Manuscript accepted on :08-07-2025
Published online on: 06-08-2025
Plagiarism Check: Yes
Reviewed by: Dr. Satya Namani
Second Review by: Dr. Sonali Manwatkar
Final Approval by: Dr. Jihan Seid Hussein
Nagaraju Bandaru* , Mohiyuddin Ikramuddin Shaikh
, Mohiyuddin Ikramuddin Shaikh , Bagal Tanmay Yogendra
, Bagal Tanmay Yogendra , Wagh Sakshi Krishna
, Wagh Sakshi Krishna , Krunal bhai Rameshbhai
, Krunal bhai Rameshbhai , Makarand Suresh Gambhire
, Makarand Suresh Gambhire
Department of Pharmacology, School of Pharmaceutical Sciences (SOPS), Sandip University, Nasik,, Maharashtra, India.
Corresponding Author Email id: bnrajupharma@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3219
Abstract
Current treatment options for Alzheimer’s disease target neurotransmitters following the disease onset, and they offer limited efficacy without slowing down the disease progression. There has been an elevating concern in recent years targeting Histamine H3 receptor in treating cognitive disorders, including dementia. Preclinical studies have shown that antagonists of H3 receptor or inverse agonists enhances the cognitive function in animal models with dementia by increasing the release of neurotransmitters associated with learning and memory. The primary aim of this study is to explore the pathophysiological mechanisms underlying Alzheimer’s disease (AD), with a specific focus on the role of the histamine H3 receptor (H3R) and its modulators. This review employed a systematic literature search across databases including PubMed, Scopus, Google Scholar, and ClinicalTrials.gov, selecting peer-reviewed studies published between 2000 and 2024. Results of the study illustrate the complex landscape of research on H3 receptor modulators in dementia, highlighting both promising findings and ongoing challenges in translating preclinical discoveries into effective clinical interventions. Knowing the Histamine H3 receptors role in dementia and developing novel pharmacological interventions targeting these receptors represent a promising avenue for future research leading to new treatments development to this devastating condition.
Keywords
Dementia; Findings; Histamine; Modulators; Receptors; Research
Download this article as:| Copy the following to cite this article: Bandaru N, Shaikh M. I, Yogendra B. T. Krishna W. S, Rameshbhai K. B. Gambhire M. S. Review on H3 Receptor Modulators: Future Pioneering approaches in Dementia Treatment. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Bandaru N, Shaikh M. I, Yogendra B. T. Krishna W. S, Rameshbhai K. B. Gambhire M. S. Review on H3 Receptor Modulators: Future Pioneering approaches in Dementia Treatment. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/45xc1oH |
Introduction
Alzheimer is a degenerative & incapacitating neuro condition marked by gradual Cognitive abilities deterioration, specifically impairment of memory & diminished reasoning. This is the primary cause of cognitive decline in elderly individuals, affecting many people globally. The disease, named after the German neurologist Alois Alzheimer, who initially documented the condition in 1906,1 primarily impacts the brain by causing the buildup of atypical protein deposits like beta-amyloid plaques and tau tangles and the demise of nerve cells.These atypical formations interfere with the regular transmission of signals between neurons, leading to the progressive decline of cognitive functions like memory, language, problem-solving, and daily activities.2
The precise etiology of Alzheimer’s disease remains elusive; however, advancing age, genetic predisposition, and lifestyle factors are considered potential determinants in its pathogenesis. The disease exhibits individual variability in its progression, typically commencing with mild symptoms that gradually deteriorate over time.2 Alzheimer’s disease not only impacts the individuals who are diagnosed but also imposes a considerable emotional and physical strain on their families and caregivers. Despite thorough investigations, there is presently no remedy for Alzheimer’s disease. The treatment options have the goal of mitigating symptoms and improving the quality of life for individuals who are impacted. Current scientific endeavors are concentrated on comprehending the fundamental mechanisms of the disease, creating early diagnostic assays, and investigating potential therapeutic interventions.
The primary aim of this study is to explore the pathophysiological mechanisms underlying Alzheimer’s disease (AD), with a specific focus on the role of the histamine H3 receptor (H3R) and its modulators. This study aims to examine how H3R modulation influences cognitive decline and neurodegeneration in AD, evaluate its potential as a therapeutic target, and investigate the implications of H3R modulators in the treatment of Alzheimer’s disease. Additionally, the study will aim to analyze the relationship between H3 receptor activity and neurotransmitter systems involved in AD, providing insights into novel therapeutic strategies for improving cognitive function and slowing disease progression.
Drug targets for the treatment of dementia
Ongoing research into drug targets for Alzheimers is advancing, with an exploration of the multiple potential targets. It is crucial to acknowledge that drug development is an intricate procedure, and numerous potential treatments undergo a thorough examination in preclinical and clinical trials prior to being released in the market. Scientists are constantly investigating new methods, and our knowledge of the underlying causes of Alzheimer’s disease is developing. The primary drug targets that are being studied for Alzheimer’s disease are summarized below:
Beta-Amyloid Plaques
Beta-amyloid is a protein that creates deposits in people brains who have Alzheimer’s disorder. Scientists have been investigating medications that specifically target beta-amyloid, with the goal of decreasing its production or improving its removal. Nevertheless, clinical trials focused on beta-amyloid have yielded inconclusive outcomes, and the beta amyolid importance in this disease continues to be actively studied.
Post-translational modification of Amyloid Precursor Protein -APP involves cleavage at α- or β-side enzymes, and processing of C-terminal fragment produced by gamma secretase. If cleavage is from the α-secretase product, it results in a non-toxic protein fragment (p3), but the products of the cleavages by the Beta site cleavage enzyme – BACE & the Gamma secretase yield 38-43 Amino acid flibrillogenic Amyloid-beta protein (Aβ), though Aβ42 which is the very much prone to deposition identified in the Core neuritic plaques.3 To reduce Aβ production, efforts have been made to target drugs that can activate alpha secretase, 4 or inhibit beta and gamma secretase, 5 Degradation of toxic form of Aβ is also a target for dementia management as Aβ degradation enzymes are low in human 6 & animal 7 AD models, and the endogenous pathways to achieve this include neutral endopeptidase (neprilysin),8 metalloproteinase, endothelin-converting enzyme,angiotensin-converting enzyme.
Studies have shown that constitutive monomer form of Aβ or fibrillar network form aggregated in plaques is not the synaptotoxic form,9 but rather the oligomerization of monomeric Aβ into dimer, trimer & other higher molecular mass combinations at aggregation stage. Thus, oligomeric inhibition is a top target for the prevention of AD. Some agents that are at different stages of clinical trial for the prevention of oligomerization Aβ include grape-derived polyphenols, 10 curcumin & Omega-3 fatty acids.11
Tau Protein
Intracellular Neuro Fibrillay Tangle, which is a condensed from of cycloskeletal structure consisting hyper phosphorylated helical paired filaments of microtubule associated with Protein Tau, is the second pathological marker of AD. While Tau phosphorylation is important for its functioning, the hyper phosphorylated Tau does no longer binds the microtubule but rather aggregate into paired helical filaments,12 ultimately leading to microtubule instability and disruption of axonal transport. Various kinases, including Glycogen synthase kinase 3 beta (GSK-3β ) & Cyclin dependant kinase – 5, that is involved in tau hyper phosphorylation have been identified and are of therapeutic target. Agents like lithium and valproate inhibit GSK-3β to stabilize tau. Protein phosphatases, which dephosphorylate tau, are also important target for the inhibition of hyperphosphorylation.13
Cholinergic System
Inhibition of cholinergic function leads to attention deficit, while facilitation of cholinergic transmission improves it. 14 The major role of cholinergic system on learning proces, and memory 15 have been established, as endogenous acetylcholine modulates the acquisition, encoding, 16 consolidation,reconsolidation,17 extinction,18 & memory retrieval.19 In fact, memory loss in AD patients is associated with cholinergic neurons degeneration from nucleus basalis of Meynert.20 There is evidence showing cholinergic system disruption impairs memory, attention, and learning. 21 Some examples of drugs that target the cholinergic system are acetylcholinesterase inhibitors and neuronal nicotinic receptors’ agonists. 22
Serotonergic modulation
The expression of serotonin receptors in the brain areas important for memory & learning, and their decline in AD and dementia have been well-established. 23 Moreover, the beneficial effect of selective agonists and antagonists of serotonin receptors on cognition in animal 24 & human 25 models have been reported.
Inflammation
Neuroinflammation is thought in contribution to advancement of Alzheimer’s Disease. Researchers are currently studying anti-inflammatory medications and substances that specifically target inflammatory pathways as potential treatments. Some drugs involved in the regulation of inflammation that are under different clinical trial phases on AD are Masitinib, NE3107, 26Semaglutide, AL002, 27 Bacillus Calmette-Guerin, 28 Baricitinib,29 Canakinumab 30, Daratumumab 31, Lenalidomide 32, Montelukast 33, Pepinemab 34, Proleukin 35, Rapamycin ,36 Sargramostim, 37 Senicapoc, 38 TB006,39 Tdap vaccine, 40 Valacyclovir, 41 XPro1595, 42 CpG1018, 43 Emtricitabine, 44 IBC-Ab002,45 Salsalate,46 VT301.47
Neuroprotective Factors
Certain studies focus on investigating factors that enhance the resilience of nerve cells, shielding them from harm and boosting their longevity. This encompasses neurotrophic factors and other molecules that have the potential to augment neuronal resilience, enhancing synaptic plasticity, or produce neuroprotective effects. Some of the drugs at different phases of clinical trial include AGB101,48 Blarcamesine,49 Fosgonimeton, Simufilam, 50Tertomotide,51 AL001,52 Bryostatin1,53 CY6463, 54 Dalzanemdor, 55 Edonerpic, 56 Elayta, 57 EX039, 58ExPlas,59 MW150,60 Neflamapimod ,61 and Centella asiatica.62
Neurogenesis
Agents that promote neurogenesis are among those that are in the treatment pipeline for the treatment of AD. An example is allopregnanolone, that is an allosteric modulator of inhibitory Gamma aminobutyric acid-A receptors, which reduced deposition of amyloid and enhanced memory and learning. 63 Another example is sovateltide, an Endothein -B receptor antagonist that helps in promoting differentiating neuronal progenitors for production of mature neuronal cells which exhibits anti-apoptotic, antioxidant, & enhancement of functional properties of mitochondria .64
Genetic factor
Persons carrying apolipoprotein E Ɛ 4 are at high risk of developing AD at earlier age, as it leads to increased Aβ deposition in the brain by regulating the passage of Aβ from the blood to the brain. Some drugs target apolipoprotein E Ɛ 4 gene carrier (APOE4), being a very influential risk factor after the age of an individual & the most vital genetic factor for the AD development. This protein makes strong interaction with Aβ, thereby reducing the amyloid brain accumulation age & elevating the total Aβ burden in gene carriers. 65 Furthermore, APOE4 exacerbates Tau neuro-fibrillary tangle-related blood brain barrier disruption neurofibrillary tangle-dusruption, neurodegeneration, microglial responses, astroglial activity and neuroinflammation.66 Some therapies to targeting this marker are hydroxypropyl-beta-cyclodextrin,67 LX 1001, and obicetrapib.68
Oxidative stress
Evidence have shown that enhancement of antioxidant status and attenuation of oxidative stress can reduce, prevent or treat AD, as the food rich polyphenols, antioxidants & poly unsaturated fatty acids reduce AD risk. 69 Drugs like hydralazine,70 edaravone,71 among others, are at various clinical trials stages for management of AD.
Others
There are other mediators and pathways that are targeted for drug development on the management of AD. Some of these are drugs that regulate vascular factors (telmisartan, perindopril), circadian rhythm (piromelatine), 72 and epigenetics (lamivudine).73
Lifestyle and Supportive Interventions
Non-pharmacological interventions are essential for effectively managing the symptoms of Alzheimer’s disease. These factors encompass cognitive stimulation, physical activity, and a nutritious diet. Establishing a nurturing and organized setting can additionally improve the general welfare of individuals who have AD.
Clinical trials and Research on Histamine-receptor in AD treatment
Ongoing research is being conducted to formulate novel treatments and therapies for Alzheimer’s disease. Clinical trials provide the opportunity to access experimental medications or interventions that are currently undergoing testing to determine their efficacy.
Alzheimer’s research is constantly evolving, with continuous endeavors to discover novel therapeutic targets and create more efficient interventions. It is recommended that individuals suffering from Alzheimer’s, their families and caregivers, remain updated on the most recent research discoveries and treatment choices by consulting with healthcare professionals and reliable sources.
Histamine
Histamine is a neurotransmitter that performs various physiological functions in the body through four receptor subunits, including G-protein coupled H1, H2, H3, & H4, and various agonists and antagonists were developed for these receptors.74 Allergic indications like asthma, rhinitis, conjunctivitis, & atopic dermatitis are treated with H1 histamine receptor antagonists.75 Activation of H2 receptors stimulates the gastric secretion, and H1 & H2 receptors mediate the opposing pharmacological and physiological effects on lungs and heart.76 H3 histamine receptor is a presynaptic autoreceptor that inhibits the production & release of histamine in histaminergic neurons,77 while the H4-receptor is expressed in the immune cells. Evidence has shown that both H3 and H4 have homology and some H3 agonists & antagonists equally bind to H4 receptor.78
Arecent nationwide cohort study in Taiwan showed that the use of H1 receptor antagonist is linked with increased dementia risk.79
H3 histamine receptor is encoded on chromosome 20 and it is displayed in many regions of brain like Cerebral cortex, CNS basal ganglia, & hypothalamus, all of which play some important roles in cognition.80 It binds to Gi protein to negatively control the intracellular second messenger cAMP.81 Histamine is known to downregulate the acetylcholine-induced calcium signaling of the muscarinic receptor via H3 histamine receptor-mediated mechanisms. 82 It is also known that H3 receptor antagonists can stimulate histamine, dopamine, acetylcholine & norepinephrine, all of which are participated in some specific cognition aspects, making H3 receptor antagonists important drug targets to improve cognition in dementia patients.
H3 receptor inverse agonists can increase histaminergic neuron activity by inhibiting the H3-receptor mediated suppression of histamine release in brain, making them a target for AD treatment drug development.
Although there are current studies investigating the involvement of histamine in neurodegenerative diseases, such as dementia, the connection between the two is intricate, and our comprehension is still incomplete. Below are several key factors concerning the potential role of histamine in dementia progression role.83
 |
Figure 1: Overview of Histamine regulation |
Table 1: Characteristics of Histamine Receptors
| S.No | Type | Location | Function | Binding Affinity toHistamine (pKi) | SignallingPathway(Figure 2) | Ref | |
| Central | Peripheral | ||||||
| 1 | H1R | Forebrain, cerebral cortex,Hippocampus,Thalamus |
Heart and smooth muscles | Decreasing BP, inflammatory response, and increased wakefulness | 4.2 | Phospholipase C (PLC) | 84 |
| 2 | H2R | SubstantiaNigra, raphe nuclei, Hippocampus and Basal ganglia |
Intestine smooth muscles, heart, and lungs | Regulation of hormone release, fluid balance, excitation, relaxation of airway smooth muscles, blood pressure regulation and gastric acid regulation | 4.3 | Protein kinase C activation(PKC) | 85 |
| 3 | H3R | Cerebral cortex, basalganglia, hypothalamus |
Lung, CVS, intestine | Histamine release regulation and stimulation | 8.0 | Inhibition of PKA, activation of Phospholipase 2, MAPK |
86 |
| 4 | H4R | Cerebellum &hippocampus | Hematopoietic Cells | Modulation of Immune system | 7.8 | PKA Inhibition, Phospholipase C & MAPK activation. |
87 |
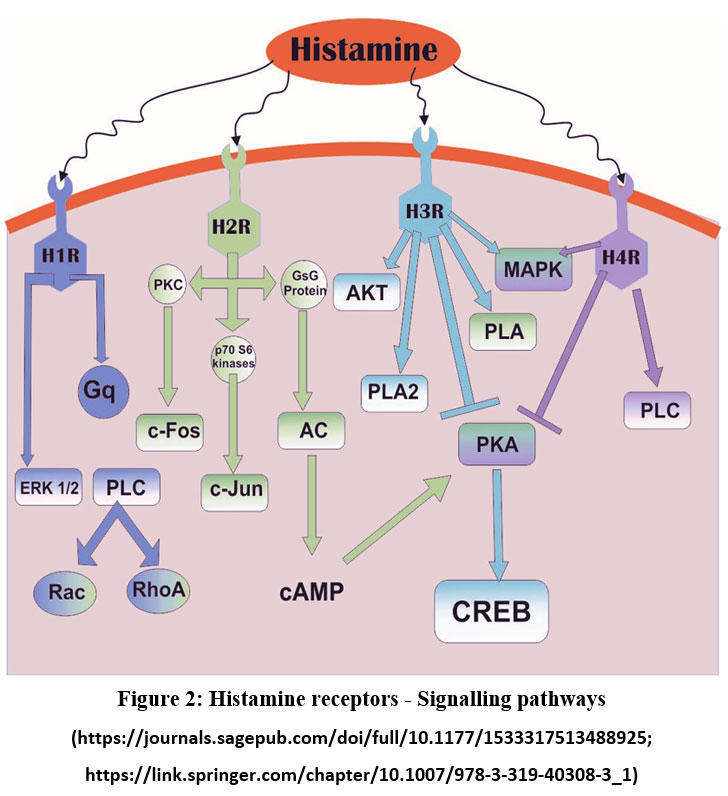 |
Figure 2: Histamine receptors – Signalling pathways. |
Role of the Histamnie in the pathogenesis of dementia
Histamine interacts with histamine receptors, specifically receptors 1, 2, and 3. Cognitive deficits occur when histamine is unable to bind to receptors. The H3 Rreduces histamine release in brain, resulting Alzheimer’s disease. Inverse agonists of H3R are crucial in counteracting the effects of histamine-induced Alzheimer’s disease. 88-89
Antagonists of Histamine 3 receptors stimulate the production of histamine, ACh, and other neurotransmitters, thereby enhancing cognitive function. Histamine plays a role in both short-term and long-term cognitive processes.90 Recent studies indicate that the deterioration of histamine neurons is the causative factor in Alzhemer’s development. The presence of histamine in the brain improves cognitive function and memory, although the specific mechanism by which it does so remains unclear.91 The histaminergic neuron system in the brain regulates various roles i.e., homeostasis, learning arousal & memory, and circadian rhythms. Furthermore, certain studies explained that histamine plays a part in regulating specific behavioral tasks, although the underlying mechanism remains unclear.92 To treat Alzheimer’s disease (AD), various pre-clinical methods have been developed to specifically target H3 receptors.93
The development of AD is attributed to the induction of neurotoxicity by beta-amyloid 1-42. However, this neurotoxicity can be mitigated by histamine acting on histamine receptors (H2 and H3).94 Using H2 receptor agonists leads to a gradual reduction in AD (Anthony et al. 2000). Using H2 receptor agonists leads to a gradual reduction in AD (Anthony et al. 2000). HIR-KO mice exhibit cognitive symptoms because of alterations in brain levels of AChE and dopamine.95 The involvement of Histamine 1 & 2 receptors in cognitive function is substantiated by the presence of cognitive deficits resulting from null mutations in the genes encoding these receptors. Both H1 and H2 are excitatory neurotransmitters, whereas H3 functions differently as an inhibitory neurotransmitter and acts as an auto heteroreceptor. Interactions between histaminergic, peptidergoc & aminergic systems can be able to regulate homeostatic functions like Sleep wake cycles, cognition & synaptic plasticity.96 The compound GSK189254, a high-affinity histamine 3 receptor antagonist, has shown therapeutic potential for Alzheimer’s disease in both rats and humans.97 Histamine is involved in cognitive processes, but the interaction between histamine and histamine 3 receptors lowers the cholinergic function in hippocampus and frontal cortex. 98 The H3 antagonists (ABT-239) enhanced the symptoms of Alzheimer’s by stimulating the biochemical signaling and inducing tau hyperphosphorylation.(Figure 3)99
Histamine receptors are involved in regulating the functional activity of dendritic cells subsets. The H2R antagonist characterizes the specific mechanisms of histamine induced decrease of CD1a (+) DCs, IL6 & IL10 increased production, upregulation of chemokines, expression of C5aR1 through the CD1a (−) & DC subset, and increased migration of activated DC subsets, which is stimulated by secretion of MMP-9 & MMP-12 enzymes. 100 SOCE or store-operated calcium entry, is the main mechanism by which DCs (dendritic cells) allow Ca2+ ions to enter.
DCs that have been primed with histamine can initiate the Th2 immune response by interacting with several types of histamine receptors. Dendritic cells (DCs) are activated by histamine, which trigger the release of calcium ions (Ca2+) from their intracellular reservoirs. Histamine elevates IL-10 levels while decreasing the IL-12p70 levels that are produced by DCs. Pretreating dendritic cells (DCs) with H1R antagonists, SOC blockers, and H4R antagonists can prevent the histamine-induced Th2 polarisation of T-helper cells in the mixed responses of lymphocytes. Recent research indicates that SOCE (Store-Operated Calcium Entry) is crucial in the Th(2) response and histamine-induced maturation of dendritic cells through activation of both H1R and H4R.101 Research has demonstrated that young individuals who produce elevated levels of IL-2 and IFN-γ possess a specific type of T-cell memory called beta (1-42)-specific Th1-type T-cell memory. There is evidence indicating that as individuals age, there is a decline in the production of IFN-γ and IL-2, while there is a noticeable increase in the release of regulatory IL-10 by CD4(+) T-cells. However, despite of the absence of an effector cytokine, individuals with AD can still generate IL-10.102 The proinflammatory cytokine IL-32 can activate nuclear factor κB & p38 mitingen activated protein kinase (p38MAPK) pathways. IL-32 can induce histamine synthesis in human derived core blood mast cells (HDCBMCs; Figure 1). Therefore, it can be demonstrated that IL-32 is specific to a particular species and functions in fully developed human mast cells (LAD 2 cells).103 Research has demonstrated that IL-32 plays role in controlling neuro inflammatory responses in various neuro diseases, including Alzheimer’s disease.104
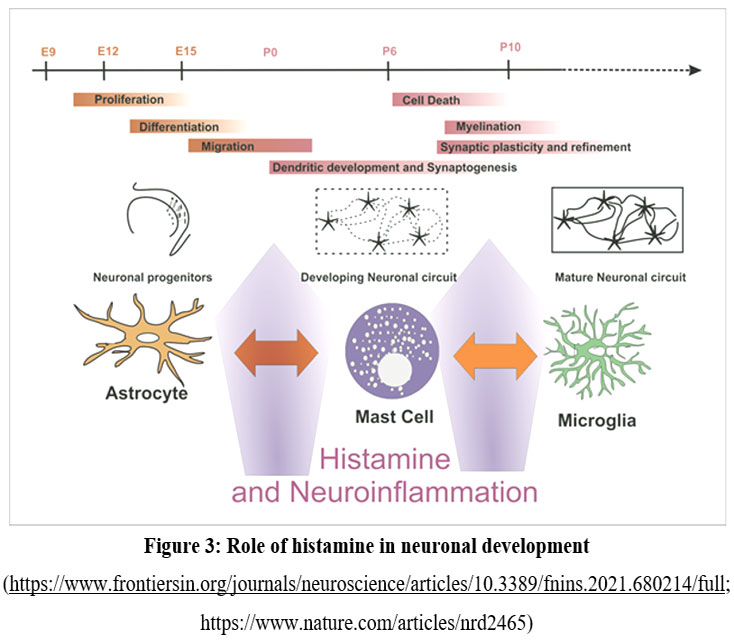 |
Figure 3: Role of histamine in neuronal development |
Recently developed drugs for the treatment of dementia by blocking H3 receptors
Thioperamide
Thioperamide is a highly potent and specific antagonist of imidazole. It was primarily developed to improve wakefulness and address issues related to learning and memory. According to recent research, thioperamide, despite its hepatotoxicity, has been found to have a significant impact on patients with circadian rhythm disorders & Parkinson’s disorder. 105 (Figure 4).
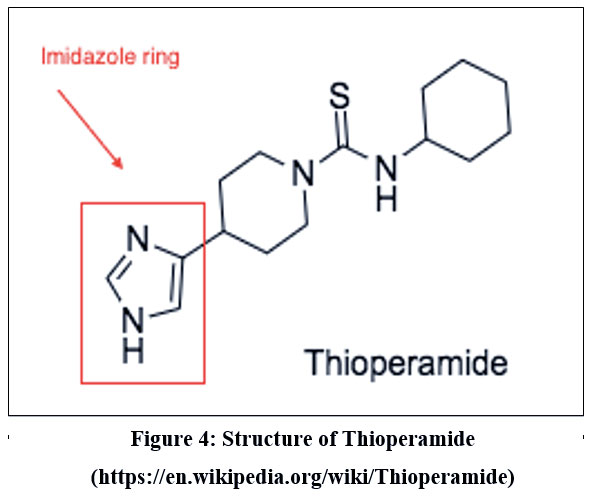 |
Figure 4: Structure of Thioperamide |
Pitolisant
Pitolisant is the potent H3 receptor antagonist/inverse agonist that has been approved by regulatory agencies in the United States and Europe. Its high oral bioavailability allows easy access to the brain. It undergoes metabolism by the enzyme CYP4A in the gastrointestinal tract. It is employed in managing narcolepsy to sustain wakefulness during the daytime. Headache, anxiety, and QT prolongation have been documented as adverse effects in clinical trials. 106 Wakix is a proprietary name for a product that has been commercially available since March 2016. The dosage is available in tablets of 4.5 mg and 18 mg.
Conclusion
Neuropharmacology has a lot of potential due to the fact that H3 receptor modulators are being used to treat dementia after being experimentally developed. Investigating these modulators has yielded valuable understandings of the intricate mechanisms that underlie dementia and has unveiled fresh prospects for therapeutic interventions. The bench-to-bedside approach prioritizes the smooth conversion of scientific findings into tangible implementations for patient treatment.
The combined endeavors of researchers, clinicians, and pharmaceutical developers are instrumental in creating groundbreaking solutions that could revolutionize dementia treatment in the future. As research and clinical trials progress, our understanding of H3 receptor modulators will improve, leading to a better understanding of their role in the comprehensive care and management of dementia patients.
Future Prospective
While H3 receptor modulators seem to be promising potential therapeutic agents for dementia, further research and development efforts are needed to realize their full clinical potential and impact on patient outcomes. Collaboration between academia, industry, regulatory agencies, and patient advocacy groups will be essential in advancing this field and addressing the growing burden of dementia worldwide.
Conclusion
H3 receptor modulators represent a promising frontier in dementia treatment, offering potential therapeutic benefits by modulating histaminergic neurotransmission. Their role in cognitive enhancement, neuroprotection, and synaptic plasticity underscores their significance in addressing dementia-related pathophysiology. Preclinical and clinical studies highlight their efficacy, yet challenges such as selectivity, safety, and long-term effects remain to be addressed. Future research should focus on optimizing drug design, exploring combination therapies, and conducting extensive clinical trials to ensure translational success. With continued advancements, H3 receptor modulators may emerge as pivotal agents in the fight against dementia.
Acknowledgment
All authors are thankful to Management of Sandip University, Nasik, for providing facilities.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author Contributions
- Nagaraju Bandaru. and Mohiyuddin Ikramuddin Shaikh: Contributed to conceptualization and methodology,
- Nagaraju Bandaru, Wagh Sakshi Krishna and Krunal bhai Rameshbhai: Performed the literature research, analyzed the data and drafted the manuscript,
- Makarand Suresh Gambhire: Critically revised the work,
- All the authors have read and agreed to the published version of the manuscript.
References
- Ahmad FB, Cisewski JA, Xu J, Anderson RN. Provisional Mortality Data – United States, 2022. MMWR Morb Mortal Wkly Rep. 2023;72(18):488-492. doi:10.15585/mmwr.mm7218a3.
CrossRef - Mendez MF. Early-Onset Alzheimer Disease. Neurol Clin. 2017;35(2):263-281. doi:10.1016/j.ncl.2017.01.005.
CrossRef - Zetterberg H, Bendlin BB. Biomarkers for Alzheimer’s disease-preparing for a new era of disease-modifying therapies. Mol Psychiatry. 2021;26(1):296-308. doi:10.1038/s41380-020-0721-9.
CrossRef - Porsteinsson AP, Isaacson RS, Knox S, Sabbagh MN, Rubino I. Diagnosis of Early Alzheimer’s disease: Clinical Practice in 2021. J Prev Alzheimers Dis. 2021;8(3):371-386. doi:10.14283/jpad.2021.23.
CrossRef - Therriault J, Zimmer ER, Benedet AL, et al. Staging of Alzheimer’s disease: past, present, and future perspectives. Trends Mol Med. 2022;28(9):726-741. doi:10.1016/j.molmed.2022.05.008.
CrossRef - Tang Y, Lutz MW, Xing Y. A systems-based model of Alzheimer’s disease. Alzheimers Dement. 2019;15(1):168-171.
CrossRef - Zilberzwige-Tal S, Gazit E. Go with the Flow-Microfluidics Approaches for Amyloid Research. Chem Asian J. 2018;13(22):3437-3447.
CrossRef - Maccioni RB, González A, Andrade V, Cortés N, Tapia JP, Guzmán-Martínez L. Alzheimer’s Disease in the Perspective of Neuroimmunology. Open Neurol J. 2018;12:50-56.
CrossRef - Jatoi S, Hafeez A, Riaz SU, Ali A, Ghauri MI, Zehra M. Low Vitamin B12 levels: An underestimated cause of minimal cognitive impairment and dementia. 2020;12:e6976. doi:10.7759/cureus.6976.
CrossRef - Wulff BS, Hastrup S, Rimvall K. Characteristics of recombinantly expressed rat and human histamine H3 receptors. Eur J Pharmacol. 2002;453(1):33-41.
CrossRef - Fernández-Novoa L, Cacabelos R. Histamine function in brain disorders. Behav Brain Res. 2001;124(2):213-233.
CrossRef - Zarrindast MR, Eidi M, Eidi A, Oryan S. Effects of histamine and opioid systems on memory retention of passive avoidance learning in rats. Eur J Pharmacol. 2002;452(2):193-197.
CrossRef - Higuchi M, Yanai K, Okamura N, et al. Histamine H(1) receptors in patients with Alzheimer’s disease assessed by positron emission tomography. 2000;99(4):721-729.
CrossRef - Shi M, Chu F, Zhu F, Zhu J. Impact of Anti-amyloid-β Monoclonal Antibodies on the Pathology and Clinical Profile of Alzheimer’s Disease: A Focus on Aducanumab and Lecanemab. Front Aging Neurosci. 2022;14:870517.
CrossRef - Cummings J, Lee G, Nahed P, et al. Alzheimer’s disease drug development pipeline: 2022. Alzheimers Dement (N Y). 2022;8(1):e12295.
CrossRef - Mintun MA, Lo AC, Duggan Evans C, et al. Donanemab in Early Alzheimer’s Disease. N Engl J Med. 2021;384(18):1691-1704.
CrossRef - Rosenberg A, Mangialasche F, Ngandu T, Solomon A, Kivipelto M. Multidomain Interventions to Prevent Cognitive Impairment, Alzheimer’s Disease, and Dementia: From FINGER to World-Wide FINGERS. J Prev Alzheimers Dis. 2020;7(1):29-36.
CrossRef - Swaddiwudhipong N, Whiteside DJ, Hezemans FH, et al. Pre-diagnostic cognitive and functional impairment in multiple sporadic neurodegenerative diseases. Alzheimers Dement. 2023;19(5):1752-1763.
CrossRef - van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in Early Alzheimer’s Disease. N Engl J Med. 2023;388(1):9-21.
CrossRef - Aevarsson O, Skoog I. A population-based study on the incidence of dementia disorders between 85 and 88 years of age. J Am Geriatr Soc. 1996;44(12):1455-1460.
CrossRef - Aisen PS, Cummings J, Schneider LS. Symptomatic and nonamyloid/tau based pharmacologic treatment for Alzheimer disease. Cold Spring Harb Perspect Med. 2012;2(3):a006395.
CrossRef - Akbaraly TN, Portet F, Fustinoni S, et al. Leisure activities and the risk of dementia in the elderly: results from the Three-City Study. 2009;73:854–861.
CrossRef - Andersen K, Nielsen H, Lolk A, Andersen J, Becker I, Kragh-Sørensen P. Incidence of very mild to severe dementia and Alzheimer’s disease in Denmark: the Odense Study. 1999;52(1):85-90.
CrossRef - Angevaren M, Aufdemkampe G, Verhaar HJ, Aleman A, Vanhees L. Physical activity and enhanced fitness to improve cognitive function in older people without known cognitive impairment. Cochrane Database Syst Rev. 2008;16(3):CD005381.
CrossRef - Arbus C, Soto ME, Andrieu S, et al. The prevalence of clinically significant depressive symptoms in Alzheimer’s disease: relationship with other psychological and behavioural symptoms. Int J Geriatr Psychiatry. 2008;23(11):1209-1211.
CrossRef - Athan ES, Williamson J, Ciappa A, et al. A founder mutation in presenilin 1 causing early-onset Alzheimer disease in unrelated Caribbean Hispanic families. 2001;286(18):2257-2263.
CrossRef - Atti AR, Palmer K, Volpato S, Winblad B, De Ronchi D, Fratiglioni L. Late-life body mass index and dementia incidence: nine-year follow-up data from the Kungsholmen Project. J Am Geriatr Soc. 2008;56(1):111-116.
CrossRef - Bachman DL, Wolf PA, Linn RT, et al. Incidence of dementia and probable Alzheimer’s disease in a general population: the Framingham Study. 1993;43(3 Pt 1):515-519.
CrossRef - Bondi MW, Edmonds EC, Salmon DP. Alzheimer’s Disease: Past, Present, and Future. J Int Neuropsychol Soc. 2017;23(9-10):818-831.
CrossRef - Alzheimer’s disease facts and figures. Alzheimers Dement. 2018;14:367–429. doi:10.1016/j.jalz.2018.02.001.
CrossRef - Brookmeyer R, Gray S, Kawas C. Projections of Alzheimer’s disease in the United States and the public health impact of delaying disease onset. Am J Public Health. 1998;88(9):1337-1342.
CrossRef - Tanzi RE. The genetics of Alzheimer disease. Cold Spring Harb Perspect Med. 2012;2(10):a006296.
CrossRef - Lambert JC, Amouyel P. Genetics of Alzheimer’s disease: new evidence for an old hypothesis? Curr Opin Genet Dev. 2011;21(3):295-301.
CrossRef - Giri M, Zhang M, Lü Y. Genes associated with Alzheimer’s disease: an overview and current status. Clin Interv Aging. 2016;11:665-81.
CrossRef - Mahley RW. Apolipoprotein E: cholesterol transport protein with expanding role in cell biology. 1988;240(4852):622-30.
CrossRef - Bales KR, Liu F, Wu S, Lin S, Koger D, DeLong C, Hansen JC, Sullivan PM, Paul SM. Human APOE isoform-dependent effects on brain beta-amyloid levels in PDAPP transgenic mice. J Neurosci. 2009;29(21):6771-9.
CrossRef - Ye S, Huang Y, Müllendorff K, Dong L, Giedt G, Meng EC, Cohen FE, Kuntz ID, Weisgraber KH, Mahley RW. Apolipoprotein (apo) E4 enhances amyloid beta peptide production in cultured neuronal cells: apoE structure as a potential therapeutic target. Proc Natl Acad Sci U S A. 2005;102(51):18700-5.
CrossRef - Brodbeck J, McGuire J, Liu Z, Meyer-Franke A, Balestra ME, Jeong DE, Pleiss M, McComas C, Hess F, Witter D, Peterson S, Childers M, Goulet M, Liverton N, Hargreaves R, Freedman S, Weisgraber KH, Mahley RW, Huang Y. Structure-dependent impairment of intracellular apolipoprotein E4 trafficking and its detrimental effects are rescued by small-molecule structure correctors. J Biol Chem. 2011;286(19):17217-26.
CrossRef - Serrano-Pozo A, Qian J, Monsell SE, Betensky RA, Hyman BT. APOEε2 is associated with milder clinical and pathological Alzheimer disease. Ann Neurol. 2015;77(6):917-29
CrossRef - Yamagata K, Urakami K, Ikeda K, Ji Y, Adachi Y, Arai H, Sasaki H, Sato K, Nakashima K. High expression of apolipoprotein E mRNA in the brains with sporadic Alzheimer’s disease. Dement Geriatr Cogn Disord. 2001;12(2):57-62.
CrossRef - Raygani AV, Zahrai M, Raygani AV, Doosti M, Javadi E, Rezaei M, Pourmotabbed T. Association between apolipoprotein E polymorphism and Alzheimer disease in Tehran, Iran. Neurosci Lett. 2005;375(1):1-6.
CrossRef - Hudry E, Dashkoff J, Roe AD, Takeda S, Koffie RM, Hashimoto T, Scheel M, Spires-Jones T, Arbel-Ornath M, Betensky R, Davidson BL, Hyman BT. Gene transfer of human Apoe isoforms results in differential modulation of amyloid deposition and neurotoxicity in mouse brain. Sci Transl Med. 2013;5(212):212ra161.
CrossRef - Pankiewicz JE, Guridi M, Kim J, Asuni AA, Sanchez S, Sullivan PM, Holtzman DM, Sadowski MJ. Blocking the apoE/Aβ interaction ameliorates Aβ-related pathology in APOE ε2 and ε4 targeted replacement Alzheimer model mice. Acta Neuropathol Commun. 2014;2:75.
CrossRef - Selkoe DJ. The molecular pathology of Alzheimer’s disease. 1991;6(4):487-98.
CrossRef - Frost B, Jacks RL, Diamond MI. Propagation of tau misfolding from the outside to the inside of a cell. J Biol Chem. 2009;284(19):12845-52.
CrossRef - Olufunmilayo EO, Gerke-Duncan MB, Holsinger RMD. Oxidative stress and antioxidants in neurodegenerative disorders. Antioxidants (Basel). 2023;12(2):517.
CrossRef - Adachi N. Cerebral ischemia and brain histamine. Brain Res Brain Res Rev. 2005;50(2):275-86.
CrossRef - Haas HL, Sergeeva OA, Selbach O. Histamine in the nervous system. Physiol Rev. 2008;88(3):1183–1241.
CrossRef - Alvarez EO. The role of histamine on cognition. Behav Brain Res. 2009;199(2):183–189.
CrossRef - Wulff BS, Hastrup S, Rimvall K. Characteristics of recombinantly expressed rat and human histamine H3 receptors. Eur J Pharmacol. 2002;453(1):33–41.
CrossRef - Mazurkiewicz-Kwilecki IM, Prell GD. Brain histamine: plasma corticosterone, spontaneous locomotor activity, and temperature. Pharmacol Biochem Behav. 1980;12(4):549–553.
CrossRef - Walter M, Stark H. Histamine receptor subtypes: a century of rational drug design. Front Biosci (Schol Ed). 2012;4:461–488.
CrossRef - Philippu A, Prast H. Importance of histamine in modulatory processes, locomotion, and memory. Behav Brain Res. 2001;124(2):151–159.
CrossRef - Sundvik M, Kudo H, Toivonen P, Rozov S, Chen YC, Panula P. The histaminergic system regulates wakefulness and orexin/hypocretin neuron development via histamine receptor H1 in zebrafish. FASEB J. 2011;25(12):4338–4347.
CrossRef - Stasiak A, Mussur M, Unzeta M, Lazewska D, Kiec-Kononowicz K, Fogel WA. The central histamine level in a rat model of vascular dementia. J Physiol Pharmacol. 2011;62(5):549–558.
- Fleck MW, Thomson JL, Hough LB. Histamine-gated ion channels in mammals? Biochem Pharmacol. 2012;83(9):1127–1135.
CrossRef - Woodruff GN, Oniwinde AB, Kerkut GA. Histamine in tissues of the snail, crab, goldfish, frog, and mouse. Comp Biochem Physiol. 1969;31(4):599–603.
CrossRef - Wang KY, Tanimoto A, Yamada S, et al. Histamine regulation in glucose and lipid metabolism via histamine receptors: model for nonalcoholic steatohepatitis in mice. Am J Pathol. 2010;177(2):713–723.
CrossRef - Kyriakidis DA, Theodorou MC, Tiligada E. Histamine in two-component system-mediated bacterial signaling. Front Biosci. 2012;17:1108–1119.
CrossRef - Schneider E, Rolli-Derkinderen M, Arock M, Dy M. Trends in histamine research: new functions during immune responses and hematopoiesis. Trends Immunol. 2002;23(5):255–263.
CrossRef - Yanai K, Tashiro M. The physiological and pathophysiological roles of neuronal histamine: an insight from human positron emission tomography studies. Pharmacol Ther. 2007;113(1):1-8.
CrossRef - Montoro J, Mullol J, Dávila I, et al. Bilastine and the central nervous system. J Investig Allergol Clin Immunol. 2011;21(suppl 3):9–15.
- Webster SJ, Wilson CA, Lee CH, Mohler EG, Terry AV Jr, Buccafusco JJ. The acute effects of dimebolin, a potential Alzheimer’s disease treatment, on working memory in rhesus monkeys. Br J Pharmacol. 2011;164(3):970–978.
CrossRef - Akdis CA, Simons FE. Histamine receptors are hot in immunopharmacology. Eur J Pharmacol. 2006;533(1-3):69–76.
CrossRef - Pan’kova MN, Lobov GI, Chikhman VN, Solnyshkin SD. Effects of histamine on contractile activity of lymphatic node capsules. The NO role [in Russian]. Ross Fiziol Zh Im I M Sechenova. 2011;97(6):633–640.
- Mizuguchi H, Ono S, Hattori M, Fukui H. Inverse agonistic activity of antihistamines and suppression of histamine H1 receptor gene expression. J Pharmacol Sci. 2012;118(1):117–121.
CrossRef - Notcovich C, Diez F, Tubio MR, et al. Histamine acting on H1 receptor promotes inhibition of proliferation via PLC, RAC, and JNK-dependent pathways. Exp Cell Res. 2010;316(3):401–411.
CrossRef - Leurs R, Traiffort E, Arrang JM, Tardivel-Lacombe J, Ruat M, Schwartz JC. Guinea pig histamine H1 receptor. II. Stable expression in Chinese hamster ovary cells reveals the interaction with three major signal transduction pathways. J Neurochem. 1994;62(2):519–527.
CrossRef - O’Mahony L, Akdis M, Akdis CA. Regulation of the immune response and inflammation by histamine and histamine receptors. J Allergy Clin Immunol. 2011;128(6):1153–1162.
CrossRef - Jolly S, Desmecht D. Functional identification of epithelial and smooth muscle histamine-dependent relaxing mechanisms in the bovine trachea, but not in bronchi. Comp Biochem Physiol C Toxicol Pharmacol. 2003;134(1):91–100.
CrossRef - Fukushima Y, Shindo T, Anai M, et al. Structural and functional characterization of gastric mucosa and central nervous system in histamine H2 receptor-null mice. Eur J Pharmacol. 2003;468(1):47–58.
CrossRef - Ogawa S, Yanai K, Watanabe T, et al. Histamine responses of large neostriatal interneurons in histamine H1 and H2 receptor knock-out mice. Brain Res Bull. 2009;78(4-5):189–194.
CrossRef - Easton AS, Dorovini-Zis K. The kinetics, function, and regulation of P-selectin expressed by human brain microvessel endothelial cells in primary culture. Microvasc Res. 2001;62(3):335–345.
CrossRef - Baudry M, Martres MP, Schwartz JC. H1 and H2 receptors in the histamine-induced accumulation of cyclic AMP in guinea pig brain slices. 1975;253(5490):362–364.
CrossRef - Hegstrand LR, Kanof PD, Greengard P. Histamine-sensitive adenylate cyclase in mammalian brain. 1976;260(5574):163–165.
CrossRef - Sheng M, Thompson MA, Greenberg ME. CREB: a Ca²⁺-regulated transcription factor phosphorylated by calmodulin-dependent kinases. 1991;252(5011):1427–1430.
CrossRef - Xu AJ, Kuramasu A, Maeda K, et al. Agonist-induced internalization of histamine H2 receptor and activation of extracellular signal-regulated kinases are dynamin-dependent. J Neurochem. 2008;107(1):208–217.
CrossRef - Arrang JM, Garbarg M, Schwartz JC. Auto-inhibition of brain histamine release mediated by a novel class (H3) of histamine receptor. 1983;302(5911):832–837.
CrossRef - Maeda K, Haraguchi M, Kuramasu A, et al. CLIC4 interacts with histamine H3 receptor and enhances the receptor cell surface expression. Biochem Biophys Res Commun. 2008;369(2):603–608.
CrossRef - Wellendorph P, Goodman MW, Burstein ES, Nash NR, Brann MR, Weiner DM. Molecular cloning and pharmacology of functionally distinct isoforms of the human histamine H3 receptor. 2002;42(7):929–940.
CrossRef - Gemkow MJ, Davenport AJ, Harich S, Ellenbroek BA, Cesura A, Hallett D. The histamine H3 receptor as a therapeutic drug target for CNS disorders. Drug Discov Today. 2009;14(9-10):509–515.
CrossRef - Thompson CM, Troche K, Jordan HL, Barr BL, Wyatt CN. Evidence for functional, inhibitory, histamine H3 receptors in rat carotid body Type I cells. Neurosci Lett. 2010;471(1):15–19.
CrossRef - Sakurai E, Sakurai E, Tanaka Y, Watanabe T, Singh Jossan S, Oreland L. Effects of histamine H3-receptor ligands on brain monoamine oxidase in various mammalian species. Brain Res. 2001;906(1-2):180–183.
CrossRef - Moreno-Delgado D, Torrent A, Gómez-Ramírez J, de Esch I, Blanco I, Ortiz J. Constitutive activity of H3 autoreceptors modulates histamine synthesis in rat brain through the cAMP/PKA pathway. 2006;51(3):517–523.
CrossRef - Nuutinen S, Vanhanen J, Pigni MC, Panula P. Effects of histamine H3 receptor ligands on the rewarding, stimulant, and motor-impairing effects of ethanol in DBA/2J mice. 2011;60(7-8):1193–1199.
CrossRef - Millan-Guerrero RO, Baltazar-Rodrıguez LM, Cardenas-Rojas MI. A280V polymorphism in the histamine H3 receptor as a risk factor for migraine. Arch Med Res. 2011;42(1):44–47.
CrossRef - Sheng Y, Lee JH, Medhurst AD, et al. Preservation of cortical histamine H3 receptors in ischemic vascular and mixed dementias. J Neurol Sci. 2012;315(1-2):110–114.
CrossRef - Bongers G, Bakker RA, Leurs R. Molecular aspects of the histamine H3 receptor. Biochem Pharmacol. 2007;73(8):1195–1204.
CrossRef - He G, Ma X, Lu J, et al. Alpha2 adrenoceptors modulate histamine release from sympathetic nerves in the guinea pig vas deferens. 2009;57(5-6):506–510.
CrossRef - Yu S, Stahl E, Li Q, Ouyang A. Antigen inhalation induces mast cells and eosinophils infiltration in the guinea pig esophageal epithelium involving histamine-mediated pathway. Life Sci. 2008;82(5-6):324–330.
CrossRef - Shaw JB, Cai Q, Mtshali C, Myles EL, Washington B. Heterogeneity of histamine H3 receptor genomic expression in the cerebral cortex of spontaneously hypertensive rats. Cell Mol Biol (Noisy-le-grand). 2007;53(4):45–50.
- Aquino-Miranda G, Osorio-Espinoza A, Escamilla-Sánchez J, et al. Histamine H3 receptors modulate depolarization-evoked [(3) H]-noradrenaline release from rat olfactory bulb slices. 2012;62(2):1127–1133.
CrossRef - Coruzzi G, Adami M, Pozzoli C. Role of histamine H4 receptors in the gastrointestinal tract. Front Biosci (Schol Ed). 2012;4:226–239.
CrossRef - Gibbs BF, Levi-Schaffer F. H4 receptors in mast cells and basophils: a new therapeutic target for allergy? Front Biosci. 2012;17:430–437.
CrossRef - Mommert S, Gschwandtner M, Koether B, Gutzmer R, Werfel T. Human memory Th17 cells express a functional histamine H4 receptor. Am J Pathol. 2012;180(1):177–185.
CrossRef - Gschwandtner M, Mommert S, Köther B, Werfel T, Gutzmer R. The histamine H4 receptor is highly expressed on plasmacytoid dendritic cells in psoriasis, and histamine regulates their cytokine production and migration. J Invest Dermatol. 2011;131(8):1668–1676.
CrossRef - Ikawa Y, Shiba K, Ohki E, et al. Comparative study of histamine H4 receptor expression in human dermal fibroblasts. J Toxicol Sci. 2008;33(4):503–508.
CrossRef - Jiang W, Lim HD, Zhang M, et al. Cloning and pharmacological characterization of the dog histamine H4 receptor. Eur J Pharmacol. 2008;592(1-3):26–32.
CrossRef - Bagyinszky E, Giau VV, Shim K, Suk K, An SSA, Kim S. Role of inflammatory molecules in Alzheimer’s disease progression and diagnosis. J Neurol Sci. 2017;376:242–254. doi:10.1016/j.jns.2017.03.031.
CrossRef - Tournier BB, Tsartsalis S, Ceyzériat K, Garibotto V, Millet P. In vivo TSPO signal and neuroinflammation in Alzheimer’s disease. 2020;9(9):1941. doi:10.3390/cells9091941.
CrossRef - Venkatachalam K, Eissa N, Awad MA, Jayaprakash P, Zhong S, Stölting F, Stark H, Sadek B. The histamine H3R and dopamine D2R/D3R antagonist ST-713 ameliorates autism-like behavioral features in BTBR T+tf/J mice by multiple actions. Biomed Pharmacother. 2021;138:111517. doi:10.1016/j.biopha.2021.111517.
CrossRef - Eissa N, Azimullah S, Jayaprakash P, Jayaraj RL, Reiner D, Ojha SK, Beiram R, Stark H, Łażewska D, Kieć-Kononowicz K, et al. The dual-active histamine H3 receptor antagonist and acetylcholine esterase inhibitor E100 alleviates autistic-like behaviors and oxidative stress in valproic acid-induced autism in mice. Int J Mol Sci. 2020;21(11):3996. doi:10.3390/ijms21113996.
CrossRef - Abdalla S, Eissa N, Jayaprakash P, Beiram R, Kuder KJ, Łażewska D, Kieć-Kononowicz K, Sadek B. The potent and selective histamine H3 receptor antagonist E169 counteracts cognitive deficits and mitigates disturbances in the PI3K/AKT/GSK-3β signaling pathway in MK801-induced amnesia in mice. Int J Mol Sci. 2023;24(16):12719. doi:10.3390/ijms241612719.
CrossRef - Wang J, Liu B, Xu Y, Luan H, Wang C, Yang M, Zhao R, Song M, Liu J, Sun L, You J. Thioperamide attenuates neuroinflammation and cognitive impairments in Alzheimer’s disease via inhibiting gliosis. Exp Neurol. 2022;347:113870.
CrossRef - Hirano K, Morishita Y, Minami M, et al. The impact of pitolisant, an H3 receptor antagonist/inverse agonist, on perirhinal cortex activity in individual neuron and neuronal population levels. Sci Rep. 2022;12:7015. doi:10.1038/s41598-022-11032-y.
CrossRef - Esbenshade TA, Browman KE, Bitner RS, Strakhova M, Cowart MD, Brioni JD. The histamine H3 receptor: an attractive target for the treatment of cognitive disorders. Br J Pharmacol. 2008 Jul;154(6):1166-81. doi: 10.1038/bjp.2008.147.
CrossRef - Rusjan P, Sabioni P, Di Ciano P, Mansouri E, Boileau I, Laveillé A, Capet M, Duvauchelle T, Schwartz JC, Robert P, Le Foll B. Exploring occupancy of the histamine H3 receptor by pitolisant in humans using PET. Br J Pharmacol. 2020 Aug;177(15):3464-3472. doi: 10.1111/bph.15067
CrossRef







