Manuscript accepted on :10-02-2026
Published online on: 05-03-2026
Plagiarism Check: Yes
Reviewed by: Dr. Zena Sideeq tawfeek
Second Review by: Dr. Mohamad R. Abdullah
Final Approval by: Dr. Prabhishek Singh
Nurali Ergashev1 , Boburbek Yuldoshev1*
, Boburbek Yuldoshev1* , Esokhon Komilov1
, Esokhon Komilov1 , Mavluda Kenjayeva1
, Mavluda Kenjayeva1 , Mukhlisa Ikromova2
, Mukhlisa Ikromova2 , Yulduz Rakhmatillayeva2
, Yulduz Rakhmatillayeva2 , Hulkar Hazratova2
, Hulkar Hazratova2 , Komila Eshbakova3
, Komila Eshbakova3 , Doniyor Siddikov³
, Doniyor Siddikov³ , Bakhrom Komilov4
, Bakhrom Komilov4 , Rustam Makhmudov5
, Rustam Makhmudov5 and Muzaffar Asrarov1
and Muzaffar Asrarov1 .
.
1Department of Molecular Biophysics, Institute of Biophysics and Biochemistryat the National University of Uzbekistan, Tashkent, Republic of Uzbekistan.
2Faculty of Chemistry-Biology, Karshi State University, Karshi, Republic of Uzbekistan.
3Department of Сhemistry of Terpenoids and Phenolic Compounds, S. Yu. Yunusov Institute of the Chemistry of Plant Substances Academy of Sciences of the Republic of Uzbekistan.
4Faculty of Exact and Natural Sciences, Namangan State Pedagogical Institute, Namangan, Republic of Uzbekistan.
5Department of proteins and peptides, Institute of Bioorganic Chemistry, Academy of Sciences of the Republic of Uzbekistan, Tashkent, Republic of Uzbekistan.
Corresponding author’s e-mail: yuldoshevboburbek10@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3393
Abstract
The inner mitochondrial membrane contains various ion channels and protein complexes that participate in cellular bioenergetics by synthesizing ATP. In addition, these structures are involved in cellular signaling through the generation of reactive oxygen species, thereby contributing to the regulation of physiological processes and the development of pathological conditions. Thus, mitochondria play a crucial role in the functioning of cells and tissues. Therefore, in in vitro studies, we investigated the effects of various flavonoids on the mPTP of rat liver mitochondria, as well as on the activities of succinate–ferricyanide reductase (succinate dehydrogenase) and NADH–ferricyanide reductase (NADH dehydrogenase). The results demonstrated that flavonoids exert an inhibitory effect on the mPTP of rat liver mitochondria, forming the following order based on their half-maximal inhibitory concentrations: apigenin > quercetin > isorhamnetin > oroxylin A > pulicarin > chrysoeriol > kaempferol > kaempferol-7-O-rhamnoside > hyperoside. Moreover, pre-incubation of liver mitochondria with 50 µM NADH for 10 minutes enhanced the inhibitory effect of flavonoids on the mPTP. Under these conditions, apigenin, oroxylin A, quercetin, kaempferol, chrysoeriol, hyperoside, and pulicarin exhibited strong inhibitory effects, whereas the remaining flavonoids showed weaker inhibition. In addition, at concentrations of 10 and 50 µM, the flavonoids hyperoside, kaempferol, kaempferol-7-O-rhamnoside, isorhamnetin, quercetin, afzelin, and pulicarin increased succinate–ferricyanide reductase activity, while the other flavonoids exhibited very weak or no effect. At the same time, a strong inhibitory effect of quercetin, isorhamnetin, kaempferol-7-O-rhamnoside, hyperoside, and kaempferol on NADH–ferricyanide reductase activity was observed, whereas the remaining studied flavonoids demonstrated only weak inhibitory effects. Overall, the results indicate that the flavonoids investigated in vitro possess distinct and compound-specific properties.
Keywords
Afzelin; Apigenin; Chrysoeriol; Hyperoside; Isorhamnetin; Kaempferitrin; Kaempferol; Kaempferol-7-O-rhamnoside; Liver mitochondria; Mitochondrial permeability transition pore; NADH dehydrogenase; Oroxylin; Pulicarin; Quercetin; Succinate dehydrogenase
Download this article as:| Copy the following to cite this article: Ergashev N, Yuldoshev B, Komilov E, Kenjayeva M, Ikromova M, Rakhmatillayeva Y, Hazratova H, Eshbakova K, Siddikov D, Komilov B, Makhmudov R, Asrarov M. Effect of Flavonoids on PTP and Dehydrogenase Activity in Liver Mitochondria. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Ergashev N, Yuldoshev B, Komilov E, Kenjayeva M, Ikromova M, Rakhmatillayeva Y, Hazratova H, Eshbakova K, Siddikov D, Komilov B, Makhmudov R, Asrarov M. Muzaffar A. Effect of Flavonoids on PTP and Dehydrogenase Activity in Liver Mitochondria. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/46Fjwdk |
Introduction
Various types of ion channels are located in the inner membrane of mitochondria. Among them, the mitochondrial permeability transition pore (mPTP), situated between the outer and inner mitochondrial membranes, plays a role in both physiological and pathological processes.1 The characteristics of the mPTP were first described by R.A. Haworth and D.R. Hunter in 1979.2 In mitochondria, excessive accumulation of Ca²⁺ ions leads to a pathological increase in reactive oxygen species (ROS).3-5 Naturally, excessive accumulation of Ca²⁺ ions in mitochondria induces the opening of the mPTP. This allows the free passage of small molecules with a molecular weight of up to 1,500 Daltons into the mitochondrial matrix.6,7 As a result, this leads to depolarization of the mitochondrial membrane and a decrease in ATP production, ultimately triggering cell death.7 Moreover, the mPTP regulates the efflux of Ca²⁺ ions from mitochondria and participates in Ca²⁺ signaling during normal cellular function. The mPTP is a complex structure, and its composition has been reported to include cyclophilin D (CypD), adenine nucleotide translocase (ANT), hexokinase-2 (HK2), voltage-dependent anion channel (VDAC), benzodiazepine receptor (BzR), phosphate carrier (Pi), and glycogen synthase kinase-3β (GSK-3β).8 However, recent studies suggest that F₀F₁-ATP synthase may also be a component of the mPTP.6 However, debates regarding the structure of the mPTP are still ongoing. In mitochondria, CypD, considered a regulatory protein of the mPTP, promotes its opening, whereas cyclosporin A (CsA), a specific inhibitor of the mPTP, interacts with CypD to maintain the pore in a closed state.9,10
Ca²⁺ ion homeostasis in mitochondria is considered one of the key factors regulating oxidative metabolism and cell death.3 MPTP-related cell death occurs as a result of the pore transitioning to an open state.4 The redox couple NAD⁺ (β-nicotinamide adenine dinucleotide)/NADH (reduced β-nicotinamide adenine dinucleotide) also participates in the regulation of Ca²⁺ ion homeostasis in mitochondria. A decrease in the NAD⁺/NADH ratio is considered a condition associated with mitochondrial dysfunction.11 In diabetic cardiomyopathy, a decrease in the NAD⁺/NADH ratio and an increase in the acetyl-CoA/CoA ratio have been reported. NAD⁺ serves as a co-substrate for deacetylases and sirtuins, playing a crucial role in redox signaling. Acetyl-CoA acetylates lysine residues in proteins, leading to the disruption of mitochondrial integrity.12 Pyridine nucleotides NADH and NADPH play a crucial role in mitochondrial energy production. The intracellular NAD⁺/NADH ratio regulates the rate of ATP synthesis by controlling NADH-dependent dehydrogenases and modulates post-translational protein modifications through the activation of NAD⁺-dependent enzymes.13 Furthermore, the intracellular NAD⁺/NADH ratio has been shown to maintain redox balance and antioxidant activity. It has been demonstrated that the NAD⁺/NADH ratio in the cytosol and mitochondria responds differently to acute metabolic disturbances.14 It has been shown that inhibition of soluble adenylyl cyclase (sAC) leads to an increase in the extracellular lactate/pyruvate ratio and the cytosolic NADH/NAD⁺ ratio. Additionally, sAC inhibition has been found to specifically suppress complex I of the mitochondrial respiratory chain. This effect supports ATP production via glycolysis and oxidative phosphorylation, thereby maintaining cellular energetic homeostasis.15 In chronic obstructive pulmonary inflammation in rats, administration of NADH has been shown to provide protection against disease development and to stabilize the antioxidant defense system of the lungs.16
Mitochondrial complex I (NADH:ubiquinone oxidoreductase, NADH dehydrogenase) is the largest multimeric enzyme complex of the mitochondrial respiratory chain, responsible for electron transport and for establishing a proton gradient across the inner mitochondrial membrane to drive ATP synthesis.17,18 Mitochondrial complex I exists in a supercomplex with complexes III and IV of the respiratory chain and is considered a major source of deleterious ROS.18 It is well established that mitochondrial complex I serves as a key entry point for electrons into oxidative metabolism within the respiratory chain. Consequently, various mutations in nuclear and mitochondrial genomes can lead to NADH dehydrogenase dysfunction, which contributes to the development of diverse pathologies.19,20 Specifically, alterations or the absence of the NUIM subunit (NADH dehydrogenase [ubiquinone] iron-sulfur protein 8) of mitochondrial complex I lead to a reduction in enzyme activity and an increase in ROS production.21
Furthermore, regarding complex II of the respiratory chain – succinate dehydrogenase (SDH), this enzyme is unique in that it participates both in electron transport within the respiratory chain and in the tricarboxylic acid (TCA) cycle.22,23 Recent findings indicate that SDH functions as a tumor suppressor in carcinogenesis, whereas succinate is regarded as an oncometabolite.23 Dysfunction of the SDH enzyme has been shown to contribute to the development of both cancer and neurodegenerative diseases.24 During ischemia, the accumulation of succinate occurs as a result of the reverse activity of the SDH enzyme. Additionally, this process is influenced by the breakdown of purine nucleotides and the partial reverse operation of the malate/aspartate cycle, which correlates with the amount of fumarate. Upon reperfusion, the accumulated succinate is rapidly oxidized by SDH, leading to the overproduction of ROS through reverse electron transport at mitochondrial complex I.25 In in vivo studies, 30 minutes of renal ischemia-reperfusion has been shown to induce mitochondrial dysfunction, and prolonging the ischemic period exacerbates this process. Following ischemia-reperfusion, the accumulation of succinate in the cytosol leads to an increase in H₂O₂ production, which is dependent on mitochondrial complex II. Changes in LDH activity during ischemia-reperfusion indicate the development of cell necrosis.26 During cardiac ischemia/reperfusion, reverse electron transport (RET)-dependent ROS production at mitochondrial complex I, driven by succinate, has been proposed to induce the opening of the mPTP, thereby contributing to cardiac injury. It has been demonstrated that in rabbit heart mitochondria, the opening of the mPTP, induced by Ca²⁺ or alamethicin, leads to static ROS generation at complex II. Simultaneously, ischemia inhibits mitochondrial complexes II and III, resulting in an accumulation of succinate and fumarate within the mitochondrial matrix and promoting their efflux. Upon reperfusion, the activation of static ROS production facilitates the transition of the mPTP to the open state. Notably, this process has been reported to occur following ROS generation via reverse electron transport in the mitochondrial respiratory chain.27 ROS generated in mitochondria play a critical role in cellular signaling. However, pathological conditions such as ischemia-reperfusion injury disrupt ROS homeostasis, leading to cell death.28
Thus, it has been emphasized that, in the study of various pathological processes developing in the organism, mitochondrial respiratory chain complexes I, II, and IV, ATP-sensitive potassium channel activity (mitoKATP), adenine nucleotide translocase (ANT), and the mPTP may serve as pharmacological targets for various drugs and biologically active compounds.29 Therefore, in the present study, the effects of flavonoids on Ca2+–induced mitochondrial swelling (mPTP opening), as well as on the activities of succinate–ferricyanide reductase (succinate dehydrogenase) and NADH–ferricyanide reductase (NADH dehydrogenase), were investigated.
Materials and Methods
Chemicals
Kaempferol, kaempferol-7-O-rhamnoside, kaempferitrin and afzelin, belonging to the group of flavonoids, were isolated from Geranium rotundifolium.30 The purity of these compounds ranged between 86–90%. Other flavonoids and all reagents which are used in the experiments purchased by Sigma Aldrich company.
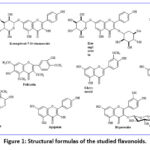 |
Figure 1: Structural formulas of the studied flavonoids. |
Isolation of liver mitochondria.
Liver mitochondria were isolated by differential centrifugation.31,32 The isolation medium consisted of 250 mM sucrose, 10 mM Tris-HCl, and 1 mM EDTA (pH 7.4). After decapitation, the rat’s abdominal cavity was opened, and the liver was excised and placed in a beaker containing ice-cold isolation medium. The liver mass was measured, then mechanically homogenized. The tissue was further homogenized in a Teflon homogenizer at a 1:6 g/mL ratio with the isolation medium. The homogenate was subjected to the first centrifugation at 1500 rotation/min (400 × g) for 7 min at 0-2°C using an RS-6MC centrifuge with an angled rotor. This step removed large tissue fragments and cell debris. The resulting supernatant was centrifuged again at 6,000 rpm (5000 × g) for 15 min at 0-2°C. The mitochondria, which had settled at the bottom of the tube, were separated from the supernatant. Any remaining liquid and lipid droplets on the tube walls were removed using filter paper. The purified mitochondria were resuspended in isolation medium without EDTA at a 10:1 g/mL dilution to prepare a mitochondrial suspension, which was stored in a special ice-cooled container for experiments. Protein concentration in mitochondria was determined using the Biuret method.33
Determination of mPTP.
The state of the Ca2+-dependent CsA-sensitive mPTP was assessed spectrophotometrically at 540 nm by monitoring changes in mitochondrial swelling kinetics.34 The incubation medium contained 200 mM sucrose, 20 μM EGTA, 20 mM Tris, 20 mM HEPES, 1 mM KH2PO4, 5 mM succinate, and 2 μM rotenone (pH 7.4). Mitochondrial protein concentration was 0.3-0.4 mg/mL, and the temperature was maintained at 26°C.
Measurement of succinate-ferricyanide-reductase (succinate dehydrogenase) activity.
Succinate ferricyanide-reductase activity in rat liver mitochondria was studied using a spectrophotometric method based on the change in optical density at 420 nm.35 The enzyme activity was determined by measuring the reduction of K3[Fe(CN)6] over a 5 min period at 37°C. The composition of the incubation medium used for determining enzyme activity included: 100 mM phosphate buffer, 1 mM sodium azide, 100 μM K3[Fe(CN)6], and 5 mM succinate (pH 7.8). The total volume of the incubation medium was 3 ml.
Measurement of NADH-ferricyanide-reductase (NADH dehydrogenase) activity.
The activity of the mitochondrial NADH-ferricyanide-reductase enzyme was determined spectrophotometrically by recording the change in optical density at 420 nm.36 This change occurred during the reduction of K3[Fe(CN)6] at 25 °C over 5 min. The incubation medium composition (mM) was as follows: tris-HCl – 100, NaN3 – 1, K3[Fe(CN)6] – 0.1–0.2, NADH – 0.27, with a total volume of 3 ml
Animal ethics.
The Animal Use Committee of our institution approved all experimental procedures and preoperative care guidelines. Animals were housed in standard vivarium conditions (humidity: 55-65%, temperature: 22 ± 2°C) with free access to drinking water and laboratory food. All manipulations with animals were carried out in accordance with the European Convention for the Protection of Animals Used for Scientific Purposes (1998) and the International Bioethical Guidelines of the Institute of Biophysics and Biochemistry of the National University of Uzbekistan (BEC/IBB-N44/2024/14-1). All operations were performed under sodium pentobarbital anesthesia, and all efforts were made to minimize suffering. Albino Wistar rats weighing 180-220g were used in the experiments.
Statistics.
Statistical analysis and graphical illustrations were performed using OriginPro 8.5 (USA). Data were obtained from 5 to 8 independent experiments (n = 5-8). A paired t-test was used for analyzing combined data, while an unpaired t-test was applied for comparisons between individual groups. Statistical significance was set at levels: p< 0.05.
Results
Initially, the study focused on the state of mPTP located in the inner membranes of rat liver mitochondria. In this setup, the response of mitochondria to Ca2+ ions (10 μM) in the absence of any biologically active substances was assessed, and no reaction was observed (data not shown). Continuing the experiments, when the mPTP inducer, Ca2+ ions (10 μM),27 was added to the incubation medium together with liver mitochondria, mitochondria exhibited high-amplitude swelling (Fig. 2).
 |
Figure 2: Effect of flavonoids on mPTP in rat liver. Ca2+-dependent high-amplitude mitochondrial swelling, and complete inhibition of mPTP with CsA (5 μM). And also, a study of the inhibitory effect of flvonoids and polyphenols with various concentrations |
To confirm that the object under investigation was indeed mPTP, CsA, 5 μM, a specific inhibitor of the mPTP modulator CypD,37 was applied, which prevented 95–100% of the high-amplitude Ca2+-induced mitochondrial swelling (Fig. 2). These results clearly indicate that the object studied in rat liver mitochondria is mPTP.
Subsequently, the effects of flavonoids isolated from plants on mPTP were investigated. The influence of different concentrations of the tested flavonoids on mPTP in rat liver mitochondria was studied in the presence of the mPTP inducer, Ca2+ ions (10 μM). Initially, the effects of kaempferol at concentrations of 50, 100, 150, and 200 μM on mPTP were examined (Fig. 2, A). At a concentration of 50 μM, kaempferol inhibited mPTP by 23.8±0.52% relative to the control. At 100 μM, it suppressed Ca2+-induced mitochondrial swelling by 37.6±1.49% compared to the control. At 150 μM, kaempferol inhibited the transition of mitochondria to a high-amplitude open conformational state by 52.5±1.32%, and at 200 μM, it inhibited the opening of mPTP by 69.8±3.5% relative to the control. The half-maximal inhibitory concentration (IC50) of kaempferol was determined to be 138.6±4.91 μM (Fig. 3, A).
 |
Figure 3: The half-maximal inhibitory concentration of flavonoids kaempferol (A), kaempferol-7-О-rhamnoside (B), Pulicarin (C), Isorhamnetin (D), Luteolin |
These findings indicate that, despite the high antioxidant potential of kaempferol,38 complete inhibition of mPTP was not achieved at the concentrations tested. The effects of kaempferol glycosides on mPTP opening induced by Ca2+ ions were also investigated. Kaempferol-7-O-rhamnoside, a glycoside of kaempferol, exhibited concentration-dependent inhibition of Ca2+-induced mPTP opening (Fig. 2, B). At a concentration of 50 μM, this flavonoid inhibited high-amplitude mitochondrial swelling induced by Ca2+ by 16.8±0.68% relative to the control. At concentrations of 100, 150, and 200 μM, kaempferol-7-O-rhamnoside suppressed mPTP transition to the open conformational state by 36.0±1.25%, 51.8±1.07%, and 59.2±3.66%, respectively. The IC50 of this kaempferol glycoside was determined to be 143.0±2.83 μM (Fig. 3, B). The effects of another kaempferol glycoside, kaempferitrin, on rat liver mitochondria mPTP were also studied at concentrations of 50, 100, 150, and 200 μM (Fig. 2, C). Initially, mitochondria were incubated with the mPTP inducer Ca2+ (10 μM) and the blocker CsA (5 μM). At 50 μM, kaempferitrin inhibited the transition of mPTP to the open conformational state by 9.45±0.17% in the presence of the inducer. At 100 μM, it suppressed Ca2+-induced high-amplitude mitochondrial swelling by 26.4±0.62% relative to the control. At 150 and 200 μM, kaempferitrin inhibited the transition to the open conformational state by 33.7±0.8% and 40.3±1.94%, respectively, in the presence of Ca2+ ions (10 μM). The next kaempferol glycoside studied was afzelin (kaempferol-3-rhamnoside, 50–200 μM), which exhibited a distinct effect on mPTP in liver mitochondria induced by Ca2+ ions (10 μM) (Fig. 2, D). Afzelin inhibited high-amplitude mitochondrial swelling induced by Ca2+ ions by only 31.6 ± 0.61% at the highest tested concentration of 200 μM. During the study, the effects of pulicarin on Ca2+-induced high-amplitude swelling of rat liver mitochondria were examined. Pulicarin inhibited mitochondrial swelling by 12.0±0.57% at 50 μM, 33.2±0.57% at 100 μM, 59.1±1.2% at 150 μM, and 67.1±0.8% at 200 μM (Fig. 2, E). The IC50 of pulicarin on mPTP was determined to be 131.8±1.2 μM (Fig. 3, C). The effects of isorhamnetin on mPTP in rat liver mitochondria were also investigated. At 50 μM, isorhamnetin inhibited Ca2+-induced high-amplitude mitochondrial swelling by 36.2±0.3%. At 100, 150, and 200 μM, inhibition increased to 40.3±0.45%, 66.4±1.1%, and 75.7±2.2%, respectively (Fig. 2, F). The IC50 value for isorhamnetin was 116.2 ± 1.0 μM (Fig. 3, D). Oroxylin A was also evaluated for its effect on mPTP in Ca2+-stimulated rat liver mitochondria. At 50 μM, Oroxylin A inhibited mPTP opening by 12.8±1.12% relative to the control. At 100, 150, and 200 μM, inhibition of high-amplitude mitochondrial swelling reached 38.5±1.4%, 67.8±1.3%, and 75.3±2.1%, respectively (Fig. 2, G). The IC50 of Oroxylin A was determined to be 117.1±0.63 μM (Fig. 3, H). Quercetin inhibited Ca2+-induced high-amplitude swelling of rat liver mitochondria more effectively than the previously tested flavonoids. At 50 μM, quercetin reduced mitochondrial swelling by 46.4±0.8%, while at 100, 150, and 200 μM, inhibition increased to 55.0±1.1%, 68.0±1.5%, and 77.9±1.5%, respectively (Fig. 2, H). The IC50 of quercetin was 67.5±2.97 μM (Fig. 3, F). The effects of chrysoeriol on mPTP in Ca2+-stimulated rat liver mitochondria were also examined. At 50 μM, chrysoeriol inhibited the open transition of mPTP by 21.1±1.1% relative to the control; at 100 μM, inhibition reached 32.3±1.7%; at 150 μM, 56.9±1.3%; and at 200 μM, 67.1±0.8% (Fig. 2, I). The IC50 of chrysoeriol was 133.8±2.2 μM (Fig. 3, G). Apigenin was also tested for its effect on Ca2+-induced high-amplitude mitochondrial swelling. At 10 μM, apigenin inhibited swelling by 10.1±1.74%; at 20 μM by 20.1±0.93%; at 30 μM by 50.2±1.4%; and at 40 and 50 μM, inhibition reached 65.7±2.3% and 75.2±0.8%, respectively (Fig. 2, J). The IC50 of apigenin was 29.9±0.16 μM (Fig. 3, H). Finally, the quercetin glycoside hyperoside was evaluated. At 50 μM, hyperoside inhibited Ca2+-induced mitochondrial swelling by only 7.8±0.4%; at 100 μM, inhibition was 22.0±1.6%; and at 150 and 200 μM, inhibition reached 36.9±1.4% and 66.2±1.68%, respectively (Fig. 2, K). The IC50 of hyperoside was 170.6±0.3 μM (Fig. 3, I).
The results indicate that the flavonoids studied exhibited a characteristic inhibitory effect on Ca2+-induced opening of mPTP in rat liver mitochondria. Apigenin inhibited high-amplitude mitochondrial swelling at low concentrations up to 50 μM, whereas quercetin exerted a dose-dependent inhibitory effect on mPTP at 50, 100, 150, and 200 μM, showing a lower IC50 compared to all flavonoids except apigenin. Under the same concentration range (50–200 μM), kaempferitrin and afzelin failed to reach 50% inhibition of mPTP. In the case of the remaining flavonoids, the inhibition of mPTP in rat liver mitochondria ranged on average from 60% to 75% at maximal concentrations.
It is well established that pyridine nucleotides exert specific effects on the state of mPTP located in the inner mitochondrial membrane. It has been shown that pyridine nucleotides, particularly NADH, can increase the Ca2+-retention capacity (CRC) in mitochondria from various tissues by 1.5- to 2.5-fold. This effect did not significantly alter mitochondrial Ca2+ uptake or the concentration of free Ca2+ in the medium. Moreover, while reducing Ca2+-induced mitochondrial swelling, NADH did not trigger mitochondrial contraction or repolarization. These findings indicate the existence of an external NADH-dependent regulatory site for mPTP.39 Based on this, the effects of flavonoids on mPTP in the presence of the pyridine nucleotide NADH were investigated. Experiments were conducted using flavonoid concentrations of 50 and 100 μM, while for apigenin, 10–20 μM was used. In these studies, mitochondria were pre-incubated with 50 μM NADH for 10 minutes, after which the effects of Ca2+ (10 μM) and the flavonoids were assessed. Initially, kaempferol at a concentration of 50 μM inhibited high-amplitude Ca2+-induced swelling of rat liver mitochondria by 45.5±1.3%, whereas at 100 μM, inhibition reached 77.6±2.4% (Fig. 4, A). Kaempferol glycoside kaempferitrin at a concentration of 50 μM inhibited Ca2+-induced swelling of mitochondria by 33.6±1.79%, and at 100 μM, inhibition reached 66.8±1.47% (Fig. 4, B). Another kaempferol glycoside, kaempferol-7-O-rhamnoside, inhibited Ca2+-induced mPTP opening by 22.9±1.29% and 55.8±1.49% at 50 and 100 μM, respectively (Fig. 4, C). Afzelin at 50 μM reduced Ca2+-induced mitochondrial swelling by 22.6±1.27%, and at 100 μM, by 41.8±1.2% (Fig. 4, D). Pulicarin at 50 and 100 μM inhibited Ca2+-induced mPTP opening by 63.7±0.27% and 74.9±0.64%, respectively (Fig. 4, E). Isorhamnetin at 50 and 100 μM inhibited Ca2+-induced mPTP opening by 62.1±0.92% and 46.2±0.9%, respectively (Fig. 4, F). Similarly, Oroxylin A at concentrations of 50 and 100 μM inhibited Ca2+-induced mPTP opening by 63.7±0.86% and 86.9±1.15%, respectively (Fig. 4, G). Quercetin at 50 and 100 μM inhibited Ca2+-induced mPTP opening by 54.0±0.86% and 94.0±1.28%, respectively (Fig. 4, H). Chrysoeriol at 50 and 100 μM inhibited Ca2+-induced mitochondrial swelling by 59.7±1.03% and 70.7±1.3%, respectively (Fig. 4, I). Apigenin at 10 and 20 μM inhibited Ca2+-induced mPTP opening by 37.6±0.9% and 61.0±1.4%, respectively (Fig. 4, J). Hyperoside at 50 and 100 μM inhibited Ca2+-induced mPTP opening by 31.5±0.3% and 58.15±0.4%, respectively (Fig. 4, K). The obtained results indicate that in the presence of NADH, flavonoids more effectively inhibit Ca2+-induced mPTP opening compared to their individual effects. This suggests that the pyridine nucleotide modulates the influence of flavonoids on mPTP status.
 |
Figure 4: Effect of various concentrations of flavonoids on Ca2+-dependent mPTP in rat liver, associated with β – Nicotinamide adenine dinucleotide. A – Kaempferol; B – Kaempferol-7-O-rhamnoside; C – Kaempferitrin; D – Afzelin; |
In the study, the effects of flavonoids on succinate–ferricyanide reductase activity were investigated. It was found that flavonoids at concentrations of 10 and 50 μM differentially modulated the enzyme activity. Under the influence of 10 μM kaempferol, the enzyme activity increased by 25.0±0.67%, while at 50 μM, it increased by 47.1±1.12% (Fig. 5, A). Kaempferol-7-O-rhamnoside enhanced succinate–ferricyanide reductase activity by 11.6±0.98% at 10 μM and 25.7±1.1% at 50 μM (Fig. 5, B). Afzelin also exerted a modest effect, increasing enzyme activity by 7.24±1.5% at 10 μM and 25.8±1.5% at 50 μM (Fig. 5, C). Kaempferitrin displayed a weaker activation, increasing enzyme activity by 3.35±0.52% at 10 μM and 15.3±1.15% at 50 μM (Fig. 5, D). Pulicarin increased enzyme activity by 17.5±0.3% at 10 μM and 30.4±1.04% at 50 μM (Fig. 5, E). Isorhamnetin enhanced the enzyme activity by 22.9±1.2% at 10 μM and 37.7±1.6% at 50 μM (Fig. 5, F). Oroxylin A increased the activity by 22.5±1.1% at 10 μM and 30.3±0.85% at 50 μM (Fig. 5, G). Quercetin showed a modest effect, enhancing enzyme activity by 9.43±0.51% at 10 μM and 11.9±0.74% at 50 μM (Fig. 5, H). Chrysoeriol (Fig. 5, I), apigenin (Fig. 5, J), and hyperoside (Fig. 5, K) increased succinate–ferricyanide reductase activity by 11.0±1.26%, 11.2±0.2%, and 28.1±0.86% at 10 μM, respectively, and by 16.7±1.4%, 14.6±0.88%, and 32.3±0.7% at 50 μM, respectively.
The obtained results indicate that the studied flavonoids possess the ability to moderately activate succinate–ferricyanide reductase, an enzyme localized in the mitochondrial respiratory chain. Literature data suggest that the NAD+-dependent deacetylase sirtuin-3 (SIRT3), a protein deacetylase complex, deacetylates the succinate dehydrogenase subunit SdhA, thereby enhancing the activity of mitochondrial respiratory chain complex II.
 |
Figure 5: Effects of selected flavonoids on succinate–ferricyanide reductase activity. A – Kaempferol; B – Kaempferol-7-O-rhamnoside; C – Kaempferitrin; D – Afzelin; |
Moreover, treatment of K562 cell lines with nicotinamide and kaempferol was shown to stimulate SIRT3 expression, reduce acetylation of the SdhA subunit, and increase the activity of complex II of the mitochondrial respiratory chain in these cells. Additionally, acetylation of the hydrophilic surface of SdhA has been proposed to regulate substrate entry into the enzyme’s active center.40 These findings suggest that kaempferol participates in this process, promoting the activation of succinate dehydrogenase. In the conducted experiments, flavonoids generally exhibited a trend toward activating succinate–ferricyanide reductase; however, apigenin, chrysoeriol, kaempferitrin, and oroxylin A displayed very weak activating effects at the concentrations tested.
In the conducted experiments, the effects of flavonoids on NADH–ferricyanide reductase activity were investigated. It was found that flavonoids inhibited enzyme activity in a concentration-dependent manner. Specifically, kaempferol inhibited NADH–ferricyanide reductase activity by 59.7±2.8% at a concentration of 10 µM and by 84.8±1.85% at 50 µM (Fig. 6, A). Kaempferol-7-O-rhamnoside inhibited the enzyme activity by 54.8±2.1% at 10 µM and by 72.3±3.1% at 50 µM (Fig. 6, B). The kaempferol glycoside, kaempferitrin, exhibited weaker inhibitory effects, reducing enzyme activity by 40.5±2.03% at 50 µM and by 51.1±1.13% at 200 µM (Fig. 6, C). Afzelin inhibited NADH–ferricyanide reductase activity by 33.8±1.9% at 10 µM and by 41.9±1.9% at 50 µM (Fig. 6, D). Furthermore, pulicarin inhibited enzyme activity by 34.8±2.05% at 50 µM and by 49.3±2.5% at 200 µM (Fig. 6, E).
 |
Figure 6: Effects of selected flavonoids on NADH–ferricyanide reductase activity. A – Kaempferol; B – Kaempferol-7-O-rhamnoside; C – Kaempferitrin; D – Afzelin; |
Isoramnetin inhibited NADH–ferricyanide reductase activity by 63.5±1.5% at a concentration of 10 µM and by 83.5±2.6% at 50 µM (Fig. 6, F). Oroxylin A inhibited enzyme activity by 33.6±1.0% at 10 µM and by 43.6±0.95% at 50 µM (Fig. 6, G). Quercetin exhibited a strong inhibitory effect, reducing enzyme activity by 78.8±1.0% at 10 µM and almost completely inhibiting it at 50 µM (Fig. 6, H). Chrysoeriol inhibited enzyme activity by 27.8±1.2% at 50 µM and by 60.2±0.8% at 200 µM (Fig. 6, I). Apigenin inhibited enzyme activity by 33.1±2.4% and 59.4±2.3% at 10 and 50 µM, respectively (Fig. 6, J). Finally, hyperoside inhibited NADH–ferricyanide reductase activity by 68.6±1.4% at 10 µM and by 88.2±1.7% at 50 µM (Fig. 6, K).
The obtained results indicate that flavonoids exhibited a concentration-dependent inhibitory effect on NADH–ferricyanide reductase activity.
Discussion
The obtained results demonstrate that flavonoids at various concentrations exert inhibitory effects on NADH–ferricyanide reductase activity. Among the tested compounds, quercetin, isorhamnetin, kaempferol-7-O-rhamnoside, hyperoside, apigenin, and kaempferol exhibited stronger inhibitory effects on enzyme activity, whereas the remaining flavonoids showed relatively weaker inhibitory effects. Overall, the studied flavonoids differentially modulated mPTP, succinate–ferricyanide reductase, and NADH–ferricyanide reductase activities. In particular, they led to inhibition of mPTP and NADH–ferricyanide reductase activities, while causing an increase in succinate–ferricyanide reductase activity. Furthermore, during the assessment of mPTP status, the use of exogenous NADH was shown to enhance the mPTP-inhibitory properties of the flavonoids.
It is well known that flavonoids are polyphenolic biologically active compounds synthesized in plants, and represent substances possessing antioxidant, anti-inflammatory, antidiabetic, antibacterial, antiviral, immunomodulatory, anticancer, cytoprotective, cytotoxic, lipid-lowering, and other biological activities. 41-44 It was revealed that quercetin, apigenin, and kaempferol, by inhibiting mitochondrial complex I, suppress H2O2 formation at low concentrations, while at the same time exhibiting no significant inhibitory effects on mitochondrial complexes II and III.45 The flavonoid (+)-catechin, in the presence of glutamate–malate substrates, inhibited mitochondrial complex I at nanomolar concentrations, leading to a decrease in membrane potential as well as a reduction in the rates of H2O2 and NO formation. The observed decrease in NO production was suggested to be associated with a direct effect of the flavonoid on mitochondrial nitric oxide synthase (mtNOS) activity.46 Indeed, the studied flavonoids were shown to strongly inhibit mitochondrial complex I, namely NADH–ferricyanide reductase activity (Fig. 6). However, a slight discrepancy was observed between the effects of the studied flavonoids on mitochondrial complex II, i.e., succinate–ferricyanide reductase activity, and the data reported in the literature (Fig. 5). This may be attributed to the use of ferricyanide as an electron acceptor in the determination of SDH activity.47,48 Furthermore, the increase in succinate–ferricyanide reductase activity in rat muscle mitochondria under the influence of flavonoids suggested that the presence of a hydroxyl group on the C-ring of flavonoids may be one of the main factors contributing to enzyme activation.49
It has been shown that peroxidized cardiolipin in mitochondria can act as an inducer for mPTP, similar to Ca2+ ions, by lowering the threshold of Ca2+-induced mPTP opening, leading to cytochrome c release from mitochondria and initiating the early phase of apoptosis.50 At the same time, flavonoids were shown to exert a restorative effect on cytochrome c activity by inhibiting cardiolipin-induced pro-apoptotic peroxidase activity of cytochrome c,51 and the results obtained in these studies (Fig. 2) indicate that flavonoids may play an important role in correcting mPTP-related mitochondrial dysfunction. In addition, polyphenols and other biologically active compounds have been shown to exert inhibitory effects on mPTP status.52-56 Flavonoids have been shown to modulate mitochondrial potassium channels, thereby exhibiting cytoprotective properties.43
Conclusion
Flavonoids were found to exert inhibitory effects on mPTP in rat liver mitochondria, with the following rank order based on their half-maximal inhibitory concentrations: apigenin > quercetin > isorhamnetin > oroxylin A > pulicarin > chrysoeriol > kaempferol > kaempferol-7-O-rhamnoside > hyperoside. In the presence of exogenous NADH, the inhibitory effects of flavonoids on mPTP were pronounced for apigenin, oroxylin A, quercetin, kaempferol, chrysoeriol, hyperoside, and pulicarin, whereas the remaining flavonoids exhibited relatively weaker effects. Moreover, the flavonoids hyperoside, kaempferol, kaempferol-7-O-rhamnoside, isorhamnetin, quercetin, afzelin, and pulicarin increased succinate–ferricyanide reductase activity, whereas the remaining flavonoids exhibited very weak or no effect. At the same time, quercetin, isorhamnetin, kaempferol-7-O-rhamnoside, hyperoside, and kaempferol showed strong inhibitory effects on NADH–ferricyanide reductase activity, while the other flavonoids exhibited relatively weaker inhibitory effects.
Acknowledgement
We would like to express our appreciation to the scientific team of the flavonoids laboratory of the Institute of Plant Substances Uzbek Academy of Sciences for kindly provided flavonoids for the research.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics Statement
All manipulations with animals were carried out in accordance with the European Convention for the Protection of Animals Used for Scientific Purposes (1998) and the International Bioethical Guidelines of the Institute of Biophysics and Biochemistry of the National University of Uzbekistan (BEC/IBB-N44/2024/14-1).
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable.
Author Contributions
- Boburbek Yuldoshev: Conceptualization and methodology, investigation, writing-original draft, writing-review, english editing, supervision.
- Nurali Ergashev: Conceptualization and methodology, writing-original draft, writing-review, english editing, supervision and formal analysis.
- Esokhon Komilov: Conceptualization and methodology, investigation, funding acquisition, supervision.
- Mavluda Kenjayeva: Investigation, funding acquisition.
- Mukhlisa Ikromova: Investigation, funding acquisition.
- Yulduz Rakhmatillayeva: Investigation, funding acquisition.
- Hazratova Hulkar: Funding acquisition.
- Komila Eshbakova: Investigation.
- Donyor Siddikov: Investigation.
- Bakhrom Komilov: Investigation.
- Rustam Makhmudov: Investigation.
- Asrarov Muzaffar Islamovich: Conceptualization and methodology.
References
- Endlicher R, Drahota Z, Štefková K, Červinková Z, Kučera O. The Mitochondrial Permeability Transition Pore-Current Knowledge of Its Structure, Function, and Regulation, and Optimized Methods for Evaluating Its Functional State. 2023;12(9):1273.
CrossRef - Haworth R, Hunter D. Control of the mitochondrial permeability transition pore by high-affinity ADP binding at the ADP/ATP translocase in permeabilized mitochondria. J Bioenerg Biomembr. 2000;32(1):91-96.
CrossRef - D’Angelo D, Vecellio Reane D, Raffaello A. Neither too much nor too little: mitochondrial calcium concentration as a balance between physiological and pathological conditions. Front Mol Biosci. 2023;10:1336416.
CrossRef - Robichaux D, Harata M, Murphy E, Karch J. Mitochondrial permeability transition pore-dependent necrosis. J Mol Cell Cardiol. 2023;174:47-55.
CrossRef - Zhou Y, Jing S, Liu S, et al. Double-activation of mitochondrial permeability transition pore opening via calcium overload and reactive oxygen species for cancer therapy. J Nanobiotechnology. 2022;20(1):188.
CrossRef - Bernardi P, Gerle C, Halestrap A, et al. Identity, structure, and function of the mitochondrial permeability transition pore: controversies, consensus, recent advances, and future directions. Cell Death Differ. 2023;30(8):1869-1885.
CrossRef - Elustondo P, Nichols M, Negoda A, et al. Mitochondrial permeability transition pore induction is linked to formation of the complex of ATPase C-subunit, polyhydroxybutyrate and inorganic polyphosphate. Cell Death Discov. 2016;2:16070.
CrossRef - Zorov D, Juhaszova M, Yaniv Y, Nuss H, Wang S, Sollott S. Regulation and pharmacology of the mitochondrial permeability transition pore. Cardiovasc Res. 2009;83(2):213-225.
CrossRef - Halestrap A, Richardson A. The mitochondrial permeability transition: a current perspective on its identity and role in ischaemia/reperfusion injury. J Mol Cell Cardiol. 2015;78:129-141.
CrossRef - Mishra J, Davani A, Natarajan G, Kwok W, Stowe D, Camara A. Cyclosporin A Increases Mitochondrial Buffering of Calcium: An Additional Mechanism in Delaying Mitochondrial Permeability Transition Pore Opening. 2019;8(9):1052.
CrossRef - Chen Z, Wang H, Wang Q. Therapeutic potential of NADH: in neurodegenerative diseases characterizde by mitochondrial dysfunction. Lin Chuang Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2024;38(1):57-62.
- Berthiaume J, Kurdys J, Muntean D, Rosca M. Mitochondrial NAD+/NADH Redox State and Diabetic Cardiomyopathy. Antioxid Redox Signal. 2019;30(3):375-398.
CrossRef - Walker M, Tian NAD(H) in mitochondrial energy transduction: implications for health and disease. Curr Opin Physiol. 2018;3:101-109.
CrossRef - Hu Q, Wu D, Walker M, Wang P, Tian R, Wang W. Genetically encoded biosensors for evaluating NAD+/NADH ratio in cytosolic and mitochondrial compartments. Cell Rep Methods. 2021;1(7):100116.
CrossRef - Chang J, Go S, Gilglioni E, et al. Soluble adenylyl cyclase regulates the cytosolic NADH/NAD+ redox state and the bioenergetic switch between glycolysis and oxidative phosphorylation. Biochim Biophys Acta Bioenerg. 2021;1862(4):148367.
CrossRef - Slama N, Abdellatif A, Bahria K, et al. NADH Intraperitoneal Injection Prevents Lung Inflammation in a BALB/C Mice Model of Cigarette Smoke-Induced Chronic Obstructive Pulmonary Disease. 2024;13(10):881.
CrossRef - Mimaki M, Wang X, McKenzie M, Thorburn D, Ryan M. Understanding mitochondrial complex I assembly in health and disease. Biochim Biophys Acta. 2012;1817(6):851-62.
CrossRef - Stroud D, Surgenor E, Formosa L, et al. Accessory subunits are integral for assembly and function of human mitochondrial complex I. 2016;538(7623):123-126.
CrossRef - Grivennikova V, Gladyshev G, Zharova T, Borisov V. Proton-Translocating NADH-Ubiquinone Oxidoreductase: Interaction with Artificial Electron Acceptors, Inhibitors, and Potential Medicines. Int J Mol Sci. 2024;25(24):13421.
CrossRef - Kahlhöfer F, Gansen M, Zickermann V. Accessory Subunits of the Matrix Arm of Mitochondrial Complex I with a Focus on Subunit NDUFS4 and Its Role in Complex I Function and Assembly. Life (Basel). 2021;11(5):455.
CrossRef - Kahlhöfer F, Kmita K, Wittig I, Zwicker K, Zickermann V. Accessory subunit NUYM (NDUFS4) is required for stability of the electron input module and activity of mitochondrial complex I. Biochim Biophys Acta. 2017;1858(2):175-181.
CrossRef - Cao K, Xu J, Cao W, et al. Assembly of mitochondrial succinate dehydrogenase in human health and disease. Free Radic Biol Med. 2023;207:247-259.
CrossRef - Dalla Pozza E, Dando I, Pacchiana R, et al. Regulation of succinate dehydrogenase and role of succinate in cancer. Semin Cell Dev Biol. 2020;98:4-14.
CrossRef - Moosavi B, Zhu X, Yang W, Yang G. Genetic, epigenetic and biochemical regulation of succinate dehydrogenase function. Biol Chem. 2020;401(3):319-330.
CrossRef - Chouchani E, Pell V, Gaude E, et al. Ischaemic accumulation of succinate controls reperfusion injury through mitochondrial ROS. 2014;515(7527):431-435.
CrossRef - Kamarauskaite J, Baniene R, Trumbeckas D, Strazdauskas A, Trumbeckaite S. Increased Succinate Accumulation Induces ROS Generation in In Vivo Ischemia/Reperfusion-Affected Rat Kidney Mitochondria. Biomed Res Int. 2020;2020:8855585.
CrossRef - Korge P, John S, Calmettes G, Weiss J. Reactive oxygen species production induced by pore opening in cardiac mitochondria: The role of complex II. J Biol Chem. 2017;292(24):9896-9905.
CrossRef - Duong Q, Levitsky Y, Dessinger M, Strubbe-Rivera J, Bazil J. Identifying Site-Specific Superoxide and Hydrogen Peroxide Production Rates From the Mitochondrial Electron Transport System Using a Computational Strategy. Function (Oxf). 2021;2(6):zqab050.
CrossRef - Jin Z, Wu J, Yan L. Chemical Conditioning as an Approach to Ischemic Stroke Tolerance: Mitochondria as the Target. Int J Mol Sci. 2016;17(3):351.
CrossRef - Siddikov, Kh. Bobakulov, A. Batoshov, S. Nishanbaev, N. Abdullaev. Phenolic compounds from the aerial part of Geranium rotundifolium. Chemistry of Natural Compounds. 2021; 57(3): 539-541.
CrossRef - Directive 2010/63/EU on the protection of animals used for scientific purposes. Official Journal of the European Union, Luxembourg, Luxembourg, 2010.
- W Schneider and G Hogeboom. Cytochemical studies of mammalian tissues: The isolation of cell components by differential centrifugation. Cancer Research. 1951; 11(1): 1-22.
- Y Umidakhon, B Erkin, G Ulugbek, N Bahadir and A Karim. Correction of the mitochondrial NADH oxidase activity, peroxidation and phospholipid metabolism by haplogenin-7-glucoside in hypoxia and ischemia. Trends in Sciences. 2022; 19(21): 6260.
CrossRef - N Ergashev, K Sayfieva, R Makhmudov and M Asrarov. Effect of tannins isolated from Plantago major L. and Plantago lanceolata L. on mitochondrial permeability transition pore in rat liver. Trends in 2024; 21(7): 7661.
CrossRef - Вдовиченко Л. О действии некоторых нейромедиаторов на сукдинатдегидрогеназную активность. Биохимия. 1973; 38(1):22-27.
- Shukolyukov S. Oxidation of pyridinnucleotides by bovine retinal rod outer segments. Journal of Evolutionary Biochemistry and Physiology. 6: 504-510.
- Panel M, Ahmed-Belkacem A, Ruiz I, et al. A Phenyl-Pyrrolidine Derivative Reveals a Dual Inhibition Mechanism of Myocardial Mitochondrial Permeability Transition Pore, Which Is Limited by Its Myocardial Distribution. J Pharmacol Exp Ther. 2021;376(3):348-357.
CrossRef - Bangar S, Chaudhary V, Sharma N, Bansal V, Ozogul F, Lorenzo J. Kaempferol: A flavonoid with wider biological activities and its applications. Crit Rev Food Sci Nutr. 2023;63(28):9580-9604.
CrossRef - Kharechkina E, Nikiforova A, Kruglov A. NAD(H) Regulates the Permeability Transition Pore in Mitochondria through an External Site. Int J Mol Sci. 2021;22(16):8560.
CrossRef - Cimen H, Han M, Yang Y, Tong Q, Koc H, Koc E. Regulation of succinate dehydrogenase activity by SIRT3 in mammalian mitochondria. 2010;49(2):304-311.
CrossRef - Hasnat H, Shompa S, Islam M, et al. Flavonoids: A treasure house of prospective pharmacological potentials. 2024;10(6):e27533.
CrossRef - Kayumov K, Kuchkarova L, Durá-Travé T, et al. Immunomodulatory and Histoprotective Effects of Luteolin and Related Flavonoids in Autoimmune Thyroiditis: Evidence from A Rat Model. Trends Sci. 2025;22(12):10981.
CrossRef - Kicinska A, Jarmuszkiewicz W. Flavonoids and Mitochondria: Activation of Cytoprotective Pathways? 2020;25(13):3060.
CrossRef - Kuchkarova L, Kayumov K, Eshbakova K, et al. Comparative Evaluation of the Antidiabetic Effect of Flavonoids Dihydroquercetin and Thamiflaside. Trends Sci. 2026;23(2):11059.
CrossRef - Lagoa R, Graziani I, Lopez-Sanchez C, Garcia-Martinez V, Gutierrez-Merino C. Complex I and cytochrome c are molecular targets of flavonoids that inhibit hydrogen peroxide production by mitochondria. Biochim Biophys Acta. 2011;1807(12):1562-72.
CrossRef - Iglesias D, Bombicino S, Boveris A, Valdez L. (+)-Catechin inhibits heart mitochondrial complex I and nitric oxide synthase: functional consequences on membrane potential and hydrogen peroxide production. Food Funct. 2019;10(5):2528-2537.
CrossRef - Masand R, Paulo E, Wu D, et al. Proteome Imbalance of Mitochondrial Electron Transport Chain in Brown Adipocytes Leads to Metabolic Benefits. Cell Metab. 2018;27(3):616-629.e4.
CrossRef - Medentsev A, Arinbasarova A, Golovchenko N, Akimenko V. Involvement of the alternative oxidase in respiration of Yarrowia lipolytica mitochondria is controlled by the activity of the cytochrome pathway. FEMS Yeast Res. 2002;2(4):519-24.
CrossRef - Ergashev N, Yuldoshev B, Paizieva O, et al. Modulating effect of some flavonoids on succinate dehydrogenase activity in skeletal muscle mitochondria. Universum: Chemistry and Biology. 2026;1(139): 21613.
CrossRef - Petrosillo G, Moro N, Ruggiero F, Paradies G. Melatonin inhibits cardiolipin peroxidation in mitochondria and prevents the mitochondrial permeability transition and cytochrome c release. Free Radic Biol Med. 2009;47(7):969-74.
CrossRef - Lagoa R, Samhan-Arias A, Gutierrez-Merino C. Correlation between the potency of flavonoids for cytochrome c reduction and inhibition of cardiolipin-induced peroxidase activity. 2017;43(3):451-468.
CrossRef - Gayibov U, Komilov E, Rakhimov R, et al. Influence of new polyphenol compound from Euphorbia plant on mitochondrial function. J Microbiol Biotechnol Food Sci. 2019;8(4):1021-5.
CrossRef - Gayibov U, Gayibova S, Pozilov M, et al. Influence of quercetin and dihydroquercetin on some functional parameters of rat liver mitochondria. J Microbiol Biotechnol Food Sci. 2021;11(1):e2924.
CrossRef - Ergashev N, Sayfieva K, Makhmudov R, Asrarov M. Effect of Polyphenols Isolated from Plantago major L. and Plantago lanceolata L. on Mitochondrial Permeability Transition Pore in Rat Liver. Trends Sci. 2024;21(7):7661.
CrossRef - Muratova D, Ergashev N, Asrarov M, Pozilov M, Berdiyeva X. Comparative Analysis of the Effects of the Diterpene Alkaloid Napelline and its Derivative on the MitoK+ATP Channel and mPTP in Rat Liver and Heart Mitochondria. Trends Sci. 2025;22(12):10483.
CrossRef - Vakhobjonovna, Jurayevich E., Ogli A.Z., Azamovich N., Rasuljonovich R., Islomovich M. Tannins as Modulators in the Prevention of Mitochondrial Dysfunction. Trends Sci. 2025;22(8):10436.
CrossRef







