Manuscript accepted on :23-04-2025
Published online on: 29-05-2025
Plagiarism Check: Yes
Reviewed by: Dr. B Kirthika
Second Review by: Dr. Ramya Rachamanti
Final Approval by: Dr. Gul Ozcan
Margret Chandira Rajappa1* , Dominic Antonysamy2
, Dominic Antonysamy2 , Karthikeyan Muthumani1
, Karthikeyan Muthumani1 , Vignesh Vezhaventhan1
, Vignesh Vezhaventhan1 , Sanjay Gnanamoorthi1
, Sanjay Gnanamoorthi1 , Ranjithkumar Sivaji1
, Ranjithkumar Sivaji1 , Sowmiya Subbiah1
, Sowmiya Subbiah1 , Visali Ravichandran1
, Visali Ravichandran1 and Jecintha Margaret Antonysamy1
and Jecintha Margaret Antonysamy1
1Department of Pharmaceutics, Vinayaka Mission’s College of Pharmacy, Vinayaka Mission’s Research Foundation (Deemed to be University), Yercaud main road, Salem, Tamil Nadu, India.
2Faculty of Engineering, Sona College of Technology, Salem, Tamil Nadu, India.
Corresponding Author E-mail: mchandira172@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3160
Abstract
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) syndrome represents a rare yet grave consequence of medication usage, among which mefenamic acid, a widely utilized nonsteroidal anti-inflammatory drug (NSAID), stands as a notable culprit. This review carefully examines the complex relationship between mefenamic acid and DRESS disease, exploring the subtleties of its epidemiology, underlying pathophysiological mechanisms, diverse clinical manifestations, diagnostic challenges, therapeutic modalities, prognostic implications, and preventive strategies. This article aims to strengthen medical community awareness, identification, and effective therapy of mefenamic acid-induced DRESS syndrome by combining current knowledge with emerging research initiatives.
Keywords
DRESS Syndrome; Epidemiology; Mefenamic Acid; NSAID; Pathophysiological Mechanisms; Preventive Strategies
Download this article as:| Copy the following to cite this article: Rajappa M. C, Antonysamy D, Muthumani K, Vezhaventhan V, Gnanamoorthi S, Sivaji R, Subbiah S, Ravichandran V, Antonysamy J. M. DRESS Syndrome: A Comprehensive Review of Its Clinical and Systemic Complexities. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Rajappa M. C, Antonysamy D, Muthumani K, Vezhaventhan V, Gnanamoorthi S, Sivaji R, Subbiah S, Ravichandran V, Antonysamy J. M. DRESS Syndrome: A Comprehensive Review of Its Clinical and Systemic Complexities. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/3SqpK9u |
Introduction
The relationship between mefenamic acid and Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) syndrome represents a captivating yet daunting terrain for clinicians and researchers alike. Among the extensive array of medications implicated in this syndrome, mefenamic acid, a prevalent nonsteroidal anti-inflammatory drug (NSAID), emerges as a significant protagonist. Despite its widespread utilization for pain relief and inflammatory conditions, mefenamic acid’s association with DRESS syndrome underscores the imperative for heightened vigilance and comprehensive understanding among healthcare providers. 1 This review aims to unravel the intricate dynamics between mefenamic acid and DRESS syndrome, elucidating its multifaceted dimensions ranging from epidemiology to preventive interventions. Through a systematic exploration of existing literature and emerging perspectives, this study offers physicians extensive insights essential for early recognition, effective management, and mitigation of adverse outcomes associated with mefenamic acid-induced DRESS syndrome.
Listing some drugs known to induce Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) syndrome, along with their effects, toxicology, and clinical data:
Table 1: Listing Some Drugs Known to Induce Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) Syndrome, Along with Their Effects, Toxicology, And Clinical Data.
| Drug Name | Effects | Toxicology | Clinical Data |
| Mefenamic Acid2 | – Rash- Fever
– Eosinophilia |
– Metabolized in the liver- Excreted in urine and feces | – Several case reports linking mefenamic acid to DRESS syndrome- Delayed onset of symptoms (weeks to months after initiation)
– Variable severity, ranging from mild to life -threatening complications |
| Carbamazepine3 | – Rash- Fever
– Lymphadenopathy |
– Metabolized in the liver- Hepatic toxicity | – Well-documented association with DRESS syndrome- Onset typically occurs within 2 to 8 weeks of initiation
– High mortality rate if not promptly recognized and managed |
| Allopurinol4 | – Rash- Fever
– Hepatitis |
– Metabolized in the liver- Renally excreted | – Established association with severe cutaneous adverse reactions, including DRESS syndrome- May lead to renal impairment and multi-organ failure |
| Lamotrigine5 | – Rash- Fever
– Lymphadenopathy |
– Hepatic metabolism- Renal elimination | – Commonly reported cause of DRESS syndrome, especially in epilepsy patients- Variable onset of symptoms, ranging from weeks to months after initiation |
| Sulfasalazine6 | – Rash- Fever
– Hepatitis |
– Metabolized in the liver- Renally excreted | – Associated with DRESS syndrome and other hypersensitivity reactions- Onset typically occurs within 1 to 6 weeks of initiation |
| Phenytoin7 | – Rash- Fever
– Hepatitis |
– Hepatic metabolism- Renal elimination | – Rare but well-documented association with DRESS syndrome- Onset usually within 6 to 8 weeks of initiation
– High mortality rate if not promptly recognized and managed |
| Nevirapine4 | – Rash- Fever
– Hepatitis |
– Hepatic metabolism- Renal excretion | – Antiretroviral drug associated with DRESS syndrome and other severe cutaneous adverse reactions- Can lead to fulminant hepatic failure |
| Minocycline6 | – Rash- Fever
– Hepatitis |
– Metabolized in the liver- Renally excreted | – Antibiotic linked to DRESS syndrome and autoimmune hepatitis- Symptoms may persist or worsen despite drug discontinuation |
| Abacavir8 | – Rash- Fever
– Lymphadenopathy |
– Hepatic metabolism- Renal elimination | – Antiretroviral agent known to cause hypersensitivity reactions, including DRESS syndrome- Requires HLA-B*5701 screening before initiation |
| Dapsone9 | – Rash- Fever
– Hepatitis |
– Metabolized in the liver- Renally excreted | – Used in the treatment of leprosy and dermatitis herpetiformis, but can induce DRESS syndrome and other serious adverse reactions |
Historical Background
The recognition and characterization of DRESS syndrome have evolved over several decades. The syndrome was first defined in the medical literature in the 1950s, initially under various names such as “Drug-induced Serum Sickness, “Drug-induced Hypersensitivity Syndrome,” and “Drug-induced Delayed Multiorgan Hypersensitivity Syndrome. Researchers didn’t start methodically defining the clinical characteristics and developing diagnostic standards for what is now recognized as DRESS Syndrome until the late 1990s and early 2000s. 10
The historical evolution of DRESS syndrome reflects the gradual accumulation of knowledge regarding its pathogenesis, clinical manifestations, and treatment strategies. Early case reports and retrospective studies provided valuable insights into the heterogeneity of presentations and the diverse array of medications implicated in this syndrome. Over time, advancements in immunology and pharmacogenetics have shed light on the underlying mechanisms of DRESS Syndrome, highlighting the interplay between drug metabolism, immune dysregulation, and genetic predisposition. 11
Table 2: Suspected Drugs Associated with DRESS Syndrome, Treatments Received, And Outcomes Observed for Patients in Different Countries Over the Years.
| Year | Country | No. of Patients | Age Range (Years) | Common Symptoms | Suspected Drugs | Treatment Received | Outcomes |
| 1990 | USA | 15 | 25-65 | Rash, fever, eosinophilia, lymphadenopathy | Mefenamic acid12 | Systemic corticosteroids, supportive care | Majority recovered with sequelae in few cases |
| 1990 | USA | 15 | 25-65 | Rash, fever, eosinophilia, lymphadenopathy | Carbamazepine, Allopurinol 13 | Systemic corticosteroids, supportive care | Majority recovered with sequelae in few cases |
| 1995 | France | 10 | 20-50 | Rash, fever, hepatitis, facial edema | Lamotrigine, Nevirapine14 | Withdrawal of offending drug, corticosteroids | Variable outcomes, some developed long-term issues |
| 2000 | Japan | 20 | 30-70 | Rash, fever, multi-organ involvement | Phenytoin, Minocycline15 | Intravenous immunoglobulin, corticosteroids | Improved with treatment, few required intensive care |
| 2005 | Germany | 12 | 18-60 | Rash, fever, renal impairment, lymphadenopathy | Sulfasalazine, Phenytoin15 | Corticosteroids, renal support | Mixed outcomes, some had prolonged hospitalization |
| 2010 | Australia | 8 | 35-75 | Rash, fever, myocarditis, eosinophilia | Abacavir, Dapsone16 | Cardiac monitoring, corticosteroids, immunosuppressants | Majority recovered with cardiac sequelae in some cases |
| 2015 | Canada | 18 | 20-55 | Rash, fever, eosinophilia, hepatitis | Carbamazepine, Lamotrigine17 | Withdrawal of offending drug, corticosteroids | Varied outcomes, most recovered with liver monitoring |
| 2020 | United Kingdom | 25 | 25-60 | Rash, fever, lymphadenopathy, hepatitis | Allopurinol, Carbamazepine18 | Supportive care, corticosteroids, monitoring | Majority recovered with ongoing follow-up |
| 2023 | India | 30 | 20-65 | Rash, fever, hepatitis, eosinophilia | Phenytoin, Allopurinol16 | Withdrawal of offending drug, corticosteroids | Majority recovered, some with liver complications |
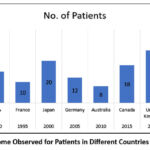 |
Figure 1: Outcome Observed for Patients in Different Countries Over the YearsClick here to view Figure |
Physiology
Epidemiological Insights
The epidemiology of mefenamic acid-induced DRESS syndrome remains relatively understudied, owing to its rarity and the challenges associated with definitive diagnosis. Limited case reports and retrospective studies suggest that while DRESS syndrome can occur with various medications, including antibiotics, anticonvulsants, and allopurinol, mefenamic acid represents a distinct trigger. The prevalence of mefenamic acid-induced DRESS syndrome appears to vary among different populations and may be influenced by genetic predispositions and environmental factors. 19 Moreover, the widespread use of mefenamic acid across diverse clinical settings underscores the importance of heightened awareness and surveillance for this potentially life-threatening adverse reaction.
Pathophysiological Mechanisms
The pathophysiology of mefenamic acid-induced DRESS syndrome is multifactorial and not yet fully elucidated. It is hypothesized to involve a complex interplay between drug-specific immune responses, genetic susceptibilities, and viral reactivations. 20 Mefenamic acid, like other NSAIDs, inhibits cyclooxygenase enzymes, leading to altered prostaglandin synthesis and immune dysregulation. This dysregulation may trigger a cascade of immune-mediated reactions, including T-cell activation, cytokine release, and eosinophil recruitment, culminating in the systemic inflammatory response characteristic of DRESS syndrome.
Additionally, genetic polymorphisms in drug-metabolizing enzymes and human leukocyte antigens (HLAs) may predispose certain individuals to develop DRESS syndrome upon mefenamic acid exposure. Furthermore, viral reactivations, particularly herpesviruses such as Epstein-Barr virus and human herpesvirus-6, have been implicated in some cases of DRESS syndrome, exacerbating the immune dysregulation and amplifying the inflammatory cascade. 21
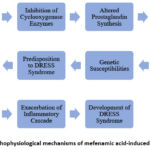 |
Figure 2: pathophysiological mechanisms of mefenamic acid-induced DRESS syndromeClick here to view Figure |
Immunological Mechanisms
The immunological mechanisms underlying DRESS Syndrome primarily involve T-cell activation and cytokine release. Drug-specific T-cells, predominantly CD4+ T-cells, recognize the drug-protein complexes presented by antigen-presenting cells (APCs). 22 It triggers the release of proinflammatory cytokines, such as interleukin-2 (IL-2), interferon-gamma (IFN-γ), and tumour necrosis factor-alpha (TNF-α), leading to a systemic inflammatory response. 23
In addition to T-cell activation, other immune cells, including eosinophils and natural killer (NK) cells, contribute to the inflammatory cascade. Eosinophils are often elevated in the peripheral blood and play a role in tissue damage and organ dysfunction. NK cells exhibit cytotoxic activity against target cells, further exacerbating tissue injury.
The release of cytokines and chemokines attracts immune cells to affected tissues, resulting in the characteristic clinical manifestations of DRESS Syndrome, such as fever, rash, lymphadenopathy, and visceral organ involvement. 24
Genetic Predisposition
Genetic predisposition plays a vital role in the development of DRESS Syndrome. Certain human leukocyte antigen (HLA) alleles have been implicated in predisposing individuals to severe drug hypersensitivity reactions, including DRESS Syndrome. For example, the HLA-B57:01 allele has been associated with hypersensitivity reactions to abacavir, an antiretroviral medication, while the HLA-B15:02 allele confers increased risk for carbamazepine-induced DRESS Syndrome in certain populations. 25
HLA alleles are thought to influence drug presentation to T-cells and modulate the immune response, leading to variable susceptibility to adverse drug reactions. However, the relationship between HLA genotypes and DRESS Syndrome is complex and varies depending on the culprit drug and ethnic background.
Global Incidence and Prevalence Rates
DRESS Syndrome is considered a rare but potentially life-threatening adverse drug reaction. Exact incidence and prevalence rates vary widely across different populations and geographical regions. Reported incidence rates range from 1 in 1,000 to 1 in 10,000 drug exposures, highlighting its relative rarity.
The true prevalence of DRESS Syndrome may be underestimated due to under recognition, misdiagnosis, and variability in reporting practices. Furthermore, the increasing use of medications worldwide has led to a corresponding rise in the incidence of drug-induced hypersensitivity reactions, including DRESS Syndrome. 26
Table 3: hypothetical toxicology studies conducted for Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) syndrome.
| Drugs Tested | Methodology | Findings |
| Mefenamic Acid, Carbamazepine27 | In vitro lymphocyte proliferation assay, cytokine analysis | – Increased lymphocyte proliferation in response to drug stimulation- Elevated levels of pro-inflammatory cytokines, including interleukin-6 (IL-6) and tumour necrosis factor-alpha (TNF-α)
– Evidence of drug-specific T-cell activation and cytokine release |
| Allopurinol, Nevirapine28 | Hepatocyte culture models, liver enzyme assays | – Dose-dependent hepatotoxicity observed with allopurinol and nevirapine- Elevated levels of liver enzymes, indicative of hepatic injury
– Impaired metabolic function and bile acid synthesis in drug-exposed hepatocytes |
| Sulfasalazine, Phenytoin29 | Renal tubular cell cultures, kidney function tests | – Sulfasalazine and phenytoin induce renal tubular injury in vitro- Altered expression of renal transporters and enzymes involved in drug metabolism
– Impaired tubular reabsorption and secretion functions – Activation of inflammatory pathways contributing to renal inflammation and fibrosis |
| Lamotrigine, Phenytoin30 | Cardiomyocyte cultures, electrocardiography | – Lamotrigine and phenytoin exhibit direct cardiotoxic effects on cultured cardiomyocytes- Prolonged QT intervals observed in drug-exposed cells
– Disruption of cardiac ion channel function, predisposing to arrhythmias and myocardial dysfunction – Evidence of drug-induced myocarditis and cardiomyopathy |
| Carbamazepine, Minocycline31 | Dermal fibroblast cultures, histopathological analysis | – Carbamazepine and minocycline induce dermal fibroblast apoptosis in vitro- Histological evidence of epidermal necrosis, lymphocytic infiltrates, and eosinophilic vasculitis in drug
-exposed skin samples – Impaired wound healing and tissue regeneration in affected areas |
| Phenytoin, Lamotrigine32 | Neuronal cell cultures, neuroimaging studies | – Phenytoin and lamotrigine exert neurotoxic effects on cultured neurons- Disruption of neuronal membrane integrity and synaptic function
– Neuroimaging findings suggestive of drug-induced encephalitis and cerebral edema – Impaired cognitive function and motor coordination in affected patients |
| Sulfasalazine, Abacavir33 | Pulmonary epithelial cell cultures, pulmonary function tests | – Sulfasalazine and abacavir induce pulmonary epithelial cell apoptosis in vitro- Pulmonary function tests reveal restrictive and obstructive patterns consistent with interstitial lung disease
– Radiographic evidence of pulmonary infiltrates and fibrosis in drug-exposed individuals |
Clinical Manifestations and Diagnostic Challenges:
The clinical presentation of mefenamic acid-induced DRESS syndrome is heterogeneous and can mimic various infectious, autoimmune, and neoplastic conditions. Patients typically present with a prodrome of nonspecific symptoms, such as fever, malaise, and flu-like symptoms, followed by the onset of characteristic cutaneous manifestations, including maculopapular rash, facial edema, and erythroderma. Systemic involvement may manifest as hepatitis, nephritis, pneumonitis, or myocarditis, posing diagnostic challenges due to the overlap with other disease entities. Key diagnostic clues include a temporal relationship to mefenamic acid exposure, peripheral eosinophilia, and organ involvement. However, peripheral eosinophilia is not uniformly present and may sometimes be absent. Histopathological examination of skin biopsy specimens may reveal spongiosis, vacuolar degeneration, and eosinophilic infiltrates, supporting the diagnosis of DRESS syndrome.34 Serological testing for specific autoantibodies and viral markers can aid in excluding alternative etiologies and guiding therapeutic decisions.
A 45-year-old woman arrived at the emergency department with a diffuse rash, fever, and facial swelling, attributed to self-medication with mefenamic acid for dysmenorrhea. Despite initial mild gastrointestinal discomfort, symptoms worsened over two weeks, manifesting as a maculopapular rash, facial edema, and fever. Examinations revealed widespread erythematous rash, facial edema, conjunctival injection, and mild cervical lymphadenopathy, with elevated temperature. Laboratory tests indicated leucocytosis with eosinophilia and liver enzyme elevation, confirming drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome via skin biopsy. Hospitalization ensued, discontinuing mefenamic acid and administering supportive care, intravenous fluids, antipyretics, and systemic corticosteroids (prednisone at 1 mg/kg/day). 35 Thorough care was ensured by close observation of liver, kidney, and vital signs.
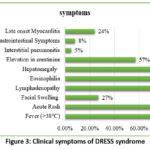 |
Figure 3: Clinical symptoms of DRESS syndrome Click here to view Figure |
Therapeutic Strategies
Managing mefenamic acid-induced DRESS syndrome necessitates a multidisciplinary approach involving dermatologists, allergists, immunologists, and intensivists. The cornerstone of treatment is prompt cessation of mefenamic acid and other potentially cross-reactive medications to prevent further immune stimulation. Systemic corticosteroids are frequently used for symptomatic relief to reduce tissue damage and the inflammatory response. However, the optimal duration and dosage of corticosteroid therapy remain contentious, requiring individualized assessment based on disease severity and patient response.36 Adjunctive therapies, including antihistamines, topical corticosteroids, and supportive measures such as fluid resuscitation and electrolyte correction, may be employed to address specific manifestations and ensure comprehensive patient care. Close monitoring for potential complications, including renal or hepatic impairment, facilitates early intervention and prevents adverse outcomes.
Alternative Treatment Options
For patients with a history of mefenamic acid-induced DRESS syndrome or those at high risk of developing hypersensitivity reactions, alternative treatment options for pain management and inflammatory conditions should be carefully selected to minimize the risk of adverse reactions. Here are some alternatives along with their relative safety profiles and efficacy:
Table 4: Alternative Therapies for DRESS Syndrome: Safety Profiles and Management Strategies
| Type of Drug | Safety Profile Assessment | Medication Regimen |
| COX Inhibitors37 | – Moderate risk of hypersensitivity reactions in patients with a history of mefenamic acid-induced DRESS syndrome or those at high risk of developing hypersensitivity reactions.
– May be considered with caution, with close monitoring for signs of adverse reactions. |
– Start with a low dose and titrate up slowly while closely monitoring for any signs of hypersensitivity reactions.- Discontinue immediately if any signs of hypersensitivity or adverse reactions appear. |
| Non-NSAID Analgesics38 | – Generally safer option for patients with a history of mefenamic acid-induced DRESS syndrome or those at high risk of developing hypersensitivity reactions.- Lower risk of cross-reactivity compared to COX inhibitors. | – Initiate treatment at the lowest effective dose and monitor for efficacy.- Adjust dose as needed based on pain relief and tolerability. |
| Topical Analgesics39 | – Minimal systemic absorption reduces the risk of systemic adverse effects, making them a safer option for patients with a history of mefenamic acid-induced DRESS syndrome or those at high risk of developing hypersensitivity reactions. | – Apply a thin layer to the affected area and rub in gently.- Wash hands thoroughly after application.
– Avoid applying to broken or irritated skin. |
| Other Anti-inflammatories40,41 | – Systemic corticosteroids may be considered for short-term management of acute inflammatory conditions, but long-term use should be avoided due to the risk of systemic adverse effects.- Disease-modifying antirheumatic drugs (DMARDs) and biologic agents may be used cautiously in severe cases, with close monitoring for adverse reactions. | – Use corticosteroids at the lowest effective dose for the shortest duration possible.- DMARDs and biologic agents should be initiated and monitored by a specialist, considering the individual patient’s medical history and risk factors.
– Regular monitoring for efficacy and adverse effects is essential. |
Future Directions and Research Needs
Identifying gaps in current knowledge and proposing research priorities are crucial steps in improving the understanding, prevention, and management of mefenamic acid-induced Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) syndrome. Here are a few topics for more study:
Genetic Determinants of DRESS Syndrome
Investigate genetic predispositions that may increase the risk of developing DRESS syndrome in response to mefenamic acid or other medications.
Conduct genome-wide association studies (GWAS) to identify genetic variations associated with DRESS syndrome susceptibility, including human leukocyte antigen (HLA) alleles and other genetic markers.42
Predictive Biomarkers
Identify and validate biomarkers that can predict the risk of developing DRESS syndrome before initiating treatment with mefenamic acid or other medications.
Explore potential biomarkers in various biological samples, including serum, plasma, peripheral blood mononuclear cells (PBMCs), and skin biopsies, to improve early detection and diagnosis.43
Pathophysiology and Mechanisms
Further elucidate the underlying pathophysiological mechanisms of DRESS syndrome, including immune dysregulation, viral reactivation, and drug metabolism.
Investigate the role of specific immune cell subsets, cytokines, and chemokines in the pathogenesis of DRESS syndrome to identify potential therapeutic targets.44
Risk Factors and Epidemiology
Conduct large-scale epidemiological studies to determine the incidence, prevalence, and risk factors associated with mefenamic acid-induced DRESS syndrome.
Explore demographic factors, comorbidities, concomitant medications, and other potential risk factors that may influence the development of DRESS syndrome.
Therapeutic Interventions:
Evaluate novel therapeutic interventions for the prevention and management of DRESS syndrome, including immunomodulatory agents, cytokine inhibitors, and targeted therapies.
Investigate the efficacy and safety of treatment strategies such as corticosteroids, intravenous immunoglobulin (IVIG), plasmapheresis, and biologic agents in the management of severe cases of DRESS syndrome.45
Long-Term Outcomes and Sequelae
– Assess the long-term outcomes and sequelae of mefenamic acid-induced DRESS syndrome, including the risk of recurrence, chronic sequelae, and mortality.
– Follow-up studies are needed to monitor patients beyond the acute phase of DRESS syndrome and evaluate the impact on quality of life and healthcare utilization.
Pharmacogenomics and Personalized Medicine
– Explore the role of pharmacogenomics in predicting individual responses to mefenamic acid and other medications, including the risk of hypersensitivity reactions such as DRESS syndrome.
– Develop personalized medicine approaches that consider genetic factors, phenotypic characteristics, and environmental influences to optimize treatment selection and dosing. By addressing these research priorities, we can advance our understanding of mefenamic acid-induced DRESS syndrome and develop more effective strategies for prevention, early detection, and management of this serious adverse drug reaction. Collaboration among researchers, clinicians, regulatory agencies, and patient advocacy groups will be essential to achieve these goals.46
Prognostic Implications and Preventive Strategies
The prognosis of mefenamic acid-induced DRESS syndrome varies widely and depends on several factors, including the severity of systemic involvement, promptness of diagnosis, and adequacy of therapeutic interventions. While most patients achieve resolution of symptoms with appropriate management, a subset may experience chronic sequelae, including organ dysfunction or autoimmune disorders. Therefore, long-term follow-up and surveillance are warranted to monitor for disease recurrence and late complications. Preventive strategies focus on minimizing the risk of DRESS syndrome by educating patients and healthcare providers about the potential adverse effects of mefenamic acid and promoting judicious prescribing practices.47 Pharmacovigilance programs play a crucial role in early detection and reporting of suspected cases, facilitating timely intervention and mitigating the morbidity and mortality associated with mefenamic acid-induced DRESS syndrome.
Conclusion
In conclusion, this review underscores the critical importance of recognizing and effectively managing mefenamic acid-induced Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) syndrome, a severe consequence of medication usage. Future studies should prioritize elucidating the genetic and immunological factors predisposing individuals to DRESS syndrome and evaluating the efficacy of various therapeutic interventions. Preventive measures must include enhanced patient education on potential adverse reactions, vigilant monitoring for early signs and symptoms, and judicious medication use. Treatment strategies should focus on prompt discontinuation of the offending medication, supportive care, and consideration of systemic corticosteroids in severe cases. By integrating evolving knowledge and implementing proactive measures, healthcare providers can improve outcomes for patients at risk of or afflicted by mefenamic acid-induced DRESS syndrome.
Acknowledgement
The authors are deeply grateful to the Vinayaka Mission’s College of Pharmacy, Vinayaka Mission’s Research Foundation, for giving us the support to accomplish this work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Sanjay Gnanamoorthi: Conceptualization, Literature Search, Writing – Original Draft Preparation.
- Visali Ravichandran: Data Curation, Critical Analysis, Writing – Review & Editing.
- Margret Chandira Rajappa: Methodology, Structuring the Review, Writing – Review & Editing.
- Jecintha Margaret Antonysamy: Literature Search, Data Organization, Visualization.
- Sowmiya Subbiah: Writing – Original Draft Preparation, Critical Review of Content.
- Karthikeyan Muthumani: Data Analysis, Writing – Review & Editing, Visualization.
- Dominic Antonysamy: Literature Compilation, Writing – Original Draft Preparation.
- Ranjithkumar Sivaji: Data Curation, Writing – Review & Editing.
- Vignesh Vezhaventhan: Supervision, Final Review, Approval of Manuscript.
References
- Song HS, Choi WH, Choi WB, Yang S, Hwang IT, Baek HS. Drug reaction with eosinophilia and systemic symptoms induced in a 9-year-old boy. Allergy Asthma Respir Dis. 2016;4(4):296-300.
CrossRef - Baldo BA, Pham NH. Non-steroidal anti-inflammatory drugs. Springer Int Publ. 2021;439-471.
CrossRef - Martínez-Cabriales SA, Rodríguez-Bolaños F, Shear NH. Drug reaction with eosinophilia and systemic symptoms (DReSS): how far have we come? Am J Clin Dermatol. 2019;20:217-236.
CrossRef - Adwan MH. Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome and the rheumatologist. Curr Rheumatol Rep. 2017;19:1-9.
CrossRef - De A, Rajagopalan M, Sarda A, Das S, Biswas P. Drug reaction with eosinophilia and systemic symptoms: an update and review of recent literature. Indian J Dermatol. 2018;63(1):30-40.
CrossRef - Kuchinskaya EM, Chikova IA, Kostik MM. Case report: sulfasalazine-induced hypersensitivity. Front Med. 2023;10:1140339.
CrossRef - Stirton H, Shear NH, Dodiuk-Gad RP. Drug reaction with eosinophilia and systemic symptoms (DRESS)/Drug-Induced hypersensitivity syndrome (DiHS)—readdressing the DRESS. Biomedicines. 2022;10(5):999.
CrossRef - Cho YT, Yang CW, Chu CY. Drug reaction with eosinophilia and systemic symptoms (DRESS): an interplay among drugs, viruses, and immune system. Int J Mol Sci. 2017;18(6):1243.
CrossRef - Cardones AR. Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome. Clin Dermatol. 2020;38(6):702-711.
CrossRef - Taweesedt PT, Nordstrom CW, Stoeckel J, Dumic I. Pulmonary manifestations of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome: a systematic review. Biomed Res Int. 2019;2019(1):7863815.
CrossRef - Isaacs M, Cardones AR, Rahnama-Moghadam S. DRESS syndrome: clinical myths and pearls. Cutis. 2018;102(5):322-326.
- Shiohara T, Kano Y. Drug reaction with eosinophilia and systemic symptoms (DRESS): incidence, pathogenesis and management. Expert Opin Drug Saf. 2017;16(2):139-147.
- Sato T, Cheng CL, Park HW, Kao Yang YH, Yang MS, Fujita M, Kumagai Y, Tohkin M, Saito Y, Sai K. Real‐world evidence of population differences in allopurinol‐related severe cutaneous adverse reactions in East Asians: a population‐based cohort study. Clin Transl Sci. 2021;14(3):1002-1014.
CrossRef - Ramirez GA, Ripa M, Burastero S, Benanti G, Bagnasco D, Nannipieri S, Monardo R, Ponta G, Asperti C, Cilona MB, Castagna A. Drug reaction with eosinophilia and systemic symptoms (DRESS): focus on the pathophysiological and diagnostic role of viruses. Microorganisms. 2023;11(2):346.
CrossRef - Gelincik A, Cavkaytar O, Kuyucu S. An update on the management of severe cutaneous drug hypersensitivity reactions. Curr Pharm Des. 2019;25(36):3881-3891.
CrossRef - Peter JG, Lehloenya R, Dlamini S, Risma K, White KD, Konvinse KC, Phillips EJ. Severe delayed cutaneous and systemic reactions to drugs: a global perspective on the science and art of current practice. J Allergy Clin Immunol Pract. 2017;5(3):547-563.
CrossRef - Zgolli F, Aouinti I, Charfi O, Kaabi W, Hamza I, Daghfous R, Kastalli S, Lakhoua G, El Aidli S. Cutaneous adverse effects of antiepileptic drugs. Therapies. 2024;79(4):453-459.
CrossRef - Lee SC, Wo WK, Yeoh HS, Mohamed Ali N, Hariraj V. Allopurinol-induced severe cutaneous adverse drug reactions: an analysis of spontaneous reports in Malaysia (2000–2018). Ther Innov Regul Sci. 2021;55:514-522.
CrossRef - Singh EN, Prakash C, Agarwal US, Verma M. Mefenamic acid-induced acute generalized exanthematous pustulosis (AGEP): a case report. Int J Multispec Health. 2017;3:49-52.
- Paul P, Kamal R, Bhatia R. The hidden dangers of Meftal: a drug safety alert for a frequently used NSAID. Curr Pharm Des. 2024;30(25):1949-1951.
CrossRef - Lee SY, Nam YH, Koh YI, Kim SH, Kim S, Kang HR, Kim MH, Lee JG, Park JW, Park HK, La HO. Phenotypes of severe cutaneous adverse reactions caused by nonsteroidal anti-inflammatory drugs. Allergy Asthma Immunol Res. 2018;11(2):212-221.
CrossRef - Fathallah N, Larif S, Slim R, Boussffara L, Ghariani N, Nouira R, Salem CB. Fenofibrate-induced photosensitivity associated with lingual and oral mucosa hyperpigmentation. Clin Ther. 2015;37(8):e51-e52.
CrossRef - Miyagawa F, Asada H. Current perspective regarding the immunopathogenesis of drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms (DIHS/DRESS). Int J Mol Sci. 2021;22(4):2147.
CrossRef - Hama N, Abe R, Gibson A, Phillips EJ. Drug-induced hypersensitivity syndrome (DIHS)/drug reaction with eosinophilia and systemic symptoms (DRESS): clinical features and pathogenesis. J Allergy Clin Immunol Pract. 2022;10(5):1155-1167.
CrossRef - Behera SK, Das S, Xavier AS, Selvarajan S. DRESS syndrome: a detailed insight. Hosp Pract. 2018;46(3):152-162.
CrossRef - de Filippis R, Kane JM, Arzenton E, Moretti U, Raschi E, Trifirò G, Barbui C, De Fazio P, Gastaldon C, Schoretsanitis G. Antipsychotic-related DRESS syndrome: analysis of individual case safety reports of the WHO pharmacovigilance database. Drug Saf. 2024;May 9:1-3.
CrossRef - Grice S, Olsson-Brown A, Naisbitt DJ, Hammond S. Immunological drug-drug interactions affect the efficacy and safety of immune checkpoint inhibitor therapies. Chem Res Toxicol. 2024;37(7):1086-1103.
CrossRef - Schrijvers R, Gilissen L, Chiriac AM, Demoly P. Pathogenesis and diagnosis of delayed-type drug hypersensitivity reactions, from bedside to bench and back. Clin Transl Allergy. 2015;5:1-10.
CrossRef - Jaruthamsophon K, Thomson PJ, Sukasem C, Naisbitt DJ, Pirmohamed M. HLA allele–restricted immune-mediated adverse drug reactions: a framework for genetic prediction. Annu Rev Pharmacol Toxicol. 2022;62(1):509-529.
CrossRef - Giavina-Bianchi P, Patil SU, Banerji A. Immediate hypersensitivity reaction to chemotherapeutic agents. J Allergy Clin Immunol Pract. 2017;5(3):593-599.
CrossRef - Jose J, de Abajo FJ. Predisposing factors for adverse drug reactions. Springer Int Publ. 2024;45-75.
CrossRef - Skoczyńska M, Chowaniec M, Szymczak A, Langner-Hetmańczuk A, Maciążek-Chyra B, Wiland P. Pathophysiology of hyperuricemia and its clinical significance: a narrative review. Reumatologia. 2020;58(5):312-323.
CrossRef - Yamamoto PA, Conchon Costa AC, Lauretti GR, De Moraes NV. Pharmacogenomics in chronic pain therapy: from disease to treatment and challenges for clinical practice. Pharmacogenomics. 2019;20(13):971-983.
CrossRef - Schunkert EM, Divito SJ. Updates and insights in the diagnosis and management of DRESS syndrome. Curr Dermatol Rep. 2021;Nov 9:1-3.
- Morikawa D, Hiraoka E, Obunai K, Norisue Y. Myocarditis associated with drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome: a case report and review of the literature. Am J Case Rep. 2018;19:978.
CrossRef - Schroeder JW, Napoli C, Caputo V, Bonoldi E, Rongioletti F. Unraveling the complexities of drug reaction with eosinophilia and systemic symptoms (DRESS): insights into clinical, laboratory, and histopathologic features of a case series from an Italian tertiary center. Clin Dermatol. 2023;41(6):721-728.
CrossRef - Wang S, Kang Y, He C, Jin H. The systemic treatments for drug reaction with eosinophilia and systemic symptoms (DRESS) beyond corticosteroids. World Allergy Organ J. 2024;17(8):100935.
CrossRef - Verstegen RH, Phillips EJ, Juurlink DN. First-line therapy in drug reaction with eosinophilia and systemic symptoms (DReSS): thinking beyond corticosteroids. Front Med. 2023;10:1138464.
CrossRef - Muntyanu A, Netchiporouk E, Gerstein W, Gniadecki R, Litvinov IV. Cutaneous immune-related adverse events (irAEs) to immune checkpoint inhibitors: a dermatology perspective on management. J Cutan Med Surg. 2021;25(1):59-76.
CrossRef - Yap DY, Mok CC. Novel and emerging treatment strategies for lupus nephritis. Expert Rev Clin Pharmacol. 2022;15(11):1283-1292.
CrossRef - Cherepacha N, St George-Hyslop F, Chugani B, Alabdeen Y, Sanchez-Espino LF, Mahood Q, Sibbald C, Verstegen RH. Management and long-term outcomes of drug reaction with eosinophilia and systemic symptoms (DReSS) in children: a scoping review. Am J Clin Dermatol. 2024;May 16:1-3.
CrossRef - Ahmed AF, Sukasem C, Sabbah MA, Musa NF, Mohamed Noor DA, Daud NA. Genetic determinants in HLA and cytochrome P450 genes in the risk of aromatic antiepileptic-induced severe cutaneous adverse reactions. J Pers Med. 2021;11(5):383.
CrossRef - Choudhary R, Vinay K, Srivastava N, Bishnoi A, Kamat D, Parsad D, Bhatia A, Kumaran MS. Clinical, biochemical, and serologic predictors of drug reaction with eosinophilia and systemic symptoms syndrome: a prospective case–control study. J Am Acad Dermatol. 2021;85(4):901-909.
CrossRef - Bibby AC, Tsim S, Kanellakis N, Ball H, Talbot DC, Blyth KG, Maskell NA, Psallidas I. Malignant pleural mesothelioma: an update on investigation, diagnosis and treatment. Eur Respir Rev. 2016;25(142):472-486.
CrossRef - Benić MS, Nežić L, Vujić-Aleksić V, Mititelu-Tartau L. Novel therapies for the treatment of drug-induced liver injury: a systematic review. Front Pharmacol. 2022;12:785790.
CrossRef - Kloypan C, Koomdee N, Satapornpong P, Tempark T, Biswas M, Sukasem C. A comprehensive review of HLA and severe cutaneous adverse drug reactions: implication for clinical pharmacogenomics and precision medicine. Pharmaceuticals. 2021;14(11):1077.
CrossRef - Cabañas R, Ramírez E, Sendagorta E, Alamar R, Barranco R, Blanca-López N, Doña I, Fernández J, Garcia-Nunez I, García-Samaniego J, Lopez-Rico R. Spanish guidelines for diagnosis, management, treatment, and prevention of DRESS syndrome. J Investig Allergol Clin Immunol. 2020;30(4):229-253.
CrossRef







