Manuscript accepted on :09-04-2025
Published online on: 09-05-2025
Plagiarism Check: Yes
Reviewed by: Dr. Akshada Koparde
Second Review by: Dr.. Thirunahari Sravanthy
Final Approval by: Dr. Anton R Keslav
Jayaprakash Thirunavukarasu and Abdul Rahim Shajahan*
and Abdul Rahim Shajahan*
Department of Chemistry, B S Abdur Rahman Crescent Institute of Science and Technology, Vandalur, Chennai, Tamil nadu, India.
Corresponding Author E-mail: ashajahan@crescent.education
DOI : https://dx.doi.org/10.13005/bpj/3188
Abstract
N-phenyl Piperazine derivatives have increasingly been recognized for their profound medicinal properties. This study embarks on the synthesis and study of such derivatives, specifically focusing on their in vitro α-amylase inhibitory and anti-inflammatory potential. Employing in silico molecular docking strategies, we assessed the interactions of these derivatives with the α-amylase enzyme (1HNY.pdb). Compounds P6, P7, and P22 emerged with commendable docking scores as -8.44, -8.37, and -8.49 kcal/mol, prompting their synthesis and assessment of in vitro α-amylase activity assessment. Among the studied compounds, P7 demonstrated robust inhibitory effects against both α- amylase and inflammation. Complementing the docking studies, this comprehensive investigation underscores the potential of N-phenyl Piperazine derivatives as potent bioactive molecules.
Keywords
Amylase inhibitor; Anti-diabetic activity; Anti-inflammatory; N-phenyl piperazines
Download this article as:| Copy the following to cite this article: Thirunavukarasu J, Shajahan A. R. Antidiabetic and Anti-inflammatory Properties of N-Phenyl Piperazine Derivatives through In Vitro Evaluations and Molecular Docking Studies. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Thirunavukarasu J, Shajahan A. R. Antidiabetic and Anti-inflammatory Properties of N-Phenyl Piperazine Derivatives through In Vitro Evaluations and Molecular Docking Studies. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/44uFz6k |
Introduction
Global community is witnessing a significant rise in carbohydrate metabolism issues like diabetes, obesity, etc.1 Among them, diabetes Mellitus, is a non-communicable metabolic disorder, gaining growing attention due to its prevalence and challenging public health issues globally. The global DM count was around 537 million in 2023, and forecasts suggest a rise to 700 million by 2045. Approximately 10.5% of this demographic comprises adults aged 20 to 79 years.2 DM can be classified into three categories: Type-1, or insulin depending diabetes; Type- 2, or insulin resistant diabetes. Insulin resistant diabetes is particularly alarming, accounting for roughly 98 percent of all diabetes diagnoses worldwide, although this ratio shows considerable variation across different countries. A noteworthy point of concern is the rising incidence rate of type 2 diabetes in young adults. The global data reveals that this figure jumped from 117 to 183 per 100,000 population between 1990 and 2019.3 This increasing prevalence of diabetes causes severe economic implications for many nations. Countries are finding that between 5% and 13% of their total budget is dedicated to managing this disease. For instance, in 2010, global diabetes- related expenditure was around USD 376 billion. This figure is projected to escalate to nearly USD 490 billion by 2030.4 Besides, untreated diabetic patients are highly likely for the development of chronic complications, including retinopathy, foot problem, heart diseases, nephropathy, neuropathy, gum disease and etc.5
Type 2 diabetes emerges as a result of prolonged, abnormal regulation of blood sugar levels, often caused by inadequate insulin segregation from the pancreas or the body’s defiance to insulin. Insulin serves as a critical enzyme that facilitates the transport of glucose from the blood into the cells. Failure or resistance to insulin invariably leads to a surge in blood sugar levels, potentially instigating severe health complications such as blindness, heart attacks, strokes, and kidney damage. Managing blood glucose levels in individuals with diabetes mellitus can be complicated due to personal factors like diet and physical activity, and the non-specific effects of oral antihyperglycemic medications.6 A common strategy for preventing and managing diabetes involves reducing the rise of post-prandial glucose levels that is dependent on diet. Thus, considering the enzyme inhibitors as an alternative target is a valid strategy because of their target specificity.
The digestive enzyme Amylase, which are secreted from salivary glands and pancreas. Among all amylase enzymes, α-amylase also known as 1,4-α-D glucan glucanohydrolase, has gained significant attraction as a therapeutic pick out for type-2 diabetes mellitus. α-amylase is a calcium metalloenzyme that catalyze the cleaving the glycosidic bonds in complex carbohydrates and converting them in to simple carbohydrates which can be absorbed.7
Inhibiting the action of α-amylase regulates blood sugar level, which cures obesity and overweight conditions by reducing postprandial measures of glucose.8 Numerous amylase inhibitors have also been found in natural sources.9-10 Several plants extracts found to be effective on inhibiting the α-amylase. For instance, natural products, particularly plant extracts, have been widely studied for their α-amylase inhibitory activity. Acarbose, a pseudo-tetra saccharide, is a commercially available drug that functions as an α-amylase inhibitor, delaying carbohydrate digestion to mitigate postprandial hyperglycemia. However, scaling up the plant extracts in bulk scale to distribute commercially is challenging task due to their limited availability,11-12 poor stability, low purity levels and high-cost factors.
Several oral antidiabetic agents, including biguanides, sulfonylureas, and acarbose are utilized to control elevated blood sugar levels.13 However, acarbose, and miglitol are the only FDA approved α-amylase inhibitors for curing the diabetics, revealing the scarcity in the availability of α-amylase inhibitors.14 Besides many synthetic inhibitors that block the enzyme’s activity by binding to its active site are effective for diabetes management. However, side effects like flatulence, diarrhea, bloating, meteorism, and stomach pain have spurred the quest for safer alpha-amylase inhibiting molecules.15
N-phenyl piperazine core derivatives are pivotal scaffold in medicinal chemistry that were reported to display various medicinal activities, including anti-inflammatory,16 dopamine D3 receptor,17 anticonvulsant agents,18and protein kinase inhibitor Inspired by the wide spectrum of medicinal properties, herein this work, we report that we designed and synthesis N-phenyl piperazine based derivatives and evaluated for their α-amylase inhibiting properties based on the molecular docking studies.
Materials and Methods
Molecular docking
Auto-Dock (version 4.2) was used to perform Molecular docking studies. Protein Data bank was used for the structure of the enzymes. To avoid the unwanted interactions, bounded solvent molecules were denied with interactions of alpha amylase.
General procedure for preparation of P6, P7, P22
Synthesis of 4-(4-acetylpiperazin-1-yl)phenyl 2-chlorobenzoate (P6)
To the reaction mixture of 2-chlorobenzoic acid (2.3 g, 14.68 mmol) in DMF (20mL), TBTU (5.66 g, 17.62 mmol) and triethylamine (2.22 g, 22.02 mmol) were added and mixed for 30 minutes at 30±5ºC. Further, 1-(4-(4-hydroxyphenyl)piperazin-1-yl)ethanone (3.23 g, 14.68 mmol) was added. Quench the mass in chilled water and filter to get the product. Isolate the material in ethyl acetate and ether (3:7) by using chromatography. Yield: 82%. 1H NMR: ppm: δ 2.08 (S, 3H), 3.13 – 3.20 (d, 4H), 3.62 (S, 4H), 7.08 (d, 2H), 7.19 (d, 2H), 7.57 (S, 1H), 7.69 (d, 2H) 8.07 (S, 1H). m/z 358.82, (M++1) 359.2. IR: 3456, 2834, 1739,1661,1232,1034,749 cm -1.
Synthesis of 4-(4-acetylpiperazin-1-yl)phenyl picolinate (P7)
To the reaction mixture of picolinic acid (1.8 g) in DMF (20mL, TBTU (5.66 g, 17.62 mmol) and triethylamine (2.22 g, 22.02 mmol) were added and mixed for 30 minutes at 30±5ºC. Then 1-(4-(4-hydroxyphenyl)piperazin-1-yl)ethanone (3.23 g) was charged. Quench the mass in chilled water and filter to get the product. Isolate the material in ethyl acetate and ether (3:7) by using chromatography. Yield: 80%. 1H NMR: ppm: δ 2.05 (S, 3H), 3.10 – 3.17 (d, 4H), 3.59 (S, 4H), 7.05 (d, 2H), 7.15 (d, 2H), 7.73 (S, 1H), 8.07 (d, 1H) 8.20 (d, 1H), 8.80 (S, 1H). m/z 325.36, 326.2. IR: 3431, 2817, 1762,1640,1231,1197 cm -1.
Synthesis of 4-(4-acetylpiperazine-1-yl)phenyl 4-methylbenzoate (P22)
TBTU (5.66 g, 17.62 mmol) and triethylamine (2.22 g, 22.02 mmol) was charged to DMF (20mL) containing 4- methyl benzoic acid (2 g, 14.68 mmol) and stirred for 30 minutes at 30±5ºC. Then 1-(4-(4-hydroxyphenyl)piperazin-1-yl)ethanone (3.23 g,) was added. Quench the mass in chilled water and filter to get the product. Isolate the material in ethyl acetate and ether (3:7) by using chromatography. NMR: ppm: δ 2.04 (S, 3H), 2.42 – 2.50 (S, 3H), 3.09 – 3.16 (d, 4H), 3.58 (S, 4H), 7.03 (d, 2H), 7.11 (d, 2H), 7.39 – 7.41 (d, 2H), 7.99-8.01 (d, 2H). ESI/LC-MS Calculated mass 338.4 found. IR : 3408, 2858, 1724,1630,1514,1444,1237,1179 cm -1.
Anti-inflammatory activity
BSA denaturation technique
α-amylase inhibition
Anti-inflammatory (BSA denaturation technique) was performed as per the reported methods of Sribalan et al.19
α-amylase inhibition was performed as per the reported methods of Sribalan et al.2
Density Functional Theory
FMO’s was performed for synthesized compounds using DFT methods. The electronic structural characterization of compounds was obtained from the structure of the compound. Gaussian 09W software package was used with B3LYP/6.31 G, resulted optimized structure was analyzed in the Gauss view software and the same software was used for the further process that mentioned initially.
Results
Molecular docking
Considering the medicinal significances of piperazine analogues in medicinal chemistry, we designed 22 piperazine derivatives (figure 1) were studied with interactions of alpha amylase.
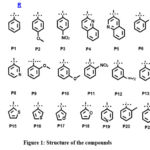 |
Figure 1: Structure of the compoundsClick here to view Figure |
Table 1: Molecular docking interconnections of prepared compounds with 1HNY
| S.No | Chemical name | Binding energyKacal/mol | Inhibition constant(µM) | Number of bonding(Hydrogen) | Interconnected amino acid |
| 1. | P1 | -6.9 | 8.73 | 4 | HIS201,GLN63,GLN63,TRP59 |
| 2. | P2 | -6.99 | 7.54 | 4 | TRP59,TYR62,ARG195,ASP197 |
| 3. | P3 | -7.98 | 1.43 | 4 | HIS201,GLN63,GLN63,HIS305 |
| 4. | P4 | -6.26 | 25.83 | 2 | ALA198, TRP59 |
| 5. | P5 | -7.27 | 4.7 | 3 | ASP300,TYR62,HIS299 |
| 6. | P6 | -8.44 | 0.65 | 2 | HIS299, TYR62 |
| 7. | P7 | -8.37 | 0.73 | 4 | TYR151,LYS200,HIS299,TYR62 |
| 8. | P8 | -6.45 | 18.57 | 7 | GLN63,GLN63,GLU233,GLU233,TRP59,LYS,200,HIS201 |
| 9. | P9 | -6.23 | 27.02 | 3 | TRP59,ALA198,GLU233 |
| 10. | P10 | -6.44 | 19.04 | 5 | HIS101,ASP197,LYS200,LYS200,TYR151 |
| 11. | P11 | -6.27 | 25.27 | 7 | GLN63,TRP59,TYR151,TYR151,LYS200,LYS200,HIS201 |
| 12. | P12 | -6.84 | 9.76 | 3 | GLN63,GLN63,LYS200 |
| 13. | P13 | -6.47 | 18.23 | 4 | THR163,HIS299,TYR62,ASP197 |
| 14. | P14 | -7.22 | 5.07 | 2 | GLN63,LYS200 |
| 15. | P15 | -6.26 | 25.91 | 4 | TRP59,TRP59,TRP59,TRP62 |
| 16. | P16 | -6.37 | 21.29 | 5 | TTYR62,HIS299,ASP197,GLU233,GLU233 |
| 17. | P17 | -6.58 | 15.06 | 3 | LYS200,LYS200,TYR151 |
| 18. | P18 | -6.45 | 18.56 | 4 | GLN63,GLY104,TYR62,HIS299 |
| 19. | P19 | -6.66 | 13.08 | 3 | GLN63,ARG195,ASP300 |
| 20. | P20 | -7.06 | 6.64 | 4 | TYR62,TYR151,TYR151,LYS200 |
| 21. | P21 | -7.39 | 3.82 | 2 | HIS299,TYR62 |
| 22 | P22 | -8.49 | 0.60 | 3 | GLN63,LYS200,ILE235 |
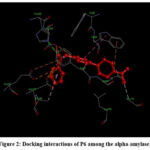 |
Figure 2: Docking interactions of P6 among the alpha-amylaseClick here to view Figure |
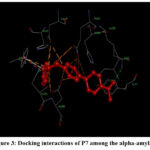 |
Figure 3: Docking interactions of P7 among the alpha-amylaseClick here to view Figure |
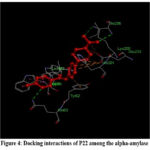 |
Figure 4: Docking interactions of P22 among the alpha-amylaseClick here to view Figure |
Chemistry
After the successful validation of 22 piperazine derivatives, we identified P6, P7 and P22 as potent compounds and intended for the synthesis of compound. As shown in scheme, 4- phenolic-piperizine (2) was treated with various aromatic carboxylic acids (1a-1c) with HBTU and triethyl amine yielded the desired esters P6, P7 and P22 in noticeable yields.
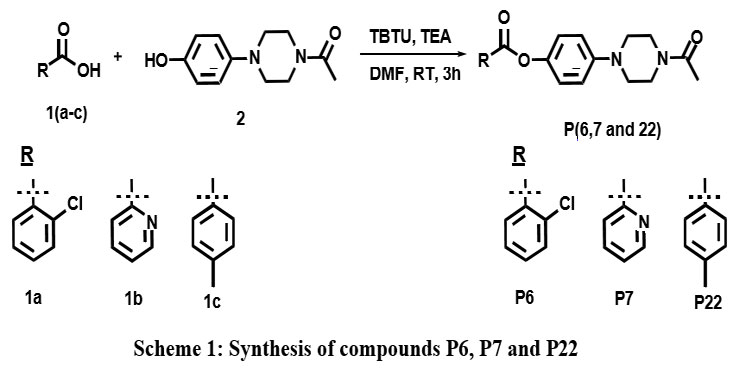 |
Scheme 1: Synthesis of compounds P6, P7 and P22Click here to view Scheme |
Biological studies
Alpha-Amylase inhibition studies
The prepared P6, P7 and P22 were evaluated of their α-amylase inhibitory potential of various strengths of (10, 50, 100, 250 and 500 µg/mL), by adopting acarbose as a Reference drug. As shown in figure 5, treatment of compounds P6, P7 and P22 demonstrated the dose dependent inhibitory effect against α-amylase.
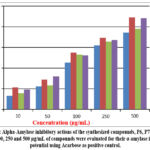 |
Figure 5: Alpha-Amylase inhibitory actions of the synthesized compounds, P6, P7 and P22. Click here to view Figure |
Anti-inflammatory activity
The inhibitors P6, P7, and P22 were analyzed for their anti-inflammatory activities, using diclofenac sodium as a reference through the albumin denaturation method. When subjected to various concentrations alongside of Bovine serum albumin, inflammation has been measured by the level of denaturation.
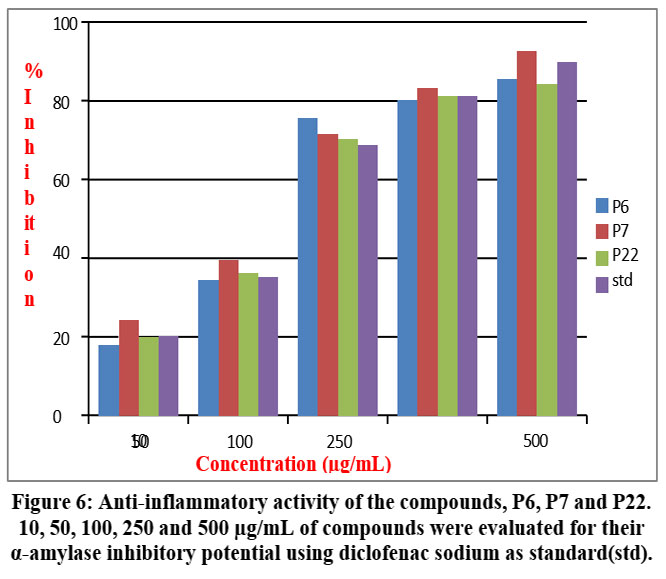 |
Figure 6: Anti-inflammatory activity of the compounds, P6, P7 and P22. Click here to view Figure |
Frontier Molecular Orbitals
Energy of Highest Occupied Molecular Orbital (HOMO) or the Lowest Unoccupied Molecular Orbital (LUMO) are specifically analysed. Frontier orbitals influence all chemical reactivity of a substance. The energy gap between them, called the band gap, indicates molecular reactivity: a smaller gap implies higher reactivity. Beyond the energy levels, DFT parameters offer insights into a molecule’s reactivity and electronic behaviours, crucial for predicting molecular interactions and designing new molecules.
Table 2: Density Functional Theroy values of P6,P7,P22.
| S.No | Chemical name | HOMO | LUMO | Band gap(DE) | Chemical potential | Globalhardness | Global softness | Electrophilicity index |
| 1. | P6 | -5.4864 | -1.3908 | 4.0956 | -3.4386 | 2.0478 | 0.2441 | 2.8870 |
| 2. | P7 | -5.4921 | -1.8327 | 3.6594 | -3.6624 | 1.8297 | 0.2732 | 3.6654 |
| 3. | P22 | -5.2478 | -1.2471 | 4.000 | -3.2474 | 2.0003 | 0.2499 | 2.6360 |
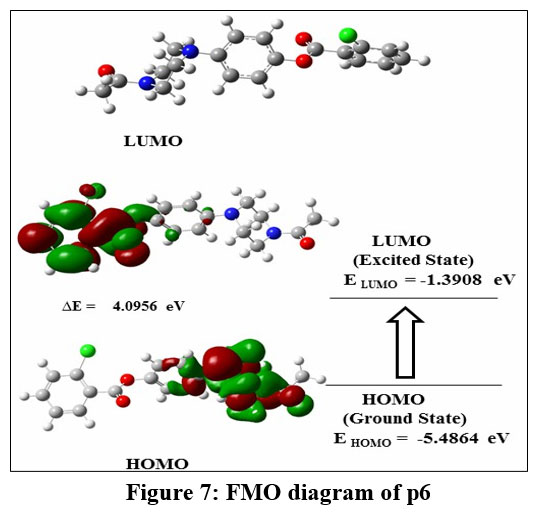 |
Figure 7: FMO diagram of p6Click here to view Figure |
 |
Figure 8: FMO diagram of p7Click here to view Figure |
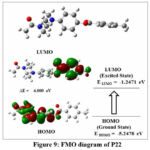 |
Figure 9: FMO diagram of P22Click here to view Figure |
Molecular Electrostatic Potential
Molecular Electrostatic Potential helps as an insightful tool for elucidating the binding interactions between nucleophilic and electrophilic interventions within the prepared compounds. Such mappings grant an unparalleled visualization of electron density distributions on molecular surfaces.21
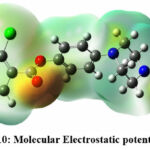 |
Figure 10: Molecular Electrostatic potential of p6Click here to view Figure |
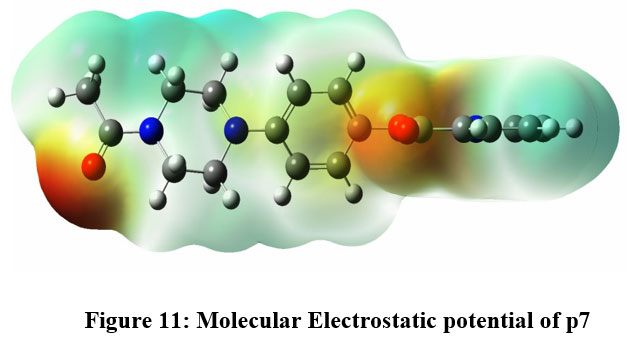 |
Figure 11: Molecular Electrostatic potential of p7Click here to view Figure |
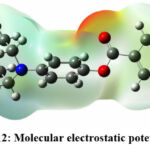 |
Figure 12: Molecular electrostatic potential of p22Click here to view Figure |
Discussions
Molecular docking
The docking studies reveals that all compounds were found to demonstrate significant binding energies and inhibition constants. In particular, compounds P6, P7, and P22 exhibited the strongest binding energy with notable inhibition constants among the tested compounds as shown in table 1. For instance, the binding energies and inhibition constants of P6, P7, and P22 were -8.44, -8.37, and -8.49 kcal/mol and 0.65, 0.73, and 0.60 µM respectively as shown in table 1.
When P6 docks with 1HNY, it was found to show two bonds with HIS299 and TYR62 of 1HNY. The carbonyl oxygen in P6 engages bonding (hydrogen) with HIS299, having bond distance of 2.88 Å. Besides, the same carbonyl-oxygen atom also underwent bonding with the phenyl ring of TYR62, having a bond distance of 3.09 Å. (Figure 2)
For P7 docking with 1HNY, four bonding are observed with TYR151, LYS200, HIS299, and TYR62 of 1HNY. The halogen in the phenyl group was found to show a conventional bond interaction with LYS200, with the bond length of 3.26 Å. The phenylene ring of TYR62 engages in a pi-donor bonding with the acetyl CH3, having the bond distance of 3.34 Å. Additionally, the piperazinamide carbonyl oxygen in the ligand having bonding with HIS299, with a bond length of 3.97 Å. (Figure 3)
Similarly, in its docking with 1HNY, P22 having multiple inter connections, -NH2 had interacted were with GLN63, LYS200, and ILE235. The carboxamide nitrogen of GLN63 formed a bonding interaction with the tolyl- CH and the bond distance was measured at 3.09 Å. The piperazinamide oxygen underwent a bonding with LYS200, with a bond distance of 3.01 Å. Additionally, the NH of ILE235 had the bonding with piperazinamide CH3, with a bond measurement of 3.14 Å. (Figure 4)
Alongside hydrogen bonding, these piperazine derivatives also demonstrated other interactions with the α-amylase enzyme, including Pi-cation, Pi-pi stacking, Pi-alkyl, Pi-pi T- shaped, and attractive charge interactions. The results of docking has been detailed as below, and visual depictions of these interactions were presented in figures. (Figure 2,3,4).
Molecular docking results can be used to study the interactions of small molecules with proteins by interpreting strong binding energies. The binding energies of P6, P7 and P22 were – 8.44, -8.37, and -8.49 kcal/mol, the results were comparable with the reference drug diclofenac sodium results -7.07 and -4.2 kcal/mol.
Biological studies
Alpha Amylase inhibition studies
It is imperative to note that P7 displayed significant inhibitory effects compared to P6 and P22. Impressively, at several concentrations, P7 outperformed the control acarbose by displaying potent inhibitory effects. Moreover, all the three compounds showed maximum inhibitory potential at the treatment of 500 µg/mL. for instance, P6 and p7 showed respectively 74 and 78% of α-amylase inhibitions, whereas P7 showed approximately 90% inhibition, which is better than the control, agarbose. At the 100 µg/mL mark, both P7 and P22 achieved 50% inhibition of α-amylase, while P6 showed slightly decreased potential. The α-Amylase inhibitory order lies in the pattern of P7> P22> P6. Collectively, the results suggest the potential of piperazine-based compounds to evolve as a model for the discovery of the α-amylase inhibitor.
Anti-inflammatory activity
As shown in figure 6, a dose-dependent anti-inflammatory response was evident upon treatment with P6, P7, and P22. In comparison to the control, all three compounds exhibited comparable anti-inflammatory effects across the tested concentrations. Notably, P7 consistently demonstrated the highest anti-inflammatory potential across most concentrations. However, at 100 µg/mL, P6 surpassed the efficacy of other compounds and even the standard control. Remarkably, at 500 µg/mL, all compounds displayed 85-90% anti-inflammatory effects, describing their significant anti-inflammatory potential. Collectively, the synthesized phenyl piperazine derivatives not only inhibit α-amylase but also inhibit inflammation, giving the new opportunities in development of drugs targeting both α-amylase and inflammation.
Frontier Molecular Orbitals
Amongst the evaluated compounds, P6 exhibited Highest occupied molecular orbital of -5.4864 eV and a Lowest unoccupied molecular orbital of -1.3908 eV (as evident in Figure 7). This results in an energy gap (ΔE) of 4.0956 eV and a chemical potential of -3.4386 eV. In contrast, compounds P7 and P22 presented almost similar HOMO values of -5.4921 eV and -5.2478 eV, respectively, while their LUMO values were measured at -1.8327 eV and -1.2471 eV, respectively (refer to Figures 8 and 9). It’s well-established that a larger energy gap between HOMO and LUMO levels corresponds to higher kinetic energy, leading to enhanced chemical or molecular interactions. Apparently, P6 was found to have more band gap. Further, the global hardness values for the synthesized compounds P6, P7, and P22 were determined to be 2.0478, 1.8297, and 2.0003 eV, respectively. Their global softness values were reported as 0.2441, 0.2732, and 0.2499 eV, respectively.
Molecular Electrostatic Potential
In the context of compounds P6, P7, and P22, this distinction in electron density becomes evident upon observing Figures 10, 11, and 12. Specifically, regions demarcated in red portray electron-rich locales, prime for electrophilic encounters, whereas those highlighted in blue signify electron-deficient zones, conducive for nucleophilic interactions. For instance, in all the three compounds, piperazine amide carbonyls and phenyl esters were found to show the electron deficiency by displaying red color, which indicate that they can be a potential hydrogen bond acceptor while interacting with the target. In contrast, electron-rich domains play a pivotal role in hydrogen bond formations, particularly with enzymes and proteins.
Conclusion
Our comprehensive study has reinforced the medicinal significance of N-phenyl Piperazine derivatives, emphasizing their potential in α-amylase inhibition and anti-inflammatory activities. Docking studies given crucial inputs about the interactions of N-phenyl Piperazine derivatives with alpha -amylase enzyme. The concurrent application of Density Functional Theory and molecular electrostatic potential investigations further complemented our understanding of these interactions. Collectively, our findings spotlight the P7 derivative as a promising candidate in the realm of bioactive molecules, potentially catalyzing advancements in therapeutic interventions targeting α-amylase and inflammation. Since N-phenyl piperazine has very good α-amylase inhibition results, The further chemical derivatives of N-Phenyl piperazine can play a key role in drug discovery.
Acknowledgement
Dr. A. Shajahan greatly acknowledges the Department of Chemistry, B.S. Abdur Rahman Crescent institute of Science and Technology, Chennai 600 048, India,
Funding Sources
This work was supported by the B.S. Abdur Rahman Crescent institute of Science and Technology, Chennai 600 048, India, for the Crescent seed grant CSD/CSM/2022/12.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Jayaprakash Thirunavukarasu : Conceptualization, Data collection, Writing – Original Draft.
- Abdul Rahim Shajahan: Visualization, Supervision, Project Administration – Review & Editing.
References
- P. M. Sales, P.M. Souza, L.A. Simeoni, P.O. Magalhães, D. Silveira. α-Amylase Inhibitors: A Review of Raw Material and Isolated Compounds from Plant Source. Journal of Pharmacy & Pharmaceutical Sciences. 2012; 15(1) : 141-183.
CrossRef - Dianna J Magliano , Edward J Boyko , Beverley Balkau, Abstract for Reference 1 of ‘Type 2 diabetes mellitus: Prevalence and risk factors, International diabetics federation, 2022; 10th edition : 10 th edition. 1-141.
- J. Xie, M. Wang, Z. Long, H. Ning, J. Li, Y. Cao, Y. Liao, G. Liu, F. Wang, A. Pan. Global burden of type 2 diabetes in adolescents and young adults, 1990-2019 systematic analysis of the Global Burden of Disease Study 2019. BMJ. 2022; 379: 072385.
CrossRef - P. Zhang, X. Zhang, J. Brown, D. Vistisen, R. Sicree, J. Shaw, G.J.D.r. Nichols. Global healthcare expenditure on diabetes for 2010 and 2030. Diabetetics Res Clin Prac. 2010 ; 87(3): 293-301.
CrossRef
- Dunya Tomic, Jonathan Shaw & Dianna J. Magliano. The burden and risks of emerging complications of diabetes mellitus. Nature Reviews Endocrinology. 2022; 18: 525–539.
CrossRef - M. Alp, A. Misturini, G. Sastre, M. Gálvez-Llompart. Drug screening of α-amylase inhibitors as candidates for treating diabetes. Journal of Cellular and Molecular Medicine. 2023 ; 15 : 2249-2260.
CrossRef - D. Lin, M. Xiao, J. Zhao, Z. Li, B. Xing, X. Li, M. Kong, L. Li, Q. Zhang, Y. Liu, H. Chen, Qin, H. Wu, S. Chen. An Overview of Plant Phenolic Compounds and Their Importance in Human Nutrition and Management of Type 2 Diabetes. Molecules. 2016; 21 (10) : 1374.
CrossRef - Y. Y.C. Alice, I.G. Fantus. Oral antihyperglycemic therapy for type 2 diabetes mellitus. Canadian Medical Association Journal. 2005 ; 172(2) : 213.
CrossRef
- M. J. Nanjan, M. Mohammed, B.R. Prashantha Kumar, M.J.N. Chandrasekar. Thiazolidinediones as antidiabetic agents: A critical review. Bioorganic Chemistry. 2018; 77 : 548-567.
CrossRef
- F. A.J.V.h. Van de Laar, r. management. Alpha-glucosidase inhibitors in the early treatment of type 2 diabetes. Vasc Health Risc Mang. 2008 ; 4(6) : 1189-1195.
CrossRef - H. Ali, P.J. Houghton, A. Soumyanath. α-Amylase inhibitory activity of some Malaysian plants used to treat diabetes; with particular reference to Phyllanthus amarus. Journal of Ethnopharmacology. 2006; 107(3): 449-455.
CrossRef - P. K. Mukherjee, K. Maiti, K. Mukherjee, P.J. Houghton. Leads from Indian medicinal plants with hypoglycemic potentials. Journal of Ethnopharmacology. 2006; 106(1) : 1-28.
CrossRef - M. Omar, D.F. AlKharboush, K.A. Mohammad, G.A. Mohamed, H.M. Abdallah, S.R.M. Ibrahim. Mangosteen Metabolites as Promising Alpha-Amylase Inhibitor Candidates: In Silico and In Vitro Evaluations. Metabolites. 2022 ;
CrossRef - A. D. Dahlén, G. Dashi, I. Maslov, M.M. Attwood, J. Jonsson, V. Trukhan, H.B. Schiöth. Trends in Antidiabetic Drug Discovery: FDA Approved Drugs. New Drugs in Clinical Trials and Global Sales. Front Pharmocol. 2022 ; 12 (12) : 1229.
CrossRef
- M. Hanefeld. The Role of Acarbose in the Treatment of Non–Insulin-Dependent Diabetes Mellitus. Journal of Diabetes and its Complications. 1998 ; 12(4) : 228-237.
CrossRef - C. Papadopoulou, A. Geronikaki, D. Hadjipavlou-Litina. Synthesis and biological evaluation of new thiazolyl/benzothiazolyl-amides, derivatives of 4-phenyl-piperazine. Il Farmaco. 2005 ; 60(11) : 969-973.
CrossRef - W. Chu, Z. Tu, E. McElveen, J. Xu, M. Taylor, R.R. Luedtke, R.H. Mach, Synthesis and in vitro binding of N-phenyl piperazine analogs as potential dopamine D3 receptor ligands, Bioorganic & Medicinal Chemistry. 2005 ; 13(1) : 77-87.
- S. Kumari, C.B. Mishra, M. Tiwari, Design, synthesis and pharmacological evaluation of N- [4-(4-(alkyl/aryl/heteroaryl)-piperazin-1-yl)-phenyl]-carbamic acid ethyl ester derivatives as novel anticonvulsant agents, Bioorganic & Medicinal Chemistry Letters. 2015 ;25(5) : 1092- 1099.
CrossRef - R. Sribalan, M. Kirubavathi, G. Banuppriya, and V. Padmini, Synthesis and biological evaluation of new symmetric curcumin derivatives, Bioorganic & Medicinal Chemistry Letters,2015; 25 (19); 4282–4286.
CrossRef - Govindharasu Banupriya,Rajendran Sribalan. Sulthan Alavudeen Rizwan Fathima, Vediappen Synthesis of Beta-Ketoamide Curcumin Analogs for Anti-Diabetic and AGEs Inhibitory Activities. Chemistry & Biodiversity,2018; 15 (8);e1800105.
CrossRef - R. Sribalan, G. Banuppriya, M. Kirubavathi, V. Padmini, Synthesis, biological evaluation and in silico studies of tetrazole-heterocycle hybrids. Journal of Molecular Structure. 2019 ; 1175 : 577-586.
CrossRef







