Manuscript accepted on :26-08-2025
Published online on: 22-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Preeti Yaduvanshi Parmar
Second Review by: Dr. Wang Yue
Final Approval by: Dr. Mariia Shanaida
Tasnia Binte Bari Kabbo1 , Pritesh Ranjan Dash1,2
, Pritesh Ranjan Dash1,2 , Adila Jahan Anika3
, Adila Jahan Anika3 , Najiha Tarannum Timon3
, Najiha Tarannum Timon3 , Tasnova Nowrin4
, Tasnova Nowrin4 , Shammi Binte Bashar2
, Shammi Binte Bashar2 and Md. Sohel Rana1*
and Md. Sohel Rana1*
1Department of Pharmacy, Jahangirnagar University, Savar, Dhaka, Bangladesh
2Department of Pharmacy, Primeasia University, Banani,Dhaka, Bangladesh
3Department of Pharmacy, University of Science and Technology Chittagong (USTC) Chittagong, Bangladesh
4Department of Management, Southern Utah University, Utah, United States
Corresponding Author Email:pritesh.ju@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3263
Abstract
Recent studies have focused on the pharmacological potentials of natural compounds as potent medicinal agents. Annona reticulata Linn. (Family: Annonaceae) has long been used as a remedy for a wide range of ailments by many tribes and ethnic groups worldwide. According to reports, the plant has a variety of medicinal properties, such as anthelmintic, antipyretic, antihyperglycemic, antiulcer, and antinociceptive activities. In addition to evaluating the plant's antioxidant, cytotoxic, and anti-cancer potentials, the current study used GC-MS analysis to identify the compounds causing these effects. Six established methodologies were utilized to evaluate the antioxidant potential. Good antioxidant properties were demonstrated by the extract in DPPH and nitric oxide radical scavenging tests, with IC50 measurements of 96.15 µg/ml and 124.85 µg/ml. In both DPPH and nitric oxide radical scavenging tests ascorbic acid was used as standard; the observed IC50 values for ascorbic acid in these two tests were 14.38 µg/ml and 67.31 μg/ml. In reducing power assay, the extract's antioxidant activity increased with increasing concentration. 136.4±6.35 mg/gm Ascorbic acid equivalents was the total antioxidant capacity value that was determined. The aqueous extract demonstrated total flavonoid content value of 89.15±0.4 mg/gm Quercetin equivalents and total phenol content value of 61.48±0.17 mg/gm Gallic acid equivalents.Good cytotoxic potential was demonstrated by the extract in the brine shrimp lethality test. This test yielded LC50 value of 93.37 μg/ml for the aqueous leaf fraction and 1.63 μg/ml for standard vincristine sulphate; while the obtained LC90 values for the aqueous leaf extract and standard vincristine sulphate were 516.65 μg/ml and 7.17 μg/ml.Additionally, the plant extract exhibited notable anti-cancer potential against the EAC cell line in in-vivo study. The groups of mice treated with 400 mg/kg dose of aqueous leaf fraction and 20 mg/kg dose of 5-FU demonstrated tumour weight and EAC cell number values of 3.00±0.38 g and 7.40±0.81; 5.08±0.35 g and 9.20±0.86, respectively. Therefore, the values obtained from aqueous extract were less than those of the standard medication 5-FU.
Keywords
Antioxidant effect; Anti-cancer activity; Bullock’s heart; Cytotoxic effect; Extract; Leaves; Tumor
Download this article as:| Copy the following to cite this article: Kabbo T. B. B, Dash P. R, Anika A. J, Timon N. T, Nowrin T, Bashar S. B, Rana M. S. Evaluation of Antioxidant and Cytotoxic Potentials against the EAC cell line of Aqueous Extract of Annona reticulata Linn.leaves. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Kabbo T. B. B, Dash P. R, Anika A. J, Timon N. T, Nowrin T, Bashar S. B, Rana M. S. Evaluation of Antioxidant and Cytotoxic Potentials against the EAC cell line of Aqueous Extract of Annona reticulata Linn.leaves. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/4gFDnwW |
Introduction
It is well known that oxidative stress is one of the main causes of a large number of degenerative and chronic illnesses. Antioxidants are the most efficient method of removing free radicals, which are the source of oxidative stress. By scavenging free radicals or encouraging their breakdown, antioxidants—natural or synthetic—can effectively inhibit the development of such illnesses. The use of herbal resources as natural antioxidants is currently gaining popularity.1
Cancer, which is a collective term for a group of malignant illnesses, remains a major global health concern. The etiology of cancer involves varying degrees of control. Normal cells undergo genetic and epigenetic modifications during this process, which leads to unchecked cell division and, ultimately, cancer. It is also recognised that reactive oxygen species (ROS) can cause DNA mutations, immune system evasion, the tumour microenvironment, metastasis, and angiogenesis, all of which can aid in the development of cancer. The ability of cancer cells to avoid dying due to unidentified mutations is one of their main problems; this causes a buildup of cells that then move to other parts of the body.2
Clinically significant anticancer chemicals are known to be produced by plants. Plants were used to make several of the chemotherapeutic medications that are currently in use.3 Numerous methods are employed to identify plants that might harbour novel physiologically active chemotherapeutic chemicals. One method in use is the ethnomedical data approach, which bases the choice of a plant on what is currently known about the plant’s traditional medicinal usage.3
Furthermore, GC-MS analysis is frequently conducted on plant extracts to identify and quantify volatile and semi-volatile organic components. A thorough chemical profile of the plant is obtained by comparing its chemical profile with an existing library of compounds. It facilitates the identification of plant constituents and the comprehension of the biological roles played by the substances derived from plants.4-5
The current study focused on A.reticulata.because numerous studies have indicated that this herbal medicinal plant has cytotoxic, anti-inflammatory, antioxidant, and anti-cancer properties. Over 100 species make up the genus Annona, and A.reticulata belongs to the Annonaceaefamily.A.reticulata is said to have originated naturally in West Indies, South and Central America.Known by most as the custard apple, A.reticulata is a tiny tree. On average, it reaches a height of 5 to 10 meters. Spreading crown, deciduous, oblong or narrow-lanceolate leaves, and a trunk that can be 25–35 cm thick are the tree’s distinguishing features.6Also known as Bullock’s heart, numerous illnesses have been treated using this traditional plant, such as epilepsy, bacterial infections, worm infestations, fever, ulcers, haemorrhage, dysuria, digestive issues, and more. Indians, Filipinos, and people from a few other countries have also utilised this plant as an anti-inflammatory, anti-stress, and anti-helminthic remedy.7Therefore, decision was taken to look into the antioxidant, cytotoxic, and anti-cancer properties of this traditional herb, A.reticulata.
Materials and Methods
Preparation of plant extract
A.reticulata leaves were gathered from Savar, Dhaka, Bangladesh.In order to prepare it for grinding, the plant material (leaves) was sun-dried separately. After that, drying was conducted in Gallenkamp (Size 1) oven at lower than 50°C temperature.Grinding mill was used to convert the plant components into coarse powders.Dark-colored flasks containing 538 grams of processed dry plant material were sealed with 10.76 liters of water (solvent). Then the container was kept at room temperature. 24 hourslater,Whatman No. 1 filter paper was used to filter the infusions. Then, equivalent amount of solvent(water) was utilized to further extract any leftover residue. The procedure was repeated 48 hours later. Using a rotary evaporator set to 40°C, the combined supernatants were vacuum-dried. The extraction process was considered complete when it was determined that the plant materials were depleted. After extraction, the plant material was dried off and submerged in one liter of deionized water. The plant parts were kept in a sealed container filled with water and shaken and stirred occasionally for a period of seven days. A fresh cotton bed filter was used for each extract. A sticky crude extract concentration was obtained by drying the filtrates at a temperature of 40±2°C. After being extracted, the materials were labeled appropriately and stored at 4°C in sterile sample containers.8
GC-MS Analysis of Aqueous Extract from A.reticulataLeaves
The Shimadzu GCMS-TQ8040 was used to conduct the GC-MS analysis. As the carrier gas, helium was employed. With hold times of 1, 2, and 7 minutes, respectively, the column oven was initially set attemperature of 50°C, then increased to 200°C and finally reached at 300°C, in accordance with the GC settings.At 250°C, the injection was performed in splitless mode, with a sampling duration of one minute, a flow control mode set to pressure at 53.5 kPa, and a total flow of 11 ml/min. Under MS circumstances, the temperature of the interface was 250°C, whereas the temperature of the ion source was 230°C.By comparing with library compounds, this approach provides excellent sensitivity and specificity for identifying and quantifying volatile and semi-volatile compounds in complicated plant extracts.
In-vitro Studies
In-vitro Evaluation of Antioxidant Activity
DPPH Radical Scavenging Assay:
Serial dilution of stock solution was conducted to obtain different concentrations (400 µg/ml, 200 µg/ml, 100 µg/ml, 50 µg/ml, 25 µg/ml). 1 ml of each concentration, appropriately labelled, was contained in test tube. Addition of 2 ml of DPPH solution was done for reaching volume of 3 ml. Incubation (25°C) for 30 min was then conducted. Next, absorbance of diluted extract solution in the solvent was done at 517 nm. Percentage of scavenging activity and IC50 were measured.9-10
Test tubes were filled with 4.0 ml of test fraction and the standard (ascorbic acid) in varying concentrations (400μg/ml, 200μg/ml, 100μg/ml, 50μg/ml, 25μg/ml). The test tubes received 1.0 ml of a Sodium nitroprusside (5mM solution added to them). To finish the reaction, the test tubes were incubated at 30°C for two hours. After taking 2.0 ml of the solution out of the mixture, 1.2 ml of Griess reagent was added. Next, a spectrophotometer was used to assess the solutions’ absorbance at 550 nm in comparison to a blank. The blank was distilled water.11
Reducing Power Assay
2.0 ml of standard solutions of various concentrations or plant extractwas taken in different test tubes. Then, 2.5 ml potassium ferricyanide solution (1%) was added. After that, incubation for 10 minutes at 50°C was conducted and then filled with 2.5 ml of trichloroacetic acid solution (10%). Centrifugation for 10 minutes at 3000 rpm of the mixture was performed.After mixing with 2.5 ml of distilled water, extraction of 2.5 ml of the supernatant solution was done. Then 0.5 ml of ferric chloride solution (0.1%) was added. The absorbance of the solutions was measured at 700 nm using a spectrophotometer. The absorbance of the blank solution at 700 nm was measured in relation to the solvent that was used to make it. Greater reducing power was demonstrated by the reaction mixture’s increased absorbance.
Determination of Total Antioxidant Capacity
Test fraction or standard (300 µl) in various strengths was combined with reagent solution (3 ml). Incubation (95°C) for 90 minutes was conducted. Absorbance of the solution was measured at 695 nm against blank. Antioxidant activity was measured and expressed in amount of ascorbic acid equivalents.12
Determination of Total Phenolics Content
Gallic acid solutions at various concentrations or plant extract (200μg/ml) both in volume of 1.0 ml were taken and kept in test tubes. Folin-Ciocalteu reagent solution (5 ml)that was diluted 10 times was added. After the addition of 4 ml sodium carbonate solution (7.5%), the test tubes were well mixed. The reaction was completed by incubating test tubes containing extract solutions for one hour at the same temperature (20°C). The solutions’ absorbance was measured at 765 nm by using spectrophotometer. A standard blank solution contained ethanol, which dissolved the plant extract.13-14Utilizing standard formula; the total phenolic contents of the plant fraction was measured and expressed as gallic acid equivalents (GAE).16
Determination of Total Flavonoids Content
One ml of reference solution in various concentrations or plant extract (200μg/ml) was taken in test tubes. 3 ml of chloroform was placed in test tubes. Then, eventually added aluminium chloride solution (200 μl), potassium acetate solution (200 μl) and purified water (5.6 ml). Then for completion of the reaction 30 minutes incubation period at room temperature was carried out.Utilizing a spectrophotometer the absorbance was measured at 415 nm and compared to a blank. Chloroform served as the blank.15-17To calculate total amount of flavonoid components standard formula was used and represented in quercetin equivalents.
In-vitro Evaluation of Cytotoxic Potential
Brine Shrimp Lethality Bioassay for Assessing Cytotoxic Activity
38 gm of iodine-free sea salt was dissolved in 1liter distilled water and the mixture was filtered for creation of transparent seawater solution. Artemiasalina leach served as the test organism. Seawater was then poured in small tank and its one side was covered after shrimp eggs were added. It took two days for the shrimps to hatch and become nauplii. During the whole hatching stage, oxygen was continuously supplied. Due to hatchling shrimp’s phototaxis (attraction to light), nauplii free of egg shells were gathered from the tank’s lit section. To enhance vision, the nauplii were taken out of the fish tank and diluted with fresh, clear seawater using a pipette. Ten nauplii were then gently removed using a micropipette. The weight of each test sample was 32 mg.It was dissolved in 200 μl of pure DMSO and then mixed with 20 ml of saltwater. Therefore, 1600 μg/ml was the stock solution’s concentration. By performing serial dilution with sea water the concentration was converted to 800, 400, 200, 100, 50, 25, 12.5, and 6.25 μg/ml. Vincristine sulphate was used in this experiment. Vincristine was tested at extremely low concentrations (10, 5, 1, 0.5, 0.25, 0.125, and 0.06 μg/ml) because to its high cytotoxicity.Three pre-marked test tubes containing 4.95 ml of simulated sea water and ten shrimp nauplii intended to serve as control groups were each filled with 50 μl of DMSO. Magnifying glass was used to examine the test tubes 24 hours later. Then, quantity of survivors was computed.
Microsoft Excel was utilized to analyse concentration-mortality data and calculation of the proportion of mortality was carried out. LC50 measurement demonstrates the efficacy of plant products. Plotting the percentage mortality against the corresponding log of concentration, the linear regression technique estimates the chemical concentration at which 50% of test participants die after a specified exposure time. Additionally, the LC90values for each fraction and standard cytotoxic drug vincristine sulphate were also calculated similarly.18-20
In-vivo Assessment
Experimental Animals
In this investigation, Swiss albino mice having weight of 25–30 g were employed. The mice were taken from Jahangirnagar University’s pharmacy department’s animal home.Pharmacological studies were conducted on A.reticulataleaf extracts inaqueous form at the Jahangirnagar University’s Pharmacology Laboratory. The animals were housed in polypropylene cages with temperatures of 25±2°C, a suitable dark-light cycle, and relative humidity (RH 55±5%) in regular laboratory settings.The mice were fed pelletised mouse feed from ICDDR, B., and they had unrestricted access to water.Every action that involved handling animals was carried out by following the rules set forth by Jahangirnagar University’s animal ethics committee[Ref No: BBEC, JU/M2024/11 (137)].
Assessment of In-vivo Anti-cancer Property against Ehrlich’s Ascites Carcinoma (EAC) Cell Line in Mice-
Collection of Tumor Cells
For transplantation, Ehrlich Ascites Carcinoma (EAC) cells were collected from Jahangirnagar University in Savar, Dhaka. To keep EAC cells alive, Swiss albino mice were given intraperitoneal transplants of 2 × 106 EAC cells every 10 days.
Transplantation of Tumor Cells
The initial step was to use ketamine hydrochloride to make the cancerous mice unconscious. After that, the ascitic fluid was collected using a 1 ml syringe. Following two dilutions with 0.9% NaCl saline solution and a final dilution with phosphate buffer solution, it was ready for cell counting under an electron microscope. After that, tumour cells were injected intraperitoneally into the mice in each group. Each mouse received 0.5 ml of treated ascitic fluid containing 2×106 tumour cells intraperitoneally.21-23
Treatment Protocol
Four groups of five mice each were created from the animals. EAC cells were transplanted intraperitoneally into each mice group. This day of implantationwas considered as the firstday.Treatment started from the next day. Each group received the following treatments in due order:negative control (distilled water, orally), positive control (5-Fluorouracil, 20 mg/kg, intraperitoneally), two doses of aqueous extract (200 and 400 mg/kg, by oral route). Treatment was continued for the following fourteen days. Over the course of the treatment, the mice’s body weight was closely observed.22
Evaluation of Anti-cancer Activity
When the treatment period was over, mice were undergone through overnight fasting and dissected the following day by cervical dislocation. The extraction of ascitic fluid was conducted from peritoneal cavity and blood was collected via the retro-orbital route. Several anti-tumor parameters were meticulously documented, along with some hematological and biochemical markers.23-24
Anti-cancer Parameters
Tumorweight
To ascertain the tumour weight, mice were weighed both prior to and following the ascitic fluid collection.23
Tumor Cell Count
From the mice’s peritoneal cavity, the ascitic fluid was extracted. The fluid was then diluted 100 times using phosphate buffer solution, and a WBC pipette was used to remove the diluted fluid. In order to determine the number of cells, placement of droplet of cell solution (diluted) on Neubauer’s counting chamber was conducted.25For counting the cells, the formula mentioned below was utilized:
Cells/μL = [(Nc × Dc) ÷ (Ns × Vs)]
Here, Nc = Number of cells
Dc = Correction for dilution
Ns = Number of squares counted
Vs = Volume of one square
Hematological Parameters
The blood drawn from each mouse was used to estimate the number of red blood cells (RBC), white blood cells (WBC), and platelets and haemoglobin (Hb%).
Biochemical Parameters
Using the technique outlined by Kumar et al. (2015), the measurement of serum biochemical markers like alanine aminotransferase (ALT), aspartate aminotransferase (AST), bilirubin and creatinine, were done. Every analysis was conducted using commercially available kits.
The SPSS 27.0 software was used to examine the observed data from in-vivo investigations. Using Mean±SEM, the experiment’s results were displayed. Utilising ANOVA, statistical significance was assessed and contrasted with the control at various P value thresholds (*p<0.05, **p<0.01, ***p<0.001).
Results
GC-MS Analysis Report of Aqueous Extract of A.reticulataLeaves
 |
Figure 1: Total ionic chromatogram of A. reticulatacrude Aqueous extractClick here to view Figure |
The library search report indicated that the crude aqueous extract of A.reticulata leaves included 77 components.
Table 1: Constituents detected by GC-MS analysis in A.reticulata’s crude aqueous extract
| Serial No. | Compound Name | Molecular Formula & Molecular Weight | Area |
| 1 | Cyclopropylcarbinol | C4H8O;72.10 g/mol | 17830 |
| 2 | Thiophene-3-ol, tetrahydro-, 1,1-dioxide | C4H8O3S;136.17 g/mol | 17830 |
| 3 | Octodrine | C8H19N;129.24 g/mol | 17830 |
| 4 | 1-(5-Bicyclo[2.2.1]heptyl)ethylamine | C9H19N;141.25 g/mol | 17830 |
| 5 | 1-Methyldecylamine | C11H25N;171.32 g/mol | 17830 |
| 6 | 2-Heptanamine, 5-methyl- | C8H19N;129.23 g/mol | 17830 |
| 7 | Benzeneethanamine, 2,5-difluoro-.beta.,3,4-trihydroxy- | C8H12F2NO3;208.18 g/mol | 17830 |
| 8 | Cystine | C6H12N2O4S2;240.32 g/mol | 17830 |
| 9 | Acetic acid, [(aminocarbonyl)amino]oxo- | C3H6N2O3;118.10 g/mol | 17830 |
| 10 | 4-Fluorohistamine | C5H8FN3;129.14 g/mol | 17830 |
| 11 | Cyclobutanol | C4H8O;72.10 g/mol | 9329 |
| 12 | 1-Methyldecylamine | C11H25N;171.32 g/mol | 9329 |
| 13 | Pentanal | C5H10O;86.13 g/mol | 9329 |
| 14 | D-Alanine | C3H7NO2;89.10 g/mol | 9329 |
| 15 | Eicosanoic acid, methyl ester | C21H42O2;326.55 g/mol | 56805 |
| 16 | Triacontanoic acid, methyl ester | C31H62O2;466.81 g/mol | 56805 |
| 17 | Docosanoic acid, methyl ester | C23H46O2;354.60 g/mol | 56805 |
| 18 | Tridecanoic acid, 12-methyl-, methyl ester | C15H30O2;242.39 g/mol | 56805 |
| 19 | Octadecanoic acid, 17-methyl-, methyl ester | C20H40O2;312.52 g/mol | 56805 |
| 20 | 1,2-Ethanediamine, N-(2-aminoethyl)- | C4H13N3;103.17 g/mol | 15238 |
| 21 | 2-Methylaminomethyl-1,3-dioxolane | C5H11NO2;117.15 g/mol | 15238 |
| 22 | Alanine | C3H7NO2;89.10 g/mol | 15238 |
| 23 | 1,2-Ethanediamine, N-methyl- | C3H10N2;74.13 g/mol | 15238 |
| 24 | Propanamide | C3H7NO;73.10 g/mol | 22359 |
| 25 | 2-Propanamine, 1-methoxy- | C4H11NO;89.14 g/mol | 22359 |
| 26 | dl-Alanine | C3H7NO2;89.10 g/mol | 22359 |
| 27 | Hydroxyurea | CH4N2O2;76.06 g/mol | 22359 |
| 28 | 1-Propanol, 2-amino-, (.+/-.)- | C3H9NO;75.11 g/mol | 22359 |
| 29 | 2-Hexanamine, 4-methyl- | C7H17N;115.22 g/mol | 22359 |
| 30 | Ethanol, 2-(methylamino)- | C3H9NO;75.11 g/mol | 10141 |
| 31 | 1,3-Dioxolane-4-methanol | C4H8O3;104.10 g/mol | 5571 |
| 32 | N-dl-Alanylglycine | C5H10N2O3;146.15 g/mol | 27186 |
| 33 | 1-Octadecanamine, N-methyl- | C19H41N;283.53 g/mol | 27186 |
| 34 | dl-Alanyl-dl-norleucine | C9H18N2O3;202.25 g/mol | 27186 |
| 35 | (S)-(+)-1-Cyclohexylethylamine | C8H17N;127.23 g/mol | 27186 |
| 36 | 1-Methyldecylamine | C11H25N;171.32 g/mol | 27186 |
| 37 | Benzeneethanamine, 2-fluoro-.beta.,3-dihydroxy- | C8H10FNO;155.17 g/mol | 27186 |
| 38 | Propanamide, N-methyl-2-amino- | C4H9N2O;101.13 g/mol | 5151 |
| 39 | 2-Methylaminomethyl-1,3-dioxolane | C5H12NO2;118.16 g/mol | 33954 |
| 40 | Formamide, N,N-dimethyl- | C3H7NO;73.10 g/mol | 17803 |
| 41 | Propanediamide, 2-amino- | C3H10N3;88.14 g/mol | 17803 |
| 42 | L-Alanine, TMS derivative | C9H25NO2Si2;235.48 g/mol | 17803 |
| 43 | 1,3-Dioxolane-4-methanol | C4H8O3;104.10 g/mol | 17803 |
| 44 | 2-(Methylamino)ethanol, O-trimethylsilyl | C6H18NOSi;148.30 g/mol | 17803 |
| 45 | 13-Docosenamide, (Z)- | C22H43NO;337.57 g/mol | 506528 |
| 46 | 3-Ethoxy-1,1,1,5,5,5-hexamethyl-3-(trimethylsilyl)trisiloxane | C11H30O3Si4;322.71 g/mol | 6749 |
| 47 | Trisiloxane, 1,1,1,5,5,5-hexamethyl-3,3-bis(trimethylsiloxy) | C12H36O4Si5;384.86 g/mol | 6749 |
| 48 | Heptasiloxane, 1,1,3,3,5,5,7,7,9,9,11,11,13,13 | C16H48O6Si7;533.17 g/mol | 6749 |
| 49 | 2,4,6-Cycloheptatrien-1-one, 3,5-bis-trimethylsilyl | C13H24OSi2;252.50 g/mol | 6749 |
| 50 | Hexestrol, 2TMS derivative | C24H40O2Si2;416.74 g/mol | 6749 |
| 51 | 3-Butoxy-1,1,1,5,5,5-hexamethyl-3-(trimethylsilyl)trisiloxane | C13H36O3Si4;352.78 g/mol | 6749 |
| 52 | Methyltris(trimethylsiloxy)silane | C10H30O3Si4;310.70 g/mol | 6749 |
| 53 | 1-Isopropoxy-5-propyl-2,3-bis(trimethylsilyl)benzene | C18H38OSi2;326.66 g/mol | 6749 |
| 54 | Thymol, TMS derivative | C13H22OSi;222.40 g/mol | 6749 |
| 55 | Silane, trimethyl[5-methyl-2-(1-methylethyl)phenyl] | C13H24Si;208.41 g/mol | 6749 |
| 56 | Trimethylsilyl-di(trimethylsiloxy)-silane | C9H27O2Si4;279.67 g/mol | 21459 |
| 57 | 3-Isopropoxy-1,1,1,5,5,5-hexamethyl-3-(trimethylsilyl)trisiloxane | C12H34O3Si4;338.75 g/mol | 21459 |
| 58 | 1,4-Bis(trimethylsilyl)benzene | C12H22Si2;222.48 g/mol | 21459 |
| 59 | Ethyl homovanillate, TMS derivative | C13H20O4Si;268.38 g/mol | 21459 |
| 60 | 1,2-Bis(trimethylsilyl)benzene | C12H22Si2;222.48 g/mol | 21459 |
| 61 | 3-Ethoxy-1,1,1,5,5,5-hexamethyl-3-(trimethylsilyl)trisiloxane | C11H32O3Si4;324.73 g/mol | 29787 |
| 62 | Trisiloxane, 1,1,1,5,5,5-hexamethyl-3,3-bis[(trimethylsilyl)oxy] | C12H36O4Si5;384.86 g/mol | 29787 |
| 63 | Silane, trimethyl[5-methyl-2-(1-methylethyl)phenyl] | C13H24Si;208.41 g/mol | 29787 |
| 64 | 1-Isopropoxy-5-propyl-2,3-bis(trimethylsilyl)benzene | C18H35OSi2;323.64 g/mol | 29787 |
| 65 | 3-Isopropoxy-1,1,1,5,5,5-hexamethyl-3-(trimethylsilyl)trisiloxane | C12H34O3Si4;338.75 g/mol | 13013 |
| 66 | Methyltris(trimethylsiloxy)silane | C10H30O3Si4;310.70 g/mol | 13013 |
| 67 | 4-Hydroxyphenyllactic acid, ethyl ester, di-TM | C17H28O4Si2;352.57 g/mol | 20582 |
| 68 | Tetrasiloxane, decamethyl- | C10H30O3Si4;310.70 g/mol | 20582 |
| 69 | Benzoic acid, 4-methyl-2-trimethylsilyloxy-, trimethylsilyl ester | C14H24O3Si2;296.51 g/mol | 20582 |
| 70 | Hexasiloxane, 1,1,3,3,5,5,7,7,9,9,11,11-dodeca- | C12H36O5Si6; 428.95 g/mol | 18997 |
| 71 | 7,7,9,9,11,11-Hexamethyl-3,6,8,10,12,15-hexaoxaheptadecane | C17H36O6;336.46 g/mol | 18997 |
| 72 | (R)-(-)-Phenylephrine, bis(trimethylsilyl) ether | C15H29NO2Si2;311.57 g/mol | 22043 |
| 73 | Silicic acid, diethyl bis(trimethylsilyl) ester | C10H28O4Si3;296.59 g/mol | 22043 |
| 74 | 3-Methylsalicylic acid, 2TMS derivative | C14H24O3Si2;296.51 g/mol | 22043 |
| 75 | Octasiloxane, 1,1,3,3,5,5,7,7,9,9,11,11,13,13,15,15-octamethyl- | C16H48O7Si8;577.26 g/mol | 25366 |
| 76 | Methyltris(trimethylsiloxy)silane | C10H30O3Si4;310.70 g/mol | 25811 |
| 77 | Tetrasiloxane, decamethyl- | C10H30O3Si4;310.70 g/mol | 25811 |
Antioxidant Activity
DPPH Radical Scavenging Assay
IC50 value obtained for aqueous fraction from A. reticulata leaves was 96.15 µg/ml; while for ascorbic acid observed IC50 value was 14.38 µg/ml. The aqueous leaf fraction was found to have a notable level of DPPH radical scavenging function. Aqueous leaf fraction demonstrated dose-dependent DPPH radical scavenging as ascorbic acid (standard) (Figure 2).
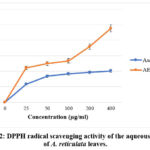 |
Figure 2: DPPH radical scavenging activity of the aqueous extract of A. reticulata leaves.Click here to view Figure |
Nitric Oxide (NO) Radical Scavenging Assay
Aqueous leaf fraction showed good scavenging (observed IC50 value 124.85 µg/ml) in comparison to ascorbic acid, the standard used in this investigation (obtained IC50 value 67.31 µg/ml). Test fraction demonstrated dose-dependent NO scavenging, as ascorbic acid (standard) (Figure 3).
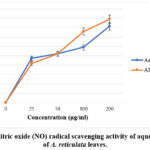 |
Figure 3: Nitric oxide (NO) radical scavenging activity of aqueous extract of A. reticulata leaves.Click here to view Figure |
Reducing Power Assay
The conversion of ferric to ferrous state indicates the reduction activity, which is obtained from the creation of complex having Perl’s Prussian blue colour, was measured spectrophotometrically to evaluate the extracts’ reducing power. The fraction showed good reducing power. As the extract concentration rose, it was discovered that the reduction power also rose (Figure 4).
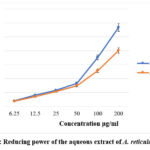 |
Figure 4: Reducing power of the aqueous extract of A. reticulata leavesClick here to view Figure |
Total Antioxidant Capacity:
Phosphomolybdenum method was utilized to determine total antioxidant capacity of test fraction, and represented in ascorbic acid equivalents. Total antioxidant capacity was ascertained by utilizing ascorbic acid standard curve (y=0.0182x+0.4455; R2= 0.9784) (Figure 5). Total antioxidant capacity obtained for aqueous leaf fraction was 136.4±6.35 mg/gm, ascorbic acid equivalents.
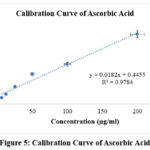 |
Figure 5: Calibration Curve of Ascorbic AcidClick here to view Figure |
Determination of Total Phenolics Content
Gallic acid equivalents (GAE) per gram of fraction were used to express the total phenolic content of the aqueous extract of A. reticulataleaves, which was determined using the Folin-Ciocalteu reagent. Utilizing gallic acid standard curve, the total phenolic contents of the test extract was determined (y=0.0194x+0.1148; R2=0.9997) (Figure 6). In the aqueous extract of A.reticulataleaves, phenols were found to be plentiful, with a numerical value of 61.48±0.17 mg/gm, Gallic acid equivalents.
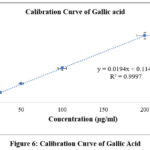 |
Figure 6: Calibration Curve of Gallic AcidClick here to view Figure |
Determination of Total Flavonoid Content
A. reticulata aqueous leaf fraction was tested for total flavonoid content using the aluminium chloride colorimetric technique. Quercetin standard curve (y=0.019x+0.0683; R2=0.9738) (Figure 7) was utilised to calculate quercetin equivalents (QE) per gram of plant fraction in order to ascertain the overall flavonoid content. The aqueous extract of A. reticulata leaves had a quercetin equivalents value of 89.15±0.4 mg/gm of extract, indicating a high quantity of flavonoids.
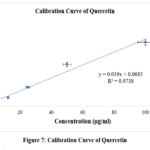 |
Figure 7: Calibration Curve of QuercetinClick here to view Figure |
Cytotoxic Activity
In-vitro Brine Shrimp Lethality Bioassay for Cytotoxic Activity
The potential cytotoxic effect of the extract was investigated using Brine Shrimp Lethality Bioassay. The anticancer medication vincristine sulphate demonstrated LC50 and LC90 values of 1.63 μg/ml and 7.17 μg/ml, while the aqueous leaf extract was shown to be extremely poisonous to brine shrimp nauplii, with LC50 and LC90 values of 93.37 μg/ml and 516.65 μg/ml (Figure 8).
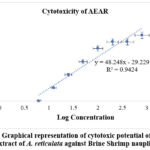 |
Figure 8: Graphical representation of cytotoxic potential of aqueous extract of A. reticulata against Brine Shrimp nauplii.Click here to view Figure |
Evaluation of In-vivo Anti-cancer Potential against Ehrlich’s Ascites Carcinoma (EAC) Cell Line-
Result of Anti-cancer study:
Table 2: Effect of Standard and AEAR on the changes in body weight and tumor weight and No. of EAC cell in EAC induced tumor in mice.
| Group | Body Weight Changes (g) (Mean±SEM) | Tumor Weight (g) (Mean±SEM) | No. of EAC Cell(Mean±SEM) |
| Disease Control | 8.20±0.66 | 8.72±0.46 | 22.20±2.08 |
| Standard | 16.20±0.38*** | 5.08±0.35*** | 9.20±0.86*** |
| AEAR 200 | 3.60±0.51*** | 3.16±0.84*** | 8.20±0.86*** |
| AEAR 400 | 4.60±0.81** | 3.00±0.38*** | 7.40±0.81*** |
*p<0.05 (Significance); **p<0.01 (Highly significance); ***p<0.001 (Very highly significance) against control; Standard is 5-Fluorouracil.
Here, AEAR = Aqueous extract of A.reticulata.
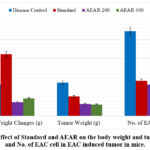 |
Figure 9: Effect of Standard and AEAR on the body weight and tumor weight and No. of EAC cell in EAC induced tumor in mice.Click here to view Figure |
Result of Hematological Parameters in Anti-cancer Study:
Table 3: Effect of Standard and AEAR on the hematological parameters in EAC induced tumor in mice.
| Group | Hb (g/dl) (Mean±SEM) | RBC (106/cmm)(Mean±SEM) | WBC (103/cmm) (Mean±SEM) | Platelet (106/cmm)(Mean±SEM) |
| Disease Control | 8±0.4 | 3.6±0.2 | 15.3±0.6 | 131.2±5.1 |
| Standard | 10.4±0.3* | 2.3±0.1*** | 13.2±0.5* | 88±2.3*** |
| AEAR 200 | 11.9±0.5*** | 4.5±0.2** | 9.3±0.3*** | 166±4.3*** |
| AEAR 400 | 12.6±0.5*** | 4.9±0.1*** | 8.4±0.2*** | 150.2±2.1* |
*p<0.05 (Significance); **p<0.01 (Highly significance); ***p<0.001 (Very highly significance) against control; Standard is 5-Fluorouracil.
Here, AEAR = Aqueous extract of A.reticulata.
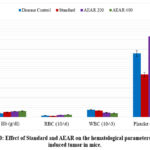 |
Figure 10: Effect of Standard and AEAR on the hematological parameters in EAC induced tumor in mice.Click here to view Figure |
Result of Liver and Kidney Markers in Anti-cancer Study
Table 4: Effect of Standard and AEAR extract on the ALT, AST, Bilirubin& Creatinine level in EAC induced tumor bearing mice.
| Group | ALT (IU/L) (Mean±SEM) | AST (IU/L) (Mean±SEM) | Bilirubin (mg/dl) (Mean±SEM) | Creatinine (mg/dl) (Mean±SEM) |
| Disease Control | 13.9±1.4 | 135.6±12.5 | 4.4±0.7 | 1.9±0.05 |
| Standard | 17.2±1.5 | 90±2.9*** | 8.1±0.4*** | 1.3±0.04*** |
| AEAR 200 | 18.9±0.7* | 87.5±1.2*** | 8.5±0.2*** | 0.9±0.07*** |
| AEAR 400 | 20.9±0.5** | 84.3±0.8*** | 8.9±0.3*** | 0.8±0.05*** |
*p<0.05 (Significance); **p<0.01 (Highly significance); ***p<0.001 (Very highly significance) against control; Standard is 5-Fluorouracil.
Here, AEAR = Aqueous extract of A.reticulata.
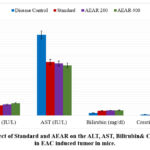 |
Figure 11: Effect of Standard and AEAR on the ALT, AST, Bilirubin& Creatinine level in EAC induced tumor in mice.Click here to view Figure |
Discussion
GC-MS Analysis
Among the identified 77 compounds, some compounds were discovered to have goodantioxidant, cytotoxic, and anti-cancer activities. For example, 2,4,6-Cycloheptatrien-1-one,3,5-Bis-Trimethylsilyl- and Thiophene-3-ol, tetrahydro-, 1,1-dioxide were identified in the extract; both of these compounds are reported to possess antioxidant property.35-36Moreover, some components with good cytotoxic activity were also identified, including ethanediamine derivative, docosanoic acid-methyl ester, and Thiophene-3-ol, tetrahydro-, 1,1-dioxide.39-40Furthermore, cyclopropylcarbinol; thiophene derivative; cyclobutanol; docosanoic acid methyl ester; 1,2-ethanediamine, n-methyl; propanamide and octadecanoic acid 17-methyl-methyl ester were also found in the extract. All these substances are reported to havegood anti-cancer activity.49-53
Evaluation of Antioxidant Activity
There are several ways to evaluate the antioxidant activity of plant extracts. Free radicals have a well-established role in a variety of clinical manifestations. Because they fight free radicals, antioxidants protect us from a number of diseases. By protecting the antioxidant defences or by inhibiting the formation of reactive oxygen species, they function. It is possible to evaluate the ability of natural products to donate electrons by employing purple-colored solution bleaching with 2,20-diphenyl-1-picrylhydrazyl radical (DPPH). Scavenging DPPH is accomplished by introducing an antioxidant or radical species that decolorises the DPPH solution. The amount of colour change depends on the antioxidants’ potency and effectiveness. A significant reduction in the absorbance of the reaction mixture indicates that the tested molecule possesses potent free radical scavenging properties.26-28The superoxide radical is believed to be one of the primary biological sources of reactive oxygen species. Although the superoxide anion is a weak oxidant, it generates strong, dangerous hydroxyl radicals and singlet oxygen, both of which worsen oxidative stress.29Whether the yellow colour of the test solution turns green in a reducing power assay depends on the reducing power of the test specimen. Using reductants in the solution, the Fe3+/ferricyanide combination is reduced to the ferrous form. Fe2+ can therefore be tracked using an absorbance measurement at 700 nm.30 Plant-based phenolic compounds are important due to the scavenging effect of their hydroxyl groups. Plant phenolic compounds fall into several categories, but flavonoids are the most common because of their potent antioxidant qualities. Naturally occurring in plants, flavonoids are thought to enhance human health. Numerous investigations on flavonoid derivatives have shown that they have antibacterial, antiviral, anti-inflammatory, anticancer, and antiallergic activities. The bulk of oxidising molecules, including singlet oxygen, and other free radicals connected to various diseases have been shown to be effectively scavenged by flavonoids.31The plants are capable of synthesizing a wide variety of physiologically important compounds. Antioxidant compounds found in many plants shield cells from the damaging effects of reactive oxygen species (ROS), which can harm cells.32A.reticulata’s aqueous leaf extract was tested for antioxidant capacity using six previously mentioned methods. According to the researches, A.reticulatawas reported to have antioxidant potential.33-34Additionally, the components contained in the aqueous extract were identified using GC-MS analytical analysis. The two potent antioxidants 2,4,6-Cycloheptatrien-1-one,3,5-Bis-Trimethylsilyl- and Thiophene-3-ol, tetrahydro-, 1,1-dioxide were discovered in the aqueous extract of A. reticulata.35-362,4,6-Cycloheptatrien-1-one,3,5-Bis-Trimethylsilyl, has antioxidant properties because it may donate electrons and hydrogen atoms, scavenging free radicals and lowering oxidative stress. Its antioxidant qualities are largely dependent on its structure, especially the tropone ring.37 Because of the thiophene ring’s ability to scavenge free radicals, Thiophene-3-ol, tetrahydro-, 1,1-dioxide can have antioxidant properties. The particular substituents on the thiophene ring affect this action; in general, electron-donating groups improve antioxidant qualities.38
Brine Shrimp Lethality Bioassay for Cytotoxic Activity
Plant fractions that demonstrate LC50 values below 1000 μg/ml are deemed bioactive in this investigation.Theoutcome shows how toxic A.reticulata’saqueousleaf extract is to brine shrimpnauplii. The extract’s lethality was shown to be exactly proportional to its concentration. The percentage of Brine Shrimp nauplii mortality increased in a concentration-dependent manner, indicating the existence of cytotoxic properties. In the Hoechst 33258 fluorescence assay, flavonoids and phenolics have also been demonstrated to exhibit cytotoxicity.Asthe outcomes of total phenolic content and total flavonoid content determination studies demonstrated that, aqueous leaf extract contains considerable amount of phenol and flavonoid contents, this can be a reason behind the extract’s cytotoxic potential. Furthermore, the results of the GC-MS analysis of aqueousfractionfromA.reticulatadiscovered that certain cytotoxic components were present. Potent cytotoxic agents, including ethanediamine derivative, docosanoic acid-methyl ester, and thiophene derivative, are reported to exist in the aqueous fraction.39-40 The cytotoxic activity of several extracts from A.reticulata has also been confirmed by Biswas et al. (2012), Pham et al. (2020), and other researchers.41-42Because of their capacity to interact with and harm DNA, obstruct cell division, or interfere with cellular functions, ethylenediamine derivatives may have cytotoxic effects. These substances, which are frequently employed as ligands in metal complexes, have the ability to cause apoptosis, or cell death, in a variety of cancer cell lines, which makes them promising candidates for the development of anticancer drugs.43Docosanoic acid methyl ester is a fatty acid methyl ester that has the potential to be toxic to cells, especially cancer cells, due to its cytotoxic activity. This activity is probably caused by the compound’s capacity to cause reactive oxygen species generation and/or cell membrane disruption.44By causing apoptosis, interfering with the course of the cell cycle, and preventing microtubule assembly, thiophene derivative can demonstrate cytotoxic action. Cancer cells may be destroyed as a result of these activities.45
Anti-cancer Study:
Experimental animals’ body weight changes during the course of treatment are closely observed during the assessment of the extract’s anti-cancer potential. For ascertaining the anti-cancer property of test materials, the tumor weight and quantity of EAC cells in ascitic fluid are computed for each group.22-24Table 2 makes it evident that the standard 5-FU treated group’s body weight change outcome was quite concerning. The change in body weight for the disease control group was also noteworthy. However, the body weight change outcomes for the groups treated with aqueous extract (both for 200 mg/kg and 400 mg/kg dosages) were lower than those for the disease control and standard groups. It is notable that the body weight change values further lowers with increasing concentration of the test extract. It was found that the tumor weight values obtained from the test extract and 5-FU treatment groups were lower than those obtained from the disease control group. It is also apparent that tumor weight reduces when the aqueous extract dosage increases. Additionally, it was discovered that the quantity of EAC cells observed decreased in both the standard and test extract treated groups; moreover, it was noticed that the amount of EAC cells decreased as the dose of aqueous leaf extract increased. The test extract’s 400 mg/kg body weight dosage produced the strongest anticancer effect, surpassing that of the common medication 5-FU. It is clear from the explanation above that the extract’s anti-cancer activity rises as the dosage does (Figure 11).
Hematological Parameters in Anti-cancer Study
As the body’s homeostasis is severely disturbed during cancer treatment, animals with cancer usually have alterations in the number of red blood cells, white blood cells, and platelets. However, these changes can be reduced with good anti-cancer treatment. Mice typically have hemoglobin content value of 0.08-26.8 g/dL, and their RBC, WBC, and platelet counts are 0.016–14.3×106/cmm, 2–10×103/cmm, and 14–1670×106/cmm, respectively.46Following dissection, blood was drawn from the test animals’ bodies and analysed to ascertain the haemoglobin content as well as the quantity of red blood cells, white blood cells, and platelets. Table 3 shows that aqueous leaf extract at 200 and 400 mg/kg body weight was able to maintain haemoglobin, RBC, WBC, and platelet values within the normal range. This is a very positive indication because many chemotherapy drugs are unable to do so. Thus, it may be concluded that the extract successfully preserved haematological parameters throughout the entire therapeutic duration.
Liver and Kidney Markers in Anti-cancer Study
As vital organs for the body’s overall metabolism and excretion, the liver and kidney may be impacted during cancer treatment. Because cancer changes the body’s normal physiological state, it might impact the kidney and liver. To determine whether test samples can lessen the harmful effects of cancer on these essential organs, various significant liver and kidney markers’levels were calculated from the serum extracted from the blood of animals receiving treatment.ALT, AST, bilirubin, and creatinine levels in mice are generally 17–77 IU/L, 54–298 IU/L, 8–33 mg/dl, and 0.2–0.9 mg/dl, respectively.47Table 4 shows that the kidney marker values were altered in the disease control group, whereas the liver marker values were slightly higher. Nevertheless, the creatinine level was not maintained with the standard medication 5-FU. Aqueous leaf extract, however, efficiently maintained the levels of both liver and kidney indicators at dosages of 200 and 400 mg/kg body weight (the bilirubin level fluctuated very little at the 200 mg/kg dosage). In light of this, it can be concluded that the extract may benefit the kidney and liver when treating cancer.
Overall Discussion about Anti-cancer Study
Ehrlich Ascites Carcinoma is a highly aggressive mammary adenocarcinoma that arises spontaneously in mice (EAC). They are distinguished by fast proliferation, undifferentiation, 100% malignancy, and a shortened life span. These traits make EAC comparable to human malignancies. EAC is used in chemotherapeutic studies because, in addition to its resemblance to human tumours, it is highly susceptible to chemotherapy.48In this study, the extract exhibited strong anti-cancer property against the Ehrlich Ascites Carcinoma cell line. The extract included many anti-cancer compounds, according to the GC-MS analytical report. The aqueous extract contained the following compounds: cyclopropylcarbinol; thiophene derivative; cyclobutanol;docosanoic acid methyl ester; 1,2-ethanediamine, n-methyl;propanamide and octadecanoic acid 17-methyl-methyl ester. According to reports, each of these substances has potent anti-cancer activity.49-53The special reactivity of the cyclopropane ring allows cyclopropylcarbinol, and more generally, compounds containing cyclopropane, to have anticancer action. Strongly electrophilic, the stretched three-membered ring reacts easily with biomolecules like DNA, causing damage to the molecule and ultimately cell death.54By focussing on several cellular mechanisms implicated in the initiation and spread of cancer, thiophene derivatives demonstrate anticancer efficacy. They can cause cell cycle arrest and apoptosis, interfere with signalling pathways, block particular proteins linked to cancer, and even make cancer cells more sensitive to other treatments.55Cyclobutanol can have anticancer effects by improving the stability, water solubility, and DNA binding affinity of platinum complexes, especially when it is added to them. The shape, characteristics, and interactions of the molecule with biological targets can be influenced by the distinct structure of the cyclobutane ring, which includes its puckered conformation and bond angles. This could result in enhanced anticancer efficacy.56 Methyl behenate, another name for docosanoic acid methyl ester, has anticancer properties mainly due to its capacity to stop cancer cells from proliferating and to cause them to undergo apoptosis. In terms of regulating the proliferation of breast cancer cells (MCF-7), it has been demonstrated to be more effective than other fatty acid esters, such as hexadecanoic acid ethyl ester.57Through a variety of methods, including interactions with biological components such as DNA and proteins, 1,2-ethanediamine, N-methyl can demonstrate anticancer activity. These mechanisms can result in cell cycle arrest, apoptosis, and the prevention of tumour growth.58By causing apoptosis, preventing the growth of cancer cells, and interfering with biological functions including mRNA processing, propanamide derivatives can have anticancer effects. In particular, it has been demonstrated that certain propanamide derivatives are cytotoxic to cancer cells but have little impact on healthy cells, indicating the possibility of targeted cancer treatment.59By causing apoptosis (programmed cell death) and preventing the proliferation of cancer cells, octadecanoic acid 17-methyl-methyl ester, has demonstrated possible anticancer action.60Some researchers have investigated a number of A.reticulata extracts for anti-cancer property.61-63The anti-cancer potential of A. reticulata’s aqueous leaf fraction was, however, assessed for the first time in it.
Conclusion
The investigation highlights that aqueous leaf fractionfromA.reticulata has considerable pharmacological potentialssuch as antioxidant, cytotoxic, and anticancer activities. GC-MS analytical data indicates the compounds which lead to these bioactivities. Outcomes of six in-vitro antioxidant studies demonstrated the presence of good antioxidant properties in the test extract. Brine shrimp lethality bioassay illustrated LC50 and LC90 values for both test extract and standard drug; by comparing the values it can be said that the aqueous leaf extract has strong cytotoxic potentials. In-vivo assessment of anti-cancer activity against EAC cell line demonstrated that, aqueous leaf extract has notable anti-cancer activity as it lowers the values of tumor weight and tumor cell count even more than the standard drug 5-FU. However, isolating the bioactive compounds from the extract and testing them, may help clarify the mechanism of action that led to the various bioactivities and may open the door to drawing a firm conclusion about the current findings.
Acknowledgment
The authors express gratitude to the Department of Pharmacy, Jahangirnagar University for laboratory and logistical support.
Funding Source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
Every action that involved handling animals was carried out by following the rules set forth by Jahangirnagar University’s animal ethics committee[Ref No: BBEC, JU/M2024/11 (137)].
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Md. Sohel Rana: Supervision(lead), investigation (lead), conceptualization (lead),formal analysis (supporting), funding acquisition (equal), writing – review and editing (equal).
- TasniaBinte Bari Kabbo: Data curation (lead),formal analysis (lead), funding acquisition (equal), investigation (lead),methodology (lead), project administration (equal), resources (lead),software (lead), validation (equal), visualization (lead), writing – originaldraft (lead), writing – review and editing (equal).
- PriteshRanjanDash: Supervision(equal), formal analysis (supporting), investigation (equal),data curation (equal), writing – review and editing (equal).
- Adila Jahan Anika: Data curation (equal),formal analysis (equal),
- NajihaTarannumTimon: Data curation (equal),formal analysis (equal),
- TasnovaNowrin:Data curation (equal),formal analysis (equal),
- ShammiBinte Bashar:Data curation (equal), formal analysis (equal).
References
- Souri E, Amin R, Jalalizadeh H, Barezi S. Screening of thirteen medicinal plant extracts for antioxidant activity. Iranian Journal of Pharmaceutical Research. 2008;7(2):149-154
- Canga I, Vita P, Oliveira I, Castro A, Pinho C. In Vitro Cytotoxic Activity of African Plants: A Review. In Molecules. 2022;27(15):4989.
CrossRef - Deniz U, Güneş H, Gülneş F, Mammadov R. Cytotoxic activities of certain medicinal plants on different cancer cell lines. Turkish Journal of Pharmaceutical Sciences.2017;14(3): 222–230.
CrossRef - Mastovska K, Dorweiler K, Lehotay S, et al. Pesticide multiresidue analysis in cereal grains using modified QuEChERS method combined with automated direct sample introduction GC-TOFMS and UPLC-MS/MS techniques. Journal of Agricultural and Food Chemistry. 2010;58(10):5959-5972.
CrossRef - Ponnamma S, Manjunath K. GC-MS Analysis of phytocomponents in the methanolic extract of Justiciawynaadensis (nees). Int J Pharm Bio Sci. 2012;3(3):570-576.
- Reddy P, Shantha R, Bharathi V, Kumar K, Venkateshwarlu G. Pharmacognostical evaluation on the Medicinal & Nutritive fruits of Raamaphal-Annona reticulata L. Journal of Pharmacognosy and Phytochemistry. 2015;4(1):21-28.
- Wen W, Lin Y, Ti Z. Antidiabetic, Antihyperlipidemic, Antioxidant, Anti-inflammatory Activities of Ethanolic Seed Extract of Annona reticulata L. in Streptozotocin Induced Diabetic Rats. Frontiers in Endocrinology. 2019;10:716.
CrossRef - Stankovic S, Niciforovic N, Mihailovic V, Topuzovic M, Solujic S. Antioxidant activity, total phenolic content and flavonoid concentrations of different plant parts of Teucriumpolium L. subsp.polium. ActaSocietatisBotanicorumPoloniae. 2012; 81(2):117–122.
CrossRef - Gulcin İ, Alwasel H. DPPH Radical Scavenging Assay. In Processes. 2023;11(8):2248.
CrossRef - Benslama A, Harrar A. Free radicals scavenging activity and reducing power of two Algerian Sahara medicinal plants extracts. International Journal of Herbal Medicine. 2016;4(6):158–161.
CrossRef - Parul R, Kumar S, Saha P. In-Vitro Nitric Oxide Scavenging Activity of Methanol Extracts Of Three Bangladeshi Medicinal Plants. The Pharma Innovation Journal. 2013;1(12):83-88.
- Kamath D, Samshuddin S, Arunkumar D, Avinash G. Determination of total phenolic content and total antioxidant activity in locally consumed food stuffs in Determination of total phenolic content and total antioxidant activity in locally consumed food stuffs in Moodbidri, Karnataka, India. Advances in Applied Science Research. 2015;6(6):99-102.
- Pathak K, Das A. Assessment of antioxidant activity of different extracts of Annona reticulata (L). International Journal of Pharmaceutical Sciences and Research. 2018;9(6):2431-2437.
- Benslama A, Harrar A. Free radicals scavenging activity and reducing power of two Algerian Sahara medicinal plants extracts. International Journal of Herbal Medicine. 2016;4(6):158–161.
CrossRef - Lallianrawna S, Muthukumaran R, Ralte V, Gurusubramanian G, Kumar S. Determination of total phenolic content, total flavonoid content and total antioxidant capacity of Ageratinaadenophora (Spreng.). King & H. Science vision. 2013;13(4):149-156.
- Khan S, Rafatullah M, Sani M, Shaheen M, Sani M, Razlan, M. Determination of Total Phenolic Content, Total Flavonoid Content and Antioxidant Activity of Various Organic Crude Extracts of LicualaSpinosa Leaves from Sabah, Malaysia. ASM Science Journal. 2018;11(3):53-58.
- Khatiwora E, Kulkarni M, Adsul B, Kulkarni M, Deshpande R, Kashalkar V. Spectroscopic determination of total phenol and flavonoid contents of Ipomoea carnea. International Journal of ChemTech Research. 2010;2(3):1698-1701.
- Waghulde S, Kale K, Patil V. Brine shrimp lethality assay of the aqueous and ethanolic extracts of the selected species of medicinal plants. In Proceedings. 2019;41(1):47.
CrossRef - Tawaha A. Cytotoxicity evaluation of Jordanian wild plants using brine shrimp lethality test. Jordan journal of applied science natural sciences. 2006;8(1):12.
- Olowa F, Nuñeza M. Brine Shrimp Lethality Assay of the Ethanolic Extracts of Three Selected Species of Medicinal Plants from Iligan City, Philippines. International Research Journal of Biological Sciences. 2013;2(11).
- Prosanta P, ChakrabortyM, Karmakar I, et al. Evaluation of Anticancer Activity of Methanol Extract of Monsteradeliciosa in EAC Induced Swiss Albino Mice. International Journal of Toxicological and Pharmacological Research. 2015;7(4):165–170.
- Raju A, Christina A, Murali A. Antitumor activity of ethanol and aqueous extracts of DroseraBurmanniivahlin EAC Bearing Mice. Spatula DD. 2012;2(2):83.
CrossRef - Lavakumar V. Anticancer and antioxidant effect of Acanthophoraspicifera against EAC induced carcinoma in mice. Journal of Pharmacy Research. 2012;5(3):1503–1507.
- Reddy N. Evaluation of anticancer activity of Ruellia tuberose on eac induced solid tumor. International Journal of Phytopharmacology. 2013;4(5):340-346.
- Haldar K, Kar B, Bala A, Bhattacharya S, Mazumder K. Antitumor activity of Sansevieriaroxburghiana rhizome against Ehrlich ascites carcinoma in mice. Pharmaceutical Biology. 2010; 48(12):1337–1343.
CrossRef - Nunes X, Silva F, Guedes J, Almeida S. Biological oxidations and antioxidant activity of natural products. Phytochemicals as nutraceuticals-Global Approaches to Their Role in Nutrition and Health. 2012;278.
- Umamaheswari M, Chatterjee K. In vitro antioxidant activities of the fractions of Cocciniagrandis L. leaf extract. African Journal of Traditional, Complementary and Alternative Medicines. 2008;5(1):61-73.
CrossRef - Krishnaiah D, Sarbatly R, Nithyanandam R. A review of the antioxidant potential of medicinal plant species. Food and bioproducts processing. 2011;89(3): 217-233.
CrossRef - Alves Q, Jorge D, Juceni P, et al. Methods for determination of in vitro antioxidant activity for extracts and organic compounds. Química Nova. 2010;33:2202-2210.
CrossRef - Saeed N, Khan R, Shabbir M. Antioxidant activity, total phenolic and total flavonoid contents of whole plant extracts Torilisleptophylla L. BMC complementary and alternative medicine. 2012;12:1-12.
CrossRef - Ao C, Li A, Elzaawely A, Xuan D, Tawata S. Evaluation of antioxidant and antibacterial activities of Ficusmicrocarpa L. fil. extract. Food control. 2008;19(10): 940-948.
CrossRef - Padmaja M, Sravanthi M, HemalathaJ. Evaluation of Antioxidant Activity of Two Indian Medicinal Plants. Journal of Phytology. 2011;3(3).
- Jayaprakash A. Phytochemicals, antimicrobial and antioxidant properties of Annona reticulata Linn. JAIR. 2017;6(6):90-95.
- MondalK, Saha P, Mondal N, Mazumder U. Free radical scavenging property of Annona reticulata leaves. Oriental Pharmacy and Experimental Medicine. 2008;8(3):260–265.
CrossRef - Devi B, Barkath N, Vijayaraghavan P, Rejiniemon S. GC-MS analysis of phytochemical from Psidiumguajavalinn leaf extract and their in vitro antimicrobial activities. J. Pharma Biol. Sci. 2018; 8:583–589.
- Shah R, Verma K. Therapeutic importance of synthetic thiophene. Chemistry Central Journal. 2018;12(1).
CrossRef - Charlton C, Mastyugin M, Török B, Török M. Structural Features of Small Molecule Antioxidants and Strategic Modifications to Improve Potential Bioactivity. Molecules. 2023; 28(3):1057.
CrossRef - Tavadyan A, Manukyan H, Harutyunyan H, Musayelyan V, Sahakyan D, Tonikyan G. Antioxidant Properties of Selenophene, Thiophene and Their Aminocarbonitrile Derivatives. Antioxidants (Basel). 2017;6(2):22.
CrossRef - Musiliyu A, Veera D, Badisa M. Cytotoxic Activity of N, N’-Bis (2-hydroxybenzyl) ethylenediamine Derivatives in Human Cancer Cell Lines. Anticancer Research. 2014; 34:1601-1608.
- Nisa S, Bibi Y, Masood S, et al. Isolation, Characterization and Anticancer Activity of Two Bioactive Compounds from Arisaemaflavum (Forssk.).Schott. Molecules. 2022;27(22):7932.
CrossRef - Biswas S, Shahriar M, Khanam A, Ahsan R. Investigation of Antioxidant, In-vitro Cytotoxic, and In-vivo Antitumor Effects of Leaf Extracts of Annona Reticulata. Bangladesh Journal of Microbiology. 2012;29(2):70-74.
CrossRef - Pham V, Le Q, Le T, Vo Q, Ho V. Phytochemical constituents of Annona reticulata and their cytotoxic activity. Letters in Organic Chemistry. 2020;17(3):206-210.
CrossRef - Bracci L, Schiavoni G, Sistigu A, Belardelli F. Immune-based mechanisms of cytotoxic chemotherapy: implications for the design of novel and rationale-based combined treatments against cancer. Cell Death Differ. 2014;21(1):15-25.
CrossRef - Santos J, Calzada F, Camacho Y, et al. Evaluation of the Anti-Toxoplasma gondii Efficacy, Cytotoxicity, and GC/MS Profile of Pleopeltiscrassinervata Active Subfractions. Antibiotics (Basel). 2023;12(5):889.
CrossRef - Santos A, Pereira C, Oliveira B, Junior B, Lima A, Pitta R, Rêgo B, Pitta G. Anticancer properties of thiophene derivatives in breast cancer MCF-7 cells. Anticancer Drugs. 2018;29(2):157-166.
CrossRef - O’Connell E, Mikkola M, Stepanek M, et al. Practical murine hematopathology: a comparative review and implications for research. Comp Med. 2015;65(2):96-113.
- Santos W, Oliveira C, Hastreiter A, et al. Hematological and biochemical reference values for C57BL/6, Swiss Webster and BALB/c mice. Brazilian Journal of Veterinary Research and Animal Science. 2016;53(2):138-145.
CrossRef - Elsherbiny M, Al-GayyarH. Anti-tumor activity of arjunolic acid against Ehrlich Ascites Carcinoma cells in vivo and in vitro through blocking TGF-β type 1 receptor. Biomedicine & Pharmacotherapy. 2016;82:28–34.
CrossRef - Dembitsky M. Naturally occurring bioactive Cyclobutane-containing (CBC) alkaloids in fungi, fungal endophytes, and plants. Phytomedicine. 2014;21(12):1559–1581.
CrossRef - Nisa S, Bibi Y, Masood S, et al. Isolation, Characterization and Anticancer Activity of Two Bioactive Compounds from Arisaemaflavum (Forssk.).Schott. Molecules. 2022; 27(22):7932.
CrossRef - Musa A, Badisa L, Latinwo M. Cytotoxic activity of N, N’-Bis (2-hydroxybenzyl) ethylenediamine derivatives in human cancer cell lines. Anticancer Research. 2014;34(4), 1601-1607.
- Zhang J, Hao G, Xin W, Yang F, Zhu M, Zhou H. Design, synthesis, and structure–activity relationship of 7-propanamide benzoxaboroles as potent anticancer agents. Journal of Medicinal Chemistry. 2019;62(14):6765-6784.
CrossRef - Mathai V, Sar K, Mitra C, Jindal K, Wang F. Ethanolic extraction and GC-MS analysis of antioxidant and anticancer bioactive compounds from Menthaarvensis and Aegle marmelos. Natural Product Research. 2024:1-7.
CrossRef - Ali E, Aziz A, Bräse S, et al. Unveiling the Anticancer Potential of a New Ciprofloxacin-Chalcone Hybrid as an Inhibitor of Topoisomerases I & II and Apoptotic Inducer. Molecules. 2024,29(22):5382.
CrossRef - Pathania S, Chawla A. Thiophene-based derivatives as anticancer agents: An overview on decade’s work. Bioorganic Chemistry. 2020;101:104026.
CrossRef - Lichota A, Gwozdzinski K. Anticancer Activity of Natural Compounds from Plant and Marine Environment. J. Mol. Sci. 2018;19(11):3533.
CrossRef - Jóźwiak M, Filipowska A, Fiorino F, Struga M. Anticancer activities of fatty acids and their heterocyclic derivatives. European journal of pharmacology. 2020;871:172937.
CrossRef - Altaher M, Adris A, Aliwaini H, Awadallah M, Morjan Y. The Anticancer Effects of Novel Imidazo[1,2-a]Pyridine Compounds against HCC1937 Breast Cancer Cells. Asian Pac J Cancer Prev. 2022;23(9):2943-2951.
CrossRef - Hansen T, Ausbacher D, Zachariassen G, Anderssen T, Havelkova M, Strøm B. Anticancer activity of small amphipathic β2, 2-amino acid derivatives. European journal of medicinal chemistry. 2012;58:22-29.
CrossRef - Mostofa G, Reza A, Khan Z, et al. Apoptosis-inducing anti-proliferative and quantitative phytochemical profiling with in silico study of antioxidant-rich Leeaaequata L. leaves. Heliyon. 2024;10(1).
CrossRef - Khan N, Kumar N, Ballal A, Datta D, Belle S. Unveiling antioxidant and anti-cancer potentials of characterized Annona reticulata leaf extract in 1, 2-dimethylhydrazine-induced colorectal cancer in Wistar rats. Journal of Ayurveda and Integrative Medicine. 2021;12(4): 579-589.
CrossRef - Rao P, Shenoy V, NayakG, et al. Evaluation of the merit of ethanolic extract of Annona reticulata as an anti-cancer agent in human colon cancer cell lines (HCT-116). F1000Research. 2024;12:1571.
CrossRef - Suresh H, Shivakumar B, Shivakumar S. Phytochemical Potential of Annona reticulata Roots for Antiproliferative Activity on Human Cancer Cell Lines. Advances in Life Sciences. 2012;2(2), 1–4.
CrossRef







