Manuscript accepted on :08-03-2026
Published online on: 18-03-2026
Plagiarism Check: Yes
Reviewed by: Dr. Huzef U
Second Review by: Dr. Hany Akeel
Final Approval by: Dr. Mariia Shanaida
Yanuarita Tursinawati1* , Lukman Faishal Fatharani 1
, Lukman Faishal Fatharani 1 , Amin Samiasih2
, Amin Samiasih2 , Ardita Nadia Zahirah3
, Ardita Nadia Zahirah3 , Shafa Noor Aulia3
, Shafa Noor Aulia3 and Ahmad Reihanzaki3
and Ahmad Reihanzaki3
1Department of Biomedical, Faculty of Medicine, Universitas Muhammadiyah Semarang, Semarang, Indonesia
2 Department of Nursing, Faculty of Nursing and Health Sciences, Universitas Muhammadiyah Semarang, Semarang, Indonesia
3Faculty of Medicine, Universitas Muhammadiyah Semarang, Semarang, Indonesia
Corresponding Author E-mail: yanuarita_tursina@unimus.ac.id
DOI : https://dx.doi.org/10.13005/bpj/3372
Abstract
Melatonin in beans is known for regulating glucose absorption in muscles and intracellular transformation with Glucose Transporter type 4 (GLUT 4) through Inositol-trisphosphate 3 (IP3) kinase activation. The efficacy of functional food ingredients has not been fully explored, and there is still a lack of information about the action mechanism of the hypoglycaemic effect and the relationship to GLUT 4 gene expression in beans. Therefore, this research aims to explore the hypoglycaemic efficacy of red beans (Phaseolus vulgaris L) and GLUT 4 gene expression in diabetic rats. This experimental research with a post-test only control group design was conducted on male Wistar rats that divided into KN (without STZ induction), K- (induced by Streptozotocin (STZ) 45 mg/kgBW), K+ (induced by STZ+Metformin), P1 (induced by STZ+red beans extract dose 0.35 g/200 gBody Weight), P2 (induced by STZ+red beans extract dose 0.7 g/200 gBW). After 21 days, the rats were examined for Fasting Plasma Glucose (FPG), while GLUT 4 gene expression was analyzed by qPCR. ANOVA test used to analyze the mean differences between groups. Qualitatively, red beans extract contain melatonin with IC50 0,4057 mg/mL (Acarbose 0.0138 mg/mL). There was a significant decrease in FPG in groups P1 (p = 0.043) and P2 (p = 0.023). The red bean extract group showed up-regulation of GLUT 4 gene expression (P1 = 52.77, P2=71.75). The correlation between GLUT 4 and FPG had no significant results with p = 0.725, and r = -0.074. Red beans extract contained melatonin has α-Glucosidase inhibitory activity although the power still below than acarbose. Administration of melatonin contained in red bean extract at a dose of 0.7 g/200 gBW can increase GLUT 4 gene expression than in lower dose. Melatonin contained in melatonin may contribute to improved glucose metabolism, but the quantitative levels of melatonin needs to be measured through further research.
Keywords
Diabetes; Glucose; GLUT 4; Melatonin; Red bean
Download this article as:| Copy the following to cite this article: Tursinawati Y, Fatharani L. F, Samiasih A, Zahirah A. N, Aulia S. N. Reihanzaki A. Effects of Melatonin-Rich Extract from Red Beans (Phaseolus Vulgaris L.) on Glucose Levels and GLUT4 Gene Expression in Diabetic Rats. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Tursinawati Y, Fatharani L. F, Samiasih A, Zahirah A. N, Aulia S. N. Reihanzaki A. Effects of Melatonin-Rich Extract from Red Beans (Phaseolus Vulgaris L.) on Glucose Levels and GLUT4 Gene Expression in Diabetic Rats. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/4lJ43zm |
Introduction
Melatonin is a hormone produced by the pineal gland for regulating human physiological rhythms, insomnia, and the immune system. The hormone has anti-inflammatory and antioxidant effects to ward off free radicals.1 Melatonin plays a role in the incidence of cardiovascular disease, obesity, and diabetes mellitus (DM) while regulating carbohydrate metabolism. Previous research showed that pinealectomy caused glucose intolerance and insulin resistance in DM2. This surgical process is responsible for the absence of melatonin, decreased expression of Glucose Transporter type 4 (GLUT4), glucose intolerance, as well as insulin resistance.2
Efforts to treat and prevent effect of hyperglycaemia on DM have been carried out by seeking other alternative treatments. The use of pharmacological treatments and nutritional therapy can help prevent the progression of DM. A typical example is beans, such as red beans (Phaseolus vulgaris L.), which are a valuable source of nutrients.3 The antihyperglycemic effect of red beans is played by the flavonoids, saponins, tannins, alkaloids, and terpenoids.4 Several research have been conducted on the benefits of red beans in reducing blood glucose levels in experimental animals. Zuo A. et al. (2023) found that Fasting Plasma Glucose (FBG) levels decreased significantly in the group given red bean polyphenol metallothionein complex.5 Many food sources have been proven to contain melatonin such as in tomato, cherries, soybean seeds, red beans and pistachio.1 For instance, red beans contain 54.79 ± 0.79 ng/g levels of melatonin.6 It is suggesting that plant sources may serve as a potential natural source of melatonin. Whereas, administration of melatonin to experimental animals injected with Streptozotocin (STZ)-Nicotinamide (NA) for 6 weeks causes an increase in insulin and superoxide dismutase activity as well as a decrease in hepatic Malondialdehyde which plays a role in the pathophysiology of diabetes.7
The mechanism regulates blood sugar levels by regulating glucose uptake in adipocytes through GLUT expression modulation. Melatonin activates glucose use in muscles and intracellular transformation with GLUT 4 through IP3 kinase activation.8 GLUT4 is an insulin-regulated glucose transporter responsible for the uptake into muscle and fat cells . Research measuring expression in the soleus muscle of DM rats showed that GLUT 4 protein and gene expression decreased in the DM group. Other results found that miR-29b-3p and miR29c-3p expression were negatively correlated with GLUT 4 gene expression.9 This was supported by Tursinawati et al., 2022 which proved the overexpression of mir29b in type 2 DM Javanese patients.10 Mir29b is a regulatory factor of MTNR1B, the gene encoding the melatonin receptor MT2.
An in vivo research on type 1 DM rats given 200 mg/kg BW of red bean water extract showed an increase in GLUT 4 protein in DM rat muscles compared to rats not given red beans.11 GLUT-4 is stored in intracellular vesicles, and its function is influenced by the IRS-1/PI3K/Akt pathway, which is a key determinant of insulin sensitivity and glucose homeostasis. Other kidney bean species, such as adzuki bean (Vigna angularis), have been studied to improve insulin signalling by increasing IRS1 and AKT1 protein expression in human liver cells.12 The efficacy of functional food ingredients containing melatonin in red beans has not been fully explored, and there is still a lack of information related to the mechanism of action of the hypoglycaemic effect as well as the relationship to GLUT 4 gene expression. Therefore, this research aims to explore the hypoglycaemic efficacy of melatonin in red beans and on GLUT 4 gene expression in diabetic experimental animals.
Material and Methods
Preparation of red bean extract
A total of 3 kg of red beans purchased from local vendor in Semarang, were dried at a temperature of 105o C for 6 hours. The dried beans were manually rubbed to remove the seed coat, then grinded and sieved through a 60-mesh sieve. Furthermore, the sieved results were dried at a temperature of 105o C for 1 hour. A total of 150 grams of simplicial powder was macerated using 96% ethanol solvent (1:10).
Melatonin Test of Red Bean Extract
The melatonin test was carried out qualitatively using the thin-layer chromatography method, which was read under UV light and Rf calculation. The test was carried out by adding 1g of ethanol extract from beans and 15 ml of 15% acetic acid before filtering using filter paper. The filtrate was extracted with 50 mL of petroleum ether, after which the acetic acid extract was collected and adjusted to pH 10 using NH₄OH. The ether extract was separated and evaporated to dryness, then re-dissolved in 5.0 mL of ethanol solvent. The ethanol solution and the comparator in the form of standard melatonin were spotted on a silica gel plate for preparative purposes. The elution was conducted using the BAW mobile phase (n-Butanol: Acetic acid: Water) with a ratio of 12:3:5.
Antidiabetic test of red bean extract (α-Glucosidase inhibitory activity)
The α-Glucosidase enzyme inhibition activity test was carried out using a microplate reader with a substrate in the form of p-nitrophenyl-α-D-glucopyranoside (p-NPG) and α-glucosidase enzyme. The reaction mixture consisted of blank, control, sample, and control. A 17 μL substrate was added and incubated at 37oC for 30 minutes. The reaction was stopped by adding 100 μL of 2 M Na2CO3 and measured at 410 nm wavelength. Acarbose solution was used as a positive control and the experiment was carried out 3 times. Furthermore, the % inhibition was calculated to determine the IC50 value.
Experimental design and animal treatment
Experimental research with pre and post test randomized controlled group design was used in male Wistar rats induced by STZ (Sigma-Aldrich, USA). A single dose of 45 mg/kg was administered intraperitoneally to Wistar rats, aged 10 weeks and weighing 170 – 220 g. These animals were divided into five groups as follows KN = without STZ induction; K- = induced by STZ; K+ = induced by STZ, given metformin at a dose of 4.5 mg/kg BW; P1 = induced by STZ, given red bean extract 0.35 g/200 gBW orally every day for 21 days; P2 = induced by STZ, given red bean extract 0.70 g/200 gBW orally every day for 21 days. The dose of beans that can decrease blood sugar in humans is 28 grams. Therefore, conversion for rats is 0.35 g/200 gBW and 0.70 g/200 gBW in 10 ml of distilled water. This experimental study has received ethical approval.
Clinical Chemistry Analysis
The rats were fasted for 8 hours, and 2 ml of blood was taken from the orbital vein for Fasting Plasma Glucose (FPG). Subsequently, surgery was performed to remove the gastrocnemius muscle for GLUT 4 gene expression.
GLUT 4 Gene Expression with quantitative PCR (qPCR)
Gastrocnemius muscles taken during termination were soaked in RNA latter solution to be extracted using the Tissue Total RNA Mini Kit (Favorgen). The purity of extracted RNA was checked with nanodrop before subjection to real-time PCR using the SensiFAST SYBR No-ROX One-Step Kit Reagent. A total of 4 µL c DNA was added to 10 µL SensiFAST SYBR® No-ROX One-Step Mix (Meridian Bioscience Cat BIO-72001), 0.8 µL primers (Table 1) and 3.8 µL H2O. Amplification was carried out with a program of 40 cycles, with 1 cycle consisting of initial denaturation at 95°C for 2 minutes, denaturation at 95°C for 5 seconds, and annealing at 60°C for 30 seconds.
The relative quantification method was calculated using the Livak formula to analyze expression of GLUT 4 gene. A fold change greater than or less than 1 shows upregulation and downregulation, respectively.
Relative expression = 2-∆∆Ct
ΔCt= Ct (target gene) – Ct (housekeeping gene / β actin)
ΔΔ CT = Δ CT (case) – Δ (control)
Statistical analysis
Frequency data and graphs were analyzed using GraphPad Prism software (GraphPad Software, USA). The normality of FPG data was tested using the Shapiro-Wilk test using SPSS. The results showed a parametric data distribution continued with the ANOVA test to analyze the mean differences between groups. Pearson correlation test investigated the correlation of FPG levels with GLUT 4 gene expression values (significance p ≤ 0.05).
Results
Melatonin content of red bean extract
In this research, red beans contained melatonin with a difference of Rf 0 according to the calculation of the difference between the sample and standard Rf. Rf calculation can be carried out using the formula b/a where a is the distance traveled by the eluen between the initial limit of the liquid to the movement, and b is the distance traveled by the analyte between the initial point of the sample spot to the diameter of the circle. The Rf of the sample is 0.9733 with a standard Rf of 0.9733 and a final Rf of 0 (≤ 0.2). Qualitatively, a sample contains melatonin if the difference between the sample’s Rf and the standard Rf is ≤ 0.2. Therefore, this study can conclude that red beans positively contain melatonin because the result have an Rf difference of 0.
Table 1: Primers used in q PCR
| Gen | Forward primer | Reverse primer |
| Glut 4 | GGCCGGGACACTATACCCTA | GGAGGAAATCATGCCACCCA |
| β actin | AGGCCCCTCTGAACCCTAAG | ATGTCACGCACGATTTCCCT |
Inhibitory activity of α-Glucosidase
The test of the inhibitory activity of α-Glucosidase from extract of red beans and the acarbose standard is presented in table 2 which showed that acarbose still has a lower IC50 (0.0138 mg/mL) than the IC50 of red bean extract (0.4057 mg/mL).
Table 2: IC50 of acarbose and red beans extract
| Sample | IC50 (mg/mL) |
| Acarbose | 0.0138 |
| Red beans extract | 0,4057 |
Rat Body Weight
Based on Figure 1, the body weight (BW) of rats before being given STZ in all groups tended to increase but decreased after the injection. Meanwhile, the group of rats not injected with STZ experienced an increase in weight. At the beginning of treatment, the average weight was 164.8 – 175.8 g. At the end of treatment, the average weight of the rats was the lowest at K- (149.2 g). During treatment, the weight of DM rats tended to decrease, not significantly different with p = 0.226.
FPG Parameters for Each Group
FPG before treatment (pre test) was lowest in the KN group not injected with STZ, while the highest was in the K+ group. FPG levels were rechecked after STZ treatment on the 11th day (post test). The lowest and the highest results remained in the KN and K- groups. If FPG was compared between pre test groups, p value was 0.0007 and FPG post test between groups, p = 0.171. A decrease in FPG occurred in all groups except the K- group. A significant reduction occurred in the FPG of treatment group P1 (p = 0.043) and P2 (p = 0.023) (Figure.2).
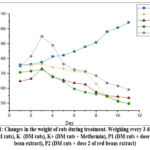 |
Figure 1: Changes in the weight of rats during treatment. Weighing every 3 days. KN (non-DM rats), K- (DM rats), K+ (DM rats + Metformin), P1 (DM rats + dose 1 of red bean extract), P2 (DM rats + dose 2 of red beans extract). |
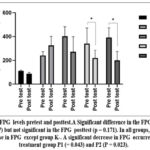 |
Figure 2: FPG levels pretest and posttest.A Significant difference in the FPG pretest (p = 0.0007) but not significant in the FPG posttest (p = 0.171). In all groups, there was a decrease in FPG except group K-. A significant decrease in FPG occurred in the treatment group P1 (= 0.043) and P2 (P = 0.023). |
GLUT 4 gene expression in each group
Based on the RNA purity test results, the A260/280 ratio value ranged from 1.8 to 2.0, hence the sample met good criteria. As shown in Table 3, the highest and lowest GLUT 4 gene expression was found in groups K+ (76.52) and K- (36.19), respectively. In all groups, GLUT 4 gene expression was upregulated. Administration of red bean extract to P2 can increase expression of GLUT 4 gene 71.75 times.
Correlation between Fasting Plasma Glucose levels and GLUT 4 expression
Based on Table 4, there was no correlation between GLUT 4 expression and FPG, with p = 0.725, r= -0.074 as well as a negative trend.
Discussion
Legumes such as red beans have a low glycaemic index and water-soluble fiber content for regulating insulin release, as well as slowing gastric emptying and absorption of sugar, with the potential to be hypoglycaemic in DM. However, there are other hypoglycaemic contents in red beans, including antioxidant contents such as melatonin ( in plants known as phytomelatonin). Red beans contain phytomelatonin even though the measurements were carried out qualitatively. In addition to phytomelatonin, red beans contain lectins, isoflavones, and trypsin, which have antihyperglycemic properties.13 Other bioactive compounds in red bean extract are predicted to produce a synergistic effect with phytomelatonin, an antidiabetic agent. Study by Aguilera et al., 2015 which measured melatonin in red bean extract with methanol using the HPLC-ESI-MS method14. However, the melatonin content is lower compared to other plant foods such as almonds, sunflowers, and mustard. The content in grains is affected by several factors, including variety, harvest time, location, and germination time. On the sixth day of germination, melatonin reached the highest level in red bean sprouts at 529.1 ng/g DW.14 Higher melatonin levels were also successfully identified by Saleh et al., 2019 at 5.85 ng/g.15 A total of 54.79 ± 0.79 ng/g melatonin was also found in red beans as reported by Sangsopa et al., 20206. Melatonin is an endogenous neurohormone synthesized from tryptophan through serotonin in the pineal gland of animals or humans. The hormone is also found on plants cultivated in Indonesia, such as ginger, as reported by Tursinawati et al., 2024.16 The role is reported as a radical scavenger, an antioxidant, and an antidiabetic agent through anti-inflammatory and antioxidant mechanisms, improving mitochondrial function or increasing insulin sensitivity.17
Table 3: Expression of GLUT 4 gene
| Group | Average ΔCt | ΔΔ CT | 2-∆∆Ct | Regulation | p |
| KN | -0.20 | 0.00 | 81.36 | upregulation | 0.0028 |
| K – | 1.77 | 1.97 | 36.19 | upregulation | |
| K + | 0.59 | 0.79 | 76.52 | upregulation | |
| P1 | -2.85 | -2.65 | 52.77 | upregulation | |
| P2 | -5.25 | -5.05 | 71.75 | upregulation |
Table 4: Correlation between GLUT 4 gene expression and FPG
| Fasting Blood Glucose | ||
| GLUT 4 expression | p | 0.725 |
| r | -0.074 |
A method used to overcome DM is by inhibiting the work of enzymes that hydrolyze carbohydrates to reduce glucose absorption. Additionally, the α-Glucosidase enzyme plays an important role in breaking down oligosaccharides or polysaccharides into monosaccharides. Inhibition of the α-Glucosidase enzyme is proven by red beans through the analysis. Determination of the magnitude of activity inhibition of the α-Glucosidase enzyme is seen from the IC50 value, where the results of acarbose still have a lower IC50 than the IC50 of red bean extract. The lower the IC50 value, the greater the inhibitory activity of a compound. The results of this study indicate that red bean extract has a lower inhibitory power of the alpha glucosidase enzyme than acarbose. These results are in line with other studies where the IC50 inhibition of the enzyme showed that red bean extract with ethanol solvent gave a significantly lower IC50 (2.60 ± 0.61 mg/ml) compared to ethyl acetate and water solvents with IC50 of 3.07 ± 0.61 mg/ml and 2.82 ± 0.14 mg/ml, respectively, although its antidiabetic ability is still lower than the standard acarbose.18 Compounds in beans suspected of having an antidiabetic effect are trypsin and isoflavones. These compounds inhibit α-glucosidase or are rich in phenol content as an antidiabetic and cardiovascular disease.13
Hypoglycaemic effect was tested in vivo using experimental rats injected with STZ to make DM rats. The weight of rats after being given STZ tended to decrease but not significantly. Meanwhile, the weight of the rats that were not injected with STZ increased. The injection of STZ 50 mg kg/BW caused a decrease in BW, but when another group was given STZ + NA 230 mg/Kg BW, The BW was higher than the STZ group. This proves that DM experimental animals with the addition of NA prevented the mortality of DM mice.19 The results are appropriate to other research on the BW of DM rats, where STZ injection could reduce BW on day 21.20 However, there was an insignificant increase after waiting for 6 weeks. This is different from the results, where the BW of rats continued to experience a decrease until day 21. The administration of metformin showed low BW in rats as reported by other study. Metformin increases fatty acid absorption and use of adipose tissue to stimulate fatty acid oxidation and inhibits fat accumulation.21,22
This research identified FPG levels as a parameter for diabetes control. Before treatment, FPG was checked, where the lowest was in the KN group that was not injected with STZ. Meanwhile, the highest was in the K+ DM group that was given metformin. The lowest FPG levels after treatment on the 21st day (post test) remained in the KN group, but the highest was in the K- group. There were significant results when compared between groups. This was consistent with Zhou et al., 2024, where there was no significant difference on the 28th day when comparing the control, DM, and DM + Metformin groups. A significant difference only occurred in the 7th week, where the DM group reached the highest FPG levels. Therefore, STZ injection has completely damaged the pancreatic beta cells, increasing FPG.23 Pre test and post test FPG were compared, and the groups with significant decreases were P1 and P2. In the K+ group, consisting of DM rats with metformin, there was an insignificant decrease in FPG. The metformin used was a dose of 500 mg/kg BW for 21 days. The administration at a dose of 300 mg/kg for 8 weeks has an anti-diabetic effect on STZ-induced DM rats and given a high-fat diet through changes in gut microbes.23 The difference in results may be attributed to the 21-day administration of metformin, which may not have been sufficient to significantly reduce FPG. Therefore, long-term administration may be required to achieve a more pronounced effect on FPG reduction. Administration of metformin facilitates the restoration of the diversity of the gut microbiota of DM rats. Zhang et al,2019 proved that metformin changed specific probiotics to increase the hypoglycaemic effect of T2DM rats.24
Red bean treatment group showed a decrease in FPG. The decrease of FPG in rats was related to the nutritional content of fiber and protein as well as flavonoids, phenolics, alkaloids, and melatonin. Flavonoids can directly decrease blood glucose levels, and alkaloids can regenerate cells. However, factors such as phytic acid content in red bean may affect the effectiveness in lowering blood glucose. Phytic acid can form complexes with minerals and reduce the absorption of essential nutrients as antinutritional compounds.25 Therefore, the presence of phytic acid may inhibit the effectiveness and limit antidiabetic ability. Melatonin also has anti-inflammatory, antioxidant, and antihyperglycemic effects. Experimental research in diabetic rats given 5 mg/kg body weight of melatonin orally for 8 weeks showed increased GSH and decreased blood glucose and HbA1c.26 Unfortunately, melatonin levels were not quantitated in this study.
The effect of decreasing FPG in group P1, which received a dose of 0.35 g/200 gBW, was less pronounced compared to the K+ group with metformin administration. However, the addition of a 0.70 g/200 gBW dose of red beans for 21 days resulted in a more significant decrease in FPG. This is different from other research that gave a single dose of 200 mg/kg red bean extract to DM rats, without affecting blood glucose levels in OGTT. There was a decrease in sugar levels when given repeatedly for 28 days.11 This repeated administration has effect on the rate of glucose use by muscle cells in rats. Red bean extract can also increase GLUT4 content in skeletal muscle cells of non-DM and DM rats. A 50% increase in GLUT content was found in the DM group given red bean extract compared to the group given water.11 Therefore, P. vulgaris extract has activated GLUT4 transcription and has affected the intracellular translocation.
GLUT4 expression in the untreated KN and the diabetes group (K-) was 81.36 and 36.19, respectively. This is different from previous research where the healthy and DM rat groups were 1 and 6.1, respectively.27 In our result,GLUT 4 expression was examined on day 21 or 3 weeks of treatment, while Yousef et al., 2024 conducted a measurement on week 4. GLUT is a transporter that responsible in the exchange of molecules through the cell membrane. This transporter is found in abundance in skeletal muscle and adipose tissue. After eating, high blood glucose levels stimulate insulin secretion from pancreatic beta cells. Insulin stimulates the translocation of GLUT4 to the cell membrane, which increases glucose uptake in cells and achieves homeostasis. In T2DM, insulin is unable to increase the translocation of GLUT4 to the membrane, causing the accumulation in the membrane compartment. GLUT4 expression is reduced in skeletal muscle, causing cells to have difficulty processing glucose in T2DM.
Research has been conducted to find natural ingredients targeting GLUT4 activation, such as red beans suspected to contain melatonin. Red bean extract could increase GLUT4 gene expression. Administration of extract to P2 can increase GLUT4 gene expression by 71.75 times. This is consistent with research conducted by providing an aqueous solution of lyophilized red bean extract of 200 mg/kg for 28 days. Therefore, P. vulgaris extract can increase the content of GLUT4 in skeletal muscle cells of control and DM rats measured by the ELISA. There was no significant difference between the GLUT4 content of the muscles of rats from the control group given water and the diabetic rat group after treatment with extract.11 The melatonin content may help the function of GLUT 4 during treatment. Administration of melatonin as much as 10 mg/kg BW/day, intraperitoneally at 6-7 PM for 6 weeks either alone or in combination with GABA (an inhibitory neurotransmitter and produced by pancreatic β cells and has an effect on DM) can increase the expression of GLUT 4 protein. The study also showed an increase in the expression of IR1β and AKT which had an impact on reducing fasting blood sugar in mice28. In rat skeletal muscle cells, melatonin activates the insulin receptor substrate 1 (IRS1)–PI3K–PKCζ pathway and stimulates glucose transport.7,29 In human PAZ6 brown adipocytes, melatonin administration inhibits cAMP and cGMP production, reduces GLUT4 expression and glucose uptake.30 The transporter included in the major facilitator superfamily (MFS) is GLUT 1 commonly found in erythrocytes and endothelial cells.31 The function of melatonin in passing through the plasma membrane is mediated by GLUT 1 encoded by the SLC2A1 gene. Tursinawati et al., 2021 analyzed the presence of rs841853 SLC2A1 polymorphism in obese DM individuals. The results showed that the TT genotype was insignificantly common in non-obese DM patients compared to other genotypes. The individuals in the TG+TT genotype group had higher blood sugar, HBA1c, triglycerides, and microalbumin levels.32
The results show no significant correlation between GLUT 4 expression and FPG levels and reports a negative trend toward FPG levels. These results is in line with Jasmin et al. 2019 who examined GLUT 4 mRNA using the semi-q PCR. The results reported that GLUT4 gene expression did not correlate with fasting glucose and HbA1c but significantly correlated with insulin and insulin resistance.33 In theory, DM patients or animals injected with STZ have high blood sugar levels with low GLUT 4 expression. There may be partial regeneration of pancreatic beta cells, allowing insulin production so that GLUT4 expression still remains.33,34 Therefore, further research is needed to conduct histopathological examinations in determining the presence or absence of the pancreatic beta cell regeneration process. This research has limitations, including the treatment duration to obtain an improvement in effect of red bean administration on the experimental animals. The modification of longer treatment duration can be performed in the future. Moreover, this research has not identified the quantitative levels of melatonin to ensure its hypoglycaemic effect. Further research is needed to measure melatonin levels in red beans to determine the exact effect of melatonin on diabetes.
Conclusion
In conclusion, red beans contained melatonin has α-Glucosidase inhibitory activity although the power still below than acarbose. The administration of melatonin contained in red bean extract could significantly reduce FPG levels and increase GLUT 4 gene expression. The administration of extract at P2 also increased GLUT 4 gene expression by 71.75 times. Administration of melatonin contained in red bean extract at a dose of 0.7 g/200 gBW can increase GLUT 4 gene expression than in lower dose. Melatonin contained in melatonin may contribute to improved glucose metabolism, but the quantitative levels of melatonin needs to be measured through further research.
Acknowledgement
The authors are grateful to the funder, the Directorate of Research, Technology, and Community Service, Ministry of Education, Culture, Research, and Technology under the Regular Fundamental Research scheme, with Number.108/E5/PG.02.00.PL/2024.
Funding Sources
This research received external funding from the Directorate of Research, Technology, and Community Service, Ministry of Education, Culture, Research, and Technology under the Regular Fundamental Research scheme in 2024, No. 0667/E5/AL.04/2024.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This experimental study has received ethical approval from the Research Ethics Committee of the Faculty of Medicine, Muhammadiyah University of Semarang, No.022/EC/KEPK-FK/UNIMUS/2024.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other Sources
Not applicable
Author contributions
- Yanuarita Tursinawati : formulating research ideas, collecting data, validating results and data analysis, compiling research articles,
- Lukman Faishal Fatharani: collecting data, analyzing the results data,
- Amin Samiasih: validating data analysis and supervising article preparation,
- Ardita Nadia Zahirah; Shafa Noor Aulia; Ahmad Reihanzaki : collecting data.
References
- Meng X, Li Y, Li S, Zhou Y, Gan RY, Xu DP, et al. Dietary sources and bioactivities of melatonin. Nutrients. 2017;9(4):1–64.
CrossRef - P.J. L, Sanchez N. AS, J.M. G, A. CV. Melatonin and Glucose Metabolism: Clinical Relevance. Curr Pharm Des. 2014;20(30):4841–53.
CrossRef - Câmara CRS, Urrea CA, Schlegel V. Pinto beans (Phaseolus vulgaris l.) as a functional food: Implications on human health. Agriculture. 2013;3(1):90–111.
CrossRef - Ocho-Anin atchibri AL, Brou KD, Kouakou TH, Kouadio YJ, Gnakri D. Screening for antidiabetic activity and phytochemical constituents of common bean (phaseolus vulgaris L.) seeds. J Med Plant Res. 2010;4(17):1757–61.
- Zuo Z, Pang W, Sun W, Lu B, Zou L, Zhang D, et al. Metallothionein–Kidney Bean Polyphenol Complexes Showed Antidiabetic Activity in Type 2 Diabetic Rats by Improving Insulin Resistance and Regulating Gut Microbiota. Foods. 2023;12(16):1–22.
CrossRef - Sangsopha J, Johns NP, Johns J, Moongngarm A. Dietary sources of melatonin and benefits from production of high melatonin pasteurized milk. J Food Sci Technol [Internet]. 2020;57(6):2026–37. Available from: https://doi.org/10.1007/s13197-020-04236-5
CrossRef - Karamitri A, Jockers R. Melatonin in type 2 diabetes mellitus and obesity. Nat Rev Endocrinol [Internet]. 2019;15(2):105–25. Available from: http://dx.doi.org/10.1038/s41574-018-0130-1
CrossRef - Rinalda C, Batubara L, Budiastuti A, Nugroho TE. Melatonin intervention effect on blood sugar levels. Diponegoro Med J. 2021;10(4):234–40.
CrossRef - Esteves J V., Yonamine CY, Pinto-Junior DC, Gerlinger-Romero F, Enguita FJ, MacHado UF. Diabetes modulates microRNAs 29b-3p, 29c-3p, 199a-5p and 532-3p expression in muscle: Possible role in GLUT4 and HK2 repression. Front Endocrinol. 2018;9(SEP):1–12.
CrossRef - Tursinawati Y, Kartikadewi A, Yuniastuti A, Susanti R. Overexpression of miR-29b in Plasma of Javanese Type 2 Diabetes Mellitus Patients in Semarang, Indonesia. Turk J Endocrinol Metab. 2022;26(4):208–12.
CrossRef - Halenova T, Raksha N, Kravchenko O, Vovk T, Yurchenko A, Vareniuk I, et al. Hypoglycemic activity of Phaseolus vulgaris (L.) aqueous extract in type 1 diabetic rats. Curr Issues Pharm Med Sci. 2019;32(4):210–8.
CrossRef - Kwan SH, Mejia EG de. Peptides From Adzuki Bean and Soybean Improved Insulin-AKT Signaling-Related Pathways in Healthy and Insulin-Resistant States in Human Liver Cells. Mol Nutr Food Res. 2025;69:e70285.
CrossRef - Xiang L, Wu Q, Osada H, Yoshida M, Pan W, Qi J. Peanut skin extract ameliorates the symptoms of type 2 diabetes mellitus in mice by alleviating inflammation and maintaining gut microbiota homeostasis. Aging (Albany NY). 2020;12(14):13991–4018.
CrossRef - Aguilera Y, Herrera T, Liébana R, Rebollo-Hernanz M, Sanchez-Puelles C, Martín-Cabrejas MA. Impact of Melatonin Enrichment during Germination of Legumes on Bioactive Compounds and Antioxidant Activity. J Agric Food Chem. 2015;63(36):7967–74.
CrossRef - Saleh HM, Hassan AA, Mansour EH, Fahmy HA, El-Bedawey AEFA. Melatonin, phenolics content and antioxidant activity of germinated selected legumes and their fractions. J Saudi Soc Agric Sci [Internet]. 2019;18(3):294–301. Available from: https://doi.org/10.1016/j.jssas.2017.09.001
CrossRef - Tursinawati Y, Rosidi A, Hajar N, Diatri D, Kurniati ID, Rahmatullah DE, et al. Effect of Different Melatonin-Rich Extract of Emprit Ginger (Zingiber Officinale Var. Amarum) Doses on Biochemical Parameters in Streptozotocin-Induced Diabetic Rats. Scr Med. 2024;55(4):399–408.
CrossRef - Tursinawati Y, Kartikadewi A, Kartikadewi A, Yuniastuti A, Yuniastuti A, Susanti R. Melatonin, A Promising Therapeutic for Diabetes Mellitus and Its Complications: A Narrative Review. J Biomed Transl res. 2021;7(3):138–45.
CrossRef - Abdulrahman BO, Bala M, Bello OM. Evaluation of in vitro antioxidant and antidiabetic potential of extracts from Phaseolus vulgaris L. seeds (Black turtle beans). FFS. 2021;1(9):23–38.
CrossRef - Sasongko H, Nurrochmad A, Rohman A, Nugroho AE. Characteristic of Streptozotocin-Nicotinamide-Induced Inflammation in A Rat Model of Diabetes-Associated Renal Injury. Open Access Maced J Med Sci. 2022;10(T8):16–22.
CrossRef - Yusuf S, Nuriyanto AR, Nadia N, Muntadhar, Muzakkir Y SN. Effects of SGLT2 inhibitor administration on blood glucose level and body weight in type 1 diabetes rat model. Narra J. 2021;1(July):1–11.
- L H. Metformin and systemic metabolism. Trends Pharmacol Sci. 2020;41(11):868–81.
CrossRef - Hideaki Kaneto , Tomohiko Kimura , Atsushi Obata , Masashi Shimoda KK. Multifaceted mechanisms of action of metformin which have been unraveled one after another in the long history. Int J Mol Sci. 2021;22(5):2596.
CrossRef - Zhou X, Zhou J, Ban Q, Zhang M, Ban B. Effects of metformin on the glucose regulation, lipid levels and gut microbiota in high-fat diet with streptozotocin induced type 2 diabetes mellitus rats. Endocrine [Internet]. 2024;163–72. Available from: http://dx.doi.org/10.1007/s12020-024-03843-y
CrossRef - M. Zhang, R. Feng, M. Yang, C. Qian, Z. Wang, W. Liu JM. Effects of metformin, acarbose, and sitagliptin monotherapy on gut microbiota in Zucker diabetic fatty rats. BMJ Open Diabetes Res Care. 2019;20(7):e000717.
CrossRef - Rahmawati S, Aji AS, Saloko S, Aprilia V, Djidin RTS, Sailendra NV, et al. Analysis of Phytic Acid and Tannin Content of Local Food-Based Analogue Rice as an Alternative Functional Food. Amerta Nutr. 2024;8(3):344–9.
CrossRef - Khorsand M. Melatonin Reduces Cataract Formation and Aldose Reductase Activity in Lenses of Streptozotocin-induced Diabetic Rat. IJMS. 2016;41(4):305–13.
- Yousef RS, Thabet SR, Ahmed NS, Osman AS. Ameliorative effect of watercress (Nasturtium officinale) aqueous extract on gene expression of Glut4 and Ampk in diabetic rats. SVU Int J Med Sci. 2024;7(1):686–97.
CrossRef - Nishant Parmar, Roma Patel, Nirali Rathwa, Mitesh Dwivedi, Rasheedunnisa Begum AR. Melatonin and GABA monotherapies are as efficacious as combination therapy in managing T1D and T2D: In-vivo studies on experimental diabetic models. bioRxiv. 2025;1–31.
CrossRef - Ha E, Yim SV, Chung JH, Yoon KS, Kang I, Yong HC et al. Melatonin stimulates glucose transport via insulin receptor substrate-1/phosphatidylinositol 3-kinase pathway in C2C12 murine skeletal muscle cells. J Pineal Res. 2006;41(1):67–72.
CrossRef - Brydon L, Petit L, Delagrange P, Strosberg AD, Jockers R. Functional expression of MT2 (Mel1b) melatonin receptors in human PAZ6 adipocytes. Endocrinology. 2001;142(10):4264–71.
CrossRef - Wang T, Wang J, Hu X, Huang X, Chen GX. Current understanding of glucose transporter 4 expression and functional mechanisms. World J Biol Chem. 2020;11(3):76–98.
CrossRef - Tursinawati Y, Kartikadewi A, Yuniastuti A, Susanti R. Association of rs10830963 MTNR1B and rs841853 SLC2A1 Polymorphism with Obesity on Type 2 Diabetes Patients: An Overview of Melatonin Receptor and Transporter. Indones Biomed J. 2021;13(2):155–62.
CrossRef - Jasmin S, Ali M, Ferdous M, Arslan MI, Biswas SK. Glucose transporter 4 gene expression in peripheral blood leukocytes in type 2 diabetes mellitus. Bangabandhu Sheikh Mujib Med Univ j. 2019;12(1):19–24.
CrossRef - Proudy Laksmana NP, Lestari RV, Sahidu MG, Hunaifi I, Harahap HS, Padauleng N, et al. Pengaruh kadar gula darah puasa terhadap ekspresi Glucose transporter type-4 (GLUT4) neuron hipokampus pada tikus model diabetes. Intisari Sains Medis. 2022;13(2):321–7.
CrossRef







