Manuscript accepted on :02-09-2025
Published online on: 09-10-2025
Plagiarism Check: Yes
Reviewed by: Dr. Hany Akeel
Second Review by: Dr. Raju Thenge
Final Approval by: Dr. Anton R Keslav
Deepali Maruti Jagdale1 , Vineeta Vivek Khanvilkar1*
, Vineeta Vivek Khanvilkar1* Abhay Ramchandra Shirode1
Abhay Ramchandra Shirode1 Ammara Hashmat Sahibole2
Ammara Hashmat Sahibole2 and Gayatri Shrikant Vinchurkar2
and Gayatri Shrikant Vinchurkar2
1Department of Pharmaceutical Chemistry, Bharati Vidyapeeth’s College of Pharmacy, University of Mumbai, Navi Mumbai, Maharashtra, India.
2Department of Quality Assurance, Bharati Vidyapeeth’s College of Pharmacy, University of Mumbai, Navi Mumbai, Maharashtra, India.
Corresponding Author E-mail: vineeta.khanvilkar@bvcop.in
DOI : https://dx.doi.org/10.13005/bpj/3282
Abstract
Nitrosamine impurities, composed of nitroso and amine functional groups, pose significant carcinogenic and mutagenic risks. An extensive literature survey was undertaken, encompassing peer-reviewed research articles, regulatory authority guidelines, and market recall data, to thoroughly explore the occurrence, sources, and regulatory perspectives related to nitrosamine impurities. The survey revealed that the formation of nitrosamine impurities in active drugs is attributed to factors like reactants and process parameters used in their synthesis and their generation in the drug products can be associated with the active drugs, excipients used, packaging material employed during manufacture and the storage conditions maintained. Stringent regulatory requirement for control of nitrosamine impurities to trace levels in pharmaceuticals necessitates the requirement of highly sensitive advanced analytical techniques for the accurate and precise detection and quantitation of nitrosamine impurities. This review is an elaborative account of the Nitrosamine impurities which gives information related to types of nitrosamine impurities, their toxicological concerns, chemistry behind their formation, and major contributors in their formation along with analytical methods used for their detection with appropriate examples wherever possible.
Keywords
Analytical techniques; Mutagenic impurities; Nitrite; Nitrosamine impurities; Secondary amine
Download this article as:| Copy the following to cite this article: Jagdale D. M, Khanvilkar V. V, Shirode A. R, Sahibole A. H, Vinchurkar G. S. Nitrosamine Impurities in Pharmaceuticals: Chemistry, Toxicology, and Advanced Analytical Approaches. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Jagdale D. M, Khanvilkar V. V, Shirode A. R, Sahibole A. H, Vinchurkar G. S. Nitrosamine Impurities in Pharmaceuticals: Chemistry, Toxicology, and Advanced Analytical Approaches. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/3IVRnWP |
Introduction
Impurities present in drug substances and drug products are detailed in the International Council for Harmonisation (ICH) quality (Q-series) guidelines. ICH Q3 defines impurity in DS as any compound from the new DS that is not the chemical entity as called the new DS. It establishes an impurity in DP as any constituent of the new DP that is neither the DP nor an excipient of the said DP.1,2 Nitrosamine impurities are a class of organic impurities that have significantly impacted in the pharmaceutical industry and have stringent regulations for their control. Impurities belonging to the nitrosamine category in drug formulations (pharmaceuticals) were observed first in 2018, where the United States Food and Drug Administration (US FDA) reported their presence in Valsartan batches manufactured by Zhejiang Huahai Pharmaceutical (ZHP).3 Since then, these impurities have become a topic of growing concern throughout the pharmaceutical industry owing to their carcinogenic potential.
Nitrosamine impurities can be observed in both DS and the DP. The formation of Nitrosamine impurities in DS is attributed to factors such as the reactants used and synthesis process parameters, including pH and temperature of the reaction. The generation of NI in the DP can be associated with the active drugs, excipients used, packaging materials employed during manufacture, and the storage conditions maintained.
Detection of NI in both DS and DP involves sensitive analytical techniques. This article aims to provide a comprehensive insight into the world of NI. It has covered information related to the types of NI, their toxicological concerns, the chemistry behind their formation, and the major contributors to their formation, along with an elaborate and detailed account of the analytical methods used for their detection, accompanied by appropriate examples wherever possible.
Materials and Methods
This review involved a comprehensive study and search through various science-based databases which included PubMed, Scopus along with Google Scholar, using keywords such as “nitrosamine impurities”, “carcinogenicity”, “conditions leading to NI formation in DS and DP”, “sources”, “analytical techniques”, “regulatory control”. No restrictions were applied regarding the publication year or language of the studies. A total of 50 studies were reviewed including case studies, original research work, industry reports, review articles. The research uses a comprehensive approach based on the unity of theory and practice.
Results
NI and their types
NI contain a nitroso and an amine functional group as shown in Figure 1
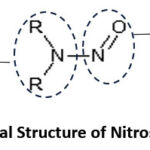 |
Figure 1: General Structure of Nitrosamine Impurity
Click here to view Figure |
US FDA has reported following seven types of NI that are most commonly encountered in DS and DP.4 These are given in Table 01:
 |
Table 1: Nitrosamine Impurities listed USFDA |
Toxicological Concerns Associated with Nitrosamines
The rising concern over the NI presence in DP is due to its mutagenic and potentially carcinogenic nature in humans.3-5 ICH multidisciplinary (M-series) guideline M7(R2) defines mutagenic impurity as “An impurity which has been demonstrated to be mutagenic in an appropriate mutagenicity test model, e.g., bacterial mutagenicity assay”.6 The National Toxicology Program, US Department of Health and Human Services, classifies NI as “reasonably anticipated to be human carcinogens”.7 NDMA comes in the class, which are termed as “probably carcinogenic to humans” by the International Agency for Research on Cancer (IARC).8
As per a study performed by Yamazaki et al.9 in 1992, NIs in the body are oxidised by cytochrome P450 (CYP450), leading to the formation of a reactive intermediate alkyldiazohydroxides (ADH), which irreversibly alkylate nucleic acids, thereby supporting the probable carcinogenic and mutagenic nature of NIs. This study shows that the liver microsomal enzymes CYP2E21 and CYP2A6 catalyse the activation of nitrosamines in the body; other microsomal enzymes, such as CYP3A4 and CYP1A2, may also contribute. The carcinogenic potential of NDMA is attributed to the formation of two reactive intermediates, namely formaldehyde and methyldiazonium ion, produced during the catalysis of nitrosamines by CYP450 enzymes, that alkylate the DNA base guanine irreversibly. The methyldiazonium ion is mainly responsible for methylating DNA, leading to carcinogenesis. This carcinogenic nature is due to the possible adduct formation which involves methylation at O6 and N7 of guanine.10
Acceptable Intake and Acceptable Daily Intake
ICH M7(R2) defines acceptable intake (AI) as “an intake level that poses negligible cancer risk, or for serious/life-threatening indications where risk and benefit are appropriately balanced”.6 Acceptable Daily Intake (ADI) is the daily intake of a substance which, over the entire life time of a human, will not have any adverse effects or will not cause any harm to the health of that human.11 Based on the threshold of toxicological concern (TTC), the US FDA defines the ADI for NI.4 According to ICH M7(R2) guideline on mutagenic impurities, “known mutagens with unknown carcinogenic potential need to be maintained at or below acceptable limits, i.e., appropriate TTC”. Known mutagens with unknown carcinogenic potential are those that exhibit bacterial mutagenicity but do not induce carcinogenicity in rodents. According to the TTC-based AI for mutagenic impurity, 1.5 µg of mutagenic impurity is allowed per person per day, which poses a negligible risk, meaning the cancer risk is less than 1 in 1,000,000 over a lifetime of exposure. This approach is utilized for mutagenic impurities present in pharmaceutical products that are to be used for long course treatment of more than 10 years and wherein no carcinogenic data is available for such pharmaceuticals.6 European Medicine Agency (EMA) also follows the ICH M7(R1) principles for NI. The AI limits for NI are based on TTC. These guidelines require all Marketing Authorization Holders (MAHs) to perform risk analysis for the occurrence of NI in pharmaceuticals. ADI limits for NI as provided by USFDA are summarized in Table 2.4
Table 2: Acceptable Intake of NI as per USFDA
| Nitrosamine Impurity | Acceptable Intake (nanograms per day) |
| NDMA | 96 |
| NDEA | 26.5 |
| NMBA | 96 |
| NMPA | 26.5 |
| NIPEA | 26.5 |
| NDIPA | 26.5 |
Sources of Nitrosamine Impurities in Pharmaceuticals
The formation of nitrosamine impurity is possible in most circumstances where both nitrite and amine sources are available.5 Different sources reported to contribute to the formation of NI in pharmaceuticals are discussed below.
Nitrosamine Impurity in Drug Substance (Active Pharmaceutical Ingredient API)
According to ICH Q1A (R2), DS is “the unformulated DS that may subsequently be formulated along with excipients to produce the dosage form”.12 Reagents and reaction conditions used during the preparation of synthetic DS contribute to the generation of NI. Two reagents may react with each other to generate NI. NDMA was reported in 2018 to originate from a reaction between sodium azide, used to form the tetrazole ring in Valsartan synthesis, and sodium nitrite, employed to quench the excess sodium azide.5,13 NI formation is possible if the manufacturing conditions are not monitored. Similarly inappropriate reaction conditions like pH, temperature have also reported to be responsible for formation of nitrosamine impurities.14
Synthesis of API sometimes involves use of recycled or recovered materials. If these recycled materials are contaminated with untreated nitrite or NI, API and other raw materials synthesized using them are definitely expected to contain these impurities.
Availability of both an amino and a nitro functional group in the molecular structure itself is a favourable condition for the NI formation as the API itself becomes the source for both amine and nitrite. The typical example of this is Ranitidine.
Nitrosamine Impurity in Drug Product
Excipients used in the formulation of the DP are sometimes contaminated with nitrite and thus contribute to the formation of NI in the final product. Few excipients commonly used in dosage form preparation, such as diluents like microcrystalline cellulose, from the class of super disintegrants, like croscarmellose sodium, sodium starch glycolate, and crospovidone, and glidants like magnesium stearate and corn starch, are reported to be contaminated with nitrite.15,16 Certain excipients, such as L-proline, meglumine, triethylamine, ethylene diamine tetraacetic acid (EDTA), having amine functionality present, can also lead to NI formation. Certain polymeric excipients, such as povidone and crospovidone, are reported to contain peroxides, which are responsible for generating nitrosating agents. These agents can react with labile amines and/or acidic degradants, catalysing nitrosamine formation.5,16 A classic example of such involvement of crospovidone is studied by N. Golob et al. 17 in Afitinib tablets (2023). They had evaluated two tablet formulations, which were prepared by using the excipient crospovidone with different nitrite content. It was observed upon accelerated storage that formulation with crospovidone having higher nitrite content showed a greater proportion of NDMA formation when analysed with liquid chromatography-high resolution mass spectrometry (LC-HRMS).17
NI are even reported to be formed during the storage of DP, for example, metformin hydroperoxide. This metformin hydroperoxide is reported to bring about oxidation of other metformin molecules or intramolecular oxidation ultimately leading to formation of NI.3
NI is also reported to occur in DP due to their interaction with the packaging material used to store it. Nitrocellulose lidding foils are commonly employed for packaging purpose which are labelled using printing ink. Literature has stated the evidence of reaction between nitrite from nitrocellulose and secondary amine functional group in drug resulting in the formation of nitrosamine impurity. During storage these may be transferred to the DP causing contamination of the DP with NI.18,19
Chemistry of Nitrosamine Impurity Formation
The reaction involved in the formation of NI includes the reaction of nitrites with amines, carbamates, amides, and urea derivatives.20 Amongst amines, the secondary amino group is found to be the most susceptible to nitrosamination as it directly forms nitrosamine upon reaction with nitrosating reagent. Aromatic secondary amines undergo faster reactions compared to aliphatic secondary amines. The primary amine group generally does not form nitrosamine upon reaction with nitrite, as it forms an unstable nitroso species and ultimately forms diazonium ion when protonated. However, in the presence of a second primary amine molecule, it can form a secondary amine, which in turn can form nitrosamine. Tertiary amino groups are significantly less susceptible to the formation of NI and form nitrosamine only after their subsequent rearrangement to secondary amine.5 Protonated amines are more susceptible than their free bases, mainly due to the reduced solubility of free bases and the need for acidic conditions to generate the active nitrosating agent.5,20 The general reaction leading to the formation of nitrosamine impurities can be depicted as follows:
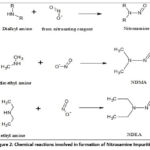 |
Figure 2: Chemical reactions involved in formation of Nitrosamine Impurities |
Nitrosamine Drug Substance-related Impurities (NDSRIs)
Along with NI listed in Table 01, nitrosamine drug substance-related impurities (NDSRIs) have also been cited to be present in DP. As mentioned in Federal Register, “NDSRIs are a class of nitrosamines having structural similarity to the API, and thus, differ in certain respects from small molecule NI. NI that donot share structural similarity to the API, and are therefore, not considered to be NDSRIs”.21 Formation of NDSRIs in DP may occur either during manufacturing of DP or during storage. This is attributed to the presence of nitrite impurities at parts per million (ppm) level in common excipients such as poly vinyl pyrrolidone (PVP), sodium starch glycolate, croscarmellose sodium and pre-gelatinized starch. The exact source of nitrites and nitrates in excipients remains unknown, but they may be attributed to their presence in trace amounts in the water used during manufacturing, processing steps involving acid titration, and air oxidation during the drying process. These excipients, when combined with APIs containing secondary, tertiary or quaternary amines, can potentially lead to the formation of NDSRI in the drug product.15 This reaction is reported to proceed under acidic pH conditions via the formation of reactive NO+ species. Therefore, depending upon the structure of the API, the excipients used and the manufacturing steps, there exists a risk of NDSRIs or simple NI formation in different DP.
One such example is the formation of N-nitroso orphenadrine, an NDSRI found in Orphenadrine citrate ER tablets. Orphenadrine is an anticholinergic agent used as a skeletal muscle relaxant. Orphenadrine citrate ER tablets were recalled from the market because the NDSRI in them was found to exceed the acceptable limits.22,23 Chemical structure of Orphenadrine and its NDSRI, N-nitroso orphenadrine, is given in Figure 3.
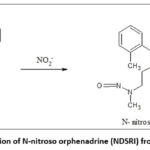 |
Figure 3: Formation of N-nitroso orphenadrine (NDSRI) from orphenadrine |
Another example of product recall from market due to NDSRI exceeding the acceptable intake, is in combination product of quinapril and hydrochlorthiazide (Accuretic™).22,24 Chemical structure of quinapril and its NDSRI, i.e. N-nitroso quinapril, are given in Figure 4.
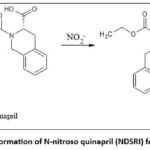 |
Figure 4: Formation of N-nitroso quinapril (NDSRI) form quinapril |
The formation of NI in selected DS and DP are discussed in detail in the following section.
Valsartan
NI in pharmaceutical drug products were for the first time observed in 2018 by the US FDA in Valsartan batches manufactured by Zhejiang Huahai Pharmaceutical (ZHP). Valsartan is an antihypertensive agent from angiotensin (AT1) receptor blocker class approved by the US FDA in 2005. According to the articles, the formation of NDMA is attributed to the change in the process employed in synthesis of Valsartan API. This change in the synthetic process was undertaken in order to improve the yield but in turn resulted in the NI contamination in the finished product. Figure 5 depicts the original route of synthesis for valsartan as per European Pharmacopoeia.25
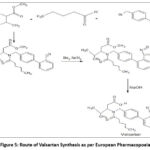 |
Figure 5: Route of Valsartan Synthesis as per European Pharmacopoeia |
As indicated, this process involved the use of tributyltin azide (Bu3SnN3) to form the tetrazole ring in valsartan, as indicated in Figure 6.25 The modified process involved the replacement of tributyltin azide with sodium azide, and dimethyl formamide (DMF) was employed as the solvent. Excess of sodium azide was quenched using sodium nitrite (NaNO2). Dimethyl amine (DMA) was reported to be present either as an impurity in DMF or was formed as a result of disproportionation reaction catalysed by ZnCl2.5,13 Figure 6 depicts the modified route of synthesis for valsartan to improve yield, but which in turn led to NDMA formation.
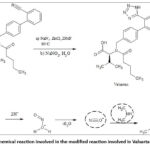 |
Figure 6: Chemical reaction involved in the modified reaction involved in Valsartan Synthesis |
Ranitidine
Ranitidine is an antihistaminic agent utilized for treatment of peptic ulcer that was approved by the US FDA in 1983. Its popular formulation Zantac® was found to be contaminated with nitrosamine impurities above acceptable limits in September 2019. The NI found was NDMA. Molecular structure of Ranitidine (Figure 7) contains both dimethyl amino (secondary amine) and nitrite functional groups.
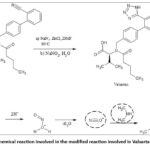 |
Figure 7: Structure of ranitidine |
Ranitidine undergoes autooxidation and generates DMA and nitrite. This availability of amine and nitrite functional groups provides favourable conditions to form NDMA by autooxidation of ranitidine which takes place in two steps with the involvement of two peroxy radials.26
Formation of DMA from Ranitidine:
The first step involves generation of ranitidine peroxyl radical formed by the reaction of ranitidine with peroxyl radical. This ranitidine peroxyl radical undergoes disproportionation to generate hydroxyl group which in turn undergoes hemiaminal rearrangement to form DMA. The sequence of the chemical reactions is given in Figure 08.
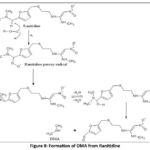 |
Figure 8: Formation of DMA from Ranitidine |
Figure 8 has been reproduced with permission from Paul Harmon, Ranitidine: A Proposed Mechanistic Rationale for NDMA Formation and a Potential Control Strategy, Journal of Pharmaceutical Sciences, Elsevier, 2022, https://doi.org/10.1016/j.xphs.2022.11.011. Permission granted by Paul Harmon.
Formation of Nitrite from Ranitidine:
Involves the reaction of the C=C bond adjacent to the nitro group with the peroxyl radical that results in the formation of an epoxide ring. This is followed by the rearrangement and liberation of the nitrite group 26 as indicated in Figure 9.
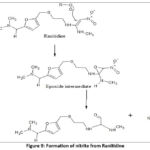 |
Figure 9: Formation of nitrite from Ranitidine |
Figure 9 has been reproduced with permission from Paul Harmon, Ranitidine: A Proposed Mechanistic Rationale for NDMA Formation and a Potential Control Strategy, Journal of Pharmaceutical Sciences, Elsevier, 2022, https://doi.org/10.1016/j.xphs.2022.11.011. Permission granted by Paul Harmon.
Afatinib
As mentioned previously in the article, Afatinib tablets are reported to generate NI due to nitrite contamination in the excipients. A comparative study was conducted by N. Golob et al.17, 2023 to evaluate the NI formation in film-coated tablets of Afatinib formulated using crospovidone obtained from two different vendors. The two groups of film-coated afatinib tablet preparations (containing crospovidone from two other companies having different nitrite contents) were subjected to accelerated stability studies, and the LC-HRMS technique was used to analyse the content of the NDMA generated. The crospovidone from one company had lower nitrite content as compared to the crospovidone manufactured by the other company. The results of the LC-HRMS analysis showed that the film-coated tablets prepared with crospovidone having a greater nitrite content exhibited greater NDMA formation compared to the group formulated with low-nitrite-containing crospovidone. This reflected that the excipient crospovidone was the nitrite source. Further study showed that secondary amine was formed due to the hydrolysis of afatinib, thereby generating DMA, which in turn reacted with nitrite from the excipient to form NDMA.17 The formation of NDMA was due to the degradation of the active ingredient and the nitrite from the excipient crospovidone. API afatinib has been shown to undergo hydrolysis, forming DMA. The presence of nitrite in crospovidone was evaluated using ion exchange chromatography, which revealed the presence of nitrite in the sample. The DMA and nitrite so formed lead to the formation of NDMA. Table 3 summarizes the formation of NI in Valsartan, Ranitidine and Afatinib.
Table 3: Summary of Nitrosamine Impurities Formation in Valsartan, Ranitidine and Afatinib
| API | Impurity Formed | Source | Recall Year |
| Valsartan | NDMA | Change in synthesis process to use of tributylin azide and DMF as solvent | 2018 |
| Ranitidine | NDMA | Autooxidation | 2019 |
| Afatinib | NDMA | Excipient crospovidone containing nitrite | Not applicable(Research study) |
Analytical Methods for Detection of NI
Due to the stringent regulatory requirements for controlling NI to a low level in drug products, sensitive analytical techniques are employed in the detection of NI in pharmaceuticals. Chromatographic methods are reported for the separation of NI from drug substances and drug products.
NI Separation using High Performance Liquid Chromatography (HPLC)
HPLC technique is used to separate NI on octadecyl silyl (ODS) columns, biphenyl columns, phenyl columns, phenylhexyl columns, and pentafluorophenyl propyl (PFP) columns. Detection is carried out using various detectors.
HPLC using UV Detector
Patil et al.27 have reported the use of a UV detector for detecting nitrosamine impurities in Losartan. The use of a diode array detector (DAD) at 230-233 nm and a UV detector at 228 nm for detecting NDMA in valsartan is reported by Li et al. and Al Kasem et al.28
HPLC using Fluorescence Detector
Dariusz Boscar et al.29 (2021) used a fluorimeter as a detector for HPLC separation of NDMA and NDEA in Enalapril maleate. The method involved precolumn derivatization wherein nitrosamines were derivatized using agents like dansyl chloride, fluorenylmethoxycarbonyl chloride. The latter was shown to give better sensitivity and selectivity. The separation was done by HPLC with PDA detector followed by an external fluorescence detector. The method reported quantitation limit of 0.038 and 0.050 µg/g for NDMA and NDEA respectively.
HPLC using Mass Spectroscopic Detector (LC-MS)
Most of the analytical methods used for detecting NI employ liquid chromatographic separation (HPLC or Ultra-Pressure Liquid Chromatography, UPLC) coupled with mass spectrometric detection. Hyphenated techniques, such as LC-MS or LC-MS/MS, are found to be the most commonly used methods for detecting NI in DPs due to their high sensitivity. Details of liquid chromatography analytical methods, including the stationary and mobile phases employed and mode of elution with MS detection, are given in Table 4.
Table 4: LC-MS methods used for separation and detection of NI in DS and DP
| Drug (s)/Drug Class | Nitrosamine Impurities Detected |
Stationary Phase | Mobile Phase | Elution Mode | Ionization Mode | LOD | LOQ | Reference Number |
| Rivoroxban(DS) | N-(2-hydroxyethyl)-N-phenylnitrous amide | ODS | Aqueous solution of methanoic acid (0.1%): methanol1:1 | Isocratic | – | 0.045 ng/mL | 0.15 ng/mL | 30 |
| Sitagliptin phosphate(DS) | 7-nitroso-3-(trifluoromethyl)-5,6,7,8-tetrahydro-[1,2,4]triazolo[4,3-a]pyrazine (NTTP) | ODS | A: 0.1% methanoic acid in 0.01M ammonium formate in water B: ACN |
Gradient | ESI | 0.037 ppm | 0.098 ppm | 31 |
| Candesartan cilexetilIrbesartanLosatan
Valsartan Olmesartan medoxomil (DS and DP) |
NDMANDEANDPA
NMBA NMEA NDiPA NDiPLA NDELA NEIPA NMOR NPYR NPIP |
HSS T3 (ODS) | A: Aqueous solution of methanoic acid (0.1%)B: 0.1% methanoic acid in acetonitrile/methanol(2:8) | Gradient | APCI | 20 ng/g | 50 ng/g | 32 |
| Mab A expressed by Chinese hamster ovary cells(DP) | MeNPNMOR NPYR NDEANPIP NEIPA NDPA NDIPA NMPA NDBA NDIBA NDBzA | – | – | – | – | 0.005-0.025 µg/L | 0.010-0.250 µg/L | 33 |
| Acebutol hydrochlorideBisoprolol fumarateMetoprolol succinate
Metoprolol tartarate Sotalol hydrochloride (DS) |
N-nitrosoacebutolol, N-nitrosobisoprolol, N-nitrosometoprolol, and N-nitrososotalol | HSS T3 | Aqueous solution of methanoic acid (0.1%) combined with methanol/ acetonitrile | – | ESI | 0.02–1.2 ppb | 2–20 ppb | 34 |
| Ranitidine(DS and DP) | NDMA | ODS | A: Aqueous solution of methanoic acid (0.1%)B: Methanol | Gradient | APCI | 0.01 ppm | 0.03 ppm | 35 |
| Varenicline tartrate(DS and DP) | NDMANDEAN-nitroso varenicline | ODS | A: Aqueous solution of methanoic acid (0.1%)B: Methanol | Gradient | APCI | 0.22 ppm | 0.66 ppm | 36 |
| Duloxetine hydrochloride(DS) | NDMA NDIPAN-nitroso duloxetine | Phenyl C18 | Methanoic acid and methanol mixture | Gradient | APCI | 0.0001 ppm | 0.003 ppm | 37 |
| Empagliflozin(DS and DP) | NDMANDEANEIA
NDIPA |
– | 0.1% aq. ammonia buffer: methanol5:95 | Isocratic | – | NDMA, NEIA, NDIPA0.03 ppmNDEA
0.02 ppm |
NDMA, NEIA, NDIPA0.03 ppmNDEA
0.02 ppm |
38 |
| Sartan group(DS and DP) | NDMANDEANDIPA
NMPA NDBA NMBA NEIPA 5-[4′-(azidomethyl)-[1,1′-biphenyl]-2-yl]-2H-tetrazole (AZBT) |
ODS | A: Aqueous solution of methanoic acid (0.1%)B: Methanol | Gradient | APCI | Range:NMBA, NDEA, NDIPA, NMPA, NDBA0.2-50 ng/mL
NDMA, NEIPA 0.5-50 ng/mL AZBT 2.0-100 ng/mL |
39 | |
| Rifampicin(DP) | 1-methyl-4-nitrosospiperazine (MNP) | Phenyl column | A: 10mM ammonium formate (pH 9)B: Methanol | Gradient | ESI | 0.51 ng/mL | 1.52 ng/mL | 40 |
| RifampinRifapentine(DS and DP) | MNPCPNP | Phenylhexyl column | A: 10mM ammonium formate (pH 9)B: Methanol | Gradient | ESI | – | 0.05 ppm | 41 |
| Metformin tablet formulation(DP) | NDMANDEANDPA
NDBA NPYR NPIP NMOR |
ODS | A: Aqueous solution of methanoic acid (0.1%)B: Acetonitrile | Gradient | – | – | 0.1-5.1 pg/mg API | 42 |
| Nizatidine(DS and DP) | NDMA | – | – | – | – | 0.25 ng/mL | 0.50 ng/mL | 43 |
| Levocetrizine(DS and DP) | NPIP | F5 column | A: 2mM ammonium formate in waterB: Acetonitrile | Gradient | ESI | – | 1 ng/mL | 44 |
NDPA: N-nitrosodipropylamine, NDIBA: N-nitrosodiisobutylbutylamine, NDBzA: N-nitrosodibenzylamine, NDiPLA: N-nitrosodiisopropanolamine, NDELA: N-nitrosodiethanolamine, NEIPA: N-nitrosoethylethylpropylamine, MeNP: 4-methy1-nitrosopiperazine, NEIA: N-nitrosoethylisopropylamine,s MNP: 1-methyl-4-nitrosopiperazine, CPNP: 1-cyclopentyl-4-nitrosopiperazine
Gas Chromatography using MS Detector (GC-MS)
Low molecular weight NI, which can be readily volatilized, are reported to be analysed by Gas chromatography using an MS detector (GC-MS). Kalauz et al.45 utilized the GC-MS method to determine 12 low-molecular-weight nitrosamine impurities, including NDMA, NDEA, NDPhA, N-nitrosodipropylamine (NDPA), NMEA, NMOR, NPIP, N-nitroso-ethylisopropylamine (EIPNA), N-nitroso-diisopropylamine (DIPNA), N-nitroso-N-methylaniline (NMPA), 1-methyl-4-nitrosopiperazine (MeNP), and NPYR. The method reported a linearity range from 12-120 ng/mL. Shu-Han Chang46 (2022) employed a GC-MS/MS method, utilising the headspace-solid phase microextraction technique (HS-SPME), for the determination of fourteen nitrosamine impurities in forty-four pharmaceuticals, with a limit of quantitation of 0.05 µg/g. Liu et al.47 (2021) developed a sensitive GC-MS/MS for the simultaneous determination of four nitrosamine impurities in pharmaceuticals with detection limits from 0.002-0.150 ppm. Anna B. Witkowska et al.48 (2022) developed a sensitive method using GC-MS for the simultaneous determination of nine NIs in Cilostazol, Sunitinib malate, and Olmesartan medoxomil, with a quantitation limit of up to 21.6 ng/mL. Tummala and Amgoth49 (2021) reported the estimation of four NIs in valsartan using the GC-MS technique, with the method having a detection limit of 0.02-0.03 ppm and a quantitation limit of 0.06-0.09 ppm.
Supercritical Fluid Chromatography with MS Detection (SFC-MS)
Schmidtsdorff et al.50 (2019) have reported Quality by Design (QbD) approach and supercritical fluid chromatography (SFC) for the detection of NI in valsartan and losartan. The method utilized ultra-performance convergence chromatography (UPC) with PDA detector. Detection was performed using MS detection. The method was capable of detecting NDMA, NDEA, NMEA, NDPA, NDBA, N-nitrosodiphenylamine (NDPhA), NPYR, NPIP and NMOR with detection limits of 4.55, 1.58, 1.81, 0.24, 0.34, 0.22, 3.71, 2.26 and 4.20 ng/mL, respectively.
A concise comparative view of different analytical techniques based on sensitivity of detection and selectivity for NI in pharmaceuticals is illustrated in Table 5.
Table 5: Brief Summary of Different Analytical Techniques for Nitrosamine Impurities Detection
| Name of the analytical technique | Sensitivity | Suitability |
| LC-MS/MS | Highly sensitivity | Ideal for non-volatile and thermolabile nitrosamines, most widely used for estimation of NI in DS and DP |
| GC-MS | High | Suitable for volatile nitrosamine such as NDMA. Good resolution, but limited to thermostable and volatile NI |
| SFC-MS | High | Useful for complex matrices and chiral separation |
| LC-UV | Low | Low sensitivity, not preferred for trace level analysis of NI in pharmaceuticals |
| LC-Fluorescence | Low | Applicable after derivatization with suitable reagent |
Discussion
NI in pharmaceuticals is a potential health threat because of the mutagenic and carcinogenic properties it possesses. This is attributed to the irreversible alkylation of DNA bases upon activation by CYP450 enzymes. NI in pharmaceuticals is attributed to APIs containing amine functionality (mainly secondary amines), excipients contaminated with nitrite, and APIs containing both amine and nitrite groups (e.g., ranitidine). Due to the mutagenic nature of NI, they need to be controlled to trace levels in pharmaceuticals. Regulatory agencies exercise strict control over levels of NI in pharmaceuticals; hence, accurate and precise quantitation of NI in DS and DP is crucial. To accurately detect and quantify NI in pharmaceuticals at such low levels, highly sensitive and advanced analytical techniques are required. There is a possibility of false positive detection of NI during analysis of pharmaceuticals due to interference from solvents such as DMF, interference from the complex matrix encountered in pharmaceuticals which can mimic NI, interference from the excipients. Mostly the separation is reported by various chromatographic techniques including LC, GC and SFC. Owing to the presence of weakly absorbing chromophoric group, mainly showing absorption maxima below 220 nm (where maximum solvents show interference), LC separation coupled with UV detection have low sensitivity. However, fluorometric methods involving post separation derivatization with agents such as dansyl chloride have been reported to provide low sensitivity detection. The most sensitive detection and quantification of NI is provided by MS detection capable of quantifying upto ng/mL level (parts per billion). Gas chromatographic methods coupled with MS detection are mainly reported for the volatile, low molecular weight NI. LC-MS methods provide the most reliable means to detect and quantify the presence of NI at such low levels in pharmaceuticals, hence LC methods coupled with MS detection have been mainly reported for the detection and quantitation of NI and NDSRIs in pharmaceuticals. It is equally important that the samples are handled properly maintaining their stability by proper storage, protection from light and heat to ensure accuracy in their detection.
Conclusion
Carcinogenic potential associated with NI has seriously impacted the pharmaceutical industry, with significant concern increasing day by day. This review provides a comprehensive overview of nitrosamine impurities, elucidating their types, toxicological concerns, and acceptable limits, alongside a detailed analysis of the chemistry underlying their formation and the critical contributors to their generation. Through an in-depth discussion of advanced analytical techniques, including HPLC-UV, HPLC-fluorescence detection, LC-MS, GC-MS, and SFC-MS, this article highlights state-of-the-art methodologies for the precise and reliable detection of nitrosamine impurities, supported by relevant case studies of Valsartan, Ranitidine, and Afatinib.
Addressing nitrosamine impurities effectively requires a combination of stringent regulatory guidelines, proactive risk assessments, and advancements in analytical capabilities. Since, formation of these impurities can take place during storage as well by migration of nitrite from packaging material, continuous monitoring of NI formation in DP and post-marketing surveillance is essential for DP susceptible to NI formation. By exploring the intricate interplay between their formation pathways and detection strategies, this review aims to equip researchers and industry professionals with the necessary insights to mitigate nitrosamine impurities contamination in pharmaceuticals. Ultimately, the collective efforts of scientists, regulatory authorities, and pharmaceutical manufacturers are pivotal in ensuring drug safety and maintaining public health.
Acknowledgement
Authors acknowledge Dr. Vilasrao J. Kadam, Principal, Bharati Vidyapeeth’s College of Pharmacy, Navi Mumbai for his contribution in drafting of the article.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Figures 8 and 9 have been reproduced with permission from Paul Harmon, Ranitidine: A Proposed Mechanistic Rationale for NDMA Formation and a Potential Control Strategy, Journal of Pharmaceutical Sciences, Elsevier, 2022, https://doi.org/10.1016/j.xphs.2022.11.011. Permission granted by Paul Harmon.
Authors Contribution
- Vineeta Khanvilkar: Selection of the title and pathway for the data search.
- Deepali Jagdale: Checking the chemistry part of the data collected
- Abhay Shirode: Drafting of the article
- Ammara Sahibole: Systematic literature search, study of the data and its compilation, typing of the manuscript, drawing the chemical structures
- Gayatri Vinchurkar: Drafting of the article
References
- International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). Impurities in New Drug Substances Q3A(R2). Quality Guidelines. https://database.ich.org/sites/ default/files/ Q3A%28R2%29%20Guideline.pdf. 15 March 1994. 25 October 2006. May 2025.
- International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). Impurities in New Drug Products Q3B(R2). Quality Guidelines. https://database.ich.org/sites/default/ files/Q3B%28R2%29%20Guideline.pdf. 29 November 1995. 2 June 2006. May 2025.
- Sedlo I, Kolonić T and Tomić S. Presence of nitrosamine impurities in medicinal products. Arh Hig Rada Toksikol. 2021;72(1):1-5.
CrossRef - United States Food and Drug Administration (US FDA). Control of Nitrosamine Impurities in Human Drugs. Guidance Document. https://www.fda.gov/media/141720/download. 24 February 2021. September 2024. May 2025.
- Cioc R, Joyce C, Mayr M and Bream R. Formation of N-nitrosamine drug substance related impurities in medicines: a regulatory perspective on risk factors and mitigation strategies. OPR&D. 2023;27(10):1736-1750.
CrossRef - International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). Mutagenic Impurities M7(R2). Multidisciplinary Guidelines. https://database.ich.org/sites/default/ files/ICH_M7%28R2%29_Guideline_Step4_2023_0216_0.pdf. 6 February 2013. 3 April 2023. May 2025.
- National Toxicology Program U.S. Department of Health and Human Services. Report on Carcinogens. 15th Report on Carcinogens. https://ntp.niehs.nih.gov/research/assessments/cancer/roc. 1980. 21 December 2021. May 2025.
- Vogel M and Norwig J. Analysis of genotoxic N-nitrosamines in active pharmaceutical ingredients and market authorized products in low abundance by means of liquid chromatography–tandem mass spectrometry. Pharm. Biomed. Anal. 2022;219:114910.
CrossRef - Yamazaki H, Inui Y, Yun C, Guengerich F and Shimada T. Cytochrome P450 2E1 and 2A6 enzymes as major catalysts for metabolic activation of N-nitrosodialkylamines and tobacco-related nitrosamines in human liver microsomes. Carcinogenesis. 1992;13(10):1789-1794.
CrossRef - Beard J and Swager T. An organic chemist’s guide to N-nitrosamines: their structure, reactivity, and role as contaminants. Org. Chem. 2021;86(3):2037-2057.
CrossRef - Joshua P. G. Acceptable Daily Intake. 2024;1:15-16.
CrossRef - International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). Stability Testing of New Drug Substances and Products Q1A(R2). Quality Guidelines.
https://database.ich.org/sites/default/files/Q1A%28R2%29%20Guideline.pdf. 16 September 1992. 6 February 2003. May 2025. - Ray A, Atal S and Sadasivam B. Understanding the molecular–pharmaceutical basis of sartan recalls focusing on valsartan. Cardiol. Sci. 2020;2020(2):4559.
CrossRef - Abe Y, Yamamoto E, Yoshida H, et al. Temperature-dependent formation of N-nitrosodimethylamine during the storage of ranitidine reagent powders and tablets. Phar. Bull. 2020;68(10):1008-1012.
CrossRef - The International Pharmaceutical Excipients Council Federation. The Role of Excipients in Determining N-Nitrosamine Risks for Drug Products. IPEC Position Paper. https://www.ipec-europe.org/uploads/ publications/20240227-if-nitrosamines-position-paper-v2-f-1709022506.pdf. March 2022. February 2024. May 2025.
- Boetzel R, Schlingemann J, Hickert S, et al. A nitrite excipient database: a useful tool to support N-nitrosamine risk assessments for drug products. Pharm. Sci. 2023;112(6): 1615-1624.
CrossRef - Golob N, Peterlin S and Roškar R. NDMA formation due to active ingredient degradation and nitrite traces in drug product. Pharm. Sci. 2023;112(5):1277-1286.
CrossRef - Golob N, Ross M and Roškar R. Nitrocellulose blister material as a source of N-nitrosamine contamination of pharmaceutical drug products. J. Pharm. 2022;618:121687.
CrossRef - Nurazzi N, Shazleen S, Norrrahim M, et al. Molecular dynamics of nanocellulose-based nanocomposites: a review. 2023:187-213.
CrossRef - Ashworth I, Blanazs A, Byrne J, et al. Approaches and considerations for the investigation and synthesis of N-nitrosamine drug substance-related impurities (NDSRIs). OPR&D. 2023;27(10):1784-1791.
CrossRef - United States Food and Drug Administration (US FDA). The daily Journal of United States Government. Identification, Assessment, and Control of Nitrosamine Drug Substance-Related Impurities in Human Drug Products; Establishment of a Public Docket; Request for Comments. Federal Register. https://www.federalregister.gov/ documents/2023/05/04/2023-09526/identification- assessment-and-control-of-nitrosamine-drug- substance-related-impurities-in- human-drug. 5 April 2023. May 2025.
- Elder D. Nitrosamine drug substance-related impurities (NDSRIs). Pharm. Rev. 2022;27(3): 5.
- US Food and Drug Administration. Sandoz, Inc. Issues Nationwide Recall of 13 Lots of Orphenadrine Citrate 100 mg Extended Release Tablets Due to Presence of a Nitrosamine Impurity. Company Announcement. https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/sandoz-inc-issues-nationwide-recall-13-lots-orphenadrine-citrate-100-mg-extended-release-tablets-due. 22 March 2022. May 2025.
- US Food and Drug Administration. Pfizer Voluntary Nationwide Recall of Lots of Accuretic (Quinapril HCl/Hydrochlorothiazide), Quinapril and Hydrochlorothiazide Tablets, and Quinapril HCl/Hydrochlorothiazide Tablets Due to N-Nitroso-Quinapril Content. Company Announcement. https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/pfizer-voluntary-nationwide-recall-lots-accuretic-quinapril-hclhydrochlorothiazide-quinapril-and. 22 March 2022. May 2025.
- Parr M and Joseph J. NDMA impurity in valsartan and other pharmaceutical products: Analytical methods for the determination of N-nitrosamines. Phar. Biomed. Anal. 2019;164:536-549.
CrossRef - Harmon P. Ranitidine: a proposed mechanistic rationale for NDMA formation and a potential control strategy. Pharm. Sci. 2023;112(5):1220-1224.
CrossRef - Aldawsari F, Alshehry Y and Alghamdi T. N-nitrosodimethylamine (NDMA) contamination of ranitidine products: a review of recent findings. JFDA. 2021;29(1):39-45.
CrossRef - Li W, Chen N, Zhao Y, et al. Online coupling of tandem liquid-phase extraction with HPLC-UV for the determination of trace N-nitrosamines in food products. Methods. 2018;10(15):1733-1739.
CrossRef - Boczar D, Wyszomirska E, Zabrzewska B, Chyła A and Michalska K. Development and validation of a method for the semi-quantitative determination of N-nitrosamines in active pharmaceutical ingredient enalapril maleate by means of derivatisation and detection by HPLC with fluorimetric detector. Sci. 2021;11(16):7590.
CrossRef - Baksam V, Saritha N, Devineni S, et al. A critical N-nitrosamine impurity of anticoagulant drug, rivaroxaban: synthesis, characterization, development of LC–MS/MS method for nanogram level quantification. Chromatographia. 2022:1-10.
CrossRef - Wang Z, Hu S, Wu X, He Z, Ke C and Hu M. A highly sensitive LC-MS/MS method for the determination and quantification of a recently identified N-nitrosamine impurity in the sitagliptin phosphate monohydrate active pharmaceutical ingredient. Methods. 2023;15(3):256-260.
CrossRef - Chang S, Chang C, Wang L, et al. A multi-analyte LC-MS/MS method for screening and quantification of nitrosamines in sartans. JFD. 2020;28(2):292-301.
CrossRef - Xie Y, Zhang L, Hou W, et al. A novel method for monitoring N-nitrosamines impurities using NH2-MIL-101 (Fe) mediated dispersive micro-solid phase extraction coupled with LC-MS/MS in biopharmaceuticals. Pharm. Sci. 2023;112(11):2783-2789.
CrossRef - Marlés‐Torres A, López‐García R, Bessa‐Jambrina S and Galán‐Rodríguez C. Ultra‐high‐speed liquid chromatography combined with mass spectrometry detection analytical methods for the determination of nitrosamine drug substance‐related impurities. Sep. Sci. 2023;46(13):2300125.
CrossRef - Rao G, Ramadevi D, Rao B, Rajana N and Basavaiah K. Novel stability indicating LC-MS method for N-Nitroso dimethyl amine genotoxic impurity quantification in ranitidine drug substance and drug product. Appl. Pharm. Sci. 2022;12(7):106-114.
CrossRef - Vidyamani U, Ramachandran D, Babu M, Raju S and Maheswar K. Development and Validation of a Rapid and Sensitive LC-APCI-MS/MS Method for the determination of three N-nitrosamine impurities in Varenicline tartrate drug substance and drug products. 2024;17(4):1703-1710.
CrossRef - Gopireddy R, Maruthapillai A and Tamilselvi M. Determination of potential nitrosamines NDMA, NDIPA and N-Nitroso Duloxetine in Duloxetine Hydrochloride by LC-MS/MS using APCI source. Today Proc. 2022;68:A7-A20.
CrossRef - Mohammed M and Gandhimathi R. A validated LC-MS method for the estimation of genotoxic nitroso amines impurities in empagliflozin. J. Health Sci. 2022;6(S9):2694-2710.
CrossRef - Luan N, Khuyen V and Tuan N. High‐Performance Liquid Chromatography–Tandem Mass Spectrometry Method Development and Validation for Simultaneous Determination of Seven Nitrosamine and Azidomethyl‐Biphenyl‐Tetrazole Impurities in Losartan. J. Sep. Sci. 2024;47(19):e202400277.
CrossRef - Witkowska A, Wołczyńska A, Lis-Cieplak A and Stolarczyk E. Development and Validation of LC-MS/MS Method for the Determination of 1-Methyl-4-Nitrosopiperazine (MNP) in Multicomponent Products with Rifampicin—Analytical Challenges and Degradation Studies. Molecules. 2023;28(21):7405-7420.
CrossRef - Li Q, Xu Q, Lo N, et al. Mitigating matrix effects for LC-MS/MS quantification of nitrosamine impurities in rifampin and rifapentine. JPBA Open. 2024;3:100027.
CrossRef - Ishizaki A, Ozawa K and Kataoka H. Simultaneous analysis of carcinogenic N-nitrosamine impurities in metformin tablets using on-line in-tube solid-phase microextraction coupled with liquid chromatography-tandem mass spectrometry. Chromatogr. A. 2023;1710:464416.
CrossRef - Dhorajiya S, Goswami J, Dudhatra B and Thummar K. Novel method for monitoring of carcinogenic impurity of N‐nitrosamine in nizatidine pharmaceutical products using ultra high‐pressure liquid chromatography triple quadrupole mass spectrometry. RCM. 2024;38(19):e9884.
CrossRef - Lim Y, Kim A, Lee Y and Cho H. Development of a sensitive LC-MS/MS method for determination of N-nitrosopiperazine in levocetirizine. Methods. 2024;16(38):6494-6500.
CrossRef - Kalauz A, Tiringer K, Horváth V and Kapui I. Simultaneous determination of low molecular weight nitrosamines in pharmaceutical products by fast gas chromatography mass spectrometry. Chromatogr. A. 2023;1708:464323.
CrossRef - Chang S, Ho H, Chang C, et al. Evaluation and optimization of a HS-SPME-assisted GC-MS/MS method for monitoring nitrosamine impurities in diverse pharmaceuticals. Phar. Biomed. Anal. 2022;221:115003.
CrossRef - Liu J, Xie B, Mai B, et al. Development of a sensitive and stable GC-MS/MS method for simultaneous determination of four N-nitrosamine genotoxic impurities in sartan substances. JAST. 2021;12:1-8.
CrossRef - Witkowska A, Giebułtowicz J, Dąbrowska M and Stolarczyk E. Development of a sensitive screening method for simultaneous determination of nine genotoxic nitrosamines in active pharmaceutical ingredients by GC-MS. J. Mol. Sci. 2022;23(20):12125.
CrossRef - Tummala S and Amgoth K. Development of GC-MS/MS Method for Simultaneous Estimation of Four Nitrosoamine Genotoxic Impurities in Valsartan. Pharm. Sci. 2022; 19(4): 455.
CrossRef - Schmidtsdorff S and Schmidt A. Simultaneous detection of nitrosamines and other sartan-related impurities in active pharmaceutical ingredients by supercritical fluid chromatography. JPBA. 2019;174:151-160.
CrossRef







