Manuscript accepted on :10-09-2025
Published online on: 25-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Emmanuel Dike and Dr. Kulvinder Kaur
Second Review by: Dr. Pratheepa Sivashankari N
Final Approval by: Dr. Anton R Keslav
Sentot Joko Raharjo1* , Dewi Ratih Tirto Sari2
, Dewi Ratih Tirto Sari2 Ernanin Dyah Wijayanti3
Ernanin Dyah Wijayanti3 and Yanty Maryanty4
and Yanty Maryanty4
1Department of Pharmacy and Food Analyst, Polytechnic of Health of Putra Indonesia Malang. Malang, Indonesia.
2Department of Pharmacy, Ibrahimy University, Situbondo, Indonesia.
3Department of Pharmacy, Polytechnic of Health of Putra Indonesia Malang. Malang, Indonesia.
4Department of Chemical Engineering, State Polytechnic of Malang, Malang, Indonesia.
Corresponding author e-mail address: sentotjoko@poltekkespim.ac,id
DOI : https://dx.doi.org/10.13005/bpj/3241
Abstract
Ginger Simplicia (Zingiber officinale Rosc.) contains various secondary metabolic active compounds, but their levels often vary due to differences in plant variety, harvesting, drying, and storage. Standardization is crucial to ensure consistent quality, efficacy, and safety. This study analyzed the chemical composition of active compounds in standardized ginger simplicia. Key quality parameters, including water- and ethanol-soluble extractives, moisture, ash content, microbial contamination, and phytochemical profiles, were evaluated. Extracts were prepared using water and n-hexane solvents, and volatile oils were analyzed by LC-MS and GC-MS. All parameters met established quality standards. Total phenolic content was 3.15 ± 0.04 mg GAE/g, flavonoid content 1.42 ± 0.46 mg QE/g, and total terpenoid content 38.82 ± 0.13 mg/g. Water extracts contained simple phenols (ellagic acid), flavonoid glycosides (quercetin-3-O-rhamnoside), flavonoids (kaempferide), diarylheptanoids ([6]-shogaol), and sesquiterpenoids (alpha-zingiberene). n-hexane extracts were rich in diarylheptanoids ([6]-gingerol), monoterpenoids ((E)-3,7-dimethyl-3,6-octadienal), and steroids (beta-sitosterol). The volatile oil fraction was dominated by sesquiterpenoids (L-zingiberene) and monoterpenoids (geranial). These findings highlight the diverse bioactive compounds in standardized ginger simplicia, supporting its therapeutic potential and emphasizing the importance of rigorous standardization for quality assurance.
Keywords
Active compounds; Essential oils; Ginger simplicia; Standardization; Water/n-hexane extracts
Download this article as:| Copy the following to cite this article: Raharjo S. J, Sari D. R. T, Wijayanti E. D, Maryanty Y. Chemical Composition and Bioactive Constituents of Standardized Ginger (Zingiber officinale Rosc.) Simplicia. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Raharjo S. J, Sari D. R. T, Wijayanti E. D, Maryanty Y. Chemical Composition and Bioactive Constituents of Standardized Ginger (Zingiber officinale Rosc.) Simplicia. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/429vEkK |
Introduction
Differences in the content of active compounds in Zingiber officinale Rosc. simplicia are influenced by several factors, namely genetic factors/ varieties,1 the growing environment,2 and post-harvest processing methods.3 Each ginger variety has different genetic properties, resulting in different active compounds.4 For example, small white ginger (Z. officinale var. amarum) contains more phenolics than other varieties, while red ginger (Z. officinale var. rubrum) contains more zingiberene and ar-curcumene compounds. This difference occurs because the genes that regulate the production of these compounds are different in each variety.5 The growing environment also has an effect, for example, ginger planted in places with an altitude of more than 200 meters and cooler temperatures will produce more defense compounds such as flavonoids. Soil type is also important,6 sandy loam soil is different from latosol soil,7 in terms of nutrients and pH, thus affecting plant metabolism. In addition, sufficient light and high humidity help the process of photosynthesis, which increases the basic materials for making active compounds such as terpenoids.8 After harvest, the processing method also determines how much active compound remains. Drying by sun drying or air drying is better in maintaining flavonoid content (around 86.81 mg QE/g), compared to drying with an oven at a temperature of 50°C which reduces flavonoid levels to 38.96 mg QE/g because heat can damage the compound. In the extraction process, using ethanol solvent is more effective in extracting zingiberene compounds compared to ethyl acetate solvent.9 Storage also affects the quality of active compounds. Storing red ginger powder at 30°C can reduce its shelf life to 212 days due to a decrease in vitamin C content and changes in color. Additionally, using kraft paper packaging is not effective in preventing the loss of volatile compounds such as geraniol.10 Overall, the difference in active compound content occurs because plants respond to environmental conditions such as temperature and soil pH by producing defense compounds. However, improper drying and storage processes can cause damage to important compounds due to enzyme reactions and oxidation.11 Therefore, it is important to choose the right variety, appropriate planting location, and good post-harvest processing techniques so that ginger simplicia has the best quality.12
Before determining the active compounds in ginger herbal medicine, it is necessary to standardize the herbal medicine. This standardization is important to ensure quality and consistency, maintain the stability of the active compounds, prevent contamination or damage, and ensure the effectiveness and safety of its use. With standardization, ginger herbal medicine used as raw material has uniform quality and meets certain requirements, such as water content, ash content, water-soluble or ethanol extract content, and active substance content according to applicable standards, such as SNI, Indonesian Materia Medika or Indonesian Herbal Pharmacopoeia.13,14 Without standardization, the quality of raw materials can vary due to variations in the harvesting, drying, or storage processes, so that the results of the analysis of active compounds are inconsistent and difficult to reproduce. The process of making herbal medicines, such as drying and grinding, aims to stabilize the material by stopping or slowing down enzymatic reactions that can damage active compounds. Standardized herbal medicines are also more resistant to the growth of microorganisms such as bacteria and fungi, so that their shelf life is longer and the content of active compounds is maintained.15,16 On the other hand, unstandardized herbal medicines are more easily contaminated by soil or microbes, and are susceptible to damage, for example due to excessively high water content, which can reduce their quality and pharmacological effectiveness. Standardization includes the process of cleaning, drying, and proper storage so that herbal medicines are safe to consume and suitable for use as research materials or medicines. In addition, standardization also ensures that the herbal medicines are effective and safe to use.17 Only simplicia that meets quality standards can provide the expected pharmacological effects and are safe for users. Standardization is also important so that the results of the active compound content test truly reflect the optimal potential of the raw material, without being affected by external factors such as damage or contamination. Thus, standardization of ginger simplicia is an important step to obtain accurate, reliable active compound analysis results that reflect the original quality of ginger.12 Ginger simplicia has a variety of secondary metabolic content that is inconsistent and difficult to reproduce due to differences in variety, harvesting process, drying, or storage. Standardization ensures the quality and uniformity of the material, maintains the stability of active compounds, prevents contamination and damage, and ensures efficacy and safety.18
The extraction of active compounds from Zingiber officinale Rosc herbal medicine can be accomplished using different techniques and solvents. Among the most frequently utilized solvents are water and n-hexane. Water is primarily used for extracting polar compounds, whereas n-hexane is more suitable for isolating non-polar compounds. Additionally, essential oils are typically obtained through steam distillation. These differences in extraction methods lead to variations in the active compound profiles found in each type of extract.19 Therefore, a thorough understanding of the active compounds present in both water and n-hexane extracts is crucial for maximizing the therapeutic potential of Zingiber officinale Rosc. To gain a comprehensive overview of the chemical constituents in Zingiber officinale Rosc, the analysis of active compounds is performed using advanced chromatographic techniques. Liquid or Gas Chromatography-Mass Spectrometry (LC-MS/GC-MS) is employed to identify active compounds present in water extracts, n-hexane extracts, and essential oils. These techniques enable the precise detection and quantification of the compounds, offering valuable insights into the chemical profile of Zingiber officinale Rosc. The objective of this study is to detail the chemical composition of active compounds in standardized Zingiber officinale Rosc by integrating standardization data with LC-MS/GC-MS analysis. By doing so, the research aims to thoroughly investigate the active compound composition of standardized ginger products. The findings are expected to support the development of higher-quality products that can be utilized in both the health and culinary sectors.
Materials and Methods
Procedures
Preparation of ginger simplicia
Ginger is washed, drained, broken to reduce size, and then dried in an oven at 50°C. Dried ginger (Zingiber officinale Rosc.) is then ground and sieved to form a simplicia powder.20
Simplicia standardization
Water content
A 5-gram sample was put into a porcelain cup that had been tared and then dried using an oven at 105°C for 5 hours. The sample was then placed in a desiccator and weighed. Drying and weighing were continued at an interval of 1 hour until the difference between two consecutive weighing’s was no more than 0.25%. The water content of simplicia is calculated by the formula:
![]()
Where w0 is the initial weight of the simplicia and w1 is the final weight of the simplicia.20
Ash content
A sample of 2 to 3 g is placed in a silicate krus that has been incinerated and tared. Next, the sample is incinerated until the charcoal runs out, cooled, and weighed. The ash content of simplicial is calculated by the formula20:
![]()
Total microbial contamination
The number of microbial contaminants was determined by total plate count (TPC) and total fungal count (TFC). Serial dilution of samples was carried out using 0.85% NaCl until a dilution of 10-5 was obtained. Each dilution was inoculated on plate count agar (PCA) media for TPC and potato dextrose agar (PDA) media for TFC. Incubation was carried out at 30°C for 24-48 hours. The number of colonies that grew was counted according to standard plate count requirements.21
Water-soluble extract
The sample was weighed as a total of 5 g, and put into a sealed flask. Then 100 mL of chloroform-saturated water (1mL chloroform:100mL distilled water) was added, shaken several times for the first 6 hours, and left for 18 hours. Then, the mixture was filtered, and 20.0 mL of the filtrate was evaporated until dry in a flat-bottomed vaporizer cup heated to 105°C and tared. The remaining was heated at 105°C to a fixed weight. The water-soluble extract is calculated by the formula22:
![]()
Ethanol-soluble extract
The sample was weighed, of 5 g, and placed in a sealed flask. Then 100 mL of ethanol was added, shaken several times for the first 6 hours, and then left for 18 hours. To prevent ethanol evaporation, rapid filtration was used. The filtrate (20.0 mL) was evaporated to dryness in a flat-bottomed vaporizer cup heated to 105°C and calibrated. The remainder was heated at 105°C to a constant weight. Ethanol-soluble extract is calculated by the same formula as the calculation of water-soluble extract.22
Phytochemical screening
Phytochemical screening was conducted on flavonoids, alkaloids, steroids, triterpenoids, saponins, and polyphenols using a standard color reaction test.23
Sample preparation for quantitative phytochemical analysis
Ginger powder (2 g) was added to 100 mL of distilled water and sonicated for 30 minutes. The mixture was filtered, and the filtrate was diluted with distilled water to yield a sample with a concentration of 4000 ppm.
Total phenolic content
The Folin-Ciocalteu method was applied to determine the total phenolic content. A 0.5-mL sample was added with 5 mL of 10% Folin-Ciocalteu reagent, followed by 4 mL of 7.5% Na2CO3, and left at room temperature for 30 minutes. The absorbance was measured at 758 nm. The calibration curve was based on a standard of gallic acid (20-60 ppm).24
Total flavonoid content
A 1 mL sample was mixed with 3 mL of 96% ethanol, 0.2 mL of aluminum chloride (AlCl3 10%), 0.2 mL of potassium acetate 1M, and 5.6 mL of distilled water, then shaken and left to stand at room temperature for 30 minutes. The absorbance was measured at 427 nm. The calibration curve used a standard of quercetin (20-100 ppm).25
Total terpenoid content
The dried extract was weighed 100mg, added 9 mL of 70% ethanol, and then allowed to stand for 24 hours. Extraction was done using a liquid-liquid method using a separatory funnel with 10 mL of petroleum ether. The extract was collected and evaporated using a water bath to dry, then weighed to determine the percentage of total terpenoid content with the following formula26:
![]()
Extraction and LC-MS analysis
Ginger simplisia weighing 25 grams was placed in a beaker glass, then 250 mL of solvent was added and stirred. The solvents used were water and n-hexane separately. The maceration was carried out for 3 hours at room temperature, then sonicated (200 W, 40 KHz) for 30 minutes at 70℃. The extract obtained was filtered, and the residue was re-macerated with each solvent. The re-macerated filtrate was combined with the initial filtrate and then concentrated to a thickness of 50 mL using a rotary evaporator under vacuum at 55℃. The concentrated extract was centrifuged for 10 minutes to remove any solids that may have escaped during filtration. The extract was stored in a freezer until frozen, then lyophilized for 62 hours to obtain a powder-form dry extract.27
The Shimadzu LCMS 8040 LC/MS was used for high-resolution MS/MS analysis.28 The LCMS properties include model Shimadzu LCMS 8040 LC/MS, column Shimadzu Shim Pack FC-ODS (2 mm x 150 mm, 3 µm), injection volume of 1 µL, capillary voltage of 3.0 kV, column temperature of 35°C, mobile phase methanol of 96% with isocratic mode, flow rate of 0.5 mL/min, sampling cone of 23.0 V, MS focused ion mode ion type [M]+, collision energy of 5.0 V, desolvation gas flow of 60 mL/hour, desolvation temperature of 350°C, fragmentation method: low energy CID, ionization: ESI, scanning of 0.6 sec/scan (mz: 10-1000), source temperature of 100°C, and run time of 60 minutes.
LC-MS was identified by comparing the mass-to-charge (m/z) data and fragmentation patterns obtained from the sample with data in compound databases or with standard compounds. This process involves matching the mass spectrum of the sample with a reference spectrum to identify the compounds contained therein. Databases used include the National Institute of Standards and Technology (NIST) Mass Spectral Library, Wiley Registry of Mass Spectral Data, MassBank, ChemSpider, PubChem, and other specific databases such as the Human Metabolome Database (HMDB).
Distillation of essential oils and GC-MS analysis
To extract the oil, 100 g of crushed ginger was placed in a distillation flask (1 L), then connected to the steam generator with a glass tube and a condenser. The essential oils were volatilized by heating the water at 100°C for 5 to 10 hours. The recovered mixture was left to settle before being extracted for oil. The product was collected and separated with a separator funnel following the steam distillation process. The essential oils settled on the bottom layer of the separator funnel and were separated several times until no oil remained in the funnel.29
Ginger oils GC-MS analysis was carried out on a Shimadzu QP2010 SE system. The operating conditions included helium as the carrier gas, 0.75 mL/min flow rate, injector, column oven temperatures: 50°C, injection temperature: 250°C, injection mode: split, flow control mode: pressure, pressure: 100.0 kPa, 0.1 μL sample volume, 36.1 kPa pressure, total flow: 681.2 mL/min, column flow: 1.69 mL/min, linear, velocity: 47.2 cm/sec, purge flow: 3.0 mL/min, split ratio: 400.0, detector temperatures: 250°C, and Willey library data.
Oven temperature program
| Rate | Temperature (°C) | Hold time (min) |
| – | 50.0 | 2.00 |
| 5.00 | 140.0 | 2.00 |
| 10.00 | 280.0 | 2.00 |
GCMS-QP2010 plus, ion source temp: 200.00°C, interface temp: 200.00°C, solvent cut time: 2.00 min, detector gain mode: relative to the tuning result, detector gain: +0.00 kV, threshold: 1000 MS table, start time: 5.00 min, end time: 37.80 min, ACQ mode: scan, event time: 0.50 sec, scan speed: 625, Start m/z: 50.00, and end m/z: 350.00.30
Result
Standardizations of Ginger (Zingiber officinale Rosc).
The standardization of ginger simplicia involved assessing both non-specific and specific parameters through physical, chemical, and biological analyses. Non-specific parameters included measurements of ash content, moisture content, total plate count, and total fungal count. Specific parameters focused on water-soluble extract, ethanol-soluble extract, and phytochemical profiling. The results are summarized in Table 1, Table 2 and illustrated in Figure 1. For the non-specific parameters, ginger simplicia showed moisture and ash contents below 10%, with total microbial contamination levels under 104 cfu/g. In terms of specific parameters, the water-soluble extract and ethanol-soluble extract were measured at 1.79 ± 0.16 and 0.47 ± 0.11, respectively. Phytochemical analysis presented in Figure 1a revealed moderate concentrations of flavonoids and saponins, and high concentrations of triterpenoids and polyphenols. Additionally, quantitative phytochemical analysis (Table 1) demonstrated that phenolic compounds were more abundant than flavonoids and terpenoids.
To analyze the profile of active compounds soluble in different solvents, ginger simplicia was extracted using both water and n-hexane. Additionally, distillation was carried out to obtain ginger essential oils. The active compounds present in the water and n-hexane extracts were identified through LC-MS, while GC-MS was employed for the analysis of the essential oils. The water extraction of ginger revealed 76 components. As illustrated in Figure 2, the major group in the water extract comprised polyphenols (51%), including flavonoid glycosides (15%), flavonoids (21%), and simple phenolics (15%), followed by terpenoids (33%) and other compounds (16%). The n-hexane extraction resulted in 67 components, with Figure 3 showing that monoterpenoids (77%), sesquiterpenoids (9%), diterpenoids (1%), and triterpenoids (13%) were the dominant groups in the n-hexane extract.
Table 1: Quuantitative of phytochemical analysis of Ginger simplicial
| Parameters | Results |
| Total Phenol Content | 3.15±0.04 (mg GAE/g), |
| Total Flavonoid Content | 1.42±0.46 (mgQE/g) |
| Total Terpenoid Content | 38.82±0.26 (mg/g) |
 |
Figure 1: Qualitative (a) and quantitative (b) phytochemical analysis of Ginger simplicial Click here to view Figure |
Table 2: Analysis of specific and non-specific parameters of simplicial
| Standardization parameter | Results | |
| Non-specific | Water Content | 7.44±0.11% |
| Ash Content | 9.69±0.03% | |
| Total Plate Count | 2.2×104 cfu/g | |
| Total Fungal Count | 5.5×104 cfu/g | |
| Specific | Water-soluble Extract | 1.79±0.16 |
| Ethanol-soluble Extract | 0.47±0.11 | |
GC-MS analysis identified 40 components in ginger essential oil. Figure 4 illustrates that sesquiterpenoids are the major constituents of the essential oil, including l-zingiberene (18.76%), alpha-curcumene (17.52%), sesquiphellandrene (12.92%), beta-bisabolene (7.59%), and alpha-farnesene (6.48%). Monoterpenoids such as citral (5.49%), camphene (4.92%), neral (3.92%), borneol (2.91%), sylvestrene (2.41%), and eucalyptol (2.34%) were also identified. A detailed composition of ginger compounds from LC-MS and GC-MS analyses can be found in the supplementary file.
 |
Figure 2: Profile of Ginger Metabolic Secondary compounds in water solvent, (a) LC-MS chromatogram, (b) summary diagram of Metabolic Secondary Composition, and (c) summary diagram of Metabolic Secondary Polarity.Click here to view Figure |
 |
Figure 3: Profile of Ginger Metabolic Secondary Compounds in n-hexane solvent, (a) LC-MS chromatogram, (b) summary diagram of Metabolic Secondary Composition, and (c) summary diagram of Metabolic Secondary Polarity.Click here to view Figure |
Discussion
Standardization aims to enhance product quality and safety by ensuring consistent pharmaceutical and therapeutic standards. This process involves determining both specific and nonspecific parameters in simplicia and extracts derived from natural ingredients.31,32 The standardized simplicia was assessed according to relevant regulations. As stated in the Indonesian Herbal Pharmacopoeia, simplicia should contain no more than 10% moisture. According to the Decree of the Minister of Health of the Republic of Indonesia No. 261/MENKES/SK/IV/2009, the ash content in extracts must not exceed 10.2%. Furthermore, the National Agency of Drug and Food Control regulations specify that microbial contamination limits for traditional medicines should not surpass 104 cfu/g for total plate count and 103 cfu/g for total fungal count.32 Water content correlates with storage duration, as lower moisture levels in simplicia extend shelf life by inhibiting microbial growth and enzymatic activity. Ash content reflects the mineral composition of ginger, with higher ash values indicating greater mineral content.31 The ginger simplicia complied with these standards, exhibiting less than 10% water content, ash content below 10.2%, total microbial contamination under 103 cfu/g, and total fungal contamination below 104 cfu/g. These results demonstrate that the simplicia processing was conducted appropriately.
In this research, the levels of soluble compounds were analyzed gravimetrically to assess the chemical composition of simplicia in ethanol and water. Water-soluble extract indicates the amount of polar compounds present, while ethanol-soluble extract reveals the number of compounds dissolved in ethanol. Measuring ethanol-soluble extract content is similar to determining water content in terms of quality assessment.33 The ginger simplicia exhibited a higher water-soluble extract, suggesting a greater concentration of compounds dissolved in water. Although determining dissolved compound levels does not directly indicate pharmacological effects, according to the Ministry of Health, it can estimate the presence of semi-polar (ethanol-soluble) and polar (water-soluble) compounds.
The World Health Organization (WHO) provides regulations for the standardization and quality control of herbal medicines by assessing the physicochemical properties of crude drugs. This includes guidelines on the selection and handling of raw materials, evaluation of safety, efficacy, and stability of finished products, documentation of safety and risks based on traditional use, consumer information provision, and promotion. Key parameters for standardization encompass macro- and microscopic examination, foreign matter detection, ash value, moisture content, extractive value, crude fiber, qualitative and quantitative chemical evaluations, chromatographic analysis, and toxicological studies.34 Quality standards for herbal medicines may differ depending on the country, intended application, and regulatory framework, making it essential to consult relevant standards when using herbal products.35 Phytochemical screening results vary slightly from previous research, which identified flavonoids, saponins, tannins, terpenoids, steroids, and phenolic compounds in ginger36. In this study, polyphenols, flavonoids, and tannins were not detected in the ginger simplicia, likely due to their low concentrations, which made them undetectable by the colorimetric reaction tests. However, more sensitive analysis using LC-MS was able to identify these three groups of compounds, as the column used was well-suited for separating polyphenols. Conversely, the LC-MS method was less effective in detecting alkaloids, saponins, and triterpenoids.
Ginger simplicia was further examined using LC-MS to identify the active compounds present. Quantification methods in HPLC with a UV detector and LC-MS differ due to their distinct detection principles. In HPLC-UV, an increase in absorbance signal directly corresponds to a higher concentration of the compound, following the Lambert-Beer law, which states that absorbance is proportional to concentration. Conversely, in LC-MS, the Total Ion Current (TIC) signal does not always accurately reflect concentration because it is influenced by the compound’s ionization efficiency. The HPLC-UV quantification relies on a UV detector measuring absorbance at a specific wavelength, where a linear correlation between signal and concentration exists if the compound has a stable molar extinction coefficient.37,38 LC-MS quantification, on the other hand, is based on TIC, which represents the total number of ions detected at each point during analysis. The intensity of the TIC signal depends on the ionization efficiency, which is affected by the compound’s chemical characteristics (such as polarity and molecular weight) and instrument settings (including voltage and temperature). Compounds that ionize more efficiently, such as polar molecules, tend to produce higher TIC signals even when present at the same concentration as less polar compounds. Using TIC for quantification has several drawbacks: Ionization variability: Different compounds have varying ionization affinities, meaning TIC may not accurately represent their true concentrations; Matrix effects: Other substances in the sample can either suppress or enhance the ionization of the target compound, leading to inaccurate TIC signals; and Limited resolution: TIC peaks may consist of overlapping signals from multiple compounds, especially in complex samples.39,40 Due to these limitations, TIC is generally not recommended for absolute quantification in LC-MS. More precise quantification methods include Selected Reaction Monitoring (SRM), the use of internal standards, and calibration curves to ensure accuracy. Nonetheless, in this study, TIC was utilized under the assumption that the ionization efficiency of the target compounds had been sufficiently characterized.
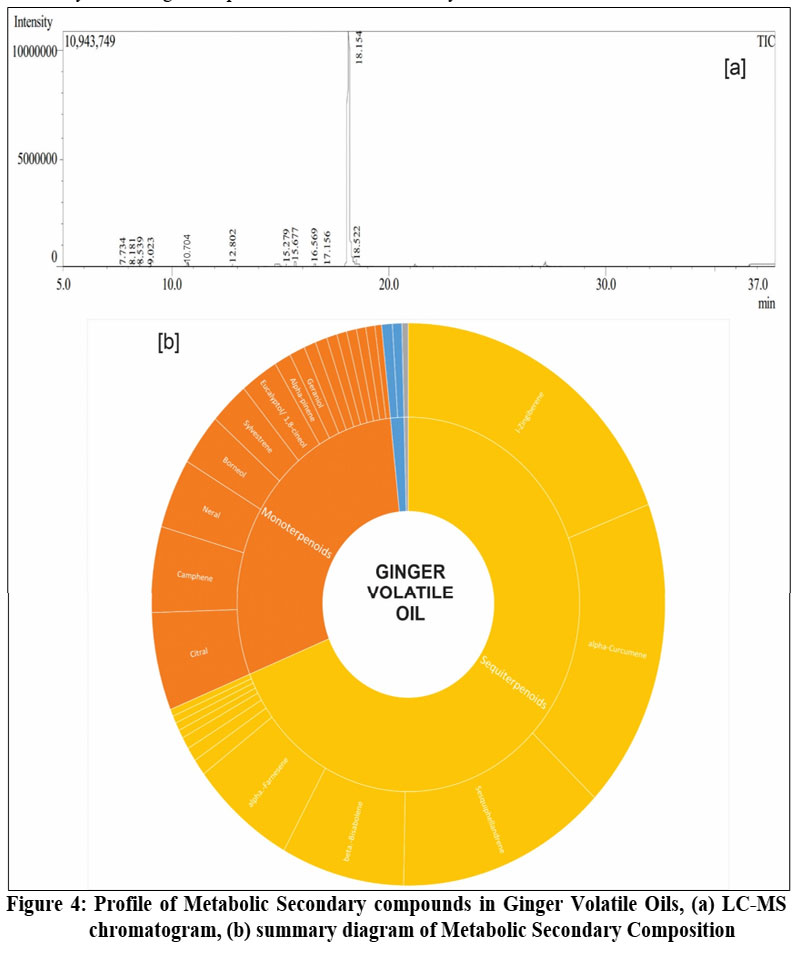 |
Figure 4: Profile of Metabolic Secondary compounds in Ginger Volatile Oils, (a) LC-MS chromatogram, (b) summary diagram of Metabolic Secondary Composition Click here to view Figure |
The extraction of ginger for LC-MS analysis was carried out using two solvents water and n-hexane resulting in distinct profiles of active chemical constituents. Similarly, the chemical composition of the essential oils also varied depending on the extraction method. The ginger extracts obtained with water and n-hexane exhibited differences in the solubility of compounds, reflecting the unique properties of each solvent. In the water extract, secondary metabolites were predominantly organized according to their polarity, ranging from polar to semi-polar and finally to non-polar compounds. These included organic acids (2%), simple phenols (22%), flavonoid glycosides (37%), flavonoids (18%), diarylheptanoids (22%), and terpenoids (3%), as illustrated in Figure 2. Conversely, the n-hexane extract contained compounds arranged from non-polar to semi-polar, including diarylheptanoids (51%); steroids (1%); terpenoids (monoterpenoids (15%), sesquiterpenoids (24%), and diterpenoids (0.5%)); steroids (4%); and other compounds (5%), as shown in Figure 3. The solubility of these compounds during extraction with water or n-hexane depends on their respective physicochemical properties. This understanding is crucial for strategically targeting specific compounds during extraction, as well as for isolating and evaluating the potential bioactivities of ginger-derived compounds. Notably, all compounds found in ginger essential oil are terpenoids, which are non-polar in nature.
Diarylheptanoids are an important group of bioactive compounds found in ginger (Zingiber officinale). They possess a characteristic structure consisting of two aromatic rings (aryl groups) connected by a seven-carbon (heptane) chain. This structure may contain various functional groups such as hydroxyl (-OH) and methoxy (-OCH3), which influence their polarity and biological activity. Structurally, diarylheptanoids generally have two aromatic rings linked by a seven-carbon chain. Most are linear in form, although some cyclic variants exist. The functional groups attached to the aromatic rings and the carbon chain determine their chemical properties and bioactivity.41 Diarylheptanoids are present in ginger extracts obtained with both polar and non-polar solvents and can be identified using LC-MS due to their molecular characteristics. However, they are not detected by GC-MS because of their low volatility. The concentration of diarylheptanoids is higher in n-hexane extracts (approximately 51%) compared to water extracts (approximately 22%), reflecting their semi-polar to non-polar nature. These compounds often co-occur with other bioactive substances such as flavonoids and terpenoids, which may produce synergistic effects. The unique structural features and biological activities of diarylheptanoids make them important targets in natural product research and the development of ginger-based herbal medicines.42 The abundance of diarylheptanoid compounds in ginger water extracts, listed from major to minor, is as follows: 1,5-bis(4-hydroxy-3-metho xyphenyl)-penta(1E,4E)-1,4-dien-3-one (CID6474893), [6]-gingerol (CID442793), [6]-gingerdione (CID16295), [6]-shogaol CID5281794, 4-hydroxycinnamoyl-(feruloyl)-methane (CID5469424), hexahydrocurcumin CID5318039, gingerenone B (CID5317592), [10]-dehydroshogaol (CID131751379), 3,5-diacetoxy-1-(4-hydroxy3,5dimethoxyphenyl) 7-(4hydroxy-3methoxy phenyl)heptane (CID5316611), and [8]-gingerol (CID168114), among others. Meanwhile, the abundance of diarylheptanoid compounds in ginger n-hexane extracts is as follows: [6]-gingerol CID442793, [6]-gingerdione (CID162952), [6]-shogaol (CID5281794), 1-(4-hydroxy-3-methoxyphenyl)-7-(3,4dihydroxyphenyl)-1,6-heptadiene-3,5-dione (CID390474), 1-(4-hydroxy-3-methoxyphenyl)-5-(-4-hydroxyphenyl)-penta-(1E,4E)1,4-dien-3-one (CID10469828), demethoxycurcumin (CID5469424), hexahydrocurcumin CID5318039, gingerenone B (CID5317592), [10]-dehydroshogaol (CID131751379), [8]-gingerol (CID168114), [8]-dehydroshogaol (CID9796428), 3,5-diacetoxy-7-(3,4-dihydroxyphenyl)-1-(4hydroxy-3methoxyphenyl)heptane (CID15118820), [6]-gingerdiol CID5275727,and [10]-gingerol (CID 168115), among others.
1,5-Bis(4-hydroxy-3-methoxyphenyl)-penta(1E,4E)-1,4-dien-3-one (CID6474893) is a diarylheptanoid derivative that has not been extensively reported in pharmacological literature; however, structurally it resembles other active compounds in ginger known for their anti-inflammatory and antioxidant activities.43 [6]-Gingerol (CID442793) is the main bioactive compound in ginger, exhibiting multiple therapeutic benefits such as anti-inflammatory, antioxidant, analgesic, and immunomodulatory effects. It effectively alleviates inflammation, muscle pain, nausea, and digestive disorders.44 This compound also plays a role in stabilizing blood glucose levels and enhancing insulin sensitivity, making it beneficial for diabetic patients. Furthermore, [6]-gingerol inhibits the production of inflammatory cytokines and reduces pain in conditions like osteoarthritis and rheumatoid arthritis. It also demonstrates tumor growth suppression and reduces oxidative stress by increasing antioxidant enzyme activity. Its immunomodulatory effects include the upregulation of key cytokines such as IFN-γ and IL-17 and the regulation of CD4+ and CD8+ immune cells.45 [6]-Gingerdione (CID16295) has limited specific data available; it is an oxidative metabolite of gingerol presumed to possess similar anti-inflammatory and antioxidant properties, contributing to ginger’s therapeutic effects.45 [6]-Shogaol (CID5281794) exhibits strong antibacterial, antiviral, anti-inflammatory, and anticancer activities. It effectively inhibits allergic reactions and cancer cell proliferation. The anti-inflammatory action of shogaol helps relieve pain and inflammation while supporting immune function.46 4-Hydroxycinnamoyl-(feruloyl)-methane (CID5469424), a curcumin derivative, is known for its anti-inflammatory and antioxidant activities, which help reduce oxidative stress and tissue inflammation, thereby supporting cellular protection.47 Hexahydrocurcumin (CID5318039), another curcumin metabolite, also exhibits anti-inflammatory and antioxidant effects by inhibiting inflammatory mediators and protecting tissues from oxidative damage.48 Gingerenone B (CID5317592) shows anti-inflammatory and anticancer activities, contributing to the reduction of systemic inflammation and potential inhibition of cancer cell growth. [10]-Dehydroshogaol (CID131751379), a shogaol derivative, is believed to possess anti-inflammatory and antioxidant properties that support ginger’s therapeutic effects in reducing inflammation and oxidative stress.49 3,5-Diacetoxy-1-(4-hydroxy-3,5-dimethoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)-heptane (CID 5316611) is a diarylheptanoid compound with potential anti-inflammatory and antioxidant activities, although specific data remain limited.43 8-Gingerol (CID 168114), structurally similar to 6-gingerol, exhibits anti-inflammatory, analgesic, and immunomodulatory effects, contributing to the reduction of inflammation and pain. 8-Dehydroshogaol (CID9796428), a shogaol derivative, has potential anti-inflammatory and antioxidant activities, aiding in inflammation reduction and cellular protection.50 3,5-Diacetoxy-7-(3,4-dihydroxyphenyl)-1-(4-hydroxy-3-methoxyphenyl) heptane (CID15118820) is another diarylheptanoid with possible anti-inflammatory and antioxidant effects, though detailed data are scarce.43 [6]-Gingerdiol (CID5275727), a metabolite of gingerol, is presumed to have anti-inflammatory and antioxidant activities that support ginger’s therapeutic properties.43 [10]-Gingerol (CID168115) exhibits similar activities to 6-gingerol, playing a role in reducing inflammation, alleviating pain, and enhancing immune function.51 1-(4-Hydroxy-3-methoxyphenyl)-7-(3,4-dihydroxyphenyl)-1,6-heptadiene-3,5-dione (CID390474) is a diarylheptanoid with antioxidant and anti-inflammatory potential that helps protect tissues from oxidative damage and reduces inflammation, although specific data are limited. Similarly, 1-(4-hydroxy-3-methoxyphenyl)-5-(4-hydroxyphenyl)-penta-(1E,4E)-1,4-dien-3-one (CID 10469828) is a diarylheptanoid derivative suspected to have anti-inflammatory and antioxidant activities, supporting cellular protection and inflammation reduction52. Overall, diarylheptanoid compounds from ginger especially gingerol, shogaol, and their derivatives contribute significantly to therapy through mechanisms including anti-inflammatory, antioxidant, analgesic, immunomodulatory, anticancer activities, as well as metabolic effects such as blood glucose stabilization and improved insulin sensitivity. These compounds play important roles in both traditional and modern treatments for inflammatory diseases, cancer, and metabolic disorders.
Ginger water extract also contains flavonoid glycosides and free flavonoids, which contribute to its promise as a multifunctional natural therapeutic agent in traditional and alternative medicine. These compounds exhibit a wide range of biological activities, including anti-inflammatory, antioxidant, antimicrobial, anticancer, immunomodulatory, and metabolic effects [5]. Some of the flavonoid glycosides found in ginger water extract include: quercetin-3-O-rhamnoside (CID5280459), kaempferol-3-(6”caffeoylglucoside) (CID44259011), kaempferol-3-(5”feruloylapioside) (CID162852962), quercetin-4′-methyl ether 3-neohesperidoside (CID44258037), kaempferol-3-O-D-glucoside (CID5282102), quercetin-3-O-neohesperidoside (CID5491657), kaempferol-3-O-rhamnoside (CID5835713), isoquercetin (CID5280804), kaempferol 3-(2”acetylrhamnoside) (CID44258953), kaempferol 3-(3”acetylrhamnoside) (CID44258954), kaempferol-3-glucoside-2”-rhamnoside-7-rhamnoside (CID10485148), kaempferide 3-O-β-D-glucopyranoside (CID9911508), kaempferol 7,4′-dimethyl ether-3-neohesperidoside (CID44259573), naringin (CID442428), quercetin 3-glucoside (CID5280804), kaempferol 4′-methyl ether 3-neohesperidoside (CID44257994), kaempferol 3-(3”,4”diacetylrhamnoside) (CID44258959), rutin (CID5280805), and kaempferol 3-(4”acetylrhamnoside) (CID44258955). Some of the free flavonoids present include kaempferide (CID5281666), kaempferol (CID5280863), naringenin (CID439246), quercetin (CID5280343),apigenin (CID5280443),3,5,7-trihydroxyflavone (CID5281616), 3,3′,5,7- tetrahydroxy-4′-methoxyflavone (CID5281699), fisetin (CID5281614), morin (CID5281670), catechin (CID9064), alpinetin (CID154279), 5-O-methylnaringenin (CID182315), and naringenin 7,4′-dimethyl ether (CID321346).
Therapeutic contributions of selected Flavonoids and their glycosides are quercetin-3-O-rhamnoside (CID5280459) exhibits strong antioxidant and anti-inflammatory activities, protecting cells from oxidative damage and reducing chronic inflammation.53 Kaempferol-3-(6”-caffeoylglucoside (CID44259011) shows antioxidant and anticancer effects by inhibiting cancer cell proliferation and inducing apoptosis.54 Kaempferol-3-(5”-feruloylapioside) (CID162852962) acts as an anti-inflammatory and antioxidant agent, reducing oxidative stress and tissue inflammation.55 Quercetin-4′-methyl ether 3-neohesperidoside (CID44258037) possesses immunomodulatory and antioxidant potential, supporting immune function and protecting against free radicals.56 Kaempferol-3-O-D-glucoside (CID5282102) have effective in reducing inflammation and protecting tissues through antioxidant mechanisms.57 Quercetin-3-O-neohesperidoside (CID5491657) are enhancing defense against oxidative stress and allergies, functions as an antioxidant and anti-allergic.58 Kaempferol-3-O-rhamnoside (CID5835713) demonstrates anti-inflammatory and anticancer activities by inhibiting tumor growth and inflammation.59 Isoquercetin (CID5280804) is known for antioxidant and anti-inflammatory properties, also supports cardiovascular health.60 Kaempferol-3-(2”-acetylrhamnoside) (CID44258953) and Kaempferol-3-(3”-acetylrhamnoside) (CID44258954) are Kaempferol derivatives with anti-inflammatory effects and cell protective properties against oxidative damage. Kaempferol-3-glucoside-2”-rhamnoside-7-rhamnoside (CID10485148) a potent antioxidant and anti-inflammatory agent. Kaempferol-7,4′-dimethyl ether-3-neohesperidoside (CID44259573) is acts as an antioxidant and immunomodulator.61 Kaempferide 3-O-β-D-glucopyranoside (CID9911508) have exhibits anticancer, anti-inflammatory, and immune-supporting activities.62 Naringin (CID442428) possesses antidiabetic, antioxidant, and anti-inflammatory properties, aiding in blood sugar regulation and inflammation reduction.63 Quercetin 3-glucoside (CID5280804) provides antioxidant, anti-inflammatory, and cardioprotective effects.64 Kaempferol 4′-methyl ether 3-neohesperidoside (CID44257994) is involved in antioxidant activity and inflammation reduction. Kaempferol 3-(3”,4”-diacetylrhamnoside) (CID44258959) and Kaempferol 3-(4”-acetylrhamnoside) (CID44258955) are Kaempferol derivatives with anti-inflammatory and cell-protective effects.65 Kaempferide (CID5281666) and Kaempferol (CID5280863) effective and exhibits anticancer, anti-inflammatory.66 Naringenin (CID 439246) shows antidiabetic, anti-inflammatory, and neuroprotective effects.67 Quercetin (CID5280343) one of the most potent flavonoids with antioxidant, anti-inflammatory, anticancer, and immunomodulatory activities.58 Apigenin (CID5280443) is functions as an anti-inflammatory, anticancer, and neuroprotective agent.68 3,5,7-trihydroxyflavone (CID5281616) exhibits antioxidant and anti-inflammatory effects. 3,3′,5,7-tetrahydroxy-4′-methoxyflavone (CID5281699) was potent antioxidant and anticancer activity.69 Fisetin (CID5281614) demonstrates neuroprotective, antioxidant, and anticancer properties.70 Morin (CID5281670) provides antioxidant, anti-inflammatory, and anticancer benefits.71 Catechin (CID9064) known for strong antioxidant activity, supports cardiovascular health, and reduces cancer risk.72 Alpinetin (CID154279) exhibits anti-inflammatory and antimicrobial effects.73 5-O-methylnaringenin (CID182315) and Naringenin-7,4′-dimethyl ether (CID321346) are naringenin derivatives with antioxidant and anti-inflammatory activities supporting metabolic and immune health.74 The flavonoids and their glycosides in ginger extracts contribute to a broad spectrum of therapeutic effects, including antioxidant, anti-inflammatory, anticancer, immunomodulatory, antidiabetic, neuroprotective, and antimicrobial activities. These compounds synergistically enhance the medicinal value of ginger as a natural remedy in both traditional and modern healthcare.
Ginger water extracts contain various simple phenolic compounds, including Salicylic acid (CID: 38), p-coumaric acid (CID637542), Vanillic acid (CID8468), Gallic acid (CID370), Shikimic acid (CID8742), Ferulic acid (CID445858), Syringic acid (CID10742), Ellagic acid (CID5281855), 5-O-caffeoylshikimic acid (CID5281762), 3-p-coumaroylquinic acid (CID9945785), Chlorogenic acid (CID1794427), and 3-O-caffeoylquinic acid methyl ester (CID6476139). These compounds contribute to various therapeutic activities. For instance, p-coumaric acid and vanillic acid exhibit a strong antioxidant effect, demonstrating a 77.30% DPPH radical scavenging capacity through hydrogen-donating mechanisms.75 Similarly, gallic acid and ferulic acid help neutralize reactive oxygen species (ROS) by virtue of their hydroxyl group structures.76 In terms of antimicrobial effects, shikimic acid and its derivatives, such as 5-O-caffeoylshikimic acid, inhibit bacterial cell wall synthesis by interfering with essential metabolic pathways. Additionally, chlorogenic acid and 3-O-caffeoylquinic acid display broad-spectrum antibacterial activity by disrupting microbial cell membranes.77 Regarding immune modulation, ellagic acid and syringic acid found in purified red ginger extract significantly enhance macrophage phagocytic response, measured at 862.88 mg GAE/g, which is comparable to the positive control Phyllanthus niruri.78 Furthermore, ferulic acid and chlorogenic acid have demonstrated anticancer potential by inducing apoptosis in cancer cells through the activation of caspase-3 and regulation of Bcl-2 expression.79 These phenolic compounds also interact synergistically within ginger water extracts. They chelate metal ions such as Fe²⁺ and Cu²⁺, preventing free radical catalysis, stabilize cell membranes through hydrophobic interactions with lipid bilayers, and regulate the expression of genes related to oxidative stress via the Nrf2/Keap1 pathway.80 To optimize extraction, aqueous methods performed at a pH between 5 and 6 with an 8:1 solvent-to-solid ratio yield the imest phenolic content, reaching up to 21.07% shogaol. However, careful temperature control during extraction is essential to prevent thermal degradation of heat-sensitive compounds like chlorogenic acid.81
Several terpenoid compounds were detected in the aqueous solvent extract, such as alpha-zingiberene (Sesquiterpenoid), ar-curcumen-15-al (Bisabolane sesquiterpenoids), 2-(2′,3′-epoxy-3′-methylbutyl)-3-methylfuran (Acyclic monoterpenoids), and galanolactone (Diterpenes). However, the abundance of terpenoid compounds in ginger n-hexane extracts is relatively high, especially in the subgroups of monoterpenoids, sesquiterpenoids, and diterpenoids. The monoterpenoid subgroup’s abundance, from major to minor, includes: (E)-3,7-dimethyl-3,6-octadienal (CID6428928), trans-carveyl acetate (CID7335), myrcene (CID31253), (-)-dihydrocarveol (CID443163), 1,3,8-p-menthatriene (CID176983), (Z)-3,7-dimethyl-3,6-octadienal (CID6428928), β-pinene (CID14896), neryl acetate (CID1549025), geranyl acetate (CID1549026), fenchyl acetate (CID107217), p-mentha-1,5,8-triene (CID527424), and (+)-2-carene (CID78249). The sesquiterpenoid subgroup includes: α-zingiberene (CID92776), ar-curcumene (CID3083834), trans-Z-α-bisabolene epoxide (CID91753504), β-bisabolene (CID10104370), α-caryophyllene (CID5281520), β-sesquiphellandrene (CID12315492), β-farnesene (CID5317319), sabinene (CID18818), farnesol (CID445070), ar-curcumen-15-al (CID10846393), β-elemene (CID6918391), and trans-nerolidol (CID5284507). The diterpenoid subgroup contains only one compound: 8(17),12-labdadiene-15,16-dial (CID9904510). In ginger oil, all terpenoid groups are present, including the monoterpenoid subgroup, such as geranial, camphene, neral, borneol, sylvestrene, eucalyptol (1,8-cineol), α-pinene, and geraniol. The sesquiterpenoid subgroup includes l-zingiberene, alpha-curcumene, sesquiphellandrene, β-cedrene, beta-bisabolene, α-farnesene, and zingiberenol.
Terpenoids are bioactive compounds widely found in ginger water/ n-hexane extract and ginger volatie oil have possess various therapeutic benefits. Several major terpenoid compounds commonly encountered, including those you mentioned, contribute significantly to health through their diverse biological activities. For instance, α-zingiberene (CID92776) exhibits anti-inflammatory, antimicrobial, and analgesic properties, helping to reduce inflammation and pain while combating bacterial and fungal infections.82 Ar-curcumene (CID3083834) acts as an antioxidant and anti-inflammatory agent, protecting cells from oxidative damage and alleviating chronic inflammation.83 Compounds such as trans-Z-α-bisabolene epoxide (CID91753504) and β-bisabolene (CID10104370) demonstrate antimicrobial, anti-inflammatory, and anticancer activities, aiding in infection control and potentially inhibiting cancer cell growth.84 α-Caryophyllene (CID5281520) is known for its anti-inflammatory, analgesic, and neuroprotective effects, which reduce pain and inflammation and safeguard the nervous system.85 Similarly, β-sesquiphellandrene (CID12315492) and β-farnesene (CID5317319) contribute antimicrobial and anti-inflammatory benefits, supporting the body’s defense against infections and inflammation. Sabinene (CID18818) provides antimicrobial and antioxidant effects, protecting the body from free radicals and infections.86 Farnesol (CID445070) shows promising anticancer and antimicrobial properties by inhibiting cancer cell proliferation and fighting pathogenic bacteria.87 Ar-curcumen-15-al (CID10846393) functions as an antioxidant and anti-inflammatory compound, reducing oxidative stress and inflammation.88 β-Elemene (CID6918391) has been studied for its anticancer potential, particularly in suppressing tumor cell proliferation.89 Trans-nerolidol (CID5284507) offers antimicrobial, antioxidant, and anti-inflammatory activities, which help protect against infections and oxidative damage.90 The diterpenoid, 8(17),12-labdadiene-15,16-dial (CID9904510) also exhibits anti-inflammatory and antimicrobial effects, reducing inflammation and combating pathogenic microorganisms.91 Both (E)-3,7-dimethyl-3,6-octadienal and (Z)-3,7-dimethyl-3,6-octadienal (CID6428928) act as antimicrobial and antioxidant agents, shielding tissues from damage and infection.92 Trans-carveyl acetate (CID7335) and myrcene (CID31253) are known for their analgesic and anti-inflammatory properties, effectively alleviating pain and inflammation.93 (-)-Dihydrocarveol (CID443163) and 1,3,8-p-menthatriene (CID176983) provide antimicrobial and antioxidant benefits, helping to fight infections and protect cells from oxidative harm.94 β-Pinene (CID14896) exhibits anti-inflammatory, antimicrobial, and bronchodilator effects, which assist in managing inflammation, infections, and respiratory issues.95 Neryl acetate (CID1549025) and geranyl acetate (CID1549026) serve as antioxidants and antimicrobials, protecting cells and combating pathogens.96 Fenchyl acetate (CID107217) and p-mentha-1,5,8-triene (CID527424) contribute antimicrobial and anti-inflammatory activities, reducing infection and inflammation.94 (+)-2-Carene (CID78249) also has anti-inflammatory and analgesic effects, helping to relieve pain and swelling.97 Other terpenoids such as l-zingiberene, alpha-curcumene, sesquiphellandrene, β-cedrene, beta-bisabolene, α-farnesene, and zingiberenol combine anti-inflammatory, antimicrobial, and anticancer properties, supporting overall health by fighting infections, reducing inflammation, and potentially exerting antitumor effects.98 Compounds like geranial, camphene, neral, borneol, sylvestrene, eucalyptol (1,8-cineol), α-pinene, and geraniol offer anti-inflammatory, expectorant, antimicrobial, and neuroprotective benefits, aiding respiratory health, reducing inflammation, combating infections, and protecting the nervous system.99 Terpenoid compounds found in ginger water/ n-hexane extracts and ginger oil exhibit a wide range of therapeutic activities with significant potential in healthcare. These include anti-inflammatory effects that reduce inflammation in various conditions, antimicrobial properties that fight bacteria, fungi, and viruses, anticancer activities that inhibit tumor growth and spread, and analgesic effects that relieve pain, antioxidant capabilities that protect cells from free radical damage, and neuroprotective functions that safeguard the nervous system from injury. Therefore, these terpenoids hold great promise for development as active ingredients in herbal medicines and modern pharmaceuticals.
Other secondary metabolite compounds found in ginger water extracts, ginger n-hexane extracts, and ginger volatile oils belong to groups of secondary metabolites such as steroids, organic acids, and cinnamic acid, which are present in relatively small amounts and contribute less to the therapeutic effects.
The present study identified a variety of active compounds in ginger, each displaying distinct chemical profiles depending on the solvent and analytical methods employed. The chemical constituents found within these active compounds, across all extract types such as aqueous extracts, n-hexane extracts, and essential oils, exhibit notable bioactivities, especially antioxidant, antimicrobial, and anti-inflammatory effects. Exploring active compounds extracted using different solvents provides a valuable basis for advancing applications in the food, cosmetic, and pharmaceutical industries. This potential is reinforced by ginger’s wide range of bioactivities, which can play a role in improving public health. However, some compounds detected in ginger extracts have yet to be associated with specific bioactivities, indicating the need for further research.
Conclusion
This study successfully standardized ginger simplicia and identified its key active compounds across different ex-tracts. All quality parameters met established standards, ensuring consistent and reliable raw material suitable for industrial applications. Water extracts were rich in simple phenols, flavonoid glycosides, flavonoids, diarylhep-tanoids, and sesquiterpenoids, while n-hexane extracts contained notable diarylheptanoids, monoterpenoids, and steroids. The volatile oil was predominantly com-posed of sesquiterpenoids and monoterpenoids. These findings lay a solid foundation for future research and the development of ginger-based therapeutic products.
Acknowledgment
The author would like to thank Ministry of Education, Culture, Research and Technology of the Republic of Indonesia for granting opportunity of research work.
Funding Source
The authors extend their appreciation to Ministry of Education, Culture, Research and Technology of the Republic of Indonesia, for supporting this work through project number (104/SPK/D.D4/PPK.01.APTV/III/2024, March 19, 2024).
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Sentot Joko Raharjo and Ernanin Dyah Wijayanti: Conceptualization, Methodology, Writing–Original Draft
- Sentot Joko Raharjo and Ernanin Dyah Wijayanti: also Funding Acquisition, Resources, Supervision
- Dewi Ratih Tirto Sari: Data Collection, Analysis, Writing- Review & Editing
- Yanty Maryanty: Visualization, Supervision, Project Administration.
Reference
- Ashraf K, Ahmad A, Chaudhary A, et al. Genetic diversity analysis of Zingiber officinale Roscoe by RAPD collected from subcontinent of India. Saudi J. Biol. Sci. 2014;21(2):159–165.doi: 10.1016/j.sjbs.2013.09.005
CrossRef - Yoon D, Choi B R, Kim H G, Lee D Y. Metabolic differences in Zingiber officinale Roscoe by geographical origin determined via multiplatform metabolomics and method for simultaneous analysis of six phenolic compounds. J. Food Sci. 2024;89(11):7452-7463. doi: 10.1111/1750-3841.17456.
CrossRef - Tientcheu YMT, Dibacto REK, Edoun FLE, et al. Comparative Study of the Effect of Five Drying Methods on Bioactive Compounds, Antioxidant Potential and Organoleptic Properties of Zingiber officinale (Ginger) Rhizome. European J. Med. Plants. 2021, 32(3):22-33. doi:10.9734/EJMP/2021/v32i330378.
CrossRef - Mao Q, Xu X Y, Cao S Y, et al. Bioactive compounds and bioactivities of ginger (Zingiber officinale roscoe). Foods. 2019;8(6):185. doi: 10.3390/foods8060185.
CrossRef - Syafitri D M, Levita J, Mutakin M, Diantini A. A Review: Is Ginger (Zingiber officinale var. Roscoe) Potential for Future Phytomedicine?. Indones. J. Appl. Sci. 2018;(8)1: 8–13. doi: 10.24198/ijas.v8i1.16466.
CrossRef - Wang G, Ren Y, Bai X, Su Y, Han J. Contributions of Beneficial Microorganisms in Soil Remediation and Quality Improvement of Medicinal Plants. 2022. Plants. 2022;11(23):3200. doi:10.3390/plants11233200
CrossRef - Ferreira J F S. Nutrient deficiency in the production of artemisinin, dihydroartemisinic acid, and artemisinic acid in Artemisia annua L. J. Agric. Food Chem. 2007;(55)5: 1686–1694 doi: 10.1021/jf063017v.
CrossRef - Rizky A and Styawan A A. Comparatif of Phenolic Content of Red Ginger (Zingiber officinale var . rubrum Theilade ) At different Growing Location. J. Farm. Sains dan Prakt. 2024;(10)3: 233–241.
- Rababah T M, Al-u’datt M, Alhamad M, et al. Effects of drying process on total phenolics, antioxidant activity and flavonoid contents of common mediterranean herbs. Int. J. Agric. Biol. Eng. 2015; 8(2):145-150. doi: 10.3965/j.ijabe.20150802.1496.
- Gamal R E, Song C, Rayan A M, Liu C, Al-Rejaie S, ElMasry G. Thermal Degradation of Bioactive Compounds during Drying Process of Horticultural and Agronomic Products: A Comprehensive Overview. Agronomy. 2023;13(6):1580. doi: 10.3390/agronomy13061580.
CrossRef - Saputri D E, Ahmad U, Pujantoro L. Shelf-life Estimation of the Red Ginger Powder in Paper Kraft Packaging using ASLT Method Arrhenius Model. J. Keteknikan Pertan. 2023;11(3):318–331. doi: 10.19028/jtep.011.3.318-331.
CrossRef - Kusumawati N, Anggarani M A, Rusijono, Setiarso P, Muslim S. Product standarization of ginger (Zingiber officinale rosc.) and Red ginger (Zingiber officinale var. rubrum) simplicia through washing time, slice thickness and raw materials drying process optimization. Int. J. Adv. Sci. Eng. Inf. Technol. 2017;(7)1:15–21. doi: 10.18517/ijaseit.7.1.910.
CrossRef - Sachan A K, Vishnoi G, Kumar R. Need of standardization of herbal medicines in modern era. Int. J. Phytomedicine. 2016; 8(3):300. doi: 10.5138/09750185.1847.
CrossRef - Gidwani B, Bhattacharya R, Shukla S S, Pandey R K. Indian spices: past, present and future challenges as the engine for bio-enhancement of drugs: impact of COVID-19. Journal of the Science of Food and Agriculture. 2022;(102)8: 3065-3077. doi: 10.1002/jsfa.11771.
CrossRef - Bhairam M, Roy A, Bahadur S, Banafar A, Turkane D. Standardization of Herbal Medicines – an Overview. J. Appl. Pharm. Res. 2013; 68(1): 213-219
- Shulammithi R, Sharanya M, Tejaswini R, Kiranmai M. Standardization and quality evaluation of herbal drugs. IOSR J. Pharm. Biol. Sci, 2016; (11)5:89-100. doi: 10.9790/3008-11050189100.
CrossRef - de Sousa Lima C M, Fujishima M A T, de Paula Lima B, Mastroianni P C, de Sousa F F O, da Silva J O. Microbial contamination in herbal medicines: a serious health hazard to elderly consumers.BMC Complement. Med. Ther. 2020;(20)1:17. doi: 10.1186/s12906-019-2723-1.
CrossRef - Manuhara G J, Mentari G P, Khasanah L U, Utami R. Aqueous Extract Composition of Spent Ginger (Zingiber officinale var. Amarum) from Essential Oil Distillation. In IOP Conference Series: Materials Science and Engineering, 2018. doi: 10.1088/1757-899X/333/1/012069.
CrossRef - Spyrou A, Batista MG F, Corazza M, Papadaki M, Antonopoulou M. Extraction of High Value Products from Zingiber officinale Roscoe (Ginger) and Utilization of Residual Biomass. Molecules. 2024; 29(4):871. doi: 10.3390/molecules29040871.
CrossRef - Djarot P, Utami N F, Putra A M. Bioactivities and Chemical Compositions of Cinnamomum burmannii Bark Extracts (Lauraceae). Sustainability, 2023; (5)1696:1–15.
CrossRef - Ekawati E R and Yusmiati S N H. Detection of Salmonella sp., Vibrio sp. and total plate count bacteria on blood cockle (Anadara granosa). IOP Conf. Ser. Earth Environ. Sci. 2018;102: 1–5.
CrossRef - Pusmarani J, Putri R J, Dewi C, Purwono S, Ikawati Z. Non Specific and Specific Parameter Standardization Of Banana Peel (Musa paradisciata Sapientum) and Andrographis Paniculata. In International Summit on Science Technology and Humanity. 2019: pp. 658–664.
- Wijayanti E D, Rahayu L O, Dzulaikha S, Ayunda F R, Djara S. Phytochemical Profile and Antioxidant Potential of Tobacco Flower Extract in Different Solvents. JSMARTech. 2024;(05)01:4–8.
- Baba S A and Malik S A. Determination of total phenolic and flavonoid content, antimicrobial and antioxidant activity of a root extract of Arisaema jacquemontii Blume. J. Taibah Univ. Sci. 2015; (9)4:449–454. doi: 10.1016/j.jtusci.2014.11.001.
CrossRef - Chandra S, Khan S, Avula B, et al. Assessment of Total Phenolic and Flavonoid Content, Antioxidant Properties, and Yield of Aeroponically and Conventionally Grown Leafy Vegetables and Fruit Crops: A Comparative Study. Evidence-based Complement. Altern. Med. 2014; 1–9, 2014, doi: 10.1155/2014/253875.
CrossRef - Malik S K, Ahmad M, Khan F. Qualtitative and quantitative estimation of terpenoid contents in some important plants of Punjab, Pakistan. Pak. J. Sci. 2017; (69)2:150–154. doi: 10.57041/pjs.v69i2.364.
CrossRef - Andishmand H, Masoumi B, Torbati M, Homayouni-Rad A, Azadmard-Damirchi S, Hamishehkar H. Ultrasonication/dynamic maceration‐assisted extraction method as a novel combined approach for recovery of phenolic compounds from pomegranate peel. Food Sci. Nutr. 2023;11:7160–7171.
CrossRef - Teoh W Y, Yong Y S, Razali F N, Stephenie S. LC-MS/MS and GC-MS Analysis for the Identification of Bioactive Metabolites Responsible for the Antioxidant and Antibacterial Activities of Lygodium microphyllum (Cav.) R. Br. Separations. 2023;(10)215:1–12. doi: 10.3390/separations10030215.
CrossRef - Wong Y C, M. Ahmad-Mudzaqqir Y, Wan-Nurdiyana W A. Extraction of essential oil from cinnamon (Cinnamomum zeylanicum). Orient. J. Chem. 2014; (30)1:37–47. doi: 10.13005/ojc/300105.
CrossRef - Chairunnisa, Tamhid H A, Nugraha A T. Gas chromatography – Mass spectrometry analysis and antibacterial activity of Cinnamomum burmanii essential oil to Staphylococcus aureus and Escherichia coli by gaseous contact. AIP Conf. Proc. 2017; 1823. doi: 10.1063/1.4978146.
CrossRef - Taurina W and Andrie M. Standardization of Simplicia Golden Sea Cucumber (Stichopus hermanii) from Pelapis Island, West Kalimantan. Trad. Med. J. 2022;(27)2:1–7. doi: 10.22146/mot.74667.
CrossRef - Ngibad K, et al. Standardization of Simplicia and Extracts of Arabic Bidara (Ziziphus spina-christi (L.) Desf.) and Tree Saga (Adenanthera pavonina L.) Leaves. J. Chem. Heal. Risks. 2024;(14)2:1747–1758.
- Husni E, Ismed F, Awaliana. Standardization Extracts and Simplicia of Limau Sundai Peel (Citrus x aurantiifolia ’sundai’), Determine Content of Nobiletin and Antibacterial Activity Test. Pharmacogn. J. 2021;(13)6:1323–1331.
CrossRef - Pradhan N, Gavali J, Waghmare N. WHO (World Health Organization) guidelines for standardization of herbal drugs. Int. Ayuverdic Med. J. 2015; (3)8:2238–2243.
- Kunle O F, Egharevba H O, Ahmadu P O. Standardization of herbal medicines – A review. Int. J. Biodivers. Conserv. 2012; (4)3:101–112. doi: 10.5897/IJBC11.163.
CrossRef - Leslie A G J and Gunawan S. Red Ginger extract (Zingiber officinale Var. Rubrum): phytochemical test, fingerprint analysis, total antioxidant capacity, and determination of phenolic content. J. Kesehat. Tambusai. 2023;(4)2:2007–2016.
- Tumanduk R, Massi M N, Agus R, Hamid F. Analisis Residu Amoksisilin Pada Hepar dan Ventrikulus Ayam Petelur di Pasar Tradisional Makassar. J. Ilmu Alam dan Lingkung. 2023; (14)2:20–28.
- Chawla G and Ranjan C. Principle, Instrumentation, and Applications of UPLC: A Novel Technique of Liquid Chromatography. Open Chemistry Journal. 2016; 3:1–16. doi: 10.2174/1874842201603010001.
CrossRef - Tang L, Swezey R R, Green C E, Mirsalis J C. Enhancement of sensitivity and quantification quality in the LC–MS/MS measurement of large biomolecules with sum of MRM (SMRM). Anal. Bioanal. Chem. 2022; p.1–15.
CrossRef - Suhendi A, Rohman A, Wahyono D, Nurrochmad A, Manggo T F. Validation of Analytical Method LC MS / MS for Determination Isoniazid in Rats Serum. Pharmacon. 2023;(20)2: 96–103.
CrossRef - Liv H and She G. Naturally occurring diarylheptanoids. Natural Product Communications. 2010;(5)10:1687-1708. doi: 10.1177/1934578×1000501035.
CrossRef - Jiang T, Ghosh R, Charcosset C. Extraction, purification and applications of curcumin from plant materials-A comprehensive review. Trends in Food Science & Technology. 2021;112:419-430. doi: 10.1016/j.tifs.2021.04.015.
CrossRef - Ganapathy G, Preethi R, Moses J A. Diarylheptanoids as nutraceutical: A review. Biocatalysis and Agricultural Biotechnology. 2019;19. doi: 10.1016/j.bcab.2019.101109.
CrossRef - Almatroodi S A, Alnuqaydan A M, Babiker A Y, Almogbel M A, Khan A A, Rahmani A H. 6-gingerol, a bioactive compound of ginger attenuates renal damage in streptozotocin-induced diabetic rats by regulating the oxidative stress and inflammation. Pharmaceutics. 2021; (13)3:317. doi: 10.3390/pharmaceutics13030317.
CrossRef - Pázmándi K, Szöllősi A G, Fekete T. The ‘root’ causes behind the anti-inflammatory actions of ginger compounds in immune cells. Front. Immunol. 2024; (15)6:1–17. doi: 10.3389/fimmu.2024.1400956.
CrossRef - Bischoff-Kont I, Primke T, Niebergall L S, Zech T, Fürst R. Ginger Constituent 6-Shogaol Inhibits Inflammation and Angiogenesis-Related Cell Functions in Primary Human Endothelial Cells. Front. Pharmacol. 2022; 13. doi: 10.3389/fphar.2022.844767.
CrossRef - Peng Y, Ao M, Dong B. Anti-inflammatory effects of curcumin in the inflammatory diseases: Status, limitations and countermeasures. Drug Des Devel Ther. 2021;2(15):4503-4525. doi: 10.2147/DDDT.S327378.
CrossRef - Nowak I, Kubina R, Strzałka-Mrozik B. Therapeutic Potential of Hexahydrocurcumin in the Regeneration and Protection of the Retinal Pigment Epithelium. Pharmaceuticals. 2025; (18)4:1–18. doi: 10.3390/ph18040554.
CrossRef - Habib SHM, Makpol S, Hamid NAA, Das S, Ngah WZW, Yusof YAM. Ginger extract (Zingiber officinale) has anti-cancer and anti-inflammatory effects on ethionine-induced hepatoma rats. Clinics, 2008; 63(6):807-13. doi: 10.1590/S1807-59322008000600017.
CrossRef - Ayustaningwarno F, Anjani G, Ayu AM, Fogliano V. A critical review of Ginger’s (Zingiber officinale) antioxidant, anti-inflammatory, and immunomodulatory activities. Front. Nutr. 2024; 11(6):1–16. doi: 10.3389/fnut.2024.1364836.
CrossRef - Dugasani S, Pichika MR, Nadarajah VD, Balijepalli MK, Tandra S, Korlakunta JN. Comparative antioxidant and anti-inflammatory effects of [6]-gingerol, [8]-gingerol, [10]-gingerol and [6]-shogaol. J. Ethnopharmacol. 2010; 127(2): 515-20. doi: 10.1016/j.jep.2009.10.004.
CrossRef - Deeb D, Jiang H, Gao X, Al-Holou S, Danyluk AL, Dulchavsky SA, Gautam SC. Curcumin [1,7-bis(4-hydroxy-3-methoxyphenyl)-1-6-heptadine-3,5-dione; C21H20O6] sensitizes human prostate cancer cells to tumor necrosis factor-related apoptosis-inducing ligand/Apo2L-induced apoptosis by suppressing nuclear factor-κB via inhibition of th. J. Pharmacol. Exp. Ther. 2007;(321)2:616-25. doi: 10.1124/jpet.106.117721.
CrossRef - Septembre-Malaterre A, Boumendjel A, Seteyen A S, et al. Focus on the high therapeutic potentials of quercetin and its derivatives. Phytomed Plus. 2022;2(1):100220. doi: 10.1016/j.phyplu.2022.100220.
CrossRef - Imran M, Salehi B, Sharifi-Rad J, Gondal T A. Kaempferol: A key emphasis to its anticancer potential. Molecules. 2019; 24(12), 2277. doi: 10.3390/molecules24122277.
CrossRef - Hussain MS. Kaempferol: Paving the path for advanced treatments in aging-related diseases. Food. 2024. doi: 10.1016/j.exger.2024.112389.
CrossRef - Iwashina T, Smirnov SV, Damdinsuren O, Kondo K. Flavonoids from Reaumuria soongarica (Tamaricaceae) in Mongolia. Bull. Natl. Mus. Nat. Sci. Ser. B. 2012;(38)4:189–195.
- Periferakis A, Periferakis K, Badarau I A, Petran E M, Popa D C, Caruntu A. Kaempferol: Antimicrobial Properties, Sources, Clinical, and Traditional Applications. Int J Mol Sci. 2022;23(23):15054. doi: 10.3390/ijms232315054.
CrossRef - Vollmannová A, Bojňanská T, Musilová J, Lidiková J, Cifrová M. Quercetin as one of the most abundant represented biological valuable plant components with remarkable chemoprotective effects. Review Heliyon, 2024; (10)12. doi: 10.1016/j.heliyon.2024.e33342.
CrossRef - Akter M, Parvin M S, Hasan M M, Rahman M A A, Islam M E. Anti-tumor and antioxidant activity of kaempferol-3-O-alpha-L-rhamnoside (Afzelin) isolated from Pithecellobium dulce leaves. BMC Complement. Med. Ther. 2022; 22(1):169. doi: 10.1186/s12906-022-03633-x.
CrossRef - Zhang W, Zheng Y, Yan F, Dong M, Ren Y. Research progress of quercetin in cardiovascular disease. Front Cardiovascular Medications. 2023; 16:10:1203713.2023. doi: 10.3389/fcvm.2023.1203713.
CrossRef - Kashyap D, Sharma A, Tuli H S, Sak K, Punia S, Mukherjee T K. Kaempferol: A dietary anticancer molecule with multiple mechanisms of action: Recent trends and advancements. J Funct Foods. 2017; 30:203-219 2017. doi: 10.1016/j.jff.2017.01.022.
CrossRef - Wang J, Fang X, Ge L, Cao F, Zhao L. Antitumor, antioxidant and anti-inflammatory activities of kaempferol and its corresponding glycosides and the enzymatic preparation of kaempferol. PLoS One. 2018;13(5):e0197563. doi: 10.1371/journal.pone.0197563.
CrossRef - Shilpa V S, Shams Rafeeya, Dash K K. Phytochemical Properties, Extraction, and Pharmacological Benefits of Naringin: A Review. Molecules. 2023; (28)15:5623.2023. doi: 10.3390/molecules28155623.
CrossRef - Nile A, Nile S H, Shin J, Park G, Oh J W. Quercetin-3-glucoside extracted from apple pomace induces cell cycle arrest and apoptosis by increasing intracellular ROS levels. Int. J. Mol. Sci. 2021; (22)19. doi: 10.3390/ijms221910749.
CrossRef - Ramzan M, Haider S T A, Hussain M B, Ehsan A, Datta R. Potential of kaempferol and caffeic acid to mitigate salinity stress and improving potato growth. Sci. Rep. 2024;(14)1:21657. doi: 10.1038/s41598-024-72420-0.
CrossRef - Qattan MY, Idreesh Khan M, Alharbi SH, Verma AK. Therapeutic Importance of Kaempferol in the Treatment of Cancer through the Modulation of Cell Signalling Pathways. Molecules. 2022;(27)24:88642022. doi: 10.3390/molecules27248864.
CrossRef - Hartogh DJD and Tsiani E. Antidiabetic properties of naringenin: A citrus fruit Polyphenol. Biomolecules. 2019; 9(3): 99. doi: 10.3390/biom9030099.
CrossRef - Allemailem K S, Almatroudi A, Obaid H, Alharbi A, AlSuhaymi N. Apigenin: A Bioflavonoid with a Promising Role in Disease Prevention and Treatment. Biomedicines. 2024:12(6):1353 doi: 10.3390/biomedicines12061353.
CrossRef - Gomes A, Couto D, Alves A, Dias I, Freitas M, Porto G. Trihydroxyflavones with antioxidant and anti-inflammatory efficacy. BioFactors. 2012; (38)5:378-862012. doi: 10.1002/biof.1033.
CrossRef - Hassan SS, Samanta S, Dash R, Karpiński TM. The neuroprotective effects of fisetin, a natural flavonoid in neurodegenerative diseases: Focus on the role of oxidative stress. Front Pharmacol. 2022; 10:13:1015835. doi: 10.3389/fphar.2022.1015835.
CrossRef - Choudhury A, Chakraborty I, Banerjee TS, Vana DR, Adapa D. Efficacy of Morin as a Potential Therapeutic Phytocomponent: Insights into the Mechanism of Action. Int. J. Med. Res. Heal. Sci. 2017; (6)11:175–194.
- Chen X Q, Hu Ting, Han Yu, Huang Wei, Yuan Hai-Bo, Zhang Yun-Tian, Du Yu. Preventive effects of catechins on cardiovascular disease. Molecules. 2016; (21)12:1759 2016. doi: 10.3390/molecules21121759.
CrossRef - Zhao G, Tong Y, Luan F, Zhu W, Zhan C, Tian Q. Alpinetin: A Review of Its Pharmacology & Pharmacokinetics. Front. Pharmacol. 2022;13(2):1–23. doi: 10.3389/fphar.2022.814370.
CrossRef - Duda-Madej A, Stecko J, Sobieraj J, Szymańska N, Kozłowska J. Naringenin and Its Derivatives—Health-Promoting Phytobiotic against Resistant Bacteria and Fungi. Humans. 2022. doi: 10.3390/antibiotics11111628.
CrossRef - Shen Y, Song X, Li L, Sun J, Jaiswal Y, Huang J, Liu C. Protective effects of p-coumaric acid against oxidant and hyperlipidemia-an in vitro and in vivo evaluation. Biomed. Pharmacother. 2019;111:579-5872019. doi: 10.1016/j.biopha.2018.12.074.
CrossRef - Jasemi S V, Khazaei H, Morovati M R, Joshi T. Phytochemicals as treatment for allergic asthma: Therapeutic effects and mechanisms of action. Phytomedicine. 2024;122:155149. doi: 10.1016/j.phymed.2023.155149.
CrossRef - Bai J, Wu Y, Bu Q, Zhong K, Gao H. Comparative study on antibacterial mechanism of shikimic acid and quinic acid against Staphylococcus aureus through transcriptomic and metabolomic approaches. LWT. 2022; 153:1124412022. doi: 10.1016/j.lwt.2021.112441.
CrossRef - Sun W and Shahrajabian M H. Therapeutic Potential of Phenolic Compounds in Medicinal Plants Natural Health Products for Human Health. Molecules. 2023; 28(4):1845. doi: 10.3390/molecules28041845.
CrossRef - Wang T, Gong X, Jiang R, Li H, Du W, Kuang G. Ferulic acid inhibits proliferation and promotes apoptosis via blockage of PI3K/Akt pathway in osteosarcoma cell. Am. J. Transl. Res. 2016;15(2):968–980.
- Ribeiro D, Freitas M, Silva A M S, Carvalho F, Fernandes E. Antioxidant and pro-oxidant activities of carotenoids and their oxidation products. Food Chem Toxicol. 2018;120;681-6992018. doi: 10.1016/j.fct.2018.07.060.
CrossRef - Ok S and Jeong W S. Optimization of extraction conditions for the 6-shogaol-rich extract from ginger (Zingiber officinale Roscoe). Prev Nutr Food Sci. 2012;(17)2:166-71. doi: 10.3746/pnf.2012.17.2.166.
CrossRef - Kravchenko I, Eberle L, Nesterkina M, Kobernik А. Anti-inflammatory and analgesic activity of ointment based on dense ginger extract (Zingiber officinale). J Herbmed Pharmacol. 2019; 8(2):126-132. doi: 10.15171/jhp.2019.20.
CrossRef - Fuloria S, Mehta Jyoti, Chandel Aditi, Sekar M, Rani NNIM, Begum Y. A Comprehensive Review on the Therapeutic Potential of Curcuma longa Linn. in Relation to its Major Active Constituent Curcumin. Front Pharmacol. 2022; 25(13): 820806. doi: 10.3389/fphar.2022.820806.
CrossRef - Hasan M R, Alotaibi B S, Althafar Z M, Mujamammi A H, Jameela J. An Update on the Therapeutic Anticancer Potential of Ocimum sanctum L.: ‘Elixir of Life. Molecules. 2023, 28(3):1193. doi: 10.3390/molecules28031193.
CrossRef - Scandiffio R, Geddo F, Cottone E, Querio G. Protective effects of (E)-β-caryophyllene (bcp) in chronic inflammation. Nutrients. 2020; 12(11): 3273. doi: 10.3390/nu12113273.
CrossRef - Sitarek P, Rijo P, Garcia C, Skała E, Kalemba D. Antibacterial, Anti-Inflammatory, Antioxidant, and Antiproliferative Properties of Essential Oils from Hairy and Normal Roots of Leonurus sibiricus L. and Their Chemical Composition. Oxid. Med. Cell. Longev. 2017:73840612017. doi: 10.1155/2017/7384061.
CrossRef - Jung Y Y, Hwang S T, Sethi G, Fan L, Arfuso F, Ahn K S. Potential anti-inflammatory and anti-cancer properties of farnesol. Molecules. 2018; 23(1): 2827. doi:10.3390/molecules23112827
CrossRef - Trujillo J, Chirino Y I, Molina-Jijón E, Andérica-Romero A C, Tapia E, Pedraza-Chaverrí J. Renoprotective effect of the antioxidant curcumin: Recent findings. Redox Biol. 2013; 17(1):448-56. doi: 10.1016/j.redox.2013.09.003.
CrossRef - Feng Y, An Qi, Zhao, Wu M. Beta-elemene: A phytochemical with promise as a drug candidate for tumor therapy and adjuvant tumor therapy. Biomed Pharmacother. 2024;172:116266. doi: 10.1016/j.biopha.2024.116266.
CrossRef - Javed H, Azimullah S, Abul Khair S B, Ojha S, Haque M E. Neuroprotective effect of nerolidol against neuroinflammation and oxidative stress induced by rotenone. BMC Neurosci. 2016;(17)58:1-12. doi: 10.1186/s12868-016-0293-4.
CrossRef - Ghosh S, Indukuri K, Bondalapati S, Saikia A K, Rangan L ChemInform Abstract: Unveiling the Mode of Action of Antibacterial Labdane Diterpenes from Alpinia nigra (Gaertn.) B. L. Burtt Seeds. ChemInform. 2014; 58(8):527-30. doi: 10.1002/chin.201401215.
CrossRef - Saddiq A A and Khayyat S A. Chemical and antimicrobial studies of monoterpene: Citral, Pestic. Biochem. Physiol. 2010; 98(1):89-93. doi: 10.1016/j.pestbp.2010.05.004.
CrossRef - McDougall J J and McKenna M K. Anti-Inflammatory and Analgesic Properties of the Cannabis Terpene Myrcene in Rat Adjuvant Monoarthritis. Int. J. Mol. Sci. 2022;(23)14: 7891. doi: 10.3390/ijms23147891.
CrossRef - Bhatti HN, Khan S S, Khan A, Rani M, Ahmad VU, Choudhary MI. Biotransformation of monoterpenoids and their antimicrobial activities. Phytomedicine. 2014; (21)12:1597-626. doi: 10.1016/j.phymed.2014.05.011.
CrossRef - Salehi B, Orhan I E, Jugran A K, Sumali, Jayaweera L D, Dias D A. Therapeutic potential of α-and β-pinene: A miracle gift of nature. Biomolecules. 2019;(9)11:738. doi: 10.3390/biom9110738.
CrossRef - Farhath MSS, Vijaya PP, Mumtaj P, Vimal M. A comparative study on antioxidant activity of essential oils and curcumin using thiobarbituric acid reactive substances. Journal of Chemical and Pharmaceutical Research. 2013; 5(9):219-221.
- Chavan MJ, Wakte PS, Shinde DB. Analgesic and anti-inflammatory activity of Caryophyllene oxide from Annona squamosa L. bark. Phytomedicine. 2010; (17)2:149-151. doi: 10.1016/j.phymed.2009.05.016.
CrossRef - Chauhan K. Commercial significance of medicinal and aromatic plants of India. South Florida Journal of Environmental and Animal Science. 2024;4(1):2-23. doi:10.53499/sfjeasv4n1-001.
CrossRef - Akande AA. and Aboaba SA. Natural Products and Therapeutics: What to know about Essential oils Essential Oils Therapeutic Effects as Anthelmintics. Book: Complimentary Contributor Copy. Publishing: Nova Medicine & Health, New York. December







