Manuscript accepted on :11-03-2026
Published online on: 19-03-2026
Plagiarism Check: Yes
Reviewed by: Dr. Heamn Noori Abduljabbar and Dr Hind Shakir
Second Review by: Dr. WANG YUE
Final Approval by: Dr. Prabhishek Singh
Sameena Parveen1* , Mohammed Mousa Hassan Bakri1
, Mohammed Mousa Hassan Bakri1 , Samar Saeed Khan1
, Samar Saeed Khan1 , Swetha Vempalli1, Reghunathan S.Preethanath2
, Swetha Vempalli1, Reghunathan S.Preethanath2 , Mohammed Nazish Alam2
, Mohammed Nazish Alam2 , Mohamed Hamdi Mansy2
, Mohamed Hamdi Mansy2 , Hind Ali Osman2
, Hind Ali Osman2 , Husham E. Homeida1
, Husham E. Homeida1 , Ahmed Mostafa Ahmed Mostafa1
, Ahmed Mostafa Ahmed Mostafa1 and Solima Ibrahim Yagoob3
and Solima Ibrahim Yagoob3 .
.
1Department of Maxillofacial Surgery and Diagnostic Sciences, College of Dentistry, Jazan University, Jazan, Kingdom of Saudi Arabia.
2Department of Preventive Dental Sciences, College of Dentistry, Jazan University, Jazan, Kingdom of Saudi Arabia.
3Department of Physical Sciences, College of Science, Jazan University, Jazan, Kingdom of Saudi Arabia.
Corresponing Author E-mail:sperveen@jazanu.edu.sa
DOI : https://dx.doi.org/10.13005/bpj/3335
Abstract
Periodontal disease is initiated by dysbiosis of the oral microbiome and sustained host inflammatory responses. Dietary patterns and intermittent fasting may influence oral microbial ecology and periodontal inflammation through microbiological and host-mediated mechanisms. This review synthesizes current evidence on how dietary patterns and intermittent fasting affect the oral microbiome and modulate periodontal inflammatory processes. A narrative review was conducted using PubMed, Embase, Scopus, Web of Science, and Google Scholar to identify literature published between January 2000 and October 2025. The search included studies addressing dietary patterns and nutrition, periodontal disease and the oral microbiome, and intermittent fasting or caloric restriction. Human clinical studies, observational studies, animal models, and in vitro investigations examining associations between diet or fasting, oral microbial composition, periodontal inflammation, and host immune pathways were reviewed and narratively synthesized. Diets high in refined carbohydrates were associated with oral dysbiosis, oxidative stress, and increased expression of inflammatory cytokines, whereas fiber-rich, antioxidant-dense, and micronutrient-adequate dietary patterns supported microbial diversity, maintained epithelial barrier integrity, and reduced periodontal inflammation. Intermittent fasting and caloric restriction influenced periodontal inflammatory pathways by improving insulin sensitivity, mitochondrial efficiency, redox balance, and autophagy. Animal studies demonstrated reductions in periodontal inflammation and alveolar bone loss, while limited human studies reported favorable changes in inflammatory markers. Dietary modulation and intermittent fasting may therefore influence periodontal inflammation through microbiome-mediated and host-metabolic mechanisms, however well-designed clinical studies integrating standardized fasting protocols, oral microbiome profiling, and validated periodontal outcomes are required to clarify their translational relevance.
Keywords
Disease; Diet; Fasting; Intermittent; Inflammation; Oral microbiome; Oral gut axis; Periodontal disease; Nutrition
Download this article as:| Copy the following to cite this article: Parveen S, Bakri M. M. H, Khan S. S, Vempalli S, Preethanath R. S, Alam M. N, Mansy M. H, Osman H. A, Homeida H. E, Mostafa A. M. A, Yagoob S. I. Dietary Patterns and Intermittent Fasting as Modulators of the Oral Microbiome and Periodontal Inflammation: A Narrative Review. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Parveen S, Bakri M. M. H, Khan S. S, Vempalli S, Preethanath R. S, Alam M. N, Mansy M. H, Osman H. A, Homeida H. E, Mostafa A. M. A, Yagoob S. I. Dietary Patterns and Intermittent Fasting as Modulators of the Oral Microbiome and Periodontal Inflammation: A Narrative Review. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/4uKlUdl |
Introduction
Periodontal disease is a chronic inflammatory condition initiated by dysbiosis of the oral microbiome and sustained host immune activation, leading to progressive destruction of the supporting periodontal tissues.1 This ecological imbalance favors the overgrowth of pathogenic microbial consortia that disrupt host–microbe homeostasis and perpetuate inflammatory tissue breakdown.2 The oral microbiome is among the most diverse microbial ecosystems in the human body and functions as a critical host–microbe interface essential for maintaining oral tissue integrity.3
Accumulating evidence indicates that periodontal inflammation is not confined to the oral cavity. Microbial components and inflammatory mediators originating from periodontal tissues can enter systemic circulation and contribute to broader immune activation, providing mechanistic links between periodontal disease and systemic inflammatory conditions.4,5 These associations underscore the need to elucidate the biological mechanisms through which modifiable factors, such as diet and feeding patterns, influence oral microbial ecology and host inflammatory regulation.
Diet represents a key environmental determinant shaping the composition and functional activity of the oral microbiome.6 Diets enriched in refined carbohydrates and pro-inflammatory fats have been associated with oral dysbiosis, increased oxidative stress, and elevated expression of inflammatory cytokines, including IL-1β, IL-6, and TNF-α. In contrast, dietary patterns rich in fibre, polyphenols, and antioxidants are associated with greater microbial diversity, improved epithelial barrier function, and reduced gingival inflammation.7,8 Beyond changes in microbial abundance, dietary modulation has been shown to influence microbial metabolic pathways, suggesting effects that extend beyond plaque accumulation alone.6 Adequate intake of micronutrients, including vitamins C and D, is also essential for collagen synthesis, immune regulation, and alveolar bone metabolism; deficiencies compromise periodontal tissue resilience.9
Intermittent fasting (IF) and caloric restriction (CR) have attracted increasing attention due to their effects on metabolic and inflammatory pathways relevant to periodontal pathophysiology. Experimental studies indicate that fasting-induced metabolic adaptations, including improved insulin sensitivity, enhanced mitochondrial efficiency, reduced oxidative stress, and activation of autophagy, may influence host responses to periodontal inflammation.10,11 Animal models demonstrate reductions in periodontal inflammation and alveolar bone loss following caloric restriction,12 while emerging human data suggest potential benefits in inflammatory profiles.13 However, direct evidence assessing the effects of fasting regimens on human periodontal outcomes remains limited. The oral–gut microbiome axis provides an additional mechanistic framework linking dietary and metabolic factors to periodontal inflammation. Oral bacteria may translocate to the gastrointestinal tract, disrupt intestinal barrier integrity, and amplify systemic inflammatory responses, thereby contributing to metabolic and inflammatory dysregulation.14 Despite its potential relevance, the role of the oral microbiome in nutritional and metabolic research has received comparatively less attention.
Given these gaps, this narrative review synthesizes current evidence on how dietary patterns and intermittent fasting may influence oral microbial ecology and periodontal inflammation through microbiological and host-mediated mechanisms.
Methods
Aim and scope
This narrative review synthesizes current evidence on how dietary patterns and intermittent fasting influence the oral microbiome, periodontal inflammation, and systemic inflammatory responses. The primary outcomes were periodontal clinical parameters and inflammatory biomarkers. Secondary outcomes included oral and gut microbial diversity, metabolic indicators, and mechanistic pathways linking diet or fasting to periodontal pathophysiology.
Databases and time frame
Electronic searches were performed in PubMed/MEDLINE, Embase, Scopus, Web of Science, Google Scholar, and the Saudi Digital Library. Searches covered English-language publications from January 2000 through October 2025. The end date reflects the most recent month in which the database was searched for this manuscript.
Search strategy
Controlled vocabulary and free-text keywords were combined using Boolean operators. Search terms covered three core concepts:
Diet, nutrition, refined carbohydrates, dietary patterns, prebiotics, and probiotics;
Periodontal diseases, oral microbiome, dysbiosis, oxidative stress, inflammatory mediators; Intermittent fasting, time-restricted eating, caloric restriction, autophagy, metabolic inflammation, and oral–gut axis.
Citation chaining was conducted using key reviews in oral microbiology and periodontal immunopathology to ensure comprehensive coverage of mechanistic and microbial literature.2,6
Study selection
Eligible publications included human clinical trials, observational studies, animal models, and in vitro investigations that examined associations between diet or fasting and oral microbiota, periodontal parameters, or inflammatory markers. Exclusion criteria were narrative commentaries without primary data, conference abstracts lacking full results, and non-English publications. Two reviewers independently screened titles and abstracts. After applying the inclusion and exclusion criteria, relevant studies were selected for synthesis. Disagreements were resolved through discussion. As a narrative review, this synthesis prioritizes conceptual integration and mechanistic insight over quantitative meta-analysis; therefore, no PRISMA flowchart or formal risk-of-bias assessment was performed.
Data extraction and variables
Data extraction focused on the key characteristics of each study, including design, sample size, participant profile, dietary or fasting exposure, intervention duration, and reported outcomes. Periodontal outcomes included bleeding on probing, probing depth, clinical attachment level, and the area of inflamed periodontal surfaces. Inflammatory markers, such as gingival crevicular fluid, Interleukin-1 beta (IL-1β), IL-6, and Tumor Necrosis Factor-alpha (TNF-α), and serum C-reactive protein, were recorded when available. Microbiome-related outcomes included changes in microbial diversity, differential taxon abundance, and, in recent studies, shifts in functional pathways and oral–gut microbial interactions. Metabolic indicators, including HbA1c, lipid profile, insulin sensitivity, and oxidative stress markers, were also extracted. Fasting regimens were classified into three categories: 16/8 time-restricted eating, alternate-day fasting, and continuous caloric restriction, following standard definitions in the literature.11
Synthesis and quality considerations
Findings were organized under dietary and fasting interventions, with emphasis on results consistent across higher-quality studies and supported by standard clinical periodontal measures.7,15 Mechanistic interpretations were cross-checked with established frameworks of periodontal immunopathology and microbiome ecology.2,6 No formal risk-of-bias tool was applied. Evidence quality was assessed qualitatively based on study design, sample adequacy, intervention clarity, and measurement validity. Common limitations included small samples, inconsistent dietary definitions, variable fasting schedules, and short follow-up durations. After applying inclusion and exclusion criteria, relevant literature published between January 2000 and October 2025 was reviewed and synthesized.
The Oral Microbiome and Periodontal Inflammation
The oral cavity harbors a diverse microbial ecosystem composed of bacteria, fungi, archaea, and viruses, organized into structured biofilms on dental and mucosal surfaces. In health, this ecosystem maintains stability through microbial competition, metabolic cooperation, and controlled immune surveillance, collectively preserving epithelial integrity and preventing unnecessary inflammation. This homeostasis sustains a stable, low-inflammatory environment in periodontal tissues through regulated neutrophil surveillance and controlled immune activation.16–18
When ecological pressures disrupt this balance, the microbiome shifts toward dysbiosis. Key drivers include frequent sugar exposure, salivary hypofunction, systemic inflammation, metabolic disorders, and impaired host immunity. Dysbiotic communities become enriched with inflammatory Gram-negative taxa, including Porphyromonas gingivalis, Treponema denticola, and Tannerella forsythia. These organisms reshape the biofilm through virulence factors that impair neutrophil chemotaxis, alter complement activity, and manipulate cytokine signaling, creating an environment that favors persistent inflammation.2,19
Host recognition of dysbiotic biofilms activates potent intracellular inflammatory pathways. Pattern-recognition receptors initiate Nuclear Factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling, which promotes the release of IL-1β, IL-6, TNF-α, prostaglandins, and matrix metalloproteinases. These mediators contribute to fibroblast apoptosis, collagen degradation, epithelial barrier breakdown, and osteoclastic bone resorption. Clinically, this manifests as gingival bleeding, periodontal pocket formation, and progressive loss of clinical attachment.20,21
Emerging evidence indicates that microbial metabolites, outer membrane vesicles, and proteolytic byproducts also significantly affect epithelial permeability, oxidative stress, and immune regulation, further complicating periodontal pathogenesis. These insights align with recent studies demonstrating that functional metabolic pathways within the dysbiotic biofilm, rather than individual bacterial species alone, drive the inflammatory phenotype associated with disease.22,23
Critically, periodontal inflammation extends beyond local tissues. Bacterial components, such as lipopolysaccharide and proinflammatory cytokines, enter the systemic circulation, contributing to endothelial dysfunction, oxidative stress, hepatic metabolic changes, and altered insulin sensitivity.24 These mechanisms provide a biological basis for the established epidemiological links between periodontitis and diabetes, cardiovascular disease, and other chronic inflammatory disorders.25 The interplay between dysbiotic biofilms and host immune responses that drives progressive periodontal breakdown is illustrated in Figure 1.
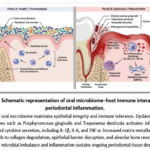 |
Figure 1: Schematic representation of oral microbiome–host immune interactions in periodontal inflammation. |
A balanced oral microbiome maintains epithelial integrity and immune tolerance. Dysbiosis initiated by pathogens such as Porphyromonas gingivalis and Treponema denticola activates inflammatory signaling and cytokine secretion, including IL-1β, IL-6, and TNF-α. Increased matrix metalloproteinase activity leads to collagen degradation, epithelial barrier disruption, and alveolar bone resorption. The cycle of microbial imbalance and inflammation sustains ongoing periodontal tissue destruction.
Carbohydrates and Refined Sugars
Dietary carbohydrates, particularly refined sugars, significantly affect the ecology of the oral microbiome. Frequent intake of fermentable sugars lowers plaque pH and drives rapid ecological shifts that favor aciduric, metabolically active bacteria, thereby increasing overall biofilm activity and inflammatory potential within gingival tissues.6 Beyond their effects on caries-associated taxa, high-sugar environments create ecological conditions that favor the growth of proteolytic and endotoxin-producing anaerobes within periodontal pockets.
Refined carbohydrates increase the abundance of taxa linked to periodontal dysbiosis, including Prevotella and Fusobacterium, which activate Toll-like receptors and upregulate NF-κB signaling, and other inflammation-promoting anaerobes that activate Toll-like receptors and upregulate NF-κB signaling. This results in elevated production of IL-1β, IL-6, TNF-α, and matrix metalloproteinases, accelerating connective tissue degradation and alveolar bone resorption.4 Large epidemiological studies consistently show that high intake of refined sugars is associated with greater gingival bleeding and deeper probing depths, independent of oral hygiene or body mass index.8,15
Excess sugar consumption also amplifies systemic inflammation through hyperglycemia-induced oxidative stress and advanced glycation end-product formation. These mechanisms impair neutrophil function, weaken antioxidant defenses, and increase endothelial inflammation, thereby heightening periodontal susceptibility.26 Recent microbiome studies demonstrate that reducing sucrose intake lowers the prevalence of periodontopathogens such as Prevotella intermedia and Fusobacterium nucleatum and reduces their virulence-related gene expression, indicating that carbohydrate-driven dysbiosis is reversible.27
Intervention trials confirm that restricting refined carbohydrates decreases plaque accumulation and inflammatory biomarkers even when mechanical oral hygiene remains unchanged.7,28 Overall, these findings show that the quality and frequency of carbohydrate intake strongly influence periodontal microbial ecology and host inflammatory responses. Incorporating dietary counseling that limits refined sugars and promotes complex carbohydrates should be considered an essential adjunct to periodontal therapy and long-term disease prevention.
Dietary Fiber, Prebiotics, and Whole Foods
Dietary fiber exerts significant effects on the oral and gut microbiomes and plays a vital role in regulating local and systemic inflammation. High-fiber diets increase salivary flow, improve buffering capacity, and reduce plaque acidification, all of which support a microbial profile associated with periodontal health.29 Although fibrous foods provide some mechanical disruption of biofilm, their primary impact is biological. Fiber-derived polysaccharides and prebiotic substrates selectively enhance the growth of beneficial microbial groups that produce short-chain fatty acids (SCFAs) such as acetate, propionate, and butyrate.30
SCFAs have well-documented anti-inflammatory functions relevant to periodontal tissues. These metabolites inhibit NF-κB signaling, suppress IL-1β and TNF-α expression, and stabilize epithelial barrier function by strengthening tight-junction proteins.31 By modulating these pathways, SCFAs help reduce gingival inflammatory burden and interrupt the cycle of dysbiosis and chronic inflammation that characterizes periodontal disease. Recent microbiome studies show that higher fiber intake reduces the expression of biofilm-associated virulence genes and downregulates pathways involved in lipopolysaccharide biosynthesis, suggesting functional microbial benefits beyond taxonomic shifts.22,23
Evidence from randomized controlled trials demonstrates that increasing dietary fiber intake reduces bleeding on probing and improves clinical attachment levels, even when oral hygiene or caloric intake remains unchanged.32 Longitudinal studies in older adults also show that habitual fiber intake is associated with slower periodontal progression.29 These outcomes are partly explained by systemic effects, including reductions in C-reactive protein and improvements in glycemic control, both of which influence periodontal healing and immune regulation.33
Whole-food dietary patterns rich in fruits, vegetables, legumes, and whole grains enhance oral microbial diversity and reduce the abundance of dysbiotic periodontal taxa when compared with refined carbohydrate–dominant diets.27 Their high levels of antioxidants and polyphenols limit oxidative stress and support epithelial barrier integrity, providing additional protection against periodontal inflammation.21 Overall, the evidence demonstrates that fiber-dense, prebiotic-rich, whole-food diets offer both local and systemic anti-inflammatory benefits through SCFA production, improved microbial balance, and reduced oxidative stress. Incorporating structured dietary counseling that promotes these patterns should be considered a key adjunct to comprehensive periodontal care.
Probiotics and Synbiotics
Probiotics are live microorganisms that, when administered in adequate amounts, confer health benefits to the host by modulating the microbial composition and immune responses. They have been investigated as adjunctive approaches for controlling periodontal inflammation.34 In the oral cavity, Lactobacillus and Bifidobacterium species inhibit pathogenic bacteria through competitive adhesion, bacteriocin production, hydrogen peroxide release, and local pH modulation.35 These actions suppress the growth of keystone periodontal pathogens such as Porphyromonas gingivalis and Tannerella forsythia and interfere with biofilm maturation and virulence expression. Emerging evidence shows that probiotics can also suppress biofilm virulence gene expression and downregulate metabolic pathways associated with proteolysis and lipopolysaccharide synthesis, indicating functional effects beyond simple changes in microbial composition.22,23
Probiotics also influence host inflammatory pathways. They attenuate NF-κB activation and reduce the secretion of IL-1β, IL-6, and TNF-α, while promoting the secretion of anti-inflammatory cytokines such as IL-10 and transforming growth factor-β.31,36 Specific strains, including Lactobacillus reuteri and Lactobacillus brevis, have demonstrated measurable clinical benefits. Randomized clinical trials report reductions in gingival crevicular fluid cytokines and improvements in bleeding scores, probing depth, and plaque indices when probiotics are used alongside conventional mechanical therapy.15,28
Synbiotics, which combine probiotics with prebiotic substrates, further enhance therapeutic potential by improving microbial colonization and metabolic activity.30 This synergy promotes short-chain fatty acid formation and supports epithelial barrier integrity while reducing oxidative stress and modulating local immune activation.21 Synbiotics may attenuate alveolar bone loss more effectively than probiotics alone through combined microbial and host-mediated pathways.37
Although the magnitude of clinical improvement is modest compared with scaling and root planing, probiotics and synbiotics consistently demonstrate benefits as adjunctive therapies that support microbial balance, reduce inflammatory burden, and enhance tissue healing.34,35 Their use may be especially valuable for patients with persistent inflammation, high recurrence risk, or systemic conditions that impair periodontal stability.
Anti-Inflammatory Dietary Patterns
Anti-inflammatory dietary patterns provide a clinically relevant means of modulating both periodontal inflammation and systemic immune responses. Among these, the Mediterranean and whole-food plant-based dietary models consistently demonstrate the strongest associations with improved periodontal parameters and reduced inflammatory burden. These diets emphasize omega-3 fatty acids, antioxidants, vitamins, polyphenols, and minimally processed plant foods, all of which exert direct and systemic effects on oxidative stress and immune regulation.15,28 These diets reduce oxidative stress by enhancing antioxidant enzyme activity and lowering lipid peroxidation, thereby limiting activation of inflammatory pathways such as NF-κB.21
Omega-3 fatty acids, particularly eicosapentaenoic acid and docosahexaenoic acid, generate specialized pro-resolving mediators such as resolvins, protectins, and maresins. These molecules actively terminate inflammation, enhance neutrophil clearance, and promote tissue repair rather than simply blocking cytokine production.38 Clinical trials show that omega-3 supplementation, especially when combined with scaling and root planing, reduces probing depth, gingival bleeding, and local concentrations of IL-1β and TNF-α, supporting their role as adjunctive periodontal therapies.13
Micronutrients are integral components of these dietary patterns. Vitamin C supports collagen synthesis, neutrophil activity, and epithelial healing, while vitamin D regulates antimicrobial peptide expression, bone turnover, and immune modulation.9,39 Deficiencies in these vitamins are associated with delayed wound healing, barrier dysfunction, and increased susceptibility to periodontal breakdown. Supplementation in individuals with deficiencies improves clinical parameters and enhances the response to periodontal therapy.40
Antioxidants from fruits, vegetables, teas, and whole grains, including flavonoids, carotenoids, and polyphenols, neutralize reactive oxygen species and suppress inflammatory signalling.41 Higher dietary intake of these bioactive compounds is consistently linked with lower gingival inflammation and reduced bleeding indices in clinical studies.28 These effects derive from improved redox balance, enhanced epithelial stability, and protection against cytokine-mediated tissue damage.
These findings show that anti-inflammatory dietary patterns influence periodontal health through several coordinated mechanisms, including reduced oxidative stress, improved immune regulation, and enhanced microbial balance. Integrating counselling on these dietary patterns with routine periodontal therapy provides a practical, evidence-supported approach for enhancing clinical outcomes.
Intermittent Fasting, Caloric Restriction, and Periodontal Health
Intermittent fasting (IF) and caloric restriction (CR) are increasingly recognised for their metabolic and anti-inflammatory effects. Their relevance to periodontal health is supported by mechanistic evidence and a growing number of clinical and experimental studies. IF involves structured intervals of fasting and feeding, while CR reduces overall caloric intake without malnutrition. Both strategies influence oxidative stress, cytokine activity, immune cell behaviour, and metabolic pathways that modulate periodontal inflammation.10,11,42
Biological Mechanisms Linking IF/CR to Inflammation
Fasting alters several biological pathways linked to periodontal tissue breakdown. Reduced nutrient availability improves mitochondrial efficiency and increases antioxidant enzyme activity, which lowers reactive oxygen species responsible for gingival and alveolar bone damage.43 Improved redox balance limits activation of NF-κB and Mitogen-Activated Protein Kinase (MAPK) pathways that drive osteoclastic bone resorption.21
IF also modifies cytokine networks. Multiple fasting interventions reduce circulating IL-1β, IL-6, and TNF-α, while increasing IL-10, reflecting a shift toward an anti-inflammatory state.13,44 Improved insulin sensitivity during fasting further reduces systemic inflammation and modulates immune cell behaviour within periodontal tissues.45 Autophagy is a conserved intracellular degradation pathway with a complex role in periodontal inflammation. Evidence from human gingival fibroblasts and inflamed periodontal tissues demonstrates that autophagy is upregulated in response to bacterial and cytokine stimulation, as evidenced by increase in the microtubule-associated protein 1 light chain 3 (LC3)-II/LC3-I ratio and LC3B expression.46 Rather than suppressing inflammation, this activation enhances IL-6, TNF-α, Cyclooxygenase-2 (COX-2), and Intercellular Adhesion Molecule-1 (ICAM-1) expression, indicating that autophagy can amplify inflammatory signalling in periodontal tissues. Inhibition of autophagy reduces these cytokines, showing that autophagy acts upstream of pro-inflammatory pathways.
Periodontal pathogens also suppress Mechanistic Target of Rapamycin (mTOR) signalling and decrease phosphorylation of Protein Kinase B (AKT) and Ribosomal Protein S6 Kinase beta-1 (S6K1), both of which promote autophagy and further reinforce inflammatory responses within gingival cells.46 A recent bibliometric analysis identified hotspots of autophagy-related research in periodontitis, including oxidative stress, apoptosis, immune regulation, and osteoclast activity. These findings confirm that autophagy contributes to periodontal tissue destruction when activated under dysbiotic or chronic inflammatory conditions.47
In summary, current evidence indicates that autophagy may play complex, context-dependent roles in inflammatory regulation. While fasting has been shown to modulate autophagic pathways, the implications of these effects for periodontal inflammation remain uncertain. Further experimental and clinical studies are needed to clarify whether modulation of autophagy through metabolic interventions has meaningful relevance to periodontal disease. These coordinated mechanisms provide strong biological justification for investigating IF and CR as modulators of periodontal inflammation. The mechanistic pathways are summarized in Figure 2.
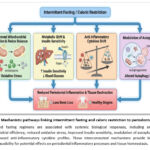 |
Figure 2: Mechanistic pathways linking intermittent fasting and caloric restriction to periodontal health. |
Structured fasting regimens are associated with systemic biological responses, including enhanced mitochondrial efficiency, reduced oxidative stress, improved insulin sensitivity, modulation of autophagy, and shifts toward anti-inflammatory cytokine profiles. These interconnected mechanisms provide biological plausibility for potential effects on periodontal inflammatory processes and tissue homeostasis.
Evidence from Human and Animal Studies
Human evidence directly assessing periodontal outcomes under intermittent fasting or caloric restriction remains limited, but emerging findings show meaningful biological improvements. A multimodal fasting intervention conducted in overweight adults with metabolic syndrome demonstrated significant reductions in bleeding on probing and gingival crevicular fluid volume, despite no change in plaque levels. These benefits persisted for four months and were accompanied by reductions in fasting glucose, waist circumference, oxidative stress markers, and systemic inflammatory cytokines, indicating that fasting-induced metabolic improvements can translate into measurable periodontal benefits.48
Another structured program combining caloric restriction with supervised lifestyle modification reported reductions in C-reactive protein, improved insulin sensitivity, and decreased oxidative stress.13 Additional clinical studies in metabolic syndrome populations show consistent reductions in C-reactive protein, IL-6, oxidative stress parameters, and fasting glucose following structured fasting regimens.45,49 Although these studies do not always measure periodontal endpoints, the systemic improvements they produce are closely linked to pathways regulating neutrophil function, endothelial integrity, and alveolar bone metabolism.
Animal models provide stronger direct evidence of periodontal effects. In ligature-induced periodontitis, alternate-day fasting reduces inflammatory cell infiltration, lowers gingival IL-1β and TNF-α expression, and preserves alveolar bone height.44 Long-term caloric restriction in non-human primates reduces plaque accumulation, probing depth, and attachment loss while lowering systemic inflammatory markers and improving immune regulatory profiles.12,50 Rodent models of caloric restriction similarly show reduced osteoclastic activity and slower alveolar bone resorption without impairing wound healing.43
Overall, evidence from controlled animal studies and early human interventions supports the concept that structured intermittent fasting or moderate caloric restriction can reduce periodontal inflammatory burden. The benefits appear to arise primarily through improvements in systemic inflammation, oxidative stress, and metabolic control. However, long-term randomized controlled trials with standardized periodontal outcomes and microbiome profiling are still required before fasting can be recommended as a routine adjunctive strategy in periodontal therapy.
Risks and Adverse Considerations
Although intermittent fasting and caloric restriction show therapeutic potential, certain risks warrant attention, particularly in patients with chronic oral conditions. Extended fasting can reduce salivary secretion, leading to xerostomia and increased risk of mucosal irritation and caries.51 Inadequate nutrient intake may lead to deficiencies in vitamins and antioxidants that are essential for maintaining epithelial integrity, collagen synthesis, and immune competence.9 Severe or unsupervised fasting can also impair immune defense and delay periodontal wound healing, especially in elderly or systemically compromised individuals.
Clinical implementation should therefore prioritize individualized regimens that ensure adequate micronutrient intake and hydration. Periodontal patients with diabetes, pregnancy, or metabolic disorders require medical supervision before initiating fasting interventions. In such cases, fasting should be integrated with nutritional counseling and standard periodontal therapy to optimize safety and outcomes.
Summary
Intermittent fasting and caloric restriction influence periodontal inflammation by reducing oxidative stress, regulating cytokines, and improving immune homeostasis. Evidence from animal models and preliminary human studies supports their anti-inflammatory potential, though long-term clinical validation remains limited. Controlled trials combining fasting regimens with standard periodontal care are needed to define effective, safe, and patient-specific strategies.
The Oral–Gut Microbiome Axis and Systemic Inflammation
The oral and gastrointestinal microbiomes are connected through continuous swallowing of saliva, which contains large numbers of viable oral bacteria. Increasing evidence shows that oral microbes can translocate to the gut, particularly under conditions of oral dysbiosis, impaired mucosal immunity, or increased intestinal permeability.52 Periodontal pathogens such as Porphyromonas gingivalis and Fusobacterium nucleatum can resist gastric acidity, adhere to intestinal epithelial cells, and modify the gut microbial community. Their presence disrupts tight junction proteins, weakens epithelial barrier function, and promotes T helper 1 cells (Th1) and Th17-skewed immune responses. Th17 cells are particularly implicated in the pathogenesis of chronic inflammatory and autoimmune conditions, driving systemic inflammation.14
Diet influences both the oral and gut microbiomes through shared ecological pressures. Diets rich in refined sugars and saturated fats promote inflammatory microbial profiles and increase gut permeability. In contrast, fiber-rich and polyphenol-containing foods increase microbial diversity and enhance short-chain fatty acid (SCFA) production. SCFAs such as butyrate strengthen epithelial barrier integrity and exert anti-inflammatory effects across both ecosystems.28,31 Nutritional improvement, therefore, represents a common intervention point for rebalancing the oral–gut microbial axis.
This bidirectional pathway helps explain epidemiological associations linking periodontitis with insulin resistance, cardiovascular disease, and other chronic inflammatory conditions. Microbial metabolites and endotoxins, such as lipopolysaccharide, can enter the systemic circulation from periodontal pockets, amplifying cytokine production and endothelial inflammation.53
Intermittent fasting appears to beneficially influence this axis by reducing systemic inflammation, improving gut barrier integrity, and altering nutrient availability to limit the growth of oral and gut pathobionts. The metabolic shift toward ketone-based energy utilisation may also reshape microbial activity in both niches.54 These connections are illustrated in Figure 3.
Together, these findings position the oral–gut axis as a shared therapeutic target. Integrating dietary modification and fasting-based metabolic strategies with standard periodontal care may reduce local periodontal inflammation and broader systemic inflammatory burden, supporting improved metabolic health.55–57
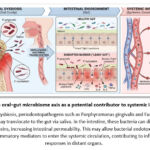 |
Figure 3: The oral–gut microbiome axis as a potential contributor to systemic inflammation. |
During oral dysbiosis, periodontopathogens such as Porphyromonas gingivalis and Fusobacterium nucleatum may translocate to the gut via saliva. In the intestine, these bacteria can disrupt tight junction proteins, increasing intestinal permeability. This may allow bacterial endotoxins (e.g., LPS) and pro-inflammatory mediators to enter the systemic circulation, contributing to inflammatory responses in distant organs.
Clinical Implications for Periodontal Practice
The integration of nutritional and metabolic assessment into periodontal care is becoming essential. Dietary behaviors influence microbial ecology, inflammatory tone, and healing capacity, and therefore should be routinely evaluated during periodontal examinations. Short, structured questions targeting refined sugar intake, fruit and vegetable consumption, and possible micronutrient deficiencies enable clinicians to identify modifiable contributors to inflammation rapidly.28,58 For patients with complex nutritional needs or comorbid systemic disease, referral to a registered dietitian or a qualified nutrition specialist provides more comprehensive management, improves patient adherence, and improves outcomes.57
Nutritional guidance should complement, not replace, standard mechanical therapy. Advising patients to reduce refined carbohydrates and processed foods while increasing whole grains, fruits, vegetables, and omega-3–rich foods has been shown to improve subgingival microbial profiles and reduce gingival bleeding, even without changes in oral hygiene practices.32,56 Vitamin C and vitamin D supplementation are appropriate only when deficiencies are confirmed, as low levels impair collagen synthesis, antimicrobial defenses, and bone metabolism.39
For medically stable adults, particularly those with metabolic syndrome, intermittent fasting (IF) or mild caloric restriction may serve as an adjunctive strategy. Approaches such as 16:8 time-restricted eating have demonstrated improvements in systemic inflammation, oxidative stress, and insulin sensitivity, all of which are closely linked to periodontal health.13,44 However, fasting should be introduced cautiously. Patients with diabetes, pregnancy, frailty, eating disorders, or significant systemic illness require medical clearance, and any fasting regimen should ensure adequate hydration and micronutrient intake.
Clinically, dentists should incorporate brief yet consistent dietary discussions during maintenance visits, use simple educational tools that illustrate diet–microbiome interactions, and document dietary and fasting habits as part of the patient’s periodontal risk profile. Reinforcement at follow-up visits is more effective than a single counseling session. In summary, reducing refined sugars, increasing fiber and antioxidant intake, and correcting micronutrient deficiencies are supported by substantial evidence and should be standard adjunctive strategies in periodontal maintenance. Data on fasting are encouraging but still preliminary; larger, longer-term clinical trials are needed. Until then, clinicians should apply IF selectively and within evidence-based boundaries to enhance control of inflammation and long-term periodontal stability.
Limitations and Gaps in Current Evidence
Current evidence linking diet, intermittent fasting, and caloric restriction to periodontal health remains promising but incomplete. A primary limitation is the predominance of small-scale, short-term studies with heterogeneous designs. Although several human trials have evaluated dietary modification and a smaller number have assessed structured fasting, direct measurements of periodontal outcomes remain limited, preventing broad generalizations. A significant gap is the lack of robust randomized controlled trials specifically designed to evaluate the impact of intermittent fasting or caloric restriction on the oral microbiome and periodontal health.
Dietary intervention research faces methodological challenges, including reliance on self-reported food intake, which is susceptible to recall bias, and a general lack of microbial sequencing or functional profiling to validate mechanistic changes in the oral microbiome. Furthermore, study populations exhibit substantial heterogeneity in baseline periodontal status, systemic health, and dietary habits, complicating cross-study comparisons.7,15
The short duration of most human trials, typically spanning only weeks to a few months, is likely insufficient to observe meaningful periodontal tissue remodeling or sustained microbial shifts. The field also lacks standardized fasting protocols, with significant variation in duration, timing, and energy restriction, resulting in inconsistent metabolic and inflammatory outcomes.45,49
Finally, mechanistic research directly linking dietary or fasting-induced metabolic shifts to changes in oral microbial ecology and gingival immunology remains sparse. Most studies focus on systemic biomarkers or animal models rather than integrating clinical, microbiological, and molecular endpoints. High-quality longitudinal trials that combine detailed periodontal assessments, microbiome sequencing, and metabolic profiling are essential to establish causality and define clinically relevant recommendations.
Future Directions
Future research must transition from observational associations to well-controlled clinical trials that directly evaluate the impact of diet and fasting on periodontal outcomes. Interventions based on anti-inflammatory, fiber-rich, or whole-food dietary patterns should be tested alongside conventional periodontal therapy to determine their additive value in reducing inflammation, improving clinical attachment levels, and modulating the subgingival microbiome. These trials must employ objective dietary assessments and standardized clinical endpoints to overcome the limitations of self-reported data. Future studies should employ integrated multi-omics methodologies, combining 16S rRNA gene sequencing, metatranscriptomics, and host metabolomic profiling, to move beyond correlation and establish causal pathways linking nutritional interventions to microbial function and periodontal tissue responses. Longitudinal studies should incorporate salivary and subgingival microbiome sequencing, metabolomics, and inflammatory proteomics to elucidate how nutritional and fasting regimens alter microbial ecology, host immune pathways, and tissue healing. Such comprehensive profiling is key to identifying patient-specific responses and developing personalized nutritional strategies for periodontal care. Gender-specific differences observed in animal models of caloric restriction underscore the need to investigate sex-specific metabolic and microbial responses in humans. Understanding the interaction between hormonal factors and nutritional interventions could enhance treatment precision.
Comparative trials examining different fasting regimens, such as time-restricted eating, alternate-day fasting, and moderate caloric restriction, are needed to identify the most effective and safest protocols for reducing periodontal inflammation. Standardizing these protocols is vital for reproducibility and for translating metabolic benefits into clinically meaningful periodontal outcomes. Nutritional and fasting strategies must be evaluated as adjuncts, not replacements, for established periodontal therapy.
Overall, future work must emphasize rigorous trial design, multi-omics integration, standardized interventions, and long-term follow-up. Such studies will be indispensable for defining evidence-based nutritional and metabolic interventions that can be safely and effectively incorporated into periodontal practice. The integrated pathways through which dietary and fasting interventions modulate metabolic and periodontal health are summarized in Figure 4.
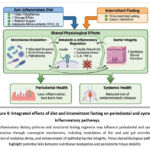 |
Figure 4: Integrated effects of diet and intermittent fasting on periodontal and systemic inflammatory pathways. |
Anti-inflammatory dietary patterns and structured fasting regimens may influence periodontal and systemic inflammation through convergent mechanisms, including modulation of the oral and gut microbiomes, reduction of oxidative stress, and enhancement of epithelial barrier integrity. These shared biological pathways highlight potential links between nutritional modulation and periodontal tissue stability
Conclusion
Diet and metabolic status appear to play an important role in periodontal health through multiple biological pathways, including modulation of the oral microbiome, epithelial barrier function, oxidative stress, and host inflammatory responses. Diets characterized by higher intake of fiber, antioxidants, and essential micronutrients are generally associated with greater microbial diversity and reduced gingival inflammation, whereas frequent consumption of refined sugars and pro-inflammatory nutrients has been linked with microbial dysbiosis and increased inflammatory activity.
Intermittent fasting and caloric restriction have been proposed as metabolic interventions that may influence pathways relevant to periodontal disease, including redox balance, cytokine signaling, insulin sensitivity, and cellular autophagy. Emerging experimental and limited clinical evidence suggests potential anti-inflammatory effects; however, current human data remain sparse and heterogeneous. Many studies involve small sample sizes, short follow-up periods, and inconsistent periodontal outcome measures, limiting definitive conclusions regarding clinical benefit.
Future research should prioritize well-designed longitudinal and interventional studies integrating standardized dietary assessment, clearly defined fasting protocols, microbiome profiling, and validated periodontal endpoints. Such work is necessary to clarify whether metabolic interventions meaningfully influence the progression of periodontal disease or treatment outcomes. At present, nutritional guidance focusing on reducing refined sugars and promoting balanced, nutrient-dense dietary patterns remains a reasonable component of periodontal health promotion, while the role of intermittent fasting should be considered exploratory and warrants further investigation.
Acknowledgement
All the authors thanks Abdullah Ali Abdullah Daghriri for his assistance in creating the figures used in this review.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author contributions
- Sameena Parveen: Conceptualization, Study Design, Methodology, Supervision, Writing – Original Draft, Writing – Review & Editing.
- Mohammed Mousa H. Bakri: Literature Search, Data Curation, Formal Analysis, Interpretation of Data, Writing – Review & Editing.
- Samar Saeed Khan: Methodology Support, Validation, Supervision, Writing – Review & Editing.
- Swetha Vempalli: Data Collection, Data Curation, Literature Review, Writing – Review & Editing.
- Reghunathan S. Preethanath: Data Collection, Formal Analysis, Visualization, Writing – Review & Editing.
- Mohammed Nazish Alam: Data Interpretation, Validation, Writing – Review & Editing.
- Mohamed Hamdi Mansy: Clinical Input, Data Interpretation, Critical Revision of Manuscript.
- Hind Ali Osman: Data Interpretation, Conceptual Input on Systemic Health Context, Writing – Review & Editing.
- Husham E. Homeida: Clinical Validation, Critical Revision, Writing – Review & Editing.
- Ahmed Mostafa Ahmed Mostafa: Methodological Review, Critical Revision, Writing – Review & Editing.
- Solima Ibrahim Yagoob: Data Verification, Literature Support, Writing – Review & Editing.
References
- Tonetti MS, Greenwell H, Kornman KS. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J Periodontol. 2018;89 Suppl 1:S159-S172. doi:10.1002/JPER.18-0006
CrossRef - Sedghi L, DiMassa V, Harrington A, Lynch SV, Kapila YL. The oral microbiome: Role of key organisms and complex networks in oral health and disease. Periodontol 2000. 2021;87(1):107-131. doi:10.1111/prd.12393
CrossRef - Deo PN, Deshmukh R. Oral microbiome: Unveiling the fundamentals. J Oral Maxillofac Pathol. 2019;23(1):122-128. doi:10.4103/jomfp.JOMFP_304_18
CrossRef - Hajishengallis G. Periodontitis: from microbial immune subversion to systemic inflammation. Nat Rev Immunol. 2015;15(1):30-44. doi:10.1038/nri3785
CrossRef - Elzayat H, Mesto G, Al-Marzooq F. Unraveling the Impact of Gut and Oral Microbiome on Gut Health in Inflammatory Bowel Diseases. Nutrients. 2023;15(15):15. doi:10.3390/nu15153377
CrossRef - Takahashi N. Oral Microbiome Metabolism: From “Who Are They?” to “What Are They Doing?” J Dent Res. 2015;94(12):1628-1637. doi:10.1177/0022034515606045
CrossRef - Woelber JP, Bremer K, Vach K, et al. An oral health optimized diet can reduce gingival and periodontal inflammation in humans – a randomized controlled pilot study. BMC Oral Health. 2016;17. doi:10.1186/s12903-016-0257-1
CrossRef - Alhassani AA, Hu FB, Li Y, Rosner BA, Willett WC, Joshipura KJ. The associations between major dietary patterns and risk of periodontitis. J Clin Periodontol. 2021;48(1):2-13. doi:10.1111/jcpe.13380
CrossRef - Ustianowski Ł, Ustianowska K, Gurazda K, Rusiński M, Ostrowski P, Pawlik A. The Role of Vitamin C and Vitamin D in the Pathogenesis and Therapy of Periodontitis—Narrative Review. Int J Mol Sci. 2023;24(7):6774. doi:10.3390/ijms24076774
CrossRef - Mattson MP, Longo VD, Harvie M. Impact of intermittent fasting on health and disease processes. Ageing Res Rev. 2017;39:46-58. doi:10.1016/j.arr.2016.10.005
CrossRef - Di Francesco A, Di Germanio C, Bernier M, de Cabo R. A time to fast. Science. 2018;362(6416):770-775. doi:10.1126/science.aau2095
CrossRef - Branch-Mays GL, Dawson DR, Gunsolley JC, et al. The effects of a calorie-reduced diet on periodontal inflammation and disease in a non-human primate model. J Periodontol. 2008;79(7):1184-1191. doi:10.1902/jop.2008.070629
CrossRef - Pappe CL, Steckhan N, Hoedke D, et al. Prolonged multimodal fasting modulates periodontal inflammation in female patients with metabolic syndrome: A prospective cohort study. J Clin Periodontol. 2021;48(4):492-502. doi:10.1111/jcpe.13419
CrossRef - Park SY, Hwang BO, Lim M, et al. Oral–Gut Microbiome Axis in Gastrointestinal Disease and Cancer. Cancers (Basel). 2021;13(9):2124. doi:10.3390/cancers13092124
CrossRef - Jeong J, Kim HS, Lee D, Kim K, Kim YH. Association between Four Dietary Patterns and the Risk of Periodontal Diseases: A Systematic Review and Meta-Analysis. Nutrients. 2022;14(20):4362. doi:10.3390/nu14204362
CrossRef - Avila M, Ojcius DM, Yilmaz O. The oral microbiota: living with a permanent guest. DNA Cell Biol. 2009;28(8):405-411. doi:10.1089/dna.2009.0874
CrossRef - Harrandah AM. The Oral–Gut–Systemic Axis: Emerging Insights into Periodontitis, Microbiota Dysbiosis, and Systemic Disease Interplay. Diagnostics. 2025;15(21):2784. doi:10.3390/diagnostics15212784
CrossRef - Parveen S, Alqahtani AS, Aljabri MY, et al. Nationwide exploration: assessing oral microbiome knowledge among dental professionals in Saudi Arabia and its implications for oral health care. BMC Oral Health. 2024;24(1):1028. doi:10.1186/s12903-024-04770-0
CrossRef - Cekici A, Kantarci A, Hasturk H, Van Dyke TE. Inflammatory and immune pathways in the pathogenesis of periodontal disease. Periodontol 2000. 2014;64(1):57-80. doi:10.1111/prd.12002
CrossRef - Chapple ILC, Matthews JB. The role of reactive oxygen and antioxidant species in periodontal tissue destruction. Periodontol 2000. 2007;43:160-232. doi:10.1111/j.1600-0757.2006.00178.x
CrossRef - Greabu M, Giampieri F, Imre MM, et al. Autophagy, One of the Main Steps in Periodontitis Pathogenesis and Evolution. Molecules. 2020;25(18):18. doi:10.3390/molecules25184338
CrossRef - Lira-Junior R, Aogáin MM, Crncalo E, et al. Effects of intermittent fasting on periodontal inflammation and subgingival microbiota. J Periodontol. 2024;95(7):640-649. doi:10.1002/JPER.23-0676
CrossRef - Chen TP, Yu HC, Lin WY, Chang YC. Bidirectional association between obesity and chronic periodontitis: Inflammatory pathways and clinical implications. J Dent Sci. 2025;20(4):2021-2025. doi:10.1016/j.jds.2025.07.009
CrossRef - Berlin-Broner Y, Febbraio M, Levin L. Association between apical periodontitis and cardiovascular diseases: a systematic review of the literature. Int Endod J. 2017;50(9):847-859. doi:10.1111/iej.12710
CrossRef - Schulze-Späte U, Wurschi L, van der Vorst EPC, et al. Crosstalk between periodontitis and cardiovascular risk. Front Immunol. 2024;15. doi:10.3389/fimmu.2024.1469077
CrossRef - Allen EM, Matthews JB, O’ Halloran DJ, Griffiths HR, Chapple IL. Oxidative and inflammatory status in Type 2 diabetes patients with periodontitis. J Clin Periodontol. 2011;38(10):894-901. doi:10.1111/j.1600-051X.2011.01764.x
CrossRef - Pang L, Zhi Q, Jian W, Liu Z, Lin H. The Oral Microbiome Impacts the Link between Sugar Consumption and Caries: A Preliminary Study. Nutrients. 2022;14(18):3693. doi:10.3390/nu14183693
CrossRef - Woelber JP, Gärtner M, Breuninger L, et al. The influence of an anti-inflammatory diet on gingivitis. A randomized controlled trial. J Clin Periodontol. 2019;46(4):481-490. doi:10.1111/jcpe.13094
CrossRef - Nielsen SJ, Trak-Fellermeier MA, Joshipura K, Dye BA. Dietary Fiber Intake Is Inversely Associated with Periodontal Disease among US Adults. J Nutr. 2016;146(12):2530-2536. doi:10.3945/jn.116.237065
CrossRef - Gibson GR, Hutkins R, Sanders ME, et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol. 2017;14(8):491-502. doi:10.1038/nrgastro.2017.75
CrossRef - Cani PD, Van Hul M, Lefort C, Depommier C, Rastelli M, Everard A. Microbial regulation of organismal energy homeostasis. Nat Metab. 2019;1(1):34-46. doi:10.1038/s42255-018-0017-4
CrossRef - Kondo K, Ishikado A, Morino K, et al. A high-fiber, low-fat diet improves periodontal disease markers in high-risk subjects: a pilot study. Nutr Res. 2014;34(6):491-498. doi:10.1016/j.nutres.2014.06.001
CrossRef - LaMonte MJ, Williams AM, Genco RJ, et al. Association between metabolic syndrome and periodontal disease measures in postmenopausal women: the Buffalo OsteoPerio study. J Periodontol. 2014;85(11):1489-1501. doi:10.1902/jop.2014.140185
CrossRef - Hill C, Guarner F, Reid G, et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11(8):506-514. doi:10.1038/nrgastro.2014.66
CrossRef - Sampaio-Maia B, Caldas IM, Pereira ML, Pérez-Mongiovi D, Araujo R. Chapter Four – The Oral Microbiome in Health and Its Implication in Oral and Systemic Diseases. In: Sariaslani S, Michael Gadd G, eds. Advances in Applied Microbiology. Vol 97. Academic Press; 2016:171-210. doi:10.1016/bs.aambs.2016.08.002
CrossRef - Parveen S, Alqahtani AS, Aljabri MY, et al. Exploring the Interplay: Oral–Gut Microbiome Connection and the Impact of Diet and Nutrition. European Journal of General Dentistry. 2024;13:165-176. doi:10.1055/s-0044-1786154
CrossRef - Al-Habsi N, Al-Khalili M, Haque SA, Elias M, Olqi NA, Al Uraimi T. Health Benefits of Prebiotics, Probiotics, Synbiotics, and Postbiotics. Nutrients. 2024;16(22):3955. doi:10.3390/nu16223955
CrossRef - Lee CT, Tribble GD. Roles of specialized pro-resolving mediators and omega-3 polyunsaturated fatty acids in periodontal inflammation and impact on oral microbiota. Front Oral Health. 2023;4:1217088. doi:10.3389/froh.2023.1217088
CrossRef - Lešić S, Ivanišević Z, Špiljak B, Tomas M, Šoštarić M, Včev A. The Impact of Vitamin Deficiencies on Oral Manifestations in Children. Dent J (Basel). 2024;12(4):109. doi:10.3390/dj12040109
CrossRef - Bačun B, Galić D, Pul L, Tomas M, Kuiš D. Manifestations and Treatment of Hypovitaminosis in Oral Diseases: A Systematic Review. Dent J (Basel). 2024;12(6):152. doi:10.3390/dj12060152
CrossRef - Rahaman MdM, Hossain R, Herrera‐Bravo J, et al. Natural antioxidants from some fruits, seeds, foods, natural products, and associated health benefits: An update. Food Sci Nutr. 2023;11(4):1657-1670. doi:10.1002/fsn3.3217
CrossRef - Patterson RE, Sears DD. Metabolic Effects of Intermittent Fasting. Annu Rev Nutr. 2017;37:371-393. doi:10.1146/annurev-nutr-071816-064634
CrossRef - Reynolds MA, Dawson DR, Novak KF, et al. Effects of caloric restriction on inflammatory periodontal disease. Nutrition. 2009;25(1):88-97. doi:10.1016/j.nut.2008.07.003
CrossRef - Wulansari L, Kaboosaya B, Khan M, et al. Beneficial effects of fasting regimens on periodontal tissues in experimental periodontitis mice model. Journal of International Dental and Medical Research. 2018;11:362-369.
- Jamshed H, Steger FL, Bryan DR, et al. Effectiveness of Early Time-Restricted Eating for Weight Loss, Fat Loss, and Cardiometabolic Health in Adults With Obesity: A Randomized Clinical Trial. JAMA Internal Medicine. 2022;182(9):953-962. doi:10.1001/jamainternmed.2022.3050
CrossRef - Kim WJ, Park SY, Kim OS, Park HS, Jung JY. Autophagy upregulates inflammatory cytokines in gingival tissue of patients with periodontitis and lipopolysaccharide‐stimulated human gingival fibroblasts. J Periodontol. 2022;93(3):380-391. doi:10.1002/JPER.21-0178
CrossRef - Zhang B, Wang Y, Chang H, Wang C, Fan H, Ren X. The role of autophagy in periodontal diseases: a bibliometric analysis from 2006 to 2023. Front Med. 2025;12. doi:10.3389/fmed.2025.1493459
CrossRef - Pappe CL, Maetschker J, Dujardin S, et al. Intermittent Fasting Regimes Reduce Gingival Inflammation: A Three‐Arm Clinical Trial. J Clin Periodontol. 2025;52(5):681-694. doi:10.1111/jcpe.14151
CrossRef - Teong XT, Liu K, Vincent AD, et al. Intermittent fasting plus early time-restricted eating versus calorie restriction and standard care in adults at risk of type 2 diabetes: a randomized controlled trial. Nat Med. 2023;29(4):963-972. doi:10.1038/s41591-023-02287-7
CrossRef - Ebersole JL, Steffen MJ, Reynolds MA, et al. Differential Gender Effects of a Reduced Calorie Diet on Systemic Inflammatory and Immune Parameters in Nonhuman Primates. J Periodontal Res. 2008;43(5):500-507. doi:10.1111/j.1600-0765.2008.01051.x
CrossRef - Attinà A, Leggeri C, Paroni R, et al. Fasting: How to Guide. Nutrients. 2021;13(5):1570. doi:10.3390/nu13051570
CrossRef - Kitamoto S, Nagao-Kitamoto H, Hein R, Schmidt TM, Kamada N. The Bacterial Connection between the Oral Cavity and the Gut Diseases. J Dent Res. 2020;99(9):1021-1029. doi:10.1177/0022034520924633
CrossRef - Olsen I, Yamazaki K. Can oral bacteria affect the microbiome of the gut? J Oral Microbiol. 2019;11(1):1586422. doi:10.1080/20002297.2019.1586422
CrossRef - Graef FA, Celiberto LS, Allaire JM, et al. Fasting increases microbiome-based colonization resistance and reduces host inflammatory responses during an enteric bacterial infection. PLOS Pathogens. 2021;17(8):e1009719. doi:10.1371/journal.ppat.1009719
CrossRef - Parveen S, Alhazmi YA. Impact of Intermittent Fasting on Metabolic Syndrome and Periodontal Disease—A Suggested Preventive Strategy to Reduce the Public Health Burden. International Journal of Environmental Research and Public Health. 2022;19(21):14536. doi:10.3390/ijerph192114536
CrossRef - Parveen S, Ara SA, Preethanath RS, et al. Exploring the knowledge of oral health and nutrition among Saudi Arabian dental professionals: a nationwide survey. BMC Oral Health. 2024;24(1):1543. doi:10.1186/s12903-024-05363-7
CrossRef - Parveen S. Investigating the Role of Personalized Nutritional Counseling in Enhancing Oral Health Management: A Cross-Sectional Survey among Saudi Arabian Dental Practitioners. European Journal of General Dentistry. Published online February 3, 2025. doi:10.1055/s-0044-1801803
CrossRef - Anchidin-Norocel L, Lobiuc A, Covasa M. Diet–Oral Microbiota Interactions and Salivary Biomarkers of Nutritional Health: A Narrative Review. Nutrients. 2026;18(3). doi:10.3390/nu18030396
CrossRef







