Manuscript accepted on :02-02-2026
Published online on: 18-03-2026
Plagiarism Check: Yes
Reviewed by: Dr. Exbrayat Jean-Marie
Second Review by: Dr. Sarraa Dhiaa Kasim
Final Approval by: Dr. Patorn Piromchai
Mohammad Shah Amran1* , Nasrin Akhter1
, Nasrin Akhter1 , Fareha Anan Shristy2
, Fareha Anan Shristy2 , Sumaiya Rahman1
, Sumaiya Rahman1 , Runa Akter1
, Runa Akter1 and Mohammad Josim Uddin1
and Mohammad Josim Uddin1
1Department of Pharmacy, School of Pharmacy and Public Health, Independent University, Bangladesh, Bashundhara, Dhaka, Bangladesh.
2Department of Pharmacy, University of Dhaka, Dhaka, Bangladesh.
Corresponding Author E-mail:amranmsspph@iub.edu.bd
DOI : https://dx.doi.org/10.13005/bpj/3334
Abstract
β-blockers retain their authoritative position in the therapy of all phases of ischemic heart disease. β-blockade is regarded as the established therapy for effort, mixed effort, unstable, and rest angina. β-blockers have been shown to significantly lower long-term mortality following a myocardial infarction and provide substantial therapeutic benefit for individuals experiencing congestive heart failure. They are also indicated for hypertension. When administered appropriately, β-blockers are considered to have a favorable safety profile. Atenolol, a second-generation and predominantly β1-selective agent targeting cardiac receptors, represents a key member of this drug class. Originally patented in 1969 and entering clinical use in 1975, atenolol is included on the World Health Organization’s List of Essential Medicines and is widely available in generic form. Although developed earlier, it received approval from the United States Food and Drug Administration (FDA) in August 1981 for the treatment of hypertension and coronary artery disease. Its continued relevance is reflected in prescribing patterns; in 2023, atenolol ranked as the 75th most dispensed medication in the United States, with over nine million prescriptions. This review summarizes the pharmacological characteristics of the atenolol, evaluates its position in the local pharmaceutical market, and discusses its clinical importance, as well as associated adverse effects, contraindications, and recommended dosing regimens.
Keywords
Arrhythmia; Atenolol; β-adrenergic receptor; Cardiac disorder; Coronary artery disease; Hypertension
Download this article as:| Copy the following to cite this article: Amran M. S, Akhter S, Shristy F. A, Rahman S, Akter R, Uddin M. J. Pharmacology of Atenolol: A β1-selective Adrenoceptor Inhibitor. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Amran M. S, Akhter S, Shristy F. A, Rahman S, Akter R, Uddin M. J. Pharmacology of Atenolol: A β1-selective Adrenoceptor Inhibitor. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/4btV1RV |
Introduction
Chemistry and physical properties of Atenolol
Atenolol is a selective β1-adrenergic receptor blocker. The chemical formula is C14H22N2O3. Its structure (Figure 1) consists of a benzeneacetamide core linked to a 2-hydroxy-3-(isopropylamino)propoxy group. Key chemical properties include a molecular weight of 266.3 g/mol and a melting point between 146-148∘C. Its chemical name is [4-(2-hydroxy-3-[(1-methylethyl) amino] propoxy)benzeneacetamide]. Its IUPAC name is 2-[4-[2-hydroxy-3-(propan-2-ylamino)propoxy]phenyl]acetamide1-2.
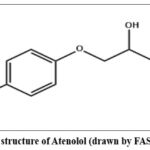 |
Figure 1: Chemical structure of Atenolol (drawn by FAS using ChemDraw). |
Atenolol is characterized by a highly polar and hydrophilic nature, as reflected by its low octanol–water partition coefficient (log P ≈ 0.23), which denotes minimal lipophilicity. At physiological temperature (37 °C), it exhibits an aqueous solubility of approximately 26.5 mg/mL. The compound dissolves readily in methanol and shows good solubility in solvents such as acetic acid and dimethyl sulfoxide (DMSO). In contrast, its solubility is limited in organic alcohols, being only sparingly soluble in 96% ethanol and slightly soluble in isopropanol. Atenolol demonstrates very poor solubility in acetone and dioxane and is essentially insoluble in solvents of lower polarity, including acetonitrile, ethyl acetate, and chloroform. Physically, it is obtained as a white to off-white crystalline powder. Overall, the solubility behavior of atenolol highlights its strong preference for polar solvents, with moderate solubility reported in ethanol (~750 g/L, TS), slight solubility in dichloromethane, and comparatively restricted solubility in water.1
History of β-blockers (Atenolol)
The history of atenolol is part of the broader development of β-blocker. The first β-blocker, dichloroisoprenaline, was discovered in 1958, but it had limitations. The Imperial Chemical Industries (ICI) developed propranolol, the first widely used β-blocker, in 1964.
Sir James Black2, a Scottish pharmacologist was the first to introduce β-blockers for the treatment of angina pectoris in 1958, a pioneering contribution that earned him the Nobel Prize. Following their initial success, β-blockers rapidly gained widespread clinical adoption and were subsequently evaluated for the management of myocardial infarction, cardiac arrhythmias, and hypertension throughout the 1960s. Research into their therapeutic potential continued over the following decades, with studies in the 1970s and 1980s exploring their role in treating of heart failure.
Following the success of propranolol, ICI researchers aimed to develop more selective β-blockers (specifically targeting the β1-receptors in the heart) with fewer side effects. Atenolol was synthesized as a second-generation, β1-selective antagonist by Alvogen Malta and received U.S. Food and Drug Administration (FDA) approval in September 1981 under the trade name Tenormin.3-5 It was developed by the pharmaceutical company Imperial Chemical Industries (ICI), based on the pioneering work of Sir James Black on β-blockers in the 1960s. Atenolol was patented in 1969 and approved for medical use in 1975 in the United Kingdom and subsequently in other parts of the globe. Atenolol is recognized by the World Health Organization as an essential medicine and is broadly accessible in generic formulations. In 2023, it ranked as the 75th most frequently prescribed medication in the United States.6-10
General Description of Atenolol
Atenolol is a member of the β-blocker class of medications, which comprises drugs that share similar mechanisms of action and are commonly used to manage related cardiovascular conditions. Specifically, atenolol is a cardioselective β1-adrenergic antagonist, targeting the heart primarily. It is among the most widely prescribed β-blockers globally, with over 40 million prescriptions issued annually in the United States alone. In patients, atenolol is utilized to control hypertension, relieve angina, manage arrhythmias, and treat myocardial infarction, while also serving a preventive role against migraines. Designed to cross the blood-brain barrier, atenolol’s cardioselectivity minimizes adverse effects in patients with bronchial asthma or diabetes mellitus. Its once-daily dosing regimen improves patient adherence. Beyond cardiovascular indications, atenolol is also utilized in the treatment of dysautonomia, anxiety, and hyperthyroidism.11-16
Aims and objectives of current work
This work aims to highlight the – (a) General pharmacology of β-blockers with special emphasis on atenolol, (b) Significance of atenolol and other β-blockers in health science, (c) Side effects, contraindications and dosage of atenolol and other β-blockers and (d) Analyze the market situations of atenolol and other β-blockers in Bangladesh.
Materials and Methods
PubMed, ResearchGate and Google Scholar and other search engines including Artificial intelligence tools, were searched to identify suitable literature to find relevant research works. The keywords “Atenolol”, “β-blocker”, “β-adrenergic receptor”, “Hypertension”, “Cardiac disorder”, “Arrhythmia,” “Coronary artery disease” etc. have been applied to find expected research articles and collect information on various aspects. Some of our related previous papers17-23 have also been consulted to enrich the whole text.
Findings and Discussion
General Pharmacology of β-blocker
Atenolol is a cardioselective β1-adrenergic receptor inhibitor, which is mainly located in the heart and kidneys. The drug decreases heart rate, reduces the strength of heart contractions, and lowers arterial pressure. It is a hydrophilic drug with low lipid solubility and undergoes minimal liver metabolism, being primarily excreted unchanged by the kidneys.
Three Generations and Classification of β-blockers
Three Generations
β-Adrenergic antagonists, or β-blockers, are agents that block the actions of catecholamines such as adrenaline and noradrenaline by competing for β-adrenergic receptor binding sites. The development of this class of drugs was pioneered by Sir James Black. Today, more than twenty β-blockers are commercially available for clinical use. Within the cardiovascular system, these agents produce negative inotropic and chronotropic effects, resulting in decreased cardiac output. Furthermore, blockade of β1-receptors in the juxtaglomerular cells diminishes renin release, thereby suppressing the renin-angiotensin-aldosterone system and contributing to a decrease in blood pressure.24-31 Although all β-blockers share a common mechanism of action, they differ in their specific pharmacological activities. Differences in adrenergic receptor subtype selectivity (α1A/B/D, α2A/B/C, β1/2/3) are central to β-blocker pharmacodynamics and inform the conventional three-generation classification. Nonselective β-blockers of the first generation inhibit both β₁ and β₂ receptors. Second-generation agents exhibit relative β1-selectivity, also referred to as cardioselectivity, although this selectivity is often dose-dependent. Third-generation β-blockers possess additional vasodilatory properties due to their broader effects on the cardiovascular system. For instance, some third-generation drugs block α1-adrenoreceptors and stimulate β3-receptors, which enhances nitric oxide synthase activity and promotes nitric oxide production. A clear understanding of these generational differences is essential for the appropriate clinical application of β-blockers.31 The classification of β-adrenergic blockers and the tissue-specific responses mediated by β-adrenergic receptors are presented in Tables 1 and 2, respectively.
Table 1: Three categories of β-adrenergic blockers.
| Generations | Mechanism of action | Examples | |
| 1 | First Generation | Non-selective, blocking both𝛽1 and β2 receptors | Propranolol, timolol, and nadolol. |
| 2 | Second Generation | Cardioselective, primarily blockingβ1-receptors | Atenolol, metoprolol, and bisoprolol. |
| 3 | Third Generation | Not only block 𝛽1-receptors but also have additional vasodilating properties, achieved through mechanisms like blocking α1-receptors or activating β3-receptors. | Carvedilol, labetalol, and nebivolol. |
Table 2: Tissue-specific responses mediated by β-adrenergic receptors.32
| Tissue/Receptors | β1 Response | β2 Response | β3 Response |
| Heart | Increases both rate (chronotropism) and force (inotropism) of contraction. | Also increases both rate (chronotropism) and force (inotropism) of contraction. | Causes a decrease in the force of contraction (negative inotropism). |
| Adipocytes (Fat Cells) | No direct effect listed. | No direct effect listed. | Induces Lipolysis (fat breakdown) and thermogenesis (heat production). |
| Vascular Smooth Muscle | Causes relaxation (vasodilation). | Causes relaxation (vasodilation). | Causes relaxation (vasodilation). |
| Lung | No direct effect listed. | Causes relaxation (bronchodilation). | Causes relaxation (bronchodilation). |
| Ileum and colon | No direct effect listed. | No direct effect listed. | Causes relaxation (decreased motility). |
| Kidney Juxtaglomerular Cells | Stimulates the secretion of renin. | No direct effect listed. | No direct effect listed. |
| Peripheral sympathetic nerves | Stimulates the release of norepinephrine. | Stimulates the release of norepinephrine. | No direct effect listed. |
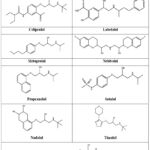 |
Figure 2: Chemical structures of available β-blockers except atenolol (drawn by FAS using ChemDraw). |
Classification of β-blockers
β-blockers are divided into four main categories based on the beta receptors they block: non-selective, β1-selective, combined α and β, and β2-selective (which are primarily used in research). They can also be further categorized by their generation, or by whether they have additional properties like intrinsic sympathomimetic activity (ISA) or vasodilating properties.33
By receptor selectivity
Non-selective β-blockers
Inhibit both β1 and β2 receptors. They are used for conditions like hypertension and angina. Examples include propranolol and timolol.
β1 selective (cardio selective) blockers
Primarily target beta-1 receptors in the cardiac tissues, which is beneficial for patients with respiratory issues like asthma, though their selectivity is dose-dependent. Examples include atenolol, metoprolol, and bisoprolol.
Combined alpha and beta blockers
Block both alpha and beta receptors to provide both vasodilation and reduced heart rate. They are used for conditions like heart failure and hypertension, and labetalol is atypical example often used in hypertensive emergencies and pregnancy.
β2 selective blockers
Primarily used in experimental settings, as they are less common in clinical practice.
Other classifications
β-blockers possessing intrinsic sympathomimetic activity (ISA)
These act as both an agonist and antagonist on β receptors. Examples include acebutolol and pindolol.34-38
Pharmacokinetics (ADME)
Absorption: Oral administration of atenolol results in gastrointestinal absorption of approximately 50–60%, with peak blood levels occurring within 2 to 4 hours.
Distribution: As a hydrophilic compound, atenolol exhibits low plasma protein binding (6–16%) and because it crosses the blood-brain barrier minimally, the likelihood of central nervous system effects is lower than with lipophilic β-blockers.
Metabolism: The drug undergoes minimal hepatic metabolism, with only around 5% being metabolized by the liver.
Elimination: Atenolol is primarily excreted unchanged via the kidneys (approximately 85–100%) following intravenous administration and roughly 50% after oral dosing. The drug’s half-life is about 6–7 hours in people with normal renal function, but it may be markedly prolonged in those with kidney dysfunction, requiring dose modifications.39-40
Pharmacodynamics (Mechanism of action)
Adrenergic receptors (AR)
Adrenergic receptors are specialized protein structures located on the surface of cells that interact with catecholamines or sympathomimetic agonists to trigger specific physiological responses. In contrast, adrenergic antagonists (blockers) bind to these receptors and inhibit the actions normally induced by endogenous or exogenous agonists, thereby producing their pharmacological effects. Adrenergic receptors are broadly classified into two main categories: α-receptors, which generally mediate excitatory responses, and β-receptors, which typically produce inhibitory effects. Each category contains multiple subtypes, including α1A, α1B, α1D; α2A, α2B, α2C; and β1, β2, and β3. All adrenergic receptors are members of the G-protein-coupled receptor (GPCR) family, functioning through their interaction with heterotrimeric G-proteins to transduce extracellular signals into cellular responses.41 In 1906, Dale 42 was the first to introduce the concept of a receptor in association with the sympathetic nervous system (SNS). In 1948, Ahlquist classified adrenergic receptors into two main types α and β based on their distinct physiological actions, with α-receptors primarily mediating vasoconstriction and β-receptors promoting vasodilation. Approximately twenty years later, Richardson and colleagues demonstrated that stimulation of β-receptors in cardiac tissue produces positive chronotropic and inotropic effects, enhancing both heart rate and contractile force.43-44 In the same year, Lands et al. subdivided β-adrenergic receptors into β1 (cardiac effects) and β2 (bronchodilator and vasodilator effects).45 Later, Yarden et al. demonstrated that β-adrenergic receptors consisted of seven transmembrane domains, and Dixon et al. described that those were G-protein-coupled receptors.46-47 In 1989, Emorine et al. observed the existence of a third isoform of β-adrenergic receptors (β3), which also belongs to the family of G-protein-coupled receptors.48
β-adrenergic receptor (β-AR)
The sympathetic division of the autonomic nervous system is responsible for orchestrating multiple homeostatic processes, including cardiovascular regulation, and initiating the fight-or-flight response. Catecholamines act as the principal signaling molecules, with epinephrine secreted by the adrenal medulla and norepinephrine released from sympathetic nerve endings as well as in minor amounts by the adrenal medulla. These catecholamines circulate systemically and interact with adrenergic receptors in target tissues, including the heart, where they elicit positive inotropic and chronotropic effects. However, in pathological conditions such as heart failure, chronically elevated catecholamine levels can exert harmful effects on cardiac tissue, contributing to maladaptive hypertrophy and cardiomyocyte apoptosis.49-51 The general structural architecture of seven-transmembrane receptors is illustrated in Figure 3.
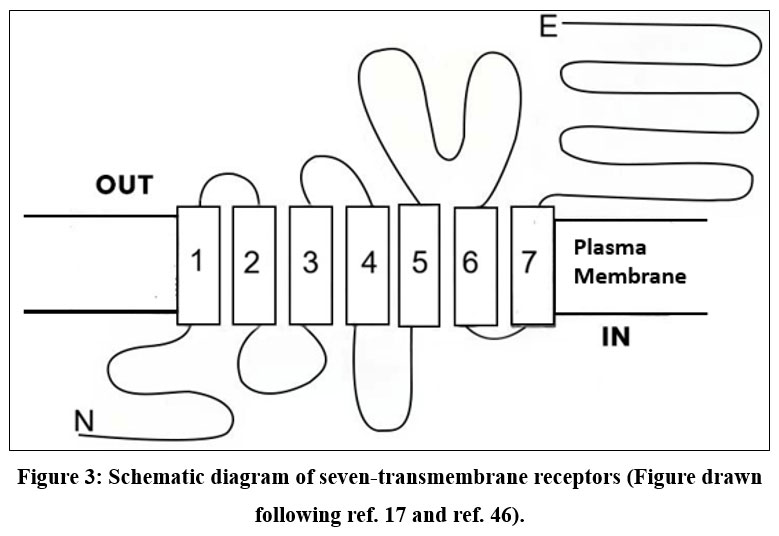 |
Figure 3: Seven-transmembrane receptors (Figure drawn by SR). |
β-adrenergic receptors belong to the large family of G protein-coupled receptors. and are essential mediators of cardiovascular regulation, influencing both cardiac function and vascular processes. To date, three β-AR subtypes β1, β2, and β3 have been well characterized, while the existence of a potential fourth subtype, β4-AR, remains a subject of scientific debate.52-53 Intracellular signaling of each β-AR is summarized in Figure 4.
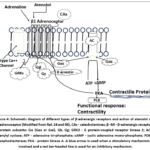 |
Figure 4: Schematic diagram of different types of β-adrenergic receptors and action of atenolol on β1 adrenoceptor (Redrawn from Ref. 24). |
The three receptor subtypes exhibit distinct ligand-binding affinities, enabling differential activation of each subtype.54-55 In healthy cardiac tissue, β1-ARs outnumber β2-ARs by about fourfold, and β3-ARs are only minimally present.55-56 Recent studies have confirmed that β1-adrenergic receptors (β1-ARs) are universally expressed in all cardiomyocytes. In contrast, β2-ARs and β3-ARs are generally absent in most myocytes, being detected in only approximately 5% of these cells, but they are highly expressed in non-myocyte populations, particularly endothelial cells. Conversely, β1-ARs are present at relatively low levels in non-myocyte cells.57 β-adrenergic receptors (β-ARs), like other G protein-coupled receptors (GPCRs), are composed of seven transmembrane domains and are linked to an intracellular heterotrimeric G protein complex. Upon binding of an agonist, the receptor undergoes a conformational shift that facilitates the exchange of guanosine diphosphate (GDP) for guanosine triphosphate (GTP) on the Gα subunit, leading to the dissociation of the activated Gα and Gβγ subunits. The Gα subunit subsequently modulates downstream effector proteins, with the specific effectors engaged being determined by the β-AR subtype that has been activated.58 Although all β-adrenergic receptors (β-ARs) typically signal through the stimulatory G protein (Gαs), evidence indicates that β2-AR and β3-AR can also couple to inhibitory G proteins. This conclusion is supported by studies demonstrating that their signaling is sensitive to pertussis toxin, implicating the involvement of Gαi proteins.58-59 Activation of Gαs stimulates adenylyl cyclase to transform ATP into cAMP. The elevated cAMP binds to PKA’s regulatory subunits, freeing the catalytic subunits to perform their functions. PKA serves as a central mediator, phosphorylating multiple effector proteins and eliciting downstream physiological responses. Key targets of PKA phosphorylation include phospholamban, L-type calcium channels, contractile proteins, and the β-adrenergic receptor itself, among others. Mammalian tissues express seven isoforms of adenylyl cyclase, with isoforms 5 and 6 being the most abundant in cardiac tissue. AC activity is positively regulated by Gαs signaling and inhibited by Gαi signaling.60-61 cAMP signaling compartmentalization has become an accepted mechanism by which the molecule can target and modulate specific signaling routes (Figure 3, Figure 4, Figure 5, and Table 3).
Table 3: Important seven-transmembrane-spanning receptors with their major characteristics.
| Receptor | β1 | β2 | β3 | α1A/B/D/AT1/ET | α2 | M2 |
| Primary G protein | Gs | Gs/Gi | Gs/Gi | Gq/G11 | Gi | Gi |
| Tissue distribution | Heart | Heart, lung, vessels, kidney | Adipose, heart | Heart, vessels, smooth muscle | Coronary vessels, CNS, Pancreas, Platelets | Hear |
| Primary effector in heart tissue | AC, L-type Ca2+ channel | AC, L-type Ca2+ channel | AC | PLC-beta | AC | AC, K+channels |
| Signals | ↑cAMP/PKA | ↑cAMP/PKA, MAPK | ↑cAMP/PKA | ↑DAG/InsP3, PKC, MAPK | ↓cAMP/PKA | ↓cAMP/PKA, PI(3)K |
| Endogenousagonist | NA, A | NA, A | NA, A | NA, A, Angiotensin-II, Endothelin | NA, A | ACh |
| α, α-adrenergic receptor subtypes A, B, D; β, β-adrenergic receptor subtypes 1, 2, 3; AC, adenylyl cyclase; ACh, acetylcholine; A, adrenaline; AT1, angiotensin-II receptor subtype 1; cAMP; adenosine 3´,5´ monophosphate; DAG, diacylglycerol; ET, endothelin receptor; InsP3, inositol-1,4,5-trisphosphate; M2, muscarinic cholinergic receptor subtype 2; MAPK, mitogen-activated protein kinase; NA, noradrenaline; PKA, protein kinase A; PKC, protein kinase C; PLC- β, phospholipase C- β. 62 | ||||||
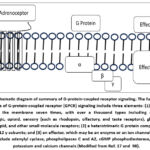 |
Figure 5: Summary of G-protein-coupled receptor signaling. The fundamental components of G-protein-coupled receptor (GPCR) signaling include three elements: |
In brief, β-adrenergic receptors are cell surface receptors in the sympathetic nervous system that are activated by adrenaline and noradrenaline, which render the “fight-or-flight” effect in response to internal or external factors. There are three main subtypes of β-adrenergic receptors. These are – β1, β2, and β3 (Figure 4). β1 receptors are present in the heart and kidneys. They increase heart rate and contractility, and release renin. β2 receptors are found in smooth muscle. They cause relaxation and are targeted for treating conditions such as asthma through bronchodilation. β3 receptors are in adipose tissue and the bladder muscle. They play a role in metabolism and relaxation.
Atenolol and β1-adrenergic receptor
Atenolol is a β1-adrenergic receptor blocking agent. Therefore, it acts selectively on the cardiac system and gives beneficial effects (Figure 5). Atenolol exerts a milder impact on the airways compared to other cardioselective β-blockers. Consequently, it may be cautiously used in patients with obstructive airway disease, provided they are concurrently receiving optimal doses of a β₂-agonist bronchodilator.
 |
Figure 6: The many signaling roles of seven-transmembrane-spanning receptors. (Figure drawn by SR). Agonist binding triggers dissociation of G proteins into Gα-GTP and Gβγ subunits, which activate downstream effectors. |
In patients with mild to moderate essential hypertension, atenolol effectively lowers both systolic and diastolic blood pressure by approximately 15%. Clinical studies indicate a relatively flat dose-response relationship, suggesting that a fixed dose may be adequate for most patients. Maximum antihypertensive effects are generally observed with doses of 100–200 mg, with most of the blood pressure reduction occurring within the first three days of therapy. Atenolol, like other β-blockers, reduces blood pressure and concurrently lowers heart rate (around 15–25%) and cardiac output (approximately 20%).
Acting on β₁-adrenergic receptors in the heart and vascular smooth muscle, atenolol mitigates sympathetic effects. Heart rate, cardiac output, and arterial pressures are reduced by this mechanism, with a slow decrease in peripheral vascular resistance occurring during sustained therapy. Beyond cardiovascular effects, atenolol influences endocrine and metabolic parameters, including suppression of renin release and decrease in circulating free fatty acids. Compared to non-selective β-blockers, atenolol has a smaller effect on extending insulin-induced hypoglycemia. Its impact on lipid metabolism is relatively mild compared to non-selective agents; While β-blockers typically exert little influence on LDL cholesterol, they can raise plasma levels of VLDL and triglycerides and decrease HDL cholesterol.
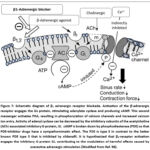 |
Figure 7: β1 adrenergic receptor blockade. Activation of the β-adrenergic receptor engages the Gs protein, stimulating adenylate cyclase and producing cAMP. This second messenger activates PKA, resulting in phosphorylation of calcium channels and increased calcium ion entry. |
Atenolol is a hydrophilic compound, resulting in relatively low penetration into brain tissue. Following oral administration, its absorption is often incomplete, peak blood levels are typically observed between 2 and 4 hours. The plasma half-life ranges from 6 to 8 hours under normal conditions, but it can be significantly prolonged in patients with renal impairment.63-64
Clinical uses
Labeled indications
Atenolol is a relatively safe drug. It is indicated for-65-66
Management of Hypertension
Atenolol is utilized both as monotherapy and alongside other antihypertensive medications to manage high blood pressure. However, they are not commonly recommended as the primary treatment option, as they do not provide the same cardioprotective benefits against coronary artery disease as other classes of antihypertensives.67
Angina pectoris
Angina pectoris, a key clinical manifestation of ischemic heart disease, arises primarily from myocardial ischemia. Atenolol is commonly employed in the management of angina pectoris, particularly when associated with coronary atherosclerosis.68
Acute MI
Early coronary reperfusion therapy, combined with evidence-based pharmacological interventions, is strongly recommended for patients experiencing acute myocardial infarction (AMI) to minimize the risk of subsequent cardiac events. In hospitalized patients, the prescription of β-blockers at discharge is included as a key indicator in clinical performance and quality-of-care measures and serves as one of the quality metrics for AMI management. Following AMI, medical therapy aimed at reducing myocardial oxygen demand while enhancing oxygen supply is critical for both immediate and long-term treatment. For hemodynamically stable individuals exhibiting a heart rate over 50 bpm and systolic blood pressure higher than 100 mmHg, β-blocker therapy is indicated to decrease cardiovascular mortality.69-72
Guideline recommendations
β-blockers lower myocardial oxygen demand by decreasing heart rate, blood pressure, and contractility. They are beneficial in stable heart failure with LVEF <40% and post-myocardial infarction. ACC/AHA guidelines recommend initiating oral β-blockers within 24 hours in STEMI or NSTE-ACS patients without heart failure, low cardiac output, risk of cardiogenic shock, or other contraindications. While the guidelines do not recommend a specific β-blocker for optimal management of NSTE-ACS, clinical judgment and experience guide the selection of therapy. Overall, evidence supports starting patients without contraindications on a low dose of oral β-blockers shortly after ACS diagnosis (<24 hours), with gradual dose escalation as tolerated based on heart rate and blood pressure73-76.
Off label uses of Atenolol
Beyond its approved indications, atenolol is utilized off-label for a variety of cardiovascular and non-cardiovascular conditions. Its off-label applications include:
Used in secondary prevention to prevent repeat heart attacks.
Management of ventricular arrhythmias, including inherited conditions.
Treatment of supraventricular tachycardia and atrial fibrillation to help control heart rate and rhythm.
Adjunct therapy in symptomatic thyrotoxicosis, often combined with antithyroid agents such as methimazole.
Prophylactic treatment of migraine headaches.
Management of heart failure in selected patients.
Use in alcohol withdrawal to mitigate autonomic symptoms and reduce cardiovascular stress73-77.
Dosage
Hypertension
Adults: Oral administration may begin at 25–50 mg once daily, with potential escalation to 100 mg/day based on efficacy.
Pediatric (off-label): Given by mouth at 0.5–1 mg/kg daily, not to surpass 2 mg/kg/day or 100 mg in total.
Geriatric: Initiate treatment at 25 mg/day orally and titrate upwards to a maximum of 100 mg/day as tolerated.
Angina Pectoris
Adults: Begin with 50 mg/day orally, escalating to 100 mg/day after seven days, while some patients may require up to 200 mg/day. Intravenously, 2.5 mg may be infused at 1 mg/min, repeated every five minutes up to a maximum of 10 mg.
Geriatric: Start with 25 mg/day orally; after one week, the dose may be raised to 100 mg/day, with higher requirements (up to 200 mg/day) in some patients.
Post-Myocardial Infarction (Secondary Prevention)
Adults and Geriatric: Oral administration of 100 mg daily, given either once or in divided 12-hour intervals, for 6–9 days post-myocardial infarction.
Alcohol Withdrawal Syndrome (off-label): Administered orally at a daily dose of 50 to 100 mg.
Supraventricular Arrhythmias (off-label, prevention): An oral dose of 50 mg per day can be initiated up to three days prior to surgery and maintained for seven days following the procedure, with the possibility of increasing the dose to 100 mg per day if needed.
Thyrotoxicosis (off-label): A total daily oral dose of 25–100 mg may be administered either as a single daily dose or divided into two doses given at 12-hour intervals.
Management of Adverse Events:
Excessive Bradycardia: Management may involve the administration of intravenous atropine sulfate in total doses ranging from 0.6 to 2.4 mg, given in divided increments of 600 micrograms.
Arrhythmias: Treatment may be achieved through administration of an intravenous infusion at a dose of 150 mg per kilogram of body weight delivered over a 20-minute period, with the option to repeat the infusion at 12-hour intervals if clinically indicated.78-79
Dosage forms
Atenolol is available for oral administration as tablets in doses of 25 mg, 50 mg, and 100 mg. It is additionally formulated for intravenous use in vials containing 5 mg of the drug dissolved in 10 mL of a citrate-buffered vehicle. The intravenous preparation is intended for administration by trained medical professionals in a controlled clinical setting only. Oral therapy typically begins with a 25 mg dose once or twice daily, with subsequent adjustments made according to the patient’s clinical response.79
Side effects
Atenolol is usually well tolerated in most patients, and its overall adverse effect profile is comparable to that of propranolol. However, fewer patients discontinue treatment with atenolol compared to propranolol (2.2% vs. 9.7%).79-81
Common Side Effects:
Mild side effects may occur, often resolving within a few days to weeks. Commonly reported effects include:
Cold extremities (hands and feet)
Fatigue or unexplained tiredness
Dizziness
Headache
Shortness of breath
Gastrointestinal disturbances, such as constipation or diarrhea
Leg pain
Reduced libido or impotence
Lowered blood pressure
If these symptoms are persistent or severe, consultation with a healthcare provider is recommended.79-81
Serious Side Effects
Though less frequent, atenolol may cause serious adverse reactions, including:
Cardiovascular effects: bradycardia, hypotension, conduction abnormalities, heart failure
Respiratory effects: bronchospasm; Caution is recommended when administering the drug to patients with obstructive airway disease, with concurrent use of a full-dose β2-agonist bronchodilator
Peripheral vascular effects: Aggravation of peripheral ischemic pain during exertion
Allergic reactions: Marked cutaneous redness, throat or tongue swelling, fever, difficulty breathing
Neuropsychiatric effects: depression, characterized by persistent sadness, anxiety, fatigue, or difficulty concentrating
Other: unusual weight gain associated with edema in the feet, ankles, or arms.79-81
Atenolol in Pregnancy, breastfeeding, and fertility
Atenolol is classified as a Pregnancy Category D medication by the FDA and Category C by the Therapeutic Goods Administration (TGA) in Australia, indicating that its use during pregnancy may pose risks to the developing fetus. The drug readily crosses the placenta, potentially exposing the fetus to adverse effects. Studies have reported that atenolol use during the second trimester can result in Infants with evidence of impaired fetal growth resulting in reduced birth weight for gestational age. Fetal exposure during the later stages of pregnancy may further increase the risk of complications in the newborn, including bradycardia, hypotension, and hypoglycemia, either immediately after birth or within several hours postpartum.82-85
Studies show a significantly higher rate of fetal growth restriction in mothers treated with atenolol compared to carvedilol (26% vs. 7%). Atenolol, a pregnancy class-D β-blocker, has been associated with reduced birth weight and placental weight when compared with placebo in pregnant women with hypertension. Evidence also indicates that exposure to atenolol early in pregnancy, particularly before 20 weeks, results in smaller infants and is linked to all stillbirths reported in the study. Therefore, this drug should be avoided during pregnancy, especially in the first trimester.86-88
Atenolol is further discouraged due to its extensive excretion into breast milk, making it unsuitable for lactating mothers. Although sometimes used in women with pre-existing hypertension, its risks, such as fetal growth restriction, reduced uteroplacental blood flow, and low birth weight, outweigh potential benefits. Early pregnancy exposure carries the greatest danger, contributing to short- and medium-term complications in infants. Hence, atenolol is generally not recommended during pregnancy or breastfeeding. Clinical studies have shown that atenolol is excreted into breast milk, with concentrations ranging from 1.5 to 6.8 times those observed in maternal plasma.89-90
Precautions
Cardiac Failure
Sympathetic nervous system activity plays a critical role in sustaining hemodynamic stability in patients with congestive heart failure (CHF); therefore, β-blocker therapy may further reduce myocardial contractility and potentially worsen cardiac function. In the setting of acute myocardial infarction (AMI), the presence of heart failure that cannot be rapidly controlled with 80 mg of intravenous furosemide, or an equivalent therapeutic intervention, constitutes a contraindication to β-blocker administration.91
In patients without a history of cardiac failure, prolonged myocardial depression caused by β-blockers may occasionally induce heart failure. At the first indication of worsening cardiac function, appropriate treatment should be initiated following current clinical guidelines, with close monitoring of patient response. If heart failure persists despite adequate therapy, atenolol should be discontinued.91
Cessation of Atenolol Therapy
Patients with coronary artery disease should be cautioned against abrupt discontinuation of atenolol. Sudden withdrawal has been associated with severe angina exacerbation, myocardial infarction, and ventricular arrhythmias, which may occur even without preceding angina. Gradual tapering is recommended, and patients should limit physical activity during this period. Given the prevalence of undiagnosed coronary artery disease, abrupt discontinuation of atenolol even when used solely for hypertension should generally be avoided.91
Concomitant Use with Calcium Channel Blockers
Concomitant use of β-blockers with non-dihydropyridine calcium channel blockers, including verapamil or diltiazem, can precipitate bradycardia, atrioventricular block, and elevations in left ventricular end-diastolic pressure. Individuals with underlying conduction defects or impaired left ventricular function are especially vulnerable to these adverse effects.91
Bronchospastic Disease
β-blockers are generally contraindicated in patients with bronchospastic conditions. Owing to its relative β₁-selectivity, atenolol may be used with caution in patients who have an inadequate response to, or are unable to tolerate, alternative antihypertensive agents. Therapy should start at the lowest effective dose (typically 50 mg), with a β2-agonist bronchodilator readily available. If dose escalation is necessary, splitting the dose may help maintain lower peak plasma concentrations and reduce risk.91
Major Surgery
Chronic β-blocker therapy should generally be continued before major surgical procedures. Nevertheless, attenuation of the heart’s responsiveness to reflex adrenergic stimulation may heighten the risks associated with general anesthesia and operative interventions.91
Diabetes and Hypoglycemia
Atenolol should be administered with caution in individuals with diabetes mellitus. While β-blockers can mask hypoglycemia-induced tachycardia, other signs such as sweating and dizziness usually remain detectable. At recommended doses, atenolol does not exacerbate insulin-induced hypoglycemia and, unlike non-selective β-blockers, does not delay the normalization of blood glucose.92
Thyrotoxicosis
β-blocker therapy may obscure observable physical findings indicative of thyroid hormone overproduction, including tachycardia. Abrupt withdrawal of atenolol in patients with thyrotoxicosis could precipitate a thyroid storm. Careful monitoring is essential when discontinuing therapy in patients suspected of developing thyrotoxicosis.92
Untreated Pheochromocytoma
Atenolol is contraindicated in patients with untreated pheochromocytoma.
Drug interactions
Atenolol and Levocetirizine
Levocetirizine is a third-generation, non-sedative antihistamine derived from cetirizine. It acts by blocking histamine receptors, thereby preventing histamine from exerting its effects, although it does not inhibit histamine release from mast cells. Levocetirizine can form a charge-complex with atenolol, and for this reason, the two drugs should not be administered together. Further in vivo and large-scale studies are recommended, as this interaction may reduce or even eliminate the therapeutic effectiveness of both medications.93
Atenolol and alcohol
Atenolol and alcohol may interact to produce an enhanced blood pressure-lowering effect. This can lead to symptoms such as fainting, headache, dizziness, lightheadedness, or changes in heart rate. These effects are observed at the start of therapy, after a dosage increase, or when treatment is resumed following a break. Patients should inform their physician if these symptoms persist beyond a few days or become bothersome. Caution is advised when driving, operating machinery, or rising quickly from a sitting or lying position until the effects of the medication are known. It is also essential to inform your doctor of all other medications, including over-the-counter drugs, and to avoid discontinuing any medication without prior medical consultation.94
Atenolol and multivitamins with minerals
Concurrent use of atenolol and multivitamins containing minerals may reduce the effectiveness of atenolol. To minimize this interaction, it is recommended to separate the administration of these products by at least two hours. If both medications are prescribed together, your doctor may need to adjust the dose of atenolol. You should always inform your healthcare provider about all medications, supplements, and herbal products you are taking, and do not discontinue any therapy without medical guidance.95
Atenolol and food
Atenolol should be taken either with meals or on empty stomach; however, it is important to be consistent in how you take it each day. Limit the intake of large quantities of orange juice, as it may interfere with the drug’s absorption and reduce its therapeutic effectiveness.96
High Cholesterol (Hyperlipoproteinemia, Hypertriglyceridemia, Sitosterolemia)
β-adrenergic receptor antagonists, commonly referred to as β-blockers, can influence blood lipid levels. Certain β-blockers have been associated with elevated levels of VLDL, LDL cholesterol, and triglycerides, along with reduced HDL cholesterol. Individuals who already have high cholesterol may need more frequent monitoring while on β-blocker therapy, and their lipid-lowering treatments may need to be modified based on these changes.96
Table 4: Interaction of Atenolol with other drugs.
| Category | Interacting Drugs |
| Minor Interaction | Diazepam |
| Moderate Interaction | Adrenaline, Diltiazem (HCl), Digoxin, Metformin (HCl), Thioridazine (HCl), Nifedipine, Disopyramide, Indomethacin, Verapamil (HCl) |
| Major Interaction | Prazosin, Alfuzosin, Clonidine (HCl) |
| Severity (High) | Amlodipine (Besylate), Indacaterol, Aldesleukin, Naphazoline, Flurbiprofen, Lacidipine, Amiodarone (HCl) |
Atenolol in Bangladeshi Medicine Market
β-blockers constitute a heterogeneous class of medications, each exhibiting distinct pharmacodynamic and pharmacokinetic characteristics. They have demonstrated long-term advantages in reducing mortality and managing cardiovascular disease. In Bangladesh, several pharmaceutical companies produce a variety of β-blocker formulations.97 The market situations of β-blockers in Bangladesh are presented below –
Local Manufacturers
In Bangladesh, local pharmaceutical industries manufacture all nine (9) generics of β-blockers as shown in Figure 2. It was observed that atenolol is manufacture by 23 manufacturers, bisoprolol by 24 manufacturers, carvedilol by 20 manufacturers, celiprolol and sotalol by one manufacturer, labetalol by 2 manufacturers, metoprolol by 12 manufacturers, Nebivolol by 4 manufacturers and propranolol by 5 manufacturers (Figure 8).
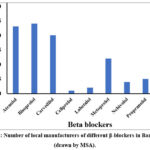 |
Figure 8: Number of local manufacturers of different β-blockers in Bangladesh (drawn by MSA). |
Price Variations in local Market
Figure 9 shows the price variation of beta blockers in the local market produced by different manufacturers.
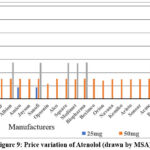 |
Figure 9: Price variation of Atenolol (drawn by MSA). |
Conclusion
In summary, atenolol is a highly significant medication used in the management of various medical conditions. It is classified as a β-blocker and is among the most frequently prescribed drugs within this category worldwide. Atenolol is a cardio selective β-blocker, specifically targeting β1-adrenergic receptors. Introduced in 1976, atenolol received approval from the U.S. Food and Drug Administration in August 1981 for the treatment of both hypertension and coronary artery disease. Despite its widespread use, atenolol has relatively low bioavailability, which presents a notable limitation in its therapeutic effectiveness.
Acknowledgement
The authors express their gratitude to authors of articles and books cited in this work. The authors also express their gratitude to authors the articles and books from where the concepts of some Figures were borrowed.
Funding Source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author contributions:
- Mohammad Shah Amran: Conceptualized the review topic and planned the main skeleton of the manuscript. Guided the overall work and reviewed the manuscript several times. Drew Figures 8 to 9. Made the final corrections before submission.
- Nasrin Akhter: Accumulated the related literatures and selected relevant articles from different sources. Helped to organize the reviewed articles and revised the manuscript.
- Fareha Anan Shristy: Read and summarized research papers. Wrote the original draft based on the main skeleton of the manuscript. Drew the chemical structures of Beta blockers (Figure 1 and 2). Perform the plagiarism check and edited the text for clarity.
- Sumaiya Rahman: Assisted in writing the original draft. Compared the findings from different articles. Assisted in structuring the review sections and revised the manuscript. Drew Figures 3 to7.
- Runa Akter: Verified reference articles. Summarized key findings and contributed to manuscript writing and editing.
- Mohammad Josim Uddin: Confirmed important information. Contributed to interpretation and structuring of the findings and manuscript review.
References
- Atenolol. PubChem Compound Database. Published 2025. Accessed December 15, 2025. https://pubchem.ncbi.nlm.nih.gov
- Nobel Prize winning scientist dies. Archived News Report. 2012.
- Frängsmyr T. Sir James W. Black: the Nobel Prize in Physiology or Medicine. Les Prix Nobel. 1989.
- Black JW. James Black tells his life story. Web of Stories. 2025. Accessed December 15, 2026. https://www.webofstories.com/play/james.black/1
- Black JW, Crowther AF, Shanks RG, Smith LH, Dornhorst AC. A new adrenergic beta-receptor antagonist. Lancet. 1964;283(7342):1080–1081.
CrossRef - com. Atenolol monograph for professionals. AHFS Drug Information. 2019. Accessed December 15, 2026. https://www.drugs.com/monograph/atenolol.html
- Fischer J, Ganellin CR. Analogue-Based Drug Discovery. John Wiley & Sons; 2006:461.
CrossRef - World Health Organization. World Health Organization Model List of Essential Medicines: 22nd List. Geneva, Switzerland: WHO Press; 2021.
- Top 300 drugs of 2023. ClinCalc Drug Database. Published 2023. Accessed December 15, 2026. https://clincalc.com/DrugStats/Top300Drugs.aspx
- Atenolol drug usage statistics, United States (2013–2023). ClinCalc Drug Database. Published 2025. Accessed December 15, 2026. https://clincalc.com/DrugStats/Drugs/Atenolol.aspx
- Gunda RK, Kumar JNS, Brahma CK, Satyanarayana V, Prashant KN. Design, formulation and evaluation of atenolol gastro-retentive floating tablets. Asian J Pharm. 2015;9(4):34–42.
- Earle RR, Ayalasomayajula LU, Venkatesh P, Naidu PG, Sagar SV, Vani BS. Formulation and evaluation of atenolol orodispersible tablets by co-processed super-disintegrants. Int J Adv Pharm. 2016;5(2):46–51.
- Heel RC, Brogden RN, Speight TM, Avery GS. Atenolol: a review of its pharmacological properties and therapeutic efficacy in angina pectoris and hypertension. Drugs. 1979;17(6):425–460.
CrossRef - University of Illinois Chicago Drug Information Group. Atenolol oral tablet. Drug Information Review. 2018;12.
- Bangalore S, Messerli FH, Kostis JB. Cardiovascular protection using beta-blockers. J Am Coll Cardiol. 2007; 50:563–572.
CrossRef - Chrysant SG, Chrysant GS. Current and future study of beta-blockers in the treatment of hypertension. Clin Cardiol. 2008;31(6):249–252.
CrossRef - Amran MS, Homma N, Hashimoto K. Pharmacology of KB-R7943: a Na⁺–Ca²⁺ exchange inhibitor. Cardiovasc Drug Rev. 2003;21(4):255–276.
CrossRef - Homma N, Amran MS, Nagasawa Y, Hashimoto K. Involvement of Na⁺–Ca²⁺ exchange system in cardiac triggered activity. J Pharmacol Sci. 2006.
CrossRef - Amran MS, Hashimoto K, Homma N. Effects of sodium-calcium exchange inhibitors on aconitine-induced arrhythmias. J Pharmacol Exp Ther. 2004;310(1):83–89.
CrossRef - Amran MS, Bahar NB, Akash S. Physiology and pathology of the cardiovascular system. IntechOpen; 2023.
- Amran MS, Homma N, Hashimoto K. Effects of SEA0400 on ouabain-induced arrhythmias in guinea pigs. J Sci Res. 2012;4(1):213–225.
CrossRef - Dixon RA, Kobilka BK, Strader DJ, et al. Cloning of the gene and cDNA for mammalian beta-adrenergic receptor homology with rhodopsin. Nature. 1986;321(6065):75–79.
CrossRef - Emorine LJ, Marullo S, Briend-Sutren MM, et al. Molecular characterization of the human beta-3 adrenergic receptor. Science. 1989;245(4922):1118–1121.
CrossRef - Johnson JA, Davis JO, Gotshall RW, et al. Evidence for an intrarenal beta receptor in control of renin release. Am J Physiol. 1976;230(2):410–418.
CrossRef - Kaumann AJ, Hall JA, Murray KJ, Wells FC, Brown MJ. Effects of adrenaline and noradrenaline on the human heart: role of β1- and β2-adrenoceptors. Eur Heart J. 1989;10(Suppl B):29–37.
CrossRef - Berg T. Beta1-blockers lower norepinephrine release by inhibiting presynaptic β1-adrenoceptors. Front Neurol. 2014; 5:51.
CrossRef - Liggett SB. Molecular and genetic basis of beta-2 adrenergic receptor function. J Allergy Clin Immunol. 1999;104(2 Pt 2): S42–S46.
CrossRef - Roberts SJ, Papaioannou M, Evans BA, Summers RJ. Beta-adrenoceptor-mediated smooth muscle relaxation in rat ileum. Br J Pharmacol. 1999;127(4):949–961.
CrossRef - Peterson L, Ismond KP, Chapman E, Flood P. Therapeutic use of β2-adrenergic receptor agonists in neuroprotection and Parkinson’s disease. J Immunol Res. 2014;2014: 1037–1080.
CrossRef - Skeberdis VA. Structure and function of beta-3 adrenergic receptors. Medicina (Kaunas). 2004;40(5):407–413.
- Amran MS, Homma N, Hashimoto K. Experimental arrhythmia model and computer simulation action potential study on Andersen syndrome. J Pharmacol Sci. 2003:154P–154P.
- Yesmin F, Sultana A, Saha A, et al. Genetic basis of hypertension: an overview. Avicenna J Pharm Res. 2021;2(2):e.
CrossRef - do Vale GT, Ceron CS, Gonzaga NA, Simplicio JA, Padovan JC. Three generations of β-blockers: history, class differences and clinical applicability. Curr Hypertens Rev. 2019;15(1):22–31.
CrossRef - Chrysant SG, Chrysant GS. Antihypertensive and cardioprotective effects of three generations of beta-blockers. Hosp Pract. 2022;50(3):196–202.
CrossRef - Reiter MJ. Cardiovascular drug class specificity: β-blockers. Prog Cardiovasc Dis. 2004;47(1):11–33.
CrossRef - Pedersen ME, Cockcroft JR. Latest generation of beta-blockers: new pharmacologic properties. Curr Hypertens Rep. 2006;8(4):279–286.
CrossRef - Lopatinschi V. Three generations of β-blockers: class differences and clinical applicability. MedEspera Congress Proceedings. 2024.
- Gammone MA, Efthymakis K, D’Orazio N. Effect of third-generation beta blockers on weight loss. J Nutr Metab. 2021; 5767306.
CrossRef - Buck ML, Wiest D, Gillette PC, Trippel D, Krull J, O’Neal W. Pharmacokinetics and pharmacodynamics of atenolol in children. Clin Pharmacol Ther. 1989;46(6):629–633.
CrossRef - Wan SH, Koda RT, Maronde RF. Pharmacokinetics, pharmacology of atenolol and effect of renal disease. Br J Clin Pharmacol. 1979;7(6):569–574
CrossRef - Westfall TC. Sympathomimetic drugs and adrenergic receptor antagonists. Encyclopedia of Neuroscience. 2009:685–695.
CrossRef - Dale HH. On some physiological actions of ergot. J Physiol. 1906;34(3):163–206.
CrossRef - Ahlquist RP. A study of the adrenotropic receptors. Am J Physiol. 1948;153(3):586–600.
CrossRef - Richardson DW, Kontos HA, Raper AJ, Patterson JL. Modification of circulatory responses to acute hypoxia by beta-adrenergic blockade. J Clin Invest. 1967;46(1):77–87.
CrossRef - Lands AM, Arnold A, McAuliff JP, Luduena FP, Brown TGJ. Differentiation of receptor systems activated by sympathomimetic amines. Nature. 1967;214(5088):597–598.
CrossRef - Yarden Y, Rodriguez H, Wong SK, et al. Avian beta-adrenergic receptor: structure and membrane topology. Proc Natl Acad Sci U S A. 1986;83(18):6795–6799.
CrossRef - Dixon RA, Kobilka BK, Strader DJ, et al. Cloning of mammalian beta-adrenergic receptor gene. Nature. 1986;321(6065):75–79.
CrossRef - Emorine LJ, Marullo S, Briend-Sutren MM, et al. Human beta-3 adrenergic receptor characterization. Science. 1989;245(4922):1118–1121.
CrossRef - Day JL, Metcalf J, Simpson CN. Adrenergic mechanisms in control of plasma lipid concentrations. BMJ. 1982; 284:1145–1148.
CrossRef - Chauhan A. A review on atenolol. World J Pharm Pharm Sci. 2020;9(3):902–914.
- de Lucia C, Eguchi A, Koch WJ. New insights in cardiac β-adrenergic signaling during heart failure and aging. Front Pharmacol. 2018; 9:904.
CrossRef - Lymperopoulos A, Rengo G, Koch WJ. Adrenergic nervous system in heart failure: pathophysiology and therapy. Circ Res. 2013; 113:739–753.
CrossRef - de Lucia C, Femminella GD, Gambino G, et al. Adrenal adrenoceptors in heart failure. Front Physiol. 2014; 5:246.
CrossRef - Gauthier C, Tavernier G, Charpentier F, Langin D, Le Marec H. Functional beta-3 adrenoceptor in the human heart. J Clin Invest. 1996; 98:556–562.
CrossRef - Zhu W, Woo AY, Zhang Y, Cao CM, Xiao RP. Beta-adrenergic receptor subtype signaling in the heart: from bench to the bedside. Curr Top Membr. 2011; 67:191–204.
CrossRef - Bristow MR, Ginsburg R, Umans V, et al. Beta-1 and beta-2 adrenergic receptor subpopulations in nonfailing and failing human ventricular myocardium. Circ Res. 1986; 59:297–309.
CrossRef - Lohse MJ, Engelhardt S, Eschenhagen T. Role of beta-adrenergic signaling in heart failure. Circ Res. 2003; 93:896–906.
CrossRef - Moniotte S, Kobzik L, Feron O, Trochu JN, Gauthier C, Balligand JL. Upregulation of beta-3 adrenoceptors and altered contractile response in failing myocardium. Circulation. 2001; 103:1649–1655.
CrossRef - Rockman HA, Koch WJ, Lefkowitz RJ. Seven transmembrane-spanning receptors and heart function. Nature. 2002; 415:206–212.
CrossRef - Myagmar BE, Flynn JM, Cowley PM, et al. Adrenergic receptors in individual ventricular myocytes. Circ Res. 2017; 120:1103–1115.
CrossRef - Kohout TA, Takaoka H, McDonald PH, et al. Augmentation of cardiac contractility mediated by beta-3 adrenergic receptors. Circulation. 2001; 104:2485–2491.
CrossRef - Post SR, Hammond HK, Insel PA. Beta-adrenergic receptors and receptor signaling in heart failure. Annu Rev Pharmacol Toxicol. 1999; 39:343–360.
CrossRef - Chen J, Levin LR, Buck J. Role of soluble adenylyl cyclase in the heart. Am J Physiol Heart Circ Physiol. 2012;302:H538–H543.
CrossRef - Hoffman BB, Lefkowitz RJ. Adrenergic receptor antagonists. In: Goodman LS, Gilman A, eds. The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill; 1996:199–248.
- Chen CH, Ting CT, Lin SJ, et al. Different effects of fosinopril and atenolol on wave reflections in hypertensive patients. Hypertension. 1995;25(5):1034–1041.
CrossRef - Aursnes I, Osnes JB, Tvete IF, Gåsemyr J, Natvig B. Does atenolol differ from other β-adrenergic blockers? BMC Clin Pharmacol. 2007;7(1):4.
CrossRef - com. Atenolol drug profile. Drugs.com. Published 2019. Accessed January 15, 2026. https://www.drugs.com/atenolol.html
- Mancia G, Kjeldsen SE, Kreutz R, et al. Individualized beta-blocker treatment for high blood pressure. Hypertension. 2022;79(6):1153–1166.
CrossRef - De Caterina AR, Leone AM. Why beta-blockers should not be used as first choice in uncomplicated hypertension. Am J Cardiol. 2010;105(10):1433–1438.
CrossRef - Zafar MS, Khan MN, Rawat A, et al. Therapeutic use of beta-blockers in cardiovascular diseases. J Pharm Technol Clin Pharmacol. 2023;30(17):857–870.
- Diaconu CC, Marcu DR, Bratu OG, et al. Beta-blockers in cardiovascular therapy: a review. J Mind Med Sci. 2019;6(2):216–223.
CrossRef - Joo SJ. Beta-blocker therapy in acute myocardial infarction: not all patients need it. Acute Crit Care. 2023;38(3):251–260.
CrossRef - Jneid H, Addison D, Bhatt DL, et al. AHA/ACC clinical performance and quality measures for myocardial infarction. J Am Coll Cardiol. 2017; 70:2048–2049.
CrossRef - Rao SV, O’Donoghue ML, Ruel M, et al. ACC/AHA guideline for management of acute coronary syndromes. Circulation. 2025; 151:e771–e862.
- Pfeffer MA, Braunwald E, Moye LA, et al. Effect of captopril on mortality after myocardial infarction. N Engl J Med. 1992; 327:669–677.
CrossRef - Ambrosioni E, Borghi C, Magnani B. Effect of zofenopril after anterior myocardial infarction. N Engl J Med. 1995; 332:80–85.
CrossRef - Køber L, Torp-Pedersen C, Carlsen JE, et al. Trandolapril in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med. 1995; 333:1670–1676.
CrossRef - Methusala J, Madhuvani, Vyshnavi D. Antihypertensive atenolol. J Emerg Technol Innov Res. 2024;11(12): a67–a82.
- Atenolol (Tenormin) drug information. RxList. Published 2019. Accessed December 15, 2026. https://www.rxlist.com/tenormin-drug.htm
- Reiter MJ. Cardiovascular drug class: beta-blockers. Prog Cardiovasc Dis. 2004;47(1):11–33.
CrossRef - McGuiness M, Frye RA, Deng JS. Atenolol-induced lupus erythematosus. J Am Acad Dermatol. 1997;37(2):298–299.
CrossRef - National Health Service. Atenolol: pregnancy, breastfeeding and fertility. NHS. Published 2025. Accessed December 15, 2026. https://www.nhs.uk/medicines/atenolol/pregnancy-breastfeeding-and-fertility
- com. Atenolol use during pregnancy. Drugs.com. Published 2025. Accessed December 15, 2026. https://www.drugs.com/pregnancy/atenolol.html
- Cruickshank DJ, Campbell DM. Atenolol in essential hypertension during pregnancy. BMJ. 1990; 301:1103.
CrossRef - Lydakis C, Lip GY, Beevers M, Beevers DG. Atenolol and fetal growth in hypertensive pregnancies. Am J Hypertens. 1999; 12:541–547.
CrossRef - Kaye AB, Bhakta A, Moseley AD, et al. Review of cardiovascular drugs in pregnancy. J Womens Health. 2019; 28:686–697.
CrossRef - Shah M, Mulley V, Jethava FV, et al. Treatment of hypertension during pregnancy and lactation. Int J Pharm Sci Rev Res. 2022; 72:101–109.
CrossRef - Thorley KJ, McAinsh J, Cruickshank JM. Atenolol in pregnancy-induced hypertension. Br J Clin Pharmacol. 1981; 12:725–730.
CrossRef - Butters L, Kennedy S, Rubin PC. Atenolol in essential hypertension during pregnancy. Br Med J. 1990; 301:587–589.
CrossRef - Lip GY, Beevers M, Churchill D, Shaffer LM, Beevers DG. Atenolol and fetal growth in hypertensive pregnancies. Am J Hypertens. 1999; 12:541–547.
CrossRef - Gordon NF, Scott CB, Duncan JJ. Effects of atenolol versus enalapril on cardiovascular fitness and serum lipids. Am J Cardiol. 1997; 79:1065–1069.
CrossRef - Mehboob S, Mughal MA, Aftab K, et al. Drug–drug interaction of levocetirizine with atenolol. J Pharm Pharmacol. 2017; 5:118–124.
CrossRef - Thomas BA, Regan TJ. Interactions between alcohol and cardiovascular medications. Alcohol Res Health. 1990; 14:333.
- Kupari M, Heikkilä J, Tolppanen EM, et al. Acute effects of alcohol and beta-blockade on ventricular function. Eur Heart J. 1983; 4:463–471.
CrossRef - Melander A, Stenberg P, Liedholm H, et al. Food-induced reduction of atenolol bioavailability. Eur J Clin Pharmacol. 1979; 16:327–330.
CrossRef - Wolinsky H. Effects of beta-adrenergic blockers on blood lipid levels. Clin Cardiol. 1987; 10:561–566.
CrossRef - Directorate General of Drug Administration. Bangladesh National Formulary. 4th ed. Dhaka, Bangladesh: Ministry of Health and Family Welfare; 2015.







