Manuscript accepted on :27-05-2025
Published online on: 30-06-2025
Plagiarism Check: Yes
Reviewed by: Dr. Stephen Adepoju
Second Review by: Dr. Areej Mussab
Final Approval by: Dr. Anton R Keslav
Hiren Kumar Mewada1 , Lingala Syam Sundar2
, Lingala Syam Sundar2 and Nandala Thippa Reddy Ravi Kumar3*
and Nandala Thippa Reddy Ravi Kumar3*
1Electrical Engineering Department, Prince Mohammad Bin Fahd University, Al Khobar, Saudi Arabia.
2Department of Mechanical Engineering, College of Engineering, Prince Mohammad Bin Fahd University, Al-Khobar 31952, Saudi Arabia.
3Department of CSR, Greenko Energies Foundation, Hyderabad, India.
Corresponding Author E-mail: nandalathippareddyravikumar@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3169
Abstract
This paper presents a thorough analysis of the application of convolutional neural networks (CNNs) for sophisticated X-ray fracture classification. Traditional fracture detection and classification methods frequently suffer from limited accuracy and effectiveness, necessitating the investigation of more sophisticated approaches. With their exceptional ability to automatically identify and acquire new characteristics from imaging data, CNNs have become a powerful tool in the field of medical diagnosis. This paper presents open-sourced datasets and their characteristics for bone fracture images, which play a crucial role in classification algorithms. Then, the paper showcases the most recent developments in CNN-based fracture classification, demonstrating gains in speed and accuracy of diagnosis. This work analyzes the resilience, accuracy, and performance of many CNN architectures used for X-ray fracture classification. It is observed that ResNet and ensemble methods demonstrated superior performance relative to conventional CNNs and machine learning algorithms, attaining a maximum accuracy of 94%. Nevertheless, these models are computationally demanding. A significant limitation is that the generalization of these models has not been thoroughly evaluated, which represents a major weakness. Finally, the potential benefits of CNN technology in clinical settings, emphasizing its potential to enhance patient care through faster and more precise diagnosis, have been discussed.
Keywords
Biomedical; Bone Fracture; Classification; CNN analysis; X-ray image
Download this article as:| Copy the following to cite this article: Mewada H. K Sundar L. S, Kumar N. T. R. R. Evaluating the Efficacy of Convolutional Neural Networks for Bone Fracture Detection from X-ray Images. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Mewada H. K Sundar L. S, Kumar N. T. R. R. Evaluating the Efficacy of Convolutional Neural Networks for Bone Fracture Detection from X-ray Images. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/46lMSOo |
Introduction
In order to accurately diagnose diseases, computer-aided systems are crucial. These systems analyze different parts of the body.1 When it comes to detecting bone fractures, healthcare providers have a variety of imaging modalities at their disposal. While traditional X-ray imaging remains the first-line and most widely used technique, several alternative approaches can provide additional information or be useful in specific situations. Computed Tomography (CT) scans, for instance, can offer more detailed, cross-sectional images of the bone, particularly helpful for evaluating fractures in complex anatomical structures like the spine or pelvis. Magnetic Resonance Imaging (MRI) can detect fractures that may not be visible on standard X-rays and provide insights into potential soft tissue injuries. Ultrasound, a portable and relatively inexpensive option, can be valuable for assessing certain fractures close to the skin surface, such as those in the ribs or extremities. Bone scintigraphy, or bone scans, can help identify early-stage fractures not yet apparent on X-rays by detecting areas of increased bone metabolism.
There are various types of bone fractures. The most common is the wrist fracture, which occurs due to a fall on the outstretched hand. This causes a break in the radial part of the bone. Spine fracture is another type of bone fracture that affects the vertebrae of the spinal column. A dislocation and compression of bone also cause spine fractures. The break in the ankle joint is called an ankle fracture, and therefore, the patient cannot walk properly. Similarly, breaks in the hand part include a break in the finger bone or palm’s long bone. Lastly, a major bone fracture among all is a skull fracture, where bone structuring surrounding of brain breaks and causes head injury. Figure 1 shows these all bone fracture’s sample images.
 |
Figure 1: Sample X-ray images presenting bone fracture across various anatomical regions.
|
Although these alternative imaging techniques have their merits, X-ray imaging remains the primary and most widely utilized method for detecting bone fractures. The ability of X-rays to visualize the dense, mineralized bone structure, along with their widespread availability, real-time evaluation capabilities, and relatively low cost, make them an indispensable tool in the assessment and management of skeletal injuries. The high-contrast imaging provided by X-rays allows healthcare providers to readily identify disruptions in bone continuity, accurately diagnose the type and extent of a fracture, and develop an appropriate treatment plan. While traditional X-ray imaging is the cornerstone of fracture detection, recent advancements in machine learning and convolutional neural networks (CNNs) have introduced new opportunities to enhance the accuracy and efficiency of this process. Figure 2 shows the overall structure of bone fracture classification using machine learning and the CNN network. The dataset covers both fractured and non-fractured images of bones. Classifying this model using either machine learning or a CNN network requires a unified image size. So, image resizing is adopted as a pre-processing step. Then, data is split into training and test datasets, where train data is used to train the model and test data are the unknown set of images for the model. The test data is used to evaluate the model performance, measuring its classification accuracy. The machine learning model needs an additional step of feature extraction to discriminate or identify the fracture bone. The CNN network can work directly on images. Many literatures use manual feature extraction and its classification using CNN network.
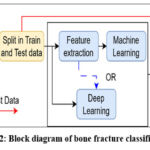 |
Figure 2: Block diagram of bone fracture classification.
|
This paper studies advancements in bone fracture classification, emphasizing on CNN network’s recognition capability. First, it provides a comprehensive overview of existing datasets, critically analyzing their strengths and weaknesses, which serves as a foundational resource for researchers looking to select appropriate data for their studies. Second, the paper systematically discusses various machine learning and convolutional neural network (CNN) models, highlighting their respective advantages and limitations in the context of bone fracture classification. By comparing accuracy rates and methodological approaches, it elucidates the performance gaps in current models. Additionally, the review addresses emerging trends, such as wearable sensor-based approaches, exploring their potential to enhance diagnostic accuracy and patient monitoring. Finally, the paper emphasizes the challenges and limitations faced by existing methodologies, offering insights that can guide future research directions in this evolving field.
Material and Methods
Data set
Recently, various domains of medical science have experienced swift advancements in automated methods for diagnosis, utilizing extensive datasets and sophisticated machine learning algorithms. This section presents some of the datasets which is available publicly and focus on fracture detection.
MURA2 is a compilation of 2D musculoskeletal radiographs comprising 14656 images from various anatomical locations, including the finger, hand, shoulder, elbow, forearm, humerus, and wrist. The dataset has one or more images classified manually by radiologists as either normal or pathological. MedPix3 is an online repository of 2D and 3D medical scans about various disorders for 12000 patients. The content is categorized by disease location (organ system), pathology kind, patient profiles, and image classification along with captions, and it has 59000 images. This dataset covers X-ray, MRI, CT scan and ultrasound images of fracture. Mislabeled images, disorganized annotation, and spam images are some of the problems with the collection.
IIEST4 comprises a modest collection of 2D X-ray pictures totaling 217, including 49 healthy scans, 99 broken scans, and 69 malignant bone scans. This valuable dataset remains inadequate for training deep learning models due to its limited size.
The MOST dataset5 comprises 4,446 X-ray and MRI images that have been annotated utilizing the Kellgren-Lawrence (KL) grading system. This method comprises five categories, from grade 0 to grade 4, with each category indicating a progressively severe image. A newly released dataset, GRAZPEDWRI-DX6, has 20,327 annotated images for localization acquired from 6,091 individuals. A number of pediatric radiologists labeled the photos by using bounding boxes to delineate nine distinct classes. While the dataset is sufficiently vast, it is limited to wrist fractures and does not include any other human body part. Similarly, VinDr-CXR7 includes 18,000 chest X-ray (CXR) images annotated by hand for localization. In this database, 28 distinct chest disorders and anomalies samples are available. This dataset is useful for detecting chest disorders, but it isn’t cut out for detecting bone fractures.
Many other prominent works used a private dataset for fracture region segmentation and classification. Thian et al8 used a dataset consisting of 7356 wrist-fractured images. Later, image augmentation was used to increase images. Similarly, 1102 fractured images and 1156 non-fractured images based on a dataset were used.9 Each image was examined by hand, and a preexisting radiological report was then marked as either normal or a fracture. One major drawback of the current datasets is that they are either not annotated correctly or are only suitable for classification tasks. Additionally, some of them have incorrect labels, making them unfit for use in machine learning applications due to poor maintenance or inadequate annotation quality. Many times, a presence of noise in X-ray images makes is challenging.10
Machine learning and CNN for bone fracture detection
The precise categorization of bone fractures in medical imaging is essential for efficient treatment planning and patient supervision. X-ray imaging is one of the most commonly used diagnostic methods for identifying fractures, which necessitates accurate interpretation to guarantee appropriate medical care. Nevertheless, conventional fracture classification techniques, which significantly rely on radiologists’ knowledge and experience, can be arbitrary and inconsistent.
Results
The steps of the suggested method for detecting bone fractures in x-ray pictures are shown in Figure 3. The preparation of the acquired data is essential because most real-life data is noisy, inconsistent, and incomplete. The first stage in achieving high picture accuracy is image preprocessing, which is followed by further procedures. Therefore, pretreatment techniques are required to eliminate these artifacts prior to additional investigation. Using preprocessing methods like red, green, blue (RGB) to grayscale conversion is the first stage. A Gaussian filter is then used to further remove noise. Figure 4 shows an example of applying noise removal and image smoothing on an x-ray image. Figure 5 indicates an example of applying adaptive histogram on an x-ray image.
 |
Figure 3: X-ray picture of the bone fracture.
|
 |
Figure 4: (a) original image, and (b) Gaussian filter.
|
 |
Figure 5: (a) original image, and (b) adaptive histogram.
|
Precise categorization of bone fractures in medical imaging is essential for efficient treatment planning and patient supervision. X-ray imaging is one of the most commonly used diagnostic methods for identifying fractures, which necessitates accurate interpretation to guarantee appropriate medical care. Nevertheless, conventional fracture classification techniques, which significantly rely on radiologists’ knowledge and experience, can be arbitrary and inconsistent.
Convolutional Neural Networks (CNNs), which are tailored for processing visual input, are a significant breakthrough in the field of deep learning. CNNs are skilled in immediately learning hierarchical representations of features from raw pixel data, a technique inspired by the human visual cortex. Convolutional layers, which use filters to extract local patterns like edges and textures, and pooling layers, which decrease spatial dimensions while preserving crucial information, are important parts of CNN architecture.11 To integrate collected characteristics for final classification or regression tasks, these networks also include fully linked layers.12 CNNs have transformed computer vision applications, paving the way for advances in medical imaging, object identification, and picture recognition. CNNs are essential in the healthcare industry for automating processes of correct identification and categorization of fractures from medical pictures in X-rays.13 Their capacity to extract complex patterns and characteristics from huge datasets has greatly improved the efficiency and accuracy of diagnosis in clinical settings. To further enhance CNNs’ performance in a variety of applications, ongoing research is investigating different architectures and training methods.14
In this section, we explore the performance, strengths, and limitations of various X-ray bone fracture classification approaches, including traditional machine learning techniques, convolutional neural networks (CNNs), residual neural networks (ResNets), Inception-based models, lightweight CNN models, ensemble methods, and semi-supervised/self-supervised learning. We discuss the specific fracture types each approach can detect, their accuracy levels, and the pros and cons of each method.
Rashid et al15 fused long short-term memory (LSTM) and CNN to identify wrist fractures from X-ray pictures. To reduce the number of fractures that go undiagnosed, it provides medical professionals with an additional alternative for diagnosing wrist fractures using computer vision. The dataset from Mendeley includes 192 wrist X-ray images. Fracture detection and classification have seen a dramatic change in direction with the introduction of artificial intelligence (AI) and machine learning (ML) technologies, especially convolutional neural networks (CNNs). A similar approach of integrating CNN and LSTM was presented.16 Model evaluation on 10580 X-ray images showed 97.33% accuracy. However, these approaches have large computational complexities and further optimization of this network is required.
Su et. al.17 developed a model to diagnose bone fractures from X-ray pictures using deep learning algorithms, which has shown considerable potential. The use of deep learning algorithms for various specialized tasks is just one of the hottest directions in bone fracture diagnosis. Another example is how deep learning algorithms have changed over time. They used to use simple bounding boxes, but now they use semantically rich representations that focus on attention mechanisms and heat maps to show fractures in more detail. According to a review by Deo et al18 the use of artificial intelligence and image-processing techniques has produced impressive outcomes in the identification, categorization, and location of human bone fractures. An external force that is far greater than a bone’s tolerance might cause breakage or fissures in the bone, which can result in dislocation that needs intensive care.
A method for diagnosing wrist fractures using deep learning was proposed,19 which employs anatomical landmark localization and image-classification algorithms. It achieved an average accuracy of 0.99. In order to classify orthopedic trauma radiographs, Jakub et al19 first used well-known deep learning networks: BVLC Reference VGG, CaffeNet, and Network-in-network. The results show that AI can do as well as humans. Mewada et al20 presented a quantum-based CNN network for bone fracture classification. They validated the model on a multi-region X-ray dataset, and their model received 96% classification rate.
Discussion
Table 1 provides a comparison of state-of-art using a machine learning algorithm. Linear Discriminant Analysis (LDA) achieved an accuracy of 88.67% using Hough line detection and Harris corner detection for feature extraction, outperforming 12 other classifiers. Support Vector Machine (SVM) demonstrated good performance in detecting fractures from X-ray images but requires extensive feature extraction. Random Forest (RF) provided robust classification accuracy for identifying bone fractures in pediatric patients. K-Nearest Neighbors (KNN) was effective in classifying fracture severity, although it was less accurate in detecting subtle fractures. Finally, the Decision Tree classifier utilized image enhancement techniques and Harris corner detection for feature extraction, achieving an accuracy limited to 82%.
Table 1: Machine learning for bone fracture detection.
|
Methodology |
Focus |
Results /Finding |
|
LDA8 |
Feature extraction using Hough line detection and Harris corner detector |
LDA achieved 88.67% accuracy compared to other 12 machine learning classifier. |
|
SVM9 |
Feature detection from X-ray images |
SVM showed good performance in detecting fracture but require significant features extraction |
|
RF10 |
Classification of bone fracture of pediatric patients |
RF provided robust classification accuracy. |
|
KNN11 |
Classification of fracture severity |
KNN was effective for classifying severity of fracture but less accurate for subtle fractures detection. |
|
Decision tree12 |
Image enhancement using unsharp masking and feature extraction using Harris corner detection. |
Accuracy is limited to 82%. |
Table 2 extends the comparison using CNN networks. The comparison highlights the significant progress made in the field of automated fracture detection and classification using deep learning techniques. The table compares and contrasts different machine learning algorithms for fracture identification based on their accuracy ranges, benefits, and drawbacks. Traditional machine learning algorithms are relatively simple and fast. But these models may struggle in finding crucial features, and hence, they have limited generalization ability. In comparison, CNN networks outperformed the machine learning model with the largest classification accuracy of 95%. However, this performance was achieved at the cost of large computational demand. In addition, tuning these models is a major challenge.
Table 2: A CNN models for fracture detection.
|
Method |
Range of Accuracy |
Pros and Cons |
|
Traditional machine learning algorithms.13,14 |
70-85% |
Pros: Relatively simple to implement, interpretable model Cons: require manual feature engineering, struggle with complex feature patterns, limited generalization |
|
CNNs.15,16,17 |
85-95% |
Pros: automated feature extraction, high accuracy, ability to generalized to diverse feature types. |
|
ResNets.18,19,20,21 |
88-94% |
Proc: deeper architecture, mitigate vanishing gradient, efficient and fast inference Cons: potentially more complex to train, still require substantial training |
|
Inception/Xception based model.22,23,24 |
87-93% |
Pros: capture multi-scale features, performed well with limited data. Cons: Large computation compared to CNNs, it may require more tuning |
|
Lightweight CNNs (i.e. MobileNet, EfficientNet).25,26,27,28 |
82-90% |
Pros: Efficient for deployment on mobile /edge computing devices, good balance between accuracy and speed. Cons: Slightly lower accuracy compared to CNNs |
|
Ensemble approaches.29,30,31,32 |
90-95% |
Pros: combine strength of multiple models, improved robustness and accuracy Cons: increased complexity and it requires more computational resources. |
Traditional machine learning approaches, while relatively simple to implement, struggle with complex fracture patterns and have limited generalization capabilities. The introduction of CNNs and their variants, such as ResNets and Inception-based models, has significantly improved the accuracy of fracture detection, with the ability to handle a wide range of fracture types, including simple and complex long bone, vertebral, and pelvic fractures.
One key advantage of the deep learning-based approaches is their ability to automatically extract relevant features from the X-ray images, eliminating the need for manual feature engineering. This has led to improved performance and better generalization across diverse fracture patterns. However, these models typically require large labeled datasets for effective training, which can be a challenge in the medical imaging domain. Lightweight CNN models, such as MobileNet and EfficientNet, have shown promising results in terms of balancing accuracy and computational efficiency, making them suitable for deployment on mobile or embedded devices. Ensemble methods, which combine the strengths of multiple models, have demonstrated further improvements in accuracy and robustness. More recently, semi-supervised and self-supervised learning techniques have emerged as a way to leverage unlabeled data and reduce the reliance on manual annotations, which can be time-consuming and expensive. These approaches have shown encouraging results, although they may not yet reach the peak performance of fully supervised methods.
CNNs have become a powerful tool for automating the categorization of fractures in medical pictures, particularly X-Rays. Radiologists’ manual evaluation, which can be laborious and inconsistent, has historically been the primary method for classifying fractures. By automatically identifying characteristics from X-ray pictures and categorizing fractures based on discovered patterns, CNNs provide a more effective and dependable substitute.33 With outstanding accuracy, these networks can discriminate between several fracture forms, including transverse, oblique, and comminuted fractures. Extensive collections of annotated medical pictures train CNNs, enabling them to identify subtle fracture patterns and abnormalities that may be difficult for humans to see on their own. This skill improves fracture classification consistency and expedites the diagnostic procedure, which benefits patient care and treatment planning. CNN technology is progressing with the integration of transfer learning and ensemble approaches, and it has great promise for improving fracture classification robustness and accuracy in clinical practice.34
Extensive research on convolutional neural networks (CNN) for enhanced X-ray fracture classification produced important results about the resilience, accuracy, and performance of the model. The CNN models outperformed conventional approaches in terms of speed and precision exhibiting a high degree of accuracy in categorizing different types of bone fractures. Table 3 shows the different types of fractures and methods used. It provides a comprehensive overview of recent studies focusing on the classification of various types of bone fractures using advanced convolutional neural network (CNN) methodologies.
Table 3: CNN model for fracture detection.
|
Fracture type |
Method |
Classification accuracy |
|
Various bone fractures46 |
CNN |
94% |
|
Wrist fracture47 |
CNN + Transfer learning |
92% |
|
Spine fracture48 |
CNN-based ensemble learning |
96% |
|
Ankle fractures49 |
CNN + Random Forest (RF) |
90% |
|
Hand fractures50 |
ResNet50 |
94.9% |
|
Skull fractures51 |
CNN + Capsule Networks |
94% |
|
Thigh bone fracture52 |
Vision transformer |
87% |
|
MURA dataset53 |
Vision transformer + CNN |
88% |
The findings showed that CNNs are especially adept at spotting intricate fracture patterns that traditional approaches frequently overlook. The hybrid and ensemble models demonstrated commendable performance, achieving an identification accuracy of 94%. The identification of fractures in wrist bones is confined to 92% accuracy. Conversely, fractures in other bone types were accurately recognized. The Vision Transformer (ViT) represents a recent advancement in convolutional neural networks (CNNs), leveraging the principles of transformer architecture to improve image classification tasks. In the context of thigh bone fracture classification, a study utilizing a ViT achieved an accuracy of 87%, demonstrating its potential in effectively identifying fractures. Furthermore, when combined with traditional CNN techniques, the hybrid model attained an even higher accuracy of 88% on the MURA dataset. The improved performance was mostly due to CNN’s capacity to recognize and extract complex information from X-ray pictures. Additionally, the models demonstrated a high degree of resilience across several datasets, retaining accuracy in spite of changes in patient demographics and picture quality. When it comes to fracture classification, CNNs have several advantages, one of which is their ability to automate the detection process. This minimizes human error and reduces the burden on radiologists. Faster diagnosis is another benefit of the automated method, which is crucial in clinical situations when prompt treatment is required.54 The conversation also highlighted the essentiality of large, diverse datasets for training effective CNN models. We must include a range of fracture types, patient ages, and imaging conditions to ensure the models translate effectively to actual clinical situations.
Wearable technologies are the new advancement in capturing fracture recovery progression.55 North et al56 showed how wearable sensors can be used in predicting the fracture healing process. Machine learning models were trained using steps and extract matrices and evaluated with -fold cross-validation. They found a model accuracy range from 74% to 79%. A wearable sensor was utilized to examine the efficacy (ideal healing) and efficiency (cost) of a treatment during the actual healing of a bone fracture. The relevant data, including stress, deformation, conductivity, and acceleration, were recorded and transformed. The author used three separate classification tests. i.e., random forest, naive Bayes, and KNNs. Among all the models, random forest fared the best.
In summary, the results highlight CNNs’ potential to transform medical imaging, especially in the area of classifying bone fractures. CNN’s sophisticated features not only improve diagnosis precision but also open the door to more dependable and effective healthcare delivery. Future research should focus on resolving highlighted issues, refining model designs, and investigating the integration of multimodal data to further enhance the efficacy of CNN-based fracture classification systems.
it is crucial to note that effective fracture healing and treatment are equally important in the clinical context. The integration of wearable technologies with CNN models offers promising avenues for monitoring recovery and optimizing treatment plans, yet this integration remains in demand and requires further study. While modern methods demand more computational resources and complexity in training, their superior performance benefits make them preferable for real-world applications. As technology continues to evolve, leveraging these advanced models will be crucial for improving diagnostic accuracy and efficiency in medical imaging, ultimately leading to better patient outcomes and enhanced care in fracture management
Conclusion
This thorough review highlights the substantial developments and promise of CNN in the field of X-ray fracture classification. The enhanced feature extraction and learning capabilities of CNNs significantly increase fracture detection efficiency and accuracy when compared to conventional techniques and machine learning algorithms. It is noted that machine learning models were limited to 85% classification rate. In contrast, advanced techniques like Convolutional Neural Networks (CNNs) and ResNets demonstrate significantly higher accuracy, with ranges of 85-95% and 88-94%, respectively. Federated learning presents a promising approach for bone fracture classification, enabling the training of machine learning models across decentralized data sources while enhancing model performance and preserving patient privacy and data security. It enables the incorporation of diverse datasets from multiple institutions, thereby enhancing the model’s generalizability and robustness in real-world clinical settings.
Through a thorough analysis of several CNN architectures and their uses in medical imaging, this paper emphasizes how crucial it is to optimize model training, hyperparameter tuning, and data preparation to achieve optimal performance. CNN integration into clinical practice has the potential to completely transform patient care by enhancing treatment results, allowing for rapid and accurate diagnosis. However, despite the encouraging results, we still need to overcome a number of obstacles to guarantee generalizability across a range of patient populations. These obstacles include the requirement for sizable annotated datasets, the possibility of model overfitting, and the requirement for reliable validation techniques. Next studies need to focus on getting around these problems, looking into new CNN structures, and adding extra data types to make CNNs better at diagnosing fractures. The medical community may look forward to a future when AI-driven tools play a significant role in improving the accuracy, efficiency, and overall quality of patient care in orthopedic diagnostics by continuing to enhance and refine these technologies.
Acknowledgement
The author would like to thank to the Prince Mohammad Bin Fahd University for doing the research work.
Funding source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Hiren Kumar Mewada: Conceptualization, Methodology, Writing – Original Draft.
- Lingala Syam Sundar: Data Collection, Analysis, Writing – Review & Editing.
- Nandala Thippa Reddy Ravi Kumar: Visualization, Supervision, Project Administration
References
- Limbasia B, Thakkar HK, and Mewada H. Optimized predictive algorithms for augmented intelligence in personalized e-health. Health 5.0, 1st Edition, CRC Press, 2025; 183–201.
CrossRef - Rajpurkar P, Irvin J, Bagul A, Ding D, Duan T, Mehta H, Yang B. MURA: Large Dataset for Abnormality Detection in Musculoskeletal Radiographs. 2017, doi: 10.48550/ARXIV.1712.06957.
- Bone Fracture MedPix, The National Library of Medicine. https://medpix.nlm.nih.gov/home, https://medpix.nlm.nih.gov/home.
- Yadav DP, and Rathor S. Bone fracture detection and classification using deep learning approach. International Conference on Power Electronics & IoT Applications in Renewable Energy and its Control (PARC), Mathura, India: IEEE, Feb. 2020; 282–285.
CrossRef - Sheehy L. Does measurement of the anatomic axis consistently predict hip-knee-ankle angle (HKA) for knee alignment studies in osteoarthritis? Analysis of long limb radiographs from the multicenter osteoarthritis (MOST) study. Osteoarthritis and Cartilage, 2011; 19: 8–64.
CrossRef - Nagy E, Janisch M, Hržić F, Sorantin E, Tschauner S. A pediatric wrist trauma X-ray dataset (GRAZPEDWRI-DX) for machine learning. Sci Data, 2022; 9: 222.
CrossRef - Nguyen HQ. VinDr-CXR: An open dataset of chest X-rays with radiologist’s annotations. Sci Data, 2022; 9: 429.
CrossRef - Thian YL, Li Y, Jagmohan P, Sia D, Chan VEY, Tan RT. Convolutional neural networks for automated fracture detection and localization on wrist radiographs. Radiology: Artificial Intelligence, 2019; 1: e180001.
CrossRef - Raisuddin AM. Critical evaluation of deep neural networks for wrist fracture detection. Scientific Report, 2021; 11, 6006.
CrossRef - Al-Asad JF, and Mewada, H. Fourier synchrosqueezed transform when used for medical OCT image enhancement. 6th International Symposium on Advanced Electrical and Communication Technologies (ISAECT), Alkhobar, Saudi Arabia: IEEE, Dec. 2024, 1–6.
CrossRef - Sarvamangala Dr, and Kulkarni RV. Convolutional neural networks in medical image understanding: a survey. Intel., 2022; 15:1–22.
CrossRef - Ahmed KD, and Hawezi R. Detection of bone fracture based on machine learning techniques. Measurement: Sensors, 2023; 27: 100723.
CrossRef - Yamashita, R, Nishio M, Do RKG, Togashi K. Convolutional neural networks: an overview and application in radiology. Insights Imaging, 2018; 9: 611–629.
CrossRef - Kourounis G, Elmahmudi AA, Thomson B, Hunter J, Ugail H, Wilson C. Computer image analysis with artificial intelligence: a practical introduction to convolutional neural networks for medical professionals. Postgraduate Medical Journal, 2023; 99: 1287–1294.
CrossRef - Rashid T, Zia MS, Meraj T, Rauf HT, Kadry S. A minority class balanced approach using the DCNM-LSTM method to detect human wrist fracture. 2023; 13: 133.
CrossRef - Mewada H, Al-Asad JF, Patel H, Mohammed N. Leveraging spatial and temporal features using CNN-LSTM for improved bone fracture classification from X-ray images. 6th International Symposium on Advanced Electrical and Communication Technologies (ISAECT), Alkhobar, Saudi Arabia: IEEE, Dec. 2024, 1–5.
CrossRef - Su Z, Adam A, Nasrudin MF, Ayob M, Punganan G. Skeletal fracture detection with deep learning: A comprehensive review. 2023; 13: 3245.
CrossRef - Deo G, Totlani J, Mahamuni CV. A survey on bone fracture detection methods using image processing and artificial intelligence (AI) approaches. AIP Conference Proceedings.2024; 2802: 1.
CrossRef - Olczak J, Fahlberg N, Maki A, Razavian AS, Jilert A, Stark A, Skoldenberg O, Gordon M. Artificial intelligence for analyzing orthopedic trauma radiographs: Deep learning algorithms-are they on par with humans for diagnosing fractures? Acta Orthopaedica. 2017; 88: 581–586.
CrossRef - Mewada H, Pires IM, Rahevar M, Khatri N. Quantum convolutional neural network for bone fracture classification from X-Ray images, Procedia Computer Science, 2025; 256, 1143–1150.
CrossRef - Sahin ME. Image processing and machine learning based bone fracture detection and classification using X-ray images. Int. J. Imaging Systems and Technology. 2023; 33: 853–865.
CrossRef - Bagaria R, Wadhwani S, Wadhwani AK. Bone fractures detection using support vector machine and error backpropagation neural network. Optik. 2021; 247: 168021.
CrossRef - Randsborg PH, and Sivertsen EA. Classification of distal radius fractures in children: good inter-and intraobserver reliability, which improves with clinical experience. BMC Musculoskeletal Disorders. 2012; 13: 1–8.
CrossRef - Mujoomdar M, Russell E, Dionne F, Moulton K, Murray C, McGill S, Lambe K. Diagnosis of fracture, optimizing health system use of medical isotopes and other imaging modalities [Internet]. Canadian Agency for Drugs and Technologies in Health, 2012.
- Myint WW, Tun KS, Tun HM, Myint H. Analysis on leg bone fracture detection and classification using X-ray images. Machine Learning Research, 2018; 3: 49–59.
CrossRef - Urakawa T, Tanaka Y, Goto S, Matsuzawa H, Watanabe K, Endo N. Detecting intertrochanteric hip fractures with orthopedist-level accuracy using a deep convolutional neural network. Skeletal Radiology. 2019; 48: 239–244.
CrossRef - Lindsey R, Daluiski A, Chopra S, Lachapelle A, Mozer M, Sicular S, Hanel D, Gardner M, Gupta A, Hotchkiss R et al. Deep neural network improves fracture detection by clinicians. Proceedings of the National Academy of Sciences. 2018; 115: 11591–11596.
CrossRef - Small J, Osler P, Paul A, Kunst M, CT cervical spine fracture detection using a convolutional neural network. American J. Neuroradiology. 2021; 42: 1341–1347.
CrossRef - Yeh LR, Zhang Y, Chen JH, Liu YL, Wang AC, Yang JY, Yeh WC, Cheng CS, Chen LK, Su MY. A deep learning-based method for the diagnosis of vertebral fractures on spine MRI: Retrospective training and validation of ResNet. European Spine J. 2022; 31: 2022–2030.
CrossRef - Naeem S, Naseer A, Rehman S, Gruhn V, Akram S. Enhancing finger fracture diagnosis: A deep learning approach using ResNet and VGG, https://doi.org/10.20944/preprints202311.1990.v1
CrossRef - Karanam SR, Srinivas Y, Chakravarty S. A systematic approach to diagnosis and categorization of bone fractures in X-ray imagery. J. Healthcare Management. 2022; 1–12.
CrossRef - Kim D, and MacKinnon T. Artificial intelligence in fracture detection: Transfer learning from deep convolutional neural networks. Clinical Radiology. 2018; 73: 439–445.
CrossRef - Abbas W, Adnan SM, Javid MA, Majeed F, Ahsan T, Hassan SS et al. Lower leg bone fracture detection and classification using faster RCNN for X-rays images. IEEE 23rd Multitopic Con. (INMIC). 2020; 1–6.
CrossRef - Mallam P, Mehta A, Singh B. Bone fracture classification comparison of ResNet, DenseNet, inception V3 with ADAM optimizer & mobile net. AIP Conference Proceedings.2024; 2900: 1.
CrossRef - Cheng CT, Ho TY, Lee TY, Chang CC, Chou CC, Chen CC, Chung I, Liao CH. Application of a deep learning algorithm for detection and visualization of hip fractures on plain pelvic radiographs. European radiology, 2019; 29: 5469– 5477.
CrossRef - El-Saadawy H, Tantawi M, Shedeed HA, Tolba MF. A two-stage method for bone X-rays abnormality detection using MobileNet network. Proceedings of the Int. Conference on Artificial Intelligence and Computer Vision (AICV2020). Springer. 2020; 372–380.
CrossRef - Mittal K, Gill KS, Chauhan R, and Kapruwan A. Innovative fracture diagnosis: MobileNet CNN approach for precise bone fracture detection and classification. Conference on Intelligent Systems for Cybersecurity (ISCS). 2024; 1–5.
CrossRef - Luong HH, Le LTT, Nguyen HT, Hua VQ, Nguyen KV, Bach KNP, Nguyen TNA, Nguyen HTQ. Transfer learning with fine-tuning on mobilenet and grad-cam for bones abnormalities diagnosis. Computational Intelligence in Security for Information Systems Conference. Springer. 2022; 171–179.
CrossRef - Mahendran S, and Baboo SS. Ensemble systems for automatic fracture detection. J. Engineering and Technology. 2012; 4: 7.
CrossRef - Badgeley MA, Zech JR, Oakden-Rayner L, Glicksberg BS, Liu M, Gale W, McConnell MV, Percha B, Snyder TM, Dudley JT. Deep learning predicts hip fracture using confounding patient and healthcare variables. NPJ Digital Medicine. 2019; 2: 31.
CrossRef - Liu Q, Cui X, Chou YC, Abbod MF, Lin J, Shieh JS. Ensemble artificial neural networks applied to predict the key risk factors of hip bone fracture for elders. Biomedical Signal Processing and Control. 2015; 21: 146–156.
CrossRef - Kilic N, and Hosgormez E. Automatic estimation of osteoporotic fracture cases by using ensemble learning approaches. Medical Systems. 2016; 40: 1–10.
CrossRef - Li Q, Cai W, Wang X, Zhou Y, Feng DD, Chen M. Medical image classification with convolutional neural network. 13th International Conference on Control Automation Robotics & Vision (ICARCV). IEEE, 2014; 844–848.
CrossRef - Yadav SS, and Jadhav SM. Deep convolutional neural network based medical image classification for disease diagnosis. Big Data.2019; 6: 1–18.
CrossRef - Thaiyalnayaki K, Kavyaa K, and Sugumar J. Automated bone fracture detection using convolutional neural network. Physics: Conference Series.2023; 2471: 012003.
CrossRef - Yadav SS, and Jadhav SM. Wrist fracture prediction using transfer learning, a case study. Population Therapeutics and Clinical Pharmacology.2023; 30: 1050–1062.
- Javed R, Abbas T, Janjua JI, Muhammad MA, Ramay SA, Basit MK. wrist fracture prediction using transfer learning, a case study. JPTCP, 2023, doi: 10.53555/jptcp.v30i18.3161.
CrossRef - Maki S, Furuya T, Inoue M, Shiga Y, Inage K, Eguchi Y, Orita S, Ohtori S. Machine learning and deep learning in spinal injury: A narrative review of algorithms in diagnosis and prognosis. Clinical Medicine. 2024; 13: 705.
CrossRef - Kitamura G, Chung CY, Moore BE. Ankle fracture detection utilizing a convolutional neural network ensemble implemented with a small sample, denovo training, and multi view incorporation. Digital Imaging. 2019; 32: 672–677.
CrossRef - Ureten K, Sevinc HF, Igdeli U, Onay A, Maras Y. Use of deep learning methods for hand fracture detection from plain hand radiographs. Turkish Journal of Trauma & Emergency Surgery. 2022; 28: 196.
- Emon MM, Ornob TR, and Rahman M. Predicting skull fractures via CNN with classification algorithms. Proceedings of the 2nd Conference on Computing Advancements. 2022; 442–449.
CrossRef - Islam MU, Fathima A, Ghosh D. An improved technique for bone fracture classification using visual transformers and convolutional neural networks. SSRN Journal, 2025, doi: 10.2139/ssrn.5191555.
CrossRef - Sharma S. Artificial intelligence for fracture diagnosis in orthopedic X-rays: Current developments and future potential. SICOT-J. 2023; 9.
CrossRef - Gabriel CL. Mobile and wearable technologies for the analysis of Ten Meter Walk Test: A concise systematic review. Heliyon, 2023; 9: e16599.
CrossRef - North K, Simpson G, Geiger W, Cizik A, Rothberg D, Hitchcock R. Predicting the healing of lower extremity fractures using wearable ground reaction force sensors and machine learning. Sensors, 2024; 24: 5321.
CrossRef - Misic D, Zdravkovic M, Mitkovic M, Vitkovic N, Mitkovic M. Real-time monitoring of bone fracture recovery by using aware, sensing, smart, and active orthopedic devices. IEEE Internet Things J. 2018; 5: 4466–4473.
CrossRef
Abbreviations List
|
AI |
Artificial Intelligence |
|
CNN |
Convolutional Neural Networks |
|
CT |
Computed Tomography |
|
EfficientNet |
EfficientNet |
|
KNN |
k-nearest neighbors |
|
LDA |
Linear Discriminant Analysis |
|
LSTM |
Long Short-Term Memory |
|
ML |
Machine Learning |
|
MobileNet |
MobileNet |
|
MRI |
Magnetic Resonance Imaging |
|
ResNets |
Residual Neural Networks |
|
RF |
Random Forest |
|
SVM |
Support Vector Method |







