Manuscript accepted on :30-12-2025
Published online on: 21-01-2026
Plagiarism Check: Yes
Reviewed by: Dr. Swastika Maity
Second Review by: Dr. Sathyanarayana Namani
Final Approval by: Dr. Prabhishek Singh
Sadriddin Nurillo ugli Boboev1.3* , Inoyat Zulfiqorovich Zhumaev1
, Inoyat Zulfiqorovich Zhumaev1 , Pulat Bekmuratovich Usmanov1
, Pulat Bekmuratovich Usmanov1 , Abdisalim Abdikarimovich Zaripov1
, Abdisalim Abdikarimovich Zaripov1 , Shavkat Yusubovich Rustamov1
, Shavkat Yusubovich Rustamov1 , Shakhnoza Bakhtiyorovna Qurbonova1
, Shakhnoza Bakhtiyorovna Qurbonova1 , Eldor Bakhtiyor ugli Ibragimov1
, Eldor Bakhtiyor ugli Ibragimov1 , Sardor Bakhtiyor ugli Sobirov1
, Sardor Bakhtiyor ugli Sobirov1 , and Sherzod Niyatqobulovich Zhurakulov2
, and Sherzod Niyatqobulovich Zhurakulov2 .
.
1Department of Cell Biophysics, Institute of Biophysics and Biochemistry, National University of Uzbekistan, Tashkent, Uzbekistan
2Department of Alkaloids Chemistry, Institute of Chemistry of Plant Substances, Academy Sciences of Uzbekistan, Tashkent, Uzbekistan
3Faculty of Biology end ecology, National University of Uzbekistan named after Mirzo Ulugbek, Uzbekistan, Tashkent, Uzbekistan
*Corresponding Author’s E-mail sadriddin-2022@mail.ru
DOI : https://dx.doi.org/10.13005/bpj/3377
Abstract
Cardiovascular diseases are among the leading causes of death worldwide, according to the World Health Organization. A key pathogenic factor in ischemia and myocardial infarction is the disruption of Ca²⁺ ion homeostasis and impaired function of the sarcoplasmic reticulum (SR) proteins RyR2 and SERCA2a. Therefore, identifying biologically active substances that modulate Ca²⁺ transport via SERCA2a is of significant scientific and practical importance. In this study, the inotropic effects of the isoquinoline alkaloid F-4, the flavonoid dihydroquercetin (DHQ), and their conjugate DKV-6 were investigated using isolated rat heart papillary muscle preparations. Mechanographic results showed that F-4 (120 μM), DHQ (60 μM), and DKV-6 (50 μM) increased papillary muscle contractility by 32.9±3.1%, 51.4±3.4%, and 110.3±3.2%, respectively, compared to control. The post-rest potentiation (PRP) method was employed to assess changes in SR Ca²⁺ levels. The effects of these biologically active compounds (BACs) on RyR2 were studied using tetracaine and ruthenium red, while the role of SERCA2a was evaluated with cyclopiazonic acid (CPA). The results indicated that the positive inotropic effect is closely associated with SERCA2a function. Furthermore, the DKV-6 conjugate exhibits distinct anti-hypoxic activity, effectively counteracting hypoxia-induced impairments in myocardial contractility. Its antiarrhythmic effect appears to be mediated through SERCA2a activation.
Keywords
Conjugate; Dihydroquercetin; Hypoxia; Isoquinoline alkaloid; Papillary muscle; SERCA2a
Download this article as:| Copy the following to cite this article: Boboev S. N. U, Zhumaev I. Z, Usmanov P. B, Zaripov A. A, Rustamov S. Y, Qurbonova S. B, Ibragimov E. B. U, Sobirov S. B. U, Zhurakulov S. N. Role of F-4 Isoquinoline Alkaloid, Dihydroquercetin Flavonoid and Conjugate DKV-6 in Myocardial RyR2 and SERCA2a under Normal and Hypoxic Conditions. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Boboev S. N. U, Zhumaev I. Z, Usmanov P. B, Zaripov A. A, Rustamov S. Y, Qurbonova S. B, Ibragimov E. B. U, Sobirov S. B. U, Zhurakulov S. N. Role of F-4 Isoquinoline Alkaloid, Dihydroquercetin Flavonoid and Conjugate DKV-6 in Myocardial RyR2 and SERCA2a under Normal and Hypoxic Conditions. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/45lf5DZ |
Introduction
Cardiovascular diseases (CVD) are among the leading causes of disability and mortality worldwide. In this context, cardiovascular disorders pose a significant threat to human health and daily functioning. Among cardiovascular ailments, ischemic heart disease and cardiac arrhythmias are particularly prevalent and contribute substantially to the risk of sudden cardiac death.¹,² The progression of arrhythmias and ischemia leads to disturbances in key myocardial parameters, including heart rate and contractility.³,⁴
In the pathogenesis of these diseases, increased reactive oxygen species (ROS) production disrupts the functional activity of intracellular signaling systems and ATP-dependent processes, including Ca²⁺-ATPase, Na⁺/K⁺-ATPase, membrane K⁺ATP-channels, and Na⁺/Ca²⁺ exchange.⁵⁻⁷ Substantial evidence indicates that the effects of acute hypoxia on cardiomyocyte ion channels are associated with changes in channel phosphorylation states or cellular redox balance. The influence of hypoxia on channel regulation extends beyond direct effects, involving indirect pathways such as alterations in ROS levels.⁸⁻¹⁰
While most cardioprotective drugs modulate these parameters, many are unsuitable for clinical use due to adverse effects. Therefore, developing novel, safe, and potent cardioprotective agents is critically important.¹⁴⁻¹⁷ Biologically active compounds derived from plants represent promising candidates for developing effective therapeutics with minimal side effects.¹⁸ Among these, flavonoids¹⁹ and alkaloids²⁰ have been extensively studied.
In this study, we investigated the effects of the flavonoid dihydroquercetin and the isoquinoline alkaloid 1-(4′-methoxyphenyl)-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline (F-4), isolated by researchers at the Institute of Plant Chemistry, Academy of Sciences of the Republic of Uzbekistan, as well as their conjugate 2-(3,4-Dihydroxyphenyl)-6-{[6,7-dimethoxy-1-(4-methoxyphenyl)-3,4-dihydroisoquinolin-2(1H)-yl]methyl}-3,5,7-trihydroxychroman-4-one (DKV-6),²¹ on the contractile activity of cardiac papillary muscle under normal and hypoxic conditions (Figure 1).
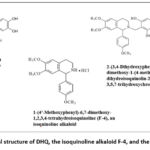 |
Figure 1: Chemical structure of DHQ, the isoquinoline alkaloid F-4, and the conjugate DKV-6.Click here to view Figure |
Materials and Methods
The experiments were conducted in the Cell Biophysics Laboratory of the Institute of Biophysics and Biochemistry at UzMU. All procedures involving animals were performed in accordance with the ethical principles of the Declaration of Helsinki and followed the guidelines established by the Council for International Organizations of Medical Sciences (CIOMS, 1985). The study strictly adhered to the institutional protocol titled “Bioethical Regulations for the Use of Laboratory Animals in Scientific Research” issued by the Institute of Biophysics and Biochemistry.
Myocardial contractile function was assessed in vitro using a mechanographic system. The contractile activity of isolated papillary muscles was recorded with a SI–BAM21–LC mechanographic setup (World Precision Instruments Inc., USA). The system included an SI–OHO2F force transducer. The mechanical signal from the muscle preparation, attached via an SI–KG20 piezoelectric sensor, was amplified by a SI–BAM21–LCB signal amplifier. Data were digitized using a Lab–Trax–4/16 converter (WPI, USA) and recorded in WLabScribe2 (*.iwxdata) format on a computer running the iWorx LabScribe2 software (iWorx Systems, Inc., USA) for subsequent primary mathematical and statistical analysis.
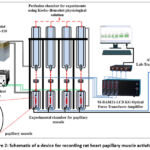 |
Figure 2: Schematic of a device for recording rat heart papillary muscle activity in vitro. |
Experimental Protocols
Male white outbred rats (200–250 g) were used to anesthetize the experimental subject, using sodium pentobarbital (40 mg/kg). Under deep anesthesia, animals were euthanized by cervical dislocation and subsequent decapitation in accordance with international ethical guidelines. The rib cage was then opened, the heart was carefully removed, and immediately placed in a petri dish containing Krebs’ solution. The left ventricle of the heart was opened and the papillary muscle was isolated. The prepared heart preparations were placed in a cell filled with Krebs solution with the following composition (mM): NaCl – 150; KCl – 4; CaCl2 – 1.8; MgCl2 – 1; NaHCO3 – 14; NaH2PO4 – 1.8; C6H12O6 – 11.5; (pH=7.4).
The flavonoid dihydroquercetin, isoquinoline alkaloid F-4, and conjugates DKV-6 studied during the research were provided by the staff of the Institute of Chemistry of Plant Substances of the Academy of Sciences of the Republic of Uzbekistan. In vitro, the PRP value, the SR RyR2 agonists tetracaine and ruthenium red, and the SERCA2a inhibitor cyclopiazonic acid (CPA) (Sigma Ltd) were used to assess the contractile activity of rat cardiac papillary muscle under normal and hypoxic conditions.
Statistical analysis
In the experiments, the force of contraction of the papillary muscle at the excitation frequency of 0.1-1 Hz was calculated as a percentage (%) of the maximum, and the average arithmetic value of 5 experiments was calculated. Statistical significance for the observed differences between the experimental and control groups was established using the Student’s t-test, with results considered significant if * –р<0.05, ** –р<0.01. The results of the study were analyzed using the statistical software OriginPro 2021 (OriginLab Corporation, USA).
In vitro Hypoxia model
A hypoxia model was used to evaluate the cardioprotective activity of the flavonoid dihydroquercetin, the isoquinoline alkaloid F-4, and the conjugates DKV-6. The hypoxia model was created by aerating a 95% O2; 5% CO2 Krebs solution with a 95% N2/5% CO2 gas for 60 minutes.22 The cardioprotective activity of the dihydroquercetin, the isoquinoline alkaloid 23 F-4, and the conjugates DKV-6 was studied under conditions where maximal changes in the parameters of rat papillary muscle contractile activity were observed after oxygen in Krebs solution was replaced with nitrogen for one hour. The heart muscle’s contractility is significantly impaired when it becomes hypoxic. The main reason for this is the disruption of calcium (Ca²⁺) transport systems and Ca²⁺ homeostasis in cardiomyocytes.24,25 This condition is mainly associated with the loss of function of the sarcoplasmic reticulum Ca²⁺-ATPase enzyme (SERCA2a), which plays a key role in maintaining Ca²⁺ homeostasis in cardiomyocytes.26-28
Results
Initial studies investigated how the conjugate of DHQ affected outcomes at varying concentrations29 (10-60 μM), the isoquinoline alkaloid 25 F-4 (5-120 μM), and DKV-6 (5-50 μM) on the contractile activity of rat cardiac papillary muscle were studied. At all doses of the studied biologically active substances, a positive inotropic effect was observed, increasing the force of cardiac papillary muscle contraction by 51.4±3.4%, 32.9±3.1%, and 110.3±3.2%, respectively, compared to the control (control was taken as 100%) (Figure. 3 A and B).
 |
Figure 3: A. Positive inotropic effect of biologically active compounds on papillary muscle contractile activity (Representative trace of isometric contraction). B. Dose-dependent positive inotropic effect of the isoquinoline alkaloid F-4, DHQ, and DKV-6 conjugate on papillary muscle contractile activity. |
The sarcoplasmic reticulum (SR) is an intracellular membrane-bound organelle that plays a key role in regulating Ca2+ concentration during contraction and relaxation. There are two important components of the SR involved in Ca2+ homeostasis: the SERCA2a and the RyR play a key role in regulating Ca2+ dynamics during myocardial contraction and relaxation. Ca2+ ions released from the SR is instrumental in the cardiac muscle’s contractile activity.30,31 Taking this into account, in the next experiments, the effect of the studied compounds on the SR Ca2+ transport systems were carried out using the post-rest potentiation method. In this case, when stimulation is stopped for 30 seconds and then given, the force of the first contraction (B1) increases, which occurs as a result of an increase in the amount of Ca2+ released from the SR. Therefore, the effect of isometric force on resting muscle contraction after cessation of stimulation is considered an index of the amount of Ca2+ released from the SR.32,33
In these experiments, in the presence of F-4 alkaloid (120 μM), DHQ (60 μM), and DKV-6 (50 μM) conjugate, the amplitude of papillary muscle contraction was observed to increase by 43.2±4.8%, 82.4±4.1%, and 93.1±3.8%, respectively, compared to the control after a rest period (30 sec). (Figure 4 A and B).
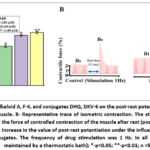 |
Figure 4: Effect of alkaloid A, F-4, and conjugates DHQ, DKV-6 on the post-rest potentiation value of rat cardiac papillary muscle. B- Representative trace of isometric contraction. |
The studies indicate that the F-4 alkaloid, DHQ, and DKV-6 conjugate exerts its positive inotropic action by enhancing the uptake and subsequent release of Ca2+ ions by the SR. RyR2 is a predominantly Ca2+ channel that plays a key role in cardiac contractility. Alterations in RyR2 function result in an increase in the inward potential.34-37 This process is associated with the release of Ca2+ ions from the SR.38-41 The probability of RyR opening depends on the cytosolic Ca2+ concentration, and Ca2+ ions bind to the activating part of the RyR. The opening of L-type Ca2+ channels and the increased probability of RyR opening following dyadic Ca2+ entry are the main mechanisms for the activation of Ca2+-induced Ca2+ release (CICR) during physiological activity. 42-44
Thus, to investigate whether RyR2 is implicated in the positive inotropic response to the F-4 alkaloid, DHQ, and DKV-6 conjugate, the effect on PRP values under tetracaine and ruthenium red incubation conditions was examined. In this case, the PRP values of F-4 alkaloid, DHQ, and DKV-6 conjugate in the presence of ruthenium red (10 μM) were 55,3±2,9%, 67.1±3.2%, and 72.1±4.1% (Figure 5 A).
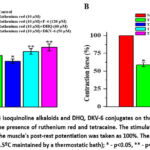 |
Figure 5: A, B. Effect of F-4 isoquinoline alkaloids and DHQ, DKV-6 conjugates on the value of papillary muscle post-rest potentiation in the presence of ruthenium red and tetracaine. |
In subsequent experiments, when tested in the presence of tetracaine (15 μM) in the medium, it was found that the effect of the F-4 alkaloid, DHQ, and DKV-6 conjugate on the post-rest potentiation value had a similar effect to the results of experiments conducted with ruthenium red (Figure 5B).
The findings of this research suggest that the DKV-6 conjugate exhibits a beneficial impact on cardiac contractility, which is based on the F-4 alkaloid and the DHQ flavonoid, is due to the low involvement of RyR2.
The study showed that the F-4 alkaloid increased the force of heart muscle contraction (positive inotropic effect). Furthermore, the DHQ and DKV-6 conjugate appears to influence how Ca2+ are stored within the SR. In order to validate this premise, we assessed the impact of the investigated BFB on SERCA2a. Experiments were conducted in the presence of the SERCA2a inhibitor cyclopiazonic acid (CPA).45-47
Research suggests that the isoquinoline alkaloid F-4, a compound formed by linking DHQ and DKV-6, enhances the contractile strength of rat heart papillary muscle by influencing how Ca2+ ions are stored within the sarcoplasmic reticulum.
To clarify this assumption, we investigated the effect of the studied isoquinoline alkaloid F-4, a conjugate of DHQ and DKV-6, on SERCA2a. Experiments were conducted in the presence of the SERCA2a inhibitor cyclopiazonic acid (CPA).48,49 To investigate how varying concentrations of CPA (from 1 to 15 μM) influenced papillary muscle contraction, control experiments were conducted. It was found that CPA at a concentration of 15 μM reduced the force of papillary muscle contraction by 80.7±4.8%. The half-maximal inhibitory concentration of CPA was IC50-5.6 μM (Figure 6).
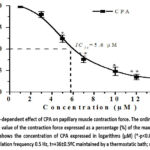 |
Figure 6: Dose-dependent effect of CPA on papillary muscle contraction force. |
In the next experiments, the effects of F-4 alkaloid (120 μM), DHQ (60 μM), and DKV-6 conjugate (50 μM) were examined in the presence of the half-maximal inhibitory concentration (IC50-5.6 μM) of the SERCA2a inhibitor CPA. Under these conditions, the amplitude of the contraction force of the papillary muscle of the rat heart was 57.6±3.5%, 60.2±5.3%, and 84.1±3.9%, respectively, compared to the control (Figure. 7 A and B).
 |
Figure 7: A. Effect of the isoquinoline alkaloid F-4, DHQ, and DKV-6 conjugate on papillary muscle contraction force in the presence of the SERCA2a inhibitor CPA (IC50-5.6 μM) in the incubation medium. |
According to the experimental results, F-4 alkaloid’s ability to increase heart muscle contractility, DHQ, and DKV-6 conjugate on papillary muscle contractile activity is explained by the fact that SR Ca2+ transport systems play an important role, with a small participation of RyR2 and a predominantly important role played by SERCA2a. Hypoxia causes a significant decrease in the force of heart muscle contraction. The main role in this is played by the disruption of Ca2+ homeostasis in cardiomyocytes, the function of Ca2+ transport systems, and the production of ATP and creatine phosphate, the main energy sources in cardiomyocytes.50-51
In this regard, an experimental model of hypoxia was used to study the effects of the F-4 alkaloid, DHQ, and DKV-6 conjugate on hypoxia-induced impairments in rat heart papillary muscle contractility. This model was performed by perfusion of rat heart papillary muscle preparations with modified Krebs saline (N2 95%/O2 5%). When rat heart papillary muscle preparation was perfused with modified Krebs solution for 60 minutes, it was found that the contractile force decreased to 30.4±3.1% compared to the control (Figure 8 A and B).
 |
Figure 8: A. Effect of hypoxia on rat heart papillary muscle contractile activity (Representative trace of isometric contraction). |
Further investigations examined how the F-4 alkaloid, DHQ, and the DKV-6 conjugate influenced papillary muscle contraction strength during hypoxic conditions, it was found that they restored the impairment of papillary muscle contraction activity caused by hypoxia to 47.3±3.1%, 52.2±3.8%, and 80.9±3.6% (Figure 9 A and B).
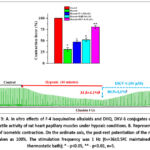 |
Figure 9: A. In vitro effects of F-4 isoquinoline alkaloids and DHQ, DKV-6 conjugates on the contractile activity of rat heart papillary muscles under hypoxic conditions. B. Representative trace of isometric contraction. |
Based on the results of the above experiments, it can be said that the F-4 alkaloid, DHQ, and DKV-6 conjugate effectively eliminates disorders in the contractile activity of the heart muscle as a result of hypoxia, demonstrating strong cardioprotective properties.
Ischemic heart disease leads to impaired ATP synthesis, including impaired SR Ca2+-ATPase (SERCA2a) activity. In this regard, the effect of the indole alkaloids under study on SERCA2a in myocardial cells under hypoxic conditions was examined. When the effects of F-4 alkaloid (120 μM), DHQ (60 μM), and DKV-6 conjugate (50 μM) in the presence of the SERCA2a inhibitor CPA (IC50-5.6 μM) under hypoxic conditions were tested, the papillary muscle contraction force was 19.6±3.5%, 25.2±5.3%, and 35.6±3.4%, respectively, compared to the control (Figure. 10 A and B).
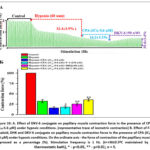 |
Figure 10: A. Effect of DKV-6 conjugate on papillary muscle contraction force in the presence of CPA (IC50-5.6 μM) under hypoxic conditions. (representative trace of isometric contraction) B. Effect of F-4 alkaloid, DHK and DKV-6 conjugate on papillary muscle contraction force in the presence of CPA (IC50-5.6 μM) under hypoxic conditions. |
The results of these experiments showed that the F-4 alkaloid, DHQ, and DKV-6 conjugate can eliminate hypoxia-induced dysfunction of SERCA2a and normalize changes in rat heart papillary muscle contractile activity.
Discussion
Our results demonstrate that the isoquinoline alkaloid F-4, dihydroquercetin (DHQ), and the DKV-6 conjugate synthesized from these compounds exert a positive inotropic effect on the contractile activity of rat heart papillary muscles. Analysis revealed that the DKV-6 conjugate had the most pronounced effect among the tested substances, significantly increasing papillary muscle contraction force. This enhanced effect may be attributed to a synergistic interaction between the flavonoid and alkaloid components within the conjugate.
The sarcoplasmic reticulum proteins RyR2 and SERCA2a play crucial roles in maintaining Ca²⁺ homeostasis and regulating intracellular Ca²⁺ dynamics in cardiomyocytes. In experiments examining the effects of F-4, DHQ, and DKV-6 on RyR2 function, we found that the positive inotropic effects of these compounds in the presence of tetracaine and ruthenium red were not primarily mediated by RyR2. Conversely, experiments using cyclopiazonic acid (CPA) indicated that the positive inotropic effects of F-4, DHQ, and DKV-6 on papillary muscle contractility are mediated through modulation of SERCA2a function.
Under hypoxic conditions, the force of cardiac papillary muscle contraction decreased by up to 70%. This reduction is primarily due to disruption of the Ca²⁺ transport system and impairment of ATP-dependent processes. However, treatment with F-4, DHQ, and particularly the DKV-6 conjugate partially restored contractile force during hypoxia, suggesting their potential role in restoring SERCA2a activity and normalizing Ca²⁺ homeostasis.
These data further indicate that the DKV-6 conjugate exhibits superior cardioprotective properties under hypoxic conditions compared with F-4 and DHQ alone. The findings suggest that these compounds, especially the DKV-6 conjugate, hold therapeutic potential for the treatment of ischemic heart disease.
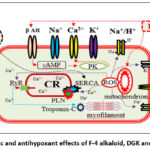 |
Figure 11: Inotropic and antihypoxant effects of F-4 alkaloid, DGK and DKV-6 conjugate. |
Conclusion
Compared to the F-4 isoquinoline alkaloid, DHQ flavonoid, the DKV-6 conjugate is recognized to increase the force of contraction in the papillary muscle of a rat heart to a greater extent. Under conditions of hypoxia, changes occur in the function of ion channels, which are mainly associated with ATP. Considering this, the F-4 isoquinoline alkaloid, DHQ and DKV-6 conjugate studied above was induced by hypoxia. SR effectively eliminates disorders in the function of the Ca2+-ATPase system and normalizes changes in the contractile activity of rat cardiac papillary muscles. This can restore the activity of special signaling systems that provide communication between the SR, regulate the concentration of intracellular Ca2+ ions, and the function of the SR.
Acknowledgment
This work was supported by the Science and Technology Development Coordination Committee.
Funding source
This work was supported by the Ministry of Higher Education, Science and Innovation of Uzbekistan (grant AL-9224104346).
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The experimental protocols complied with the standards and requirements for the humane treatment of animals and the provisions of the Ethical Commission of the IBB at the National University of Uzbekistan. (Protocol No. 7 BEC/IBB-NUU of 04/07/2022) on the use of laboratory animals. Isolated cardiac papillary muscle preparations were obtained using a specific method.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Boboev Sadriddin Nurillo Ugli: Conceptualization, Methodology, Writing – review & editing, Writing – Original Draft, Supervision.
- Zhumaev Inoyat Zulfiqorovich: Methodology, Writing – review & editing, Writing – original draft, Validation, Formal analysis, Project Administration.
- Usmanov Pulat Bekmuratovich: Conceptualization, Methodology, Writing – review & editing, Supervision, Resources, Funding acquisition.
- Zaripov Abdisalim Abdikarimovich: Writing – review & editing, Writing – Original Draft.
- Rustamov Shavkat Yusubovich: Visualization, Resources.
- Qurbonova Shakhnoza Bakhtiyorovna: Visualization, Resources
- Eldor Bakhtiyor ugli Ibragimov: Visualization, Resources
- Sardor Bakhtiyor ugli Sobirov: Visualization, Resources
- Zhurakulov Sherzod Niyatkobulovich: isoquinoline alkaloid 1-(4¢-methoxyphenyl)-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline (F-4) isolated by the staff of the Institute of Chemistry of Plant Substances of the Academy of Sciences of the Republic of Uzbekistan, as well as the conjugate 2-(3,4-Dihydroxyphenyl)-6-{[6,7-dimethoxy-1-(4-methoxyphenyl-3,4-dihydroisoquinolin-2(1H)-yl]methyl}-3,5,7-trihydroxychroman-4-one (DKV-6).
Reference
- Di Cesare M, Perel P, Taylor S. et al. The Heart of the World. Global Heart. 2024;19(1):1-13.
CrossRef - Uzbekov V V, Abdullaev B F, Jumayev I Z, et al. Comparative study of the antiarrhythmic activity of liposomal forms of lappaconitine hydrobromide and its complex with glycyrrhizic acid monoammonium salt in the aconitine arrhythmia model // Pharmaceutical Chemistry Journal. 2023;56:1327–1332.
CrossRef - Oshchepkova Yu I, Uzbekov V V, Jumayev I Z, et al. Antiarrhythmic Activity Of Ethacizine Hydrochloride And Its Supramolecular Complexes With Glycyrrhizic Acid And Its Monoammonium Salt // Pharmaceutical Chemistry Journal, 2025;59(6):21-26.
CrossRef - Oshchepkova Yu I, Uzbekov V V, Jumayev I Z, et al. Comparative study of antiarrhythmic and inotropic activity of amiodarone hydrochloride and its complexes with glycyrrhizic acid and monoammonium salt of glycyrrhizic acid. Eksperimental’naya i Klinicheskaya Farmakologiya. 2023:86(4):15–22.
- Gorenek B, Lundqvist C B, Terradellas J B, et al. Cardiac arrhythmias in acute coronary syndromes: position paper from the joint EHRA, ACCA, and EAPCI task force. 2014;16(11):1655-73.
CrossRef - Thygesen K, Alpert J S, Jaffe A S, et al. Fourth Universal Definition of Myocardial Infarction. Circulation. 2018;138(20):e618-e651.
CrossRef - Michiels C. Physiological and Pathological Responses to Hypoxia. American Journal of Pathology. 2004;164(6):1875-1882.
CrossRef - Mukandala G, Tynan R, Lanigan S and O’Connor J.J. The Effects of Hypoxia and Inflammation on Synaptic Signaling in the CNS. Brain Sci. 2016;6(6):1-14
CrossRef - Gusarova G A, Trejo H E, Dada L A, et al. Hypoxia Leads to Na, K-ATPase Downregulation via Ca2+ Release-Activated Ca2+ Channels and AMPK Activation. Molecular and cellular biology, 2011;31:3546–3556.
CrossRef - Xuan C A, Peres de la Lastra J M, Plou F J, Peres-Lebena E. The Chemistry of Reactive Oxygen Species (ROS) Revisited: Outlining Their Role in Biological Macromolecules (DNA, Lipids and Proteins) and Induced Pathologies. J. Mol. Sci. 2021;22(9), 4642.
CrossRef - Zhumaev I Z, Boboev S N, Usmanov P B, et al. Role of RyR2 and SERCA2a in the Cardioprotective Effects of Vincanine and Pyrazoline Alkaloids. Trends in Sciences, 2024; 22(1):8626.
CrossRef - de Almeida AJPO, de Oliveira JCPL, da Silva Pontes LV, et al. ROS: Basic Concepts, Sources, Cellular Signaling, and its Implications in Aging Pathways. Oxidative Medicine and Cellular Longevity. 2022;1225578:1-23.
CrossRef - Veit F, Pak O, Brandes RP, Weissmann N. Hypoxia-Dependent Reactive Oxygen Species Signaling in the Pulmonary Circulation: Focus on Ion Channels. Antioxidants & redox signaling. 2015;22(6):537-552.
CrossRef - Zaripov A A, Zhumaev I Z, Usmanov PB, et al. Mechanism of Vasorelaxant Action of Isoquinoline Alkaloid F-19 on Rat Aorta. Biomedical & Pharmacology Journal, 2025;18(2):1580-1589.
CrossRef - Forrester SJ, Kikuchi D S, Hernandes M S, et al. Reactive Oxygen Species in Metabolic and Inflammatory Signaling. Circ Res. 2018;122(6):877–902.
CrossRef - Ziyavitdinov Zh F, Ishimov U Zh, Berdiev N Sh, et al. Supramolecular complex of lappaconitine hydrobromide and the monoammonium salt of glycyrrhizic acid: synthesis, physicochemical characteristics, and antiarrhythmic activity // Pharmaceutical Chemistry Journal, 2022;56(2):25–32.
CrossRef - Chaachouay N, Zidane L. Plant-Derived Natural Products: A Source for Drug Discovery and Development. Drugs Drug Candidates. 2024;3:184–207.
CrossRef - Zhumaev I Z, Boboev S N, Usmanov P B, et al. Mechanism of Positive Inotropic Effect of Vincanine on Cardiac Muscle Contraction Activity. Biomedical & Pharmacology Journal, 2022;15(4):2309-2316.
CrossRef - Zaripov A A, Jumayev I Z, Usmanov P B, et al. Protective Effect of DHQ-11 against Hypoxia-induced Vasorelaxation. Trends in Sciences, 2024;21(11):8192.
CrossRef - Bakhtiyorovna Q S, Zulfiqorovich Z I, Ugli B S N, et al. Antiarrhythmic Effect of 1-(3ˊ-Bromophenyl)-6,7-methylenedioxy-1,2,3,4-tetrahydroisoquinoline Biomed Pharmacol J 2025;18(3):2463-2473.
CrossRef - Zhurakulov Sh N, Babkin V A, Chernyak E I, et al. Aminomethylation of 1-aryl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinolines by dihydroquercetin. Chemistry of natural compounds. 2015;51(1):57-61.
CrossRef - Batista P R, Vassallo DV, Simões M R, Lima M L. Cardioprotective Solutions Exposure For 1 Hour in Hypoxia and Low Temperatures Affects Vascular Reactivity Differently. Braz J Cardiovasc Surg. 2021;36(2):201-211.
CrossRef - La Padula P H, Czerniczyniec A, Bonazzola P, et al. Acute hypobaric hypoxia and cardiac energetic response in prepubertal rats: Role of nitric oxide. Exp Physiol. 2021;106(5):1235-1248.
CrossRef - Jumayev I, Usmanov P, Rustamov Sh, Zhurakulov Sh. Comparative inotropic effects of the some isoquinoline alkaloids // Biomedical and Pharmacology Journal. 2020;13(1):325-333
CrossRef - Wang R, Wang M, He S, Sun G, Sun X. Targeting Calcium Homeostasis in Myocardial Ischemia/Reperfusion Injury: An Overview of Regulatory Mechanisms and Therapeutic Reagents. Front Pharmacol. 2020;11:872.
CrossRef - Lipskaia L, Chemaly ER, Hadri L, Lompre AM, Hajjar RJ. Sarcoplasmic reticulum Ca2+ ATPase as a therapeutic target for heart failure. Expert Opin Biol Ther. 2010;10(1):29-41.
CrossRef - Xu H, Van Remmen H. The SarcoEndoplasmic Reticulum Calcium ATPase (SERCA) pump: a potential target for intervention in aging and skeletal muscle pathologies. Skelet Muscle. 2021;11(1):25.
CrossRef - Zimaand AV, Mazurek SR, Functional impact of ryanodine receptor oxidation on intracellular calcium regulation in the heart. Reviews of Physiology, Biochemistry and Pharmacology. 2016;171:39-62.
CrossRef - Usmanov PB,Jumayev IZ, Rustamov Sh Yu,et al. The combined inotropic and vasorelaxant effect of DHQ-11, a conjugate of flavonoid dihydroquercetin with isoquinoline alkaloid 1-aryl-6,7-dimethoxy-1,2.3,4-tetrahydroisoquinoline. Biomedical and Pharmacology Journal. 2021;14(02):651-661
CrossRef - Pieske B, Maier LS, Schmidt-Schweda S. Sarcoplasmic reticulum Ca2+ load in human heart failure. Basic Res Cardiol. 2002;97(1):63-71.
CrossRef - Kho C. Targeting calcium regulators as therapy for heart failure: focus on the sarcoplasmic reticulum Ca-ATPase pump. Front Cardiovasc Med. 2023;10:1185261.
CrossRef - Posterino GS, Lamb GD. Effect of sarcoplasmic reticulum Ca2+ content on action potential-induced Ca2+ release in rat skeletal muscle fibres. J Physiol. 2003;551(1):219-37.
CrossRef - Pieske B, Sütterlin M, Schmidt-Schweda S, et al. Diminished post-rest potentiation of contractile force in human dilated cardiomyopathy. Functional evidence for alterations in intracellular Ca2+ J Clin Invest. 1996;98(3):764-76.
CrossRef - Yagi S, Becker PL, Fay FS. Relationship between force and Ca2+ concentration in smooth muscle as revealed by measurements on single cells. Proc Natl Acad Sci U S A. 1988;85(11):4109-4113.
CrossRef - Yano M, Ikeda Y, Matsuzaki M. Altered intracellular Ca2+ handling in heart failure. J Clin Invest. 2005;115(3):556-564.
CrossRef - Gyurke S, Carnes C. Dysregulated sarcoplasmic reticulum calcium release: potential pharmacological target in cardiac disease. Pharmacol Ther. 2008;119(3):340-354.
CrossRef - Belevych AE, Radwański PB, Carnes CA, Gyurke S. ‘Ryanopathy’: causes and manifestations of RyR2 dysfunction in heart failure. Cardiovasc Res. 2013;98(2):240-247.
CrossRef - Connell P, Word TA, Wehrens XHT. Targeting pathological leak of ryanodine receptors: preclinical progress and the potential impact on treatments for cardiac arrhythmias and heart failure. Expert Opin Ther Targets. 2020;24(1):25-36.
CrossRef - Lanner JT, Georgiou DK, Joshi AD, Hamilton SL. Ryanodine receptors: structure, expression, molecular details, and function in calcium release. Cold Spring Harb Perspect Biol. 2010;2(11):a003996.
CrossRef - Walweel K, Laver DR. Mechanisms of SR calcium release in healthy and failing human hearts. Biophys Rev. 2015;7(1):33-41.
CrossRef - Overend CL, O’Neill SC, Eisner DA. The effect of tetracaine on stimulated contractions, sarcoplasmic reticulum Ca2+ content and membrane current in isolated rat ventricular myocytes. J Physiol. 1998;507(3):759-769.
CrossRef - Laver DR. Regulation of the RyR channel gating by Ca2+ and Mg. Biophys Rev. 2018;10(4): 1087-1095.
CrossRef - Frank KF, Bulck B, Erdmann E, Schwinger RH. Sarcoplasmic reticulum Ca2+-ATPase modulates cardiac contraction and relaxation. Cardiovasc Res. 2003;57(1):20-7.
CrossRef - Periasamy M, Bhupathy P, Babu GJ. Regulation of sarcoplasmic reticulum Ca2+ ATPase pump expression and its relevance to cardiac muscle physiology and pathology. Cardiovasc Res. 2008;77(2):265-273.
CrossRef - Kaasik A, Kuum M, Joubert F, et al. Energetic state is a strong regulator of sarcoplasmic reticulum Ca2+ loss in cardiac muscle: different efficiencies of different energy sources. Cardiovascular Research. 2009;83(1):89–96.
CrossRef - Sitsel A, De Raeymaecker J, Drachmann N.D., et al. Structures of the heart specific SERCA2a Ca2+-ATPase. The EMBO Journal. 2019;38:1-17
CrossRef - Zhao Y, Xiong W, Li C, et al. Hypoxia-induced signaling in the cardiovascular system: pathogenesis and therapeutic targets. Signal Transduction and Targeted Therapy 2023;8(431):1-42.
CrossRef - Rosa A O, Movafagh S, Cleemann L, Morad M. Hypoxic regulation of cardiac Ca2+ channels: possible role of haem oxygenase. J Physiol 2012;590(17):4223–4237.
CrossRef - Eisner D A, Caldwell J L, Kistamás K, Trafford A W. Calcium and Excitation-Contraction Coupling in the Heart. Circ Res. 2017;121(2):181–195.
CrossRef - Pikor D, Hurła M, Słowikowski B, et al. Calcium Ions in the Physiology and Pathology of the Central Nervous System. J. Mol. Sci. 2024;25(13133):1-24.
CrossRef







