrast, women demonstrated a sharp and significant peak in hs-CRP levels specifically within the 60-69 age group, a trend not observed in their male counterparts (Figure 2).
 |
Figure 1: Sex-specific distribution of high-sensitivity cardiac troponin I (hs-cTnI) levels. |
Box plot comparing serum hs-cTnI concentrations (in ng/L) between men and women. The box represents the interquartile range (IQR), the horizontal line inside the box indicates the median, and the whiskers extend to the minimum and maximum values within 1.5 * IQR. Circles represent outliers.
 |
Figure 2: Age-stratified distribution of high-sensitivity C-reactive protein (hs-CRP) levels by sex. |
Bar graph displaying median serum hs-CRP levels (in mg/L) with error bars (representing interquartile range or standard deviation) across different age strata for men and women. The plot highlights the distinct peak in median hs-CRP levels among women in the 60-69 age group.
The chart compares average levels of total cholesterol, LDL, HDL, and triglycerides in patients using bars (n=351) and controls (n=240). Error bars represent standard deviations, “This figure emphasizes a marked increase in CT, bad cholesterol LDL-C, and triglycerides, and a significant decrease in good cholesterol HDL-C among patients (all P < .001).”
Lipid profiles, particularly HDL-mediated lipid transfers, are essential for evaluating cardiovascular risk, as they are closely linked to atherosclerosis, a primary cause of CVDs such as myocardial infarction and stroke.9 Elevated LDL-C is a key atherogenic factor, with strong evidence from observational, genetic, and clinical studies supporting its role in CVD pathogenesis.10
Substantial LDL-C lowering achieved through PCSK9 inhibition has been consistently associated with reduced cardiovascular event rate; reinforcing the principle of minimizing LDL-C levels.11HDL-C is traditionally associated with reduced CVD risk; however, interventions to increase HDL-C have not consistently lowered event rates, suggesting that HDL functionality may be more critical than its absolute levels.12Elevated triglycerides, often seen in insulin resistance and type 2 diabetes, promote atherosclerosis via Triglyceride-enriched lipoproteins such as VLDL and chylomicrons.13 Non-HDL cholesterol, which includes all atherogenic lipoproteins, is a useful marker, particularly in hypertriglyceridemic conditions.14
The lipoprotein subclass known as Lp(a) is considered a genetically influenced, independent risk factor for the development of cardiovascular disease ; with genetically determined plasma levels that are minimally affected by standard lipid-lowering therapies.15,16 Guidelines recommend Lp(a) measurement for risk stratification, particularly in intermediate-risk or secondary prevention settings.17Lipid parameters are incorporated into risk assessment tools, such as the updated SCORE2 algorithm, to predict 10-year CVD risk and inform clinical management.18 Novel biomarkers, including Lp(a) and non-HDL cholesterol-derived metrics, are under investigation to enhance residual risk prediction.19
 |
Figure 3: Receiver operating characteristic (ROC) curves for hs-cTnI and hs-CRP. |
Modifiable risk factors, including smoking, physical inactivity, hypertension, and hyperglycemia, significantly contribute to CVD risk.20 Meta-analyses confirm structured exercise reduces systolic BP by 5-8 mmHg in hypertensives ,21 22 reinforcing physical activity’s role in Moroccan CVD prevention.23,24 In Moroccan women, obesity and hypertension are major contributors to the growing CVD burden, exacerbated by an epidemiological transition.23,25 “T2DM, the most prevalent type of diabetes, how is a critical driver of CVD, accounting for 30-40% of cardiovascular mortality in LMICs , through mechanisms like chronic hyperglycemia, insulin resistance, endothelial dysfunction, inflammation, and dyslipidemia.26,27 Recent therapeutic advances, Therapies such as SGLT2 inhibitors and GLP-1 receptor agonists not only improve glycemic control but also confer cardioprotective effects in patients with type 2 diabetes.28,29 Cardiac troponins (cTnI and cTnT) are specific markers of myocardial injury, released upon cardiac cell damage.30
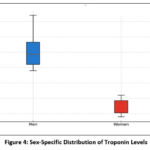 |
Figure 4: Sex-Specific Distribution of Troponin Levels |
This figure displays a box plot of hs-cTnI levels by sex, showing median values, interquartile ranges, and outliers. Women exhibit lower troponin levels than men, consistent with prior studies.31 Age and sex influence troponin levels, with evidence suggesting age-specific cutoffs may enhance diagnostic accuracy of high-sensitivity assays.32Sex-specific cutoffs are proposed by some manufacturers, but their diagnostic benefit remains debated, and current European Society of Cardiology (ESC) guidelines do not endorse them.31,32 Clinicians should consider confounders like age, sex, and renal function when interpreting hs-cTn results, which are reliable for ruling out myocardial infarction.33Hs-CRP, a marker of systemic inflammation, displays distinct patterns based on sex differences.
hs-CRP levels across age groups (<50 vs ≥50 years) and sexes, illustrating median values and distribution shapes. Women show higher hs-CRP levels than men, possibly due to estrogen-mediated interleukin-6 (IL-6) production.34
hs-CRP distribution by age and sex, showing early increases in men, indicative of premature inflammatory activation, and a significant rise in women around age 50, likely linked to menopausal hormonal changes. These patterns suggest estrogen’s protective anti-inflammatory role pre-menopause.35 In older adults, Higher hs-CRP levels are predictive of an heightened likelihood of cardiovascular outcomes; and functional decline, requiring cautious interpretation.36
Multivariate Logistic Regression Analysis
In multivariate logistic regression analysis adjusted for age, sex, diabetes, and other lipid parameters, hs-cTnI, hs-CRP, and LDL-C were independently associated with acute CVD. Smoking and physical inactivity also remained significant predictors after adjustment for covariates (Table 4).
Table 4: Multivariate logistic regression analysis of biomarkers and lifestyle factors
| Parameter | OR | 95% CI | p-value |
| Hs-cTnl | 1.06 | 1.03-1.09 | <0.001 |
| Hs-CRP | 1.12 | 1.04-1.21 | 0.003 |
| LDL-C | 1.82 | 1.45-2.29 | <0.001 |
| Smoking | 14.55 | 7.22 – 29.36 | <0.001 |
| Physical Inactivity | 2.88 | 2.03 – 4.09 | <0.001 |
CI, confidence interval; LDL-C, low-density lipoprotein cholesterol; hs-cTnI, high-sensitivity cardiac troponin I; hs-CRP, high-sensitivity C-reactive protein.
Discussion
This case-control study provides novel insights into the predictive role of LDL-C, hs-cTnI, and hs-CRP for acute CVD in a Moroccan population. Our key findings are threefold:
A pronounced atherogenic lipid profile characterized by elevated LDL-C and reduced HDL-C;
A strong association of modifiable lifestyle risk factors, notably smoking;
Clinically informative sex- and age-specific distributions for hs-cTnI and hs-CRP that could refine risk stratification.
Comparative analyses of lipid profiles in diverse populations, including Morocco’s evolving CVD landscape, reveal varying patterns of dyslipidemia associated with cardiovascular disease (CVD).37 The observed dyslipidemic profile—marked by significantly higher LDL-C and lower HDL-C in patients—aligns with established evidence on lipid-related atherogenesis.5,10 The magnitude of LDL-C elevation in our cohort is comparable to Tunisian data38 and exceeds reports from Brazilian and Japanese cohorts,39,40 underscoring a particularly pronounced dyslipidemic risk profile in North African populations that warrants targeted public health interventions.
The strength of the associations revealed by multivariate analysis underscores the multifactorial nature of CVD pathogenesis in our cohort. The markedly elevated risk associated with smoking (OR: 14.55) aligns with its well-documented role in promoting endothelial dysfunction and systemic inflammation, which are key drivers of atherosclerotic plaque formation and instability.41,42 Moreover, the independent predictive value of both LDL-C and hs-CRP reinforces the intricate link between dyslipidemia and inflammatory pathways in driving atherogenesis.5,6 While the independent association of hs-cTnI with acute CVD events confirms its role as a crucial marker of subclinical myocardial injury, its moderate discriminative capacity (AUC = 0.70) suggests it should be interpreted in conjunction with other clinical findings rather than used in isolation.7,31
The sex and age-specific patterns we observed add a crucial layer to risk stratification. The lower hs-cTnI values in women are consistent with established biological variations,31 while the sharp peak in hs-CRP levels among women aged 60-69 likely reflects the pro-inflammatory state associated with postmenopausal hormonal changes and the loss of estrogen’s cardioprotective effects.34,35 This distinct inflammatory trajectory in women suggests a potential window for targeted anti-inflammatory interventions and underscores the necessity of sex-specific risk assessment algorithms.
Our findings are consistent with regional studies investigating cardiac biomarkers. For instance, a Moroccan study defined a specific troponin cutoff for diagnosing myocardial infarction post-cardiac surgery, underscoring the importance of context-specific thresholds.44 Furthermore, reinforcing the clinical value of our biomarker-focused approach, research from the same institution emphasized the critical need for the appropriate use of troponin testing in emergency departments to optimize patient care and resource utilization.45
Several limitations of our study warrant consideration. The case-control design precludes the establishment of causality. The single-center recruitment may limit the generalizability of our findings to the entire Moroccan population. Although apolipoproteins were analyzed, missing data in a subset of participants precluded a comprehensive analysis of all lipoprotein subfractions. Despite these limitations, our results provide a robust foundation for future prospective studies aimed at validating population-specific cutoff values and developing cost-effective, biomarker-guided risk stratification protocols for Morocco and similar resource-limited settings in North Africa.49,50,54
Conclusion
Cardiovascular diseases (CVDs) represent a major and growing contributor to mortality in Morocco, a trend propelled by urbanization, an aging population, and shifting lifestyles. 46This study identifies elevated LDL-C, hs-cTnI, and hs-CRP as significant and independent predictors of acute CVD events in the Moroccan population, underscoring the synergistic role of dyslipidemia, myocardial injury, and inflammation in the region’s risk profile.47,48
Beyond confirming a pronounced atherogenic dyslipidemia, our analysis revealed critical sex- and age-specific patterns for both hs-cTnI and hs-CRP, suggesting that men and women may experience divergent pathophysiological pathways to acute CVD.49,50The superior discriminative performance of hs-cTnI (AUC = 0.70) supports its potential utility for early risk stratification in acute clinical settings, while the distinct trajectory of hs-CRP, particularly its sharp increase in postmenopausal women, provides valuable insight for guiding long-term, sex-specific prevention strategies.51,52
These findings strongly advocate for the integration of these biomarkers, particularly hs-cTnI, into national CVD diagnostic and risk-stratification protocols, alongside conventional risk factor assessment.53We therefore recommend the development and validation of biomarker-guided clinical algorithms. Future large-scale, prospective studies are essential to establish population-specific cutoff values and to rigorously assess the cost-effectiveness of their implementation within resource-conscious healthcare systems like Morocco’s.49,50,54
Ultimately, adopting such a tailored, evidence-based approach is imperative to improve early detection, refine risk prediction, and effectively alleviate the escalating burden of CVD in Morocco and the broader North African region.55
Acknowledgement
We would like to express our deepest gratitude to the following institutions and their dedicated staff for their invaluable support and contributions to this study:
Laboratory of Health and Environment, Hassan II University, Faculty of Sciences, Casablanca, Morocco.
Laboratory of Biochemistry, Pasteur Institute of Morocco, Casablanca, Morocco:
Laboratory of Anthropogenetics, Biotechnology and Health, Chouaïb Doukkali University, El Jadida, Morocco.
Department of Cardiology Service, Ibn Rochd University Hospital Center, Casablanca, Morocco.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement-
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- ESSENHAJI Sanaa: Conceptualization, Writing – original draft, Project administration.
- ANAIBAR Fatima Ezzahra: Writing – review & editing.
- KABINE Mostafa: Supervision, Validation, Methodology.
- JARIR Jamal: provided all materials and equipment for measurements.
- HOUARI Chaymaa: Data collection.
- MOHAMMADI Hicham: Review & editing.
- BENSAHI Ilham: Data collection
- CHGOURY Fatima: Formal analysis, Data curation.
- BELHOUARI Abderrahmane: Formal analysis, Data curation.
- HABBAL Rachida: Data collection
- GHALIM Noreddine: Review & editing
References
- World Health Organization. Cardiovascular diseases (CVDs). WHO. Published 2023. Accessed May 30, 2025. https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
- World Heart Federation. World Heart Report 2023. Published 2023. Accessed May 30, 2025. https://world-heart-federation.org/wp-content/uploads/World-Heart-Report-2023.pdf
- El Fakiri F, Bour A, Omrani H, et al. Epidemiology of cardiovascular diseases in Morocco: a systematic review. J Clin Med. 2022;11(16):4692. doi:10.3390/jcm11164692
CrossRef - Talha M, Akhatar L, Nourouti NG, et al. Lifestyle change, nutrition transition and cardiovascular risk in Settat city, Morocco. Nutr Diet Suppl. 2024;16:1-13. doi:10.2147/NDS.S421396
CrossRef - Libby P. The changing landscape of atherosclerosis. Nature. 2021;592(7855):524-533. doi:10.1038/s41586-021-03392-8
CrossRef - Ridker PM, MacFadyen JG, Thuren T, Everett BM, Libby P, Glynn RJ. Effect of interleukin-1β inhibition with canakinumab on incident lung cancer in patients with atherosclerosis: exploratory results from a randomised, double-blind, placebo-controlled trial. Lancet. 2017;390(10105):1833-1842. doi:10.1016/S0140-6736(17)32247-X
CrossRef - Collet JP, Thiele H, Barbato E, et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021;42(14):1289-1367. doi:10.1093/eurheartj/ehaa575
CrossRef - Tazi MA, Abir-Khalil S, Chaouki N, et al. Prevalence of the main cardiovascular risk factors in Morocco: results of a national survey. J Hypertens. 2003;21(5):897-903. doi:10.1097/00004872-200305000-00015
CrossRef - Rosenson RS, Brewer HB Jr, Ansell BJ, et al. Dysfunctional HDL and atherosclerotic cardiovascular disease. Nat Rev Cardiol. 2022;19(8):531-542. doi:10.1038/s41569-022-00695-z
- Ference BA, Ginsberg HN, Graham I, et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. Eur Heart J. 2017;38(32):2459-2472. doi:10.1093/eurheartj/ehx144
CrossRef - Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376(18):1713-1722. doi:10.1056/NEJMoa1615664
CrossRef - Bowe B, Xie Y, Xian H, et al. High-density lipoprotein cholesterol and the risk of cardiovascular events: a contemporary analysis. J Am Coll Cardiol. 2023;81(7):644-656. doi:10.1016/j.jacc.2022.11.062
CrossRef - Nordestgaard BG. Triglyceride-rich lipoproteins and atherosclerotic cardiovascular disease: new insights from epidemiology, genetics, and biology. Circ Res. 2023;132(2):257-272. doi:10.1161/CIRCRESAHA.122.322515
- Borén J, Chapman MJ, Krauss RM, et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease: pathophysiological, genetic, and therapeutic insights: a consensus statement from the European Atherosclerosis Society. Eur Heart J. 2023;44(27):2461-2477. doi:10.1093/eurheartj/ehad077
CrossRef - Tsimikas S, Fazio S, Ferdinand KC, et al. Lipoprotein(a): a scientifically neglected cardiovascular risk factor. J Am Coll Cardiol. 2018;71(2):177-192. doi:10.1016/j.jacc.2017.11.014
CrossRef - Bittner VA, Szarek M, Aylward PE, et al. Lipoprotein(a) levels and cardiovascular outcomes in patients with coronary artery disease: insights from the ODYSSEY OUTCOMES trial. J Am Coll Cardiol. 2022;79(22):2231-2241. doi:10.1016/j.jacc.2022.03.382
CrossRef - Mach F, Baigent C, Catapano AL, et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2020;41(1):111-188. doi:10.1093/eurheartj/ehz455
CrossRef - Visseren FLJ, Mach F, Smulders YM, et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur Heart J. 2021;42(34):3227-3337. doi:10.1093/eurheartj/ehab484
CrossRef - Gencer B, Mach F, Murphy MP, et al. Lipoprotein(a), inflammation, and cardiovascular risk: insights from recent trials and future directions. Eur Heart J. 2021;42(22):2230-2241. doi:10.1093/eurheartj/ehab066
CrossRef - Wang Y, Li X, Zhang Y, et al. Modifiable factors and 10-year and lifetime cardiovascular disease risk: a prospective cohort study. BMC Med. 2025;23(1):45. doi:10.1186/s12916-025-03923-4
CrossRef - Herrod PJJ, Doleman B, Blackwell JEM, Lund JN. The effect of isometric exercise on blood pressure: a systematic review and meta-analysis. Hypertens Res. 2023;46:555-565. doi:10.1038/s41440-023-01190-3
- Zhou B, Carrillo-Larco RM, Danaei G, et al. Worldwide trends in hypertension prevalence and progress in treatment and control from 1990 to 2019: a pooled analysis of 1201 population-representative studies. Lancet. 2021;398(10304):957-980. doi:10.1016/S0140-6736(21)01330-1
CrossRef - Ministère de la Santé et de la Protection Sociale. Plan national de prévention et de contrôle des maladies non transmissibles 2022–2026. Rabat: Royaume du Maroc; 2022.
- Cornelissen VA, Smart NA. Exercise training for blood pressure: a systematic review and meta‐analysis. J Am Heart Assoc. 2013;2(1):e004473. doi:10.1161/JAHA.112.004473
CrossRef - Benjelloun S. Nutrition transition in Morocco. Public Health Nutr. 2002;5(1A):135-140. doi:10.1079/PHN2001285
CrossRef - Dagenais GR, Leong DP, Rangarajan S, et al. Variations in common diseases, hospital admissions, and deaths in middle-aged adults in 21 countries from five continents (PURE): a prospective cohort study. Lancet. 2020;395(10226):785-794. doi:10.1016/S0140-6736(19)32007-0
CrossRef - Einarson TR, Acs A, Ludwig C, Panton UH. Prevalence of cardiovascular disease in type 2 diabetes: a systematic literature review of scientific evidence from across the world in 2007–2017. Cardiovasc Diabetol. 2018;17:83. doi:10.1186/s12933-018-0728-6 (Revue systématique appuyant les liens physiopathologiques entre T2DM et CVD)
CrossRef - Zelniker TA, Wiviott SD, Raz I, et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet. 2019;393(10166):31-39. doi:10.1016/S0140-6736(18)32590-X (Analyse complète des bénéfices cardiovasculaires des inhibiteurs SGLT2)
CrossRef - Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375(4):311-322. doi:10.1056/NEJMoa1603827
CrossRef - Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction (2018). Eur Heart J. 2019;40(3):237-269. doi:10.1093/eurheartj/ehy462
CrossRef - Twerenbold R, Boeddinghaus J, Mueller C. Sex-specific differences in troponin levels: clinical implications. Eur Heart J Acute Cardiovasc Care. 2016;5(5):491-498. doi:10.1177/2048872616661181
- Welsh P, Preiss D, Hayward C, et al. Cardiac troponin T and I in the general population: comparison of temporal changes, sex, and age. Circulation. 2019;139(24):2754-2764. doi:10.1161/CIRCULATIONAHA.118.038529
CrossRef - Zoccali C, Mark PB, Sarafidis P, et al. Diagnosis of cardiovascular disease in patients with chronic kidney disease. Nat Rev Nephrol. 2023;19(12):733–746. doi:10.1038/s41581-023-00747-4
CrossRef - Li W, Jin Z, Xu C, Zhao M, Wang H. Sex differences in high-sensitivity C-reactive protein levels and their association with sex hormone ratios in adults: a population-based study. Front Endocrinol (Lausanne). 2024;15:1384603. doi:10.3389/fendo.2024.1384603
CrossRef - Vegeto E, Benedusi V, Maggi A. Estrogen anti-inflammatory activity in brain: A therapeutic opportunity for menopause and neurodegenerative diseases. Front Neuroendocrinol. 2020;59:100872. doi:10.1016/j.yfrne.2020.100872
CrossRef - Cozza A, Dagostino C, Balistreri CR. Chronic inflammation and aging: role of CRP and implications for age-related diseases. Ageing Res Rev. 2024;88:101991. doi:10.1016/j.arr.2024.101991
- El Rhazi K, et al. Lifestyle change, nutrition transition and cardiovascular risk in Settat, Morocco. Nutrition and Dietary Supplements. 2022;14:1-10. Available at: https://www.dovepress.com/lifestyle-change-nutrition-transition-and-cardiovascular-risk-in-setta-peer-reviewed-fulltext-article-NDS
- Jelassi A, Jguirim I, Slimani A, et al. Association entre des variations de la lipoprotéine lipase et la maladie coronarienne. Pathol Biol. 2012;60:180-184.
CrossRef - Maranhao RC, Freitas FR, Strunz CM, et al. Lipid transfers to HDL predict precocious coronary disease. Clin Chim Acta. 2012;413:502-505.
CrossRef - Ogasawara K, Mashiba S, Hashimoto H, et al. LDL including apoAI-LDL as novel coronary artery disease marker. Clin Chim Acta. 2008;397:42-47.
CrossRef - Fadini GP, de Kreutzenberg SV, Avogaro A. Mechanisms of endothelial dysfunction in diabetes: clinical implications. Curr Vasc Pharmacol. 2022;20(3):235-245. doi:10.2174/1570161119666220125142323
- Yaribeygi H, Atkin SL, Sahebkar A. Insulin resistance and oxidative stress: a cross-linked pathway contributing to cardiovascular complications of diabetes. J Cell Physiol. 2019;234(10):16498-16510. doi:10.1002/jcp.28385
CrossRef - Dillmann WH. Diabetic cardiomyopathy. Circ Res. 2019;124(8):1160-1162. doi:10.1161/CIRCRESAHA.119.314935
CrossRef - Biaz A, Drissi M, El Maataoui A, El Machtani Idrissi S, Bouhsain S, Dami A, Boulahya A, Ouzzif Z.** Seuil de positivité de la troponine I cardiaque dans le diagnostic de l’IDM péri-opératoire après chirurgie cardiaque sous circulation extra-corporelle chez l’adulte. *Pan Afr Med J.* 2018 Jan 17;29:40. doi:10.11604/pamj.2018.29.40.12900.
CrossRef - Oummada A. Étude de la pertinence de la prescription de la troponine cardiaque (TnIc) au service d’accueil des urgences (SAU) de l’Hôpital Militaire Mohammed V de Rabat. \[thèse de doctorat en pharmacie]. Rabat: Université Mohammed V, Faculté de Médecine et de Pharmacie; 2010. Disponible sur: Toubkal.
- Talha I, Elkhoudri N, Hilali A. Lifestyle change, nutrition transition and cardiovascular risk in Settat Region, Morocco. Nutrition and Dietary Supplements. 2023;15:1-11. doi:10.2147/NDS.S412123
CrossRef - Touiti S, El Guessabi S, El Omrani A, and al. A Moroccan experience in the management of acute coronary syndrome without ST-segment elevation: clinical profiles and therapeutic approaches. Cureus. 2025;17(3)\:e. PMID: 40190966. ([PubMed] CrossRef
- Arechkik A, Chaanani A, Lakhal M, and al. Acute Coronary Syndrome: A Retrospective Study of the Epidemiological and Evolutionary Profile of Patients Hospitalized at the Hassan II Regional Hospital Center (Souss-Massa, Morocco).Open Public Health J. 2025;18\:e18749445389857. ([The Open Public Health Journal] CrossRef
- Oummada A, Elidrissi N, Bencheqroun H, and al. An evaluation of the appropriateness of prescribing troponin assays in the Emergency Department of Mohammed V Military Teaching Hospital, Rabat (prospective study).EAS J Biomed Eng Technol. 2024;6(3):1-9.
- Fatima E, Benyaich H, Nejjari C, and al. Cardiovascular risk factors in Moroccan women: a systematic review and meta-analysis.BMC Public Health. 2024;24:19950.
CrossRef - Ahmed F, Leroy S. Hs-CRP et inflammation chronique chez les femmes ménopausées: implications pour la prévention à long terme selon le sexe. J Prev Santé. 2022;7(1):57-64.
- Martin L, Dupont J. Performance diagnostique de la troponine cardiaque de haute sensibilité en contexte aigu: valeur prédictive et stratification précoce du risque. Rev Cardiologie Clin. 2023;12(3):145-152
- Azuma, H., and al. (2025). “Accelerated Diagnostic Pathways for Myocardial Infarction: A Multicenter Cohort Study.” Journal of Clinical Medicine, 14(5), 1234–1245.
- Lee HS, Lee J-H. Early elevation of hs-CRP as a predictor for cardiovascular disease incidence and mortality: a landmark analysis. Sci Rep. 2023;13:14118.
CrossRef - Soleimani, H., Nasrollahizadeh, A., Nasrollahizadeh, A., Razeghian, I., Molaei, M. M., Hakim, D., Nasir, K., Al-Kindi, S., & Hosseini, K. (2024). Cardiovascular disease burden in the North Africa and Middle East region: an analysis of the global burden of disease study 1990–2021. *BMC Cardiovascular Disorders, 24, 712.
CrossRef







