Manuscript accepted on :23-01-2026
Published online on: 19-02-2026
Plagiarism Check: Yes
Reviewed by: Dr. Dhara Patel and Dr. Shah Tapas
Second Review by: Dr. Aditya Vikram Jain Parmar
Final Approval by: Dr. Prabhishek Singh
Mohira Nuraddinova1 , Akmal Islomov2
, Akmal Islomov2 , Arofat Inkhonova3
, Arofat Inkhonova3 , Shukhrat Khakberdiyev4
, Shukhrat Khakberdiyev4 , Risolat Norboboyeva5
, Risolat Norboboyeva5 , Akmal Khaitov 6
, Akmal Khaitov 6 and Nodira Kurbanova7
and Nodira Kurbanova7
1Department of Medical and Biological Chemistry, Tashkent State Medical University, Tashkent, Uzbekistan
2Department of Physicochemical Methods of Research, Proteins and Peptides, and Pharmacology, A. S. Sadykov Institute of Bioorganic Chemistry of the Academy of Sciences of Uzbekistan, Uzbekistan.
3Department of Pharmacy, Alfraganus University, Tashkent, Uzbekistan.
4Department of Chemical Engineering, Jizzak Polytechnic Institute, Jizzak, Uzbekistan.
5Department of Biology, Faculty of Natural Sciences, National Pedagogical University of Uzbekistan named after Nizami, Tashkent, Uzbekistan.
6 Head of the Department of Surgical Sciences, Faculty of Medicine, PhD., Termez University of Economics and Service, Termez, Uzbekistan.
7Department of Medical and Biological Chemistry, Urgench State Medical University, Urgench, Uzbekistan
Corresponding Author E-mail:nmohira470@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3390
Abstract
This article presents methods and results for predicting the pharmacological and biological activity of amyrin substances isolated from the Euphorbia Milii plant using molecular docking and SWISSADME calculations, a quantum-chemical technique. By studying the nature of amyrin substances to determine their binding to proteins through molecular docking, the areas in which these substances can be applied in pharmacological processes have been identified.
Keywords
Amyrin; Euphorbia; Ligands; Molecular docking; SWISSADME calculations; Receptor
Download this article as:| Copy the following to cite this article: Nuraddinova M, Islomov A, Inkhonova A, Khakberdiyev S, Norboboyeva R, Khaitov A, Kurbanova N. Comparative Study of the Pharmacological Properties of Amyrin Compounds Derived from the Composition of Euphorbia Milii: Molecular Docking and SWISSADME Calculations. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Nuraddinova M, Islomov A, Inkhonova A, Khakberdiyev S, Norboboyeva R, Khaitov A, Kurbanova N. Comparative Study of the Pharmacological Properties of Amyrin Compounds Derived from the Composition of Euphorbia Milii: Molecular Docking and SWISSADME Calculations. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/4czqyE6 |
Introduction
As you know, medicinal plants are considered the main raw materials for producing medicines.27,28 For this reason, large-scale efforts are underway worldwide to cultivate medicinal plants, protect them, and identify new natural sources of medicines. In particular, in the Republic of Uzbekistan, In recent years, systematic reforms have been implemented to enhance the conservation of medicinal plants, promote the sustainable utilization of natural resources, improve the cultivation and processing of medicinal plants, and develop plantations for their propagation and reproductionThe local flora comprises over 4,300 plant species, approximately 750 of which are recognized for their medicinal properties.29 Among these, 112 species are officially documented for use in applied medicine, while around 70 species are actively utilized in the pharmaceutical industry. Representatives of the family Euphorbiaceae (mammals) are precisely those species that grow wild and are currently the most cultivated.4 The family Euphorbiaceae contains about 300 genera and about 7,500 species, with most representatives being grasses. In tropical and subtropical regions, it can also be found in tree, shrub, and semi-shrub views. Their original homeland was the subtropics of Africa, which is now cultivated in almost all countries of the earth.31 Representatives of this genus are trees, shrubs, and herbivores with stems growing erect or on their sides, sometimes large, serrated, leafless sometimes prickly, and of various shapes.6 Euphorbia is a genus of plants known from ancient times. Such a scientific name was given to the plant in honor of a person who lived in the 4th century BC. The physician Ephorbus, who discovered the healing properties of Euphorbia, was the first to use it for medicinal purposes. Plants of the order Euphorbia have long been known, many of which have been used by humans in their various fields. The classification of 6 species in this order was given by Theophrastus in his work “Studies on plants” (Theophrastus, 1951).8
Most plants in the family Euphorbiaceae are medicinal. For example, the Acalypha indica type is mainly used to treat bronchitis, asthma, and pneumonia. The type Euphorbia hirta is mainly used in the treatment of glandular inflammation as well as as as a expectorant. Euphorbia thymifolia is used in eye diseases and diseases associated with the breast, while Euphorbia tiruclli is used as an effective remedy for liver diseases, gallbladder diseases, and gallstones. In addition, representatives of this family also began to be used to treat various tumor diseases.24 In folk medicine of many countries, Euphorbia species are currently used to obtain drugs against tumors and hepatitis.
The data of many publications about the composition of polyphenols of representatives of the family Euphorbiaceae and their biological activity, studying the chemical composition of plants of this family growing on the territory of Uzbekistan, aroused interest in scientists and served as an impetus for research on this topic.32
The family Euphorbiaceae, order Euphorbia, is a species of Euphorbia milii of great interest worldwide and little studied in exactly Uzbekistan. Euphorbia milii, the original homeland of the” crown of thorns “plant, is the Madagascar islands, first introduced into Plant Systematics by Charles Des Moulins in 1826, hence the scientific name “Euphorbia milii des moulins”. Therefore, the plant name Euphorbia milii is also listed in some reviews as Euphorbia Splendes, Euphorbia bojeri, Euphorbia hislpoii, Euphorbia breonii.
Euphorbia milii is usually 60-90 cm tall, a plant that always maintains its greenness and is a shrub or semi-shrub according to its life form. The length of the neck of the representatives of the Euphorbia milii plant, distributed in tropical and subtropical regions, sometimes reaches 1.8-2 meters.27 The Root has an axial root system, white. The stem type is woody, erect, and branching. The leaves are sequentially positioned along the stem and coated with a waxy layer. They measure approximately 3.5 cm in length and 1.5 cm in width. On the lower part of the Euphorbia milii stem, the leaves are long and thick, whereas those near the stem apex are shorter and thinner.32
The quantum-chemical research carried out is based on the theoretical assumption of the pharmacological properties of the amyrin substance isolated from the Euphorbia milii plant.
Amyrin is a pentacyclic triterpenoid abundant in plants, corresponding to the general formula C30H50O3 types of amyrin: α-amyrin, β-amyrin and δ-amyrin have been identified. Ursolatic acid, common in nature, is a derivative of α-amyrin, while oleic acid is a derivative of β-amyrin, and δ-amyrin is very rare and little studied. The substance amyrin, found mainly in plants, and in small quantities in animal products, was mainly isolated from the stem and leaf of plants. The β-type of the substance amyrin is relatively widely used in medicine.41 Because β-amyrin is a biologically active substance, it is currently used in the treatment of oncological diseases. In addition to showing various microbes, fungi, and anti-inflammatory activity, the substance amyrin also has digestive system activating and antioxidant effects. Amyrin is mainly extracted from natural raw materials due to its relative cost as well as the difficulty in extracting the substance in laboratory conditions.
Materials and Methods
Qualitative and quantitative analysis of biologically active compounds in plants requires prior extraction, as this step is fundamental for assessing the composition of medicinal herbs. The accuracy of the final results largely depends on the extraction method employed.
For this study, air-dried roots, stems, and leaves of Euphorbia milii were utilized. Plant samples were collected from the Fergana, Tashkent, and Khorezm regions of the Republic of Uzbekistan.
Preparation of raw materials
Roots, stems, and leaves were harvested, washed with cold water, and spread in a thin layer on paper to dry.
Drying was performed in shaded conditions, avoiding direct sunlight, at a humidity of 78.8%. Adequate drying was assessed based on the texture and color of the plant material; leaves should not retain a dark green hue to prevent chlorophyll contamination in the extract.
For the extraction process, 3 types of organic solvents: acetone, chloroform, and ethanol (96%) were selected. For the extraction, plant samples and solvents in a ratio of 1:10 were used. The stems and leaves of each type of plant imported from the Fergana, Tashkent, and Khorezm regions were extracted separately. The composition of the extracts was verified by the GC-MS method and the presence of an amyrene substance was established, and in subsequent steps, the amyrine substance was isolated from the plant composition. Extraction, filtration, vacuum drive, purification, and precipitation methods were used to extract β-amyrin from the E.Milii plant. To start the process, the stem and leaves of the Fergana species E.Milii plant are dried and crushed (until it becomes a powder). In the next step, the crushed sample was extracted in chloroform. The extraction procedure took 3h and a return cooler was used. The extract was dissolved in 80% ethanol, diluted by adding boiled water in a 1:1 ratio, and then filtered and poured under vacuum, evaporation. The purified extract from chlorophyll and additives was re-dissolved in 96% ethanol and diluted again by adding distilled water in a ratio of 2:1. With the help of gasoline, the neutral substance was separated and put into the infusion. Preliminary theoretical pharmacological reports were carried out based on quantum chemistry as a pre-stage to investigate the pharmacological properties of the extracted amyrin. This allows you to know in advance what should be done in the laboratory.
Molecular docking is a computational method used to predict the mode and affinity of a small molecule (ligand) to bind to a protein or nucleic acid (receptor). This involves predicting the optimal spatial orientation of the ligand at the receptor binding site and calculating the energy of interaction between the two molecules.
First of all, 1uz9 protein, which originally belonged to insulin pdb.com downloaded from the site, the waters in the protein were removed and polar hydrogens and Kollman charges were placed. The area of interaction around the active center was defined in the AutoGrid Section at 78×67×62 (A3). α-amyrin and β-amyrin were selected as ligands and stored in PDB format by plotting them in the Avogadro program. Ligand-protein binding energies were studied in the AutoDock 4.2 program. The molecular docking of amyrin-to-insulin binding has been studied.
SWISSADME is a web-based tool developed by the Swiss Institute of Bioinformatics (SIB) and the École Polytechnique Fédérale de Lausanne (EPFL). It allows users to predict various molecular properties and assess the drug-likeness of small molecules. It provides valuable information for drug discovery and development processes. In SWISSADME, Log P represents the logarithm of a molecule’s partition coefficient between n-octanol and water. It was found that β-amyrin differs from α-amyrin in its Log P (lipophilicity) value. This measures the compound’s lipophilicity or hydrophobicity. The Log P value indicates how well a molecule can dissolve in lipid-based environments compared to water.
Additionally, differences in Log S (ESOL), Log S (Ali), and Log S (SILICOS-IT) values can be observed from Table 3.6. Log S, derived from the English term “Estimated Solubility,” refers to the predicted solubility of a compound. This value is based on a linear regression approach and helps determine the solubility level of the studied substance. A lower Log S value indicates lower solubility in water. In pharmaceutical and medical fields, the solubility of drugs is very important. Therefore, theoretically studying the water solubility of proposed biologically active compounds is essential.
Results
Results of molecular docking between amyrin molecules and insulin receptors.
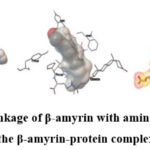 |
Picture 1: The linkage of β-amyrin with amino acid residues in the β-amyrin-protein complex |
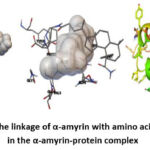 |
Picture 2: The linkage of α-amyrin with amino acid residues in the α-amyrin-protein complex |
Binding energy (ΔGbinding) is a key parameter of molecular docking used to determine the strength of the interaction between the ligand and the receptor. This represents the change in energy that occurs when the ligand binds to receptors and is usually expressed in kcal/mol per mole) or kJ/mol.
Table 1: In the complexes obtained by molecular docking research, the ligand molecules with protein amino acid residues (H-bond, VdV, etc.) exposures
| Ligand | Active amino acids involved in hydrogen bonding | Non-active amino acids involved in hydrogen bonding |
| b-amyrin | HIS4 | HIS5, PHE1, GLN4, VAL3, TYR26, GLU4, PRO28 |
| α-amyrin | GLN4, VAL3, GLU4, GLY8, GLY1, TYR26, PRO28, HIS5, PHE1 |
In molecular docking, more negative binding energy indicates a stronger interaction between the ligand and the receptor. This is because the binding energy represents the free energy change that occurs when the ligand binds to the receptor.
Table 2: Ligand-protein binding energies (ΔGbinding, kcal/mol)
| № | b–amirin(ΔGbinding) | α-amirin(ΔGbinding) |
| 1. | -10.06 | -9.08 |
| 2. | -10.06 | -9.01 |
| 3. | -10.06 | -9.01 |
| 4. | -10.06 | -9.00 |
| 5. | -10.06 | -9.00 |
| 6. | -10.05 | -9.00 |
| 7. | -10.05 | -9.00 |
| 8. | -10.03 | -9.00 |
| 9. | -10.03 | -8.99 |
| 10. | -10.01 | -7.64 |
Table 3:
| № | α-amirin(ΔGbinding) | β-amirin(ΔGbinding) |
| 1 | -8.06 | -8.67 |
| 2 | -8.05 | -8.09 |
| 3 | -8.04 | -8.05 |
| 4 | -8.04 | -8.03 |
| 5 | -7.91 | -7.84 |
| 6 | -7.79 | -7.80 |
| 7 | -7.61 | -7.79 |
| 8 | -7.61 | -7.79 |
| 9 | -7.48 | -7.74 |
| 10 | -7.31 | -7.67 |
Among the ligands above, the binding energy between β-amyrin and protein can be seen to be the smallest, and this means that the binding between the ligand and the 1uz9 protein is strong. The smallest energy value representing bond strength was ΔGbinding=-10.06.
α-amyrin and β-amyrin are triterpenoid compounds. The results of molecular docking made it possible to theoretically study the biological activities of both substances selected as objects and compare the biological activities of substances among themselves. One of the theoretical computations on α-amyrin and β-amyrin is that molecular docking studies have also shown differences in their interactions with specific proteins.
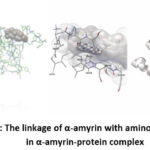 |
Picture 3: The linkage of α-amyrin with amino acid residues in α-amyrin-protein complex |
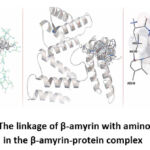 |
Picture 4: The linkage of β-amyrin with amino acid residues in the β-amyrin-protein complex |
Table 4: Studying the pharmacokinetic properties of alpha and beta amyrin using computer modeling
| Pharmacological properties | α-amyrin | β-amyrin |
| Molecular weight g/mol | 426.72 | 426.72 |
| Lipophilicity Log Po/w XLOGP3 | 9.01 | 9.15 |
| TPSA | 20.23 Ų | 20.23 Ų |
| Solubility | -8.16-9.33
-6.71 |
-8.25-9.47
-7.16 |
| Log S ESOL | 0.93 | 0.93 |
| Log S Ali | -2.51 | -2.41 |
| Log S SILICOS-IT | less | less |
| Saturation Csp3 | no | no |
Discussion
In drug development, ligands with more negative binding energy are generally considered stronger and are more likely to be recommended for production as drugs. Molecular docking results have shown that the substance β-amyrin has higher biological activity than α-amyrin. Calculations using modern computer programs will help predict the biological activity of the studied substance in advance, but at the same time, it is advisable to carry out practical examinations after theoretical calculations.
One of the diseases that is currently growing in high percentages around the world is diabetes. Hence, the identification of novel compounds with insulin-modulating activity is crucial for maintaining glucose homeostasis and for the effective management of diabetes. Studies using molecular docking have shown that β-amyrin binds rapidly and easily to insulin promoter proteins relative to α-amyrin. This helps β-amyrin to activate or ingress insulin production and thus regulate glucose metabolism. In addition, the strong binding between β-amyrin and bacterial proteins indicates its antibacterial activity. So, theoretical calculations have proven that the substance β-amyrin can be used in the production of drugs that prevent diabetes and have antibacterial properties, as well as biologically active additives.
Log Kp (membrane permeability) represents the logarithm of the compound’s membrane permeability coefficient. It measures the molecule’s ability to penetrate the skin and enter systemic circulation. A higher Log Kp value indicates a greater degree of cellular uptake through membrane transport. These theoretical calculations are approximate and should be validated in the laboratory before practical application.
Although SWISSADME is a good information tool, it is advisable to test theoretically obtained data in the laboratory before applying it in practice. In conclusion, in today’s developing information era, using digital technologies is a necessity. Various quantum-chemical calculations save scientists’ time and prevent unnecessary use of substances. Therefore, modeling or theoretically studying the biological activity of a compound or extract on a computer first allows researchers to choose a clear direction for their work. This ensures both economic efficiency and work productivity. However, since theoretical data do not always match practical results, it is recommended to first verify the results obtained from software before applying them in practice.
Conclusion
Using the molecular docking program, it is possible to predict in advance the biological and pharmacological properties of substances isolated from the plant, thereby understanding exactly what laboratory work needs to be carried out practically. According to this project, molecular docking has shown that β-amyrin has higher biological activity than α-amyrin.
Acknowledgement
The authors wish to thank Tashkent State Medical University and National University of Uzbekistan for the opportunity and support during the research.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed Biomedical and Pharmacology Journal consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
The sole author was responsible for the conceptualization, the second author for methodology, the third one for data collection, the fourth one for analysis, the fifth author for writing, and the sixth and seventh authors were responsible for final approval of the manuscript.
References
- Arafa NM, Amer AM, Girgis ND, El-Shenawy R, Helmy NM. Effect of different carbon sources on callus formation, pigment accumulation, and antiviral activity in cell cultures of Euphorbia milii. Egypt Pharm J. 2023;22(3):432-439. doi:10.4103/epj.epj_19_23
CrossRef - Matsunaga S, Tanaka R, Akagi M. Triterpenoids from Euphorbia maculata. 1988;27(2):535-537. doi:10.1016/0031-9422(88)83136-4
CrossRef - Hoang L, Dong P, Nguyen V, Thao VTM, Ramadhan R, Jutakanoke R, Sichaem J. β-Amyrin heptadecanoate, a new oleanane triterpenoid with α-glucosidase inhibitory and cytotoxic activities from the leaves of Averrhoa bilimbi Nat Prod Res. 2024;1-9. doi:10.1080/14786419.2024.2425045
CrossRef - Silihe KK, Zingue S, Yeshak MY, Bisrat D, Kemboi D, Bräutigam K, Rody A, Michel T, Asres K, Njamen D, Bishayee A, Köster F. Purification of α-amyrin-acetate and phenylpropanoid compounds from Ficus umbellata Vahl (Moraceae) stem bark and evaluation of their anti-breast cancer potential. S Afr J Bot. 2024;177:445-456. doi:10.1016/j.sajb.2024.12.015
CrossRef - Song Q, Guo Y, Sun P, Fan Y, Ji K. Skeleton rearranged and oxygenated ent-Rosane diterpenoids with antiadipogenic activity from Euphorbia milii. Chin J Chem. doi:10.1002/cjoc.202400749
CrossRef - Negm WA, Elekhnawy E, Mokhtar FA, Binsuwaidan R, Attallah NG, Mostafa SA, Moglad E, Ibrahim S, Al-Fakhrany OM, Eliwa D. Phytochemical inspection and anti-inflammatory potential of Euphorbia milii Des Moul. integrated with network pharmacology approach. Arab J Chem. 2023;17(2):105568. doi:10.1016/j.arabjc.2023.105568
CrossRef - Al-Qrimli AF, B SH, J KE. Antiangiogenic activity and the isolation of five phenolic compounds from Euphorbia milii ethyl acetate solvent extract. Res J Pharm Technol. 2023:3083-3091. doi:10.52711/0974-360x.2023.00507
CrossRef - Chandrashekarappa SB, Assi S, Jayabalan M, Al-Hamid A, Al-Jumeily D. Exploring high opioid prescriptions among nephrologists in the United States using machine learning algorithms. Emerg Trends Drugs Addict Health. 2024;5:100165. doi:10.1016/j.etdah.2024.100165
CrossRef - Wang Y, Qi X, Hou J, Xu B. Quantum coherence of Fermionic Gaussian states. Results Phys. 2025;108107. doi:10.1016/j.rinp.2025.108107
CrossRef - Zhao C, Huang X, Yang K, Wang X, Wang Q. Generalizable 3D Gaussian splatting for novel view synthesis. Pattern Recognit. 2024;111271. doi:10.1016/j.patcog.2024.111271
CrossRef - Serhatlioglu I, Kilic I, Yaman O, Kacar E, Oz ZD, Ozdede MR, Yol F, Kelestimur H. A new method based on Local Binary Gaussian Pattern for classification of rat estrous cycle stages using smear images. Biomed Signal Process Control. 2024;103:107390. doi:10.1016/j.bspc.2024.107390
CrossRef - Khadka M, Sah M, Chaudhary R, Sahani SK, Sahani K, Pandey BK, Pandey D. Spectroscopic, quantum chemical, and topological calculations of the phenylephrine molecule using density functional theory. Sci Rep. 2025;15(1). doi:10.1038/s41598-024-81633-2
CrossRef - Zhou G, Xu Q, Liu X, Wang K. Quantitative analysis of chondroitin sulfate and dermatan sulfate using capillary electrophoresis with Gaussian distribution-based peak fitting for improved peak resolution. Sep Sci Plus. 2024;7(10). doi:10.1002/sscp.202300244
CrossRef - Li Y, Li Y, Gao W, Fang C, Lv J, Yue J, Yu J. Jatrophane and ingenane diterpenoids with anti-inflammatory activity from Euphorbia esula. Phytochemistry. 2024;232:114369. doi:10.1016/j.phytochem.2024.114369
CrossRef - Idrees S, Abbasi BA, Iqbal J, Kazi M. Euphorbia serpens extracts mediated synthesis of copper oxide nanoparticles and their biological applications. Heliyon. 2024;e41397. doi:10.1016/j.heliyon.2024.e41397
CrossRef - Zhao X, Zhang C, Qian X, Zhang J, Wang G, Wang Z. Research progress on the anti-tumor effects of Euphorbia humifusa. Discover Oncol. 2024;15(1). doi:10.1007/s12672-024-01624-7
CrossRef - Ferrer-Gallego PP, Riina R. (3055) Proposal to conserve the name Euphorbia portulacoides (Euphorbiaceae) with a conserved type. Taxon. 2024. doi:10.1002/tax.13271
CrossRef - Escobar-Montaño F, Gómez-Oliva R, Ezzanad A, De Górgolas SV, Zorrilla D, Macías-Sánchez AJ, Botubol-Ares JM, Nunez-Abades P, Castro C, Durán-Patrón R, Hernández-Galán R. Effect of lathyrane-type diterpenoids in neural stem cell physiology: Microbial transformations, molecular docking and dynamics studies. Bioorg Chem. 2024;153:107769. doi:10.1016/j.bioorg.2024.107769
CrossRef - Hassan AZ, Sweelam HM, Abd-Alla HI, Zohair MM, Ashour WE, Shaker KH. Phytochemical analysis and antimicrobial activity of Euphorbia milii. Egypt J Chem. 2023. doi:10.21608/ejchem.2023.192476.7575
CrossRef - Riet K, Adegoke A, Mashele S, Sekhoacha M. Effective use of Euphorbia milii DCM root extract encapsulated by thermosensitive immunoliposomes for targeted drug delivery in prostate cancer cells. Curr Issues Mol Biol. 2024;46(11):12037-12060. doi:10.3390/cimb46110714
CrossRef - Saidu MB, Krstić G, Barta A, Hunyadi A, Berkecz R, Gallah US, Cholke K, Gertsch J, Rédei D, Hohmann J. Euphane and tirucallane triterpenes with trypanocidal activity from Euphorbia desmondii. J Nat Prod. 2024. doi:10.1021/acs.jnatprod.4c00730
CrossRef - Aly SH, Elbadry AMM, Doghish AS, El-Nashar HAS. Unveiling the pharmacological potential of plant triterpenoids in breast cancer management: An updated review. Naunyn Schmiedebergs Arch Pharmacol. 2024. doi:10.1007/s00210-024-03054-2
CrossRef - Golik M, Titko T, Shaposhnyk A, Suleiman M, Drapak I, Sych I, Perekhoda L. QSAR analysis and molecular docking study of pyrrolo- and pyridoquinolinecarboxamides with diuretic activity. SciRise Pharm Sci. 2021;3(31):19-27. doi:10.15587/2519-4852.2021.234493
CrossRef - Paredes-Doig A, Pinedo-Flores A, Aylas-Orejón J, Obregón-Valencia D, Kou S. The interaction of metallic ions onto activated carbon surface using computational chemistry software. Adsorp Sci Technol. 2020;38(5-6):191-204. doi:10.1177/0263617420919234
CrossRef - Pasdaran A, Azarpira N, Aghdaie MH, Zare M, Sheidaie N, Fard FH, Hamedi A. Three new spirocyclic terpenoids from Euphorbia amygdaloides exhibit cytotoxicity against cancerous cell lines through early and late apoptosis. Arab J Chem. 2024;18(1):106049. doi:10.1016/j.arabjc.2024.106049
CrossRef - Pío-León JF, Salomón-Montijo B, Millán-Otero MG, Díaz JS. Jatropha marquezii (Euphorbiaceae), especie nueva, recuento de las especies de Jatropha en Sinaloa, México, y notas sobre J. purpurea. Bot Sci. 2024;103(1). doi:10.17129/botsci.3593
CrossRef - Ishmuratova AS, Abdugafurova DG, Islomov AKh, Mahmudov LU, Baratov KR, Azimova AQ. The study of the biologically active effect of Rubia tinctorum plant on rats with experimental kidney stone disease and issues of introduction. Biomed Pharmacology J. 2024;17(3):2035-2042. https://biomedpharmajournal.org/ vol17no4/determination-of-vitamins-and-pharmacological-properties-of-vitis-vinifera-l-plant-fruit-part-mixed-varieties-syrup-honey/
CrossRef - G’aybullayeva OO, Islomov AX, Abdugafurova DG, Elmurodov B, Mirsalixov BA, Mahmudov LU, Abdullaev IZ, Baratov KR, Omonturdiev SZ, Sa’dullayeva SA. Inula helenium root extract in sunflower oil: Determination of its content of water-soluble vitamins and immunity-promoting effect. Biomed Pharmacology J. 2024;17(4):2729-2737. https://biomedpharmajournal.org/vol17no4/inula-helenium-l-root-extract-in-sunflower-oil-determination-of-itscontent-of-water-soluble-vitamins-and-immunity-promoting-effect
CrossRef - Azimova AQ, Islomov AX, Maulyanov SA, Abdugafurova DG, Mahmudov LU, Abdullaev IZ, Ishmuratova AS, Siddikova SQ, Askarov IR. Determination of vitamins and pharmacological properties of Vitis vinifera plant fruit part (mixed varieties) syrup-honey. Biomed Pharmacology J. 2024;17(4):2779-2786. https://biomedpharmajournal.org/vol17no4/determination-of-vitamins-and-pharmacological-properties-of-vitis-vinifera-l-plant-fruit-part-mixed-varieties-syrup-honey
CrossRef - Saidova XA, Asqarov IR, Islomov AX, Ashurov JM, Abdugafurova D, Mahmudov L, Inkhonova A, Ishimov UJ. Development and HPLC characterization of a water-soluble supramolecular complex acid monoammonium salt: Composition, toxicity, and pharmaceutical potential. Biomed Pharmacology J. 2025;18(1):1017-1029. https://biomedpharmajournal.org/vol18no1/development-and-hplc-characterization-of-a-water-soluble-supramolecular-complex-of-propolis-and-glycyrrhizic-acid-monoammonium-salt-composition-toxicity-and-pharmaceutical-potential
CrossRef - Azimova A, Asqarov I, Islomov A, Inxonova A, Ishimov U, Mahmudov L, Xakberdiyev S, Oripdjanov O, Bekturdiyev G. Chemical composition and anti-inflammatory effects of Taifi grape (Vitis). Biomed Pharmacology J. 2025;18(4):1017-1029. https://biomedpharmajournal.org/vol18no4/chemical-profile-and-anti-inflammatory-effects-of-taifi-grapes-vitis-l/
CrossRef - Pirimova MA, Nuraddinova MB, Islomov AX, Asqarov IR. Mineral composition of Zingiber officinale Roscoe rhizomes and their applications in medicine. Universum: Khimiya i Biologiya. 2025;25(9). https://7universum.com/ru/nature/archive/item/20756.P.21-27
CrossRef







