Manuscript accepted on :08-01-2026
Published online on: 28-01-2026
Plagiarism Check: Yes
Reviewed by: Dr. Amit Gupta and Dr. Ana Golez
Second Review by: Dr. Oyeka Daniel
Final Approval by: Dr. Mariia Shanaida
Kumari Soniya1 , Sanjit Boora1
, Sanjit Boora1 , Suman Yadav1
, Suman Yadav1 , Abhishekh Kumar Bharti2
, Abhishekh Kumar Bharti2 , Manisha Sharma1
, Manisha Sharma1 , Poonam Patil2
, Poonam Patil2 , Kalichamy Alagarasu2
, Kalichamy Alagarasu2 , Deepti Parashar2
, Deepti Parashar2 and Samander Kaushik1*
and Samander Kaushik1*
1Centre for Biotechnology, Maharshi Dayanand University, Rohtak, India
2Dengue and Chikungunya Group, ICMR-National Institute of Virology, Pune, India
Corresponding Author Email: samanderkaushik@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3367
Abstract
Chikungunya is an infectious, mosquito-borne viral disease that poses a significant threat to global public health. No licensed antiviral drugs or vaccines are available to treat CHIKV infection. The present study aims to investigate the in vitro antiviral and anti-inflammatory potential of Cynodon dactylon aqueous extract. FTIR and GC-MS phytochemical studies were used to identify the primary bioactive component. The maximum non-toxic dose of the plant was determined through an MTT assay on Vero cells. Subsequently, the antiviral potential was evaluated through pre-, co-, and post-treatment assays to determine the stage at which the extract is most effective. Cell morphological features were microscopically examined for cytopathic effects, followed by the evaluation of virus titre via the focus-forming unit using culture supernatant. The phytochemical study revealed 24 bioactive chemicals, the most common of which was hexadecanoic acid eicosyl ester, accounting for 24.64% of the total. The maximum non-toxic dose was 62.5 µg/mL, with an IC50 of 420.67 µg/mL. Post-treatment showed the highest antiviral activity, reducing the viral titre from 8.10 (virus control) to 7.36 mean log10 FFU/mL, corresponding to 82.25% inhibition. The anti-inflammatory assay of the plant extract showed a percentage inhibition of 72.38% at 500 μg/ml, compared to the standard diclofenac sodium (75.14%). Medicinal plants and their bioactive compounds serve as a vital source of antimicrobial agents, offering promising alternatives for the development of natural therapeutics. This study demonstrates that Cynodon dactylon can effectively suppress CHIKV-induced infection and inflammation. Furthermore, hexadecanoic acid eicosyl ester has the potential to be developed as an anti-Chikungunya therapeutic candidate; however, it requires further validation in in vivo models.
Keywords
Antiviral; Anti-inflammatory; Chikungunya Virus; Cynodon dactylon; Cytotoxicity; FFU assay
Download this article as:| Copy the following to cite this article: Soniya K, Boora S, Yadav S, Bharti A. K Sharma M, Patil P, Alagarasu K, Parashar D, Kaushik S. Anti-Chikungunya and Anti-Inflammatory Therapeutic Potential of Cynodon dactylon Aqueous Extract. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Soniya K, Boora S, Yadav S, Bharti A. K Sharma M, Patil P, Alagarasu K, Parashar D, Kaushik S. Anti-Chikungunya and Anti-Inflammatory Therapeutic Potential of Cynodon dactylon Aqueous Extract. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/4t3pMoR |
Introduction
Chikungunya is a mosquito-borne Alphavirus belonging to the Togaviridae family, which was first identified in Africa between 1952 and 1953.1 It exhibits clinical manifestations such as severe arthritis, as well as back and joint pain, which may persist for weeks or even years.2 The term “Chikungunya” comes from the Makonde language, signifying “that which bends up,” which accurately describes the bent posture of patients afflicted with severe arthralgia.3 In 1958, the first Asian outbreak occurred in Bangkok, and followed in various Asian countries. In India, the first CHIKV epidemic was documented in 1963 in Kolkata, West Bengal, killing over 200 people, primarily children.4 After 32 years, CHIKV emerged and infected 13 states, including about 1.4 million people. Additionally, the outbreak had unexpected clinico-pathological complications such as CHIKV-related deaths and mother-to-child transmission.5 The global spread of CHIKV may increase the public health risk of Chikungunya. The extensive transmission could increase cases and outbreaks, highlighting the need for effective prevention, supervision, and management.
Chikungunya fever is primarily treated symptomatically, as there are currently no effective vaccines or antivirals available.6,7 Despite its global health impact, Chikungunya has no specific antiviral treatment. Patient care predominantly focuses on symptomatic treatment, including analgesics, anti-inflammatory drugs, and physiotherapy for joint pain. Despite clinical testing, no vaccine candidates have received regulatory approval for broad usage. The lack of effective treatments underscores the urgent need for the development of antiviral drugs. Medicinal plants and natural items are regarded as the sole method for therapies or proper treatment for various severe illnesses.8 C. dactylon, a popular perennial indigenous grass known as Bermuda grass, occurs naturally and ornamentally in tropical and humid temperate climates.9,10 Historically, Indian herbalists have used C. dactylon as an oral supplement against fevers, diabetes, ulcers, cardiac arrhythmia, diarrhea, gout, rheumatism, and chronic inflammatory conditions. C. dactylon, a member of the Poaceae family, is known for its antibacterial, antidiabetic, anti-hyperlipidemia, anti-inflammatory, and antiemetic properties.11,12 Furthermore, C. dactylon extract has been reported to have promising antiviral activity against shrimp’s white spot syndrome virus (WSSV) via in vitro and in vivo approaches.13 A previous study demonstrated that apigenin and luteonin ethanolic fractions from C. dactylon at 50 µg/mL suppressed 98% of CHIKV activity in Vero cells.42
The present research explores the in vitro antiviral potential of an aqueous extract of C. dactylon against CHIKV infection. The various chemical compositions of plant extracts may yield synergistic effects superior to those of single-compound methods, resulting in greater therapeutic potential.
Materials and Methods
Preparation of Plant Extract
The entire plant (C. dactylon) was obtained from the Maharshi Dayanand University campus in Rohtak, India, and authenticated by a university expert. The geographic coordinates of the collection site are latitude 29.670019°N and longitude 75.817738°E. The plant material was cleaned, air-dried, ground into powder, and 20 g was subjected to Soxhlet extraction using double-distilled water as solvent. The extract was filtered through Whatman filter paper No. 1, and the solvent was evaporated using a hot air oven at 45°C to yield a dried, concentrated crude extract, which was then preserved at -20°C until use.
Stock preparation of C. dactylon
For in vitro studies, a stock solution of C. dactylon (100 mg/ml) was prepared in DMSO (50%) and purified by filtration using a syringe filter (0.22 μm pore size). The working solution was prepared from stock in Minimum Essential Medium (MEM) at different concentrations ranging from 500 to 1.9 µg/mL.
Phytochemical Analysis
Fourier transform infrared (FTIR) spectroscopy.
The FTIR is an efficient technique for identifying and characterizing compounds or functional groups present in an unknown plant extract mixture. It is carried out within the range of 4000–400 cm-1. The obtained spectrum data were analyzed against reference graphs to evaluate the functional groups present in the extract.
Gas chromatography-mass spectrometry (GC–MS)
The Shimadzu GC-MS-QP2020 was used to identify volatile organic compounds in a plant extract sample. Helium, a carrier gas (1.0 mL/min flow rate), is used on the SH-Rxi-5Sil MS column (I.D.: 30 m × 0.25 mm; film thickness: 0.25 µm). A 1 μL volume was used to deliver a splitless infusion. The initial oven temperature was set at 40°C for 2 minutes, then subsequently raised (10°C/min) for 5 minutes, not exceeding 150°C, and then elevated (20°C/min) up to 250°C for 10 minutes. The ion source and interface temperatures were sustained at 230°C and 280°C, respectively. The mass spectrometer was configured to 70 eV in electron impact mode, scanning from 35 to 500 m/z (2000 amu/sec), with a solvent delay of 3 minutes. The NIST Library’s ionization spectra were used to identify the chemicals and to determine their area and proportion in the total composition.
In vitro analysis of plant extract
Cells and Viruses
Vero CCL-81 cells were used and cultured in MEM enriched with FBS (10 %, heat-inactivated) and 1% antimycotic antibiotic solution (Sigma Aldrich, US) under standard conditions (37°C, 5% CO2). The CHIKV strain was cultured in T-25 tissue culture flasks and subjected to two passages in Vero cells for virus propagation. Cell supernatants were collected after observing 75% CPE, then prepared into aliquots and stored at -80 °C. The viral titers were quantified via the Foci-forming assay.
In vitro Cytotoxicity Assays
The MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] test determines the cell viability.14,15 Briefly, vero CCL-81 cells (35×10³ cells/well) were treated with C. dactylon extracts (500-3.90 µg/mL) at two-fold serial dilutions in MEM for 48 hours in 5% CO2 at 37°C. Introduce MTT dye (10 µL; 5mg/mL in PBS) into respective wells and further incubate for 3 hours. Subsequently, the MTT solution was removed, and DMSO (100 µL) was added to dissolve the formazan crystals. Following 30 minutes of constant stirring, the absorbance was measured at 570 nm using a reference filter of 690 nm. The viability percentage was determined in contrast to untreated cells. The highest concentration of the plant extract that did not show cellular toxicity was determined as the Maximum Non-Toxic Dose (MNTD).
Antiviral assay
In order to evaluate the antiviral potential of aqueous C. dactylon extracts against Chikungunya virus (0.01 MOI), three methods were employed using Vero cells: pre-treatment (5 hours treatment with plant extract before virus infection), co-treatment (1 hour treatment of plant extract with virus before infection), and post-treatment (cells were infected before extract treatment). A virus control (VC) was maintained for all treatment conditions in untreated cells. Following 48 hours of incubation under each treatment condition, the morphology of cells was microscopically examined for cytopathic effects (CPE). Culture Supernatants were obtained after three freeze-thaw cycles and used to evaluate the virus particle titre via the focus-forming unit assay. All experiments were conducted in triplicate.
Foci-forming unit assay
The virus particles were quantified using the Foci-forming unit assay (FFU), as described by 16,17. Vero cells (35000 cells/well) were introduced in a 96-well plate and allowed to grow a monolayer. The cells were infected for approximately one hour using a tenfold dilution of the supernatants from the antiviral assay. Following incubation, the washed cells were cultured with 2% MEM containing 1.8% carboxymethyl cellulose and incubated for 24 hours at 37°C. The cells were rinsed with PBS supplemented with Tween 20 detergent, and then fixed with a chilled acetone and methanol solution at a 1:1 ratio. A blocking buffer (1% bovine serum albumin in PBS) was used for 40 minutes at 37 °C to block. After washing, cells were treated with anti-CHIKV monoclonal antibody (MAb ClVE4/D9 clone; 1:300 dilution) for 40 minutes. Following this, anti-mouse IgG HRP conjugate (1:1000) (Sigma-Aldrich, USA) was added, and the cells were incubated for an additional 40 minutes. Following the addition of True-Blue Peroxidase Substrate (LGC Sercare, USA) under dark conditions, the mixture was kept at room temperature for 15 minutes. Once the blue tinge developed, the foci were visualized and counted to determine the virus titer.
Anti-inflammatory Activity
The egg albumin denaturation assay determined the anti-inflammatory activity of C. dactylon extract.18 Prepare a 5 mL reaction mixture including various concentrations of plant extract (100-500 μg/ml), and the reference drugs (diclofenac sodium), 1 mL solution of bovine serum albumin (1%), and 4.78 mL of PBS (pH 6.4). The mixture was then incubated for 20 minutes at 37°C and subsequently incubated for 30 minutes at 70°C in a water bath to denature the BSA. The reaction mixture was then cooled to room temperature. Measured the absorbance at 660 nm and calculated protein denaturation activity based on the following formula:
Statistical Analysis
GraphPad Prism 10 (GraphPad Inc., USA) was used for the Statistical analyses. Multiple group comparisons were performed using one-way ANOVA, followed by Tukey’s post hoc test, whereas two-group comparisons were conducted using Student’s t-test. The threshold for statistical significance was established at p ≤ 0.05. All data are represented as mean ± SEM.
Results
Our research shows that C. dactylon aqueous extract exhibits potent antiviral activity against the Chikungunya virus, particularly displaying high efficacy in post-treatment administration.
Phytochemical Analysis
Fourier transform infrared (FTIR) spectroscopy.
The functional group of bioactive compounds of C. dactylon aqueous extract was determined using an FTIR spectrophotometer. Eight major peaks were observed for the FTIR analysis of C. dactylon, as presented in Figure 1. The IR spectrum showed an absorption band at 3850.36 cm-1, 3739.75 cm-1, 3675.08 cm-1, and 3611.88 cm-1 attributed to O-H stretching, 2356.68 cm-1 corresponds to C≡O triple bond vibration, 1744.12, and 1694.45 cm-1 correspond to C=O stretching, and 1520.05 cm-1 represents N-O asymmetric stretch. As represented in Table 1, these absorbance peak values have been correlated to the online standard infrared spectrum.
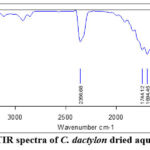 |
Figure 1: FTIR spectra of C. dactylon dried aqueous extract |
Table 1: FTIR spectrum analysis of C. dactylon aqueous extract
| S. No. | Wave number | Wave number range (cm-1) | vibration | Functional group |
| 1. | 3850.35 | 4000-3800 | O-H stretching and N-H stretching | – |
| 2. | 3739.75 | 3800-3700 | O-H stretching | Alcohols and phenols |
| 3. | 3675.08 | 3700-3600 | O-H stretching | Alcohols and phenols |
| 4. | 3611.88 | 3700-3600 | O-H stretching | Alcohols and phenols |
| 5. | 2356.68 | 3400-2300 | Triple bonds (C≡O) | – |
| 6. | 1744.12 | 1760-1690 | C=O stretching | δ-lactone, esters, cyclopentanone, |
| 7. | 1694.45 | 1700-1600 | C=O stretching | Amides, Ketones, Aldehydes, and Carbonyl |
| 8. | 1520.05 | 1650–1580 | N–O asymmetric stretching | Nitro Compounds |
Gas chromatography-mass spectrometry (GC-MS)
The result of the GC-MS revealed that the plant extracts recorded over 24 compounds, as shown in Figure 2. The bioactive compounds of the aqueous extract of C. dactylon were identified through GC/MS analysis and are listed, along with their retention times, area percentages, classes, molecular formulas, and molecular weights, in Table 2. The most abundant compound was hexadecanoic acid, eicosyl ester. According to a study, structural and kinetic investigations of n-hexadecanoic acid, which is responsible for inhibiting phospholipase A2 (an enzyme involved in inflammation), suggest that it has anti-inflammatory potential. This finding supports the conventional use of n-hexadecanoic acid-enriched medicinal oils in the Ayurvedic system of medicine for treating rheumatic conditions.19
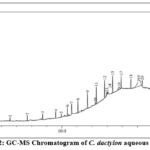 |
Figure 2: GC-MS Chromatogram of C. dactylon aqueous extract |
Table 2: Bioactive compounds found in C. dactylon aqueous extract
| Peak# | Retention Time | Area% | Compounds | Class | Molecular formula | Molecular weight (g/mol) |
| 1 | 6.566 | 1.35 | Cyclohexasiloxane, dodecamethyl- | organosilicon | C12H36O6Si6 | 444.92 |
| 2 | 7.709 | 2.20 | Cycloheptasiloxane, tetradecamethyl- | Cyclic Siloxane | C14H42O7Si7 | 519.07 |
| 3 | 8.720 | 2.21 | Cyclooctasiloxane, hexadecamethyl- | cyclic siloxane | C16H48O8Si8 | 593.2 |
| 4 | 9.589 | 2.45 | Cyclononasiloxane, octadecamethyl- | siloxanes | C18H54O9Si9 | 667.4 |
| 5 | 9.963 | 1.68 | Neophytadiene | diterpene | C20H38 | 278.5 |
| 6 | 10.360 | 2.95 | Cyclononasiloxane, octadecamethyl- | siloxanes | C18H54O9Si9 | 667.4 |
| 7 | 10.645 | 4.90 | Dibutyl phthalate | benzoic acid esters | C16H22O4 | 278.34 |
| 8 | 11.061 | 4.21 | Cyclooctasiloxane, hexadecamethyl- | cyclic siloxane | C16H48O8Si8 | 593.2 |
| 9 | 11.693 | 5.37 | Cyclononasiloxane, octadecamethyl | siloxanes | C18H54O9Si9 | 667.4 |
| 10 | 12.207 | 1.00 | Glycidyl palmitate | glycidyl ester | C19H36O3 | 312.5 |
| 11 | 12.275 | 5.75 | Cyclononasiloxane, octadecamethyl- | siloxanes | C18H54O9Si9 | 667.4 |
| 12 | 12.822 | 6.12 | Cyclononasiloxane, octadecamethyl- | siloxanes | C18H54O9Si9 | 667.4 |
| 13 | 12.957 | 1.66 | (R)-(-)-14-Methyl-8-hexadecyn-1-ol | fatty alcohols | C17H32O | 252.4 |
| 14 | 13.011 | 1.01 | Palmitic Acid, TMS derivative | organosilicon | C19H40O2Si | 328.6 |
| 15 | 13.354 | 6.37 | Cyclononasiloxane, octadecamethyl- | siloxanes | C18H54O9Si9 | 667.4 |
| 16 | 13.773 | 3.08 | 9,12-Octadecadienoic acid (Z, Z)-, TMS derivative | linoleic acid | C21H40O2Si | 352.6 |
| 17 | 13.923 | 5.76 | Cyclononasiloxane, octadecamethyl- | siloxanes | C18H54O9Si9 | 667.4 |
| 18 | 14.563 | 4.48 | Tetracosamethyl-cyclododecasiloxane | organometalloid | C24H72O12Si12 | 889.8 |
| 19 | 15.079 | 24.64 | Hexadecanoic acid, eicosyl ester | wax ester | C36H72O2 | 537 |
| 20 | 15.314 | 3.19 | Tetracosamethyl-cyclododecasiloxane | organometalloid | C24H72O12Si12 | 889.8 |
| 21 | 16.221 | 3.43 | Tetracosamethyl-cyclododecasiloxane | organometalloid | C24H72O12Si12 | 889.8 |
| 22 | 16.778 | 1.97 | Stigmast-5-en-3-ol, oleate | phytosterol | C47H82O2 | 679.2 |
| 23 | 17.355 | 2.52 | Tetracosamethyl-cyclododecasiloxane | organometalloid | C24H72O12Si12 | 889.8 |
| 24 | 18.774 | 1.70 | Tetracosamethyl-cyclododecasiloxane | organometalloid | C24H72O12Si12 | 889.8 |
In vitro Antiviral Screening
Cell Cytotoxicity Assays
Cell viability was evaluated using the MTT test to determine the cytotoxic effects of C. dactylon aqueous extract on Vero cells. The results of cell viability percentages are represented in Figure 3. The highest dose of extract that resulted in more than 90% cell viability was used for evaluating antiviral activity. Cell viability exceeded 90% at concentrations ≤ 62.5 µg/mL, with an estimated IC50 of 420.67 µg/mL. Further investigations used 62.5μg/mL concentrations, which proved non-toxic to cells. All tests with each concentration of plant extract and cell control were performed in triplicate.
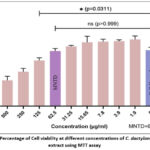 |
Figure 3: Percentage of Cell viability at different concentrations of C. dactylon aqueous extract using MTT assay |
Microscopic morphological screening of cytopathic effects
The antiviral effectiveness of C. dactylon extract in Vero cells was evaluated by microscopic morphological screening 48 hours post-infection to determine the CPE. The uninfected control cells formed a confluent monolayer of fibroblastic, elongated, spindle-shaped cells with evident intercellular boundaries. In contrast, virus-infected cells exhibited severe CPE, which resulted in multinucleated syncytia, cell rounding, and substrate detachment, leading to monolayer degradation. The pre-treatment cells exhibited syncytium development and cellular damage similar to those in the virus control. However, co-treatment indicated that the extract provided moderate protection. Post-treatment notably provided substantial cellular protection, maintaining nearly normal fibroblastic shape and inhibiting syncytia development. The extract successfully arrested the Chikungunya-induced cellular damage when supplied post-infection. Figure 4 illustrates the comparative CPE of various treatment conditions of C. dactylon extract against cell and viral controls, indicating that post-treatment is more efficient.
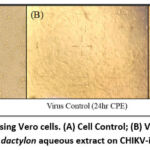 |
Figure 4: Post-treatment using Vero cells. (A) Cell Control; (B) Virus control showing CPE; (C) Effect of C. dactylon aqueous extract on CHIKV-infected cells. |
Effect of C. dactylon extract on CHIKV replication
CHIKV-infected vero cells were treated with MNTD (62.5 μg/ml) of the extract under pre (prophylactic effect), co (virucidal activity), and post (therapeutic effect) treatment. The results revealed that the extract was effective only under post-treatment conditions at a concentration of 62.5 µg/ml. A graph for anti-CHIKV potential at maximum non-toxic dose under post-treatment condition, compared with the virus control shown in Figure 5. Following the antiviral test, the FFU assay was conducted 48 hours later to determine the virus titer. In the FFU assay, the treatment of the cells 4 hours post-infection with extract at a 62.5 μg/ml concentration resulted in foci reduction from 8.10 (Virus control) to 7.36 Mean log10 FFU/ml (82.25% inhibition in virus titre; p = 0.005).
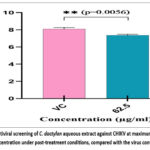 |
Figure 5: Antiviral screening of C. dactylon aqueous extract against CHIKV at maximum non-toxic concentration under post-treatment conditions, compared with the virus control. |
In Vitro Anti-inflammatory Activity
The anti-inflammatory effects of C. dactylon aqueous extract were determined using the egg albumin denaturation assay, performed in triplicate. The extract showed dose-dependent (100-500 μg/ml) inhibition between 25.90% and 72.38%, with an IC50 of 2.4 μg/ml compared to diclofenac sodium (a standard drug), which exhibited 38.64% to 75.14% inhibition with an IC50 of 3.2 μg/ml, as shown in Figure 6. The results indicate the strong anti-inflammatory potential of the plant extract in contrast to diclofenac, suggesting its therapeutic capability as a natural anti-inflammatory candidate.
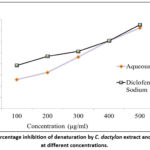 |
Figure 6: Percentage inhibition of denaturation by C. dactylon extract and diclofenac at different concentrations. |
Discussion
In recent years, significant viral infections like influenza,20,21 hepatitis,22 COVID-19,23 Zika,24 Nipah,25 Marburg,26 Monkeypox,27 dengue,28 Chikungunya, and others have also raised serious public health concerns. Chikungunya fever is a global public health challenge in tropical and subtropical countries. Currently, there are no licensed medications or vaccines effective against Chikungunya. Mosquito management is the primary preventive strategy for CHIKV. A wide range of drug candidates has been evaluated for anti-CHIKV activity, and the search for effective therapies is ongoing due to safety concerns associated with the expedited vaccination licensing procedure and potential long-term adverse consequences.29 Chemically produced antivirals have a wide range of adverse effects, in contrast to traditional therapies.30 The developments of plant-based medicine are being researched and developed, offering many potential uses and benefits.31,32 The emergence of viral diseases required the exploration of alternative therapeutic approaches. A broad spectrum of traditional medicinal plants and herbs demonstrating significant antiviral potential against various pathogenic viruses, including influenza,33,34 dengue,35,36 Chikungunya,37,38,39, and COVID-19.23,40 Therefore, plant-based anti-Chikungunya medications may function as an alternative therapy for mosquito-transmitted diseases. Several plant extracts exhibit strong antiviral properties against CHIKV, such as silymarin from Silybum marianum, which inhibits viral replication. Additionally, Ipomoea aquatica and Persicaria odorata also inhibit viral replication. Other species, such as Tinospora cordifolia and Andrographis paniculata prevent genome replication, while Tradescantia spathacea and Rhapis excelsa demonstrate direct virucidal effects. Additional plants, including Ocimum tenuiflorum, Zingiber officinale, Picrorhiza kurroa, Terminalia chebula, Cedrus deodara and Commiphora wightii also disrupt CHIKV replication or attachment. Key active compounds identified are andrographolides and nobiletin, with the polyherbal formulation Nilavembu kudineer showing significant antiviral potential.41 Certain isolated flavonoids, such as silymarin, baicalein, nobiletin, quercetagetin, and fisetin, exhibited strong anti-CHIKV action.42 The ethyl acetate extracts of Stillingia lineata having highest efficiency against CHIKV, suggesting its potential as a natural antiviral candidate.43 According to the Ayurvedic medicine system, C. dactylon is employed as a traditional remedy to treat a wide range of health problems, including cough, diarrhea, hemorrhage, bronchitis, dysentery, cramps, epilepsy, measles, piles, dropsy, cystitis, convulsions, calculus, warts, vitiligo, high blood pressure, hysteria, asthma, cancer, tumors, sores, anasarca, rubella, snakebites, carbuncles, stones, urogenital problems, wounds, eye problems, weak eye sight, headache, and dermatitis. It also effectively alleviates pain, inflammation, dental discomfort, and influenza in youngsters. The astringent properties of the extracted plant juice are used to stop bleeding from cuts and wounds. A combination of leaf paste and honey is beneficial against nosebleeds. Doses of plant extract mixed with honey, taken two to three times a day, can effectively treat heavy menstrual bleeding. Acute vaginal hemorrhage is alleviated by applying a thin layer of a plant extract-based paste to the lower abdomen. A combination of C. dactylon and sugar works well in the treatment of urine retention.44
In this research, we examined the anti-CHIKV potential of the aqueous extract of C. dactylon to identify CHIKV targets and pathways for developing novel traditional antivirals. Numerous phytochemicals are responsible for its antiviral action and other therapeutic qualities.45 Phytochemical research using FTIR spectroscopy revealed that the diverse bioactive chemicals, including phenols, alkaloids, flavonoids, carboxylic acids, and other organic compounds, may be responsible for its biological action. The GC-MS chromatogram displays 24 peaks. The most-abundant compound identified in the GC-MS analysis was hexadecanoic acid eicosyl ester (24.64%). It exhibits a significant anti-inflammatory property and is used in Ayurvedic medicine to treat rheumatic conditions.19
The in vitro anti-chikungunya activity of C. dactylon plant extract was investigated using Vero cells. To evaluate its potential adverse effects, it is necessary to conduct an in vitro evaluation of the maximum non-toxic dose of the aquatic medicinal plant extract before its therapeutic use. Aqueous medicinal plant extract displays a cytopathic effect on Vero cells. The MTT assay demonstrated the MNTD of C. dactylon aqueous extract, which is non-toxic to Vero cells, showing cell viability above 90% at concentrations up to 62.5 µg/mL, with an estimated IC50 value of 420.67 µg/mL. The concentration of DMSO in the working solution of plant extract was less than 1%, which is considered safe for the cells and does not cause cytotoxicity.46
The three different antiviral approaches showed different cytopathic effects on Vero cells. The extract’s protective properties were demonstrated through a microscopic morphological examination of CPE. The co-treatment and pre-treatment showed minimal protection to Vero cells. The post-treatment with C. dactylon extract effectively prevented the formation of multinucleated syncytia and preserved the nearly normal fibroblastic morphology in virus-infected control cells. This pattern indicates that the extract primarily disrupts active viral replication rather than providing prophylactic protection, highlighting its therapeutic potential. The quantitative antiviral results, which correlate with the cellular protection, further confirm the extract’s potential for medical use. The antiviral results from post-treatment were further confirmed by the FFU assay, which measures functional infectious virus titre. Post-treatment has the most effective antiviral action (82.25% reduction in FFU titre). The anti-inflammatory activity is assessed using the egg albumin denaturation assay. The extract exhibited 72.38% inhibition at 500 μg/mL, with an IC50 of 2.4 μg/mL, compared to diclofenac sodium (75.14%; IC50: 3.2 μg/mL). Chikungunya induces debilitating arthralgia due to viral replication and substantial inflammatory reactions; hence, this antiviral and anti-inflammatory combination is necessary for treatment.
Combining antiviral and anti-inflammatory activities in a single extract signifies a therapeutic benefit. By targeting both viral replication and its inflammatory responses, C. dactylon extract may provide a comprehensive treatment approach against Chikungunya fever. This dual-action mechanism is consistent with the traditional use of medicinal plants, which often contain multiple bioactive compounds that work together to act as antiviral agents against complex viral diseases.
Conclusion
Chikungunya is a mosquito-borne disease that causes severe joint pain and inflammation, resulting in long-term disability. There are currently no widely approved antiviral drugs or vaccines available for the effective treatment of CHIKV infection. Patient management is largely symptomatic, relying on general analgesics, anti-inflammatory medications, and supportive care to relieve joint pain and fever. The current research highlights the antiviral and anti-inflammatory effects of C. dactylon aqueous extract against the chikungunya virus (CHIKV) and its potential uses in medicine. The ability of extract to reduce virus titer by 82% and its anti-inflammatory activity make it a promising candidate for developing natural antiviral therapies. Hexadecanoic acid helps alleviate rheumatological symptoms by reducing the severity of inflammation associated with CHIKV. These findings could have a significant impact on the management of Chikungunya fever and other viral diseases, particularly in areas where these infections are endemic and medical resources are limited.
Acknowledgement
Kumari Soniya & Sanjit Boora acknowledge the Council of Scientific & Industrial Research (CSIR), New Delhi, for providing a Senior Research Fellowship.
Funding Sources
The author(s) received no financial support for this article’s research, authorship, and/or publication.
Conflict of Interest
The author(s) declare no conflict of interest or competing interest related to this research.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve any studies with human participants or animals performed by any authors.
Informed Consent Statement
This study did not involve human participants, so informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable
Authors Contributions
- Kumari Soniya: Drafted the manuscript and experimental work.
- Sanjit Boora, Suman Yadav, and Manisha Sharma are drafting and reviewing the manuscript.
- Poonam Patil, Abhishekh Kumar Bharti: protocol drafting, data collection, and analysis.
- Kalichamy Alagarasu, Deepti Parashar, and Samander Kaushik: Conceptualization and manuscript drafting.
References
- Higgs S, Vanlandingham D. Chikungunya virus and its mosquito vectors. Vector-Borne and Zoonotic Dis. 2015;15(4):231-240.
CrossRef - Schwartz O, Albert ML. Biology and pathogenesis of chikungunya virus. Nat Rev Microbiol. 2010;8(7):491-500.
CrossRef - Peper SM, Monson BJ, Van Schooneveld T, Smith CJ. That which bends up: a case report and literature review of chikungunya virus. J Gen Intern Med. 2016;31(5):576-581.
CrossRef - Naresh Kumar CV, Sai Gopal DV. Reemergence of Chikungunya virus in Indian subcontinent. Indian J Virol. 2010;21(1):8-17.
CrossRef - Sudeep AB, Parashar D. Chikungunya: an overview. J Biosci. 2008;33(4):443-449.
CrossRef - Cunha RV, Trinta KS. Chikungunya virus: clinical aspects and treatment-A Review. Memórias do Instituto Oswaldo Cruz. 2017;112(8):523-531.
CrossRef - Soniya K, Yadav S, Boora S, Kaushik S, Yadav JP, Kaushik S. The Cat Que Virus: a resurfacing orthobunyavirus could lead to epidemics. 2021;32(4):635-641.
CrossRef - Dahiya H, Sonia K, Boora S, et al. Trachyspermum ammi (Ajwain): A sacred plant with high medicinal and therapeutic potential. Res J Pharm Technol. 2023;16(7):3285-3288.
CrossRef - Garjani A, Afrooziyan A, Nazemiyeh H, Najafi M, Kharazmkia A, Maleki-Dizaji N. Protective effects of hydroalcoholic extract from rhizomes of Cynodon dactylon (L.) Pers. on compensated right heart failure in rats. BMC Complement Altern Med. 2009;9(1):28.
CrossRef - Auddy B, Ferreira M, Blasina F, et al. Screening of antioxidant activity of three Indian medicinal plants, traditionally used for the management of neurodegenerative diseases. J Ethnopharmacol. 2003;84(2-3):131-138.
CrossRef - Singh SK, Kesari AN, Gupta RK, Jaiswal D, Watal G. Assessment of antidiabetic potential of Cynodon dactylon extract in streptozotocin diabetic rats. J Ethnopharmacol. 2007;114(2):174-179.
CrossRef - Immanuel G, Uma RP, Iyapparaj P, et al. Dietary medicinal plant extracts improve growth, immune activity and survival of tilapia Oreochromis mossambicus. J Fish Biol. 2009;74(7):1462-1475.
CrossRef - Balasubramanian G, Sarathi M, Venkatesan C, Thomas J, Sahul Hameed AS. Studies on the immunomodulatory effect of extract of Cyanodon dactylon in shrimp, Penaeus monodon, and its efficacy to protect the shrimp from white spot syndrome virus (WSSV). Fish Shellfish Immunol. 2008;25(6):820-828.
CrossRef - Bruggisser R, von Daeniken K, Jundt G, Schaffner W, Tullberg-Reinert H. Interference of plant extracts, phytoestrogens and antioxidants with the MTT tetrazolium assay. Planta medica. 2002 May;68(05):445-8.
CrossRef - Parashar D, Paingankar MS, Kumar S, Gokhale MD, Sudeep AB, Shinde SB. Administration of E2 and NS1 Sirnas Inhibit Chikungunya Virus Replication In Vitro and Protects Mice Infected With the Virus. PLoS Negl Trop Dis. 2013;7(9):e2405.
CrossRef - Patil P, Agrawal M, Almelkar S, Jeengar MK, More A, Alagarasu K. In vitro and in vivo studies reveal a-mangostin, a xanthonoid from garcinia mangostana, as a promising natural antiviral compound against chikungunya virus. Virol J. 2021;18(1):47.
CrossRef - Panda K, Alagarasu K, Patil P, et al. In Vitro Antiviral Activity of α-Mangostin against Dengue Virus Serotype-2 (DENV-2). 2021;26(10):3016.
CrossRef - Abbad S, Aissaoui N, Gana FZ. Marrubium vulgare Leaf Extract-Assisted Green Synthesis of Silver Nanoparticles: Optimization, Characterization and In Vitro Exploration of Their Antimicrobial, Antioxidant and Anti-Inflammatory Potential. Pharmacogn J. 2025;17(1).
CrossRef - Aparna V, Dileep KV, Mandal PK, Karthe P, Sadasivan C, Haridas M. Anti‐inflammatory property of n‐hexadecanoic acid: structural evidence and kinetic assessment. Chem Biol Drug Des. 2012;80(3):434-439.
CrossRef - Hom GG, Chous AP. The prospect of pandemic influenza: Why should the optometrist be concerned about a public health problem?. Optometry- J Am Optom Assoc. 2007;78(12):629-643.
CrossRef - Garcia-Garcia J, Ramos C. Influenza, an existing public health problem. Salud publica Mex. 2006;48(3):244-267.
- Boora S, Sharma V, Kaushik S, Bhupatiraju AV, Singh S, Kaushik S. Hepatitis B virus-induced hepatocellular carcinoma: a persistent global problem. Braz J Microbiol. 2023;54(2):679-689.
CrossRef - Ayenigbara IO. COVID-19: an international public health concern. Cent Asian J Glob Health. 2020;9(1):e466.
CrossRef - Demir T, Kilic S. Zika virus: a new arboviral public health problem. Folia Microbiol. 2016;61(6):523-527.
CrossRef - Cui Z, Li J, Meng L, Zhang Z. Nipah virus: a re-emerging public health concern. The Lancet Microbe. 2024;5(3):e212.
CrossRef - Mujahid U, Ahmad M, Mujahid A, Narayan E, Rehman SU, Iqbal HM, Ahmed I. Recent outbreak of Marburg virus; a global health concern and future perspective. Eur J Clin Microbiol Infect Dis. 2024;43(1):209-211.
CrossRef - Boora S, Yadav S, Soniya K, et al. Monkeypox virus is Nature’s wake-up call: a bird’s-eye view. 2023;34(2):191-203.
CrossRef - Vijayakumar TS, Chandy S, Sathish N, Abraham M, Abraham P, Sridharan G. Is dengue emerging as a major public health problem. Indian J Med Res. 2005;121(2):100-107.
- US Food and Drug Administration. FDA Approves First Vaccine to Prevent Disease Caused by Chikungunya Virus. 2023. https://www.fda.gov/news-events/press-announcements/fda-approves-first-vaccine-prevent-disease-caused-chikungunya-virus
- Goncalves BC, Lopes Barbosa MG, Silva Olak AP, et al. Antiviral therapies: advances and perspectives. Fundam Clin Pharmacol. 2021;35(2):305-320.
CrossRef - Sharma R, Bhattu M, Tripathi A, et al. Potential medicinal plants to combat viral infections: A way forward to environmental biotechnology. Environ Res. 2023;227:115725.
CrossRef - Najmi A, Javed SA, Al Bratty M, Alhazmi HA. Modern approaches in the discovery and development of plant-based natural products and their analogues as potential therapeutic agents. 2022;27(2):349.
CrossRef - Akram M, Tahir IM, Shah SM, et al. Antiviral potential of medicinal plants against HIV, HSV, influenza, hepatitis, and coxsackievirus: A systematic review. Phytother Res. 2018;32(5):811-822.
CrossRef - Rajasekaran D, Palombo EA, Chia Yeo T, et al. Identification of traditional medicinal plant extracts with novel anti-influenza activity. PloS one. 2013;8(11):e79293.
CrossRef - Kaptein SJ, Neyts J. Towards antiviral therapies for treating dengue virus infections. Curr opin pharmacol. 2016;30:1-7.
CrossRef - Lim SY, Chieng JY, Pan Y. Recent insights on anti-dengue virus (DENV) medicinal plants: review on in vitro, in vivo and in silico All Life. 2021;14(1):1-33.
CrossRef - Dhama K, Karthik K, Khandia R, et al. Medicinal and therapeutic potential of herbs and plant metabolites/extracts countering viral pathogens-current knowledge and future prospects. Curr drug metab. 2018;19(3):236-263.
CrossRef - Pebam M, Sushma MV, Sankaranarayanan SA, Thanekar AM, Koyande N, Rengan AK. Antiviral perspectives of economically important Indian medicinal plants and spices. Proc of the Indian National Science Academy. Indian Natl. Sci. Acad. 2022;88(3):392-416.
CrossRef - Soniya K, Boora S, Yadav S, Sharma M, Miglani M, Kaushik S. A review of Cassia fistula Linn potential uses in medicine and health management. World Journal of Biology Pharmacy and Health Sciences. 2025;22(2):480-508.
CrossRef - Adhikari B, Marasini BP, Rayamajhee B, et al. Potential roles of medicinal plants for the treatment of viral diseases focusing on COVID‐19: A review. Phytother Res. 2021;35(3):1298-1312.
CrossRef - Raghavendhar S, Tripati PK, Ray P, Patel AK. Evaluation of medicinal herbs for anti-CHIKV activity. 2019;533:45-49.
CrossRef - Kumar S, Garg C, Kaushik S, Buttar HS, Garg M. Demystifying therapeutic potential of medicinal plants against chikungunya virus. Indian J. Pharmacol.2021;53(5):403-411.
CrossRef - Millsapps EM, Underwood EC, Barr KL. Development and application of treatment for chikungunya fever. Res Rep Trop Med. 2022;13:55.
CrossRef - Chandra DM, Shama S, Satish C. Overview of Cynodon dactylon (Doob Grass) in modern medicine as an antidiabetic herb. J Drug Deliv Ther. 2013;3(6):117-120.
CrossRef - Murali KS, Sivasubramanian S, Vincent S, et al. Anti-Chikungunya activity of luteolin and apigenin rich fraction from Cynodon dactylon. Asian Pac J Trop Med. 2015;8(5):352-358.
CrossRef - Siddiqui MS, Giasuddin M, Chowdhury SM, Islam MR, Chowdhury EH. Comparative effectiveness of Dimethyl Sulphoxide (DMSO) and Glycerol as cryoprotective agent in preserving Vero cells. Bangladesh Vet. 2015;32(2):35-41.
CrossRef
Abbreviations List
CHIKV: Chikungunya Virus; DMSO: dimethyl sulfoxide; MEM: minimum essential medium; FTIR: Fourier transform infrared; GC-MS: Gas chromatography-mass spectrometry; NIST: National Institute of Standards and Technology; FBS: Fetal bovine serum; CPE: cytopathic effect; PBS: phoshphate buffer saline; MNTD: maximum Non-Toxic Dose; MOI: multiplicity of Infection; FFU: foci forming unit; BSA: Bovine Serum Albumin; ANOVA: analysis of variance; IC50: half maximal inhibitory concentration







