Manuscript accepted on :04-06-2025
Published online on: 19-06-2025
Plagiarism Check: Yes
Reviewed by: Dr. Ramdas Bhat
Second Review by: Dr. Supratim Datta
Final Approval by: Dr. Anton R Keslav
Vandana Esht1* , Abhishek Sharma2
, Abhishek Sharma2 , Marissa Javier Bautista3
, Marissa Javier Bautista3 , Shazia Malik1
, Shazia Malik1 , Ana Maria Escolano-Castillo3
, Ana Maria Escolano-Castillo3 and Jermilyn Tiglao-Adriano3
and Jermilyn Tiglao-Adriano3
1Physical Therapy Department, College of Nursing and Health Sciences, Jazan University, Jazan, Kingdom of Saudi Arabia.
2Department of Physiotherapy, Arogyam Institute of Paramedical and Allied Sciences (Affiliated to H.N.B. Uttarakhand Medical Education University) Roorkee, Uttarakhand, India.
3Nursing Department, College of Nursing and Health Sciences Jazan University, Jazan, 45142, Kingdom of Saudi Arabia.
Corresponding Author E-mail: vandanaesht@jazanu.edu.sa
DOI : https://dx.doi.org/10.13005/bpj/3156
Abstract
The anti-malarial and immunomodulatory drug hydroxychloroquine (HCQ) was seen as a viable option for therapeutic repurposing in coronavirus illness (COVID-19). However, based on clinical research, the concentration-dependent effects of HCQ are inconclusive. Web of Science, PubMed, Embase, MEDLINE, Global Health databases and ClinicalTrials.gov were searched from December 10, 2022, to February 15, 2024. The factors used for eligibility requirements were Population, Intervention, Comparison: pre-post/placebo/standard care, retrospective data or randomized clinical studies. Modified Newcastle-Ottawa Quality Assessment Score and Cochrane criteria was used. Three retrospective studies (386 patients with a baseline and follow up ECG) and four randomized trials (283 patients: HCQ with standard care; azithromycin= 188; standard care/placebo= 95) used HCQ as treatment for hospitalized COVID-19 patients, one randomized trial examined chloroquine (CQ) (81 patients: CQ high dose= 41; low dose CQ=40) to treat hospitalized patients with severe COVID-19 and one case series (98 patients: HCQ= 10; azithromycin= 27; HCQ along with azithromycin = 61) studied HCQ as treatment for hospitalized confirmed/ suspected COVID-19 cases. Findings showed significant prolongation of QTc Interval in 12% to 93% of patients. The administration of HCQ combined with or without azithromycin requires continuous ECG surveillance to prevent risk of cardiotoxicity.
Keywords
Azithromycin; Chloroquine; Coronavirus; Electrocardiogram; QT prolongation
Download this article as:| Copy the following to cite this article: Esht V, Sharma A, Bautista M. J, Malik S, Castillo A. M. E, Adriano J. T. Impact of Repurposed Hydroxychloroquine and Chloroquine on Cardiovascular Health During COVID-19: A Systematic Review. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Esht V, Sharma A, Bautista M. J, Malik S, Castillo A. M. E, Adriano J. T. Impact of Repurposed Hydroxychloroquine and Chloroquine on Cardiovascular Health During COVID-19: A Systematic Review. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/4k7YmbS |
Introduction
SARS-CoV-2 (severe acute respiratory syndrome coronavirus), also called COVID-19, first appeared in late 2019 in Wuhan, China.1 On January 6, 2023, there were 657,977,736 COVID-19 cases, as reported by WHO, including 6,681,433 deaths. A total of 13,073,712,554 vaccine doses have been delivered as of December 21, 2022. Efforts to establish viable treatment techniques for this dreaded pandemic have been continuing. There are currently no particular therapeutics for infection of SARS-CoV-2, and a number of medicinal drugs (e.g., antivirals, antibiotics, inhaled nitric oxide, immune modulators, and convalescent plasma) have been repurposed with negative to equivocal evidence.2 To treat COVID-19, two anti-malarial medications from the amino-quinoline group, namely chloroquine as well as hydroxy-chloroquine) have gained more attention. This is due to their inhibition of other coronaviruses, namely SARS-CoV-1.2,3
Chloroquine raises the pH of acidic vesicles. It is a 9-aminoquinoline that was discovered in 1934. When given extracellularly, the non-protonated chloroquine penetrates the cell and becomes protonated, and concentrates in acidic, low-pH organelles. Its antiviral action depends on how much the virus uses endosomes for entry. Chloroquine has a wide range of effects on virus infection. Without causing noticeably harmful side effects, chloroquine has been used extensively to treat autoimmune disorders, HIV, malaria, and amoebiasis in humans.4 In COVID-19 pneumonia, chloroquine or hydroxychloroquine was evaluated, and it was successful in reducing the duration of the illness. Chloroquine and Remdesivir work well together to treat COVID-19 infection in a lab setting, and they have been used in patients with positive outcomes.5While recent trials in COVID-19 have indicated severe cardiac effects, antimalarials have a tolerable safety profile for treating malaria. The most common cardiac side effect is a lengthening of the QT interval, which raises the risk of sudden death or torsade de pointes. This effect can be brought on by concurrent use of macrolides, as was the case in the early months of the pandemic.6,7 The widespread usage of chloroquine (CQ) or hydroxychloroquine (HCQ) in nations where malaria is endemic may be to blame for the negative correlation among malaria and COVID 19 prevalence. This is due to the possibility that these drugs can treat and prevent COVID 19 infection through three key mechanisms: (i) slowing the progression by decreasing T cell activation and preventing cytokine storm; (ii) altering cellular pH and prevention in viral replication; and (iii) Antiviral activity of CQ in both infected and normal cells, suggesting that HCQ may also have this property. The use of CQ has been discontinued as an anti-malarial in most of Africa in favour of artemisinin-based combination therapy, which has no indication of ineffectiveness.8Antimalarials are not typically recommended by any COVID-19 treatment guidelines, despite the fact that it may appear evident that the evidence against their usage is strong. Instead, actual data seem to support the contrary. Recently, despite their ineffectiveness, Brazil reported using them. In the context of a developing nation, this study offers: a real-life/pharmacovigilance experience with the use of these medications.9 The prospect of utilising CQ in the treatment of SARS-CoV-2 has been vigorously debated in the fields of science, politics, and economics due to recent advancements and potential side effects of the medication discovered in prior attempts to treat acute viral illnesses. The potential contribution of CQ and HCQ to the fight against the virus has undoubtedly been overemphasised, despite some promising early results that were subject to significant restrictions, simplification, and overinterpretation of the data. The effectiveness of CQ and HCQ to treat COVID-19 is not directly supported by any data currently. Despite encouraging in vitro outcomes, the most recent and largest international RCTs for COVID-19 therapies released by WHO found little to no impact on overall mortality, the beginning of ventilation, and the development of a ventilator-associated pneumonia (VAP) score.10Due to the severity of the condition, recent studies have investigated a large fraction of critically sick patients (who finally needed mechanical ventilation, or MV), a population that is particularly vulnerable to drug-induced QT prolongation.11 Additionally, it is well known that pneumonia with a higher degree of severity may itself be a risk factor for QT prolongation.12 Many national papers have recently been produced to instruct clinicians on QT monitoring during this pandemic in order to prevent any potentially fatal drug-induced incident in the setting of COVID-19 sickness.13,14 Prior research has shown that automated QT measurement, or QT generated by electrocardiographic (ECG) devices, is more accurate than manual QT assessment. Nevertheless, the majority of these studies used healthy volunteers.15This systematic review will analyse the benefits and adverse effects of using chloroquine or hydroxychloroquine in COVID 19 cases and association of HCQ alone or combined with Azithromycin (AZT) withQT variation/prolongation48 hr after initiation of HCQ/AZT inthis specific population of SARS-CoV-2.
Materials and Methods
Search strategy
From December 10, 2022, to February15, 2024, searches were conducted in Google Scholar, PubMed, Scopus, Web of Science and ClinicalTrials.gov. Stats data were collected from World Health Organization Official website. The search was constrained to full text papers published in English language. Only completed studies with results were examined at ClinicalTrials.gov. To find additional papers for inclusion, the references of all pertinent studies and reviews were checked.
Study selection and eligibility criteria
Based on the studies titles and abstracts, reviewers separately assessed the search results to find research that might be relevant. The eligibility requirements were considered as we carefully studied and chose the pertinent papers. The factors listed below were used to specify eligibility requirements:
(1) Population: People who has tested positive or is suspected of having COVID-19 (post-exposure), and hospitalised COVID-19 patients.
(2) Antimalarial drug (HCQ intervention).
(3) Results: Suspected or confirmed cases of COVID-19 who are hospitalised: negative conversion of virus, mortality, ECG surveillance, QTc interval changes and any other negative outcomes.
Data Extraction
The authors pulled data from the studies included and cross-checking it to ensure correctness. A standard data extraction sheet was used to pool- registry of study protocol, patient demographics, any pre-existing health conditions, treatment modalities, protocol for administering HCQ, any simultaneous medication, duration of follow-up and outcome data. The following symptoms were also recorded from the data: QTc interval > 500 ms, and other cardiac events
Risk of bias assessment
Risk of bias assessment was approached following Cochrane guidelines for RCTs,16 and the domains evaluated were selection bias, performance bias, detection bias, attrition bias, reporting bias and other bias.
Data synthesis
Mean Difference (MD) or Relative Risk (RR) for dichotomous (negative conversion of virus and adverse events) and continuous variables were described at 95% Confidence Intervals (CI). Although funnel plots have a limited ability to identify effects when there are few trials, they may be effective tools for evaluating meta-analyses for minute study effects.17 The effect sizes and the 95% CI were presented using funnel plots and forest plots, and significance was determined using a 2-tailed p 0.05 using Review Manager version 5.3 for the analyses (Cochrane IMS).
Methodological Quality assessment
Modified Newcastle-Ottawa Quality Assessment Score were assessed for determination of exposure, outcomes and follow ups of all nine studies18.
Dealing with missing data
When possible, the number of patients who completed the follow-up to the moment of outcome assessment was examined as part of the primary analysis for efficacy outcomes. In cases where this wasn’t possible, we investigated the effects of missing data using an intention-to-treat approach. We intended to include all participants receiving at least one dose of either the intervention medicine or a placebo in the safety outcomes. To investigate the effects of missing information on the major outcomes, we have conducted sensitivity analyses.
Results
The search yielded 1,077 potentially relevant records. After evaluation, 30 full-text articles were scrutinized for eligibility and 9 studies included in the review bases on study titles, abstracts and completed trials from ClinicalTrials.gov (Table 1).
Table 1: Summary of included studies (n=9)
| Author (yr) | HCQ dosage | Results | Conclusion |
| Eveleens Maarse BC et al 27 | At t = 0, 12, 24, 48, 72, and 96 hr, Dosage HCQ 400 mg | The RR-, QRS-, and QTcF-intervals of the ECG did not significantly correlate with HCQ (P >0.05) | At plasma concentrations as high as 200 ng/mL, HCQ had no effect on ventricular repolarization, including the QTcF-interval and T-wave shape. |
| Chorin E. et al 19 | HCQ 400 mg BD/1 day; 200 mg BD /4days | In 23% of cases, a severe new QTc interval lengthening to 500 ms | HCQ dramatically lengthens the QTc interval in COVID-19 cases. |
| Tang W. et al 20 | HCQ1200 mgOD/3day; 800 mg OD/ 2-3 weeks | QTc prolongation cardiac arrhythmia | In mild to moderate COVID-19 cases, HCQ did not showed significant probability of negative conversion |
| Borba MGS. et al 21 | HCQ 2.7 g OD /10days; AZT 500g | 28 days of ventricular arrhythmia or QTc > 500 milliseconds | High dose is not suggested for critically sick patients with COVID-19 |
| van den Broek et al22 | 600 mg of CQ OD/5 days | Mean QTc prolongation of 35ms (95% CI 28-43ms) | In a clinically significant way, chloroquine considerably lengthens the QTc interval. |
| Bessière F. et al 23 | HCQ 200 mg BD/10 days | 93% of the patients displayed an elevation in QTc | QTc intervals increased in more than 90% of patients. |
| Ramireddy et al 24 | HCQ and azithromycin | Baseline average QTc of 448±29 ms to rise to 459 ±36 ms (P=0.005) | Critical QTc prolongation was present in 12% of patients overall |
| Cipriani et al 25 | HCQ 200 mg BD/3 days; AZT 500mg | The QTc-interval on the ECG was longer after treatment (p=.02) | Azithromycin and HCQ prolong the QTc interval in COVID-19 patients, especially in those with high transaminase levels. |
| Bun SS et al 26 | HCQ 200 mg TID/10 days | The average automated QTc prolonged after 48 hr | Combination therapy with HCQ/AZT may be given to more than of inpatients who had Lower Respiratory Tract Infection. |
Of the included trials, three were retrospective studies in which HCQ was used as a medication to COVID-19 hospitalized patients along with standard care and ECG was monitored at baseline and post therapy,19-21 six randomized trials included HCQ to treat hospitalized patients with COVID-19.22-27Modified Newcastle-Ottawa Quality Assessment Score of 1 indicated a positive affirmation as an original score of 1 star (Table 2).
Table 2: Modified Newcastle-Ottawa Quality Assessment Score*
| Author (year) | Ascertainmentof exposure | Outcomes Assessment | Length of Follow-up | Loss tofollow-up rate |
| Chorin E. et al 19 | 1 | 1 | 1 | 1 |
| Tang W. et al 20 | 1 | 1 | 1 | 1 |
| Borba MGS. et al 21 | 1 | 1 | 1 | 1 |
| van den Broek MPH et al 22 | 1 | 1 | 1 | 1 |
| Bessière F. et al 23 | 1 | 1 | 1 | 1 |
| Ramireddy A. et al 24 | 1 | 1 | 1 | 1 |
| Cipriani et a l25 | 1 | 1 | 1 | 1 |
| Bun SS et al 26 | 1 | 1 | 0 | 1 |
| Eveleens Maarse BC et al 27 | 1 | 1 | 0 | 1 |
Details of study selection process and specific reasons for exclusion are described as a flow diagram in Figure 1.
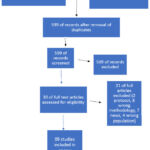 |
Figure 1: Flow of article selectionClick here to view Figure |
HCQ as treatment for hospitalized patients with COVID-19
The studies were carried out in the USA, China, Brazil, France and the Netherlands. Trials enrolled individuals with confirmed or suspected cases of COVID-19 and included critically as well as non-critically ill COVID 19 patients, requiring hospitalization. All trials had a parallel design, classified as Phase 2/ Phase 3 and were registered on ClinicalTrials.gov. Dosage regimens of HCQ were different between studies and QTc Interval was assessed at baseline and up to 2- or 3-days post therapy. Most studies had a low risk for random sequence selection and attrition bias, there was potential risk for allocation concealment, performance, detection, and reporting bias (Figures 2a, 2b).19-27
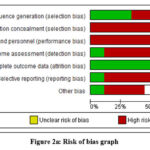 |
Figure 2a: Risk of bias graphClick here to view Figure |
 |
Figure 2b: Risk of bias summary Click here to view Figure |
Statistical heterogeneity was unavailable due to variety of studies included in review. Any meta-analysis result for QTc interval > 500ms was restricted due to inclusion of retrospective studies in review The forest plot shows reduced QTc prolongation in the experimental group, indicating hydroxychloroquine’s relatively safer cardiovascular profile in controlled settings. The x-axis represents effect size (e.g., mean QTc change) and the y-axis shows study precision, typically the inverse standard error (Figures3a, 3b). Tang W. et al (2020) found no difference in negative conversion of virus by using HCQ than the standard of care alone.20 COVID-19 treatment with HCQ and azithromycin enhanced the threat of TdP by raising the likelihood of excessive QTc interval prolongation in a sizable majority of individuals.19 Studies only reported a moderate improvement in QTc Interval after treatment.26 When compared to the other group, a higher dose of CQ in particular increased the occurrence of QTc intervals longer than 500 milliseconds.21
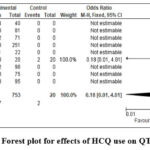 |
Figure 3a: Forest plot for effects of HCQ use on QTc interval.Click here to view Figure |
 |
Figure 3b: Funnel plot for effects of HCQ use on QTc interval.Click here to view Figure |
Studies showing a generalized increase in the probability of unfavorable outcomes in COVID-19 hospitalized patients (QTc> 500ms) reported after HCQ treatment19, 21- 24,26. When given HCQ along with normal therapy, 30% of patients experienced adverse events, compared to 9% of patients in the control group. These occurrences included diarrhea, impaired vision, and other gastrointestinal issues.20Contrary to findings of other studies a randomized controlled trial found no significant association between HCQ concentrations and QTc interval at plasma concentrations as high as 200 ng/mL, hydroxychloroquine27 (Table 1).
Discussion
Globally, the COVID-19 pandemic has a significant negative impact on people, communities, healthcare systems, markets, and governments. There is currently no best method for managing COVID-19, and there are no 100% effective vaccinations to protect against SARS-CoV-2 infection. Several drugs including colchicine, CQ, favipiravir, interferon, ivermectin, lopinavir-ritonavir, ribavirin, remdesivir and HCQ have been studied for their potential to be repurposed for the prevention and treatment of this illness. Observations of reduced viral replication in vitro are the basis for the argument that HCQ should be repurposed,28 however clinical data show different outcomes.29-31 Based on the findings of RCTs and retrospective investigations, we combined the available data in this systematic review and meta-analysis to determine the effectiveness and safety of HCQ for the prevention and treatment of COVID-19. A trial included in this review evaluated the safety and efficacy of high and low CQ dose regime and showed no effect of CQ in COVID-19 management instead, there was a higher risk of adverse drug events. The high-dosage group presented more instance of QTc interval (> 500 milliseconds); henceforth higher dosage of CQ should not be recommended for severe cases.21
Our results support best practices’ conclusions that HCQ is ineffective against COVID-19 at the current state of the art. Hydroxychloroquine (HCQ) has demonstrated effectiveness in vitro against SARS-CoV-2 by altering intracellular pH and disrupting the glycosylation of both the ACE2 receptor and the viral spike protein, thereby inhibiting the virus’s ability to infect host cells.28 Despite encouraging preclinical findings, it has not been proven to be a successful COVID-19 therapy. QTc interval monitoring is clinically essential when administering hydroxychloroquine/chloroquine, as these agents are associated with a risk of QT prolongation, potentially leading to life-threatening arrhythmias. This is particularly critical in populations with pre-existing cardiovascular conditions or those receiving other QT-prolonging drugs. Cautious use, regular ECG monitoring and individualized risk assessment is strongly recommended to mitigate cardiac complications and ensure patient safety during treatment with these repurposed COVID-19 therapies.29,32
The QT interval represents the time taken for the heart’s ventricles to depolarize and then repolarize, and when prolonged, it increases the risk of developing serious arrhythmias, most notably Torsades de Pointes, which can progress to sudden cardiac death.33 HCQ exert their antiviral effects by altering endosomal pH and interfering with viral entry, replication, and immune modulation. However, these drugs also affect cardiac ion channels, particularly by blocking the hERG (human Ether-à-go-go-Related Gene) potassium channels, which play a crucial role in cardiac repolarization.34 This blockade delays repolarization and prolongs the QT interval. COVID-19 patients, especially those with pre-existing cardiovascular disease, electrolyte imbalances (e.g., hypokalemia, hypomagnesemia), renal dysfunction or those receiving other QT-prolonging medications (such as azithromycin), are at even higher risk of adverse cardiac events. Elderly patients and those in critical care settings are particularly vulnerable. In such cases, QTc prolongation can quickly become clinically significant and life-threatening. Routine QTc monitoring—ideally via serial ECGs before, during, and after drug administration—is therefore recommended.35
The current study adds weight to the data that HCQ is ineffective in many clinical contexts and gives crucial information to those who make healthcare decisions. In clinical studies, there had been no link between HCQ treatment and decrease in the frequency, intensity, or duration of COVID-19 symptoms,29-31 nor has the drug’s use been related with a decreased risk of hospitalization in COVID-19 outpatients. Although a medicine specifically for SARS-CoV-2 has not yet been discovered, management techniques and a few drugs have improved patient outcomes. Without any supporting data and in disregard of their negative side effects, a number of medications have been promoted for usage. The US Food and Drug Administration (FDA) and other health organizations have just recently approved the use of remdesivir and tocilizumab for the treatment of hospitalized COVID-19 patients. The symptomatic COVID-19 patients require medical examination and risk factor categorization for worse clinical outcomes.36,37. Analgesics and antipyretics are included in the symptomatic therapy for outpatients. Moreover, precautions to lessen the possibility of SARS-CoV-2 transmission should be taken by isolating confirmed cases. Prone posture and respiratory physiotherapy may be recommended for individuals who have dyspnea.38,39 In order to prevent dehydration, regular fluid consumption should also be encouraged, and walking should be promoted while taking into account the patient’s tolerance. The use of SARS-CoV-2 specific monoclonal antibodies has been investigated for the treatment of out-patients with mild to severe COVID-19.40-41 Clinical trial participants who received HCQ have attained HCQ concentrations that are lower than the goal levels determined by in vitro experiments.31 Nevertheless, the in vivo concentration of HCQ must be high enough for in vivo antiviral activity to be possible, which increase the risk of systemic exposure, adverse effects, and toxicity.42 According to our findings, there was no difference between patients receiving HCQ and a placebo when it came to the negative conversion of virus in the hospital context. There is contradictory evidence about the HCQ effectiveness in COVID-19 according to previous systematic reviews and meta-analyses. A living systematic review that used data from RCTs, cohort studies, and case studies, stated that HCQ can improve respiratory related CT findings with low-quality evidence, without any impact on mortality, the requirement for mechanical ventilation, the severity of progression, symptomatic relief, or the clearance of upper respiratory viruses.43 Nevertheless, a recent study that included findings from both open-label and blinded trials revealed that the impact of HCQ on hospitalization and death was minimal to nonexistent.44
Limitations
Our findings have drawbacks, such as early study termination and increased potential of detection and performance bias. Also, we discovered that non-peer reviewed studies had a significant impact on the quality of the evidence for a few key outcomes. Although the results’ degree of certainty on the absence of a therapeutic benefit for HCQ was limited, we think the genuine impact is probably rather close to the calculated effect.
Clinical Implication
The high dose-response connection between HCQ and significant prolongation of QTc Interval require continuous ECG monitoring and caution to HCQ usage among individuals at higher risk of cardiotoxicity. The study explored the potential benefits of these drugs in combating viral infections. The findings suggested that hydroxychloroquine and chloroquine exhibited promising antiviral properties, raising the possibility of their use in viral treatment strategies. However, the review also highlighted the importance of assessing their impact on cardiovascular health, emphasizing the need for a comprehensive understanding of potential risks and benefits associated with these medications.
The study underscored the intricate balance between antiviral efficacy and cardiovascular considerations, offering insights into the complex interplay between these drugs and overall health outcomes.
Conclusion
The data that is now available, based on the findings of retrospective studies and RCTs, did not demonstrate any therapeutic advantages of HCQ and showed significant prolongation of QTc Interval hence indicating continuous ECG surveillance during the treatment protocol and at follow up.
Acknowledgement
The authors would like to thank Dr. Rohit Sharma for his contribution in editing the final version of manuscript.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article
Conflict of Interests
The author(s) do not have any conflict of interest..
Data Availability Statement
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Riegistration
This research does not involve any clinical trials.
Permission to Reproduce from Other Sources
Not applicable
Author Contributions
- Vandana Esht, Abhishek Sharma: Conceptualization, Methodology, Writing – Original Draft; Data Collection, Analysis, Writing – Review & Editing;
- Marissa J Bautista, Shazia Malik: Data Collection, Analysis, Writing – Review & Editing, Visualization, Supervision, Project Administration;
- Ana Maria Escolano-Castillo, Ermilyn Tiglao-Adriano: Data Collection, Analysis, Writing – Review & Editing, Funding Acquisition, Resources, Supervision.
References
- Das RR, Jaiswal N, Dev N, Jaiswal N, Naik SS, Sankar J. Efficacy and Safety of Anti-malarial Drugs (Chloroquine and Hydroxy-Chloroquine) in Treatment of COVID-19 Infection: A Systematic Review and Meta-Analysis. Front Med (Lausanne). 2020;7:482.
CrossRef - Zhang J, Xie B, Hashimoto K. Current status of potential therapeutic candidates for the COVID-19 crisis. Brain Behav Immun.2020;87(Apr):59-73.
CrossRef
- Vincent MJ, Bergeron E, Benjannet S, Erickson BR, Rollin PE, Ksiazek TG, et al. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J.2005;2(1):1-10.
CrossRef - Savarino A, Boelaert JR, Cassone A, Majori G, Cauda R. Effects of chloroquine on viral infections: an old drug against today’s diseases?Lancet Infect Dis.2003;3(11):722-7.
CrossRef
- Valizadeh R, Dadashzadeh N, Zakeri R, James Kellner SJ, Rahimi MM. Drug therapy in hospitalized patients with very severe symptoms following COVID-19. J Nephropharmacol.2020;9(2):e21.
CrossRef
- Chowdhury MS, Rathod J, Gernsheimer J. A rapidsystematicreview of clinicaltrialsutilizingchloroquine and hydroxychloroquine as a treatment for COVID-19. AcadEmergMed.2020;27(6):493-504.
CrossRef
- Patil VM, Singhal S, Masand N. A systematic review on use of aminoquinolines for the therapeutic management of COVID-19: efficacy, safety and clinical trials. Life Sci.2020;254(Apr):117775.
CrossRef
- Hussein MIH, Albashir AAD, Elawad OAMA, Homeida A. Malaria and COVID-19: unmasking their ties. Malar J.2020;19(1):457.
CrossRef
- Lozano-Cruz OA, Jiménez JV, Olivas-Martinez A, Ortiz-Brizuela E, Cárdenas-Fragoso JL, Azamar-Llamas D, et al.Adverse effectsassociatedwith the use of antimalarialsduringthe COVID-19 pandemic in a Tertiary Care Center in Mexico City. Front Pharmacol.2021;12(Jun):668678.
CrossRef - Gasmi A, Peana M, Noor S, Lysiuk R, Menzel A, Gasmi Benahmed A, et al. Chloroquine and hydroxychloroquine in the treatment of COVID-19: the never-ending story. ApplMicrobiolBiotechnol. 2021;105(4):1333-43.
CrossRef - EtchegoyenCV, KellerGA, MradS, ChengS, Di GirolamoG. Drug-induced QTintervalprolongation in the Intensive Care Unit. CurrClin2017;12(4):210-22.
CrossRef - GoldsteinLH, Gabin A, Fawaz A, Freedberg NA, Schwartz N, Elias Met al.Azithromycin is not associated with QT prolongation in hospitalized patients with community acquiredpneumonia. PharmacoepidemiolDrug 2015;24(10):1042-8.
CrossRef - SacherF, Fauchier L, Boveda S, de Chillou C, Defaye P, Deharo JCet al.Use of drugs with potential cardiac effect in the setting of SARS-CoV-2 infection.Arch Cardiovasc Dis.2020;113(5):293-6.
CrossRef - SappJL, Alqarawi W, MacIntyre CJ, Tadros R, Steinberg C, Roberts JDet al.Guidance on minimizing risk of drug-induced ventricular arrhythmia during treatment of COVID-19: a statement from the Canadian heart rhythm society. Can JCardiol. 2020;36(6):948-51.
CrossRef - AzieNE, Adams G, Darpo B, Francom SF, Polasek EC, Wisser JM, et al.Comparing methods of measurement for detecting drug-induced changes in the QT interval: implications for thoroughly conducted ECG studies. AnnNoninvasElectrocardiol. 2004;2:166-74.
CrossRef - Higgins JPT, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ.2011;343:d5928.
CrossRef - Simmonds M. Quantifying the risk of error when interpreting funnel plots.Syst Rev.2015;4:24.
CrossRef - Basu S, Qayyum H, Mason S. Occupational stress in the ED: a systematic literature review. Emerg Med 2017Jul;34(7):441-7.
CrossRef - Chorin E, Wadhwani L, Magnani S, Dai M, Shulman E, Nadeau-Routhier Cet al.QT interval prolongation and torsade de pointes in patients with COVID-19 treated with hydroxychloroquine/azithromycin. Heart Rhythm.2020Sep;17(9):1425-33.
CrossRef - Tang W, Cao Z, Han M, Wang Z, Chen J, Sun W, et al. Hydroxychloroquine in patients with mainly mild to moderate coronavirus disease 2019: open label, randomised controlled trial. BMJ.2020;369:m1849.
CrossRef - Borba MGS, Val FFA, Sampaio VS, Alexandre MAA, Melo GC, Brito Met al.CloroCovid-19 team. Effect of High vs Lowdoses of ChloroquineDiphosphate as adjunctivetherapy for patientshospitalizedwithsevere acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection: A randomizedclinicaltrial. JAMA Netw Open.2020Apr24;3(4):e208857.
CrossRef - van den Broek MPH, Möhlmann JE, Abeln BGS, Liebregts M, van Dijk VF, van de Garde EMW. Chloroquine-induced QTc prolongation in COVID-19 patients.Neth Heart J.2020Jul;28(7-8):406-9.
CrossRef - Bessière F, Roccia H, Delinière A, Charrière R, Chevalier P, Argaud Let al.Assessment of QTintervals in a caseseries of patientswithcoronavirusdisease 2019 (COVID-19) infectiontreated with hydroxychloroquine alone or in combination with azithromycin in an Intensive Care Unit. JAMA 2020Sep1;5(9):1067-9.
CrossRef - Ramireddy A, Chugh H, Reinier K, Ebinger J, Park E, Thompson Met al.Experience withhydroxychloroquine and azithromycin in the coronavirusdisease 2019 pandemic: implications for QTintervalmonitoring. J Am Heart 2020Jun16;9(12):e017144.
CrossRef - Cipriani A, Zorzi A, Ceccato D, Capone F, Parolin M, Donato Fet al.Arrhythmic profile and 24-hour QT interval variability in COVID-19 patients treated with hydroxychloroquine and azithromycin. Int J 2020Oct1;316:280-4
CrossRef - Bun SS, Taghji P, Courjon J, Squara F, Scarlatti D, Theodore Get al. QTinterval prolongation under hydroxychloroquine/azithromycin association for in patients withSARS-CoV-2 low errespiratory tractinfection. ClinPharmacolTher.2020Nov;108(5):1090-7.
CrossRef - EveleensMaarse BC, Graff C, Kanters JK, van Esdonk MJ, Kemme MJB, In ’t Veld AEet al.Effect of hydroxychloroquine on the cardiac ventricular repolarization: A randomized clinical trial. Br J Clin2022Mar;88(3):1054-62.
CrossRef - Liu J, Cao R, Xu M, Wang X, Zhang H, Hu H, et al. Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov.2020;6:16.
CrossRef - BoulwareDR, Pullen MF, Bangdiwala AS, Pastick KA, Lofgren SM, Okafor EC, et al.A Randomized Trial of Hy- droxychloroquine as Postexposureprophylaxis for Covid-19. N Engl J Med.2020;383(6):517-25.
CrossRef - BarnabasRV, Brown ER, Bershteyn A, StankiewiczKarita HC, Johnston C, Thorpe LE, et al. Hydroxychloroquine as Postexposure prophylaxis to Prevent severe acute respiratory syndrome coronavirus 2 In- fection. Ann Intern Med.2021;174(3):344-52.
CrossRef - Rajasingham R, Bangdiwala AS, Nicol MR, Skipper CP, Pastick KA, Axelrod ML, et al. Hydroxychloroquine as pre–exposure prophylaxis for coronavirusdisease 2019 (COVID-19) in healthcareworkers: A randomized trial. Clin Infect Dis.2021;72(11):e835-43.
CrossRef - Villa Zapata L, Boyce RD, Chou E, Hansten PD, Horn JR, Gephart SM, Subbian V, Romero A, Malone DC. QTc Prolongation with the Use of Hydroxychloroquine and Concomitant Arrhythmogenic Medications: A Retrospective Study Using Electronic Health Records Data. Drugs Real World Outcomes. 2022 Sep;9(3):415-423.
CrossRef - Antoniou CK, Dilaveris P, Manolakou P, Galanakos S, Magkas N, Gatzoulis K, Tousoulis D. QT Prolongation and Malignant Arrhythmia: How Serious a Problem? Eur Cardiol. 2017 Dec;12(2):112-120.
CrossRef - Bansal P, Goyal A, Cusick A 4th, Lahan S, Dhaliwal HS, Bhyan P, Bhattad PB, Aslam F, Ranka S, Dalia T, Chhabra L, Sanghavi D, Sonani B, Davis JM 3rd. Hydroxychloroquine: a comprehensive review and its controversial role in coronavirus disease 2019. Ann Med. 2021 Dec;53(1):117-134.
CrossRef - Schiavone M, Gasperetti A, Gherbesi E, Bergamaschi L, Arosio R, Mitacchione G, Viecca M, Forleo GB. Arrhythmogenic Risk and Mechanisms of QT-Prolonging Drugs to Treat COVID-19. Card Electrophysiol Clin. 2022 Mar;14(1):95-104.
CrossRef - Li J, Huang DQ, Zou B, Yang H, Hui WZ, Rui F, et al. Epidemiology of COVID-19: A systematic review and meta-analysis of clinical characteristics, risk factors, and outcomes. J Med Virol.2021;93(3):1449-58.
CrossRef - Martins-Filho PR, Tavares CSS, Santos VS. Factors associated with mortality in patients with COVID-19. A quantitative evidence synthesis of clinical and lab- oratory data.EurJInternMed.2020;76:97-9.
CrossRef - JaganN, Morrow LE, Walters RW, Klein LP, Wallen TJ, Chung J, et al. The POSITIONED Study: pronePosi- tioning in Nonventilatedcoronavirusdisease 2019 Patients—A Retrospective Analysis. Crit Care Explor.2020;2(10):e0229.
CrossRef - Touchon F, Trigui Y , Prud’homme E , et al. Awake prone positioning for hy- poxaemic respiratory failure: past, COVID-19 and perspectives. EurRespir Rev.2021;30:210022.
CrossRef - US Food and Drug Administration. Fact sheet for health care providersemer- gency use authorization (EUA) of bamlanivimab and etesevimab.https://www; 2021. Available from: http://gov/media/145802/download.
- Regeneron Pharmaceuticals. IncFact sheet for health care providers: emergency use authorization (EUA) of casirivimab and imdevimab. Regeneron2020https://www.regeneron.com/sites/default/files/treatment-covid19-eua-fact-sheet-for-hcp.pdf.
- Fan J, Zhang X, Liu J, Yang Y, Zheng N, Liu Q, et al.Connecting hydroxychloroquinein vitroAntivi- ral Activity to invivo Concentration for Prediction of antiviral Effect: A Crit- iCal Step in Treating Patients withcoronavirusdisease 2019. Clin Infect Dis.2020;71(12):3232-6.
CrossRef - Hernandez AV, Roman YM, Pasupuleti V, Barboza JJ, White CM. Hydroxychloroquine or chloroquine for Treatment or prophylaxis of COVID-19: A Living Systematic Review. Ann Intern Med.2020;173(4):287-96.
CrossRef - KashourZ,Kashour T, Gerberi D, Tleyjeh IM. Mortality, viral clearance, and other clinical outcomes of hydroxychloroquine in COVID-19 patients: A sys- tematic review and meta-analysis of randomized controlled trials. ClinTranslSci.2021;14:1101-12.
CrossRef







