Manuscript accepted on :22-12-2025
Published online on: 20-01-2026
Plagiarism Check: Yes
Reviewed by: Dr. Hany Akeel
Second Review by: Dr. Noora Thamer Abdulaziz
Final Approval by: Dr. Patorn Piromchai
Faruk Alam1 , Alindam Ghosh2,1
, Alindam Ghosh2,1 , Arpan Sen2,3
, Arpan Sen2,3 , Debraj Dey2
, Debraj Dey2 , Soumya Sunder Ghora2
, Soumya Sunder Ghora2 , Saurav Guchhait2
, Saurav Guchhait2 , Pakhi Pandey2
, Pakhi Pandey2 . Avik Dutta2,1*
. Avik Dutta2,1* and Prithviraj Chakraborty3
and Prithviraj Chakraborty3
1Department of Pharmaceutical Science, Assam down Town University, Guwahati, India.
2 Department of Pharmaceutical Science, DmbH Institute of Medical Science, Hooghly, West Bengal, India.
3Department of Pharmaceutical Science, Royal School of Pharmacy, The Assam Royal Global University, Guwahati- Assam, India
Corresponding Author Email: avikdutta2010@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3339
Abstract
The immune system is a simple network of natural biological systems that protects an organism from diseases. It can discriminate between the organism's healthy tissues and unhealthy ones, such as various pathogens, including viruses, parasitic worms, cancer cells, and things like wood splinters. Living thing has an immune system in the form of enzymes. Ancient plants and animals developed additional fundamental immune systems, which are still present in their contemporary offspring. The complement system, phagocytosis, and antimicrobial peptides, such as defensins, are small cysteine-rich cationic proteins found in all cellular life, including plants, fungi, vertebrates, and invertebrates. Immunotherapy is a type of treatment that uses substances to stimulate or suppress the immune system to help fight diseases, such as cancer, Autoimmune diseases, Allergies, and the rejection of transplanted organs. Immunotherapy has been applied for years to treat allergies and prevent the body from rejecting transplanted organs. Immunotherapy is a class of treatments that harnesses a person's immune system to help kill cancer cells. The Immune system's function and immune therapy for treating liver cancer and collateral cancers are covered in this article. It draws attention to growths in targeted medicines, checkpoint inhibitors, and CAR T-cell therapy, emphasizing how these may strengthen immune responses and boost patient outcomes. For these cutting-edge cancer therapies to be optimized, ongoing clinical studies are essential.
Keywords
Collateral cancer; Hepatocellular carcinoma; Immune system; Immunotherapy; Liver cancer
Download this article as:| Copy the following to cite this article: Alam F, Ghosh A, Sen A, Dey D, Ghora S. S, Guchhait S, Pandey P, Dutta A, Chakraborty P. A Comprehensive Review on the Immune System and Immune Therapy in Liver Cancer and Colorectal Cancer. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Alam F, Ghosh A, Sen A, Dey D, Ghora S. S, Guchhait S, Pandey P, Dutta A, Chakraborty P. A Comprehensive Review on the Immune System and Immune Therapy in Liver Cancer and Colorectal Cancer. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/4jNImNr |
Introduction
A sophisticated network of organs, tissues, cells, and the substances they generate to help the body fight against infections and diseases. The immune system is composed of white blood cells and tissues and organs of the lymphatic system. Primary lymphoid organs, which specifically produce lymphocytes, include the bone marrow and the thymus (Null et al., 1). Organs, cells, and proteins make up the complex organisation known as the immune system, which protects the body’s cells against infection. In order to quickly recognise and eradicate the microbe should it re-enter the body, the immune system keeps track of every germ (microorganism) it has ever eradicated. It is composed of different cells, proteins, and organs O’Brien et.al2.The body is protected against disease by the immune system and these specialized cells. The immune system combats bacteria in the body’s tissues, on the skin, and in internal fluids like blood. It consists of the adaptive (specialized) immune system and the innate (general) immune system. Although they perform separate duties, the two systems are closely related. Humans have three different types of immunity: innate, adaptive, and passive. Everyone is born with natural immunity, sometimes referred to as innate immunity, which is a comprehensive protection Matsuzaki G et.al 3. The skin, arteries, spleen, lymph nodes, bone marrow, and thymus are some of the tissues and organs that are essential to the proper functioning of the immune system. Rather than any particular heritable genes, environmental variables are linked to a large proportion of colorectal cancer (CRC) occurrences Bardhan K et. al.4. Risk factors include persistent intestinal inflammation, which occurs before tumors grow, sensitivity to certain gut bacteria, diseases, and mutagens occurring in food and the environment.
Types of the immune system
The body’s protection against infections is the immune system. The immune system, or MYOON, protects us against illnesses and maintains our health. There are two main parts to the immune system:
The innate immune system
The body uses this, often referred to as the nonspecific immune system, as its first line of defense against common infections. It includes proteins, mucous membranes, skin, and immune system cells. It starts working at birth and is inherited. Innate immunity can be classified into four categories: endocytic and phagocytic, physiological (temperature, low pH, and chemical mediators), anatomical (skin and mucous membranes), and inflammatory. In Table No. 1 shows the main nonspecific defence methods for each. Many studies have been conducted on the immune system’s cells and defense mechanisms that avoid anatomical obstacles. Innate immunity depends on pattern recognition receptors (PRRs), which are the basis for the various immune systems’ capacity to identify and respond rapidly to a variety of pathogens that have similar characteristics, or pathogen-associated molecular patterns (PAMPs).
Table 1: Non-specific host-defence mechanisms for obstacles to innate immunity Liu C et.al.5.
|
Barrier |
Mechanism |
|
Anatomic |
|
|
Skin |
The mechanical barrier prevents microorganisms from entering. An acidic environment (i.e., pH 3–5) hinders the development of microbes |
|
Mucous membrane |
Microbes and normal flora compete for attachment locations Foreign microorganisms are trapped by mucus Microbes are expelled from the body by Cilia. |
|
Physiologic |
|
|
Temperature |
Certain infections are inhibited in their proliferation by the fever response and body temperature. |
|
Low pH |
The overwhelming majority of undigested microorganisms are destroyed by the stomach’s acidic pH. |
|
The use of chemical intermediaries
|
Interferon stimulates antiviral defenses in non-infected cells; lysozyme cuts the bacterial cell wall; and complement lyses microorganisms or promotes phagocytosis. |
|
Phagocytic/endocytic barriers |
Through endocytosis, different cells absorb and break down superfluous macromolecules. |
|
swellingand pain obstacles |
When vascular fluid carrying serum protein that has antibacterial properties leaks due to tissue injury or infection, phagocytic cells infiltrate the afflicted region. |
The several cell types that comprise the immune system include innate lymphocytes, natural killer (NK) cells, mast cells, basophils, eosinophils, phagocytes (macrophages and neutrophils), and dendritic cells. Phagocytes are mostly composed of two cell types: neutrophils and macrophages. Both kinds of cells perform comparable tasks, which include phagocytosing bacteria and eliminating them via different bactericidal techniques. Granules and enzymatic pathways found in neutrophils’ phagocytic material aid in the destruction of harmful microorganisms. Unlike short-lived neutrophils, macrophages are long-lived cells that aid in phagocytosis by transferring antigens to T cells. (Table 2).
Table 2: Characterization and specific roles of cells that makeup innate immunity Avelino CD et.al.6
|
Types of Cells |
Presence quality in adults Human body(%) |
Types of Nucleus |
Role |
Time Taken |
Prime Suspects |
|
Eosinophil |
1-6% |
Bi-lobed |
Degranulation: Release of enzymes, growth factors, cytokines |
8-12 days(circulate for 4-5hours) |
Parasites Several allergic tissues |
|
Monocyte |
2-6% |
Kidney shaped |
To start an immune reaction, develop into dendritic cells and macrophages. |
Varies from hours to days |
Various |
|
Mast cell |
Common in tissues |
Central, single-lobed |
Degranulation Release of histamine, enzymes, and cytokines |
Varies from Months to Years |
Parasites; Various allergic tissues |
|
Lymphocytes (T cells) |
20-40% |
Deeply staining, eccentric |
T helper (Th) cells(CD4+): immune; Response mediatorsCytotoxic T cells(CD8+): celldestruction |
It may takes a week longer usually, but it may take more than uasual |
The cells include intracellular bacteria, tumor and virus-infected cytotoxic T cells, and natural killer cells. |
|
Neutrophil |
40-75% |
Multi-lobed |
Phagocytosis and Degranulation |
hours up to few days |
Bacteria and Fungi |
|
Natural killer (NK)cell |
15% (varies) of circulating lymphocytes and tissues |
Single-lobed |
Granzymes and perforin, which trigger apoptosis, are produced when infected cells are killed. |
7-10 days |
Viruses Tumor cells |
|
Macrophage* |
Varies |
Varies |
Phagocytosis and T cell presentation of antigens |
Months to years |
Various |
|
Basophil |
< 1% |
Bi– or tri-lobed |
Degranulation: Release of histamine, enzymes, and cytokines |
Lifetime uncertain, likely a few hours to a few days |
Numerous allergic tissues |
Dust cells (pulmonary alveolus), histiocytic (connective tissue), Kupffer cells (liver), microglial cells (neural tissue), epithelioid cells (granulomas), osteoclasts (bone), mesangial cells (kidney)
The adaptive immune system
This system, which is also known as the sophisticated immune system, is formed when the body is exposed to chemicals or microorganisms. It produces antibodies, which are unique proteins that shield the body from certain infections. It is well recognized that individuals suffering from IBD, ulcerative colitis (UC), and Crohn’s disease (CD), are more likely to develop colorectal cancer.This kind of cancer is thought to be a prime illustration of how inflammation can lead to the formation of cancer since it originates in the setting of IBD. Chronic inflammation-induced oxidative stress breaks down DNAturning on genes that encourage the growth of tumors and turning off ones that prevent it Tacke F et.al.7
The body’s immunological response and the gut microbiota and its metabolites also play a role in this inflammatory and cancer-causing process. The immune system and surrounding tissue can have an impact on the clonal proliferation of somatic epithelial cells. This process includes both genetic and epigenetic modifications. Cell of signalling survival, and encourage the development of new blood vessels can raise the risk of cancer due to inflammation. Thus, these substances create mutations in genes linked to cancer, change metabolic processes, and increase inflammatory levels. Dysplasia of the intestinal epithelial cells follows, which makes the illness worse by encouraging cell division and many forms of cell death. After the immunosuppressive TME forms, tumor cells may make it difficult for the immune system to recognize themFor the purpose of coping Low Glucose from the metabolism, they might also alter their energy metabolism. Carcinogenic programs, immune system evasion, genomic instability, and energy metabolism reprogramming, are facilitated by metalloproteinases and modification of the extracellular matrixItzkowitz S.H.et al.8The immune system is the body’s defensive mechanism, reacting to infections quickly and broadly. It encompasses the complement system, inflammation, and surface barriers.Frank MM et al, Lukasch B et al., Zouridakis EG et.al.9-11 Table 3 explains different types of immune systems.
Table 3: Various kinds of immune systems.
|
Adaptive defence |
Immune globulin |
Phagocytes
|
complement design |
Dendritic cells |
Lymphocytes
|
|
This is an even more potent and targeted reaction that follows infection. It consists of antibodies and lymphocytes. |
This is also called antibody-mediated immunity and is a component of the adaptive immune system. |
As innate immune system members, these cells aid in the activation of the adaptive immune system.
|
This innate immune system component aids in the removal of infections via phagocytes and antibodies. |
The innate and adaptive immune systems communicate with one through these cells. |
These white blood cells respond to foreign invaders in the body.
|
The human body has three primary defense mechanisms against bacteria, viruses, and fungus, among other external invaders. Physical and chemical barriers, non-specific innate reactions, and specialized adaptive responses are the three lines of defense for the immune system.
Role of the immune system
Preventing outside invaders-like germs
Eliminating intruders.
Reducing the amount of damage.
Healing physical injuries.
Adjusting to fresh difficulties and dangers.
Medications used for Immunotherapy either immune system stimulation or suppression to aid in the fight against illness, likecancer, various infections, and other illnessesOtt PA et al.12
Potential modes of action
Immunotherapies:Immunotherapies that boost the immune system to combat illness are known as activation therapies. These medications prevent T cells from activating and eliminating malignant cells by severing the link between checkpoint proteins and other proteinsOsei-Bordom et al..13Colorectal cancer (CRC), the second leading cause of cancer-related deaths globally and the third most prevalent malignant carcinoma, most often spreads to distant metastases in the liver. The majority of CRC patients with liver metastases have a miserable prognosis and are not appropriate candidates for surgery.Furthermore, inadequate results are achieved from standard anticancer techniques such as chemotherapy, radiation, targeted therapy, and surgery. By boosting the host’s anticancer immune activity, immunotherapy has demonstrated favourable results in the treatment of several malignancies in recent years. It may also prove to be a new, successful treatment for colorectal cancer metastases to the liver. Applying immunotherapy to CRC with liver metastases is still difficult, though.
Immunosuppressive therapies: Immunosuppressive therapy is used to treat autoimmune diseases, organ transplants, and aplastic anaemia by reducing the immunological response in the body. These medical interventions lower or inhibit immunological functionHaanen JB et al..14This overview looks at how the immunosuppressive environment and microenvironment of the liver promote the formation of tumors. The most current developments in immunotherapy for colorectal liver metastases are also put together, and immunotherapy has been identified as a therapeutic option with an exciting prospect in Advanced Clinical ScienceNicholson LB et al..15.
Hepatic cancer: The most prevalent kind of liver cancer, advanced hepatocellular carcinoma (HCC), may be treated with immunotherapy. Hepatocellular carcinoma cell inhibitors and specific antibodies work together to aid the immune system in eliminating cancerous cells. Immunotherapy is not always an option for people with liver damage or a history of hepatitis infections, though, as it can harm healthy liver cellsGreten TF et al..16
Colorectal cancer: Although there are still hurdles, immunotherapy has demonstrated potential in treating colorectal cancer (CRC) liver metastases. According to the literature, individuals with colorectal cancer may benefit better from immunotherapy if their liver metastases are treated first. Preliminary research indicates that CRC might not react to PD-1 or PD-L1 suppression, and early evidence implies that anti-CTLA-4 might not be useful as a stand-alone treatmentYaghoubi N et al.17
Immune System in Liver and Colorectal Cancer
Liver/Hepatic Cancer: The immune system plays a complex role in liver cancer, including:
Evasion Immune: Due to prolonged inflammation and liver cirrhosis, which can result in immunological suppression, liver cancer frequently affects the immune system’s tolerance. Tumor Microenvironment: Within the immunosuppressive components produced by malignancies.These factors combined to prevent effective immune responsesYang M et al.18
Drug Molecules: Immunological escape is facilitated by the high expression of immunological drug molecules in liver cancer cells, such as PD-L1Yaghoubi N et al.17.17
Chronic Infection: Because the hepatitis B and C viruses can affect immune system, they pose a serious riskPol Set al.19
Signaling Pathways and Immune cells in the Tumor Microenvironment
Signalling networks and immune cells. Macrophages and lymphocytes facilitate the progression from inflammation to tumor growth while also contributing to mucosal damage brought on by inflammation. Intestinal inflammation is primarily caused by alterations, along with lymphocyte dysregulation and cell proliferation. Macrophages have a crucial but complex role in the progress of colorectal cancer. While macrophages of M2 are anti-inflammatory and involved in the elimination of waste and apoptotic cells, M1 macrophages are antibacterial and pro-inflammatoryMantovani A et al.20
M1 enhances tumor immunity during carcinogenesis, but M2 accelerates tumorigenesis and metastasis, especially when present in tumor-associated macrophages (TAMs)Natoli M et al.21(Fig. 1) In Figure 1The immune system’s biological components in the tumor microenvironment of colorectal cancer are illustrated. while the cells and products released by the immune system that are involved in the anticancer response are displayed on the left. Preliminary stages of CAC, prolonged M1 macrophage over activation and pro-inflammatory responses elevate and cytokine and chemokine competition. Various environmental signals stimulate these characteristics. Macrophage metabolism restriction caused by a shortage of amino acids and glucose in the TME will lead to decreased synthesis of Metabolites of succinate. NADPH and reactive oxygen species inhibit M1 macrophage activity. In contrast, M2 macrophages have a TCA cycle with high levels of the enzyme arginase expression, which reduces L-arginine accessible for NO generation and promotes polyamine synthesis. In addition to promoting the synthesis of L-ornithine, like IL-10 through inhibition, of phosphoinositide 3-kinase (PI3K) of PI3K/Akt/mTOR cascade can be achieved. As a consequence of this, Akt activation and phosphorylation are reduced, which in turn decreases the activity of mTORC1, which is a regulator of autophagy that is negativeCham C.M. et al..22 (Figure 1).
M2 reduces M1’s effect in later phases by changing the STAT3 signalling and PI3K/AKT pathways, which lead to an immunosuppressive TME by encouraging angiogenesisBai B.et al.,Mantovani A et al.23,24 As colorectal cancer improvements. The intestinal mucosa and peripheral blood contain stimulated CD8+ and CD4+ T cells, which are crucial mediators of the inflammatory response in patients with IBD. It is thought that CD8+ T lymphocytes are the primary immune effectors that target cancer cells and are crucial in halting the growth of tumors even if their reactions worsen as the tumors get bigger. Resistance to carcinogenesis is determined by CD8+ T cell infiltration and function in the TMEWang Q. et al..25The expression of transduction by the evasion and HIF-1 genesKryczek I. et al.,26 boosts the levels of glycolytic enzymes in the hypoxic TME, has not only positive but also negative consequences on CD8+ T cells. These effects can be categorized as either advantageous or deleterious. It is possible that CD8+ T lymphocytes will be deprived of this essential source of energy, which can result in metabolic dysfunction. This is because tumor cells compete with one another for glucose in the hypoxic TME. Their capacity to create cytokines, kill tumor cells, and proliferate is being diminished. The presence of tumor acidosis, which is an acidic environment that can inhibitCD8+ T cell function, is brought on by lactic acid produced by tumor glycolysis. Both hypoxia and tumor acidosis induce CD8+ T lymphocytes to generate PD-1, which depletes and malfunctions them. In order to stop CD8+ T lymphocytes from penetrating the tumor, tumor glycolysis may produce substances to attract myeloid-derived suppressor cells (MDSCs). One of the positive effects of hypoxia is that it may cause the expression of surface molecules on tumor cells. The helper T cells (Th) to be immune response to malignancy. The growth and spread of the tumor are influenced by the complex and interrelated functions carried out by different cells in the TME of colon cancerKryczek I. et al.,.26CD8+ T cells are subject to both positive and negative effects as a result of the transduction. This expression, which increases the amounts of glycolytic enzymes in the hypoxic TME, has both good and negative effects. The consequences that are being discussed here can be classified as either beneficial or detrimental. It is likely that CD8+ T cells will be deprived of this vital source of energy, which can lead to metabolic malfunction. This is a possibility. Within the hypoxic tumor microenvironment (TME), tumor cells engage in a competition with one another for glucose. The capacity of these cells to produce cytokines, destroy tumor cells, and proliferate is being reduced. A tumor acidosis, which is an acidic environment that can limit the growth of tumor cells, is presentFormica V. et al., Hou N. et al..27,28(Fig. 1) The cell polarization, cytokine generation, and immune response are all significantly impacted by these signaling pathways. Hypoxia triggers the transcription factor commonly referred to as inducible factor 1 (HIF-1). HIF-1 controls the development of many genes, including those related to immunological signaling, metabolism, and cell survival. HIF-1 may decrease Th1 polarization and stimulate Th17 polarization in Th cells. The mTOR pathway is a different process that can reduce Th1 polarization; hypoxia can impede this route, which increases Th17 polarization. Improved Th1 polarization and reduced Th2 polarization could originate from hypoxia’s activation of AMPK.Regulatory T cells (Tregs) are crucial for controlling the immune response. When colorectal cancer takes place(CRC), which in turn accelerate the growth of the tumor.
By releasing immunosuppressive cytokines like TGF-β and IL-10, they prevent effector CD8+ and CD4+ T cells from proliferating in addition to carrying out their cytotoxic activity. By interacting with myeloid suppressor cells, they provide a suppressive state that encourages defense against the immune system and tumor angiogenesis. Tregs influence a complicated web of signaling pathways that encourage colon cancer tumor growth and immune-suppression.Tregs trigger two key pathways in effector T cells: the NF-κB route, which decreases their capacity for releasing pro-inflammatory cytokines and destroying tumor cells, Additionally, they can promote tumor cell proliferation and immune evasion by stimulating the STAT3 pathwayOlguín J.E. et al..29 Whereas the role of Tregs has been discussed in broad strokes, not all Tregs are created equal. There are several subpopulations of Tregs, each with distinct traits and roles. Indeed, in cancer, an increase in certain Treg distinct populations may be associated with a poor prognosisFantini M.C. et al..30In the CRC TME, Foxp3+ Tregs are most common and are linked to a poorer prognosisOshi M. et al..31 Although STAT6 signalling may restrict Foxp3+ Treg activity and hence encourage CAC progression, tumor growth suppression results from a temporary depletion of Foxp3+ Tregs during CAC development. Wnt-β-catenin signalling pathway Activation is linked to the production of RORγt+ Tregs promotes the development of cancerQuandt J.et al..32 As important as the adaptation response is, we also need to take into account the role of innate immune response cells in the TME. Because of their unique functions and modes of action, the two NKT cell subpopulations in IBD (NKT1 and NKT2) are crucial role in the construction of the CRC TME.The TME and neighbouring bowel of CRC patients showed early research indicates that there is a drop in type 1 NKT cells and reversely increase in type 2 cells, both of which may continue to occur while the tumor continues to undergo progression. Because type 2 cells secrete IL-13, they have the potential to make ulcerative colitis (UC) worse, but type 1 cells are capable of producing inflammatory cytokines that have an effect on the intestinal barrierLiao C.M. et al..33 By secreting IL-9, type 1 NKT cells probably provide the protection against colitis, according to some researchKim H.S. et al..34In conclusion, type 2 NKT cells in IBD are probably going to encourage intestinal inflammation, whereas type 1 cells have both pathogenic and preventive functionsLai L.J. et al..35 Neutrophils’ dualism in connection to IBD is being supported by more and more data. Pro-inflammatory agents and some subpopulations of neutrophils have anti-inflammatory qualities.
Molecules likelow-density neutrophils (LDNs) or N2, which possess an immune-suppressive role and stimulate proliferation of tumors, angiogenesis, and N1 or high-density neutrophils (HDNs).These have pro-inflammatory property and capable of destroying tumor cells, have been identified in the TME of colorectal cancer (CRC)Zheng W. et al..36 Typically present in the initial stages of colorectal cancer (CRC), neutrophils of the N1 subtype change into N2 as the disease spreads. While GM-CSF and IFN-γAndzinski L. et al.,Granot Z.et al.37, 38By preferentially secreting cytokines, LDN or N2 neutrophils may regulate the way they move while avoiding inflammation from spreading. These have acrucial role in the repair and the regeneration of tissue processes, actively engage the removal of pro-inflammatory cells, and inhibits the action of cytotoxic T cells by producing a lot of dendritic cells and arginaseLoh W. et al.39Apoptotic neutrophils (N1), on the other hand, might regulates migration by the generation of stimulation of macrophages and releasing certain cytokines, which aid in their removal and aid in the resolution of inflammationKourtzelis I. et al.40It is essential to remember that neutrophils may also be detrimental since they can release carcinogens and free radicals, such as N-nitroso compounds, that make IBD patients more susceptible to cancerHebels D.G. et al.,LakshmiV.M.,et al.41,42 . Despite the fact that studies have shown that patients with a high density of CD177+ neutrophils had higher overall survival and disease-free survival compared to controls, CD177+ neutrophils have been shown to accelerate the growth of tumors and impair the function of NK cells and cytotoxic cells.T lymphocytesZhou Getal.43.However, the role of neutrophils associated with malignancies is yet unknown. Furthermore, just cells that have undergone in vitro differentiation should be categorized using the N1/N2 M1/M2 and classifications. This is because both neutrophils and macrophages. Inflammatory bowel disease (IBD) and intestinal inflammation are significantly influenced by innate lymphoid cells (ILCs). Three major categories into which they are dividedLuo W et al..44 Increased NK cells in the intestinal mucosa are seen in IBD, which may encourage T cell differentiation and inflammation. However, by preventing neutrophils from promoting inflammation, NK cells can also protect animal models of colitisHall L.J. et al..45Crohn’s disease and ulcerative colitis are linked to ILC1 secretion of interferon gamma, which may contribute to chronic both inflammation and the development of cancerBernink J.H. et al..46 ILC2s are found in higher concentrations in the tissues of individuals with inflammatory bowel disease (IBD), and it is possible that they contribute to the preservation of the intestinal mucosal barrier. However, the precise function of these molecules remains unknown. In particular, it appears that the IL-13 that is released by these cells induces the intestinal stem cells to develop into goblet, brush cells, are necessary for the healing of injury to the digestive tractSaez A. et al..47 ILC3, and more specifically NKp44 + ILC3, is responsible for the production of IL-22. This cytokine helps to preserve the integrity of the intestinal barrier, but it may also speed the proliferation of cells and the development of colorectal cancer. The activities and effects of these cells in inflammatory bowel disease and chronic adenocarcinoma are not entirely understood, despite the fact that these cells play a key role in both of these disorders. Table 1 provides a concise description of the main cells and the cytokines that they produce in the TME and is presented here.Colorectal cancer’s microenvironment consists of tumor-associated and inflammatory cells, together with the cytokines they produce. Pro-Tumor Cells Cytokines Linked with Antitumor Cells Macrophages TGF-β and M2 IL-10 macrophages M1 INF-γ, Th2 T helper 2 cells, TNF-α, IL-1β, IL-4, IL-5, IL-13 T helper 1 cells (Th1).
 |
Figure 1. A schematic illustration of the immune system’s cellular components of the microenvironment surroundingtumors of colorectal cancer. Left hand side represents the cells and secreted products in immune system that are involved in the anticancer response, and on the right hand side molecules and cells involved in a pro-tumor response Uthaisang sook S et al48.
|
Cancer Biology Pathway
One form of sickness that arises from aberrant cell proliferation. Tumor development may be influenced by genetic mutations and environmental pollutants. The p53-mediated apoptosis pathway, the GSK3 signaling system, and the ErbB family pathway are some of the most significant pathways in cancer biology. As tumor immunotherapy research advances, much of the work is focused on discovering the mechanisms by which the immune system recognizes and eliminates tumors and the basis for this process. The body’s failure to recognize is not limited to the classical model of self-immunity or lack of immunity, but includes small differences that exist independently and modify themselves. This decision provides arguments for cancer prevention. Similar to how it combats traditional tumors, the immune system strives to inhibit tumor growth, but occasionally tumor cells escape or develop immunity. (Figure 2).
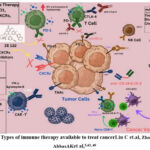 |
Figure 2: Types of immune therapy available to treat cancerLiu C et.al,Zhou G. et al.AbbasAKet al,5,43, 49
|
Cancer immunotherapy approaches include monoclonal antibodies (mAbs), cancer vaccines, small molecule drugs, adoptive cell therapy, and oncolytic viruses. CAR is an acronym for chimeric antigen receptor. Chemokine receptor with the C-X-C pattern, or CXCR. TAAs are antigens linked to tumors. cytotoxicity mediated by antigen-dependent cells, or ADCC. A. Programmed death-1, also known as PD-1. To begin, PD-L1, also known as programmed death-ligand 1. In the context of cytotoxic T-lymphocyte-associated protein 4, the acronym CTLA-4 refers to the protein.
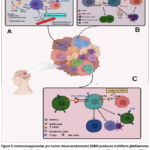 |
Figure 3: Immunosuppressive pro-tumor micro-environment [GBM produces multiform glioblastoma with a systemic or local immunosuppressive microenvironment. A significant fraction of the GBM microenvironment comprises immunosuppressive cells, and various immunotherapies that target these immune cells are presently being researched Zhang X, et al.,50,83
|
An immunosuppressive tumor microenvironment (TME) is a microenvironment that tumors create to inhibit immune cells and promote their growth. This is done by releasing metabolites and cytokines and depositing extracellular matrix (Figure 3), where (A) Cells inhibit the immune system within the glioma milieu. TGF-β, IL-6, and IL-1β are just a few of the numerous cytokines released by GAMs that support the malignant phenotype of GBM and preserve the blood-brain barrier’s high permeability. By focusing on phagocytosis checkpoints such as CCL2/CCR2, CD47/SIRP-α, and the CSF-1/CSF-1R axis, macrophages can more effectively phagocytose tumor cells. (B) Chemokines enlist Tregs, which then block cytotoxic T cell activity by blocking immunological checkpoints such as CTLA-4 or GITR. Tregs block a potent anti-tumor immune response by releasing immunosuppressive cytokines like TGF-β, IL-10, and IDO to impede dendritic cell function. (C) In the GBM microenvironment, the CCL2/CCR2 axis also recruits MDSCs, which then potently inhibit anti-tumour immunity through PD1/PD-L1. Additionally, human essential amino acids are depleted by MDSCs, which impairs T-cell activation and functionShowalter A et al., Drake CG et al..51, 52
The two immune system attacks and tumor promotion are the basis of a process known as cancer immuno-editing. Anticancer drugs suggest that external immunity can prevent cancer growth by shaping tumor immunogenicity or inhibiting host defense responses. The ability to create tumors that resist the immune system in this way is the seventh hallmark of cancer resistance and has been shown to have three stages: elimination (Also known as Prevention), homeostasis (maintenance), and escape(growth).
Equilibrium: A state of equilibrium is attained.
Escape: The presence of immunosuppressive substances and cytokines causes the balance to shift toward tumordevelopmentTan S et al..53
Treatment of cancer with immunotherapy drug-inhibited treatment:
Immune checkpoint blockade, or checkpoint blockade therapy, is a type of cancer treatment in which medications are used to disrupt proteins that prevent the immune system from attacking cancer cells. By inhibiting T cells, which are immune cells that target cancer cells, cancer cells can deceive the immune system. T cells are prevented from eliminating cancer cells by checkpoint proteins on both cancer and T cells, which transmit an “off” signal to T cells. Drugs used in checkpoint blockade therapy prevent these proteins from attaching, reactivating T cells’ onslaught on cancer cells. Patients with metastatic melanoma and numerous other malignancies have shown better survival and long-lasting responses with checkpoint blocking therapy. Not only is it generally easier on the body than most chemotherapy medications, but it’s also less poisonous. Numerous cancers, including those of the breast, bladder, cervical, colorectal, endometrial, oesophageal, head and neck, kidney, liver, lung, melanoma, and stomach, can be treated with checkpoint blockade therapyDeng H, et al.54
Treatment of colorectal and liver cancers with immunotherapy
Immune checkpoint inhibitors: Drugs like these suppress immune cell proteins that stop the body’s defenses from eradicating cancer cells.
Toll-like receptor (TLR) agonists: Innate immune cells have TLRs, and this may strengthen the immune system’s natural defenses.
Monoclonal antibodies (mAbs): By blocking immune checkpoint receptors like PD-1/PD-L1, these antibodies can increase the effectiveness of colorectal cancer therapy.
Using small molecules or modifying mAbs: Combining therapies to address several pathways using antigens unique to a given tissue that are only expressed during the stage of cell differentiation.
Using excessively expressed antigens: These are significantly higher expressed in cancerous cells than in healthy cells.
Mechanistic Approach
Immunogenicity of Colorectal Cancer:Colorectal cancer with high mutational loads can release neo-antigens that the immune system can attack.
Microsatellite instability (MSI): When several neo-antigens are present in a tumor, a high MSI status is linked to a stronger immune response and a better prognosis.
Tumour Microenvironment: Tregs and MDSCs may be present in the immunosuppressive microenvironment of colorectal cancer, which is comparable to that of liver cancer.
Inflammation:Long-term inflammation carried on by conditions like inflammatory bowel disease (IBD) may affect the immune system and increase the risk of colorectal cancer.
Immunotherapy for Colorectal and Liver CancerLichtenstern CR, et al..55
Liver Cancer Diagnostic Restrictors: Although results can vary, medications that target PD-1/PD-L1 (such as pembrolizumab and nivolumab) have shown promise in the treatment of liver cancer.
CAR-T Cell Therapy: Although research is still in progress, CAR-T treatments that target certain antigens associated with liver cancer are not yet generally accessibleAravalli RN et al.56 The application of CAR-T cells to treat colorectal cancer is being studied; these cells are specifically designed to target antigens such as CEA (carcinoembryonic antigen).
Cancer Vaccines: Efforts are being made to create vaccines that specifically target antigens associated with liver cancer. Research is being done to create vaccines that specifically target neo-antigens or antigens unique to colorectal cancerPan RY et al..57
Combination therapy: To increase efficacy, checkpoint inhibitors are being used in conjunction with other treatments, such as targeted therapy (such as tyrosine kinase inhibitors like sorafenib).To overcome resistance and enhance results, immunotherapy is being combined with chemotherapy, targeted therapy, or other immune-modulating drugs.
Checkpoint Inhibitors for Colorectal Cancer: PD-1/PD-L1 inhibitors have demonstrated good response rates in MSI-high colorectal malignancies. Examples of these medications include pembrolizumab and nivolumab.
Scientific Insight along with Mechanism
In various forms, the effectiveness of immunotherapy for cancer can be impacted by the unique features of the tumor, hereditary variables, and the general health of the patient. Its foundation lies in the use of humanized antibodies known as immune checkpoint inhibitors (ICIs) to block the cellular pathways that impede T lymphocyte activity subset of immune system cells that aid in both the body’s defense against infection and its potential to fight cancer. One way that tumor cells might proliferate and spread quickly is by suppressing, eluding, or taking advantage of the host’s immune systemWang Y, Deng et al..58By boosting immune responses, immunotherapy has demonstrated remarkable efficacy in the last several years, changing the therapeutic picture for several cancers.64 Instead of directly attacking tumor cells, immunotherapy, in contrast to traditional cancer treatments, strengthens the immune system of the patient or alters the immunological microenvironment (TME) of the tumor.Since cancerous cells can evade the immune system, the main goal of immunotherapy, including immune checkpoint inhibitors (ICIs), is to prevent tumor cells from using their immune systems to evade detection and spread of the disease. The receptors PD-1 (programmed cell death 1) and CTLA-4 (cytotoxic T-lymphocyte-associated antigen 4) block responses of T cells to preserve marginal tolerance, which allows tumor cells to proliferate instead of being destroyed by the immune systemLei X et al..59Instead of being killed by the immune system’s aid, tumor cells are allowed to grow instead of being destroyed. ICIs blocks the inhibitory receptors and enhance the anticancer immune responses, that is expressed upon T lymphocytes combinedwith superficial layer of malignant cells like PD-L1 that exhibit colorectal cancer progression. PD-L1 is responsible for the quick phosphorylation of SHP-1 and SHP-2PD-1 is able to exert an inhibitory impact as a result of its interaction with 1 / 3 Senth. Antibodies that target CTLA-4, PD-1, or PD-L1 are known as 15 immunotherapy combinations (ICIs). These antibodies have the ability to re-establish anticancer immune responses, which can result in dramatic clinical responses in a range of cancers. Establishing the blocking of pD-1/PD-L1 is the most effective immuno-therapeutic method. This technique has also been associated with significant clinical responses in a wide range of cancers, likeskin cancer, non-small cell lung cancer, head and neck cancerGao T et al..60 CTLA-4 is responsible for the modification of T lymphocyte activity. It does this by engaging in a competition with the receptors CD28, which are demonstrated by both CD8+ and CD4+ cells, for common ligands. It has been demonstrated that the presence of anti-CTLA-4 antibodies can increase the availability of CD28 ligandsKanesvaran R et al..61 This is due to the fact that effector T cell activation and Treg suppression have an effect on homeostasis and limit tumor growth. Immune receptors with Ig and ITIM domains (TIGIT) has been shown to be elevated in tissues that have been affected by colorectal cancer. Due to the fact that they play such an important part in the immune evasion of tumors, for immune-mediated therapy treatment. T cells are triggered by tenacious antigens, which results in a high level of expression from these cells. TIM-3 have a crucial role in the immunological gatekeeping process, specifically in the regulation of T-cell responses. Through the creation of an immunosuppressive milieu within the tumor, theresult of TIM-3 on both immune evasion and the progression of tumors can be promoted by Tregs. There is a significant association between TIM-3 and tumor lymph node/distant metastases in the tissues of colorectal cancer patients. It is possible to predict that T cells will fail and that the presence of TIM-3 in CRC TME will enhance tumor spread. Precision of the TIM-3+ T cellcharacterisation should improve clinical response, enable tailored therapy, and strengthen anti-tumor immunity.
TIM-3 has a direct correlation with CRC cell invasion and proliferation Ruffo Eet al.62 another essential immunoglobulin superfamily immunological checkpoint is LAG-3. It is produced by multiple immune cells and prevents T-cell activation and proliferation. It is a prime regulator that establishes the high level of Treg activity and codes for molecules on the surface that specifically enhance Treg. When LAG-3 attached with major histocompatibility complex II.The potential to restore anti-tumor immunity and activate LAG3 block. In the colorectal cancer model, anti-PD-L1 and anti-TIM-3 combined can increase T cells’ antitumor activity. The anti-LAG3 and PD-1 antibodies inhibited tumor growth in tandem in the MC38 colorectal adeno carcinoma. Thus, LAG-3-targeting treatment regimens could benefit CRC patientsHuo JL et al..63
81 TIGHT inhibits NK cells and effector T lymphocytes. Additionally, it can decrease the immunological response by supporting regulatory T cells. T Cell activation inside tumor grows when ICIs and anti-TIGIT anti bodies are combined, which makes tumor elimination easier. Variety of malignancies is now being investigated in the varioius kind of clinical studies. The blocking of TIGIT in the NK cell can reverse their functional failure and restore their powerful activity in vivoLi XF, et al..64
These novel immunological checkpoints, however, show promise as therapeutic goals for use in clinical settingsSeidel JA, et al..65
By treating transplanted T cells, adoptive T cell therapy (ACT) is used in conjunction with anti-tumor therapy. In this procedure, TILs with anticancer activity are isolated, then they are extensively expanded and activated in vitro before being given to the patients. The improved effectiveness of T cells bearing innately neo-antigen-reactive T They generated cell receptors (TCRs). Autologus or endogenous CD8+ T cells generated from TILs can both be utilized in ACT. It has been observed that TILs, an essential part of the TME, are connected to clinical outcomes and treatment responses. The highly specific anti-tumor immune response is brought about by TILs that can identify tumor antigens. Moreover, TILs are less harmful than CAR-T or TCR-modified T cells. Additionally, TILs exhibit diverse specificity, suggesting a critical benefit in impeding immune evasion. For customised cancer immunotherapy, TILs thus represents and ideal source of tumor reactive TCRs.. They can also carry ACT and TCR gene therapies. Sustained remission and positive overall response rates were associated with the use of TILs in ACT. Furthermore, immune treatments that target toll-like receptors have been shown to have an antitumor effect in colorectal cancer (CRC) by either suppressing oncogenic signalling pathways or activating anticancer immunityKawakami Y, et al.66In the context of ACT, the utilization of CAR-T cells, also known as genetically engineering antigen-specific T cells provides an advantage over earlier efforts that utilized these alterations. CAR-T cells have the potential to be therapeutically effective, particularly in the treatment of hematological cancers. CAR-T cell therapy that targets anti-CD19 was recently approved by the Food and Drug Administration (FDA) for the treatment of acute lymphoblastic leukemia and diffuse large B-cell lymphoma. There has been an improvement in the therapy of CAR-T for patients who have B-cell malignancies.Because of CAR-T cells enhanced ability to expand to tumor loci, their prolonged in vivo persistence, and their ability to work in concert with the patient’s immune system, this living, replicating therapy may be able to provide patients with prolonged anti-tumor control. It has recently been demonstrated that CAR-T cells can cure solid tumors other than haematological malignancies. T cells struggle to eradicate the bulk of solid tumors and maintain long-term remission because they are more complex than haematologicalmalignanciesMohanty R et al..67 In preclinical animals, CAR-T cell therapy has recently shown a lot of potential in preclinical animals for the treatment of CLM. However, due to drawbacks such increased toxicities, recurrence, restricted trafficking, and an adverse TME, only a small percentage of CRC cases with CLM show a sustained response to CAR-T cell treatmentMiliotou AN, et al., Zhylko A, et al..68,69Important research topics include the potential therapeutic use of anti-inflammatory therapies for colorectal cancer and the role of inflammation in predicting cancer prognosis. Numerous cancer forms, including colorectal cancer, have been linked to the onset and spread of chronic inflammation. The tumoral milieu created by this ongoing inflammatory process encourages cell division, local invasion, and distant metastasis. The procedure involves several molecular pathways, like the nuclear factor kappa-B (NF-κB) activation signaling pathway. This literature represents the crucial function of NF-κB activation and the correlation between colorectal cancer and its dysregulation. Anti-inflammatory medications that target NF-κB activation might therefore be a viable cancer therapeutic approach. Celecoxib, aspirin, and other nonsteroidal anti-inflammatory drugs (NSAIDs) have shown anticancer benefits in preclinical and clinical studies. By inhibiting the NF-κB pathway, these drugs can reduce cell proliferation, trigger apoptosis, and restrict tumoral angiogenesis.The potential use of NSAIDs, such as aspirin and sulindac, used for the investigation of colorectal cancer. It has been demonstrated that sulindac is highly involved in lowering the likelihood of developing colorectal adenomatous polyps, as precursors to colorectal cancer. A further point to consider is that aspirin has demonstrated potential in the prevention of colorectal cancer, particularly in individuals who have a family history of the disease Sun M et al.,Iftode C.et al, Huang F. et al, Luo P., et al, Kanth P., et al,Lloyd-Price J et al.70–75For a better understanding of the intricate relationship that exists between intestinal bacteria and the formation of tumors, it is possible to investigate the function that the microbiome and microbiota play in colorectal cancer (CRC)Dejea C.M., et al..76 There is a significant difference inbetweenthecomposition of the gut microbiome and healthy individuals with colorectal cancer (CRC), which has been established to have the potential to influence the development of the illness. Since dysbiosis, which reflects by the composition of bacterial imbalance, has been associated to the correlation between toa number of intestinal ailments, including colorectal cancer, it is imperative to comprehend the role of these bacteria in health and illness. In the study of colorectal cancer, both harmful and beneficial bacteria for intestinal health have been thoroughly investigated. The influences of the TME and immunological response, bacteria likeBactericides fragilis Escherichia coli, Fusobacterium nucleatum, for instance are connected to colorectal cancer (CRC).Kostic A.D., etal,Thomas R.M., et al, Shariati A et,al77–79.Considering the presence of these contaminants can cause acceleration of the development of cancer and inflammation, eliminating them specifically may be a useful therapeutic approach. Cancer vaccines were investigated and approved by the FDA as a type of cancer immunotherapySchuster M, et al..80 To increase antitumor cytotoxicity, by injecting patients with cancer-specific components, an immune response that is very specific to the patient’s tumor cells is triggered. Remarkably, vaccines modifies the antigen-specific B and T cell responses, which impacts the TME. As evidence of the important role adjuncts play in the treatment of colorectal cancer, the distinct effects of varying amounts of neo-antigen clonally on TME may present chances to improve the targets targeted in the vaccines. It was also discovered that Tregs and hepatic macrophages were sensitive targets for antitumor action, among other possible treatment approachesMcComb S et al., Cheng D et al..81,82
The absence of consistently positive clinical outcomes, despite the development of several unique therapeutic techniques in CLM, emphasizes the need for additional research to either improve the effectiveness of already available treatments or develop new ones to increase clinical benefits. The FDA some persons can be treated with approved targeted therapy medications with liver cancer and colorectal cancer. Targeted therapy drugs are listed below, along with their mechanism and dosages in Table 4 and Figure 3.
Table 4: Drugs used to treat in immunotherapy for liver cancer and colorectal cancer.YuXet al, Burgos-Molina AM et al, Jain A et al. 83,84,85
|
Therapy |
Drugs |
Mechanism |
Dosage |
|
Immunotherapy drugs used to treat liver cancer include |
Atezolizumab |
A checkpoint inhibitor that can be used in combination with bevacizumab or cabozantinib |
until the condition worsens or the toxicity becomes intolerable, 840 mg intravenously every two weeks, 1200 mg intravenously every three weeks, or 1680 mg intravenously every four weeks. |
|
Nivolumab |
An immunotherapy drug that can be used in combination with ipilimumab |
480 mg IV over 30 minutes every 4 weeks until disease development or severe toxicity, or 240 mg IV over 30 minutes every 2 weeks until the condition worsens or the toxicity becomes unbearable. |
|
|
Drugs used to treat colorectal cancer include |
Bevacizumab |
A drug that targets blood vessel formation (VEGF) |
Together with bolus-IFL (5 mg/kg IV every two weeks) with FOLFOX4 (10 mg/kg IV every two weeks), this |
|
Capecitabine |
Prodrug that interferes with the growth of cancer cells by releasing 5-fluorouracil (5-FU) |
As a Single Agent: 1250 mg/m2 orally twice a day for the first 14 days of each 21-day cycle. Duration of therapy: Maximum of 8 cycles |
|
|
Panitumumab |
A monoclonal antibody that binds to the human epidermal growth factor receptor (EGFR) |
Every fourteen days, give 6 mg/kg IV over 60 minutes; if the initial infusion is well tolerated, give further infusions over 30 to 60 minutes; and provide dosages more than 1000 mg over 90 minutes. |
|
|
Leucovorin |
A drug that helps 5-fluorouracil work better |
By gradual intravenous administration (at least 3 minutes), 200 mg/m2, and then 5-fluorouracil (the product information from the manufacturer should beconsulted), once a day for 5 days |
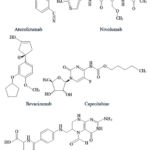 |
Figure 4: Structure of some anticancer drugs Yang XL et al..86
|
Rationality
It is widely acknowledged that cancer is one of the primary contributors to mortality worldwide, among all the non-communicable diseases (NCDs) that currently increase the world’s mortality rate, cancer is the foremost common cause of death worldwide. Immunotherapy is a potential treatment for colorectal and liver cancer because it uses the natural; immune system to target and especially eliminate the cancer cells. By boosting the immune response, these treatments help the body recognize the tumor antigen. Immuno-checkpoint inhibitors have the potential to combat immune evasion in liver cancer. These open up new therapeutic options and enhance patient outcomes in the disease’s late stage. Besides that, any chemical moiety has some serious adverse and side effects on our body function, which is a major reason to focus on natural immunity, and is the reason to study related to natural wings.
Conclusion
The anatomical position of metastatic colorectal cancer (mCRC) and the immunosuppressive environment make treatment difficult, particularly for colonic liver metastases (CLM). Even while there is increasing clinical evidence that immunotherapeutic medicines (ICIs) are effective for patients having MSI-H mCRC or dMMR, most patients with pMMR or MSS malignancies they doesn’t get any kind of benefit. This is illustrated as different suppressive networks restrict effector immune cells, allowing CRC to proliferate and spread throughout the TME.Effortsemphasizing on the liberation of CLM from the inhibitory networks.The activation barriers which may comprise the TME by mergingimmune therapy use of immunomodulatory and anti-inflammatory medications may aid in preventing the development of cancer during the phase of chronic inflammation. To effectively inhibit tumor development during the tumor stage, antitumor immunity must be strengthened. Optimizing immune cell recruitment through metabolic regulation can enhance the TME. Tumor cell and immune cell metabolites affect immunological checkpoint regulation, immune cell stimulation, and phenotypic alterations. The effect of microbiome in the field of colorectal cancer (CRC) has been emphasized by recent research, which also shows that it may be a target for therapeutic approaches. Various bacteria like Escherichia coli, Bacteroides fragilis, and Fusobacterium nucleatum etc., are crucial therapeutic targets. In this strategical way-outit has been strongly decided the alteration of the microbiome, reduction of inflammation, and enhance the efficacy of traditional therapies like chemotherapy, immunotherapy are the dietary modifications, synthetic biology, and fecal microbiota transplantation (FMT). These methods provide new opportunities for personalized and efficient CRC care by enhancing treatment response and lowering adverse effects.
Acknowledgment
The authors greatly appreciate the Faculty of Pharmaceutical Science, Assam down town University, and DmbH Institute of Medical Science, Dadpur, Puinan, Hooghlyfor granting me access to their extensive resources, which greatly enriched my research. Additionally, my heartfelt thanks to both Institute for providing access to their library and computer laboratory, enabling me to conduct critical analyses for the project.
Funding source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest
Data availability
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed consent statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author contributions
- Avik Dutta:Involved in the entire literary survey and drafted the paper or critically revised it for significant intellectual content;
- Faruk Alam and Prithviraj Chakraborty:Made a significant contribution to the conceptualization and design of the data;
- Alindam Ghosh and Soumya sunder Ghora:Helped with data gathering.
- Arpan Sen and Debraj Dey:The creation of the diagrammatic framework handeled
- Saurav Guchhait and Pakhi Pandey:General citation was handled.
References
- Null M, Agarwal M. Anatomy, Lymphatic System. Nih.gov. Published June 22, 2023. Accessed June 30, 2025. https://www.ncbi.nlm.nih.gov/books/NBK513247/
- Lerner A, Aminov R, Matthias T. Dysbiosis May Trigger Autoimmune Diseases via Inappropriate Post-Translational Modification of Host Proteins. Frontiers in Microbiology. 2016;7.
CrossRef - Matsuzaki G, Umemura M. Interleukin-17 family cytokines in protective immunity against infections: role of hematopoietic cell-derived and non-hematopoietic cell-derived interleukin-17s. Microbiology and Immunology. 2018;62(1):1-13.
CrossRef - Bardhan K, Liu K. Epigenetics and Colorectal Cancer Pathogenesis. Cancers. 2013;5(4):676-713.
CrossRef - Liu C, Yang M, Zhang D, Chen M, Zhu D. Clinical cancer immunotherapy: Current progress and prospects. Frontiers in Immunology. 2022;13.
CrossRef - Avelino CD, Hardman Y. The Role of Innate Immune System: A Crosstalk between Invertebrates and Humans. Journal of Cellular Immunology. 2025;7(3):116-121. doi:https://doi.org/10.33696/immunology. 7.230
CrossRef - Tacke F. Targeting hepatic macrophages to treat liver diseases. Journal of Hepatology. 2017;66(6):1300-1312.
CrossRef - Abdulla KN, Hassan MN, Al-Ibadah M, Mohsein OA, Oraibi AI, Al-Samydai AM, Al-hussaniy HA. Mucinous Ovarian Cancer: A Review of Diagnosis and Treatment. Malaysian Journal of Medicine & Health Sciences. 2025 May 1;21(3).
- Al-Hussaniy HA, Al-Zobaidy MJ. Effects of Mdm2 Inhibitors on Cellular Viability of Breast Cancer Cell Lines HP100, MCF7. Bratislavske lekarske listy. 2024;125(10):627-34
CrossRef - Lukasch B, Westerdahl H, Strandh M, et al. Genes of the major histocompatibility complex highlight interactions of the innate and adaptive immune system. PeerJ. 2017;5:e3679. doi:https://doi.org/10.7717/peerj.3679.
CrossRef - Zouridakis EG, Garcia-Moll X, Kaski JC. Usefulness of the blood lymphocyte count in predicting recurrent instability and death in patients with unstable angina pectoris. The American Journal of Cardiology. 2000;86(4):449-451.
CrossRef - Ott PA, Hodi FS, Kaufman HL, Wigginton JM, Wolchok JD. Combination immunotherapy: a road map. Journal for ImmunoTherapy of Cancer. 2017;5(1).
CrossRef - Osei-Bordom DC, Kamarajah S, Christou N. Colorectal Cancer, Liver Metastases and Biotherapies. Biomedicines. 2021;9(8):894.
CrossRef - Haanen JBAG, Robert C. Immune Checkpoint Inhibitors. Progress in Tumor Research. 2015;42:55-66.
CrossRef - Nicholson LB. The Immune System. Essays in Biochemistry. 2016;60(3):275-301.
CrossRef - Greten TF, Mauda-Havakuk M, Heinrich B, Korangy F, Wood BJ. Combined locoregional-immunotherapy for liver cancer. Journal of Hepatology. 2019;70(5):999-1007.
CrossRef - .Yaghoubi N, Soltani A, Ghazvini K, Hassanian SM, Hashemy SI. PD-1/ PD-L1 blockade as a novel treatment for colorectal cancer. Biomedicine & Pharmacotherapy. 2019;110:312-318.
CrossRef - Yang M, Li J, Gu P, Fan X. The application of nanoparticles in cancer immunotherapy: Targeting tumor microenvironment. Bioactive Materials. 2021;6(7):1973-1987.
CrossRef - Pol S, Nalpas B, Driss F, et al. Efficacy and limitations of a specific immunotherapy in chronic hepatitis B. Journal of Hepatology. 2001;34(6):917-921.
CrossRef - Mantovani, A, Sica, A, Locati M. Macrophage Polarization Comes of Age. Immunity. 2005;23(4):344-346.
CrossRef - Natoli M, Herzig P, PishaliBejestani E, et al. Plinabulin, a Distinct Microtubule-Targeting Chemotherapy, Promotes M1-Like Macrophage Polarization and Anti-tumor Immunity. Frontiers in Oncology. 2021;11.
CrossRef - Cham CM, Driessens G, O’Keefe JP, Gajewski TF. Glucose deprivation inhibits multiple key gene expression events and effector functions in CD8+ T cells. European Journal of Immunology. 2008;38(9):2438-2450.
CrossRef - Bai B, Wu F, Ying K, et al. Therapeutic effects of dihydroartemisinin in multiple stages of colitis-associated colorectal cancer. Theranostics. 2021;11(13):6225-6239.
CrossRef - Mantovani A, Marchesi F, Malesci A, Laghi L, Allavena P. Tumour-associated macrophages as treatment targets in oncology. Nature Reviews Clinical Oncology. 2017;14(7):399-416.
CrossRef - Wang Q, Qin Y, Li B. CD8+ T cell exhaustion and cancer immunotherapy. Cancer Letters. Published online December 2022:216043.
CrossRef - Kryczek I, Lin Y, Nagarsheth N, et al. IL-22+CD4+ T Cells Promote Colorectal Cancer Stemness via STAT3 Transcription Factor Activation and Induction of the Methyltransferase DOT1L. Immunity. 2014;40(5):772-784.
CrossRef - Formica V. Immune reaction and colorectal cancer: Friends or foes? World Journal of Gastroenterology. 2014;20(35):12407.
CrossRef - Hou N, Zhang X, Zhao L, et al. A novel chronic stress-induced shift in the Th1 to Th2 response promotes colon cancer growth. 2013;439(4):471-476.
CrossRef - Olguín JE, Medina-Andrade I, Rodríguez T, Rodríguez-Sosa M, Terrazas LI. Relevance of Regulatory T Cells during Colorectal Cancer Development. Cancers. 2020;12(7):1888.
CrossRef - Fantini MC, Favale A, Onali S, Federica Facciotti. Tumor Infiltrating Regulatory T Cells in Sporadic and Colitis-Associated Colorectal Cancer: The Red Little Riding Hood and the Wolf. International Journal of Molecular Sciences. 2020;21(18):6744-6744.
CrossRef - Masanori Oshi, Sarkar J, Wu R, et al. Intratumoral density of regulatory T cells is a predictor of host immune response and chemotherapy response in colorectal cancer. PubMed. 2022;12(2):490-503.
- Quandt J, Arnovitz S, Haghi L, et al. Wnt–β-catenin activation epigenetically reprograms Treg cells in inflammatory bowel disease and dysplastic progression. Nature Immunology. 2021;22(4):471-484.
CrossRef - Liao CM, Zimmer MI, Wang CR. The Functions of Type I and Type II Natural Killer T Cells in Inflammatory Bowel Diseases. Inflammatory Bowel Diseases. 2013;19(6):1330-1338.
CrossRef - Kim HS, Chung DH. IL-9-producing invariant NKT cells protect against DSS-induced colitis in an IL-4-dependent manner. Mucosal Immunology. 2013;6(2):347-357.
CrossRef - Lai LJ, Shen J, Ran ZH. Natural killer T cells and ulcerative colitis. Cellular Immunology. 2019;335:1-5.
CrossRef - Zheng W, Wu J, Peng Y, Sun J, Cheng P, Huang Q. Tumor-Associated Neutrophils in Colorectal Cancer Development, Progression and Immunotherapy. Cancers. 2022;14(19):4755-4755.
CrossRef - Andzinski L, Kasnitz N, Stahnke S, et al. Type IIFNs induce anti-tumor polarization of tumor associated neutrophils in mice and human. International Journal of Cancer. 2015;138(8):1982-1993.
CrossRef - Granot Z, Jablonska J. Distinct Functions of Neutrophil in Cancer and Its Regulation. Mediators of Inflammation. 2015;2015:1-11.
CrossRef - Loh W, Vermeren S. Anti-Inflammatory Neutrophil Functions in the Resolution of Inflammation and Tissue Repair. Cells. 2022;11(24):4076-4076.
CrossRef - Kourtzelis I, Hajishengallis G, Chavakis T. Phagocytosis of Apoptotic Cells in Resolution of Inflammation. Frontiers in Immunology. 2020;11.
CrossRef - Dennie H, Jennen D, Jos Kleinjans, Theo. Molecular Signatures of N-nitroso Compounds in Caco-2 Cells: Implications for Colon Carcinogenesis. Toxicological Sciences. 2009;108(2):290-300.
CrossRef - Lakshmi VM, Hsu FF, Zenser TV. Nitric Oxide-Mediated Nitrosation of 2-Amino-3,8-dimethylimidazo[4,5-f]quinoxaline Potentiated by Hemin and Myeloperoxidase. Chemical Research in Toxicology. 2005;18(6):1038-1047.
CrossRef - Zhou G, Peng K, Song Y, et al. CD177+ neutrophils suppress epithelial cell tumourigenesis in colitis-associated cancer and predict good prognosis in colorectal cancer. Carcinogenesis. 2017;39(2):272-282. doi:https://doi.org/10.1093/carcin/bgx142
CrossRef - Luo W, Tian L, Tan B, et al. Update: Innate Lymphoid Cells in Inflammatory Bowel Disease. Digestive Diseases and Sciences. 2021;67(1):56-66.
CrossRef - Hall LJ, Murphy CT, Quinlan A, et al. Natural killer cells protect mice from DSS-induced colitis by regulating neutrophil function via the NKG2A receptor. Mucosal Immunology. 2013;6(5):1016-1026.
CrossRef - Bernink JH, Peters CP, Munneke M, et al. Human type 1 innate lymphoid cells accumulate in inflamed mucosal tissues. Nature Immunology. 2013;14(3):221-229.
CrossRef - Saez A, Gomez-Bris R, Herrero-Fernandez B, Mingorance C, Rius C, Gonzalez-Granado JM. Innate Lymphoid Cells in Intestinal Homeostasis and Inflammatory Bowel Disease. International Journal of Molecular Sciences. 2021;22(14):7618.
CrossRef - Uthaisangsook S, Day NK, Bahna SL, Good RA, Haraguchi S. Innate immunity and its role against infections. Annals of Allergy, Asthma & Immunology. 2002;88(3):253-265.
CrossRef - Abbas AK, Lichtman AH. Basic Immunology : Functions and Disorders of the Immune System. Saunders/Elsevier; 2011.
CrossRef - Zhang X, Zhao L, Zhang H, et al. The immunosuppressive microenvironment and immunotherapy in human glioblastoma. Frontiers in Immunology. 2022;13.
CrossRef - Showalter A, Limaye A, Oyer JL, et al. Cytokines in immunogenic cell death: Applications for cancer immunotherapy. Cytokine. 2017;97:123-132.
CrossRef - Drake CG. Combination immunotherapy approaches. Annals of Oncology. 2012;23:viii41-viii46.
CrossRef - .Tan S, Li D, Zhu X. Cancer immunotherapy: Pros, cons and beyond. Biomedicine & Pharmacotherapy. 2020;124(124):109821.
CrossRef - Deng H, Zhang Z. The application of nanotechnology in immune checkpoint blockade for cancer treatment. Journal of Controlled Release. 2018;290:28-45.
CrossRef - Lichtenstern CR, Ngu RK, Shalapour S, Karin M. Immunotherapy, Inflammation and Colorectal Cancer. Cells. 2020;9(3):618.
CrossRef - Aravalli R, Steer C. Immune-Mediated Therapies for Liver Cancer. Genes. 2017;8(2):76.
CrossRef - Pan RY, Chung WH, Chu MT, et al. Recent Development and Clinical Application of Cancer Vaccine: Targeting Neoantigens. Journal of Immunology Research. 2018;2018:1-9.
CrossRef - Wang Y, Deng W, Li N, et al. Combining Immunotherapy and Radiotherapy for Cancer Treatment: Current Challenges and Future Directions. Frontiers in Pharmacology. 2018;9.
CrossRef - Lei X, Lei Y, Li JK, et al. Immune cells within the tumor microenvironment: Biological functions and roles in cancer immunotherapy. Cancer Letters. 2020;470:126-133.
CrossRef - Gao T, Pei R. Isolation of DNA Aptamer Targeting PD-1 with an Antitumor Immunotherapy Effect. ACS Applied Bio Materials. 2020;3(10):7080-7086.
CrossRef - Kanesvaran R, Cordoba R, Maggiore R. Immunotherapy in Older Adults With Advanced Cancers: Implications for Clinical Decision-Making and Future Research. American Society of Clinical Oncology Educational Book. 2018;(38):400-414.
CrossRef - Ruffo E, Wu RC, Bruno TC, Workman CJ, Vignali DAA. Lymphocyte-activation gene 3 (LAG3): The next immune checkpoint receptor. Seminars in Immunology. 2019;42:101305.
CrossRef - Huo JL, Wang YT, Fu WJ, Lu N, Liu ZS. The promising immune checkpoint LAG-3 in cancer immunotherapy: from basic research to clinical application. Frontiers in Immunology. 2022;13.
CrossRef - Krijgsman D, de Vries NL, Skovbo A, et al. Characterization of circulating T-, NK-, and NKT cell subsets in patients with colorectal cancer: the peripheral blood immune cell profile. Cancer Immunology, Immunotherapy. 2019;68(6):1011-1024.
CrossRef - Seidel JA, Otsuka A, Kabashima K. Anti-PD-1 and Anti-CTLA-4 Therapies in Cancer: Mechanisms of Action, Efficacy, and Limitations. Frontiers in Oncology. 2018;8(86).
CrossRef - Kawakami Y, Eliyahu S, Delgado CH, et al. Identification of a human melanoma antigen recognized by tumor-infiltrating lymphocytes associated with in vivo tumor rejection. Proceedings of the National Academy of Sciences. 1994;91(14):6458-6462.
CrossRef - Mohanty R, Chowdhury C, Arega S, Sen P, Ganguly P, Ganguly N. CAR T cell therapy: A new era for cancer treatment (Review). Oncology Reports. 2019;42(6).
CrossRef - Miliotou AN, Papadopoulou LC. CAR T-cell Therapy: A New Era in Cancer Immunotherapy. Current Pharmaceutical Biotechnology. 2018;19(1):5-18.
CrossRef - Zhylko A, Winiarska M, Graczyk-Jarzynka A. The Great War of Today: Modifications of CAR-T Cells to Effectively Combat Malignancies. Cancers. 2020;12(8):2030.
CrossRef - Sun M, Yu J, Wan J, Dou X, Chen X, Ye F. Role of aspirin in cancer prevention. Cancer Treatment and Research Communications. 2024;43:100884. doi:https://doi.org/10.1016/j.ctarc.2025.100884
CrossRef - Iftode C, Stela Iurciuc, Iasmina Marcovici, et al. Genistein–Aspirin Combination Exerts Cytotoxic and Anti-Migratory Effects in Human Colorectal Cancer Cells. Life. 2024;14(5):606-606.
CrossRef - Huang F, Guo W. Aspirin use and changes in circulating tumor DNA levels in patients with metastatic colorectal cancer. PubMed. 2024;37(1):123-128.
- Luo P, Shi W, Cheng X, Yang J, Pei G, Dong J. Which Drugs are More Effective in Preventing Familial Adenomatous Polyposis Progression based on Network Meta-analysis? Current pharmaceutical design. 2024;30(20):1548-1563.
CrossRef - Priyanka Kanth, Hazel MW, Schell JC, et al. Evaluation of EGFR and COX pathway inhibition in human colon organoids of serrated polyposis and other hereditary cancer syndromes. Familial cancer. Published online April 12, 2024.
- Lloyd-Price J, Abu-Ali G, Huttenhower C. The healthy human microbiome. Genome Medicine. 2016;8(1).
CrossRef - Dejea CM, Fathi P, Craig JM, et al. Patients with familial adenomatous polyposis harbor colonic biofilms containing tumorigenic bacteria. Science. 2018;359(6375):592-597.
CrossRef - Kostic AD, Gevers D, Pedamallu CS, et al. Genomic analysis identifies association of Fusobacterium with colorectal carcinoma. Genome Research. 2011;22(2):292-298.
CrossRef - Thomas RM, Jobin C. The Microbiome and Cancer: Is the “Oncobiome” Mirage Real?.Trends in Cancer. 2015;1(1):24-35.
CrossRef - Shariati A, Razavi S, Ghaznavi-Rad E, et al. Association between colorectal cancer and Fusobacterium nucleatum and Bacteroides fragilis bacteria in Iranian patients: a preliminary study. Infectious Agents and Cancer. 2021;16(1). doi:https://doi.org/10.1186/s13027-021-00381-4
CrossRef - Schuster M, Nechansky A, Kircheis R. Cancer immunotherapy. Biotechnology Journal. 2006;1(2):138-147.
CrossRef - McComb S, Thiriot A, Akache B, Krishnan L, Stark F. Introduction to the Immune System. Methods in Molecular Biology (Clifton, NJ). 2019;2024:1-24.
CrossRef - Cheng D, Chai J, Wang H, Fu L, Peng S, Ni X. Hepatic macrophages: Key players in the development and progression of liver fibrosis. Liver International. 2021;41(10):2279-2294.
CrossRef - Yu X, Zhu L, Liu J, Xie M, Chen J, Li J. Emerging Role of Immunotherapy for Colorectal Cancer with Liver Metastasis. OncoTargets and Therapy. 2020;Volume 13:11645-11658.
CrossRef - Antonio Manuel Burgos-Molina, Teresa Téllez Santana, Redondo M, Bravo J. The Crucial Role of Inflammation and the Immune System in Colorectal Cancer Carcinogenesis: A Comprehensive Perspective. International Journal of Molecular Sciences. 2024;25(11):6188-6188.
CrossRef - Jain A, Chitturi S, Peters G, Yip D. Atezolizumab and bevacizumab as first line therapy in advanced hepatocellular carcinoma: Practical considerations in routine clinical practice. World Journal of Hepatology. 2021;13(9):1132-1142. doi:https://doi.org/10.4254/wjh.v13.i9.1132
CrossRef - Yang X. Structural studies of atom-specific anticancer drugs acting on DNA. Pharmacology & Therapeutics. 1999;83(3):181-215.
CrossRef
List of Abbreviations
BMDCs: Bone marrow-derived cells
APM: Antigen processing machinery
ACT: Adoptive T cell treatment
CLM: Colorectal liver metastasis
CRC: Colorectal cancer
CAR: Chimeric antigen receptor
CAR-T: Chimeric antigen receptor T cell
CTLA-4: Cytotoxic T-lymphocyte-associated antigen 4
DCs: Dendritic cells
dMMR: Defective DNA mismatch repair
EMT: Epithelial-mesenchymal transition
HGPs: Histological growth patterns
EVs: Extracellular vesicles
FDA: Food and Drug Administration
ICIs: Immune checkpoint inhibitors
KCs: Kupffer cells
mCRC: Metastatic colorectal cancer
MDSCs: Myeloid-derived suppressor cellsMMR DNA mismatch repair
MSI: Microsatellite instability,
MSI-H: Microsatellite instability-high,
MSS: Microsatellite-stable,
M2-TAMs: M2 tumor-associated macrophages,
LAG-3: Lymphocyte activation gene-3
LSECs: Liver sinusoidal endothelial cells,
PD-1: Programmed cell death 1,
PD-L1: Programmed death ligand
1pMMR: Mismatch repair-proficient,
NK: Natural killer,
ORRs: Objective response rates,
TDSFs: Tumor-derived secreted factors,
TIGIT: T-lymphocyte-infiltrating lymphocytes,
TILs: Tumor-infiltrating lymphocytes,
TMF: Tumor immune microenvironment, or TME,
TIM-3: T cell immunoglobulin mucin-3,
TNF: Tumor necrosis factor
TCRs: T cell receptors.







