Manuscript accepted on :10-06-2025
Published online on: 19-06-2025
Plagiarism Check: Yes
Reviewed by: Dr. Mehdihasan Ijubhai
Second Review by: Dr. Nikesh Narang
Final Approval by: Dr. Anton R Keslav
Department of Mechanical Engineering, College of Engineering, Prince Mohammad Bin Fahd University, Al-Khobar, Saudi Arabia
Corresponding Author E-mail: sslingala@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3153
Abstract
The potential of biodegradable materials and their alloys for use in biomedical applications has drawn a lot of attention. The most common biodegradable materials are graphene oxide (GO), iron (Fe), magnesium (Mo), and zinc (Zn), which has led to a great deal of research into the creation of new alloys. These metals are appropriate for bio-implant use due to their excellent mechanical integrity, adequate biocompatibility, and inherent biodegradability. The recent developments in biodegradable implants in the biomedical field in this thorough study, concentrating mostly on orthopedic applications were examined. The mechanical characteristics, corrosion mechanism, and degrading behavior of these materials both in-vitro and in-vivo are thoroughly examined in this work. The surface modification and fabrication methods, which are essential for maximizing implant performance are also explained. However, their extensive use has been hampered by specific constraints related to alloying materials, particularly iron and zinc. Additionally, the potential highlight of novel metals with exceptional qualities, like molybdenum, which could lead to implants with better mechanical qualities. All things considered, this study highlights the growing importance of biodegradable metallic implants and demonstrates how they can be used to meet a variety of clinical objectives. By overcoming material constraints and utilizing cutting-edge metals, the door is opened to create the implants of exceptional mechanical qualities, favourable rates of degrade, transforming the biomedical application space.
Keywords
Biodegradable; Bone regeneration; Bone healing; Graphene oxide; Metal alloys
Download this article as:| Copy the following to cite this article: Sundar L. S. In-vitro and In-vivo Responses of Functional Biodegradable Materials for Bone Implant Applications: A Comprehensive Review. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Sundar L. S. In-vitro and In-vivo Responses of Functional Biodegradable Materials for Bone Implant Applications: A Comprehensive Review. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/4e8wSkZ |
Introduction
In orthopedics, consideration of degradable materials was expanding rapidly, anticipated make considerable studies earlier to come benefits for implants are the main factors contributing to their popularity.1 Advancements in biodegradable implants created avenues for medical application that were previously unattainable, such as promoting bio response, ability of bio-materials, in response to particular issues like, pH variations, and temperatures. Implants made of synthetics improves living tissue function are referred to as biomaterials.2 Mechanical properties, biocompatibility, molecular weight, biodegradability, and physiochemical properties are the criteria used to choose biomaterials for medical applications.3
Based on their biological and cellular reactions, they are categorized as autonomous, responsive, active, or inactive.4 Surgical operations, such as dental implants, and bone settings are on the rise due to the world’s population growth.5 For overall, the necessity for medical implants has increased. They are employed in surgical procedures such as joint replacement, artificial tendons, fracture fixation plates, bone cement, and fillers for bone defects. The most widely utilized implant materials in orthopedic procedures at the moment are titanium, cobalt-chromium, and stainless steel.6 They promote healing because of their exceptional strength and resistance to corrosion.7
These materials are not biodegradable, though, which has a number of negative consequences, including the need for repeated procedures to remove the implants, growth limitations, sensitivity to temperature changes, and the possibility of cross-contamination. Although implants have side effects, they are necessary for medical procedures such bone replacement, dental surgery, and bone-fixing.8 Synthetic biodegradable polymer-based implants, which reduce the need for repeated surgeries and speed up healing, are the product of advances in medical research. Innovative biomaterials that are used by medical practitioners to enhance the quality of current therapies and life have advanced significantly during the past 10 years.9 Recent advancements in tissue engineering, regenerative medicine, and targeted/sustained drug administration have promoted the use of novel biomaterials with biodegradation properties for cutting-edge clinical applications.10
Currently, polymeric and metallic components make up bioresorbable materials. Bile duct stent applications include polymeric materials such as polydioxanone and poly (L-lactic acid).11 A range of biodegradable polymers with different biodegradation characteristics have been used to create biliary stents. The required properties (mechanical integrity, high tensile strength, non-toxicity, and rate of degradation regulation) are used to choose biodegradable polymers. Furthermore, their biocompatibility is greatly influenced by their chemical characteristics. Bioimplants should ideally be biocompatible with the human body, allowing for prolonged physiological immersion.
The functional graphene oxide (GO), magnesium (Mg), zinc (Zn), and iron (Fe) are the most prevalent metallic bio-absorbable metals.12 Following their use in medical procedures such as tissue repair, illness diagnostics, or support without causing adverse reactions in the host body, these biomaterials corrode and eventually deteriorate in-vivo.13 In cardiovascular stents and bone implants, biodegradable metals outperform polymeric materials due to large mechanical properties.14 Absorbable implant materials already have a huge market thanks to their desirable medicinal qualities. The most competitive polymers for orthopedic and medical therapies are now synthetic biodegradable poly-esters, which can be made economically and with a variety of properties.15 These specifications give rise to new difficulties in the creation of biomaterials. The breakdown products that corrosion releases into the environment are the main cause for worry. Fig. 1 shows the various orthopedic implants.16
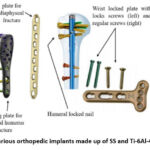 |
Figure 1: Various orthopedic implants made up of SS and Ti-6Al-4 V alloy.16 |
These goods have the potential to be harmful and cause additional harm to the body. Therefore, in order to ensure biosafety, degraded products must be optimized. Furthermore, the kinetics and progression of the healing process should be consistent with the behaviour of deteriorating biomaterials. Inflammation, granulation, and remodelling are the three phases of the healing process in cardiovascular applications. The three phases of cardiac repair after MI is shown in Fig. 2.17
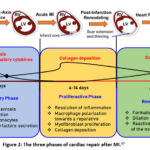 |
Figure 2: The three phases of cardiac repair after MI.17 |
Recent developments in biodegradable materials for medical usage are reviewed, critically evaluated in this paper. Potential medical uses of various re-usable materials and their characteristics were also covered. Due to their advantageous mechanical and chemical characteristics, new materials are showing promise for usage in biological applications. Thus, it is also argued if it is possible to alloys with the existing materials.
The significant advancements in the knowledge and use of biodegradable metals in implantology is discussed in this review. The current developments in the creation of new alloys, like materials based on molybdenum, which have exceptional mechanical qualities that have not yet been investigated in the context of biodegradable implants. Additionally, the state-of-the-art surface modification and fabrication methods that are essential to maximizing the functionality of biodegradable metallic implants is to be discussed. These developments mark a substantial shift from the typical research environment and present encouraging paths toward overcoming the drawbacks of standard materials like iron and zinc. This review highlights the potential of biodegradable implants to transform the field of biomedical applications and offers important insights into their future by clarifying these innovative developments.
This review thoroughly examines the intricate interactions between various conditions, material issues in order to clarify the fundamental metallic parts. These materials’ corrosion mechanism is thoroughly investigated, emphasizing the dynamic mechanisms that contribute to their gradual deterioration. Furthermore, it is clarified that the vital role in-vitro and in-vivo research plays in determining how biodegradable implants react in controlled laboratory environments and in the human body. These investigations demonstrate the complex relationships that exist over implant material and body environment. The comprehensive understanding of biodegradable metallic implants by thoroughly addressing these mechanisms.
Bone defects and healing mechanisms
Bone defects are defined as deficiencies in bone matrix brought on by trauma or surgery, which frequently result in localized body malfunction, delayed or absent healing, and non-union.18 The severity of bone abnormalities is not, however, well defined or categorized. A critically sized bone defect is generally thought to require manual surgical intervention since it cannot heal on its own. However, noted that a bone loss 50%, which is considered critical-size. Nonetheless, it needs to consider the body’s condition defect’s anatomical placement. According to Haines et al.19, the degree of infection and the extent of the defect were important variables influencing therapy effectiveness. Therefore, in order to provide individualized treatment for clinical bone deformities, we need to consider all of the elements that could influence the defects.
Two processes that are crucial to both bone development and natural bone repair following damage are intramembranous ossification (IMO) and endochondral ossification (EO). To put it briefly, IMO can enhance inner and outer periosteum, thicken and calcify the periosteum. On the other hand, EO primarily encourages bone marrow cavity, surrounding environment, hematoma at the fractured end, resulting in the formation of fibrous tissue, granulation tissue, and temporary cartilage tissue. Eventually, bone tissue is formed when osteoblasts infiltrate and replace chondrocytes.20 Following an injury, the healing process of bone differs from that of spontaneous bone growth is mentioned in Fig. 3. Mineralized biomaterials, such as ceramics based on calcium phosphate and other mineralized biomaterials, have been shown in several investigations to be efficient activators of IMO pathways.21 In contrast to calcified biomaterials, the EO route is promoted by biomaterials (such as synthetic and naturally produced polymers) improve cell treatment. Despite fact that numerous studies have documented these phenomena, it is unclear how precisely various biomaterials can trigger osteogenesis via various routes Barbieri et al.22. Due to porosity characteristics of scaffold particularly crucial because of the requirement to offer superior mechanical support as well as attachment.
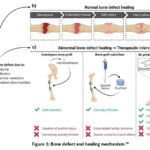 |
Figure 3: Bone defect and healing mechanism.20 |
The EO pathway equals to IMO pathway, is primarily responsible for the growth of the majority of bone in the human body. By giving undifferentiated cells external stimulation, such as a mineralized/mineralizable platform, stem cells can be stimulated to differentiate into functional osteocytes, or osteoblasts Quarto et al.23. Researchers have focused a lot of attention on bone repair by EO stimulation in recent years. Generally speaking, biomaterials stimulate cells locally through the EO pathway to induce osteogenesis.24 A recent study demonstrated that by simulating the natural extracellular matrix (ECM), EO may be successfully induced by exclusively biomaterial-based treatments to heal critical-size bone lesions Petersen et al.25. Apart from biomaterials, Nilsson Hall et al.26 discovered that the EO route may also mend critical-size bone lesions through callus organisms made of certain cells that can be spatially bio-assembled into multi-modular structures.
Biodegradable materials for bone implants
For almost 50 years, bone defect repair has been strongly associated includes biodegradable materials.27 Due to their biodegradability, those are frequently utilized in tissue engineering. It expands and breaks down, and the tiny biomolecules created during this process can control the regenerative milieu to accommodate the bone tissue’s expansion. Simultaneously, the graft’s mechanical qualities progressively deteriorate, and the body’s biological stress shifts, preventing stress-shield. As a result, the harm and associated financial burden of a second procedure are avoided by the biodegradable biomaterial. Based on the state of research, biodegradable materials are mostly made up of materials based on magnesium, biodegradable polymers, and biodegradable ceramics. Various biodegradable materials for bone implant applications are mentioned in Fig. 4.
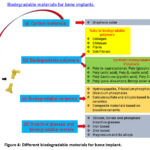 |
Figure 4: Different biodegradable materials for bone implant. |
Graphene based materials
Materials from the graphene family should undergo a thorough evaluation of their cytotoxicity and biocompatibility before being selected for clinical trials. “is graphene a material that is biocompatible?” The response remains contentious. In aqueous media, unfunctionalized graphene readily agglomerates and is hydrophobic. A thick coating of nonspecific proteins can displace water from hydrophobic surfaces and instantly build up on the materials, causing the nanoparticles to be recognized by the immune system.28 For graphene to be employed in biomedical applications, it must first undergo chemical functionalization, which includes the oxidation, reduction, and insertion of functional groups. This process increases the hydrophilicity of graphene. Materials belonging to the graphene family exhibit varying toxicities, functions, and chemical characteristics. According to Das et al.29, the reduced-Graphene Oxide (rGO) is lower harmful, intriguing to observe oxidative stress increased as the density of oxygen. One of the main elements mediating cellular cytotoxicity was the density of functional groups on the GO sheet. In addition to COOH groups were among the many variables that affected.
Graphene functionalization
In order to create composite materials, graphene must be functionalized because its clean structure. These groups from a covalent bond. Fig. 5 indicates both covalent and non-covalent functionalization of graphene.30 Using intermolecular interactions, the GO is formed. The 2D structure, XRD spectra, SEM, and FTIR spectra of GO is shown in Fig. 6(a-d). The various applications of GO were shown in Fig. 7.
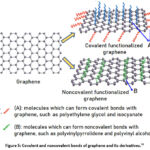 |
Figure 5: Covalent and noncovalent bonds of graphene and its derivatives.30 |
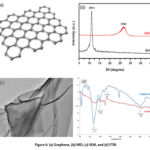 |
Figure 6: (a) Graphene, (b) XRD, (c) SEM, and (d) FTIR. |
 |
Figure 7: The wide application of graphene-based materials. |
Graphene in-vitro and in-vivo responses
In-vitro
According to Chang et al.31, the GO can produce intracellular accumulation and dose-dependent oxidative stress in a lung cancer epithelial cell line (A549), and a modest decrease of cell viability was noted at high concentrations of GO (≥ 50 μg/mL). Wei et al.32 showed that at a low dosage of 0.1 μg/mL, pure GO promoted the proliferation of BMSCs while inhibiting of 10 μg/mL. In a similar vein, 200 μg/mL of GO obviously resulted in fewer cells, but 300 μg/mL of GO was found to have a stronger cytotoxic effect.33 Preosteoblasts (MC3T3-E1) viabilities are marginally impacted over rGO at < 62.5 μg/mL, considerably (p < 0.05) reduced doses (≥ 100 μg/mL), according to Kim et al.34. Furthermore, when applied at low concentrations, CXYG and GQDs both demonstrated minimal cytotoxic potential. Simply put, materials belonging to the graphene are cyto compatible around less concentrations and have minimal detrimental effects on cell viability, proliferation, and morphology; however, concentration is not the only relevant element.
Second, it has been suggested various shapes like layers are added intricacy to the cytotoxicity of the graphene family. Talukdar et al.35 assessed the cytotoxicity, and the trend GNOs > GONRs > GONPs was followed by the CD50 values, suggesting GONRs offers larger cytotoxic than GONPs. Therefore, another important factor in mediating cytotoxicity is the form of the nanomaterials in the graphene family. For example, whereas MWCNT and graphene under various shapes MWNTs are tubular, while graphene is flat, they have a comparable crystalline structure and chemical makeup. Only at 50 μg/mL did GO exhibit cell development action of SK-N-SH. In contrast, MWCNTs demonstrated acute cytotoxicity by preventing cell proliferation at low concentrations (6.7 μg/mL). The MWCNTs showed considerable cytotoxicity on HeLa cells, while GO showed only mild growth inhibitory effect, even at concentrations as high as 50 μg/mL.36 They attributed these phenomena to their diverse shapes and physical/chemical characteristics.
The negative effects of nanostructured graphene derivatives were more noticeable in SK-N-SH cells as a brain cell line than in HeLa cells.36 Yoon et al.37 assessed particle size influence on cytotoxicity, and discovered that whereas the bigger graphene (80.9 ± 5.5 nm), primarily agglomerated over cell causes less toxicity, the smaller ones (30.9 ± 5.4 nm) caused apoptosis due to increased cell absorption.
Akhavan et al.38 described the probable size-dependent uptake mechanisms of protein-coated GO nanosheets and found that smaller nanosheets (420 ± 260 nm) entered cells primarily through clathrin-mediated endocytosis, while larger nanosheets (860 ± 370 nm) first adhered to the cell surface, then underwent membrane invagination, pseudopodia extension, and ultimately entered cells primarily through phagocytosis. Human umbilical vein endothelial cells are seeded 10 μg/mL of GO at various sizes (800 nm and 400 nm) by Das et al.39. The MTT assay results demonstrated bigger sheets are larger hazardous, subsequently, bigger GO, rGO (800 nm) were reduced down into smaller ones (70 nm) by ultrasonography. Following ultrasonication, increased cytotoxicity was noted, suggesting that the smaller GO and rGO were more hazardous. Zhao et al.40 used graphene oxide (GO) material for bone repair, and they used MC3T3-E1 cells for the culture, and they found the used cell is excellent for orthopedic applications, and optical microscope, fluorescent images of cells are mentioned in Figs. 8(a-b), and Cell apoptosis analysis was shown in Figs. 9 (a-d).
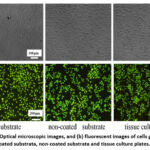 |
Figure 8: (a) Optical microscopic images, and (b) fluorescent images of cells grown on the coated substrata, non-coated substrata and tissue culture plates.40 |
 |
Figure 9: Cell apoptosis determined by Annexin V-FITC and PI assays on (a) coated substrata, (b) non-coated substrata and (c) tissue culture plates. (d) statistical analysis of cell apoptosis.40 |
In-Vivo
When Mullick et al.41 exposed zebrafish embryos to larger-sized GO dispersions, they reported decreased embryo viability in the lower GO dilution, but a larger embryo mortality against control group. Dosage of 25 mg/L, MWCNTs caused major morphological abnormalities in developing embryos, whereas GO did not significantly increase apoptosis in embryos. These investigations also showed that the sizes, had a significant impact on in vivo toxicity. Furthermore, intravenous injection are the most common ways that materials from the graphene family are introduced to animal models.
In their assessment of the toxicity of nanoscale GO in mice administered intravenously, Li et al.42 discovered GO is primarily for liver, lung caused lung fibrosis. A GO/PEG may lessen acute tissue damage and decrease GO retention for liver. Because GO was more toxic over agglomerated GO and Pluronic diluted GO given to mice’s lungs, causing long lasting lung damage, Duch et al.43 investigated ways to lessen the harmful effect of graphene nanoparticles in the lung. The production of pristine graphene by liquid phase exfoliation considerably reduced the toxicity, which was further reduced when it was distributed using the block copolymer Pluronic.
Zha et al.44 used graphene foams (GF), and graphene oxide foams (GOF) on the rat model for short term and long term for vivo toxicity studies, and they have seen that, GFs and GOFs are not caused any discernible hematologic, hepatic, or renal toxicity upon implantation, there is no change up to 7 months. For a considerable amount of time, only granulomas were seen at the implantation site. Better in vivo biocompatibility was demonstrated by HE-stained pictures (Fig. 10). The most straightforward and efficient method of evaluating was by subcutaneous experimentation have impact on patterns of GO in vivo. The GO/HA composite on MG-63 cells of Mahmoodi et al.45 and their results are shown in Fig. 11.
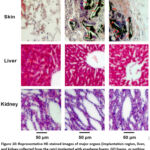 |
Figure 10: Representative HE-stained images of major organs (implantation region, liver, and kidney collected from the rats) implanted with graphene foams, GO foams, or nothing at day 14 post-implantation.44 |
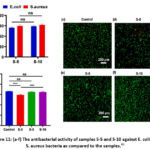 |
Figure 11: (a-f) The antibacterial activity of samples S-5 and S-10 against E. coli and S. aureus bacteria as compared to the samples.45 |
The required time, pathway of degradation and removal of GO from body
For bone implant applications, graphene oxide (GO) is frequently added to scaffolds or coatings to enhance mechanical strength, biocompatibility, and osteogenic differentiation. In contrast to intravenous or free-form GO, GO is often encapsulated in a matrix (like hydroxyapatite or polymers like PLGA or PCL), which affects how it is biodegraded and removed from the body. Depending on (i) the GO concentration, (ii) the scaffold composition, (iii) crosslinking or chemical bonding, and (iv) local biological activity (e.g., enzyme presence), the GO in bone implants degrades gradually, usually over weeks to months. Particularly in small animal models, in vivo investigations indicate partial deterioration after 8–12 weeks.
The main mechanisms of bone tissue degradation are (i) enzymatic degradation by immune cells, such as macrophages and neutrophils surrounding the implant site, which produce myeloperoxidase (MPO) and ROS, which can oxidatively cleave GO sheets; (ii) slow hydrolytic degradation (in biodegradable polymer composites), where polymers like PLGA degrade via hydrolysis, exposing GO to body fluids and enzymes over time; and (iii) phagocytosis, in which macrophages engulfy small GO fragments released from the scaffold, resulting in lysosomal processing and partial degradation. The GO is primarily trapped or embedded in the scaffold at the implant site. Local immune cells may absorb and hold onto larger or more persistent particles for an extended period of time.
Magnesium based composites and alloys
Alloys based on magnesium exhibit potential as biomaterials for orthopedic purposes. In magnesium, there is a significant mapping of anisotropic properties and Young’s modulus with human cortical bone. Magnesium also shows a notable degradation. Magnesium is widely used medical applications. The known application of this technology was in 1878, when magnesium wires were effectively used as blood vessel ligatures. However, several applications were later studied, including the use of magnesium sheets, plates, and screws for vascular anastomoses, intestinal, and nerve anastomoses, cavernous hemangiomas. Trials successful than expected for bio-degradable. Issues such post-operative later decades. The Mg-Cd composite used has been shown in studies by Espiritu et al.46 to be useful in treating fractures. These magnesium-cadmium implants were found to promote the formation of robust calluses throughout the healing process of fractures. Infections or the development of gas cysts, however, caused failures in some cases. Interestingly, none of the patients had abnormal changes in their serum magnesium levels or acute inflammatory reactions.
Magnesium alloys have been shown to be more biocompatible than bio-polymers like polyether ketone (PEEK), polyglycolic acid (PGA), and poly L-lactic acid (PLLA). Furthermore, it outperforms tricalcium phosphate (TCP), and hydroxyapatite (Hap) due to their lesser strength.47 Magnesium alloys are easily sterilized due to flexibility. The magnesium alloys by-products that break down, particularly magnesium ions, possess distinct Osteopromotive. Breakdown of magnesium-ion, made possible proteins, increases Osterix levels and bone repair. Biological limitations due to propensity to mechanically disintegrate essentially.
According to the advancements in magnesium-based research, earlier study on degradation of magnesium, the use of titanium dioxide (TiO2), hydroxyapatite (HA), and inert materials. Recent advancements indicate that in addition to corrosion protection, coating application is being researched to offer a range of therapeutic responses. For modifying the texture and grain size of magnesium alloys, a number of mechanical popularities in addition chemical coatings.48 mentioned the degradation process of Mg for bone implant applications in their review paper and process Is shown in Fig. 12.
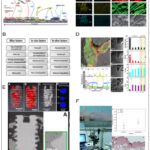 |
Figure 12: (a-f) Step by step degradation process of Mg.48 |
Magnesium alloys and their corrosion
Bioimplants must be cyto-compatible, hemo-compatible, and corrosion resistant in order to operate safely in in-vivo environments. In orthopaedic implants, the corrosion rate of magnesium and materials based on magnesium exceeds biosafety requirements. In particular, if the corrosive media contains chloride at values more than 3 mM/L, the magnesium turns into magnesium chloride (MgCl2). It has a high solubility in aqueous solution encourages normally higher levels of chloride. The magnesium implant failed before its planned service life as a result of hydrogen embrittlement brought on by H2 trapping in the corrosion voids. On the other hand, magnesium alloys that break down more slowly (less than 0.5 mm/year) are better at promoting bone repair.
According to Kirkland et al.49, galvanic coupling causes localized corrosion in most magnesium alloys, accelerating the degradation process by generating micro-galvanic cells. Magnesium alloys contain metals such as copper (Cu), nickel (Ni), and aluminium (Al) that are less electronegative than magnesium alone. They may therefore cause damage to the protective layer Mg (OH)2. When the oxide film is destroyed, corrosion increases because the magnesium matrix comes into contact with the surrounding fluid.
Witte et al.50 created rods using magnesium alloys (WE43, AZ31, LAE442, and AZ29). Magnesium alloy implants degraded in in-vivo at different rates. The main factor influencing the degradation was the alloy composition. After prolonged immersion, LAE442 showed the slowest corrosion rate of any alloy, whereas, WE43 Mg/Ca and P-rich apatite. A number of surface modification techniques have been developed in addition to alloying. The surface modification techniques of Mg and its alloys is shown in Fig. 13.
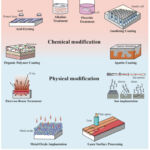 |
Figure 13: Surface modification techniques of Mg and its alloys.48 |
Magnesium based bioimplants
Magnesium based alloys using a variety of surface treatment approaches was given by Khatun et al.51. Micro arc oxidation (MAO) was widely used effective surface treatment method. When magnesium was treated with MAO, Wang et al.52 found difference in Young’s modulus and strength. The MAO coating method was examined by Chen et al.53, and proposed degree of corrosion damage is determined by the layer’s thickness as well as the homogeneity and porosity of the MAO coating. Some studies also focused on enhancing the hydrogen storage capacity of magnesium-based materials to counteract embrittlement occurrences.
Lu et al.54 created a core-shell Mg@Pt composite with a nanostructure using the plasma arc technique. Additionally, the Rieke process could be used to generate metal nanoparticles of a variety of metals, including Al, Mg, Ni, Co, and others. Suh et al.55 used a machine learning-based data-driven methodology to investigate novel materials and forecast material properties. They used mean and standard deviation numbers to enhance the data. A single data point is multiplied by twice the number of variables in the first technique, which adds or subtracts the standard deviation for each input and output variable. In this case, by iterating over a total of six variables and changing the standard deviation of one while maintaining the average value of the other variable constant, one data point can be stretched by a factor of 12.
The development of biomaterials depends on various additive manufacturing (AM) techniques since they can create intricate geometrical structures and complicated material patterns. In order to overcome the challenges presented by conventional manufacturing techniques, the application of AM technologies for the production of magnesium (Mg) alloys has increased recently. Magnesium is naturally difficult to manufacture using the latest 3D processes because it oxidizes unpredictably and is very reactive in its pure condition. Furthermore, because of their high surface energy, magnesium in liquid. When combined, these factors make magnesium alloy manufacture a challenging process that requires specialized equipment to ensure worker safety and a controlled environment.
On the other hand, powder bed fusion (PBF) is the method for making magnesium alloys that has been explored the most.56 This technique enables the creation of intricate external and internal geometries with densities as high as 96.13% by employing minimal heat flow. Even though alloying and treatments have made significant strides in enhancing the performance of magnesium-based biomaterials, much more work need to be done to make magnesium the ideal biodegradable material. Recent advances in materials science and bioengineering have given researchers more control over the mechanical properties and corrosion rates of magnesium. Magnesium alloys are increasingly being used in orthopedic and cardiovascular applications. Fig. 14 describes how different Mg degradation-triggered osteogenic pathways work in concert and communicate with one another.48
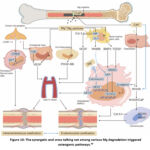 |
Figure 14: The synergetic and cross talking net among various Mg degradation triggered osteogenic pathways.48 |
Magnesium responses in in-vitro and in-vivo
The primary barrier to magnesium’s (Mg) clinical application is its deteriorating nature. Zheng et al.57 demonstrated the remarkable properties of the Mg–Sr alloy in a comprehensive study of its in vitro and in-vivo performance. These included larger tensile strength (203 MPa) qualities and lesser rate of degradation. Yao et al.58 applied Mg–Sr composite and studied the toxicity. Investigation showed that the Mg-0.5Sr alloy deteriorated more slowly, indicating the presence of Ni thrombosis after in-vivo. It has been shown that magnesium alloys become more biocompatible when calcium (Ca) and zinc (Zn) are added.
Notably, during in-vivo breakdown, calcium phosphate (Ca–P) is produced, which promotes improved bone mineralization. Lu et al.59 discovered that the addition of calcium and zinc for vitro, vivo studies when preparing magnesium alloys for orthopedic implants. Examination of magnesium and magnesium/magnesium composite to mice to analyse the in-vivo weight reduction.60 They remarkably discovered a lower rate of biocorrosion in vivo environments. Mg/Mg alloys were biocompatible with the tissues of mice’s kidneys, liver, skin, lungs, and hearts, according to the histological findings of their investigation. The Mg implants applications in fracture fixation and sports medicine is mentioned in Figs. 15 and 16.48
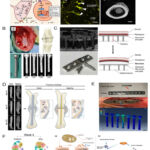 |
Figure 15: Mg implants application in fracture fixation.48 |
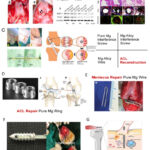 |
Figure 16: Mg implants application in sports medicine.48 |
The required time, pathway of degradation and removal of magnesium from body
Because of its mechanical qualities, biodegradability, and biocompatibility, magnesium-based materials particularly magnesium implants, like magnesium alloys are being utilized more and more in biomedical applications. The body breaks down magnesium mainly chemically, such as through corrosion in physiological environments, particularly when water and ions (like Cl⁻) are present. (ii) The biological environment influences the rate of corrosion, such as pH, local ion concentration, proteins, and cells. Near bone or tissue, osteoclasts and macrophages may hasten the breakdown of magnesium.
Pure magnesium implants (cardiovascular and orthopaedic) degrade over a period of three to twelve months, while magnesium alloys (stents, screws, and plates) such as Mg-Ca and Mg-Zn take six weeks to twelve months, and surface-treated magnesium can take up to two years. The body converts magnesium into Mg²⁺, which can be eliminated by metabolism or eliminated in urine; H₂ is eliminated through the lungs and diffusion.
Zinc and zinc composites
Zinc (Zn) is another popular biodegradable metal that is prized for its high biocompatibility and abundance in the human body. The second most common element in the human body is Zn2+, which is mostly found in muscles and bones. Compared to titanium and stainless steel, its mechanical and physical properties are more in line with those of human bones. Remarkably, around 600 enzymes depend on Zn2+ function. It is necessary to emphasize that despite its physiological importance, zinc is not widely regarded as a crucial bioresorbable drug. The potential medical applications of zinc, particularly in the areas of orthopedics and cardiovascular disease, have not received much attention. Furthermore, a number of zinc alloys’ perceived toxicity has restricted their usage in biological applications.
Zinc composites responses in in-vitro and in-vivo
Zinc (Zn) is an excellent option for bone implants, has been the subject of numerous studies to assess its corrosion behavior, cytocompatibility, and bioactive effects. Zinc is a trace element that is necessary for numerous biological functions. In numerous in-vivo investigations, zinc has demonstrated non-cytotoxicity to endothelial cells, strong antiatherogenic actions, and high hemocompatibility. Pure zinc gives corrosion when compared to Mn and Fe. Torne et al.61 showed that, in contrast the phosphate-buffered saline and Ringer’s estimations, the Zn surfaces formed passivation films composed of inorganic components. The barrier in whole blood was also examined using electromechanical measures.
Notably, the content of proteins human blood has a significant impact on implant corrosion, and proteins present or absent alters the corrosion behavior of biodegradable zinc alloys. Several studies have hypothesized that an excess of zinc ions produced during decomposition leads to fibroblasts. Su et al.62 demonstrated that pure zinc negatively impacted MC3T3-E1 cells, while MG-63 cells demonstrated a same effect. Alloying zinc with other metals, such as magnesium (Zn–3Mg), reduces the possibility of genotoxicity in the human body, claim by Krus et al.63. Kubasek et al.64 reported that the Zn-0.8 Mg alloy did not cause any appreciable damage to U-2OS cells. Zn-0.05 Mg and Zn-0.5 al-xMg alloys have been found to have a positive impact on cell. Additionally, Zn-Mg disintegration facilitates help for bone materials. Additionally, it has been noted that calcium (Ca) and strontium (Sr) improve cytocompatibility.
Yin et al.65 observed no symptoms of severe inflammation, platelet aggregation, thrombosis, or intimal hyperplasia. Similarly, Zhou et al.66 found no inflammation or thrombus formation in coronary arteries following two years of Zn-0.8Cu alloy stent implantation. Young pigs’ iliofemoral arteries were treated with Zn–3Ag alloy stents by He et al.67, and the outcomes showed satisfactory healing without thrombosis or vascular obstruction. When Chen et al.68 implanted Zn-xMg alloys in the rat abdominal aortas, they noticed comparatively little inflammation and intimal activation. Rats’ abdominal aortas were implanted with Zn-0.1Li alloys by Ma et al.69, who observed a minor inflammatory reaction. When Drelich et al.70 implanted Zn-xAl alloy strips in the rat abdominal aortas, they saw a little inflammatory reaction. The several uses of zinc-based implants for bone regeneration and repair are shown in Fig. 17.71
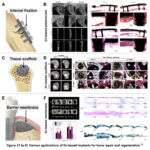 |
Figure 17: (a-f). Various applications of Zn-based implants for bone repair and regeneration.71 |
Functionalization of zinc materials
Surface modification techniques have been used by many researchers to increase the properties of Zn materials. These methods are believed to be effective because of the modification of the surface characteristics. Despite lack of research on Zn surface treatment in orthopedic applications, and wear decrease. However, because of their toxic qualities, they are prohibited throughout the world. A common chemical conversion process that enhances surface qualities like adhesion, corrosion resistance, and wear resistance is Zn–P coating.
Numerous biological processes, including DNA metabolism, signal transduction, apoptotic control, and gene expression, are impacted by zinc. Zinc was useful material in the preparation of energy, enzymes, it is said covering the metal’s surface with a protective laye. The electrophoretic deposition coating method to enhance the properties of degradable Zn alloy implants is shown in Fig. 18.71 and explains how the Zn–Cu stent two-year implant in the coronary artery is modified on the surface. The osteoconductive and osteoinductive properties of Zn-based scaffolds are shown in Fig. 19.71
 |
Figure 18: Possible molecular mechanisms of Zn-based internal fixation materials.71 |
 |
Figure 19: (a-b): Osteoconductive and osteoinductive effects of Zn-based scaffolds.71 |
Biodegradable implants made of zinc-based materials are becoming more and more common, particularly in orthopedic and cardiovascular stents. Zinc is a biocompatible and bioresorbable metal since it is necessary for biological function. In physiological settings, zinc primarily degrades by electrochemical corrosion, which yields zinc hydroxide (Zn(OH)₂) or zinc oxide (ZnO) as the end products. The zinc ions that are released into bodily fluids can also form protein-bound complexes, zinc phosphate, or carbonate. While zinc degrades more quickly than other materials, it does so more slowly than magnesium. The degradation period of pure zinc (used in stents and bone implants) is 6 to 24 months, while that of zinc alloys such as Zn-Mg and Zn-Ca for orthopaedic implants is 12 to 36 months. Zn covered with polymers degrades over a period of more than three years. According to albumin, metallothionein, and other plasma proteins, the body eliminated Zn2+ ions.
Iron (Fe) based bioimplants
Iron has been frequently used because of its affordability, machinability. The compounds and alloys are perfect option for bio implants. Fe-based materials are primarily utilized in three fields, according to the literature, stents, scaffolds (Fig. 20) and bone implant materials for surgical operations.72 The non-degradable metal implants usage like screws, plates for bone operations outcomes larger toxicity. Removing such implants may result in issues including pain or anomalies under the implant location. Because they can be used for bone regeneration, Fe-based metallic implants get around these problems. The Fe material is also another application like scaffolds and tissue repair. Because of mechanical integrity, biodegradability, porous scaffolds composed of iron are mostly utilized to repair hard tissues, such as bones.
 |
Figure 20: Cell viability and metal concentration: (a, b) direct method, (c, d) indirect method.72 |
Iron composites responses in in-vitro and in-vivo
Iron (Fe) has proven to be incredibly biocompatible in a number of in-vitro analysis. It is demonstrated that it did not inhibit halt cell growth. Furthermore, pure industrial-grade Fe showed negligible cytotoxicity and good hemocompatibility and biocompatibility, according to Chen et al.73. Lin et al.74 found that the extraction medium and oxygen availability were crucial for both incubation and extraction in their study of the L929 cells. Nonetheless, it was discovered that alloying elements affected the biocompatibility of iron alloys. The alloy content in the medium had a significant impact on the inhibitory effect, according to research on alloying cells. Additionally, multicomponent Fe alloys were examined. When Schinhammer et al.75 investigated the cytocompatibility of Fe–Mn–C (-Pd) alloy with HUVECs, they extracted medium’s composition significantly affected both metabolic activity and cell viability. Furthermore, a substantial relationship was found between the rates of corrosion of Fe alloys and cell viability. Orinak et al.76 analysed a perfect in-vitro cell for Fe based scaffolds and noticed fibroblasts died in 1-day duration. Orinakova et al.77 observed a higher cell viability for Fe/bio-ceramics.
Furthermore, surface modification has proved consideration when choosing biomaterials. They need to be slow to corrode and biocompatible. Peuster et al.78 used Fe stents in descending aortas to conduct first in vivo testing of iron stents. Throughout, there were no thromboembolic problems, and no neointimal development, were observed. After the identical stents included aortas pigs, they were observed for 1 year; neither Fe overload nor organ damage was discovered. No indication of local toxicity from corrosion products was found. In a short study, the safety and efficacy of Fe stents were assessed by implanting them in the coronary arteries of young pigs and keeping them there for 28 days. There was no sign of thrombosis or particle embolization, and histomorphometry revealed neither fibrin accumulation nor severe inflammation.
He et al.79 observed a decreased inflammatory response and endothelial cell coverage of the Fe stents used in young pigs. There was no sign of local tissue necrosis or thrombosis even a year later. Nitrite iron stents shown good biocompatibility. The Fe were implanted into the rat femur by Khan et al.80. After 1 year, no complaints of pathological issues are found. Even though there was some mild edema for a day or two following surgery, often bulging as anticipated. There was no tissue damage from the implants. Furthermore, Fe–Mn–Si implants showed remarkable biocompatibility. In certain tissue studies, Fe implants were found to be mildly toxic, while the surrounding tissues only experienced modest damage. Ulum et al.72 assessed the bioactivity of Fe-bioceramic composites in the leg bones of Indonesian sheep. The inflammation caused by the stent decreased 35 days of usage, and when the lesion healed, the tissue did not react. Rabeeh et al.81 investigated in-vitro of gold coated Fe material and observed an enhanced the degradation. Fig. 21 is the diagram of the gold coated Fe.81 Fig. 22. (a-b) represents the MTT assay for MG-63 cells.81
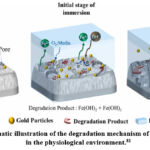 |
Figure 21: Schematic illustration of the degradation mechanism of gold sputtered iron in the physiological environment.81 |
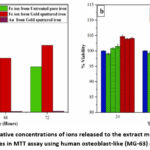 |
Figure 22: (a) Cumulative concentrations of ions released to the extract medium. (b) Cytotoxicity studies in MTT assay using human osteoblast-like (MG-63) cells.81 |
Functionalization of Fe materials
Fe based implants are little bit slow. The Fe implant is encased in tissue level even in the face of ongoing corrosion. It has been challenging to accelerate the rate of Fe breakdown in biological applications. Numerous techniques like alloying, surface treatment, adding different phases for iron. Among other elements analysed by Wermuth et al.82, it was determined that Fe bio alloys.
Cheng et al.83 claim that the addition of a second phase decreased the corrosions. Including TCP, HA, and BCP, are regularly used bioceramics because of their solubility in the body. It has been noted that bioceramics and surface modification can improve biocompatibility. Corrosion resistance is significantly increased when an oxide layer forms on the surface of pure iron. Furthermore, the mechanical stability and corrosion resistance of Fe stents are enhanced by NaCl. Although nitriding did not significantly improve corrosions. Nitride iron coronary stents exhibited a slow rate of deterioration, with almost complete corrosion occurring 13 months post-implantation and generating minimal degradation products.
Corrosion can cause Fe stents to lose their mechanical integrity. Advancements can improve radial, tensile strength, microhardness. While metallic materials produced using this technique have comparatively lesser mechanical characteristics, Fe developed with SPS offer outstanding corrosion resistance. Furthermore, the mechanical properties of Fe are enhanced by rolling and electrodeposition. Excellent biocompatibility is a feature of bioceramics. The Fe-bioceramics combination is among the best biodegradable materials. The ability to blend metal-to-non-metal is another advantage of powder metallurgy. The corrosion process may also be slowed down by these treatments. Developing porous materials to speed up corrosion is highly recommended. Iron properties made into metallic foam without losing its shape or strength.
The required time, pathway of degradation and removal of iron from body
As substitutes for permanent metal implants, iron-based biodegradable materials such as scaffolds, orthopedic pins, and iron stents are being researched. Although they deteriorate slowly, they have considerable mechanical strength. Under physiological conditions, iron corrodes electrochemically, albeit more slowly than magnesium and zinc. Because of passivation (the creation of a protective layer), these corrosion products may build up at the implant site and further slowdown degradation.
Less than 24 months pass before the pure iron utilized in cardiovascular stents degrades. Iron alloys with a degradation duration of 12 to 36 months, such as Fe-Mn, Fe-C, and Fe-P, are employed in orthopedic and vascular applications. It takes six to twenty-four months for the coated iron to decompose. The body gradually releases the Fe²⁺ and Fe³⁺ ions, which are carried by transferrin in the blood.
Biodegradable polymers
Generally speaking, polymers are macromolecules made up of repeated monomers joined by covalent bonds. Researchers have preferred biodegradable polymers among them because to their degradability, which is crucial for bone defect repair. Polymers categorized either natural or synthetic based on where they come from. Because of their biodegradability, bioactivity, and biocompatibility thoroughly researched materials for repairing bone defects. Water mixing qualities, potential is among of its drawbacks, though. Synthetic polymers made to biomaterials of superior qualities by adjusting their design and synthesis characteristics. Nevertheless, the acidic breakdown products of some synthetic polymers in vivo alter the local pH level, speeding up the rate of implant degradation and triggering inflammatory responses.
Natural biodegradable polymers
Collagen
Collagen, the primary structural protein of tissues, is crucial for controlling the extracellular matrix of the cellular milieu. Collagen is the primary component of the roughly 30% matrix that makes up bone, a complex, naturally occurring structure. One common biomaterial in the biomedical industry is collagen. Collagen and apatite crystal-based composite membranes are gaining popularity due to their superior mechanical qualities. Because collagen/bioceramic composite scaffolds are more akin to actual bone in composition, they can produce greater bone repair benefits from a biomimetic standpoint. Nevertheless, the collagen must be cross-linked, and these scaffolds frequently have poor mechanical qualities. Other approaches have been investigated to enhance the functionality of such scaffolds. By using surface biosilification technology to coat acellular, Wang et al.84 recently created novel biomimetic nanosilica-collagen scaffolds that successfully repaired the rabbit.
When constructing dependable biomedical materials for scaffolds, wound dressings, and implants, it is essential to comprehend the mechanical strength and the requirement for cross-linking in biopolymers, especially collagen. Mammals’ skin, bones, tendons, ligaments, and connective tissues all contain collagen, the most prevalent structural protein. Cross linking improves mechanical strength, increases heat stability, reduces enzymatic degradation, and preserves structural integrity in damp environments.
Chitosan
A naturally occurring polymer with a linear structure, chitosan (CS) serves as structural element. CS has been shown to improve cell adhesion, proliferation, osteoblast differentiation, and mineralization due to its biological activity, biodegradability, antibacterial and biocompatible properties, and hydrophilic surface. In other words, CS’s cationic characteristics enable it to interact with anions control, so playing physiological importance. After undergoing sophisticated preparation procedures, CS shaped to 3D scaffolds of various porosity topologies. Such as nanotechnology and 3D printing, and composite materials can be used for bone abnormalities fixing. It should be noted that the CS usage is majority of load-bearing settings was unsatisfactory, regardless of the manufacturing process employed. Therefore, the only way to create scaffolds with improved mechanical and biological qualities is to combine CS with different natural or synthetic polymers or bioceramics. Uneven bone deformities can be filled with injectable CS hydrogels. According to a recent study, Cui et al.85 created a sort of network of microporous, linked. In situ cross-linking of CS resulted in a hydrogel, the addition of nanosilicate raised the hydrogel’s Young’s modulus and reduced its rate of breakdown.
Fibrin
Thrombin acts on fibrinogen to generate fibrin, a natural biopolymer, in the final stage of the coagulation cascade. Human blood is a steady supply of fibrinogen, thrombin, and fibrin precursors, which lowers production costs and the chance of needless disease transmission. Fibrin is a viable option for inclusion in the perfect scaffold for fixing bone defects, given the crucial function that hemorrhages play in the early stages of bone healing. Additionally, fibrin has the ability to stimulate osteogenic differentiation and angiogenesis, both of which can quicken the rate of bone repair. To get around fibrin’s drawbacks, however, other materials must be used as well because of its weak mechanical qualities and quick rate of disintegration. Although fibrin can be made into injectable fibrin hydrogels, it should be used in conjunction with other biomaterials to treat bone abnormalities because it cannot treat them on its own. Nonetheless, there is ongoing debate on fibrin glue’s potential to enhance bioceramics’ ability to mend bone, and other researchers have focused on fibrin’s negative effects. The immunological reaction brought on experiments of excessive fibrin quantity are two potential causes. Fibrin utilized for repair of bone defects in addition to altering scaffolds. This helps to attract host cells and encourage angiogenesis, which in turn speeds up the repair process of bone.
Fibrin made of silk
Silkworms, spiders, and other insects are the primary producers of silk, a natural protein biopolymer that is used to make silk fiber (SF). Mulberry silk is the type of silk that has been the subject of the greatest biomedical study. Fibrin and sericin are the two primary protein components found in silkworms’ silk. Since sericin causes the host’s immune system to reject it, it is degummed during SF purification. Silk has gained importance in the field of bone tissue engineering due to its great inherent strength. Researchers in bone tissue engineering use silk-based scaffolds because they are found to have more mechanical strength than other naturally biodegradable polymer scaffolds. Silk scaffolds’ tunable and often slow pace of deterioration aids in optimal size bone healing. Recycled polymers of PLA, PGA, and PLGA were used as bone implants.
The Runx2, OCN are examples indicators that have been shown to be upregulated by silk fibroin. Degradable bioceramics can be mixed with silk fibroin to create enormous, intricately shaped scaffolds that are incredibly strong and have the right amount of porosity to enable cell proliferation, which is crucial for the healing of big bone lesions. For load-bearing bone healing, McNamara et al.86 recently created SF-hydroxyapatite (HA) ceramic scaffolds with a variety of mechanical structures.
Biodegradable synthetic polymers
Aliphatic polyesters, including PCL, PLA, PGA are most researched synthetic degradable polymers in recent years. It has been demonstrated that these materials are biocompatible and degrade at a controlled rate, and they are good and don’t harm tissues when they’re in vivo. Furthermore, through manual adjustment polymers with better mechanical qualities can be created. Even though the tissue’s natural metabolic pathway releases and produce, they can cause transplant, degradation, and detrimental impact on repairs particularly when it comes to fixing bone defects in regions that support loads.
Poly (ε-caprolactone)
Poly (ε-caprolactone) (PCL) is a flexible biologic and low-cost polymer that has FDA approval. Despite being biodegradable and biocompatible, PCL was not the best material for repairing bone defects because, according to numerous long-term tests, its mechanical qualities were weak and its rate of breakdown was sluggish. But according to a recent work by Rotbaum et al.87, 3D printed PCL scaffolds’ mechanical qualities can be improved by altering their pore geometry. According to studies, PCL can be applied through different scaffolds to improve cell-cell interactions. According to new analysis, circular lesions in the rat skull with a diameter of 5 mm might be healed in 12 weeks with the help of PLLA/PCL nanofibre scaffolds coated with hydroxyapatite.
Poly (glycolic acid)
Poly (glycolic acid) (PGA) has a straight linear molecular structure and is a basic aliphatic polyester. PGA is the polymer of glycolic acid, which human metabolism byproduct. For many years, PGA to be utilized in clinical applications due to its good tensile modulus and regulated solubility. Glycolic acid, a byproduct of PGA’s rapid breakdown, can be eliminated through the urine. PGA is more mechanically strong than other degradable polymers (such PCL and PLA). In particular, PGA, PCL, and PLA have young’s moduli of 5-7 GPA, 0.4-0.6 GPA, and 2.7 GPA, respectively. However, PGA scaffold by itself was not appropriate fixing bone deformities because of its unreasonably high rate of deterioration in vivo. For the PGA composite based on rabbit was studied by Toosi et al.88 and noticed the rabbit was cured after 12 weeks of therapy.
Poly (Lactic Acid)
A Swedish chemist by the name of Scheele made the initial discovery and naming of Poly (Lactic Acid) PLA, a polymer made of lactic acid, in 1780. PLA, on the other hand, is a biodegradable polymer composed of starch that comes from renewable plant sources, like corn and sugar cane. The most often utilized PLA in clinical settings at the moment are L-PLA and DL-PLA (combination of L-and D-lactic acid. L-PLA is frequently utilized for adequate porosity.
According to one study, scaffolds made of PLA-PCL tissue and loaded with BMP-2 shown good bone healing properties. In order to create scaffolds, PLA can also be mixed with biodegradable ceramics. Zhang et al.89 discovered that at an 8:2 PLA/HA mass ratio, the created porous scaffold performed the best overall. Recently, Ge et al.90 produced Sr-HA/PLLA that may lessen the acidic environment’s deterioration enhance the material’s ability to adsorb proteins, make its surface more hydrophobic, and make it more osteoinducible.
Poly (Lactide-co-glycolide)
The PLA, and PGA followed by degradation, results in Poly (Lactide-co-glycolide) PLGA. By altering the proportion of these two polymers, rate can be controlled. With these benefits the PLGA was perfect biodegradable polymer. Because it may be utilized to create sutures as well as cell and drug delivery systems, PLGA is therefore more widely used by researchers than PGA and PLA. Nevertheless, due to its low osteoconductivity and hydrophobicity, PLGA’s usage in bone healing is restricted, even with the aforementioned benefits. Thankfully, alternative materials can make up for these drawbacks. Using low temperature rapid prototyping (LT-RP) technology, Lai et al.91 created a porous PLGA/TCP/Mg (PTM) scaffold that greatly accelerated the creation of new bone in rabbits with bone defects by promoting both osteogenesis and angiogenesis.
3-hydroxybutyrate, or PHB Poly
The French microbiologist Maurice Lemoigne identified (PHB), a kind of polyhydroxyalkanoate (PHA), for the first time in 1925. PHB is a very biocompatible polymer that can break down both hydrolytically and enzymatically in tissue. When PHB breaks down, the local pH stays within a steady range, in contrast to other popular biodegradable polymers like PGA, PLGA, or PLA. PHB is a double-edged sword since it is less robust and less stiff than materials with stronger mechanical qualities. PHB permits a small amount of mobility upon implantation, which could aid in bone repair. After 36 weeks in vivo, Meischel et al.92 used PHB composites into the femur of SD rats and observed no discernible degradation, indicating that the material’s tensile strength, strain characteristics, and elastic modulus are comparable to those of natural bone, which may therefore be a material that shows promise for the healing of bone defect. Copolymers of 3-hydroxybutyrate and 3-hydroxyvalerate (PHBV) are another extensively researched polymer among PHA, in addition to PHB. Zhang et al.93 altered PHBV surface with hydroxyapatite and used for biocomposite and seen on the 20th day of PHBV scaffolds.
Para-dioxanone (PDS) Poly
A hydroquinone monomer undergoes ring-opening polymerization to produce (PDS), a biodegradable polyester. PDS is widely used in tissue engineering and fracture repair due to its exceptional biodegradability and biocompatibility. PDS fully absorbed when it is used for internal fracture fixation. Regretfully, no research has been done on the application of PDS to heal bone defects.
HYAFF-11
Commonly present in the extracellular matrix, HYAFF-11 is a benzyl ester. HYAFF-11 is interesting substance for tissue healing because of its good biocompatibility and controllable degradation rate based on the degree of esterification. HYAFF-11 is currently utilized in bone replacement, neuron regeneration, and skin restoration. and other domains. According to Mermerkaya et al.94 HYAFF-11 membrane improves osteogenic activity throughout beginning stages of bone healing and crucial in fixing 10-mm rabbit tibial lesions.
Biodegradable ceramics
Natural clay is primary materials used to make ceramic, which are then crushed, mixed, molded, and calcined. Because of their benefits, bioceramics have been employed extensively in the last few decades for the replacement and repair of injured tissues. of precise chemical composition, mechanical compatibility, and biocompatibility. Researchers are particularly interested in biodegradable ceramics for bone tissue engineering. up particular, they are primarily utilized to mend fractures and fill up the spaces left by bone abnormalities. Tricalcium phosphate, dicalcium phosphate, and hydroxyapatite (HA) are currently the most widely used biodegradable ceramics. The benefits of biodegradable ceramics include biological activity, corrosion resistance, and biocompatibility. The biggest benefit is that, upon implantation in the body, they are progressively broken down by cell-mediated and solution-driven mechanisms before being replaced.
Hydroxyapatite
Ca10(PO4)6(OH)2, sometimes referred to as hydroxyapatite (HA), is a commonly utilized bioactive material of over 65% of the total mass of bone. The calcium/phosphorus molar ratio of calcium orthophosphate ceramics ranges from 0.2 to 2.0, while the ratio for HA is 1.67. A high-temperature process is typically used to create the extremely crystalline form of calcium phosphate known as synthetic HA. Given that natural HA exhibits good osteoconductivity due to its remarkable chemical similarities. Their porosity, crystal size, and physical microstructure, however, varies somewhat. HA can immediately integrate with new bone tissue after being implanted into the gap of bone. Because of its comparatively sluggish rate of biodegradation, HA may be detrimental to the healing of bone defects. In particular, bone frequently covers the HA surface after implantation without connective tissue interposition, which prevents the material from degrading and absorbing. After implanting nanocrystalline HA into the rabbit’s distal femur, Brandt et al.95 found that most grafts had no discernible absorption near the edge. Researchers have looked into a number of HA changes in light of the aforementioned drawbacks. The mechanical and biological properties of HA-based bone substitutes can be improved by adding Sr2+ or Mg2+. This may be because the introduction of cations alters the physical and chemical properties of HA. HA bone grafts can improve bone repairs of both critical and non-critical sizes, according to a recent comprehensive review.
Tricalcium phosphate
With a calcium/phosphorus ratio of 1.5, tricalcium phosphate, also known as Ca3(PO4)2 was regular ceramic material. α-TCP, β-TCP, and α′-TCP are the three crystalline forms of TCP. Since of its close resemblance to bone minerals makeup, researchers like β-TCP since it is highly biocompatible and biodegradable. TCP and HA have young’s moduli of 60–75 GPA and 80–110 GPA, respectively. Despite having a somewhat lower mechanical strength than HA, β-TCP biodegrades much more quickly than HA, which promotes the formation of new bone. It is noted that HA/TCP determines the bioactivity and degradation rate of biphasic calcium phosphate (BCP), which are mostly made up of HA and TCP. When porous β-TCP, made using the goat radial shaft, 3 months following the procedure, significant bone growth was observed across the defect. Research has shown that S1P can considerably boost alkaline phosphatase activity and osteogenesis-related genes OPN, OCN, and RUNX2. Collagen is a crucial for bone, and influence vitamin D metabolism and collagen maturation. TCP can therefore be mixed with other substances like collagen, sphingosine 1-phosphate (S1P), and metal ions, to improve its osteogenic potential and biomechanical qualities.
Phosphate dicalcium
A type of acid calcium phosphate, dicalcium phosphate (DCP) has an acidic phosphorus supply and a basic calcium source. The two forms of DCP, which are the primary constituent of calcium phosphate cement (CPC), are brushite [CaHPO4·2H2O, dicalcium phosphate dihydrate (DCPD)] and monetite [CaHPO4, dicalcium phosphate anhydrous (DCPA)]. DCPD has a comparatively high solubility over calcium phosphate that are used in body. Meanwhile, brushite bone cement, also known as DCPD bone cement authorized use in clinical settings in Europe. Recent research, however, has shown that brushite bone cement breaks down quickly after implantation and that the body transforms the breakdown products into insoluble apatite forms, such HA, which impacts the cement’s ability to heal bone lesions. According to a recent study by Shariff et al.96, β-TCP’s osteoconductivity can be enhanced by applying a suitable quantity of DCPD. Four weeks after procedure, rats showed significant bone growth. Research indicates that monetite has a higher capacity for bone growth and resorption than brushite bone cement.
Bioceramics based on calcium sulfate and silicate
The mineral calcium sulfate, which made up CaSO4·2H2O is found nature as gypsum ore. Two kinds of calcium sulfate hemihydrate, called Plaster of Paris, are formed when calcium sulfate loses water during the calcination process at 110 °C. Dreesman originally reported on the use of calcium sulfate to heal bone deformities in 1959. Calcium sulfate can greatly enhance according to an experiment that used it to fix canine alveolar bone abnormalities. Furthermore, calcium sulfate can enhance osseointegration in vivo and upregulate genes linked to bone formation in vitro. Long-term research has shown that, despite the benefits mentioned above, calcium sulfate degrades too quickly to keep up with the rate at which new bone tissue grows. However, it can also result in negative effects such surface instability and inflammation. In the meanwhile, because of the low mechanical strength, calcium sulfate is unable to give the defect enough long-term mechanical support. To improve its application in tissue engineering, researchers have thus treated calcium sulfate in a number of ways.
Cui et al.97 created composite particles with superior compressive strength by coating compounding of BMP-2. osteoinductivity and strength (as supplied by BMP-2). Furthermore, Hao et al.98 created composite bone cement by combining tricalcium silicate with calcium sulfate, which was utilized to fix a rabbit’s femoral condyle defect. According to in vivo tests, composite bone cement only showed 50% degradation after 12 weeks of implantation, while calcium phosphate bone cement was totally broken down after 8 weeks. New injectable biomaterials can also be made with calcium sulfate. In order to create an injectable and controlled bone healing material, Chen et al.99 added calcium sulfate hemihydrate. The pace at which this substance degraded matched the rate at which new bone tissue grew in the rabbit’s mandibular transplant site. Other than oxygen, which is mostly found as complex silicate or silica, silicon makes up the largest portion of the earth’s crust. At the same time, silicon makes up around 0.026% of body weight, making it vital trace components of human body. In connective tissues like bone and articular cartilage, silicon is crucial. Furthermore, studies have shown that silicon can stimulate osteoblasts’ collagen manufacturing process as well as the growth and development of rat bone. Silicate bioceramics frequently employed for tissue engineering because silicon plays a crucial role in bone development and mineralization. It is noted, although CaSiO3 ceramics contain high rate of dissolution, which raises the pH of the surrounding environment and inhibits cell proliferation, therefore limiting their use in bone tissue engineering. In order to create hardystonite, a novel crystal phase of CaSiO3.
Numerous investigations have demonstrated that a variety of silicate bioceramics, including akermanite (Ca2MgSi2O7), baghdadite (Ca3ZrSi2O9), hardystonite (Ca2ZnSi2O7), and diopside (CaMg-Si2O6). In particular, studies by Gu et al.100 demonstration of akermanite (Ca, Mg, and Si) stimulate ERK pathway, which in turn into fat stem cells. Rats’ supracondylar bone defect was filled by Luo et al.101 using baghdadite (Ca3Zr-Si2O9) and diopside (CaMgSi2O6) in the form of microspheres. According to in vivo tests, the baghdadite microspheres expressed more osteopontin and contained more nascent bone.
Bioactive ceramic-based composite materials
Materials that combine the benefits of biodegradable polymers and biodegradable ceramics are referred to as composites. These composites generally have outstanding mechanical strength, osteogenic properties, osteoconductivity, and biocompatibility. At the same time, these composite materials have emerged as the most promising materials in the field of bone defect repair thanks to innovative fabrication procedures that have arisen in recent years. According to a recent study, a novel collagen/HA hybrid scaffold can promote collagen deposition, upregulate osteogenic gene expression. It is anticipated that this material will be crucial in the healing of bone defects because of another recent study that demonstrated PCL/silicon-substitute may enhance osteoblast adhesion, proliferation while inducing cell development and differentiation. Furthermore, novel materials with enhanced biological qualities that are made by mixing many elements are also appearing. Recently, Lai et al.102 created novel PLGA/TCP/Mg (PTM) using Mg powder, PLGA, and β-TCP to repair bone defect brought on by steroid associated osteonecrosis (SAON).
Effect of environmental changes on biodegradable materials
Biodegradable materials, particularly those employed in biomedical, bioimplant applications, can be greatly impacted by environmental changes. Degradation rate, mechanical integrity, biocompatibility, and toxicity are all impacted by these impacts.
Environmental factors such as pH slow down the rate of degradation and the compounds that are produced. The temperature of each material alters the mechanical stability and speeds up the process. The hydrolysis and corrosion triggers are caused by the moisture content of biodegradable materials. Oxidative degradation or stabilization is produced by oxygen, deterioration is accelerated by enzymes, and corrosion and biofouling are accelerated by salts.
After being implanted in the body, biodegradable materials particularly metallic alloys can react differently to environmental changes such as elevated body temperature (fever, inflammation), and high moisture content (which is constant in human tissues). Unexpected consequences including pain, inflammation, or rapid deterioration may result from these alterations. Pain or discomfort, swelling, warmth, or localized tissue damage are all brought on by elevated body temperatures. There is also a chance of gas accumulation, particularly with magnesium, which can result in pressure or pockets. Excessive moisture causes tissue irritation or cellular stress, pain from quick changes in local chemistry, and early loosening of implants like pins, and screws.
Impact of metal leaching on central nervous system in patients
There is serious concern about metal leaking into the body from biodegradable implants, particularly if it gets to delicate organs like the central nervous system (CNS). Even while the majority of biodegradable metals (such as iron, magnesium, and zinc) are necessary trace elements, their excessive or unregulated release, particularly in susceptible people, can be neurotoxic or disrupt regular brain function.
Because the kidneys control magnesium levels, the effects of magnesium metal leaching on the central nervous system are generally well tolerated. However, too much magnesium can produce confusion, muscle weakness, slow reflexes, hypotension, and in extreme situations, coma. Excess zinc metal leads to oxidative stress and neuronal death, especially following brain injury, convulsions, and stroke. Excess iron leads to brain-related issues such multiple sclerosis, Parkinson’s disease, and Alzheimer’s disease.
Biomaterials a potential immunomodulatory material from translational point of view
Collagen, magnesium, zinc, iron, and other biodegradable materials are examples of biomaterials that can function as immunomodulatory agents in addition to being structural scaffolds from a translational (clinical application) standpoint. This implies that they have the ability to actively influence immunological responses, which is crucial for effective tissue integration, repair, and regeneration. Depending on the therapeutic requirement, immunomodulatory biomaterials can either stimulate or decrease immunological responses by interacting with immune cells (macrophages, dendritic cells, T cells, etc.).
Immunomodulatory biomaterials provide the following benefits: they improve biocompatibility and implant longevity; they speed up healing and tissue regeneration; and they lessen the requirement for systemic immunosuppressants.
Direction of research and prospects for the future
The characteristics, qualities, and functions of biomaterials are thoroughly described in this article. The primary focus of the research has been on the production and medical applications of minerals including iron, zinc, and magnesium. Recent medical experiments revealed biodegradable metals that function better implants, despite fact that biomaterials offer a wide range of potential applications in medicine.
It is necessary to solve challenges presented with development methods. When selecting bio-metals, corrosion behavior, non-toxicity, and biocompatibility are crucial factors. However, it is a fact that the development and use of biomaterials has improved and can continue to promote life’s quality. Medical implants help people with a range of medical conditions by improving comfort and mobility. Furthermore, tissue engineering helps patients bone failure thereby preventing the requirement of implant. The operations could make people feel better about themselves if they wish to appear better. These materials are also utilized in the production of rehabilitative and assistive devices, such as prosthetics, orthotics, and hearing aids.
The currently available clinically qualified bioimplants (Co-Cr or Ti alloys) are permanent and necessitate a second procedure, which puts additional financial and physical burden on patients. Furthermore, the duration of secondary surgery reduces the healthcare system’s total efficacy. Even while Co-Cr and Ti alloys are generally regarded as biocompatible, effects, including as allergic or hypersensitive reactions. This could lead to inflammation, discomfort, and irritation around the implant site. Biomaterials present an opportunity to overcome these constraints. The in vivo performance has been greatly enhanced by the advancement of fabrication techniques. 3D bioprinting, electrospinning, self-assembly, microfabrication, nanotechnology, additive manufacturing, decellularization, and recellularization are some of the most recent methods for creating biomaterials. However, as additive printing techniques enable the integration of different materials with the shared goal of achieving biocompatibility and bioactivity, it is recommended that more attention be paid to them. Customization, design flexibility, quicker prototyping, improved functionality, waste reduction, scalability, and the development of new material characteristics are some advantages additive manufacturing techniques provide for biomaterials. These benefits have the potential to expand healthcare applications and transform the biomaterials industry. Research on biomaterials is anticipated to develop and have a major influence on a number of disciplines in the future, such as engineering, environmental sciences, and medicine. These are a few possible avenues and perspectives for additional research.
Conclusion
Different materials used for bone implant applications are reviewed in this paper. However, the possible toxicity of certain zinc alloys limits their application in biomedicine. Iron (Fe)-based materials are biocompatible and have excellent mechanical properties. Their primary use is the use of biodegradable metal porous scaffolds for the repair of hard tissues, including bones. However, there are disadvantages for biological applications due to their high corrosion resistance. Long-term stability, imaging compatibility, strength-to-weight ratio, osseointegration ability, corrosion resistance, and biocompatibility are all well-known attributes of titanium as a biomaterial. The titanium implants can withstand stress because they are more stiff than natural bone. On the other hand, excessive use or exposure to molybdenum may pose toxicity problems. Investigators were looking for ways to produce larger improvement metals, and alloys is a helpful technique. The Mo has perfect biomaterial for the future, but needed other regularities before it can be used in clinical settings. In conclusion, research on biomaterials is being advanced by interdisciplinary research involves material science, mechanical engineering, doctors and end users. Projects in this kind aim to promote new kind of materials that can address challenging issues in sustainability, engineering, and healthcare. New biomaterials could contain a significant impact on many different fields, leading to innovative medicinal techniques, sustainable technologies, and improved patient outcomes.
Acknowledgement
The author would like to thank to the Prince Mohammad Bin Fahd University for doing the research work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
The sole author was responsible for the conceptualization, methodology, data collection, analysis, writing, and final approval of the manuscript.
References
- Agarwal S, Curtin J, Duffy B, Jaiswal S. Biodegradable magnesium alloys for orthopaedic applications: A review on corrosion, biocompatibility and surface modifications. Sci. Eng. C. 2016; 68: 948–963.
CrossRef - Zhang X, Bos C, Sloof WG, Hovestad A, Terryn H. Comparison of the morphology and corrosion performance of Cr(VI)- and Cr(III)- based conversion coatings on zinc, Coat. Technol. 2005; 199: 92–104.
CrossRef
- Shi ZZ, Gao XX, Chen HT, Liu XF, Li A. Enhancement in mechanical and corrosion resistance properties of a biodegradable Zn-Fe alloy through second phase refinement, Material Science Engineering C 116 (2020) 111197.
CrossRef
- Yuan W, Xia D, Zheng Y, Liu X, Wu S. Controllable biodegradation and enhanced osseointegration of ZrO2-nanofilm coated Zn-Li alloy: in vitro and in vivo studies. Acta Biomaterials. 2020; 105: 290–303.
CrossRef - Wu K, Wang YQ, Zheng MY. Effects of microarc oxidation surface treatment on the mechanical properties of Mg alloy and Mg matrix composites. Material Science Engineering. 2007; 447: 227–232.
CrossRef
- Uo M, Watari F, Yokoyama A, Matsuno H, Kawasaki T. Tissue reaction around metal implants observed by X-ray scanning analytical microscopy. 2001; 22: 677-685.
CrossRef
- Bostman O, Pihlajamaki H. Clinical biocompatibility of biodegradable orthopaedic implants for internal fixation: A Review. 2000; 21: 2615-2621.
CrossRef - Niinomi M, Nakai M, Hieda J. Development of new metallic alloys for biomedical applications. Acta Biomaterials. 2012; 8: 3888–3903.
CrossRef
- Witte F. The history of biodegradable magnesium implants: A review. Acta Biomaterials. 2010; 6: 1680-1692.
CrossRef
- Tsakiris V, Tardei C, Clicinschi FM. Biodegradable Mg alloys for orthopedic implants-A review. Magn. Alloys. 2021; 9: 1884–1905.
CrossRef
- Ghali E, Dietzel W, Kainer KU. General and localized corrosion of magnesium alloys: A critical review. Mater. Eng. Perform. 2004; 13: 7–23.
CrossRef - Elias CN, Meyers MA, Valiev RZ, Monteiro SN. Ultrafine grained titanium for biomedical applications: an overview of performance. Mater. Res. Technol. 2013; 340–350.
CrossRef
- Shi ZZ, Bai WS, Liu XF, Zhang HJ, Yin YX. Significant refinement of coarse (Fe, Mn)Zn13 phase in biodegradable Zn–1Mn-0.1Fe alloy with minor addition of rare earth elements. Char. 2019; 158: 109993.
CrossRef
- Putra NE, Mirzaali MJ, Apachitei I, Zhou J. Multi-material additive manufacturing technologies for Ti-, Mg-, and Fe-based biomaterials for bone substitution. Acta Biomaterials. 2020; 109: 1–20.
CrossRef
- Nair LS, Laurencin CT. Biodegradable polymers as biomaterials. Polymer. Sci. 2007; 32: 762–798.
CrossRef
- Rony L, Lancigu R, Hubert L. Intraosseous metal implants in orthopedics: A review. 2018; 102: 231–242.
CrossRef
- Ghali E, Dietzel W, Kainer KU. General and localized corrosion of magnesium alloys: A critical review. Mater. Eng. Perform. 2004; 13:7–23.
CrossRef
- El-Rashidy AA, Roether JA, Harhaus L, Kneser U. Regenerating bone with bioactive glass scaffolds: a review of in vivo studies in bone defect models. Acta Biomaterials. 2017; 62: 1–28.
CrossRef - Haines NM, Lack WD, Seymour RB, Bosse MJ. Defining the lower limit of a “critical bone defect” in open diaphyseal tibial fractures. Orthop. Trauma. 2016; 30: e158–e163.
CrossRef
- Lopes D, Martins-Cruz C, Oliveira MD, Mano JF. Bone physiology as inspiration for tissue regenerative therapies. 2018; 185: 240–275.
CrossRef
- Bouler JM, Pilet P, Gauthier O, Verron E. Biphasic calcium phosphate ceramics for bone reconstruction: a review of biological response. Acta Biomaterials. 2017; 53: 1–12.
CrossRef
- Barbieri DAJSR, Bruijn JD, Yuan H. Heterotopic bone formation by nano-apatite containing poly (D,L-lactide) composites. Cell Mater. 2010; 19: 252–261.
CrossRef
- Quarto R, Mastrogiacomo M, Cancedda R, Kutepov SM, Mukhachev V. Repair of large bone defects with the use of autologous bone marrow stromal cells, Engl. J. Med., 2001; 344: 385–386.
CrossRef
- Kuhn LT, Liu Y, Boyd NL, Dennis JE. Developmental-like bone regeneration by human embryonic stem cell-derived mesenchymal cells. Tissue Eng. Part A. 2014; 20: 365–377.
CrossRef
- Petersen A, Princ A, Korus G, Ellinghaus A, Leemhuis H. A biomaterial with a channel-like pore architecture induces endochondral healing of bone defects. Nat Comm. 2018; 9(1): 4430.
CrossRef
- Nilsson Hall G, Mendes LF, Gklava C, Geris L, Luyten LP, Papantoniou L. Developmentally engineered callus organoid bioassemblies exhibit predictive in vivo long bone healing, Sci (Weinh). 2020; 7: 1902295.
CrossRef - Hench LL, Polak JM. Third-generation biomedical materials, 20002: 295:1014–1017.
CrossRef - Wei Q, Becherer T, Angioletti-Uberti S, Dzubiella J. Protein interactions with polymer coatings and biomaterials. Chem. Int. Ed. 2014; 53: 8004–8031.
CrossRef
- Das S, Singh S, Singh V, Joung D et al. Oxygenated functional group density on graphene oxide: its effect on cell toxicity. Part Syst. Charact. 30 (2013) 148–157.
CrossRef
- Jayakumar A, Surendranath A, Pv M. 2D materials for next generation healthcare applications. J. Pharm. 2018; 551: 309–321.
CrossRef
- Chang Y, Yang ST, Liu JH, Dong E, Wang Y, Cao A. In vitro toxicity evaluation of graphene oxide on A549 cells. Toxicology Lett. 2011; 200: 201–210.
CrossRef
- Wei C, Liu Z, Jiang F, Zeng B, Huang M, Yu D. Cellular behaviours of bone marrow-derived mesenchymal stem cells towards pristine graphene oxide nanosheets. Cell Prolif. 2017; 50.
CrossRef
- Ricci R, Ncs L, Da-Silva NS, Pacheco-Soares C, Canevari RA, Marciano FR. Graphene oxide nanoribbons as nanomaterial for bone regeneration: Effects on cytotoxicity, gene expression and bactericidal effect. Sci. Eng. C. 2017;78: 341.
CrossRef - Kim JW, Shin YC, Lee JJ, Bae EB, Jeon YC, Jeong CM. The effect of reduced graphene oxide-coated biphasic calcium phosphate bone graft material on osteogenesis. J. Mol. Sci. 2017; 18: 1725.
CrossRef
- Talukdar Y, Rashkow J, Lalwani G, Kanakia S, Sitharaman B. The effects of graphene nanostructures on mesenchymal stem cells. 2014; 35: 4863–4877.
CrossRef
- Chen LQ, Hu PP, Zhang L, Huang SZ, Luo LF, Huang CZ. Toxicity of graphene oxide and multi-walled carbon nanotubes against human cells and zebrafish. Science China Chem. 2012;55: 2209–2216.
CrossRef
- Yoon OJ, Kim I, Sohn IY, Kieu TT, Lee NE. Toxicity of graphene nanoflakes evaluated by cell-based electrochemical impedance biosensing. Biomed Mater. Res. A. 2014; 102: 2288–2294.
CrossRef
- Akhavan O, Ghaderi E, Aghayee S, Fereydooni Y, Talebi A. The use of a glucose-reduced graphene oxide suspension for photothermal cancer therapy. Mater. Chem. 2012; 22: 13773–13781.
CrossRef - Das S, Singh S, Singh V, Joung D, Dowding JM, Reid D. Oxygenated functional group density on graphene oxide: its effect on cell toxicity. Part Syst. Characterization. 2013; 30: 148–157.
CrossRef
- Zhao C, Lu X, Zanden C, Liu J. The promising application of graphene oxide as coating materials in orthopedic implants: preparation, characterization and cell behaviour. Mater. 2015; 10: 015019.
CrossRef
- Mullick CS, Dasgupta S, Mcelroy AE, Sitharaman B. Structural disruption increases toxicity of graphene nanoribbons. Appl. Toxicology. 2014; 34: 1235–1246.
CrossRef - Li B, Zhang XY, Yang JZ, Zhang YJ, Li WX, Fan CH. Influence of polyethylene glycol coating on biodistribution and toxicity of nanoscale graphene oxide in mice after intravenous injection. J. Nanomedicine. 2014; 9: 4697–4707.
CrossRef
- Duch MC, Budinger GR, Liang YT, Soberanes S, Urich D, Chiarella SE. Minimizing oxidation and stable nanoscale dispersion improves the biocompatibility of graphene in the lung. Nano Lett. 2011; 11:5201.
CrossRef
- Zha Y, Chai R, Song Q, Chen L, Wang X, Cheng G. Characterization and toxicological effects of three-dimensional graphene foams in rats in vivo. Nanoparticle Res. 2016; 18, 1–12.
CrossRef
- Mahmoodi M, Hydari MH, Mahmoodi L, Gazanfari L, Mirhaj M. Electrophoretic deposition of graphene oxide reinforced hydroxyapatite on the tantalum substrate for bone implant applications: In vitro corrosion and bio-tribological behaviour. Surface and Coatings Technology. 2021; 424: 127642.
CrossRef - Espiritu J, Berangi M, Cwieka H, Iskhakova K, Kuehne A. Radiofrequency induced heating of biodegradable orthopaedic screw implants during magnetic resonance imaging. Mater. 2026; 25: 86–94.
CrossRef - Li H, Yang H, Zheng Y, Zhou F, Qiu K. Design and characterizations of novel biodegradable ternary Zn-based alloys with IIA nutrient alloying elements, Mg, Ca and Sr. Des. 2015; 83: 95–102.
CrossRef - He X, Li Y, Zou D, Zu H. An overview of magnesium-based implants in orthopaedics and a prospect of its application in spine fusion. Bioactive Materials. 2024; 39: 456–478.
CrossRef
- Kirkland NT, Lespagnol J, Birbilis N, Staiger MP. A survey of bio-corrosion rates of magnesium alloys. Corrosion Sci. 2010; 52: 287–291.
CrossRef
- Witte F. The history of biodegradable magnesium implants: A review. Acta Biomaterials. 2010; 6: 1680–1692.
CrossRef
- Khatun H, Rahman M, Mahmud S, Ali MO, Akter M. Current advancements of hybrid coating on Mg alloys for medical applications. Results in Engineering. 2023; 18: 101162.
CrossRef
- Wang YM, Wang FH, Xu MJ, Zhao B, Guo LX, Ouyang JH. Microstructure and corrosion behavior of coated AZ91 alloy by microarc oxidation for biomedical application. Surf. Sci. 2009; 255: 9124–9131.
CrossRef
- Chen K, Dai J, Zhang X. Improvement of corrosion resistance of magnesium alloys for biomedical applications, Corrosion Reviews. 2015; 33: 101-117.
CrossRef
- Lu C, Ma Y, Li F, Zhu H. Visualization of fast “hydrogen pump” in core–shell nanostructured Mg@Pt through hydrogenstabilized Mg3Pt. Mater. Chem. A. 2019; 7: 14629–14637.
CrossRef
- Suh JS, Suh BC, Bae JH, Kim YM. Machine learning-based design of biodegradable Mg alloys for load-bearing implants. Des. 2023; 225: 111442.
CrossRef
- Yusop AHM, Alsakkaf A, Kadir MRA, Sukmana I, Nur H. Corrosion of porous Mg and Fe scaffolds: A review of mechanical and biocompatibility responses. Corrosion Eng. Sci. Technol. 2021; 56: 310–326.
CrossRef
- Zheng YF, Gu XN, Witte F. Biodegradable metals, Materials Science and Engineering: R. Rep. 2014; 77: 1-34.
CrossRef - Yao Z, Li L, Jiang Z. Adjustment of the ratio of Ca/P in the ceramic coating on Mg alloy by plasma electrolytic oxidation. Surf. Sci. 2009; 255: 6724.
CrossRef
- Lu P, Fan H, Liu Y, Cao L. Controllable biodegradability, drug release behavior and hemocompatibility of PTX-eluting magnesium stents. Colloids Surf. B Biointerfaces. 2011;83; 8.
CrossRef
- Virtanen S. Biodegradable Mg and Mg alloys: Corrosion and biocompatibility, Materials Science and Eng. B. 2011; 176; 8.
CrossRef
- Torne K, Larsson M, Norlin A, Weissenrieder J. Degradation of zinc in saline solutions, plasma, and whole blood, Biomed. Mater. Res. B Appl. Biomater.2016; 104: 1141–1151.
CrossRef
- Su Y, Wang K, Gao J, Yang Y. Enhanced cytocompatibility and antibacterial property of zinc phosphate coating on biodegradable Zinc materials. Acta Biomaterials.2019; 98: 174–185.
CrossRef
- Kraus T, Moszner F, Fischerauer S, Fiedler M. Biodegradable Fe-based alloys for use in osteosynthesis: outcome of an in vivo study after 52 weeks. Acta Biomaterials.2014; 10: 3346–3353.
CrossRef
- Kubasek J, Vojtech D, Jablonska E, Pospísilova I, Lipov J, Ruml T. Structure, mechanical characteristics and in vitro degradation, cytotoxicity, genotoxicity and mutagenicity of novel biodegradable Zn-Mg alloys. Sci. Eng. C. 2016; 58: 24–35.
CrossRef
- Yin YX, Zhou C, Shi YP, Shi ZZ. Hemocompatibility of biodegradable Zn-0.8 wt% (Cu, Mn, Li) alloys, Sci. Eng. C. 2019; 104: 109896.
CrossRef
- Zhou C, Li HF, Yin XX, Shi ZZ, Li T. Long-term in vivo study of biodegradable Zn-Cu stent: a 2-year implantation evaluation in porcine coronary artery, Acta Biomater. 97 (2019) 657–670.
CrossRef
- He J, Li DW, He FL, Liu YY, Liu YL. A study of degradation behaviour and biocompatibility of Zn-Fe alloy prepared by electrodeposition. Sci. Eng. C. 2020; 117: 111295.
CrossRef
- Chen Z, Jin M, He H, Dong J, Li J. Mesenchymal stem cells and macrophages and their interactions in tendon-bone healing. Orthop. Translat. 2023; 39: 63–73.
CrossRef
- Ma J, Zhao N, Zhu D. Endothelial cellular responses to biodegradable metal zinc. ACS Biomater. Sci. Eng. 2015; 1: 1174–1182.
CrossRef
- Drelich AJ, Bowen PK, LaLonde L, Goldman J, Drelich JW. Importance of oxide film in endovascular biodegradable zinc stents. Innovations. 2016; 4: 133–140.
CrossRef
- Li P, Dai J, Li Y, Alexander D, Capek J. Zinc based biodegradable metals for bone repair and regeneration: Bioactivity and molecular mechanisms. Materials Today Bio. 2024; 25: 100932.
CrossRef
- Ulum MF, Arafat A, Noviana D, Yusop AH, Nasution AK, Abdul Kadir MR, Hermawan H. In Vitro and in vivo degradation evaluation of novel ironbioceramic composites for bone implant applications. Sci. Eng. C. 2014; 36: 336–344.
CrossRef
- Chen CZ, Shi XH, Zhang PC, Bai B, Leng YX, Huang N. The microstructure and properties of commercial pure iron modified by plasma nitriding. Solid State Ionics. 2008; 179: 971–974.
CrossRef
- Lin W, Qin L, Qi H, Zhang D, Zhang G, Gao R, Qiu H. Long-term in vivo corrosion behavior, biocompatibility and bioresorption mechanism of a bioresorbable nitrided iron scaffold. Acta Biomaterials. 2017; 54: 454–468.
CrossRef
- Schinhammer M, Gerber I, Hanzi AC, Uggowitzer PJ. On the cytocompatibility of biodegradable Fe-based alloys. Sci. Eng. C. 2016; 33: 782-789.
CrossRef - Orinak A, Orinakova R, Kralova ZO, Turonov AM, Kupkova M, Hrubovcakova W. Sintered metallic foams for biodegradable bone replacement materials. Porous Mater.2014;21:131–140.
CrossRef
- Orinakova R, Gorejova R, Macko J, Orinak A, Kupkova M, Hrubovcakova M, Sevc J, Smith RM. Evaluation of in vitro biocompatibility of open cell iron structures with PEG coating. Surf. Sci. 2019; 475: 515–518.
CrossRef
- Peuster M, Hesse C, Schloo T, Fink C, Beerbaum C. Longterm biocompatibility of a corrodible peripheral iron stent in the porcine descending aorta. 2006; 27: 4955–4962.
CrossRef
- He J, He FL, Li DW, Liu YL, Yin DC. A novel porous Fe/Fe-W alloy scaffold with a double-layer structured skeleton: Preparation, in vitro degradability and biocompatibility. Colloids Surf. B Biointerfaces. 2016; 142: 325–333.
CrossRef
- Khan AR, Zhang HJ, Jun Z, Maosheng Z, Eldin SM. Electrochemical corrosion protection of neat and zinc phosphate modified epoxy coating: a comparative physical aging study on Al alloy 6101. Chem. 2023;11.
CrossRef
- Rabeeh VPM, Hanas T, Enhancing biointerfacial properties of porous pure iron by gold sputtering for degradable implant applications. Today Communications. 2022; 31: 103492.
CrossRef
- Wermuth DP, Paim TC, Bertaco I, Zanatelli C, Naasani LIS, Slaviero M. Mechanical properties, in vitro and in vivo biocompatibility analysis of pure iron porous implant produced by metal injection molding: A new eco-friendly feedstock from natural rubber. (Hevea brasiliensis) Materials Science and Engineering: C. 2021; 131: 112532.
CrossRef - Cheng J, Zheng YF. In vitro study on newly designed biodegradable Fe-X composites (X = W, CNT) prepared by spark plasma sintering, Biomed. Mater. Res. B Appl. Biomaterials.2013; 101B: 485–497.
CrossRef
- Wang SJ, Jiang D, Zhang ZZ, Chen YR, Yang ZD, Zhang JY. Biomimetic nanosilica-collagen scaffolds for in situ bone regeneration: toward a cellfree, one-step surgery. Adv Mater. 2019; 31(49): e1904341.
CrossRef
- Cui ZK, Kim S, Baljon JJ, Wu BM, Aghaloo T, Lee M. Microporous methacrylated glycol chitosan-montmorillonite nanocomposite hydrogel for bone tissue engineering. Nat Commun. 2019;10(1):3523.
CrossRef
- McNamara SL, Rnjak-Kovacina J, Schmidt DF, Lo TJ, Kaplan DL. Silk as a biocohesive sacrificial binder in the fabrication of hydroxyapatite load bearing scaffolds. 2014; 35(25): 6941–53.
CrossRef
- Rotbaum Y, Puiu C, Rittel D, Domingos M. Quasi-static and dynamic in vitro mechanical response of 3D printed scaffolds with tailored pore size and architectures. Sci. Eng. C, Mater. Biol. Appl. 2019; 96: 176–82.
CrossRef
- Toosi S, Naderi-Meshkin H, Kalalinia F, Hosseinkhani H, Heirani-Tabasi A, Havakhah S, et al. Bone defect healing is induced by collagen sponge/ polyglycolic acid. Mater. Sci. Mater. Med. 2019; 30(3): 33.
CrossRef
- Zhang Y, Wang J, Ma Y, Han B, Niu X, Liu J. Preparation of poly(lactic acid)/sintered hydroxyapatite composite biomaterial by supercritical CO2. Biomed Mater Eng. 2018; 29(1): 67–79.
CrossRef - Ge M, Ge K, Gao F, Yan W, Liu H, Xue L. Biomimetic mineralized strontium-doped hydroxyapatite on porous poly (L-lactic acid) scaffolds for bone defect repair. Int J Nanomedicine. 2018; 13: 1707–1721.
CrossRef
- Lai Y, Li Y, Cao H, Long J, Wang X, Li L. Osteogenic magnesium incorporated into PLGA/TCP porous scaffold by 3D printing for repairing challenging bone defect. 2019; 197: 207–19.
CrossRef - Meischel M, Eichler J, Martinelli E, Karr U, Weigel J, Schmöller G. Adhesive strength of bone-implant interfaces and in-vivo degradation of PHB composites for load-bearing applications. Mech. Behavior of Biomed. 2016; 53:104–18.
CrossRef
- Zhang S, Prabhakaran MP, Qin X, Ramakrishna S. Biocomposite scaffolds for bone regeneration: role of chitosan and hydroxyapatite within poly-3- hydroxybutyrate-co-3-hydroxyvalerate on mechanical properties and in vitro evaluation. Mech. Behav. Biomed Mater. 2015; 51: 88–98.
CrossRef - Mermerkaya MU, Doral MN, Karaaslan F, Huri G, Karacavuş S, Kaymaz B. Scintigraphic evaluation of the osteoblastic activity of rabbit tibial defects after HYAFF11 membrane application. Orthop. Surg. Res. 2016; 11(1): 57.
CrossRef
- Brandt J, Henning S, Michler G, Hein W, Bernstein A, Schulz M. Nanocrystalline hydroxyapatite for bone repair: an animal study. Mater. Sci. Mater Med. 2010; 21(1): 283–294.
CrossRef
- Shariff KA, Tsuru K, Ishikawa K. Fabrication of dicalcium phosphate dihydrate-coated β-TCP granules and evaluation of their osteoconductivity using experimental rats. Mater Sci Eng C Mater Biol Appl. 2017; 75: 1411–1419.
CrossRef
- Cui X, Zhang B, Wang Y, Gao Y. Effects of chitosan-coated pressed calcium sulfate pellet combined with recombinant human bone morphogenetic protein 2 on restoration of segmental bone defect. J Craniofac Surg. 2008;19(2):459–65.
CrossRef
- Hao F, Qin L, Liu J, Chang J, Huan Z, Wu L. Assessment of calcium sulfate hemihydrate-Tricalcium silicate composite for bone healing in a rabbit femoral condyle model. Mater Sci Eng C Mater Biol Appl. 2018; 88: 53–60.
CrossRef
- Chen Z, Kang L, Meng QY, Liu H, Wang Z, Guo Z. Degradability of injectable calcium sulfate/mineralized collagen-based bone repair material and its effect on bone tissue regeneration. Mater Sci Eng C Mater Biol Appl.2014; 45: 94–102.
CrossRef
- Gu H, Guo F, Zhou X, Gong L, Zhang Y, Zhai W. The stimulation of osteogenic differentiation of human adipose-derived stem cells by ionic products from akermanite dissolution via activation of the ERK pathway. 2011; 32(29): 7023–7033.
CrossRef
- Luo T, Wu C, Zhang Y. The in vivo osteogenesis of mg or Zr-modified silicate-based bioceramic spheres. Biomed Mater. Res A. 2012;100(9):2269–2277.
CrossRef
- Lai Y, Li Y, Cao H, Long J, Wang X, Li L. Osteogenic magnesium incorporated into PLGA/TCP porous scaffold by 3D printing for repairing challenging bone defect. 2019; 197: 207–219.
CrossRef








