Yuriy Nikolaevich Lezhnin1,2, Dmitry Sergeevich Kravchenko1,2, Anna Evgenyevna Ivanova1, Yulia Evgenyevna Kravchenko1 and Elena Ivanovna Frolova1
1Shemyakin-Ovchinnikov Institute of Bioorganic Chemistry of the Russian Academy of Sciences, 117997, Russian Federation, Moscow, GSP-7, Miklukho-Maklaya Str., 16/10.
2Engelhardt Institute of Molecular Biology, Russian Academy of Sciences 119991, Russia, Moscow, Str. Vavilova, 32.
DOI : https://dx.doi.org/10.13005/bpj/1087
Abstract
Dendritic cells (DC) play a key role in regulation of adaptive immune response by providing effective protection from various infectious agents and pathologies. The main function of dendritic cells is to absorb and present foreign antigens to effector cells of the immune system, such as CD8+ T-lymphocytes. To date, either studying of functions of dendritic cells or their utilization as cellular vaccines in cancer immunotherapy have been performed with various lentivirus-based genetic constructs for silencing or activation of certain genes. Successful infection of dendritic cells is constrained by expression of specific SAM domain and HD domain-containing protein 1 (SAMHD1) by these cells. In this work, we have developed an optimized helper vector for obtaining substantial titers of Vpx-containing lentiviral particles that can efficiently infect human monocytes and dendritic cells.
Keywords
Dendritic cells; lentiviral transduction; immunotherapy; vpx, SAMHD1
Download this article as:| Copy the following to cite this article: Lezhnin Y. N, Kravchenko D. S, Ivanova A. E, Kravchenko Y. E, Frolova E. I. Construction of an Optimized Helper Plasmid Containing Vpx for Lentiviral-Mediated Transduction of Dendritic Cells. Biomed Pharmacol J 2017;10(1). |
| Copy the following to cite this URL: Lezhnin Y. N, Kravchenko D. S, Ivanova A. E, Kravchenko Y. E, Frolova E. I. Construction of an Optimized Helper Plasmid Containing Vpx for Lentiviral-Mediated Transduction of Dendritic Cells. Biomed Pharmacol J 2017;10(1). Available from: http://biomedpharmajournal.org/?p=13649 |
Introduction
Dendritic cells (DC) are an important segment of the immune system that links the innate and acquired immune responses. Dendritic cells can absorb foreign antigens of various nature, and present their antigenic determinants to the effector cells of the immune system [1]. For modern clinical practice, dendritic cells are of great interest due to their ability to present specific tumor antigens, and to activate the anti-tumor response in the patient [2].
Mainly, dendritic cell vaccines are prepared according to the standard scheme, where monocytes in the peripheral blood of the patient are differentiated into dendritic cells under the influence of a set of cytokines, and are further loaded (pulsed) by purified preparation of a specific tumor antigen, or by the total lysate of tumor cells [3]. However, the most efficient method of loading tumor antigens is the transduction of dendritic cells with antigen containing viral vectors that ensure more efficient presentation of the first class major histocompatibility complex (MHCI) by molecules [4]. As a rule, lentiviral vectors based on HIV1 are used as viral expression vectors for transduction of cells; they allow to integrate the target sequence into the genomic DNA of a cell, and ensure constant and stable level of gene expression. However, using standard lentiviral vectors for creating dendritic-cell vaccines is limited by low efficiency of transduction of the dendritic cells themselves [5]. Low efficiency of transduction is associated with expression of specific protein SAMHD1 in dendritic cells, the phosphohydrolase that decomposes dNTP to inorganic phosphate and nucleoside, thereby reducing the pool of dNTPs and preventing reverse transcription of the viral genome. Besides, SAMHD1 features nuclease activity, and results in degradation of the viral RNA, reducing the probability of integrating the target sequence to the minimum [6]. To increase the level of dendritic cells transduction many times without complex optimization of the infection conditions, spinoculation and introduction of polybrene, the method of creating lentiviral virions associated with protein Vpx may be used. Vpx is a virion-associated protein characteristic of human immune deficiency virus type 2 (HIV-2) and most immune deficiency viruses of monkeys (Simian Immune Deficiency Virus SIV), but Vpx is absent in HIV-1 [7]. Getting into the cell, Vpx interacts with SAMHD1 and promotes its association with CRL4DCAF1 E3 ubiquitin ligase. Next, SAMHD1 is subjected to proteasomal degradation, thereby allowing the virus to replicate and integrate into the genome of the host cell.
In this work, we have developed a bicystronic helper lentiviral vector where the sequence of the Vpx gene is associated with the sequence of transactivator protein Rev via the T2A peptide. Such a system allows obtaining viral particles containing many molecules of the Vpx protein, which results in efficient transduction and stable expression of the target gene’s sequence in human dendritic cells.
Materials and methods
Cell culture
Adherently growing human embryonic kidney (HEK) cell lines HEK 293T were cultured in Dulbecco’s modified Eagle’s medium (DMEM, high glucose (4.5 g/L) with 2 mM L-glutamine) supplemented with 10% (v/v) heat inactivated fetal calf serum (FCS) (Thermo scientific, USA) and 1% (v/v) of a 10.000 U/mL penicillin and 10 mg/mL streptomycin stock solution (Paneco, Russia) at 37°C, 5% CO2. Human monocyte and monocyte-derived DCs were cultured in serum free AIM-V culture medium (Themo Fisher Scientific, USA) at 37°C, 5% CO2. Peripheral blood mononuclear cells were prepared from the healthy donor by Ficoll density gradient centrifugation. Monocytes population were isolated by EasySep human CD14 positive selection kit (StemCell Technologies, USA) and cultured for 4 days in medium supplemented with 50 ng/ml rhGM-CSF (Peprotech, UK) and 50 ng/ml rhIL-4 (Peprotech, UK) to generate monocyte-derived DCs. The medium was replenished every 3 days with fresh cytokines.
RNA isolation and cDNA preparation
Total RNA was extracted from PBMC using TRIzol isolation reagent (Invitrogen, USA). The RNA was reverse transcribed using oligo (dT)20 by incubation with 200 U of SuperScript III reverse transcriptase at 50°C at 50 min in the presence of 25 mM MgCl2, 10x RT Buffer, 10 mM dithiothreitol (DTT), 40 U of RNase OUT (RNase inhibitor) and 10 mM dNTP Mix. The obtained cDNA was used for amplification of EF1alpha promoter region sequence.
Plasmid construction
PLenti-EF1alpha-tagRFP The sequence of the EF1alpha promoter was amplified from the cDNA fraction of monocytes in a healthy donor’s peripheral blood with the use of Tersus HS DNA Polymerase (Evrogen, Russia) and cloned by sites ClaI and XbaI in the lentiviral vector pLenti-MCS-NP-puro. The sequence of gene tagRFP was amplified with pTagRFP-N (Evrogen, Russia) and cloned into lentiviral vector pLenti-EF1a-MCS-NP-puro by sites XbaIand BamHI.
Modified pRSV-Rev Modified vector pRSV-Rev is a pRSV-Rev vector with re-cloned sequence of transactivator protein Rev, the C-end region of which contains the sequence of “self-cleaving” T2A peptide and additional restriction site XbaI.
pCMV-Rev-T2A-Vpx The sequence of the Vpx gene was amplified and cloned by sites BglII XbaI in modified plasmid pRSV-Rev split by similar sites. To increase the efficiency of packing Vpx protein into capsid of virus in the Forward primer sequence, a sequence of the Gag/Pol of packaging motif was introduced. In addition, to identify Vpx protein packaging into the capsid of lentiviral particles with the use of Western blotting, FLAG sequence of the epitope was placed on the C-terminal region of Vpx amplified sequence (sequences of all primers are shown in Table 1).
Virus preparation and infection
To prepare lentiviral stocks, HEK293T cells were co-transfected with lentiviral vector pLenti-EF1alpha-tagRFP and helper plasmids pRSV-Rev-T2A-Vpx, pVSV-G, pMDLg/pRRE with the use of polyethyleneimine 40.000 (PEI 40) (Polyscience, USA). After 48 hours of cultivating, cell supernatant was collected, filtered through a 0.45 µm filter, and the virus was concentrated by the method of ultracentrifugation at 50,000 g for 30 min. The titer of the tagRFP virus was determined for 293T cells with the use of cytometry. The virus stock was frozen in the RPMI 1640 medium supplemented with 10% heat inactivated fetal calf serum at -80°C.
The dendritic cells were infected in serum free AIM-V culture medium for 8 hours. Then the cells were collected, centrifuged at 400 g for 10 min and placed in serum free AIM-V culture medium with addition of rhGM-CSF (50 ng/mL) and rhIL-4 (50 ng/mL). 48 hours after infection, expression of fluorescent protein tagRFP was analyzed.
SDS-PAGE electrophoresis and Western blotting
The collected fractions were mixed with sample buffer and run on a 10% SDS–PAGE gel under reducing conditions. The separated proteins were transferred to PVDF (Polyvinylidene fluoride) membrane (Hybond GE Healthcare, USA). PVDF was blocked with PBS containing 3% BSA and incubated with primary monoclonal mouse antibodies specific to FLAG-tag for 1 h at room temperature followed by three steps of washing by PBS containing 0.1% Tween 20 and incubated with peroxidase-labeled rabbit anti-mouse antibodies. Peroxidase activity was measured by picoLUCENT Luminol Solution (GBioscience, USA) and visualized by GeneGnome (Syngene, USA).
Table 1: Primer sequences used for plasmid construction
| Target | Primer sequence (5’-3’) | Restriction sites |
| EF1alpha promoter (F) | GAGAGAATCGATAGCGTGAGGCTCCGGTGCCCGTCAGTGG | ClaI |
| EF1alpha promoter (R) | AGAGAGTCTAGAAGGAATTCTCACGACACCTGAAATGGAA GAAAAAAAC | XbaI |
| tagRFP (F) | ATATATCTAGAATGGTGTCTAAGGGCGAAGAGC | XbaI |
| tagRFP (R) | ATATAGGATCCTTAATTAAGTTTGTGCCCCAGTTTGCT | BamHI |
| Rev (F) | ATATACTCGAGATGGCAGGAAGAAGCGGAGAC | XhoI |
| Rev-T2A (R1) | TGGAGGAGAATCCCGGCCCTTTCTTTAGCTCCTGACTCCAATATTGTAGG | |
| Rev-T2A (R2) | ATATAAGATCTGAGGGCAGAGGAAGTCTTCTAACATGCGGTGACGTGGAGGAGAATCCCG | BglII |
| Vpx (F) | ATATAAGATCTAAAGCAAGAGTTTTGGCTGAAGCA | BglII |
| Vpx (R) | ATATATCTAGAGAGGGCAGAGGAAGTCTTCTAACATGCGGTGACGTGGAGGAGAATCCCGGCCCTTGCTAGTCCTGGAGGGGG | XbaI |
Results
Vpx-containing lentiviral vectors ensure the most efficient transduction and stable expression of monocyte-derived human dendritic cells in vitro. In order to test this concept, we had built a versatile lentiviral vector, where the sequence of the gene Vpx was fused with the sequence of the gene of transactivating protein Rev through a specific sequence of T2A, which ensured fusing of the fused proteins during translation of the mRNA transgene (Figure 1). The Vpx gene sequence also includes the sequence of the packaging motif – the Gag/Pol packaging motif, which promotes integration of protein Vpx into the viral particle.
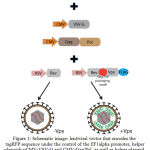 |
Figure 1: Schematic image: lentiviral vector that encodes the tagRFP sequence under the control of the EF1alpha promoter, helper plasmids pCMV-VSV-G and CMV-Gag/Pol, as well as helper plasmid containing the sequence of the transactivator protein gene fused with the sequence of protein Vpx pRSV-Rev-T2A-Vpx and helper unmodified plasmid pRSV-Rev.
|
As a result of co-transfecting cells of the HEK293T line by lentiviral vector pLenti-EF1alpha-tagRFP-NP-puro and packaging helper plasmids pVSV-G, pMDLg/pRRE, pRSV-Rev-T2A-Vpx, the virus containing Vpx protein was obtained. In addition, for analyzing the efficiency of dendritic cells transduction with Vpx-containing virus, viral particles had been obtained where Vpx was absent.
The content of Vpx protein in the composition of the virions was determined with the use of Western blot on the FLAG tag, which had been fused with the C-terminal domain of Vpx (Figure 2).
 |
Figure 2: Western blot analysis of viral stocks 1 – monitoring, virus EF1alpha-tagRFP-NP-puro without Vpx, 2 – pseudo-virus with Vpx, 3 – virus EF1alpha-tagRFP-NP-puro with Vpx
|
Monoclonal mouse FLAGtag antibodies (R and D systems, USA) and anti-mouse HRP-conjugated antibodies (Santa Cruz Biotechnology, USA) were used for the western blot.
Despite the efficiency of self-splitting sequence of the T2A peptide, not all Rev-T2A-Vpx fusion becomes split during expression of the helper plasmid pRSV-Rev-T2A-Vpx, and part of the fusion becomes packaged into viral particles. However, most Vpx protein molecules get successfully packaged as part of the virion in the form of single molecules.
Subsequently, the monocyte derived dendritic cells were divided into 2 groups and infected with Vpx+ and Vpx – viral stocks. Expression of fluorescent protein tagRFP was analyzed 48 hours after infecting by the method of flow cytometry and fluorescence microscopy (Figure 3 shows a micrograph of infected dendritic cells with tagRFP-Vpx positive and tagRFP-Vpx negative viruses). As a result of infection, 42.5% of the population of monocyte derived dendritic cells infected with Vpx-containing lentiviral particles expressed fluorescent protein tagRFP.
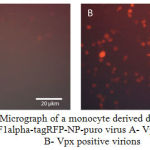 |
Figure 3: A – Micrograph of a monocyte derived dendritic cells infected with the EF1alpha-tagRFP-NP-puro virus A- Vpx negative virions, B- Vpx positive virions
|
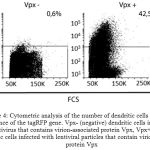 |
Figure 4: Cytometric analysis of the number of dendritic cells expressing a sequence of the tagRFP gene. Vpx- (negative) dendritic cells infected with a lentivirus that contains virion-associated protein Vpx, Vpx+ (positive) dendritic cells infected with lentiviral particles that contain virion-associated protein Vpx
|
Discussion
One of the main problems in the modern immunotherapy based on the use of dendritic cells is the low efficiency of these cells’ transduction with standard HIV-1 lentiviral vectors. This effect is primarily due to dendritic cell expressing specific protein SAMHD1, which can hydrolyze dNTP and thereby prevent reverse transcription of the viral genome, and consequently inhibits its integration into the host cell genome. As already mentioned, one of the most efficient ways of increasing transduction of dendritic cells is the introduction of the Vpx protein into lentiviral particles, which can initiate proteasomal degradation of SAMHD1.
In this paper, we have designed a universal packaging plasmid that ensures effective packaging of Vpx protein into the structure of the lentiviral virion. We have obtained the modified helper vector pRSV-Rev-T2A-Vpx, which is a fusion of sequences of transactivator protein Rev and protein Vpx, which are fused by a sequence of self-splitting peptide T2A. This structure ensures the high level of incorporating Vpx into the structure of viral particles due to expression of a sequence Rev and Vpx protein in the form a bicistronic sequence. Besides, expression of Vpx and Rev on the same plasmid allows preserving the number of helper plasmids that is characteristic of third generation lentiviral vectors, thereby increasing the probability of all constructs getting into cells of line HEK293T in the process of co-transfection.
Conclusion
We have developed a new helper lentiviral vector that makes it possible to obtain HIV1-lentiviral particles containing HIV2-virion associated Vpx protein, which greatly increases the efficiency of human dendritic cells transduction. Thus, this system may be used to obtain dendritic cells capable of expressing various genetic constructs, which is an important condition for enhancing the immune therapeutic potentiality of dendritic cell vaccines.
Acknowledgments
The work was funded by MESR, contract No. 14.607.21.0140 (RFMEFI60715X0140).
References
- Banchereau, J. and Steinman, R.M. Dendritic Cells and the Control of Immunity. Nature 1998; 392(6673): 245-52.
- Anguille, S. et al. Clinical Use of Dendritic Cells for Cancer Therapy. Lancet Oncol 2014; 15(7): 257-67.
- Lezhnin, Y.N. et al. Comparative Efficiency of Different Techniques for in Vitro Maturation of Dendritic Cells. Research Journal of Pharmaceutical, Biological and Chemical Sciences 2016; 7(1): 1961-1968.
- Lotem, M. et al. Presentation of Tumor Antigens by Dendritic Cells Genetically Modified with Viral and Nonviral Vectors. J Immunother 2006; 29(6): 616-27.
- Hrecka, K. et al. Vpx Relieves Inhibition of HIV-1 Infection of Macrophages Mediated by the SAMHD1 Protein. Nature 2011; 474(7353): 658-61.
- Beloglazova, N. et al. Nuclease Activity of the Human SAMHD1 Protein Implicated in the Aicardi-Goutieres Syndrome and HIV-1 Restriction. J Biol Chem 2013; 288(12): 8101-10.
- Fujita, M. et al. SAMHD1-Dependent and -Independent Functions of HIV-2/SIV Vpx Protein. Front Microbiol 2012; 3: 297.








