Manuscript accepted on :01-05-2025
Published online on: 20-05-2025
Plagiarism Check: Yes
Reviewed by: Dr. Pravinkumar Darji
Second Review by: Dr. Radha A
Final Approval by: Dr. Anton R Keslav
Binoy Varghese Cheriyan1 , Kaniga Pandi1*
, Kaniga Pandi1* , Jubilee Ramasamy2
, Jubilee Ramasamy2 , Rithikha Sooriyaprakash1
, Rithikha Sooriyaprakash1 and Sam Jebaraj1
and Sam Jebaraj1
1Department of Pharmaceutical Chemistry, Saveetha College of Pharmacy, Saveetha Institute of Medical and Technical Sciences, Chennai, Tamil Nadu, India.
2Department of Pharmacology, Saveetha College of Pharmacy, SIMATS, Chennai, Tamil Nadu, India.
Corresponding Author E-mail: kanigapharma@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3155
Abstract
Traditional Chinese medicine has long utilized Hedyotisdiffusa (Bai Hua She She Cao) for its diverse therapeutic properties, including anticancer potential. The study examines its mechanisms of action, bioactive compounds, and synergistic effects with conventional cancer therapies.A literature review analyzed studies on Hedyotisdiffusa, focusing on its anticancer effects, including apoptosis induction, metastasis suppression, angiogenesis inhibition, and modulation of molecular pathways like NF-κB, STAT3, and VEGF.Hedyotisdiffusa demonstrated significant anticancer activity, targeting key pathways and reducing cancer cell proliferation, metastasis, and angiogenesis. Its bioactive compounds, such as polysaccharides and flavonoids, enhanced chemotherapy and radiation therapy efficacy while mitigating side effects.By integrating traditional and modern medicine, Hedyotisdiffusa presents a promising complementary cancer therapy. Its clinical applications require more investigation and optimize its role in oncology.
Keywords
Apoptosis induction; Bioactive compounds; Cancer therapy; Hedyotisdiffusa; Traditional Chinese Medicine
Download this article as:| Copy the following to cite this article: Cheriyan B. V, Pandi K, Ramasamy J, Sooriyaprakash R, Jebaraj S. Phytochemicals, Pathways, Mechanisms and Clinical Applications: A Comprehensive review on Hedyotis diffusa(Bai Hua She She Cao) in Cancer Management. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Cheriyan B. V, Pandi K, Ramasamy J, Sooriyaprakash R, Jebaraj S. Phytochemicals, Pathways, Mechanisms and Clinical Applications: A Comprehensive review on Hedyotis diffusa(Bai Hua She She Cao) in Cancer Management. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/4j8qECx |
Introduction
Cancer, which is defined by the unchecked development and spread of aberrant cells, continues to rank among the world’s top causes of mortality. Globally, the incidence and mortality burden of cancer is rising quickly.12018 had 18.1 million cancer diagnoses and 9.6 million cancer-related deaths.2Malignancies of the colon, lungs, breast, and prostate are among the most common tumours, whereas malignancies of the liver, stomach, and lungs are among the most lethal.In China, there are 3.6 million new instances of cancer and 2.2 million deaths from the disease each year.3Despite advances in cancer diagnosis and treatment, managing the disease remains challenging. Radiation, chemotherapy, and surgery are examples of conventional therapies that frequently have serious side effects, such as toxicity, immunological suppression, and a decreased quality of life.Drug resistance and variability in patient responses further limit treatment efficacy, while high costs restrict access, particularly in resource-limited settings. These challenges underscore the need for alternative, cost-effective, and less toxic therapies.4Herbal medicine has a long history of being used to treat cancer because of its anti-inflammatory properties and wealth of anticancer ingredients. It has been demonstrated that these elements affect cancer immunology, the tumour microenvironment, and the course of treatment both directly and indirectly.5,6Natural and herbal remedies have long been used in traditional medicine to treat a range of ailments, including infections and cancer.7,8
In this context, Traditional Chinese Medicine has gained attention for its potential to address these limitations, offering natural, holistic approaches that may enhance therapeutic outcomes and improve patient well-being.Traditional Chinese medicine treats cancer holistically, emphasising the body’s natural defences and re-establishing equilibrium (Fig.1.). Rooted in concepts like Yin-Yang harmony and Qi flow, TCM views cancer as a result of internal imbalances caused by factors such as stagnation, toxins, and deficiencies. Treatment strategies aim to “clear heat,” “detoxify,” and “invigorate blood circulation,” often using a combination of herbal medicines, acupuncture, dietary adjustments, and lifestyle modifications.9Traditional Chinese Medicine herbal formulas target not only the tumor but also the surrounding microenvironment, reducing inflammation,10boosting immune function, and alleviating side effects of conventional treatments. This integrative approach has gained attention for its potential to enhance therapeutic outcomes, improve patients’ quality of life, and provide personalized care tailored to individual needs.11
 |
Figure 1: Methods involved in Traditional Chinese Medicine
|
Hedyotisdiffusa, commonly known as Bai Hua She She Cao, a popular herb in Traditional Chinese Medicine (TCM), Oldenlandiadiffusa (Willd)Roxb is known for its anti-inflammatory and detoxifying qualities.12Traditionally, it has been used to “clear heat” and “remove toxins,” aligning with TCM principles for treating conditions like abscesses, infections, and inflammations (Fig.2).13Over time, its applications have extended to cancer therapy, particularly for digestive system cancers,14liver cancer,15and lung cancer.16The herb is often incorporated into TCM formulations and prescribed alongside other medicinal plants to enhance its efficacy.Packed with beneficial substances including flavonoids, polysaccharides, and terpenoids, Hedyotisdiffusa exhibits potent anticancer,17anti-inflammatory,18and immune-modulating effects.Hedyotisdiffusa exhibits significant anticancer properties through multiple mechanisms of action. By causing apoptosis and upsetting the cell cycle, its bioactive substances which include flavonoids, polysaccharides, and terpenoidsinhibit the growth of cancer cells. The herb also stops metastasis by stopping cancer cells from migrating and invading.Hedyotisdiffusaalso has anti-angiogenic properties, which stop new blood vessels from growing and supplying tumours. By decreasing inflammation and boosting immune responses to cancer cells, it modifies the tumour microenvironment.These mechanisms collectively make it a promising natural agent for combating cancer, with the potential to complement conventional therapies while minimizing side effects. Its holistic approach not only targets cancer cells but also supports overall health, making it a valuable component of integrative cancer care in TCM practices. Investigating medicinal herbs like Hedyotisdiffusa is vital for overcoming the limitations of conventional cancer treatments. With its proven anticancer properties, such as inducing apoptosis, inhibiting metastasis, and modulating immunity,19Hedyotisdiffusa offers a natural, cost-effective alternative with fewer side effects. Due to its anti-tumor efficacy in animals with tumours and anti-proliferative activity in cancer cells, H. diffusa has drawn more and more attention.20-23It is the most commonly suggested Chinese herb for patients with breast and colon cancer, according to Taiwan’s National Health Insurance Research Database.24, 25Research into its mechanisms can enhance current therapies, promote integrative medicine, and improve patient outcomes while advancing sustainable and accessible cancer treatments.This review’s goal is to investigate Hedyotisdiffusa’s potential for therapeutic use (Bai Hua She She Cao)in cancer treatment, focusing on its role within the framework of Traditional Chinese Medicine (TCM).
 |
Figure 2: Role of Hedyotisdiffusa in Traditional Chinese Medicine (TCM)
|
The scope includes a discussion of preclinical and clinical evidence, its integration into TCM practices, and future directions for research and therapeutic development, emphasizing its promise as a natural, holistic approach to cancer care.
Materials and Methods
A comprehensive literature search was conducted using databases such as PubMed, Scopus, Web of Science, and Google Scholar up to 2025. The search utilized the keywords “Hedyotisdiffusa,” “cancer,” “anticancer activity,” “mechanism of action,” “toxicity,” “drug interaction,” and “phytochemicals,” with Boolean operators (AND, OR, NOT) to refine the results. Initially, 1,250 articles were retrieved. After removing 200 duplicates and screening titles and abstracts for relevance, 850 articles were excluded due to irrelevance or duplication. Full-text screening was conducted on the remaining 200 articles, resulting in the inclusion of 125 studies based on the inclusion and exclusion criteria. The inclusion criteria encompassed original research articles published in English, focusing on the anticancer effects of Hedyotisdiffusa through in vitro, in vivo, or clinical studies. Exclusion criteria included review articles, editorials, conference abstracts, non-English articles, and studies that did not specifically address cancer or the anticancer mechanisms of Hedyotisdiffusa. (Fig.3.), illustrate the research process based on the PRISMA guidelines, which is typically used for systematic reviews.
 |
Figure 3 Flowchart based on the PRISMA guidelines, which is typically used for systematic reviews
|
Hedyotisdiffusa (Bai Hua She She Cao) in Traditional Chinese Medicine
For millennia, Traditional Chinese Medicine (TCM) has made extensive use of Hedyotisdiffusa (Bai Hua She She Cao), primarily for its detoxifying and heat-clearing properties. Historically, it has been employed to treat conditions such as infections, abscesses, and inflammations, aligning with TCM theories that associate cancer with heat, toxin accumulation, and blood stagnation. TCM approaches to cancer therapy focus on clearing heat, removing toxins, and promoting blood circulation to restore balance and inhibit tumor growth. Bai Hua She She Cao is commonly prepared as decoctions, powders, or extracts, often combined with other herbs to enhance its efficacy. It is a key component in various TCM prescriptions, such as “Clear Heat and Detoxify Decoction” and “Anti-Toxin Combination,” which are used to manage cancers of the digestive system, liver, and lungs. These formulations are tailored to individual patients, addressing not only the tumor but also systemic imbalances, making Hedyotisdiffusa an integral part of TCM cancer therapy.26
Phytochemical Composition of Hedyotisdiffusa
China, Japan, Indonesia, and other parts of the Orient and tropical Asia are home to the annual herb H. diffusa.27Reaching a height of 50 cm, the plant is usually found growing in damp fields and farming ridges.The papilla was seen in the stem’s transverse section, which is glabrescent to glabrous and terete to slightly flattened. The opposite, linear, narrowly elliptic, sessile or subsessile leaves are 1-4 × 0.1-0.4 cm and have a drying membrane. The corolla is white, and the pedicelled blooms are paired in axillary racemes.30Techniques for distinguishing H. diffusa from closely related species, such Hedyotiscorymbosa, have been devised, to ensure proper medicinal use.These methods include DNA sequencing,31gas chromatography-mass spectrometer (GC-MS), high performance liquid chromatography (HPLC),29and thin-layer chromatography (TLC).28Numerous studies on H. diffusa have identified a number of significant phytochemicals, such as iridoids,32triterpenes,33flavonoids,34anthraquinones,35phenolic acids,36and their derivatives, sterols,37alkaloids,38volatile oils,39polysaccharides,40cyclotides,41and coumarins,42because of the advancement of analysis technologies like nuclear magnetic resonance–mass spectrometers (NMR-MS), liquid chromatograph–mass spectrometers (LC-MS), and mass spectrometers (MS) (Fig.4).
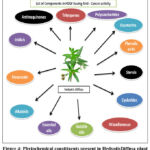 |
Figure 4: Phytochemical constituents present in HedyotisDiffusa plant
|
These compounds work synergistically to target multiple pathways involved in cancer development, including inflammation, angiogenesis, and immune evasion, making Hedyotisdiffusa a promising natural therapeutic in cancer treatment.These substances work in a number of ways, such as triggering apoptosis, preventing the growth of cancer cells, and preventing angiogenesis and metastasis. Compared to other cancer-targeting herbs like Scutellariabaicalensis, which is abundant in baicalin and wogonin, or Curcuma longa (turmeric), known for its curcuminoids, Hedyotisdiffusa offers a broader spectrum of bioactive compounds that work synergistically. While Scutellariabaicalensis is renowned for its anti-inflammatory and apoptotic effects,43and Curcuma longa excels in inhibiting metastasis and oxidative stress,44Hedyotisdiffusa uniquely integrates immune modulation, detoxification, and angiogenesis inhibition into its mechanisms. This diverse phytochemical composition makes it highly adaptable for use in both standalone and integrative cancer therapies, offering complementary benefits when combined with other natural or conventional treatments.Using a CRC mouse xenograft model, researchers showed the anticancer benefits of EEHDW in vivo in 2012. By inhibiting STAT3 phosphorylation, they were able to diminish the tumor’s weight and volume. Both in vitro and in vivo, EEHDW demonstrated strong anti-colorectal cancer efficacy against HCT-8, HT-29, HCT-116, and SW620 cells. It controlled inflammatory (IL-1β, IL-6, IL-4, IL-10, TNF-α) and angiogenic (COX-2, iNOS, eNOS, HIF-1α) factors and downregulated oncogenes (Bcl-2, Bax, Pim-1, p53), affecting tumor angiogenesis, proliferation, and apoptosis (Fig.5 & 6). These effects suggest its role in modulating key signaling pathways, including p38, AKT, STAT3, JNK, and ERK1/2.45, 46
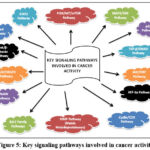 |
Figure 5: Key signaling pathways involved in cancer activity
|
Mechanisms of Action of HedyotisDiffusain Cancer Therapy
Hedyotisdiffusa, a well-known medicinal herb in traditional Chinese medicine, exhibits significant anticancer properties through multiple mechanisms (Fig.5).By controlling important signalling pathways like the PI3K/Akt and MAPK pathways, it induces apoptosis in cancer cells, which results in programmed cell death. This is one of its main mechanisms of action. Furthermore, it has been demonstrated that Hedyotisdiffusa inhibits the growth of cancer cells by inhibiting cyclins and cyclin-dependent kinases (CDKs), thereby arresting the cell cycle at critical checkpoints. The herb also plays a crucial role in impeding metastasis by downregulating matrix metalloproteinases (MMPs), which are responsible for tumor invasion and migration. Furthermore, Hedyotisdiffusa exerts anti-angiogenic effects by modulating VEGF expression, reducing the blood supply to tumors and restricting their growth. Its ability to enhance immune responses, especially by stimulating natural killer (NK) and T cells, further contributes to its anticancer efficacy. Additionally, the herb exhibits strong anti-inflammatory and antioxidant properties, mitigating oxidative stress and chronic inflammation, which are key factors in tumor progression. These multifaceted mechanisms suggest that Hedyotisdiffusa could serve as a potential complementary or alternative therapeutic agent in cancer treatment.
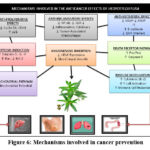 |
Figure 6: Mechanisms involved in cancer prevention
|
Anti-Proliferative Effects
Hedyotisdiffusa exhibits multifaceted mechanisms in cancer therapy, targeting various aspects of tumor development and progression. It demonstrates antiproliferative effects by inhibiting cancer cell growth and survival, often through cell cycle arrest. The herb induces apoptosis via both mitochondrial and death receptor pathways, activating key caspases and pro-apoptotic proteins. Its anti-metastatic properties suppress cancer cell invasion and migration, while its anti-inflammatory effects modulate the tumor microenvironment, reducing inflammation-driven cancer progression. Additionally, Hedyotisdiffusa boosts immune responses against cancer cells and inhibits angiogenesis, which stopsthe development of new blood vessels necessary for the growth and dissemination of tumours.These interrelated processes highlight its potential as a whole anticancer treatment.The study highlights the multifaceted anticancer effects of HedyotisdiffusaWilld (HDW), particularly its role in inhibiting theexpansion of colon cancer HT-29 cells. Using ethanol extracts of HDW (EEHDW), the research demonstrates that its antiproliferative activity is mediated through cell cycle arrest at the G1/S transition, a critical checkpoint in DNA synthesis and replication. The study provides evidence that EEHDW downregulates key regulators of the G1/S phase, including Cyclin D1, CDK4, and PCNA, while upregulating the expression of p21, a known proliferation inhibitor. These findings underline the ability of HDW to disrupt uncontrolled cell division, a hallmark of cancer progression.47The chloroform extract of HDW, CEHDW, inhibits the RAS/ERK and PI3K/AKT signalling pathways to provide anticancer effects. In SW620 CRC cells, it inhibits AKT and ERK activation,48and downregulates Bcl-2, cyclin D1, CDK4, survivin, PCNA, and Bax, which lowers proliferation and causes apoptosis. Multiple drug resistance (MDR) is one of the main causes of chemotherapy failure. In HCT-8/5-FU cells, the ethanol extract of HDW (EEHDW) inhibits P-glycoprotein (P-gp), ABCG2, and PI3K/AKT pathway phosphorylation to fight drug resistance. It inhibits CDK4, PI3K, p-Akt, Bcl-2, and cyclin D1, while upregulating Bax, p21, and PTEN, reducing cancer cell viability, colony formation, and reversing multidrug resistance (MDR).49
Apoptosis Induction
Pro-apoptotic proteins Bax, caspase-9, and caspase-3 were activated by EEHDW therapy, whereas phosphorylation of the STAT3 pathway caused by IL-6 was markedly reduced. Additionally, it downregulated CDK4, cyclin D1, and Bcl-2, contributing to an increased local inflammatory milieu and accelerated tumor growth.50In order to combat tumour angiogenesis, EEHDW may also suppress VEGF-A expression levels and stop HT-29 cells from progressing from G1 to S. In a CMX model, EEHDW lowers intra-tumor microvascular density (MVD) early on by inhibitingTarget genes of the Sonic Hedgehog (SHH) signalling pathway include VEGF-A and VEGFR2. Treatment with EEHDW may cause DNA damage, lower mitochondrial membrane potential, and raise the HT-29 cell’s Bax/Bcl-2 ratio, indicating that EEHDW suppressed HT-29 cell growth through a mitochondrion dependent mechanism.51,52Cytochrome c, encoded by the nuclear genome, is found in the mitochondria, which regulate cell death by controlling members of the Bcl-2 family, itcauses cell death.
In short, cytochrome C may be stimulated to leave the mitochondria and enter the cytoplasm by a variety of apoptosis inducers, including growth factor deficiency or DNA damage agentswhich then form the apoptotic complex by binding to the activating factor 1 of apoptotic peptidase (Apaf-1). This compound’s synthesis initiates the caspase cascade.53Morphologically, it causes cell lysis, chromatin condensation, nuclear fragmentation, and chromosomal DNA fragmentation,blebbing, and shrinkage.54
Anti-Metastatic Properties
The process by which epithelial cells undergo morphological and mesenchymal changes is known as the epithelial mesenchymal transition (EMT). Mesenchymal stem cells have the ability to migrate and invade, whereas the polarity and cell-cell adhesion of epithelial cells are lost. In particular, the epithelial sign E-cadherin is expressed less frequently in EMT cells, while the EMT marker Vimentin is expressed more frequently.55Epithelial cells contain the Ca2+-dependent adhesion molecule E-cadherin that is necessary for cell differentiation and tissue growth. It is a transmembrane glycoprotein that preserves the polarity and structural integrity of epithelial cells while mediating intercellular adhesion.56Utilising experiments on cell colony development and wound healing,Oleanolic acid inhibited tumor cell EMT by downregulating vimentin and FOXA1 while increasing E-cadherin.57EEHDW halted the progression of the cell cycle from G1 to S by upregulating p21 and downregulating cyclin D1, PCNA, and CDK4, with effects positively related to both concentration and treatment duration.58By decreasing the proportion of SP in HT-29 cells and the expression of Lgr5, PCNA, ABCB1, survivin, β-catenin, and c-Myc, which are associated with the inhibition of the Wnt/β-catenin pathway and ABC transporters,59EEHDW also inhibited CRC stem cells. Furthermore, via altering the TGF-β signalling pathway and reducing cell adhesion, migration, and invasion,60EEHDW stopped the spread of HCT-8/5-FU cells. With supplementary effects on TGF-β, Wnt/β-catenin, SHH, ABC, and mitochondrion-dependent pathways, HDW extracts mainly target the cell cycle, PI3K/AKT, RAS/ERK, and STAT3 pathways in colorectal cancer. By encouraging apoptosis and inhibiting angiogenesis, migration, invasion, proliferation, and resistance to treatment, HDW prevents colon cancer. AKT, PIK, TP53, BRAF, CDK2, and RAF are among the important targets identified by network pharmacology. The function of HDW in controlling contacts, cell motility, and tumor-related pathways is highlighted by GO analysis.61Through different apoptotic pathways, the H-EtOAc fraction and its main constituent, DMQ, demonstrated strong anticancer effects on HepG2 cells, according to the study. The H-EtOAc fraction engaged the death receptor pathway, whereas DMQ specifically triggered the mitochondrial apoptotic pathway. Both upregulated caspase-3, -8, and -9 activity, along with pro-apoptotic p53, influencing apoptosis and cell cycle arrest via Fas, Bax, Bcl-2, and p21. PFT-α inhibited p53-dependent apoptosis, highlighting the importance of functional p53 signaling. Given TP53 mutations’ role in therapy resistance,To assess the effect of these alterations on HDW’s anticancer activity, more investigation is required.62
Anti-Inflammatory Effects
Uncontrolled inflammation is highly associated with the onset of tumours, metastases, and multidrug resistance.63Tumour angiogenesis and metastasis are facilitated by inflammation, and tissue microenvironments associated with chronic inflammation can contribute to the development of cancer by causing DNA damage and gene alterations.63, 65Important molecular participants in tumor-suppressive and carcinogenic processes include androgen/estrogen receptors,66FOXM1, NF-κB, STAT3, Wnt/β-catenin, HIF-1α, and NRF2..Active HDW ingredients like kaempferol reduce inflammation by downregulating factors such as COX-2, iNOS, IKK, and blocking the PI3K/Akt, MAPK, and VEGF pathways in RAW264.7 cells.67HMA lowers inflammatory cytokine levels in A549 cells by 87–81%, 34.94%, 63.64%, 72.57%, and 21.99%.68Additionally, quercetin (10 μM) activates the RIG-I promoter,increasing the synthesis of IFN-α and IFN-β in human melanoma B16 and A375 cell lines.69
Immune Modulation
The immune system’s bioactivities and functions are demonstrated by the polysaccharides extracted from HDW. Polysaccharides can boost CD3+ andCytokine-induced killer (CIK) cells, or CD56+, which improves CIK cells’ cytolytic activity against cancer cells in vitro, according to earlier studies. Polysaccharide-treated CIK cells show increased IFN-γ and TNF-α levels and exhibit greater cancer cell-killing efficiency compared to non-treated CIK cells.70By increasing the synthesis and release of inflammatory cytokines in immune cells, HDW extracts were also shown to have anticancer activity. This suggests that such antitumor activity should be further explored for new cancer immunotherapy.
Angiogenesis Inhibition
Angiogenesis is the process by which existing blood arteries give rise to new ones.
An essential process for the development and dissemination of cancer cells is tumour angiogenesis, therefore, inhibiting tumour angiogenesis may help prevent the progression of cancer.71Isoscutellarein, quercetin, and kaempferol that are separated from HDW prevent the growth and angiogenesis of non-small cell lung cancer by inhibiting the PI3K/Akt, MAPK, and VEGF pathways.72Additionally, 4-vinylphenol, a styrene metabolite from p-coumaric acid, showed hepatotoxic and pulmonary toxicity at high doses.73In breast cancer, 4-vinylphenol (2 mg/kg) inhibited angiogenesis by suppressing the PI3K/Akt and p38 MAPK pathways, reducing fibronectin, laminin, collagen production, and VEGFR expression in HUVECs, thus decreasing endothelial cell adhesion and ECM breakdown.
Results
Hedyotisdiffusa (Bai Hua She She Cao) as a Therapeutic Agent in Cancer Treatment
Because of its extensive biological and pharmacological effects on cancer, H. diffusa has been utilised for a long time in China. This review also examines its pharmacological actions across various cancer cell lines.74
Anti-Colorectal Cancer Activity
A common cancer, colon cancer is characterised by unchecked cell proliferation in the colon or rectum and is frequently caused by environmental, nutritional, and hereditary causes. Despite improvements in screening and treatment, It continues to be a major global cause of cancer-related death.By encouraging apoptosis, preventing cell division, and lowering angiogenesis, Hedyotisdiffusa exhibits notable anti-colon cancer properties. Its bioactive chemicals inhibit tumour growth and metastasis by targeting pathways such as NF-κB and Wnt/β-catenin. H. diffusa is widely used in colorectal cancer (CRC) treatment. In vitro, its ethanol extract suppresses HT-29 cell proliferation and induces apoptosis by triggering DNA fragmentation, mitochondrial dysfunction, caspase activation, and an increased Bax/Bcl-2 ratio. It downregulates cyclin D1, CDK4, Bcl-2, and PCNA expression, inhibits G1–S progression, and upregulates pro-apoptotic Bax.These findings suggested that H. diffusa may have anti-colorectal cancer cell effects through a variety of mechanisms, including cell cycle arrest, the IL-6/STAT3 pathway, and the mitochondria-dependent route.75Animal studies also validated the mechanism.76At concentrations of 0.06 mg/mL to 0.20 mg/mL, the ethanolic extract of Hedyotisdiffusa inhibited CT-26 cells in a concentration-dependent manner, with inhibition rates ranging from 35.46% to 71.84%.77The ethanolic extract treatmentabolished 5-fluorouracil resistance in HCT-8/5-FU cells by downregulating the expression of P-gp and ABCG2.78
Ursolic acid (IC50 71 µM) and 2-hydroxymethyl-1-hydroxy anthraquinone (IC50 45 µM), two substances that were separated from H. diffusa, prevented the growth of Caco-2 cells. Ursolic acid may cleave the Poly (ADP-ribose) polymerase (PARP)in order to exert its inhibitory effect.79Without causing any discernible damage, the ethanol extract of HDW (EEHDW) inhibits the growth of cancer in vivo. EEHDW prevented STAT3 phosphorylation in CRC mice, which reduced cell division and caused cell death. Additionally, EEHDW decreased CDK4, Bcl-2, and Cyclin D1 levels, while increasing p21 and Bax expression.80
Anti-Breast Cancer Activity
Breast cancer is the most prevalent disease among women globally, which develops when breast cells grow abnormally. Because of its aggressive subtypes and metastatic potential, it continues to be a major cause of cancer-related deaths despite advancements in early identification and targeted therapy.81,82Hedyotisdiffusa has shown potential in breast cancer treatment by inducing apoptosis, inhibiting proliferation, and blocking metastasis. Its active compounds modulate key signaling pathways, including those related to inflammation and oxidative stress, reducing tumor progression.At half maximum effective dosages (EC50) of 18.62 ± 2.71 µM for 24 hours and 42.19 ± 3.84 µM for 48 hours, methylanthraquinone produced from H.Diffusa inhibited MCF7 cells, causing apoptosis through the Ca2+/calpain/caspase-4 pathway.83Furthermore, Bcap37 cell growth and the protein tyrosine kinases v-src and pp60srcwere suppressed by 2-hydroxy-3-methyl anthraquinone (IC50 57 µM) and 2-hydroxy-1-methoxy anthraquinone (IC50 65 µM).84
Anti-Liver Cancer Activity
The fifth most prevalent type of cancer is liver cancer. Hepatocellular carcinoma (HCC), the sixth most common type of cancer and the cause of almost one million fatalities each year, continues to be a significant obstacle in biomedical research. A gene mutation model serves as the foundation for much of the research and treatment development techniques for cancers and cancer-related disorders.85Hedyotisdiffusa, a traditional medicinal herb, exhibits anti-cancer effects on liver cancer by inducing apoptosis, inhibiting tumor growth, and suppressing metastasis. Its bioactive compounds, such as iridoids and flavonoids, target cancer cell proliferation and inflammatory pathways, the prevention of blood metastases in H22 mice by an aqueous preparation of H. diffusa.86After being given three doses of H. diffusa extract (0.25, 0.5, and 1.0 mg/kg), the weights of the body and immunological organs increased. In vitro, HepG2 cell proliferation was markedly inhibited by the aqueous extract of H. diffusa (1.25–10 mg/mL) through the induction of S phase delay and dose-dependent cell arrest at the G0/G1 phase.87Additionally, the invasion of MHCC97-H cells during the epithelial-mesenchymal transition induced by TGF-β1 was prevented by a 200 µg/mL dosage of H. diffusa complete flavone extract. Vimentin protein expression may be upregulated and e-cadherin protein expression may be downregulated in order to accomplish this.88HepG2 cells were subjected to apoptosis by 1,3-Dihydroxy-2-Methylanthraquinone (79 and 157 µmol/L) and ethyl acetate extract (100 and 200 µg/mL). This resulted in increased activation of caspase-3, -8, and -9 as well as dose-dependent increases in Bax, p53, Fas, FasL, p21, and cytoplasmic cytochrome C.89Nine compounds from H. diffusa, including ursolic acid, oleanolic acid, and several anthraquinones, were examined for their anti-liver cancer activities at doses ranging from 1 to 200 µM. HepG2 cell survival was considerably decreased by ursolic acid (IC50 36.63 µM).90Furthermore, the v-src and pp60src protein tyrosine kinases were inhibited by 2-hydroxy-3-methyl anthraquinone (IC50 51 µM) and 2-hydroxy-1-methyl anthraquinone (IC50 62 µM).91
Anti-Lung Cancer Activity
Cancerous cells can develop in the lungs, a condition known as lung carcinoma. One of the most deadly and challenging malignancies to cure, lung cancer often spreads to other parts of the body at an early stage. The World Health Organisation reports that for several decades, lung cancer has topped all cancers globally in terms of incidence.92Hedyotisdiffusa exhibits anti-lung cancer properties by inducing apoptosis, inhibiting tumor cell proliferation, and suppressing metastasis. Its bioactive compounds regulate signaling pathways like PI3K/Akt and MAPK, reducing cancer progression.A549 and H1355 cells were shown to be concentration-dependently inhibited by treatment with an aqueous extract of H. diffusa (0–200 µg/mL), while LLC cellswere unaffected.93Furthermore, two compounds associated with the mitochondrial apoptotic pathway, 2-hydroxy-3-methyl anthraquinone (IC50 66 µM) and 2-hydroxy-1-methoxy anthraquinone (IC50 79 µM), caused SPC-1-A cells to undergo apoptosis.94Hedyotisdiffusa injection significantly reduced the lifespan of lung adenocarcinoma cells in vitro and inhibited tumour growth in BALB/c nude mice in vivo. By inhibiting the Bcl-2 gene and controlling VDAC 2/3 through Bax, it caused ferroptosis. Furthermore, the study showed significant improvements in clinical symptoms, such as headache, dry mouth, sputum production, cough, chest tightness, wheezing, and shortness of breath, which ultimately improved quality of life.95
Anti-Leukemia Activity
Hedyotisdiffusa exhibits anti-leukemia activity by inducing apoptosis, inhibiting cell proliferation, and modulating immune responses. Its bioactive compounds target key pathways such as PI3K/Akt and MAPK, effectively reducing leukemia progression.H. diffusa aqueous and ethanolic extracts have shown anti-leukemia activity in various cancer cell lines. The aqueous extract (0.01–4150 µg/mL) affected WEHI-3 cells in murine leukemia, increased T- and B-cell proliferation in leukemic animals (16 and 32 mg/kg in vivo), and inhibited CEM cell growth by enhancing P53 expression in vitro.96, 97The ethanolic extract (IC50 4.62 mg/mL) arrested HL-60 cells at the G0/G1 phase and sub-G1 population, inducing DNA damage and activating caspase-3, -8, and -9 to inhibit cell proliferation.982-hydroxy-3-methyl anthraquinone (0–80 µM) accelerated U937 cell death by downregulating p-ERK1/2 and activating p-p38MAPK.99Another study suggested it altered Fas/FasL expression and activated caspase-8 to induce apoptosis in THP-1 cells.100
Anti-Cervical Tumor Activity
Its anti-cancer qualities have been emphasised by studies, especially in preventing cervical cancer cells from growing and proliferating. In cervical cancer cell lines, the plant’s aqueous and ethanolic extracts have been shown to trigger apoptosis and inhibit cell cycle progression. The plant’s capacity to alter important biochemical pathways involved in cell survival, proliferation, and death is thought to be responsible for these effects. Furthermore, it has been observed that Hedyotisdiffusa increases the expression of tumor-suppressor proteins such as P53, which are essential for controlling malignant cell death.Its anti-inflammatory, antioxidant, and anti-proliferative activities further contribute to its therapeutic potential, making it a promising candidate for complementary or alternative treatment strategies in cervical cancer management.When given intragastrially to a human cervical carcinoma xenograft in naked mice, the aqueous extract of H. diffusa (0.5 g/kg bw) suppressed cervical cancer cells and caused HeLa cells to undergo apoptosis.101With an IC50 of 45 µM,90anthraquinones, in particular 2-hydroxymethyl-1-hydroxy anthraquinone, demonstrated strong suppression of HeLa cells in vitro.
Anti-Prostate Cancer Activity
Several cell lines have shown that H. diffusa’s active chemicals have anti-prostate cancer properties. DU145, PC-3, and LNCaP cells were inhibited by 2-methyl-3-methoxy anthraquinone (IC50 64.72–105.90 µM), 2-hydroxy-3-methyl anthraquinone (IC50 28.82–159.20 µM), and ursolic acid (IC50 22.33–36.08 µM). Furthermore, 10(S)-hydroxypheophytin and 10-O-(E)-p-coumaroylscandoside methyl ester had dose-dependent anti-proliferative effects on PC-3 cells (0–60 µM), whereas 10(S)-hydroxypheophytin A shown a strong anti-proliferative effect on LNCaP cells (IC50 20 µM).101 Three cyclotides (DC 1-3) were found to have anti-prostate cancer characteristics; in particular, DC 3 (1 mg/kg) inhibited PC3, DU145, and LNCaP cells.102Furthermore, in the prostate xenograft model, DC3 considerably reduced the tumor’s development in both weight and size. At a dose of 1 mg/kg, it also had a significant anti-cancer effect (p < 0.01), reducing the tumour growth rate (weight) by 40.23%.103
Immunomodulatory Effect
In leukemic mice with WEHI-3-generated leukaemia, the aqueous extract of H. diffusa (16 and 32 mg/kg) increased T- and B-cell proliferation, improving immune responses.104By raising CD11b, CD19, and Mac-3 levels, improving macrophage phagocytosis, and increasing NK cell activity and B- and T-cell proliferation, the ethanolic extract (16, 32, and 64 mg/kg) also enhanced immunological responses in normal BALB/c mice.105Furthermore, in immunosuppressed mice, polysaccharide extracts (2.25, 4.5, and 9.0 mg/mL) enhanced the phagocytic index, clearance index, and thymus and spleen indices.106The immunomodulatory effect of total flavonoids of H. diffusa was demonstrated by the increased proliferation of T and B lymphocytes and the enhancement of interleukin-2 (IL-2) and interferon-γ (INF-γ) levels in immunosuppressed mice given oral doses of 15, 30, and 60 mg/kg.107
Anti-Multiple Myeloma Activity
In RPMI 8226 cells, H. diffusa has been shown to have an anti-multiple myeloma impact. RPMI 8226 cell proliferation was dose-dependently reduced by injection of H. diffusa (20, 40, and 60 µL/mL) 109, 2-hydroxymethyl-1-hydroxy anthraquinone (1–200 µM) 90, and polysaccharide extracts (1, 2, and 3 mg/mL).108
Other Anti-Cancer Effects
In numerous research, Hedyotisdiffusa has demonstrated strong anti-cancer properties. B16F10 cell growth was dose-dependently reduced by the ethanolic extract (0–200 µg/mL).109,110Its crude polysaccharide (31.25 and 62.5 mg/kg) and lipophilic extract (50 and 100 mg/kg) had anti-tumor actions on S-180 cells and provided protection against damage caused by chemotherapy.111When H. diffusa was injected into MG-63 cells, the Bax gene was upregulated (50–400 µg/mL).112Its impact was amplified when paired with cisplatin, most likely due to the regulation of Bcl-xl, Bcl-2, Bax, Bad, caspase-3, and caspase-8.114By triggering mitochondrial death through the AKT/ERK pathways, the aqueous extract (2–8 mg/mL) suppressed the proliferation of U87 cells.113Furthermore, human endothelial cells showed anti-angiogenic activity in response to 4-vinyl phenol from H. diffusa (IC50 15.31 µg/mL), breast tumor-bearing mice, and zebrafish embryos, linked to the PI3K/AKT pathway.The efficacy of Hedyotisdiffusa in cancer therapy presents a compelling alternative or complement to conventional treatments such as chemotherapy,115 and radiation.116Unlike these standard therapies, which often cause severe side effects like toxicity, immune suppression, and diminished quality of life, Hedyotisdiffusa exhibits targeted anticancer activities with lower toxicity profiles. Its bioactive compounds selectively induce apoptosis, inhibit proliferation, and suppress metastasis in cancer cells while sparing healthy tissues. Additionally, Hedyotisdiffusa demonstrates immune-modulatory and anti-inflammatory properties, addressing the tumor microenvironment a limitation often unmet by conventional therapies.117While chemotherapy and radiation are effective in many cases, their efficacy is often diminished by drug resistance, a challenge that Hedyotisdiffusa mitigates through its multifaceted mechanisms. These advantages underscore its potential as a valuable addition to cancer treatment regimens, particularly in reducing side effects and enhancing therapeutic outcomes.
Synergistic Effects with Modern Cancer Therapies
Hedyotisdiffusa demonstrates significant potential for synergistic effects when combined with modern cancer therapies, enhancing efficacy and reducing side effects. For example, studies have shown that its bioactive compounds can amplify the cytotoxic effects of chemotherapy agents like cisplatin and doxorubicin while mitigating their toxicity to healthy tissues.118Similarly, by making cancer cells more sensitive to radiation-induced damage, Hedyotisdiffusa has been shown to enhance the results of radiation therapy, possibly through mechanisms like enhanced oxidative stress and apoptosis induction.119Additionally, when used alongside targeted therapies such as tyrosine kinase inhibitors, Hedyotisdiffusa may help overcome drug resistance by modulating pathways like NF-κB and STAT3. By lowering side effects associated with treatment, these synergistic interactions not only increase therapeutic efficacy but also enhance patients’ quality of life.120
Discussion
Traditional Chinese medicine is less harmful to healthy cells than chemotherapy drugs. By upgrading regulatory procedures and authorising a number of herbal extracts and Chinese herbal formulations for clinical trials, such as BZL101 and PHY906,121the US Food and Drug Administration (FDA) has promoted the use of herbal medicine in the treatment of cancer. Hedyotisdiffusa (HDW) is a popular Chinese herbal remedy; at the moment, it is a major component in 15% of the 1,700 herbal compounds used to treat cancer.122According to the theories of traditional Chinese medicine, qi and blood stagnation and the buildup of poisonous heat are the causes of tumours. According to studies, HDW and Scutellariabarbata are essential for heat-clearing and detoxifying in therapeutic settings. At 5.1%, HDW is the most commonly used herb among the top 10 Chinese herbal formulations prescribed for patients with post-surgical colon cancer. Through in vitro investigations, contemporary research on Hedyotisdiffusa (HDW) has produced strong evidence of its anticancer properties, which are consistent with its clinical uses in traditional Chinese medicine. Among the many bioactive anticancer chemicals found in HDW are cyclopeptides, anthraquinones, flavonoids, iridoids, terpenes, and steroids.123Although these substances are also present in other botanical sources, HDW’s anticancer effects are a result of their combined existence. Its methods of action include protective autophagy, anti-inflammatory and antioxidant actions, and the inhibition of angiogenesis and cancer cell proliferation while inducing apoptosis through host immune response modulation. The main signalling pathways impacted by HDW’s active ingredients include the Wnt/β-catenin, PI3K/Akt, Jak/STAT, MAPK, Bcl-2, p53, NF-κB, TNF, and Ras-Raf pathways. By focussing on several pathways implicated in tumour growth and survival, these molecular interactions demonstrate HDW’s promise in cancer treatment. The investigation of traditional Chinese medicine’s potential anticancer effects has drawn more attention in recent years. To clarify the underlying molecular pathways and pinpoint the exact targets that control the beginning, development, and metastasis of cancer, more research is necessary because human carcinogenesis is a complicated, multi-step process. HDW’s flavonoids, volatile oils, phenolic acids, and their derivatives are mainly responsible for its anticancer properties. Although iridoids have shown strong anticancer activity in vitro, it is still unknown how exactly they work. Furthermore, even though the anticancer molecular mechanisms of HDW’s ethanol and water extracts have been thoroughly studied, more research is necessary to identify the precise active ingredients causing these effects. Future research ought to focus on elucidating their exact modes of action and pinpointing particular molecular targets for certain cancer types. The majority of Hedyotisdiffusa (HDW) formulations on the market today are injectable or oral. In order to establish a glioma-targeting system (MC-DA7R-LS),DA7R, modified with MC (MC-DA7R), was immobilised onto PEGylated liposomes.124 This approach specifically targeted glioma cells and glioma stem cells (GSCs) and successfully penetrated the blood-brain barrier. Several studies suggest that combining multiple components or extracts from HDW is more effective than using a single compound, as it can modulate multiple antitumor signaling pathways simultaneously. Future research should focus on developing improved formulations, such as integrating HDW with different drug delivery systems to enhance targeted action on specific pathways and focus on exploring its synergistic potential with other Traditional Chinese Medicine (TCM) herbs to enhance its anticancer efficacy. Additionally, combining HDW with other anticancer drugs may help improve cancer cell sensitivity to chemotherapy, potentially enhancing treatment efficacy.Future research on Hedyotisdiffusa should Modern pharmacological approaches, such as nanotechnology and advanced drug delivery systems, could be employed to improve its bioavailability and therapeutic outcomes. Comprehensive clinical trials and rigorous safety assessments are essential to validate its efficacy and establish standardized protocols for its use in cancer treatment. Further investigations into the molecular pathways targeted by Hedyotisdiffusa will provide deeper insights into its mechanisms of action and enable the development of targeted therapies. Additionally, integrating Hedyotisdiffusa into personalized cancer treatment strategies, leveraging TCM principles alongside modern medicine, could revolutionize patient-specific therapeutic approaches.
In addition to its therapeutic effects, both short-term and long-term administration of Hedyotisdiffusa may present certain adverse effects. Short-term use has occasionally been associated with gastrointestinal discomfort and allergic reactions in sensitive individuals. Long-term consumption, particularly in high doses, may lead to liver or kidney function alterations, although comprehensive human data is limited. Furthermore, Hedyotisdiffusa may interact with conventional chemotherapeutic agents by modulating cytochrome P450 enzyme activity, potentially affecting drug metabolism and efficacy. These aspects highlight the need for cautious integration with standard cancer therapies and underscore the importance of further pharmacokinetic and toxicity studies 125.
Conclusion
Hedyotisdiffusa has demonstrated significant anticancer potential in Traditional Chinese Medicine (TCM), with its ability to inhibit tumor growth, induce apoptosis, suppress metastasis, and modulate the immune response. Its low toxicity and mechanisms of action position it as a promising complementary therapy to conventional cancer treatments. However, while preclinical studies highlight its efficacy, To confirm these results in clinical settings, more investigation is required.Comprehensive trials, safety assessments, and exploration of its integration into modern therapeutic protocols are essential to establish its role in mainstream oncology. Increasing our knowledge of Hedyotisdiffusa may lead to more individualised and comprehensive cancer treatment strategies.
Acknowledgement
The authors express their thanks to Saveetha College of Pharmacy – SIMATS for providing the necessary facilities to carry out this research work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict Of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author Contributions
- Binoy Varghese Cheriyan: Visualization, Supervision, Project Admininstration.
- Jubilee Ramasamy :Funding Acquisition, Resources.
- RithikhaSooriyaprakash :Data Collection, Analysis.
- Sam Jebaraj : Analysis.
- KanigaPandi: Writing – Original Draft.
References
- Bharadwaj N, Manimuthu MS, Vimal S, Radhakrishnan N. Evaluation of In vitroAnti-Cancer Activity of Methanolic Leaf Extract of Phoenix pusillaon Colon Cancer Cell Line. J Pharm Bioallied Sci. 2024;16(Suppl 2):S1181-S1185. doi:10.4103/jpbs.jpbs_522_23
CrossRef - Yadav N, Deshmukh R, Majumder R. A Comprehensive Review on the Use of Traditional Chinese Medicine for Cancer Treatment. Pharmacological Research-Modern Chinese Medicine. 2024; 2:100423.
CrossRef - Zhang S, Sun K, Zheng R, Zeng H, Wang S, Chen R, Wei W, He J. Cancer incidence and mortality in China, 2015. Journal of the National Cancer Center. 2021;1(1):2-11.
CrossRef - Vijayalakshmi TM, Murali R, Shinde MG, Singh H, Aparadh VM. Cancer Biology And Therapeutics: A Contemporary Review. Journal of Advanced Zoology. 2024;1;45(2).
- Ye L, Jia Y, Ji KE, et al. Traditional Chinese medicine in the prevention and treatment of cancer and cancer metastasis. Oncol Lett. 2015;10(3):1240-1250. doi:10.3892/ol.2015.3459
CrossRef - Jansen C, Baker JD, Kodaira E, et al. Medicine in motion: Opportunities, challenges and data analytics-based solutions for traditional medicine integration into western medical practice. J Ethnopharmacol. 2021;267:113477. doi:10.1016/j.jep.2020.113477
CrossRef - Mercadante S, Bellavia G, Cascio AL, et al. The use of complementary alternative medicines in advanced cancer patients followed at home. Support Care Cancer. 2022;30(3):2003-2008. doi:10.1007/s00520-021-06580-4
CrossRef - Martin JH, Patel J. Complementary and alternative therapies in the palliative setting. Intern Med J. 2022;52(10):1677-1684. doi:10.1111/imj.15922
CrossRef - Nestler G. Traditional Chinese medicine. Med Clin North Am. 2002;86(1):63-73. doi:10.1016/s0025-7125(03)00072-5
CrossRef - Pan MH, Chiou YS, Tsai ML, Ho CT. Anti-inflammatory activity of traditional Chinese medicinal herbs. J Tradit Complement Med. 2011;1(1):8-24. doi:10.1016/s2225-4110(16)30052-9
CrossRef - Chan K. Progress in traditional Chinese medicine. Trends Pharmacol Sci. 1995;16(6):182-187. doi:10.1016/s0165-6147(00)89019-7
CrossRef - Tao, C.; Taylor, C.M. Rubiaceae. In Flora of China; Wu, Z.Y., Raven, P.H., Hong, D.Y., Eds.; Science Press: Beijing, China; Missouri Botanical Garden Press: St. Louis, MO, USA, 2011;19:147–174.
- Hu CJ, He J, Li GZ, et al. Analyzing hedyotisdiffusa mechanisms of action from the genomics perspective. Comput Methods Programs Biomed. 2019;174:1-8. doi:10.1016/j.cmpb.2018.10.019
CrossRef - Ou L, Li M, Hou Y. Network pharmacology, bioinformatics, and experimental validation to identify the role of Hedyotisdiffusawilld against gastric cancer through the activation of the endoplasmic reticulum stress. Heliyon. 2024;10(7):e28833. Published 2024 Mar 30. doi:10.1016/j.heliyon.2024.e28833
CrossRef - Zhang XG. Isolation and Characterisation of the Anti-Tumour Activity of Some Chinese Plants Used in Cancer Treatment. The University of Manchester (United Kingdom); 1995.
- Wang S, Yin N, Li Y, et al. Molecular mechanism of the treatment of lung adenocarcinoma by HedyotisDiffusa: an integrative study with real-world clinical data and experimental validation. Front Pharmacol. 2024;15:1355531. Published 2024 Jun 6. doi:10.3389/fphar.2024.1355531
CrossRef - Shao J, Gong G, Trombetta L. An evidence-based perspective of Hedyotisdiffusa or Oldenlandiadiffusa (spreading Hedyotis) for cancer patients. Evidence-based Anticancer MateriaMedica. 2011:179-92.
CrossRef - Xu L, Li Y, Ji J, et al. The anti-inflammatory effects of HedyotisdiffusaWilld on SLE with STAT3 as a key target. J Ethnopharmacol. 2022;298:115597. doi:10.1016/j.jep.2022.115597
CrossRef - Efferth T, Li PC, Konkimalla VS, Kaina B. From traditional Chinese medicine to rational cancer therapy. Trends Mol Med. 2007;13(8):353-361. doi:10.1016/j.molmed.2007.07.001
CrossRef - Lin J, Li Q, Chen H, Lin H, Lai Z, Peng J. HedyotisdiffusaWilld. extract suppresses proliferation and induces apoptosis via IL-6-inducible STAT3 pathway inactivation in human colorectal cancer cells. Oncol Lett. 2015;9(4):1962-1970. doi:10.3892/ol.2015.2956
CrossRef - Zhang P, Zhang B, Gu J, Hao L, Hu F, Han C. The Study of the Effect of Hedyotisdiffusa on the Proliferation and the Apoptosis of the Cervical Tumor in Nude Mouse Model. Cell BiochemBiophys. 2015;72(3):783-789. doi:10.1007/s12013-015-0532-9
CrossRef - Zhang Y, Xie RF, Xiao QG, Li R, Shen XL, Zhu XG. HedyotisdiffusaWilld extract inhibits the growth of human glioblastoma cells by inducing mitochondrial apoptosis via AKT/ERK pathways. J Ethnopharmacol. 2014;158 Pt A:404-411. doi:10.1016/j.jep.2014.10.017
CrossRef - Meng QX, Roubin RH, Hanrahan JR. Ethnopharmacological and bioactivity guided investigation of five TCM anticancer herbs. J Ethnopharmacol. 2013;148(1):229-238. doi:10.1016/j.jep.2013.04.014
CrossRef - Chao TH, Fu PK, Chang CH, et al. Prescription patterns of Chinese herbal products for post-surgery colon cancer patients in Taiwan. J Ethnopharmacol. 2014;155(1):702-708. doi:10.1016/j.jep.2014.06.012
CrossRef - Yeh YC, Chen HY, Yang SH, et al. Hedyotisdiffusa Combined with Scutellariabarbata Are the Core Treatment of Chinese Herbal Medicine Used for Breast Cancer Patients: A Population-Based Study. Evid Based Complement Alternat Med. 2014;2014:202378. doi:10.1155/2014/202378
CrossRef - Lin J, Wei L, Xu W, Hong Z, Liu X, Peng J. Effect of HedyotisDiffusaWilld extract on tumor angiogenesis. Mol Med Rep. 2011;4(6):1283-1288. doi:10.3892/mmr.2011.577
CrossRef - Li M, Wong YL, Jiang LL, et al. Application of novel loop-mediated isothermal amplification (LAMP) for rapid authentication of the herbal tea ingredient HedyotisdiffusaWilld. Food Chem. 2013;141(3):2522-2525. doi:10.1016/j.foodchem.2013.05.085
CrossRef - Lau CB, Cheng L, Cheng BW, et al. Development of a simple chromatographic method for distinguishing between two easily confused species, Hedyotisdiffusa and Hedyotiscorymbosa. Nat Prod Res. 2012;26(15):1446-1450. doi:10.1080/14786419.2011.603317
CrossRef - Wang L, Zhou C, Mai HZ. Analysis of volatile compounds in Hedyotisdiffusa and Hedyotiscorymbosa. J. Chin. Mater. Med. 2003;26:563-4.
- Wang, L., C. Zhou, and H. Z. Mai. “Analysis of volatile compounds in Hedyotisdiffusa and Hedyotiscorymbosa.” J. Chin. Mater. Med26.2003; 563-564.
- Liu Z, Hao M, Wang J. Zhong Yao Cai. 2004;27(7):484-487.
- Liu EH, Zhou T, Li GB, et al. Characterization and identification of iridoid glucosides, flavonoids and anthraquinones in Hedyotisdiffusa by high-performance liquid chromatography/electrospray ionization tandem mass spectrometry. J Sep Sci. 2012;35(2):263-272. doi:10.1002/jssc.201100780
CrossRef - Si JY, Chen DH, Pan RL. Chemical constituents of Hedyotisdiffusa. Natural Product Research and Development. 2006;18(6):942.
- Li C, Zhao Y, Guo Z, Zhang X, Xue X, Liang X. Effective 2D-RPLC/RPLC enrichment and separation of micro-components from HedyotisdiffusaWilld. and characterization by using ultra-performance liquid chromatography/quadrupole time-of-flight mass spectrometry. J Pharm Biomed Anal. 2014;99:35-44. doi:10.1016/j.jpba.2014.06.020
CrossRef - Kang XD, Li X, Mao Y, Zhao CC, Li N, Meng DL. Chemical constituents of HedyotisdiffusaWilld. J. Shenyang Pharm. Univ. 2007;24:479-81.
- Liang SY, Chen FL, Tang QF, Luo JB, Zeng YC. Study of Chemical Constituents from HerbaHedyotisdiffusa. Tradit. Chin. Drug Res. Clin. Pharm. 2012;23:655-7.
- Zhang YY, Luo JB. Zhong Yao Cai. 2008;31(4):522-524.
- Huang W, Li Y, Jiang J. ZhongguoZhong Yao ZaZhi. 2009;34(6):712-714.
- Wang L, Zhou C, Mai HZ. Analysis of volatile compounds in Hedyotisdiffusa and Hedyotiscorymbosa. J. Chin. Mater. Med. 2003;26:563-4.
- Ma H, Cheng YL, Zhang JJ, Cao GS, Yang PM. Effect of preliminary immune activity and structural identification of a polysaccharide extracted from Oldenlandiadiffusa. Chin. J. Exp. Tradit. Med. Form. 2014;20:37-40.
CrossRef - Hu E, Wang D, Chen J, Tao X. Novel cyclotides from Hedyotisdiffusa induce apoptosis and inhibit proliferation and migration of prostate cancer cells. Int J ClinExp Med. 2015;8(3):4059-4065. Published 2015 Mar 15.
- Ninghua T, Shuangming W, Yabin Y, Fang T. Anticancer activity and principles of Hedyotisdiffusa. Natural Product Research and Development. 2002;1;14(5):33-6.
- Cheng CS, Chen J, Tan HY, Wang N, Chen Z, Feng Y. Scutellariabaicalensis and Cancer Treatment: Recent Progress and Perspectives in Biomedical and Clinical Studies. Am J Chin Med. 2018;46(1):25-54. doi:10.1142/S0192415X18500027
CrossRef - Sultana S, Munir N, Mahmood Z, et al. Molecular targets for the management of cancer using Curcuma longa Linn. phytoconstituents: A Review. Biomed Pharmacother. 2021;135:111078. doi:10.1016/j.biopha.2020.111078
CrossRef - Lin M, Lin J, Wei L, et al. HedyotisdiffusaWilld extract inhibits HT-29 cell proliferation via cell cycle arrest. ExpTher Med. 2012;4(2):307-310. doi:10.3892/etm.2012.599
CrossRef - Yan Z, Feng J, Peng J, et al. Chloroform extract of HedyotisdiffusaWilld inhibits viability of human colorectal cancer cells via suppression of AKT and ERK signaling pathways. Oncol Lett. 2017;14(6):7923-7930. doi:10.3892/ol.2017.7245
CrossRef - Li S, Kuo HD, Yin R, et al. Epigenetics/epigenomics of triterpenoids in cancer prevention and in health. BiochemPharmacol. 2020;175:113890. doi:10.1016/j.bcp.2020.113890
CrossRef - Cai Q, Lin J, Wei L, et al. HedyotisdiffusaWilld inhibits colorectal cancer growth in vivo via inhibition of STAT3 signaling pathway. Int J Mol Sci. 2012;13(5):6117-6128. doi:10.3390/ijms13056117
CrossRef - Feng J, Jin Y, Peng J, et al. Hedyotisdiffusawilldextract suppresses colorectal cancer growth through multiple cellular pathways. Oncol Lett. 2017;14(6):8197-8205. doi:10.3892/ol.2017.7244
CrossRef - Lin J, Li Q, Chen H, Lin H, Lai Z, Peng J. HedyotisdiffusaWilld. extract suppresses proliferation and induces apoptosis via IL-6-inducible STAT3 pathway inactivation in human colorectal cancer cells. Oncol Lett. 2015;9(4):1962-1970. doi:10.3892/ol.2015.2956
CrossRef - Lin J, Wei L, Shen A, et al. HedyotisdiffusaWilld extract suppresses Sonic hedgehog signaling leading to the inhibition of colorectal cancer angiogenesis. Int J Oncol. 2013;42(2):651-656. doi:10.3892/ijo.2012.1753
CrossRef - Lin J, Chen Y, Wei L, et al. HedyotisDiffusaWilld extract induces apoptosis via activation of the mitochondrion-dependent pathway in human colon carcinoma cells. Int J Oncol. 2010;37(5):1331-1338. doi:10.3892/ijo_00000785
CrossRef - Goldstein JC, Waterhouse NJ, Juin P, Evan GI, Green DR. The coordinate release of cytochrome c during apoptosis is rapid, complete and kinetically invariant. Nat Cell Biol. 2000;2(3):156-162. doi:10.1038/35004029
CrossRef - Li YL, Zhang J, Min D, Hongyan Z, Lin N, Li QS. Anticancer Effects of 1,3-Dihydroxy-2-Methylanthraquinone and the Ethyl Acetate Fraction of HedyotisDiffusaWilld against HepG2 Carcinoma Cells Mediated via Apoptosis. PLoS One. 2016;11(4):e0151502. Published 2016 Apr 11. doi:10.1371/journal.pone.0151502
CrossRef - Lamouille S, Xu J, Derynck R. Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol. 2014;15(3):178-196. doi:10.1038/nrm3758
CrossRef - Beavon IR. The E-cadherin-catenin complex in tumour metastasis: structure, function and regulation. Eur J Cancer. 2000;36(13 Spec No):1607-1620. doi:10.1016/s0959-8049(00)00158-1
CrossRef - Duan L, Yang Z, Jiang X, Zhang J, Guo X. Oleanolic acid inhibits cell proliferation migration and invasion and induces SW579 thyroid cancer cell line apoptosis by targeting forkhead transcription factor A. Anticancer Drugs. 2019;30(8):812-820. doi:10.1097/CAD.0000000000000777
CrossRef - Lin M, Lin J, Wei L, et al. HedyotisdiffusaWilld extract inhibits HT-29 cell proliferation via cell cycle arrest. ExpTher Med. 2012;4(2):307-310. doi:10.3892/etm.2012.599
CrossRef - Sun G, Wei L, Feng J, Lin J, Peng J. Inhibitory effects of HedyotisdiffusaWilld. on colorectal cancer stem cells. Oncol Lett. 2016;11(6):3875-3881. doi:10.3892/ol.2016.4431
CrossRef - Chen, Wujin, et al. “HedyotisdiffusaWilld reduces migration and invasion through inhibition of TGF-β-induced EMT in colorectal cancer cells.” European Journal of Integrative Medicine23.2018; 57-63.doi:10.1016/j.eujim. 2018.09.008.
CrossRef - Liu X, Wu J, Zhang D, Wang K, Duan X, Zhang X. A Network Pharmacology Approach to Uncover the Multiple Mechanisms of HedyotisdiffusaWilld. on Colorectal Cancer. Evid Based Complement Alternat Med. 2018;2018:6517034. Published 2018 Feb 12. doi:10.1155/2018/6517034
CrossRef - Li YL, Zhang J, Min D, Hongyan Z, Lin N, Li QS. Anticancer Effects of 1,3-Dihydroxy-2-Methylanthraquinone and the Ethyl Acetate Fraction of HedyotisDiffusaWilld against HepG2 Carcinoma Cells Mediated via Apoptosis. PLoS One. 2016;11(4):e0151502. Published 2016 Apr 11. doi:10.1371/journal.pone.0151502
CrossRef - Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002;420(6917):860-867. doi:10.1038/nature01322
CrossRef - Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008;454(7203):436-444. doi:10.1038/nature07205
CrossRef - Shacter E, Weitzman SA. Chronic inflammation and cancer. Oncology (Williston Park). 2002;16(2):217-232.
- Rajagopal C, Lankadasari MB, Aranjani JM, Harikumar KB. Targeting oncogenic transcription factors by polyphenols: A novel approach for cancer therapy. Pharmacol Res. 2018;130:273-291. doi:10.1016/j.phrs.2017.12.034
CrossRef - Xu Y, Chen XX, Jiang YX, Zhang DD. Ethyl Acetate Fraction from Hedyotisdiffusaplus ScutellariabarbataExerts Anti-Inflammatory Effects by Regulating miR-155 Expression and JNK Signaling Pathway. Evid Based Complement Alternat Med. 2018;2018:3593408. Published 2018 Mar 14. doi:10.1155/2018/3593408
CrossRef - Sun C, Yang J, Cheng HB, et al. 2-Hydroxy-3-methylanthraquinone inhibits lung carcinoma cells through modulation of IL-6-induced JAK2/STAT3 pathway. Phytomedicine. 2019;61:152848. doi:10.1016/j.phymed.2019.152848
CrossRef - Peng D, Chen L, Sun Y, et al. Melanoma suppression by quercein is correlated with RIG-I and type I interferon signaling. Biomed Pharmacother. 2020;125:109984. doi:10.1016/j.biopha.2020.109984
CrossRef - Ma C, Wei Y, Liu Q, et al. Polysaccharides from Hedyotisdiffusa enhance the antitumor activities of cytokine-induced killer cells. Biomed Pharmacother. 2019;117:109167. doi:10.1016/j.biopha.2019.109167
CrossRef - Ferrara N, Kerbel RS. Angiogenesis as a therapeutic target. Nature. 2005;438(7070):967-974. doi:10.1038/nature04483
CrossRef - Su X, Li Y, Jiang M, et al. Systems pharmacology uncover the mechanism of anti-non-small cell lung cancer for HedyotisdiffusaWilld. Biomed Pharmacother. 2019;109:969-984. doi:10.1016/j.biopha.2018.10.162
CrossRef - Yue GG, Lee JK, Kwok HF, et al. Novel PI3K/AKT targeting anti-angiogenic activities of 4-vinylphenol, a new therapeutic potential of a well-known styrene metabolite. Sci Rep. 2015;5:11149. Published 2015 Jun 8. doi:10.1038/srep11149
CrossRef - Mahaveerakannan R, Choudhary SL, Dixit RS, Mylapalli S, Kumar MS. Enhancing Diagnostic Accuracy and Early Detection Through the Application of Deep Learning Techniques to the Segmentation of Colon Cancer in Histopathological Images. In2024 8th International Conference on I-SMAC (IoT in Social, Mobile, Analytics and Cloud)(I-SMAC).2024;3:1809-1815. IEEE.
CrossRef - Lin J, Wei L, Shen A, et al. HedyotisdiffusaWilld extract suppresses Sonic hedgehog signaling leading to the inhibition of colorectal cancer angiogenesis. Int J Oncol. 2013;42(2):651-656. doi:10.3892/ijo.2012.1753
CrossRef - Cai Q, Lin J, Wei L, et al. HedyotisdiffusaWilld inhibits colorectal cancer growth in vivo via inhibition of STAT3 signaling pathway. Int J Mol Sci. 2012;13(5):6117-6128. doi:10.3390/ijms13056117
CrossRef - WU Z, Jin C, LI J, Chen X, Yao Q, Zhu Q. Inhibition of colon cancer cells by ethanol extract of Oldenlandiadiffusa. Journal of Kunming Medical University. 2013:31-4.
- Li Q, Wang X, Shen A, et al. HedyotisdiffusaWilld overcomes 5-fluorouracil resistance in human colorectal cancer HCT-8/5-FU cells by downregulating the expression of P-glycoprotein and ATP-binding casette subfamily G member 2. ExpTher Med. 2015;10(5):1845-1850. doi:10.3892/etm.2015.2762
CrossRef - Ganbold M, Barker J, Ma R, Jones L, Carew M. Cytotoxicity and bioavailability studies on a decoction of Oldenlandiadiffusa and its fractions separated by HPLC. J Ethnopharmacol. 2010;131(2):396-403. doi:10.1016/j.jep.2010.07.014
CrossRef - Cai Q, Lin J, Wei L, et al. HedyotisdiffusaWilld inhibits colorectal cancer growth in vivo via inhibition of STAT3 signaling pathway. Int J Mol Sci. 2012;13(5):6117-6128. doi:10.3390/ijms13056117
CrossRef - Vishnupriya N, Jayaraman S, Veeraraghavan VP. Camptothecin Anti-cancer Activity Against Breast Cancer Cells (MDA MB-231) Targeting the Gene Expression of Wnt/Beta-catenin Pathway-An In silico and In vitro Approach.
- Nautiyal M, Ganapathy D, Ameya KP, Sekar D. Analysis of Carboplatin and STAT3 in the Breast Cancer MCF7 Cell Line. Texila International Journal of Public Health. 2024;12(2).
CrossRef - Liu Z, Liu M, Liu M, Li J. Methylanthraquinone from Hedyotisdiffusa WILLD induces Ca(2+)-mediated apoptosis in human breast cancer cells. ToxicolIn Vitro. 2010;24(1):142-147. doi:10.1016/j.tiv.2009.08.002
CrossRef - Shi Y, Wang CH, Gong XG. Apoptosis-inducing effects of two anthraquinones from Hedyotisdiffusa WILLD. Biol Pharm Bull. 2008;31(6):1075-1078. doi:10.1248/bpb.31.1075
CrossRef - Reddy, MB Sai Sreekar, et al. “Effect of Diosmin on The Expression of Epithelial-Mesenchymal Transition Signaling Molecules in Ndea-Induced Hepato-Cellular Carcinoma in Experimental Rats.”. 10.21522/TIJPH.2013.SE.23.01.Art008.
- Chen R, He J, Tong X, Tang L, Liu M. The HedyotisdiffusaWilld. (Rubiaceae): A Review on Phytochemistry, Pharmacology, Quality Control and Pharmacokinetics. Molecules. 2016;21(6):710. doi:10.3390/molecules21060710
CrossRef - Chen XZ, Cao ZY, Chen TS, et al. Water extract of HedyotisDiffusaWilld suppresses proliferation of human HepG2 cells and potentiates the anticancer efficacy of low-dose 5-fluorouracil by inhibiting the CDK2-E2F1 pathway. Oncol Rep. 2012;28(2):742-748. doi:10.3892/or.2012.1834
CrossRef - Zhang Y, Zhu J, Xiao J, Guo Y, Liao Z, XU R. Effect and mechanism of total flavones of Oldenlendiadiffusawilld on epithelial-mesenchymal transition of cell line MHCC97-H induced by TGF-β1. Journal of Xi’an Jiaotong University (Medical Sciences). 2016:279-82.
- Li YL, Zhang J, Min D, Hongyan Z, Lin N, Li QS. Anticancer Effects of 1,3-Dihydroxy-2-Methylanthraquinone and the Ethyl Acetate Fraction of HedyotisDiffusaWilld against HepG2 Carcinoma Cells Mediated via Apoptosis. PLoS One. 2016;11(4):e0151502. Published 2016 Apr 11. doi:10.1371/journal.pone.0151502
CrossRef - Meng QX, Roubin RH, Hanrahan JR. Ethnopharmacological and bioactivity guided investigation of five TCM anticancer herbs. J Ethnopharmacol. 2013;148(1):229-238. doi:10.1016/j.jep.2013.04.014
CrossRef - Shi Y, Wang CH, Gong XG. Apoptosis-inducing effects of two anthraquinones from Hedyotisdiffusa WILLD. Biol Pharm Bull. 2008;31(6):1075-1078. doi:10.1248/bpb.31.1075.
CrossRef - Divya D, Saveetha D, Bharathi VC, Shanmugam P. Transforming Lung Carcinoma Analysis: Deep Learning‘s Efficient Segmentation and Classification. In2023 International Conference on Innovative Computing, Intelligent Communication and Smart Electrical Systems (ICSES) 2023;14:1-8). IEEE.
CrossRef - Kuo YJ, Yang JS, Lu CC, Chiang SY, Lin JG, Chung JG. Ethanol extract of Hedyotisdiffusawilld upregulates G0/G1 phase arrest and induces apoptosis in human leukemia cells by modulating caspase cascade signaling and altering associated genes expression was assayed by cDNA microarray. Environ Toxicol. 2015;30(10):1162-1177. doi:10.1002/tox.21989
CrossRef - Shi Y, Wang CH, Gong XG. Apoptosis-inducing effects of two anthraquinones from Hedyotisdiffusa WILLD. Biol Pharm Bull. 2008;31(6):1075-1078. doi:10.1248/bpb.31.1075
CrossRef - Huang, Fuhao, et al. “Hedyotisdiffusa injection induces ferroptosis via the Bax/Bcl2/VDAC2/3 axis in lung adenocarcinoma.” Phytomedicine104.2022: 154319.. doi:10.1016/j.phymed.2022.154319.
CrossRef - Zhu DC, Pan RB, Wang Q. Research on the mechanisms of inhibiting effects of the aqueous extract of HedyotisdiffusaWilld on CEM cells. Lishizhen Med. Mater. Med. Res. 2014;25:827-9.
- Lin CC, Kuo CL, Lee MH, et al. Extract of HedyotisdiffusaWilld influences murine leukemia WEHI-3 cells in vivo as well as promoting T- and B-cell proliferation in leukemic mice. In Vivo. 2011;25(4):633-640.
- Chen XH, Gao RL, Qian XD, et al. Zhongguo Shi Yan Xue Ye XueZaZhi. 2008;16(5):1035-1038.
- Wang N, Li DY, Niu HY, Zhang Y, He P, Wang JH. 2-Hydroxy-3-methylanthraquinone from HedyotisdiffusaWilld induces apoptosis in human leukemic U937 cells through modulation of MAPK pathways. Archives of pharmacal research.2013;36:752-8.
CrossRef - Wang JH, Shu LH, Yang LL, Zhang M, He P. 2-Hydroxy-3-methylanthraquinone from Hedyotisdiffusa WILLD induces apoptosis via alteration of Fas/FasL and activation of caspase-8 in human leukemic THP-1 cells. Arch Med Res. 2011;42(7):577-583. doi:10.1016/j.arcmed.2011.11.003
CrossRef - Zhang P, Zhang B, Gu J, Hao L, Hu F, Han C. The Study of the Effect of Hedyotisdiffusa on the Proliferation and the Apoptosis of the Cervical Tumor in Nude Mouse Model. Cell BiochemBiophys. 2015;72(3):783-789. doi:10.1007/s12013-015-0532-9
CrossRef - Hu E, Wang D, Chen J, Tao X. Novel cyclotides from Hedyotisdiffusa induce apoptosis and inhibit proliferation and migration of prostate cancer cells. Int J ClinExp Med. 2015;8(3):4059-4065. Published 2015 Mar 15.
- Li M, Jiang RW, Hon PM, et al. Authentication of the anti-tumor herb Baihuasheshecao with bioactive marker compounds and molecular sequences. Food Chem. 2010;119(3):1239-1245. doi:10.1016/j.foodchem.2009.09.013
CrossRef - Lin CC, Kuo CL, Lee MH, et al. Extract of HedyotisdiffusaWilld influences murine leukemia WEHI-3 cells in vivo as well as promoting T- and B-cell proliferation in leukemic mice. In Vivo. 2011;25(4):633-640.
- Kuo YJ, Lin JP, Hsiao YT, et al. Ethanol Extract of HedyotisdiffusaWilld Affects Immune Responses in Normal Balb/c Mice In Vivo. In Vivo. 2015;29(4):453-460.
- Ma H, Cheng YL, Zhang JJ, Cao GS, Yang PM. Effect of preliminary immune activity and structural identification of a polysaccharide extracted from Oldenlandiadiffusa. Chin. J. Exp. Tradit. Med. Form. 2014;20:37-40.
CrossRef - Wang Y, Zhang Y, Fang M, LI Q, Jiang Q, Ming L. Immunomdulatory effects of total flavones of Oldenlandiadiffusawilld. Chinese Pharmacological Bulletin. 1986.
- Lin SY, Shen CY, Jiang JP, et al. ZhonghuaXue Ye XueZaZhi. 2013;34(4):337-340. doi:10.3760/cma.j.issn.0253-2727.2013.04.019
- Zhang X, Ye BD, Lin SY. ZhongguoZhong Xi Yi Jie He ZaZhi. 2012;32(12):1658-1662.
- Kuo YJ, Yang JS, Lu CC, Chiang SY, Lin JG, Chung JG. Ethanol extract of Hedyotisdiffusawilld upregulates G0/G1 phase arrest and induces apoptosis in human leukemia cells by modulating caspase cascade signaling and altering associated genes expression was assayed by cDNA microarray. Environ Toxicol. 2015;30(10):1162-1177. doi:10.1002/tox.21989
CrossRef - Zhao HR, Li R, Lin YN, Cheng NL. Influence of extraction process of Hedyotisdiffusa on anti-tumor activity. JOURNAL-CHINA PHARMACEUTICAL UNIVERSITY. 2002;33(6):510-3.
- Wang J, Xie K, Huang K. Effect of Hedyoticdiffusawilld injection on osteosarcoma MG-63 cells bax gene expression. Chongqing Medicine. 2014:4708-10.
- Zhang Y, Xie RF, Xiao QG, Li R, Shen XL, Zhu XG. HedyotisdiffusaWilld extract inhibits the growth of human glioblastoma cells by inducing mitochondrial apoptosis via AKT/ERK pathways. J Ethnopharmacol. 2014;158 Pt A:404-411. doi:10.1016/j.jep.2014.10.017
CrossRef - Pu F, Chen F, Lin S, et al. The synergistic anticancer effect of cisplatin combined with Oldenlandiadiffusa in osteosarcoma MG-63 cell line in vitro. Onco Targets Ther. 2016;9:255-263. Published 2016 Jan 11. doi:10.2147/OTT.S90707
CrossRef - Partridge AH, Burstein HJ, Winer EP. Side effects of chemotherapy and combined chemohormonal therapy in women with early-stage breast cancer [published correction appears in J Natl Cancer Inst 2002 Jun 5;94(11):866] [published correction appears in J Natl Cancer Inst Monogr.2003;(31):131]. J Natl Cancer InstMonogr. 2001;(30):135-142. doi:10.1093/oxfordjournals.jncimonographs.a003451
CrossRef - al-Mefty O, Kersh JE, Routh A, Smith RR. The long-term side effects of radiation therapy for benign brain tumors in adults. J Neurosurg. 1990;73(4):502-512. doi:10.3171/jns.1990.73.4.0502
CrossRef - Hung HY, Cheng KC, Kuo PC, et al. Chemical Constituents of Hedyotisdiffusaand Their Anti-Inflammatory Bioactivities. Antioxidants (Basel). 2022;11(2):335. Published 2022 Feb 9. doi:10.3390/antiox11020335
CrossRef - Ye C, Zhang B, Tang Z, Zheng C, Wang Q, Tong X. Synergistic action of HedyotisdiffusaWilld and Andrographispaniculata in Nasopharyngeal Carcinoma: Downregulating AKT1 and upregulating VEGFA to curb tumorigenesis. IntImmunopharmacol. 2024;132:111866. doi:10.1016/j.intimp.2024.111866
CrossRef - Niu Y, Meng QX. Chemical and preclinical studies on Hedyotisdiffusa with anticancer potential. J Asian Nat Prod Res. 2013;15(5):550-565. doi:10.1080/10286020.2013.781589
CrossRef - Tamanna S, Perumal E, Rajanathadurai J. Enhanced Apoptotic Effects in MDA-MB-231 Triple-Negative Breast Cancer Cells Through a Synergistic Action of Luteolin and Paclitaxel. Cureus. 2024;16(7):e65159. Published 2024 Jul 22. doi:10.7759/cureus.65159
CrossRef - Ling B, Michel D, Sakharkar MK, Yang J. Evaluating the cytotoxic effects of the water extracts of four anticancer herbs against human malignant melanoma cells. Drug Des DevelTher. 2016;10:3563-3572. Published 2016 Nov 1. doi:10.2147/DDDT.S119214
CrossRef - Li D, Schmitz OJ. Comprehensive two-dimensional liquid chromatography tandem diode array detector (DAD) and accurate mass QTOF-MS for the analysis of flavonoids and iridoid glycosides in Hedyotisdiffusa. Anal Bioanal Chem. 2015;407(1):231-240. doi:10.1007/s00216-014-8057-4
CrossRef - Chao TH, Fu PK, Chang CH, et al. Prescription patterns of Chinese herbal products for post-surgery colon cancer patients in Taiwan. J Ethnopharmacol. 2014;155(1):702-708. doi:10.1016/j.jep.2014.06.012
CrossRef - Ying M, Wang S, Zhang M, et al. Myristic Acid-Modified DA7R Peptide for Whole-Process Glioma-Targeted Drug Delivery. ACS Appl Mater Interfaces. 2018;10(23):19473-19482. doi:10.1021/acsami.8b05235.
CrossRef - Chen DH, Mao PJ, Diao WJ, Li Q. Toxicity study of compound granules of Hedyotisdiffusa: Acute toxicity and long-term toxicity. Journal of Ethnopharmacology. 2024;321:117434.
CrossRef
Abbreviations
TCM: Traditional Chinese Medicine
EEHDW: Ethanol Extract of Hedyotisdiffusa Wild
VEGF: Vascular Endothelial Growth Factor
STAT3: Signal Transducer and Activator of Transcription 3
NF-κB: Nuclear Factor Kappa B
PI3K/AKT: Phosphoinositide 3-Kinase/Protein Kinase B
EMT: Epithelial-Mesenchymal Transition
MDR: Multiple Drug Resistance
HDW: HedyotisDiffusa Wild







