Manuscript accepted on :13-01-2026
Published online on: 28-01-2026
Plagiarism Check: Yes
Reviewed by: Dr. Manikandan Samidurai
Second Review by: Dr. Abeer Gatea
Final Approval by: Dr. Kamal Upreti
Deniz M. Al-Tawalbeh1* , Wissam Al-momani2
, Wissam Al-momani2 , Amir Majdi Moh'd2
, Amir Majdi Moh'd2 , Abdelrahman Alenaizat2
, Abdelrahman Alenaizat2 , Anas Majdi Moh’d2
, Anas Majdi Moh’d2 and Bsher Mfarrej2
and Bsher Mfarrej2
1Department of Medicinal Chemistry and Pharmacognosy, Faculty of Pharmacy, Yarmouk University, Irbid, Jordan
2Faculty of Medicine, Yarmouk University, Irbid, Jordan
Corresponding Author E-mail:deniz.altawalbeh@yu.edu.jo
Abstract
Parkinson’s Disease (PD), is a neurodegenerative disease marked by motor and non-motor symptoms, with high evidence highlighting the critical role of the gut-brain axis in the disease development. Helicobacter pylori (H. pylori), a typical gastric pathogen, has been identified as a contributor to Parkinson’s Disease. Changes in H. pylori infection to gut microbiota may cause systemic inflammation and nutrient malabsorption in addition to levodopa absorption impairment which may exacerbate PD pathogenesis and worsen symptoms. This narrative review explores the correlation between H. pylori infection and PD, to bring new insight into both diseases’ pathophysiology and focus on novel therapeutic options that can inhibit H. pylori growth, enhance intestinal integrity, and influence neurodegeneration in PD. However, it is constrained by rigorous quality assessment and may be subjected to selection bias. A comprehensive research investigating the link between H. pylori and PD was the focus of an extensive literature search that spanned the years 2005–2024 and was carried out using PubMed, Scopus, Web of Science, and Google Scholar. We combed through new information on treatments based on microbiota with relevant clinical trials, meta-analyses, and models of experimentation. H. pylori infection is common in PD patients and may play a role in systemic inflammation, small intestinal bacterial overgrowth (SIBO), reduced levodopa bioavailability, and elevated α-synuclein expression. The eradication of H. pylori has shown beneficial impacts on motor symptoms and enhanced the efficacy of levodopa. Herbal compounds including epigallocatechin gallate, baicalein, curcumin, and garlic derivatives exhibit notable antimicrobial and neuroprotective properties. Synbiotic interventions and personalized nutrient supplementation enhance gastrointestinal and neurological health. H. pylori infection in PD is a unique therapeutic target for gastrointestinal and neurological therapy. Antimicrobial therapy, dietary management, and phytomedicine may reduce inflammation, restore gut-brain balance, and enhance PD treatment outcomes. To improve patient-specific therapies, longitudinal studies and bioinformatics-guided microbiome analyses should be considered.
Keywords
Gut-brain axis; Helicobacter pylori; Herbal remedies; Levodopa; Parkinson’s disease
| Copy the following to cite this article: Al-Tawalbeh D. M, Al-momani W, Moh'd A. M, Alenaizat A, Moh’d A. M, Mfarrej B. The Convergence of Parkinson’s Disease and Helicobacter pylori Infection: Review Article. Biomed Pharmacol J 2025;18(October Spl Edition). |
| Copy the following to cite this URL: Al-Tawalbeh D. M, Al-momani W, Moh'd A. M, Alenaizat A, Moh’d A. M, Mfarrej B. The Convergence of Parkinson’s Disease and Helicobacter pylori Infection: Review Article. Biomed Pharmacol J 2025;18(October Spl Edition). Available from: https://bit.ly/49Qqb52 |
Introduction
Parkinson’s disease is a chronic, progressive neurological disorder characterized by motor deficits including tremor, bradykinesia, muscle rigidity, and postural instability.1 These clinical manifestations result from the degradation of dopaminergic neurons in the substantia nigra pars compacta, resulting in dopamine insufficiency in the striatum.2 In addition to its well-known motor symptoms, Parkinson’s disease includes a wide range of non-motor signs such as cognitive decline, mood changes, sleep difficulties, autonomic dysfunction, and gastrointestinal (GI) abnormalities, all of which can considerably impact patients’ quality of life.3
Although the exact cause of Parkinson’s disease remains a mystery, it is thought to be a complicated interaction of genetic vulnerability and environmental variables.4 In recent years, there has been an increased focus on the involvement of the gastrointestinal system, particularly the gut-brain axis—a bidirectional communication network that connects the central nervous system (CNS) to the enteric nervous system and the gut microbiome.5 Gut dysbiosis and inflammation are linked to neuroinflammation, α-synuclein aggregation, and dopaminergic neurodegeneration.6
Helicobacter pylori (H. pylori) infection has emerged as a potentially significant environmental component in the pathophysiology of Parkinson’s disease.7 Helicobacter pylori is a gram-negative, helical bacterium that inhabits the stomach mucosa and is recognized for its association with peptic ulcers and gastric carcinoma.8 Numerous clinical and epidemiological investigations have indicated a greater frequency of H. pylori infection in patients with Parkinson’s disease compared to the general population.9 Proposed pathways connecting H. pylori to PD encompass systemic inflammation, 10 compromised levodopa absorption,11 modification of gut microbiota composition,7 and modified gut barrier integrity.12
This review examines the relationship between Parkinson’s disease and H. pylori infection through an analysis of epidemiological data, common pathophysiological methods, and therapeutic implications. The influence of gut microbiota, herbal and pharmacological interventions, and emerging strategies concentrating on the gut-brain axis is emphasized as a novel approach in the management of Parkinson’s disease.
Methodology
This narrative review aims to investigate the potential correlation between Helicobacter pylori infection and Parkinson’s disease, focusing on the gut-brain axis, inflammatory mechanisms, treatment obstacles, and herbal therapies. A thorough literature search was conducted across many databases, including PubMed, Scopus, Web of Science, and Google Scholar, covering publications from 2005 to 2024.
As this is a narrative review; formal systemic review protocol or quality appraisal tool was not utilized. This limitation may lead to selection bias potentially affecting the interpretation and robustness of the conclusions. However, future systematic reviews may asses the causal relationship and therapeutic implications more rigorously.
The subsequent keywords and combinations utilized include: “Parkinson’s disease”, “Helicobacter pylori”, “gut-brain axis”, “levodopa absorption”, “neuroinflammation”, “herbal medicine”, “synbiotics”, and “dopaminergic neurodegeneration”. The use of Boolean operators, including AND, OR, and NOT, was implemented to enhance the precision of search results.
Articles that have undergone peer review are published in English, clinical trials, meta-analyses, observational studies, and in vivo or in vitro experimental studies, beside research studies examining the mechanistic connections between H. pylori infection and Parkinson’s disease or its treatment were included in this review.
Exclusion criteria included:
Publications in languages other than English, studies lacking accessibility for full text and papers that do not demonstrate relevance to either Parkinson’s Disease or Helicobacter pylori.
Multiple authors (W.A., A.M.M., A.A.) independently evaluated articles for relevance by reviewing titles and abstracts. The full texts of possibly eligible studies were examined, and data were extracted concerning study type, population characteristics, key findings, and conclusions. Any disagreements were addressed through discussions with the principal investigator (D.A.).
The quality appraisal was not properly performed owing to the review’s narrative character. Preference was accorded to studies exhibiting robust methodological design, substantial sample numbers, and recent publication dates. Reference management software, such as EndNote, was utilized to organize and reference sources throughout the manuscript.
Overview and Epidemiology of Parkinson’s Disease in Relation to Helicobacter pylori
Parkinson’s disease is a progressive condition that impacts the neurological system as well as the bodily components that receive nerve impulse. This disease is brought on by degeneration or loss of nerve cells in the basal ganglia; a movement regulation region of the brain. Normally, these neurons—nerve cells—produce dopamine, an essential neurotransmitter in the brain, 13 leading to tremors, stiffness, and slowed mobility. Rigid muscles, impaired posture and loss of automatic movements, along with speech changes, and difficulties writing are additional symptoms can cause pain and make daily tasks more challenging. 14
PD is a relatively common disease; in 2019 approximately 8.5 million cases were reported worldwide. 15 This disease affects 1-2 per 1000 individuals at any time. The prevalence of the disease is increasing with age affecting 1% of the population above 60 years. 16 This neurological illness – which is the second most prevalent disease – is predicted to double over the next 30 years. 16
In 2019, the MENA region reported around 309.9 thousand cases of Parkinson’s disease, with an age-standardized prevalence rate of 82.6 per 100,000 people. 17 Since 1990, the prevalence rate in the MENA region has risen by 15.4%, highlighting a growing health concern in this area. 18
There are many etiological elements that contributes to Parkinson’s Disease as inherited and environmental factors. Smoking tendency, pesticides use as well as physical exercise are risk factors and preventive factors associated with PD. However, it is challenging to identify the culprit due to the prodromal phase’s duration. 19
A mix of unknowns, genetic and environmental variables are associated to the disease. Autosomal or recessive dominants may account for 10-20 % of cases from genetic origin. 20 The inheritance pattern of Parkinson’s disease may vary depending on the mutated gene, however the majority of diagnosed people have no family history. The development of PD is altered by genetics, but the inheritance pattern is typically unclear. 21
Environmental factors such as exposure to pesticides, heavy metals, detergents, and solvents, have been studied heavily to demonstrate the unambiguous link to PD even if idiopathic factors as age and gender predominate and among the main risk factors. On the other hand, head trauma, which involves repeated blows to the head, may not be a direct cause for Parkinson’s Disease, but it may raise the risk of developing the disease. Overall deeper understanding to the disease to discover the related factors is now more feasible because of the continuous genomic findings.22
Some of the most important genetic and environmental pathways that are involved in the pathophysiology of Parkinson’s disease are summarized in Figure 1.
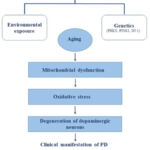 |
Figure 1. A schematic summery outlining the main causes of Parkinson’s disease. |
Helicobacter pylori a frequent bacterium that infects the stomach, and the primary etiology of peptic ulcer disease which may affect more than half of the world’s population.23 The prevalence of H. pylori infection among the MENA region countries vary from 7–50% in young children and up to 36.8–94% in adults. 10 Although the crude global prevalence of H. pylori infection among adults fell from 52.6% to 43.9% in 2022, it was still high in children and adolescents at 35.1% (95% CI, 30.5%–40.1%).24
In contrast to healthy control groups and according to several case-control studies, patients with Parkinson’s Disease had three times the frequency of positive results on an H. pylori test, and over the age of 80, the prevalence of H. pylori antibody was five-fold higher.25
After referring to pertinent papers published between January 1983 and January 2017, a comprehensive search of the PubMed, Web of Science, and EMBASE databases was carried out. The fixed or random effects model was used to pool the odds ratio from each independent study with a 95% confidence interval, and the funnel plot and Egger’s test were used to calculate publication bias.26 A total of 33,125 participants from eight different trials were included. According to the results; individuals with H. pylori infection were more prone to develop Parkinson’s disease than individuals without the infection. Overall, those carrying the bacterium are 1.6 times more likely to acquire Parkinson’s disease. Additionally, the risk in Europe was 1.5 times higher than in Asia. Regardless of whether or not additional influencing variables were considered during the studies has no bearing on the elevated risk.26
This may be explained by the secretion of pro-inflammatory cytokines, which are facilitated by H. pylori autotoxin, and lead to the development of central inflammation and consequent neuronal damage. 10 By generating small intestinal bacterial overgrowth (SIBO) or an overabundance of bacteria in the small intestine, H. pylori disturbs the balance of the gut microbiota. This dysbiosis of the gut flora affects the CNS through the microbiome-gut-brain axis. 27 Moreover, SIBO impairs the effectiveness of levodopa as a treatment of Parkinson’s disease as illustrated in Figure 2. Additionally, H. pylori stimulates the synthesis of defensins to control the blood-brain barrier’s (BBB) permeability, which makes it easier for pathogens to enter the central nervous system.28
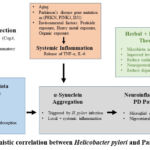 |
Figure 2: Mechanistic correlation between Helicobacter pylori and Parkinson’s disease. |
H. pylori infected patients who have gastroparesis may experience diminished efficacy of levodopa due to the longer transit time needed for levodopa to enter the small intestine. Conversely, levodopa may either promote the proliferation and cell growth, or damage gastrointestinal mucosa leading to gastrointestinal ulcers.10
Patients with Parkinson’s disease having H. pylori infection have lower motor functions than those who do not. With H. pylori eradication motor skills and levodopa absorption could be improved in Parkinson’s Disease patients.29
S1 Table presents a summary of significant preclinical, clinical studies and reviews investigating the correlation between H. pylori infection and Parkinson’s disease, encompassing processes, outcomes, and therapies included in supplementary file.
Current Medicinal Treatments for Parkinson’s Disease and Helicobacter pylori infection
Dopamine is the corner stone of Parkinson’s disease patients’ treatment. Beside dopamine other medications may be included within the regimen:
Levodopa
Restores levels of dopamine. It crosses the BBB and converted to dopamine by the presence of aromatic amino acid decarboxylase enzyme. Dopamine transporters absorb dopamine from drugs and store the exogenous dopamine from taken medications to maintain constant levels of dopamine in synaptic vesicle.30
Catechol-O-methyltransferase (COMT) Inhibitors:
Responsible for the inhibition of COMT enzyme. By preventing the methylation of catechol group containing substrates they lower levodopa levels that enters the brain by transforming it into 3-O-methyldopa. With the aid of COMT inhibitor; levodopa’s O-methylation is inhibited and converted into dopamine in the central nervous system. However, the half-life of the most frequently employed COMT inhibitors are only two to three hours.31
Monoamine Oxidase-B (MAO-B) Inhibitors:
Increase the quantity of dopamine in the synapses, thus, boosting dopamine signaling capacity blocking the MAO-B activity. When MAO-B receptor metabolizes dopamine; two harmful compounds are produced: Dihydroxyacetone and hydrogen peroxide. By preventing this; MAO-B inhibitor medicines stop this synthesis preserving dopaminergic neurons in the substantia nigra and delaying the onset of PD.31
Dopamine Agonists
The first-choice for PD patients under the age of 60 that postpone the onset of motor dysfunctions. Dopamine agonists are suggested by many authors to be used as a first line treatment. Nevertheless, dopamine agonist medications are unhelpful for patients who do not respond to levodopa.32
On the other hand, patients infected with H. pylori may benefit from a regimen comprising at least three antibiotics; containing proton pump inhibitor (PPI), amoxicillin (AMX), and clarithromycin (CLR) or metronidazole (MTZ), and to achieve the best H. pylori eradication results, this regimen should last 10 to 14 days.33 If the triple therapy proves ineffective Bismuth-Based Quadruple Therapy (BQT) consisting of a PPI, bismuth subcitrate, metronidazole, and tetracycline, serves as a treatment option for clarithromycin-resistant H. pylori infection. BQT is recommended as the first-line treatment option with high local clarithromycin and metronidazole resistance. 34
MTZ is a critical element of the triple therapy; but unfortunately has been linked to a high incidence of resistance. Bacteria’s virulence is correlated to metronidazole resistance, with strains deficient to the cagA gene exhibiting higher resistance.35 Clarithromycin resistance, which is increasing over time is a primary cause of H. pylori treatment failure. The dual-priming oligonucleotide-based multiplex polymerase chain reaction (DPO-PCR) method detected the 23S RNA gene of H. pylori as point mutation responsible for resistance to certain antibiotics, including clarithromycin. Several mutations have been identified (A2142C, A2142G, A2143G, A2144T, T2717C, and C2694A), and clinical studies reported high H. pylori eradication rates subsequent to the identification of clarithromycin resistance using the DPO-PCR method.36
Herbal Medicine and Parkinson’s disease
Carbidopa/levodopa, dopamine agonists, MAO-B inhibitors, and anticholinergics are among the prescribed medications for PD. These medications are utilized to alleviate symptoms. 37 Currently there is no licensed therapy capable of halting or slowing the progression of Parkinson’s disease despite countless trials and studies, 38 therefore assessing the neuroprotective qualities of novel drugs is essential for future effective treatments.
For the management of neurodegenerative diseases daily diet may be advantageous. The correlation between gut microbiota and neurodegenerative diseases underscores the importance of creating meals enriched with prebiotics and probiotics is increasing.39 Probiotics are live microorganisms, of beneficial bacteria and yeast that confers health benefits when consumed in adequate amounts. These microbes maintain balance for the gut microbiome, supporting digestive health by fostering the growth of beneficial bacteria and inhibiting harmful pathogens. Thus, alleviate symptoms associated with digestive disorders such as irritable bowel syndrome (IBS) and diarrhea.40 Prebiotics, on the other hand, are indigestible fibers that serve as food for the beneficial bacteria in the gut, encouraging their growth and activity. By supporting the balance of the gut microbiome, prebiotics contribute to better digestion, immune function, and nutrient absorption.41 Common sources of probiotics include fermented foods like yogurt and kefir, while prebiotics are found in fiber-rich foods such as garlic, onion, and whole grains. Together, synbiotics work synergistically to promote overall gut health and well-being.42
Furthermore, fecal DNA sequencing can help to evaluate the ratio of beneficial to detrimental bacteria and guide us to personalized medical approaches.43
On the other hand, herbal therapies represent promising treatment pathway for Parkinson’s disease, potentially offering efficient results than synthetic drugs due to their interference with the underlying disease mechanisms. Generally, polyphenols contribute to neuronal survival and serve a protective role in pro-survival cell signaling, thereby protecting mitochondria from pathological events.44
Epigallocatechin-3-gallate (EGCG), a polyphenolic compound abundant in green tea extract, protects against neuronal death in the substantia nigra pars compacta (SNpc), by reducing 6-hydroxydopamine (6-OHDA) induced apoptosis in PC12 cells.45 EGCG also lowers the levels of anti-apoptotic proteins Bcl-w, Bcl-2, and Bcl-x(L), and inhibits the mRNA synthesis of pro-apoptotic factors Bad, Bax, and Mdm2. By increasing phosphorylated protein kinase C (PKC); EGCG rescues SH-SY5Y cells from 6-OHDA toxicity. These findings suggest that EGCG may prevent oxidative stress-induced cell death by activating PKC and modulating apoptosis.46
While EGCG is a promising compound we still have limited clinical observations and bioavailability studies. Thus; variability in dosing and extract filtration and standardization may limit reproducibility.
Scutellaria baicalensis, an herb containing baicalein which is a flavonoid extracted from the plants’ roots and inhibits the levels of NO and COX-2, as well as ROS accumulation, ATP depletion, and leads to apoptosis.47
Some researchers proposed that polyphenols as curcumin, icariin, baicalein, and cannabidiol, which are naturally occurring bioactive phytochemicals have neuroprotective effects in Parkinson’s disease via regulating the PI3K/AKT pathway.48,49 However, because of their limited biological activity their use is limited. Nevertheless, there are different opinions to improve their effectiveness by changing the structure or using the nano-loading method taking the dosage, time and mode of administration in consideration as important factors for Parkinson’s disease treatment.50
Curcumin easily permeates blood-brain barrier and neuronal membranes due to its anti-amyloid and anti-inflammatory properties, rendering it a viable option for the management of Parkinson’s disease or other neurodegenerative diseases.51 Previous studies has shown that phytochemicals achieved from saffron specifically – crocus sativa, crocin, crocetin, and safranal – can slow down the neurodegenerative progress in Parkinson’s disease by reducing dopaminergic neuronal loss particularly in the substantia nigra as well as preventing the accumulation of alpha-syncline.52 On the other side, Chamomile, through its active constituent apigenin, can protect the neurons from oxidative stress and inflammation, resulting in improvement in patients with Parkinson’s Disease.52,53
Many studies linked the neuroprotective effect of Siberian ginseng Roots and Stems in Parkinson’s disease to the reduction in inflammation and activation of microglial in brain ischemia.54 However, there is also evidence pointing out to the fact that Siberian ginseng may cause neurotoxicity in the central nervous system necessitating further studies to determine the risk versus benefit ratio and the potential therapeutic applications.55
Parkinson’s disease sleep disorder (PDSD), is linked to fluctuations of serum levels of melatonin, cystatin C, vitamin D, homocysteine, and dopamine. Specific herbs used in Traditional Chinese Medicine (TCM), including Semen ziziphi spinosae, Rhizoma anemarrhenae, Poria cocos, and Rhizoma chuanxiong, are believed to nourish the liver, calm the mind, and alleviate vexation.56 Chinese herbal medicine (CHM) prescriptions unlike ordinary antidepressant drugs, may have more favorable impact on depression in PD specifically on the motor symptoms. 57
UGS plant study (Uncariae Ramulus et Cortex, Gastrodiae Rhizoma, and Schisandrae Fructus) demonstrated that this treatment enhances the quality of life for Parkinson’s patients particularly those with severe anxiety. 58 The brief duration of treatment and the lack of blinding for the participants in the control group; might inflate the results. However, the authors identified several limitations to their study, including a propensity to underestimate the effects of UGS for PD by restricting its use to patients who poorly respond to medication; nonetheless, the study advocated for further studies to elucidate benefits of UGS in Parkinson’s disease treatment, and its impact on anxiety symptoms in patients with Parkinson’s disease.59
Various publications investigated the vast components of CHM herbs, demonstrating numerous approaches for treating Parkinson’s disease symptoms as modulating the hypothalamic-pituitary-adrenal (HPA) axis, suppressing CNS inflammation, and increasing effect of Brain derived-neurotrophic factor (BDNF) release which are critical for symptoms relieve.60 Components may also act on α-synuclein, identified as a primary factor in PD, providing benefits for non-motor and motor signs. It is important to know that CHM is not fully understood and has not been clinically trailed, though it demonstrate favorable results in vivo.57
Herbal Medicine and Helicobacter pylori
Previous studies have established that plants containing flavonoids may have interesting effects on eradicating H. pylori infection. For example, the extracts of Glycirrhiza glabra including glabridin and glabrol have been demonstrated to inhibit the syntheses of dihydrofolate reductase in cells, which is crucial for H. pylori‘s DNA synthesis.61 Another examples include, quercetin, catechin, epicatechin, and rutin, which possess anti-inflammatory and antioxidant functions.62
Garlic, a common plant in Jordanian ethnopharmacology, possesses anti-oxidative and anti-inflammatory activities and exhibits tremendous antimicrobial properties due to the presence of allicin. It suppresses bacterial growth and biofilm formation regardless bacterial strain.63
A previous study that was conducted on rat model revealed that ethanolic pomegranate peel extract inhibited urease enzyme activity in H. pylori, hence preventing its gastrointestinal colonization by 39%; the combination of the extract with metronidazole had synergistic effect, as it serves adjunct therapy due to its anti-inflammatory effects and prevention of H. pylori colonization.64
The hydroalcoholic extract of sumac (Rhus coriaria L.) revealed antibacterial and anti-inflammatory effects, which retain its efficacy when subjected in vitro to gastric digesting conditions.65 By blocking Il-6 and Il-8, it can directly inhibit the growth of H. pylori, and it is noteworthy that bioactivity can be attained by oral administration of sumac or its extracts.66
Nevertheless, other lesser known plant species as Salvia species (S. eiggii, S. hierosolymitana, and S. viridis) demonstrated significant antioxidant activity. Among them S. hierosolymitana has the most potent antioxidant activity, attributed to its high total phenolic content (TPC) and total flavonoid content (TFC) along with rosmarinic acid. Also, the methanolic extract of S. hierosolymitana showed significant DPPH radical scavenging activity and high ferrous ions chelating effect due to its abundant rosmarinic acid, caffeic acid, and other bioactive components.67
A review addressing the role of herbal remedies on the eradication of H. pylori mentioned the active ingredients in cinnamon, specifically eugenol and cinnamaldehyde, exhibited inhibitory effects on H. pylori proliferation in vitro. It also included mixed results from clinical trials; in a study 98 patients administered cinnamon extract alongside antibiotics experienced lower H. pylori densities compared with the antibiotic-only group.68
Ginger (Zingiber officinale) owing to the presence of two active compounds gingerol and zingerone, provides antioxidant properties and inhibits the growth of H. pylori.69
Dioscin, a saponin glycoside present in various plants, may serve as an effective therapy against H. pylori when used in conjunction with clarithromycin, metronidazole, and levofloxacin as it enhances their antibacterial activity and disturbs H. pylori biofilm formation through direct antimicrobial action.70
In a model of non-tumoral gastric epithelium, hydroalcoholic extract of Castaneda sativa Mill leaves, which contain ellagitannin isomers as castalagin and vescalagin, were evaluated for their anti-inflammatory, anti-adhesive, and antibacterial effects in relation to H. pylori infection indicated that ellagitannins inhibited the release of IL-8 and had an effect against TNF-α, while castalagin and vescalagin potentially inhibited H. pylori proliferation.71
The integration of phenolic phytochemical-rich plants and lactic acid bacteria with probiotic capabilities can suppress H. pylori growth and address H. pylori associated peptic ulcers and active chronic gastritis.72 Lactic acid bacteria strains combined to methanolic extracts of garlic (Allium sativum L.),73 red onion (Allium cepa L.), 74 cumin (Cuminum cyminum L.),74 and fenugreek (T. foenum-graecum L.),75 was explored against H. pylori colonization to confirm the improved efficacy. The combination of extracts with probiotics suppressed H. pylori, with varied outcomes. H. pylori growth suppressed in a concentration dependent manner by phenolic substances found in plants such as gallic acid, caffeic acid, quercetin, and vanillic acid, as well as probiotics through lactic acid and bacteriocins. Additionally, combining B. breve (a beneficial bacteria resides in the intestines) with fenugreek extract effectively suppressed H. pylori by decreasing gastritis in H. pylori infected rats.72
Sixteen medicinal herbs growing in Jordan were tested against clinical isolates of H. pylori, including Aloysia triphylla (L’Her.) Britton, Anethum graveolens L., Artemisia inculata Delile, Capparis spinosa L., Crataegus aronia (L.) Bosc ex. DC., Inula viscosa (L.) Ait, Lavandula officinalis Chaix., Lepidium sativum L., Origanum syriaca L., Paronychia argentea Lam., Passiflora incarnata L., Psidium guajava L., Sarcopoterium spinosum (L.) Spach., Sesamwn indicum L., Urtica urens L., and Varthemia iphionoides Boiss. The results demonstrated that ethanolic extracts of A. triphylla and I. viscosa exhibited the highest lethal effect against H. pylori isolates by inhibiting bacterial protein synthesis.76
Parkinson’s disease and gut microbiota
Recent studies regarding Parkinson’s disease focused on the gut-brain axis, with drawing attention to the common gastrointestinal infection Helicobacter pylori. In Parkinson’s Disease, numerous pathophysiological changes take place even in early stages, including increased local inflammation, oxidative stress, neurodegeneration in the enteric nervous system and intestinal permeability.77 These changes may support the hypothesis of associating PD etiology to gastrointestinal dysfunctions.78,79
The gut-brain axis is a gaining a major interest in Parkinson’s Disease studies, and as the incidence of H. pylori is elevated in PD patients, it could have possible involvement in the disease and influence Parkinson’s Disease through several mechanisms:
pylori infection causes chronic infection which in turn may boost systemic inflammation, facilitating the systemic entry of inflammatory mediators via the secretion of pro-inflammatory cytokines including IL-6 and TNF-α. These mediators have the potential to penetrate blood brain barrier contributing the neuroinflammatory process that are implicated in dopaminergic neurodegeneration worsening neuroinflammation in PD patients and exacerbating dopaminergic death in the substantia nigra. 80 Boziki et al, (2018), conducted a study demonstrated that H. pylori-related galectin-3 may exacerbate central nervous system inflammation consequently aggravating neuronal loss in PD. 28
Dysbiosis of gut microbiota may influence gut-brain interaction which is observed in roughly 25% of patients with PD. Parkinson’s Disease clinical hallmarks may start in the gastrointestinal tract and disseminate to the brain through vagus nerve following α-synuclein aggregation. The fact that pylori infection has been associated with elevated expression of α-synuclein in gastric mucosa may link H. pylori infection to the progression of PD. 80,81 However the small sample size may limit the generalizability. A previous study by Braak and colleges hypothesized that PD could be initiated by unknown pathogen colonizes through the gastric mucosal lining and can trigger the formation of α-synuclein.82
Gastrointestinal infections may interfere with the absorption of nutrients essential for neuronal health and function as vitamin B12 and iron.83
pylori infection may hinder levodopa absorption by delaying gastric emptying, diminishing mucosal integrity and altering gastric pH. Thus, H. pylori can affect the advancement and treatment strategies of PD by making levodopa more bioavailable which improves control of motor symptoms.84
While these data collectively may indicate a complex link between H. pylori infection and the pathophysiology of Parkinson’s disease, encompassing microbial dysbiosis, systemic inflammation, compromised medication absorption, and neuroimmune communication, it is important to consider the alternating hypothesis of reverse causality.
PD may induce progressive autonomic dysfunction by altering gastric mucosa, thus, fostering an environment that facilitates H. pylori colonization, SIBO and overall dysbiosis.85 Impaired peristalsis and altered gastric acidity that may take place in PD may imbalance gut microbial and enhance H. pylori existence as well as eradication resistance.85 This bidirectional interaction still unclear and may indicate the complex interaction between PD and H. pylori.
Eradication of H. pylori as an initial step for PD improvement
pylori treatment with antibacterial regimen plus levodopa optimization: Eradication of H. pylori depends on antibacterial treatment involving triple or quadruple regimens including proton pump inhibitor, antibiotics (clarithromycin, amoxicillin, metronidazole or tetracycline) with or without bismuth subsalicylate.86 The corner stone treatment of Parkinson’s Disease; levodopa may be influenced by gastrointestinal issues because of its narrow therapeutic index.87 The absorption of levodopa may be compromised by gastric acid levels, as its bioavailability might be diminished by PPIs or delayed gastric emptying caused by the infection.88 Although eradication of H. pylori may enhance the efficiency of levodopa and improve PD symptoms, metronidazole may interfere with levodopa metabolism and block its conversion in the gut.89
pylori treatment with anti-inflammatory agents: H. pylori infection may provoke systemic inflammation via cytokine release, thereby exacerbating neuroinflammation and leading to PD.90 Anti-inflammatory drugs are utilized in H. pylori eradication, giving a dual benefit of lowering gastric inflammation and mitigating systemic neuroinflammatory process related to PD.91 Selective COX-2 inhibitors may mitigate gastric inflammation and neuroinflammation, studies indicate that such drugs may play a crucial role in enhancing levodopa therapy while aiding dopaminergic neurons preservation, hence slowing Parkinson’s disease progression.92
pylori treatment with tailored nutrient supplement and herbal remedy use: Vitamin B12, iron, magnesium and antioxidants are critical nutrients for neural function and maintenance of myelin integrity.93 H. pylori infection impairs gastric acid secretion, resulting in diminished nutrient absorption. Consequently, nutrients and supplementation may facilitate H. pylori eradication and influence the progression of PD.94
On the other hand, antioxidants as vitamin C, vitamin E, selenium and coenzyme Q10 are antioxidants available in herbal remedies can mitigate oxidative stress and provide natural anti-inflammatory and neuroprotective effects. 95 Therefore, incorporating customized nutrient supplementation and herbal remedies into H. pylori treatment protocols may offer a beneficial approach that impacts both gastrointestinal and neurological health, potentially enhancing neuroprotection.96,97
S2 Table presents a summary of phytochemicals and extracts assessed for their significance in Parkinson’s disease and H. pylori infection, detailing their mechanisms of action and the characteristics of supporting studies included in supplementary file.
(4) H. pylori treatment with synbiotic treatment: Synbiotics, comprising prebiotics (non-digestible fibers) and probiotics (living microorganisms that interact with gut microflora) may significantly contribute gastrointestinal health by restoring gut microbiota balance and increasing aspects related to the gut-brain axis factors. Probiotics, may inhibit H. pylori colonization by secreting lactic acid and blocking IL-8 secretion.98 This in turn improves H. pylori eradication and promotes gut health, leading to enhanced outcomes for
Parkinson’s disease by diminishing systemic inflammation and neuroinflammation.99
Conclusion
Elimination of Helicobacter pylori presents promising potential for alleviating its extensive systemic repercussions, particularly regarding Parkinson’s disease. The interaction between H. pylori-induced gut dysbiosis, chronic inflammation, and neurodegeneration underscores the pivotal role of the gut-brain axis in the advancement of PD. By targeting H. pylori infection with specific interventions including antibiotics, synbiotics, nutrient supplementation, and herbal treatments; it is feasible to modulate gastric and systemic inflammation, restore gut microbiota equilibrium, and enhance nutrient absorption, all of which are vital for preserving neural health in PD patients.
While some emerging data indicates that H. pylori eradication may improve both motor and non-motor symptoms of Parkinson’s disease by enhancing levodopa absorption and diminishing neuroinflammation; however, additional longitudinal studies are required to confirm this association. A multidisciplinary approach that integrates conventional medicines with personalized techniques has the potential to improve treatment outcomes, enhance quality of life, and maybe decelerate disease progression. The correlation between H. pylori eradication and PD care highlights the necessity of examining neurodegenerative illnesses from a comprehensive perspective that integrates gastrointestinal and brain health.
The link between PD and H. pylori is constrained with several limitations as most available studies are observational studies thus; it is difficult to make clear consequence of infection – neurological sequences. On the other hand, many studies inconsistently control disease confounders, which reduces the comparability and inflate possible associations. As a result; more longitudinal multidisciplinary studies are needed to explore the correlation between gastrointestinal and neurological aspects and investigate the importance of both diseases management. On the other hand, using advanced technologies including bioinformatics, metabolomics and gut microbiome sequencing may advance our understanding the complexity of relationship between gut microbiome, neurodegeneration and herbal remedies.
Acknowledgment
The authors extend their appreciation to the Deanship of Scientific Research at Yarmouk University, Irbid, Jordan.
Funding Source
This research was funded by the Deanship of Scientific Research at Yarmouk University, Irbid, Jordan, under grant number (18/2024).
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Deniz Al-Tawalbeh: principal investigator, conceived the idea, planned the review and supervised the all project.
- Wissam Al-Momani, Amir Majdi Moh’d and Abdelrahman Alenaizat: conducted the literature search and data collection.
- Anas Majdi Moh’d and Besher Mfarrej: M wrote the initial draft of manuscript, all authors edited and revised the final draft.
References
- Palakurthi B, Burugupally SP. Postural instability in Parkinson’s disease: a review. Brain sciences. 2019;9(9):239.
CrossRef - Tonduti D, Chiapparini L, Moroni I, et al. Neurological disorders associated with striatal lesions: classification and diagnostic approach. Current neurology and neuroscience reports. 2016;16:1-15.
CrossRef - Radad K, Moldzio R, Krewenka C, Kranner B, Rausch W-D. Pathophysiology of non-motor signs in Parkinsons disease: some recent updating with brief presentation. Exploration of Neuroprotective Therapy. 2023;3(1):24-46.
CrossRef - Pang SY-Y, Ho PW-L, Liu H-F, et al. The interplay of aging, genetics and environmental factors in the pathogenesis of Parkinson’s disease. Translational Neurodegeneration. 2019;8:1-11.
CrossRef - Arneth BM. Gut–brain axis biochemical signalling from the gastrointestinal tract to the central nervous system: gut dysbiosis and altered brain function. Postgraduate medical journal. 2018;94(1114):446-452.
CrossRef - Lei Q, Wu T, Wu J, et al. Roles of α-synuclein in gastrointestinal microbiome dysbiosis-related Parkinson’s disease progression. Molecular medicine reports. 2021;24(4):734.
CrossRef - McGee DJ, Lu X-H, Disbrow EA. Stomaching the possibility of a pathogenic role for Helicobacter pylori in Parkinson’s disease. Journal of Parkinson’s disease. 2018;8(3):367-374.
CrossRef - Kesharwani A, Dighe OR, Lamture Y. Role of helicobacter pylori in gastric carcinoma: A review. Cureus. 2023;15(4)
CrossRef - Huang H-K, Wang J-H, Lei W-Y, Chen C-L, Chang C-Y, Liou L-S. Helicobacter pylori infection is associated with an increased risk of Parkinson’s disease: a population-based retrospective cohort study. Parkinsonism & related disorders. 2018;47:26-31.
CrossRef - Wei B-r, Zhao Y-j, Cheng Y-f, Huang C, Zhang F. Helicobacter pylori infection and Parkinson’s Disease: etiology, pathogenesis and levodopa bioavailability. Immunity & Ageing. 2024;21(1):1.
CrossRef - Nyholm D, Hellström PM. Effects of Helicobacter pylori on levodopa pharmacokinetics. Journal of Parkinson’s Disease. 2021;11(1):61-69.
CrossRef - Kountouras J, Boziki M, Polyzos S, et al. The emerging role of Helicobacter pylori-induced metabolic gastrointestinal dysmotility and neurodegeneration. Current Molecular Medicine. 2017;17(6):389-404.
CrossRef - Parkinson’s Disease: Causes, Symptoms, and Treatments. National Institute of Aging; 2022. April 2022. https://www.nia.nih.gov/health/parkinsons-disease/parkinsons-disease-causes-symptoms-and-treatments
- Staff MC. Pkinson’s Disease. Mayo clinic United states of america: Mayo Foundation for Medical Education and Research; 2024.
- Chandan JS. Improving global surveillance of gender-based violence. health. 2020;230(38):7.
- Tysnes O-B, Storstein A. Epidemiology of Parkinson’s disease. Journal of neural transmission. 2017;124:901-905.
CrossRef - Alrefai A, Habahbih M, Alkhawajah M, et al. Prevalence of Parkinson’s disease in Northern Jordan. Clinical neurology and neurosurgery. 2009;111(10):812-815.
CrossRef - Safiri S, Noori M, Nejadghaderi SA, et al. The burden of Parkinson’s disease in the Middle East and North Africa region, 1990–2019: results from the global burden of disease study 2019. BMC Public Health. 2023;23(1):107.
CrossRef - Ben-Shlomo Y, Darweesh S, Llibre-Guerra J, Marras C, San Luciano M, Tanner C. The epidemiology of Parkinson’s disease. The Lancet. 2024;403(10423):283-292.
CrossRef - Rajan S, Kaas B. Parkinson’s disease: risk factor modification and prevention. Thieme Medical Publishers, Inc.; 2022:626-638.
CrossRef - Klein C, Westenberger A. Genetics of Parkinson’s disease. Cold Spring Harbor perspectives in medicine. 2012;2(1):a008888.
CrossRef - Ball N, Teo W-P, Chandra S, Chapman J. Parkinson’s disease and the environment. Frontiers in neurology. 2019;10:421551.
CrossRef - Al Tawalbeh D, Aburjai T, Al Balas Q, Al Samydai A. In Silico and in vitro investigation of anti Helicobacter activity of selected phytochemicals. Journal of Pharmacy and Bioallied Sciences. 2022;14(3):132-139.
CrossRef - Chen Y-C, Malfertheiner P, Yu H-T, et al. Global prevalence of Helicobacter pylori infection and incidence of gastric cancer between 1980 and 2022. Gastroenterology. 2024;166(4):605-619.
CrossRef - Weller C, Charlett A, Oxlade NL, et al. Role of Chronic Infection and Inflammation in the Gastrointestinal Tract in the Etiology and Pathogenesis of Idiopathic Parkinsonism: Part 3: Predicted Probability and Gradients of Severity of Idiopathic Parkinsonism Based on H. pylori Antibody Profile. Helicobacter. 2005;10(4):288-297.
CrossRef - Shen X, Yang H, Wu Y, Zhang D, Jiang H. Meta‐analysis: association of Helicobacter pylori infection with Parkinson’s diseases. Helicobacter. 2017;22(5):e12398.
CrossRef - Kalyan M, Tousif AH, Sonali S, et al. Role of endogenous lipopolysaccharides in neurological disorders. Cells. 2022;11(24):4038.
CrossRef - Boziki M, Polyzos SA, Deretzi G, et al. A potential impact of Helicobacter pylori-related galectin-3 in neurodegeneration. Neurochemistry International. 2018;113:137-151.
CrossRef - McGee DJ, Lu X-H, Disbrow EA. Stomaching the possibility of a pathogenic role for Helicobacter pylori in Parkinson’s disease. Journal of Parkinson’s disease. 2018;8(3):367-374.
CrossRef - Ben-Jonathan N. Dopamine: endocrine and oncogenic functions. CRC Press; 2020.
CrossRef - So Y-J, Lee J-U, Yang G-S, et al. The potentiality of natural products and herbal medicine as novel medications for parkinson’s disease: a promising therapeutic approach. International Journal of Molecular Sciences. 2024;25(2).
CrossRef - Blandini F, Armentero M-T. Dopamine receptor agonists for Parkinson’s disease. Expert opinion on investigational drugs. 2014;23(3):387-410.
CrossRef - Sambhaji Misal S, Balasaheb Bhor V, Subramanian Chelakara R. Structural Insights into Potent Anti-ulcer Agents for the Eradication of Helicobacter pylori. Anti-Infective Agents. 2024;22(5):29-42.
CrossRef - Lin Y, Shao Y, Yan J, Ye G. Antibiotic resistance in Helicobacter pylori: From potential biomolecular mechanisms to clinical practice. Journal of Clinical Laboratory Analysis. 2023;37(7):e24885.
CrossRef - Goderska K, Agudo Pena S, Alarcon T. Helicobacter pylori treatment: antibiotics or probiotics. Applied microbiology and biotechnology. 2018;102:1-7.
CrossRef - Moon S-G, Lim C-H, Kang H-J, Choi A, Kim S, Oh J-H. Seven days of bismuth-based quadruple therapy is as effective for the first-line treatment of clarithromycin-resistant confirmed Helicobacter pylori infection as 14 days of bismuth-based quadruple therapy. Journal of Clinical Medicine. 2022;11(15):4440.
CrossRef - Marino BL, de Souza LR, Sousa KP, et al. Parkinson’s disease: a review from pathophysiology to treatment. Mini reviews in medicinal chemistry. 2020;20(9):754-767.
CrossRef - Kopil CM, Asis A, Campbell C, et al. A Coalition to Advance Treatments for Parkinson’s Disease, Dementia with Lewy Bodies, and Related Disorders. Journal of Parkinson’s Disease. 2024;14(6):1105-1114.
CrossRef - Jain A, Madkan S, Patil P. The role of gut microbiota in neurodegenerative diseases: Current insights and therapeutic implications. Cureus. 2023;15(10).
CrossRef - Ji J, Jin W, Liu SJ, Jiao Z, Li X. Probiotics, prebiotics, and postbiotics in health and disease. MedComm. 2023;4(6):e420.
CrossRef - Davani-Davari D, Negahdaripour M, Karimzadeh I, et al. Prebiotics: definition, types, sources, mechanisms, and clinical applications. Foods. 2019;8(3):92.
CrossRef - Markowiak P, Śliżewska K. Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients. 2017;9(9):1021.
CrossRef - Zheng Y, Bonfili L, Wei T, Eleuteri AM. Understanding the gut–brain axis and its therapeutic implications for neurodegenerative disorders. Nutrients. 2023;15(21):4631.
CrossRef - Bhullar KS, Rupasinghe HV. Polyphenols: multipotent therapeutic agents in neurodegenerative diseases. Oxidative medicine and cellular longevity. 2013;2013(1):891748.
CrossRef - Kupershmidt L, Weinreb O, Amit T, Mandel S, Bar-Am O, Youdim M. Novel molecular targets of the neuroprotective/neurorescue multimodal iron chelating drug M30 in the mouse brain. Neuroscience. 2011;189:345-358.
CrossRef - Wang Y, Wu S, Li Q, et al. Epigallocatechin-3-gallate: A phytochemical as a promising drug candidate for the treatment of Parkinson’s disease. Frontiers in pharmacology. 2022;13:977521.
CrossRef - Amro M, Teoh S, Norzana A, Srijit D. The potential role of herbal products in the treatment of Parkinson’s disease. La Clinica Terapeutica. 2018;169(1):e23-e33.
- Kujawska M, Jodynis-Liebert J. Polyphenols in Parkinson’s disease: a systematic review of in vivo studies. Nutrients. 2018;10(5):642.
CrossRef - Long H-Z, Cheng Y, Zhou Z-W, Luo H-Y, Wen D-D, Gao L-C. PI3K/AKT signal pathway: a target of natural products in the prevention and treatment of Alzheimer’s disease and Parkinson’s disease. Frontiers in pharmacology. 2021;12:648636.
CrossRef - Tabanelli R, Brogi S, Calderone V. Improving curcumin bioavailability: Current strategies and future perspectives. Pharmaceutics. 2021;13(10):1715.
CrossRef - Maiti P, Dunbar GL. Use of curcumin, a natural polyphenol for targeting molecular pathways in treating age-related neurodegenerative diseases. International journal of molecular sciences. 2018;19(6):1637.
CrossRef - Ali F, Ahmed J, Ahmad S. Role of saffron and chamomile in Parkinson diseases. JPMA. 2023;73:2-15.
CrossRef - Salehi B, Venditti A, Sharifi-Rad M, et al. The therapeutic potential of apigenin. International journal of molecular sciences. 2019;20(6):1305.
CrossRef - Bu Y, Jin ZH, Park SY, et al. Siberian ginseng reduces infarct volume in transient focal cerebral ischaemia in Sprague‐Dawley rats. Phytotherapy Research: An International Journal Devoted to Pharmacological and Toxicological Evaluation of Natural Product Derivatives. 2005;19(2):167-169.
CrossRef - Yap KY-L, See CS, Chan A. Clinically-relevant chemotherapy interactions with complementary and alternative medicines in patients with cancer. Recent patents on food, nutrition & agriculture. 2010;2(1):12-55.
CrossRef - Liu Y-y, Yu L-h, Zhang J, Xie D-j, Zhang X-x, Yu J-m. Network Pharmacology‐Based and Molecular Docking‐Based Analysis of Suanzaoren Decoction for the Treatment of Parkinson’s Disease with Sleep Disorder. BioMed research international. 2021;2021(1):1752570.
CrossRef - Zhang Y, Xu X. Chinese herbal medicine in the treatment of depression in Parkinson’s disease: From molecules to systems. Frontiers in pharmacology. 2022;13:879459.
CrossRef - Zhao Q, Hu Y, Yan Y, et al. The Effects of Shaoma Zhijing Granules and its Main Components on Tourette Syndrome. Phytomedicine. 2024:155686.
CrossRef - Jin C, Cho K-H, Kwon S, et al. Effectiveness and safety of herbal medicine Ukgansan for clinical symptoms in Parkinson’s disease: A pilot, randomized, assessor-blinded clinical trial. Frontiers in Neurology. 2022;13:1025269.
CrossRef - Liu J, Meng T, Wang C, Cheng W, Zhang Q, Cheng G. Natural products for the treatment of depression: Insights into signal pathways influencing the hypothalamic–pituitary–adrenal axis. Medicine. 2023;102(44):e35862.
CrossRef - Sathianarayanan S, Ammanath AV, Biswas R, Anita B, Sukumaran S, Venkidasamy B. A new approach against Helicobacter pylori using plants and its constituents: A review study. Microbial Pathogenesis. 2022;168:105594.
CrossRef - Ivyna de Araújo Rêgo R, Guedes Silvestre GF, Ferreira de Melo D, et al. Flavonoids-rich plant extracts against Helicobacter pylori infection as prevention to gastric cancer. Frontiers in Pharmacology. 2022;13:951125.
CrossRef - El-Saadony MT, Saad AM, Korma SA, et al. Garlic bioactive substances and their therapeutic applications for improving human health: a comprehensive review. Frontiers in immunology. 2024;15:1277074.
CrossRef - Mayyas A, Abu-Sini M, Amr R, et al. Novel in vitro and in vivo anti-Helicobacter pylori effects of pomegranate peel ethanol extract. Veterinary World. 2021;14(1):120.
CrossRef - Martinelli G, Angarano M, Piazza S, et al. The nutraceutical properties of sumac (Rhus coriaria L.) against gastritis: Antibacterial and anti-inflammatory activities in gastric epithelial cells infected with H. pylori. Nutrients. 2022;14(9):1757.
CrossRef - Khalil M, Abdallah H, Razuka-Ebela D, Calasso M, De Angelis M, Portincasa P. The Impact of Za’atar Antioxidant Compounds on the Gut Microbiota and Gastrointestinal Disorders: Insights for Future Clinical Applications. Antioxidants. 2023;12(2):426.
CrossRef - Al-Jaber HI, Shakya AK, Elagbar ZA. HPLC profiling of selected phenolic acids and flavonoids in Salvia eigii, Salvia hierosolymitana and Salvia viridis growing wild in Jordan and their in vitro antioxidant activity. PeerJ. 2020;8:e9769.
CrossRef - Abdi S, Ataei S, Abroon M, Sanaye PM, Abbasinazari M, Farrokhian A. A Comprehensive Review of the Role of Complementary and Dietary Medicines in Eradicating Helicobacter pylori. Iranian Journal of Pharmaceutical Research: IJPR. 2022;21(1)
CrossRef - Shmuely H, Domniz N, Yahav J. Non-pharmacological treatment of Helicobacter pylori. World journal of gastrointestinal pharmacology and therapeutics. 2016;7(2):171.
CrossRef - Spiegel M, Krzyżek P, Dworniczek E, Adamski R, Sroka Z. In silico screening and in vitro assessment of natural products with anti-virulence activity against Helicobacter pylori. Molecules. 2021;27(1):20.
CrossRef - Piazza S, Martinelli G, Fumagalli M, et al. Ellagitannins from Castanea sativa Mill. leaf extracts impair H. pylori viability and infection-induced inflammation in human gastric epithelial cells. Nutrients. 2023;15(6):1504.
CrossRef - Hasna B, Houari H, Koula D, Marina S, Emilia U, Assia B. In vitro and in vivo study of combined effect of some Algerian medicinal plants and probiotics against Helicobacter pylori. Microorganisms. 2023;11(5):1242.
CrossRef - Si X-B, Zhang X-M, Wang S, Lan Y, Zhang S, Huo L-Y. Allicin as add-on therapy for Helicobacter pylori infection: A systematic review and meta-analysis. World Journal of Gastroenterology. 2019;25(39):6025.
CrossRef - BOUHENNI H. Study of combined effect of some medicinal plants and probiotics against Helicobacter pylori responsible for gastroduodenal diseases. Faculty of Nature and Life Sciences; 2023.
- Addissouky TA, Ali MM, El Sayed IET, Wang Y. Recent advances in diagnosing and treating helicobacter pylori through botanical extracts and advanced technologies. Archives of Pharmacology and Therapeutics. 2023;5(1):53-66.
CrossRef - Masadeh MM, Alkofahi AS, Alzoubi KH, Tumah HN, Bani-Hani K. Anti-Helicobactor pylori activity of some Jordanian medicinal plants. Pharmaceutical biology. 2014;52(5):566-569.
CrossRef - Shannon KM, Keshavarzian A, Dodiya HB, Jakate S, Kordower JH. Is alpha‐synuclein in the colon a biomarker for premotor Parkinson’s disease? Evidence from 3 cases. Movement Disorders. 2012;27(6):716-719.
CrossRef - Devos D, Lebouvier T, Lardeux B, et al. Colonic inflammation in Parkinson’s disease. Neurobiology of disease. 2013;50:42-48.
CrossRef - Braak H, De Vos RA, Bohl J, Del Tredici K. Gastric α-synuclein immunoreactive inclusions in Meissner’s and Auerbach’s plexuses in cases staged for Parkinson’s disease-related brain pathology. Neuroscience letters. 2006;396(1):67-72.
CrossRef - Fasano A, Bove F, Gabrielli M, et al. The role of small intestinal bacterial overgrowth in Parkinson’s disease. Movement Disorders. 2013;28(9):1241-1249.
CrossRef - Almeida C, Oliveira R, Soares R, Barata P. Influence of gut microbiota dysbiosis on brain function: a systematic review. Porto biomedical journal. 2020;5(2):1.
CrossRef - Elfil M, Kamel S, Kandil M, Koo BB, Schaefer SM. Implications of the gut microbiome in Parkinson’s disease. Movement Disorders. 2020;35(6):921-933.
CrossRef - Rizzo G, Laganà AS. A review of vitamin B12. Molecular nutrition. 2020:105-129.
CrossRef - Vosoo K. The Damages Made by Helicobacter Pylori on Parkinson and Emerging Role of Nutrition in Parkinson’s Disease. International Journal of Medical Reviews. 2023;10(2):516-522.
- Xiao X, Wang Z, Zhang H, et al. Helicobacter pylori infection might be a protective factor against Parkinson’s disease. Scientific Reports. 2025;15(1):16689.
CrossRef - Roberts LT, Issa PP, Sinnathamby ES, et al. Helicobacter pylori: A review of current treatment options in clinical practice. Life. 2022;12(12):2038.
CrossRef - Lenka A, Di Maria G, Lamotte G, Bahroo L, Jankovic J. Practical pearls to improve the efficacy and tolerability of levodopa in Parkinson’s disease. Expert Review of Neurotherapeutics. 2022;22(6):489-498.
CrossRef - Lahner E, Virili C, Santaguida MG, Annibale B, Centanni M. Helicobacter pylori infection and drugs malabsorption. World Journal of Gastroenterology: WJG. 2014;20(30):10331.
CrossRef - Beckers M, Bloem BR, Verbeek MM. Mechanisms of peripheral levodopa resistance in Parkinson’s disease. npj Parkinson’s Disease. 2022;8(1):56.
CrossRef - Wang F, Yao Z, Jin T, Mao B, Shao S, Shao C. Research progress on Helicobacter pylori infection related neurological diseases. Ageing Research Reviews. 2024:102399.
CrossRef - Beydoun MA, El-Hajj ZW. Helicobacter pylori and Cognitive Dysfunction. Infectious Diseases in Neurocognitive and Neuropsychiatric Medicine. 2024:142.
CrossRef - Gupta A, Kumar A, Kulkarni S. Targeting oxidative stress, mitochondrial dysfunction and neuroinflammatory signaling by selective cyclooxygenase (COX)-2 inhibitors mitigates MPTP-induced neurotoxicity in mice. Progress in Neuro-Psychopharmacology and Biological Psychiatry. 2011;35(4):974-981.
CrossRef - Myelin I, Zinc D, Vitamin A. Brain Requirement for Nutrient. Swaiman’s Pediatric Neurology-E-Book: Principles and Practice. 2024:384.
- Testerman TL, Morris J. Beyond the stomach: an updated view of Helicobacter pylori pathogenesis, diagnosis, and treatment. World journal of gastroenterology: WJG. 2014;20(36):12781.
CrossRef - Sindhu RK, Kaur P, Kaur P, Singh H, Batiha GE-S, Verma I. Exploring multifunctional antioxidants as potential agents for management of neurological disorders. Environmental Science and Pollution Research. 2022;29(17):24458-24477.
CrossRef - Vale FF, Oleastro M. Overview of the phytomedicine approaches against Helicobacter pylori. World Journal of Gastroenterology: WJG. 2014;20(19):5594.
CrossRef - Cai X, Li X, Jin Y, et al. Vitamins and Helicobacter pylori: an updated comprehensive meta-analysis and systematic review. Frontiers in Nutrition. 2022;8:781333.
CrossRef - Al-Tawalbeh D, Bustanji Y, Aburjai T, Al-Balas Q, Abu-Qatouseh L, Hamad I. Anti-Helicobacter activity of medicinal plants and probiotics as alternatives for Helicobacter pylori treatment. International Journal of Research in Pharmaceutical Sciences. 2020;11(3):3484-3489.
CrossRef - Gazerani P. Probiotics for Parkinson’s disease. International journal of molecular sciences. 2019;20(17):4121.
CrossRef - Kountouras J, Papaefthymiou A, Polyzos SA, et al. Impact of Helicobacter pylori-related metabolic syndrome parameters on arterial hypertension. Microorganisms. 2021;9(11):2351.
CrossRef - Ivyna de Araújo Rêgo R, Guedes Silvestre GF, Ferreira de Melo D, et al. Flavonoids-rich plant extracts against Helicobacter pylori infection as prevention to gastric cancer. Frontiers in Pharmacology. 2022;13:951125.
CrossRef - Sathianarayanan S, Ammanath AV, Biswas R, Sukumaran S, Venkidasamy B. A new approach against Helicobacter pylori using plants and its constituents: A review study. Microbial Pathogenesis. 2022;168:105594.
CrossRef - El-Saadony MT, Saad AM, Korma SA, et al. Garlic bioactive substances and their therapeutic applications for improving human health: a comprehensive review. Frontiers in immunology. 2024;15:1277074.
CrossRef - Abdi S, Ataei S, Abroon M, Sanaye PM, Abbasinazari M, Farrokhian A. A comprehensive review of the role of complementary and dietary medicines in eradicating Helicobacter pylori. Iranian Journal of Pharmaceutical Research: IJPR. 2022;21(1):e127030.
CrossRef
Abbreviations List
ATP: Adenosine Triphosphate
AMX: Amoxicillin
BQT: Bismuth-based Quadruple Therapy
BBB: Blood Brain Barrier
BDNF: Brain Derived Neurotrophic Factor
CHM: Chinese Herbal Medicine
COMT: Catechol-O-Methyl Transferase
CLR: Clarithromycin
CNS: Central Nervous System
COX-2: Cyclooxygenase-2
DPO-PCR: Dual-Priming Oligonucleotide-Based Multiplex Polymerase Chain Reaction
DPPH: 2,2-Diphenyl-1-Picrylhydrazyl
EGCG: Epigallocatechin-3-Gallate
GI: Gastrointestinal
H. pylori: Helicobacter pylori
HPA: Hypothalamic Pituitary-Adrenal
IBS: Irritable Bowel Syndrome
IL-8: Interleukin-8
MAO-B: Monoamine Oxidase-B
MENA: Middle East and North Africa
MTZ: Metronidazole
NO: Nitric Oxide
6-OHDA: 6-Hydroxydopamine
PD: Parkinson’s Disease
PDSD: Parkinson’s Disease Sleep Disorder
PPI: Proton Pump Inhibitor
PKC: Phosphorylated Protein Kinase C
ROS: Reactive Oxygen Species
SIBO: Small Intestinal Bacterial Overgrowth
SNpc: Substantia Nigra pars compacta
TCM: Traditional Chinese Medicine
TFC: Total Flavonoid Content
TNF-α: Tumor Necrosis Factor-alpha
TPC: Total Phenolic Content







