Manuscript accepted on :21-04-2025
Published online on: 29-05-2025
Plagiarism Check: Yes
Reviewed by: Dr. Akhtar Ali
Second Review by: Dr. Karuna Priyachitra
Final Approval by: Dr. Anton R Keslav
Punam Jyoti Borah1,2* and Ruma Sarma1
and Ruma Sarma1
1Department of Botany, Cotton University, Guwahati, Assam, India
2Department of Botany, Sibsagar University, Joysagar, Assam, India
Corresponding Author E-mail: punamborah1997@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3183
Abstract
FTIR and GC-MS were carried out to reveal the phytocompounds and their functional groups present in the methanolic leaf extracts of A. cathcartii. FTIR analysis resulted a total of five peaks indicated the presence of alcohol, amines, amino acids, alkynes, aldehyde, aliphatic groups, aromatics, unsaturated heterocycles, amides, nitro compound, alkanes, alkenes, phosphorous, and organosilicon compounds. GC-MS analysis resulted a total of 70 peaks consisting different compounds with different retention time, area percentage where 2,6,10,14-Hexadecatetraen-1-ol, 3,7,11,15-tetramethyl-, acetate, (E,E,E)-, Cyclohexanol, 1-methyl-4-(1-methylethyl)-, 3-Ethoxy-1,1,1,5,5,5-hexamethyl-3-(trimethylsilyloxy)trisiloxane, Anthranilic acid, N-methyl-, butyl ester, 3-Hydroxy-7,8-dihydro-.beta.-ionol, Benzophenone, 4-(trimethylsiloxy)-, Pentasiloxane, dodecamethyl-, Disiloxane, 1,3-diethoxy-1,1,3,3-tetramethyl-, 10-epi-γ-Eudesmol were some major compounds.
Keywords
A. cathcartii; FTIR; GC-MS; Medicinal Plant
Download this article as:| Copy the following to cite this article: Borah P. J, Sarma R. Phytoconstituents of Aristolochia cathcartii Hook.f. Leaf Extract; an Analysis through FTIR and GC-MS. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Borah P. J, Sarma R. Phytoconstituents of Aristolochia cathcartii Hook.f. Leaf Extract; an Analysis through FTIR and GC-MS. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/4mC6tj6 |
Introduction
Nature has bestowed upon us with the plants of medicinal importance constitute bioactive compounds and can be divided into primary and secondary metabolites. Primary metabolites directly involve in the primary living cell process and metabolism are pivotal for growth and development of the cell. Primary metabolites are also referred as central metabolites include examples of carbohydrates, fats and oils, amino acids, etc. On the other hand, the secondary metabolites are low molecular weight complex compounds in structure and produced by the biosynthesis modification such as methylation, acylation, glycosylation, and hydroxylation of primary metabolites helps in defensive mechanism of plants.1 Several factors including environmental and pathogenic stresses, genetic traits as well as nutrient availability influence the biosynthesis of these products. Based on the biosynthetic route secondary metabolites can be categorised into phenolic groups, terpenoids and nitrogen-containing compounds.2 They have been reported to perform different biological activities, such as antimicrobial, antifungal, antiviral, antiprotozoal, antioxidant, anticancer, and anti-inflammatory effects. Phenolic compounds probably the largest group of secondary metabolites includes the simple phenolics, tannins, coumarins, flavonoids, chromones and xanthones, stilbenes, and lignans are efficient in various pharmacological effects like antioxidants, anti-inflammatory, antimicrobial, antitumor, cholagogue, anthelmintic activity etc.3,4 The hidden treasure of natural remedies are the phytoconstituents of which identification is essential to reveal the actual composition of extracts behind therapeutical effects. Characterization of the bioactive compounds can be carried out employing different spectroscopy techniques such as UV-Visible, Nuclear Magnetic Resonance, Raman, and Mass spectroscopy. Fourier Transform Infrared Spectroscopy (FTIR) and Gas chromatography-mass spectrometry (GC-MS) which are emerging techniques in the present scenario in identification of functional groups as well as the structural elucidation of the compounds.5 An attempt has been made to analyse the phytoconstituents of Aristolochia cathcartii leaf extract and their functional groups through FTIR and GC-MS technique.
A. cathcartii is a species of Family Aristolochiaceae; commonly known as birthwort family belonging to the order Aristolochiales. Popular as Dutchman’s pipe for S-shaped flower, this species is distributed in the Eastern Himalayan Region (Nepal and North East India), Northern Bangladesh, North-west Burma, and South-central China. It is a large woody climber with greyish furrowed corky bark, broadly cordate or ovate leaves with small silky appressed hairs beneath the leaves. Flowers bloom in short racemes; two or three flower in each often in lower stem. Perianth are 6-8cm long, dense silky villous creamy white tubular with purplish violet vein bearded with long hairs, border of broad square mouth is occupied by violet papillose hair, limbs are yellowish with brownish or violet dots as shown in the figure 1. Presence of gynostemium is one of the characteristic features of the family. Fruits of this species are six ribbed linear obovoid with silky tomentose. Traditionally, people of North East India use the leaves, stem, rhizome, roots of this plant in treating various ailments including stomach disorders, liver problems, to promote urine flow, also for food poisoning, and as insect repellent.6
 |
Figure 1: Flower of A. cathcartiiClick here to view Figure |
Materials and Methods
Plant specimen was identified and authenticated by expert of related fields and preserved in mounted herbarium sheet as following the method described by Jain and Rao,7 and submitted to the BSI, Shillong (Accession No. 98879). were collected from Behali Wildlife Sanctuary, Assam, India after receiving proper permission from Government officials then washed under running tap water and dried under shade. The dried leaves are grinded in powder form and 10g of powder extracted in 250 ml of methanol using Soxhlet apparatus. The resulted extract was made into gel form using hot water bath at 30-40⁰c.
GC-MS analysis
The methanolic leaf extracts of A. cathcartii extract was subjected to Gas chromatography-mass spectrometry analysis in a chromatography device (model: Shimadzu GCMS 2010). Thickness of column was 0.25 μM with 30.0 m length and 0.25 mm diameter. The initial temperature of the column was set at 80ºC, then 1 μL of sample was injected into the device at injection temperature of 260⁰c with injector mode split less ratio of 20.0. The mass spectra were taken at a scan range of 50 to 1000 m/z. Ion source temperature was maintained at 200ºC. Helium was used as carrier gas at flow 1 mL/min passed through 65.9 kPa pressure. The programmed oven temperature was set from 80ºC to 280ºC in running time program hold for 54.67minutes. Handling of acquired data was done using GC-MS Software post-run analysis. Identification of the compound was based on the NIST 11 and PESTEI_3 libraries.8
FTIR analysis
Fourier-Transform Infrared Spectrometer (PerkinElmer) was used to detect the functional groups of the compound present in the plant methanolic extract of different concentration employing standard KBr pellet technique. The measured infrared absorption spectra were ranged from 450 cm-1 to 4000 cm-1. The chemical bonds in a compound were determined by the interpretation of the infrared absorption spectrum.
Results
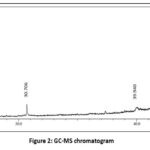 |
Figure 2: GC-MS chromatogramClick here to view Figure |
The bioactive compounds present in the methanolic leaf extract of A. cathcartii were analysed from the GC-MS chromatogram (Figure 2) accounted a total of 70 peaks and the retention time, area percentage, height percentage and m/z value are recorded. Among the identified compounds, some major compounds obtained from the GC-MS analysis are 2,6,10,14-Hexadecatetraen-1-ol, 3,7,11,15-tetramethyl-, acetate, (E,E,E)- (highest area percentage= 15.16%; retention time= 42.143)> Cyclohexanol, 1-methyl-4-(1-methylethyl)- (area percentage = 5.09%; retention time= 30.706)> 3-Ethoxy-1,1,1,5,5,5-hexamethyl-3-(trimethylsilyloxy)trisiloxane (area percentage = 4.65%; retention time= 42.327)> Anthranilic acid, N-methyl-, butyl ester (area percentage = 3.02%; retention time= 42.605)> 3-Hydroxy-7,8-dihydro-.beta.-ionol (area percentage = 2.98%; retention time= 39.940)> Benzophenone, 4-(trimethylsiloxy)- (area percentage = 2.70%; retention time= 42.255)> Pentasiloxane, dodecamethyl- (area percentage = 2.38%; retention time= 43.647)> Disiloxane, 1,3-diethoxy-1,1,3,3-tetramethyl- (area percentage = 2.25%; retention time= 43.489)> 10-epi-γ-Eudesmol (area percentage = 2.08%; retention time = 41.685)> Benzenepropanoic acid, 3,5-bis(1,1-dimethylethyl)-4-hydroxy-, methyl ester (area percentage = 1.91%; retention time = 27.178).
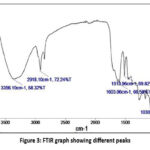 |
Figure 3: FTIR graph showing different peaksClick here to view Figure |
Functional group of phytocompounds constituted in the methanol leave extract of A. cathcartii revealed a total of five peaks as shown in Figure 3. The spectrum obtained in FTIR analysis showed frequency ranges from 3356.10 cm-1 to 1030.94 cm-1 and corresponds the presence of -OH, -NH, ≡C-H, -CH, -CH2-, -CH3, C=C, C=N, NH, NO2, CH3, CH2, Si-O and P-O functional groups. The obtained peaks indicate the presence of alcohol, amines, amides, amino acid, aldehyde, aliphatic groups, aromatics, unsaturated heterocycles, nitro compounds, alkanes, alkenes, alkynes, ethers, sulphur, phosphorous, and organosilicon compounds as listed in Table 1.
Table 1: Functional groups obtained from different peaks in FTIR analysis
| Peak Number | Peak (cm–1) | Transmittance | Functional Group | Possible Compound |
| 1 | 3356.10 | 68.32 | -OH, -NH, ≡C-H | Alcohol, amines, alkynes, aldehydes |
| 2 | 2918.10 | 72.24 | -CH, -CH2-, -CH3 | Aliphatic groups |
| 3 | 1603.06 | 66.58 | C=C, C=N, NH | Unsaturated aliphatic, aromatics, unsaturated heterocycles, amides, amines, amino acids |
| 4 | 1513.96 | 69.82 | NO2, CH3, CH2 | Nitro compound, alkanes, alkenes |
| 5 | 1030.94 | 51.67 | Si-O and P-O | Phosphorous compounds, organosilicons |
Discussion
Phytocompounds resulted from this investigation belong to different groups of secondary metabolites possesses various biological activities. For instance; 2,6,10,14-Hexadecatetraen-1-ol, 3,7,11,15-tetramethyl-, acetate, (E, E, E)- is a diterpenoid reported to has antioxidant, antibacterial, and urease enzyme inhibition potentialities while examined in the methanol extract of Strobilanthes glutinosus.9 Cyclohexanol, 1-methyl-4-(1-methylethyl)- P-menthan-1-ol is a p-menthane monoterpenoid and a volatile organic compound. The GC-MS analyses of leaf, stem, and roots of Withania somnifera revealed the presence of 3-Ethoxy-1,1,1,5,5,5-hexamethyl-3-(trimethylsilyloxy) trisiloxane which exhibits antibacterial and anticancer properties.10 Anthranilic acid, N-methyl-, butyl ester found to possess anti-apoptosis activity against targeted tumour necrosis factor receptor-1, tumour necrosis factor-related apoptosis inducing ligand-receptor 2, B-cell lymphoma 2, caspase-9 and caspase-3 proteins and anti-inflammatory activity against nuclear factor kappa B, inducible nitric oxide synthase, pregnane X receptor protein in the ethanol extract of Andrographis paniculata.11 The 3-Hydroxy-7,8-dihydro-β-ionol is a disaccharide glycoside and an aroma precursor isolated from the leaves of Camellia sinensis var. sinensis.12 Silicic acid, diethyl bis(trimethylsilyl) ester isolated from the essential oil of Citrus limon found to display antioxidant activity.13 Hexadecamethyl heptasiloxane is an important antifungal agent reported to be effective against Candida albicans and Trichophyton mentagrophytes and used in cosmetics as a film-forming polymer also has anti-inflammatory properties (www.biosynth.com). Methyl 3-bromo-1-adamantaneacetate isolated from the root parts of Bulbophyllum kaitense possess antimicrobial activity.14 The 2-(Acetoxymethyl)-3-(methoxycarbonyl) biphenylene is an aromatic compound, also act as antibacterial agent.15 Androst-4-en-9-thiocyanomethyl-11-ol-3,17-dione is a steroid which is a precursor for androgen and estrogen production.16 Strychane, 1-acetyl-20α-hydroxy-16-methylene- isolated from the methanol extract of Cassia fistula showed antibacterial activity.17 Hydrocinnamic acid, 3,4-bis(trimethylsiloxy)-, trimethylsilyl ester reported to perform anticancer and anti-inflammatory properties isolated from the leaves of Spondias mombin.18 Benzaldehyde, 2-nitro-4-trimethylsilyl- is an aromatic aldehyde which has antifungal activity, also reported to be present in Hibiscus vitifolius leaves.19 Fraction of methanolic extract of Lagenaria breviflora fruit yielded 2-(Acetoxymethyl)-3-(methoxycarbonyl) biphenylene which is said to be an antibacterial agent.20
Conclusion
The phytoconstituents resulted from our FTIR and GC-MS analysis owns different bioactive properties including antioxidant, antibacterial, antifungal, anti-inflammatory, anti-cancer, anti-apoptosis activities. Hence, the study of the phytocompounds may lead to open the door of designing new therapeutic formulations
Acknowledgement
We are grateful to the Institute of Advanced Study in Science and Technology (IASST) for providing facility to carry out the GC-MS analysis.
Funding source
The authors received no financial support for the research, authorship, or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author contributions
- Punam Jyoti Borah: Conceptualization, Methodology, Data Collection, Analysis, Writing – Original Draft
- Ruma Sarma: Editing, Visualization, Supervision
References
- Alqethami A and Aldhebiani A. Y. Medicinal plants used in Jeddah, Saudi Arabia: Phytochemical screening. Saudi J. Biol. Sci. 2021;28(1):805-812.
CrossRef - Selwal N, Rahayu F, Herwati A, Latifah E, Supriyono, Suhara C, Suastika I. B. K, Mahayu W. M and Wani A. K. Enhancing secondary metabolite production in plants: Exploring traditional and modern strategies. Agri. Food Res. 2023; 14:100702.
CrossRef - Seca A. M. L and Pinto D. C. G. A. Biological potential and medical use of secondary metabolites. Medicines (Basel). 2019;6(2):66.
CrossRef - Yeshi K, Crayn D, Ritmejeryte E and Wangchuk P. Plant secondary metabolites produced in response to abiotic stresses has potential application in pharmaceutical product development. Mol. 2022;27(1):313.
CrossRef - Balabhaskar R and Vijayalakshmi K. Identification of secondary metabolites from the ethanol extract of the leaves of Bauhinia tomentosa by GC-MS analysis. J. Pharm. Technol. 2021;14(5):2735.
CrossRef - Borah P. J, Borah D, Das U, Das T. J and Sarma R. A review on ethnopharmacological utility, traditional knowledge and phytochemistry of Aristolochia species in Assam, India. Sci. Biol. 2021;13(3):11027.
CrossRef - Jain S. K and Rao R. R. A Hand Book of Field and Herbarium Methods. Today & Tomorrow’s Printers and Publishers, New Delhi, 1977.
- Borah P.J and Sarma R. GC-MS analysis and qualitative phytochemical screening of Aristolochia assamica, a newly discovered rare medicinal plant species of India. Indian Nat. Prod. Resour. 2022;13(4):552-558.
- Aziz M, Ahmad S, Iqbal M. N, Khurshid U, Saleem H, Rehman K. U, Alamri A, Anwar S, Alamri A. S and Chohan T. A. Phytochemical, pharmacological, and In-silicomolecular docking studies of Strobilanthes glutinosus Nees: An unexplored source of bioactive compounds. Afr. J Bot. 2022; 147:618-627.
CrossRef - Lingfa L, Tirumala A and Ankanagari S. GC-MS profiling of anticancer and antimicrobial phytochemicals in the vegetative leaf, root, and stem of Withania somnifera (L.) Dunal. J. Second. Metab. 2024;11(1):63-77.
CrossRef - Manisha P and Prince S. E. An In silico approach to analyse the potential compound of Andrographis paniculata against the targets involved in the apoptotic and inflammatory pathways. Indian J. Pharm. Sci. 2023;85(5):1291-1301.
- Ma S. J, Watanabe N, Yagi A and Sakata A. The (3R,9R)-3-Hydroxy-7,8-dihydro-β-ionol disaccharide glycoside is an aroma precursor in tea leaves. Phytochem. 2001;56(8): 819-825.
CrossRef - Prabha M, Brintha M and Beena L. Molecular profiling and antioxidant potential of Citrus limon (L.) Burm. F Fruits. Vol. Essent. Oil. 2021; 8(4): 8360-8373.
- Kalaiarasan A, Kumar P and John S. A. Bio chemical investigation of Bulbophyllum kaitense root by GC-MS. Eastern Ghats of India. Nat. Sci. 2012;10(2).
- Shushizadeh M. R, Behroozi S, Behfar A and Nazemi M. Antibacterial activity and GC-Mass analysis of organic extract from Persian Gulf Haliclona sp. Pharmacophore. 2018;9(2):19-24.
- Akintunde O. G, Lairbe O. C, Abakpa S. A. V, Thomas F. C, Akinloye O. A and Ajibola E. S. Bioactive components in ethanol extract of Citrullus lanatus rind using Gas Chromatography- Mass Spectroscopy. Niger. Chem. Res. 2023;28(1):059-073.
CrossRef - Kadhim M. J, Mohammed G. J and Hameed I. H. In vitro antibacterial, antifungal and phytochemical analysis of methanolic extract of fruit Cassia fistula. J. Chem. 2016;32(3).
CrossRef - Okwute S. K, Ibikunle G. F and Ogbadoyi E. O. A 2,4,6-Trisubstituted Phenol and Trimethylsilyoxy derivatives of Hydrocinnamic Acid and Benzene Acetic Acid from cytotoxic leaf extract of Spondias mombin Linn (Anacardiaceae). Curr. Chem. Pharm. Sci. 8(1): 113.
- Krishnaveni A, Umamageshwari K, Prithivirajan S, Bairavi M and Kumar T. V. R. Gas Chromatography-Mass Spectrometry analysis of hydroalcoholic extract of Hibiscus vitifolius Linn Leaves. J. Res. Pharmacol. Pharmacother. 2024;13(3):350-356.
- Adeyemi M. A, Ekunseitan D. A, Abiola S. S, Dipeolu M. A, Egbeyale L. T and Sogunle O. M. Phytochemical analysis and GC-MS determination of Lagenaria breviflora Fruit. Int. J. Pharmacogn. Phytochem. Res. 2017; 9(7):1045-1050.







