Manuscript accepted on :04-11-2025
Published online on: 26-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Abdulrahman Rasheed Mahmood
Second Review by: Dr. Ana Golez
Final Approval by: Dr. Prabhishek Singh
Joel Padayachee1 , Ajit Kumar2
, Ajit Kumar2 , Takshita Sookan-Kassie3
, Takshita Sookan-Kassie3 and Irene Mackraj1*
and Irene Mackraj1*
1Discipline of Human Physiology, School of Basic Medical Sciences, College of Health Sciences, University of KwaZulu-Natal (Westville Campus), Durban, Republic of South Africa.
2Discipline of Microbiology, School of Life Sciences, College of Agriculture, Engineering and Science, University of KwaZulu-Natal (Westville Campus), Durban, Republic of South Africa.
3Discipline of Biokinetics, Exercise and Leisure Sciences, School of Health Science, University of KwaZulu-Natal (Westville Campus), Durban, Republic of South Africa.
Corresponding Author E-mail:mackraji@ukzn.ac.za
DOI : https://dx.doi.org/10.13005/bpj/3321
Abstract
The study aims to fill the gap in limited scientific evidence on variations in resting serum Interleukin-6 (IL-6), Interleukin-10 (IL-10), and Tumor Necrosis Factor-alpha (TNF-α) levels, as well as their associations with respiratory patterns across different recreational sports and sedentary individuals, particularly in South Africa. This cross-sectional study involved 80 male participants, comprising 20 from each of the three sport subgroups (swimming, soccer, and volleyball) and 20 from a sedentary group. Spirometry values for Forced Vital Capacity (FVC), Forced Expiratory Volume in one second (FEV1), Peak Expiratory Flow Rate (PEFR), Vital Capacity (VC), FEV1/FVC ratio, and Maximal Voluntary Ventilation (MVV) were measured according to American Thoracic Society guidelines. Fasting plasma levels of IL-6, IL-10, and TNF-α were assessed after a two-week break from training for the sports groups, and sedentary behavior was defined for the sedentary group. Significant differences in respiratory patterns were observed among the groups (p < 0.01), with recreational athletes exhibiting better lung function than the sedentary group. The cytokine expression levels for IL-10 and TNF-α in the swim, soccer, volleyball, and sedentary groups were significantly different (p < 0.01). The study confirms that an increase in anti-inflammatory cytokines, such as IL-10, is associated with improved spirometry values, while TNF-α cytokine expression is associated with reduced spirometry values. Swimming notably enhances lung function compared to soccer and volleyball. Long-term physical activity has been shown to reduce inflammation compared to sedentary lifestyles. Larger studies are needed to further explore the relationship between cytokine levels and lung function in recreational sports.
Keywords
Cytokines; Pulmonary function; Recreational athletes; Respiratory profiles; Sedentary lifestyle
Download this article as:| Copy the following to cite this article: Padayachee J, Kumar A, Kassie T. S, Mackraj I. Cytokine and Respiratory Profiles in Recreational Athletes and a Sedentary Group. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Padayachee J, Kumar A, Kassie T. S, Mackraj I. Cytokine and Respiratory Profiles in Recreational Athletes and a Sedentary Group. Biomed Pharmacol J 2025;18(4). |
Introduction
Studies investigating the effect of physical activities on pulmonary patterns consistently support the role of different physical activities in influencing these patterns.1, 2 However, some studies have reported that pulmonary values between different sports disciplines are statistically insignificant for certain parameters, such as Forced Vital Capacity (FVC) and Forced Expiratory Volume in one second (FEV1), between aerobic and anaerobic athletes.3 When comparing various cohorts of football, hockey, volleyball, swimmers, basketball, and a control group of players, significant differences in FVC, FEV1, and Maximal Voluntary Ventilation (MVV) were recorded in favor of all athletic groups,4 with swimmers consistently portraying the best pulmonary function.5 Therefore, it is generally assumed that athletes are more likely to demonstrate increased spirometry measures than their sedentary counterparts.6 The descriptive data concerning anthropometric and respiratory characteristics are documented.7 While respiratory patterns in individual sports have been extensively researched compared to sedentary groups, there is a notable scarcity in the study of multiple recreational sport groups within a single investigation.8 Further investigation is particularly required from a South African cohort in the context of recreational sport groups.9 Inconclusive findings and a lack of data for pulmonary patterns in South African recreational sport groups highlight the need to address this gap in the literature.
Regarding exercise and cytokine levels, studies have shown that exercise may alter cytokine levels,10 although the association between exercise and cytokine expression is complex and may vary across different sports branches.11 Interleukin 6 (IL-6) and tumor necrosis factor-α (TNF-α) are primarily recognized as pro-inflammatory cytokines that play a crucial role in the acute inflammatory response.11 Their levels are typically elevated when acute moderate to extreme intensity exercise (>85−90% of maximal heart rate) is performed.11 However, IL-6 is known for its dual nature, acting as both a pro- and anti-inflammatory cytokine,12 which adds to the complexity of this particular cytokine. Both IL-6 and IL-10 are associated with the activation of anti-inflammatory cascades and may also inhibit TNF-α, a known mediator of tissue damage.11 Prolonged bouts of intense exercise are characterized by higher concentrations of IL-6, followed by increases in cytokine inhibitors, such as the anti-inflammatory cytokine IL-10. Chronic training, on the other hand, can reduce the release of IL-6 by skeletal muscle because exercise improves the energy performance of the myocytes.13
There is a lack of consistent scientific evidence demonstrating possible variations in resting serum levels of IL-6, IL-10, and TNF-α between different sports disciplines. However, some findings have indicated that differences in cytokine levels between individuals who exercise and a control group may not be statistically significant.14 Given the contrasting evidence reported in studies concerning respiratory inflammation and the limited sport-specific studies, further investigation is essential. Regarding exercise and cytokine levels, inconsistent findings and a lack of available data for recreational athletes in a South African cohort necessitate determining the optimal inflammatory profile influenced by recreational sport disciplines.15
Hence, the present study investigated respiratory profiles as well as cytokine profiles (IL-6, IL-10, TNF-α) in adult males participating in three different recreational sporting branches and compared them to a sedentary group. The ongoing global health challenges, exacerbated by the recent Coronavirus Disease 2019 (COVID-19) pandemic, have underscored the crucial importance of physical activity for maintaining respiratory function and modulating the immune system.16 Sedentary lifestyles have been identified as a significant risk factor for severe outcomes in respiratory illnesses, including SARS-CoV-2 infection.17 Therefore, understanding how different recreational physical activities influence lung health and inflammatory markers is increasingly relevant for public health strategies. Recent literature emphasizes the role of physical activity in reducing systemic inflammation and improving cardiorespiratory fitness, which are crucial for mitigating the severity of various diseases, including COVID-19.18-20 Thus, the study will contribute to this growing body of knowledge, particularly within a South African context.
Materials and Methods
Participants
The study was conducted between April 2018 and March 2019. A recreational athlete for the purpose of this study was defined as a male athlete participating in active sport playing three times a week (60-minute duration per session).21 A cross-sectional study included 80 male participants comprising recreational athletes from three subgroups: soccer (20), swimming (20), and volleyball (20), as well as a sedentary group of 20. The inclusion criteria for recreational athletes were participation in recreational soccer, volleyball, or swimming, with a training frequency of three one-hour sessions per week. Adult males who are 18 years or older were required for the study. Smokers, former smokers, individuals using medication, or those with a medical history of respiratory diseases were excluded from the study. Participants were self-recruited by placing invitational notices at various sports clinics, physiotherapy clinics, and local gyms. Participants were informed of the study’s aim to motivate their interest in participating, and they were provided with the study protocol and informed consent.
Ethical approval
Regulatory, ethical, and institutional approval was obtained from the Biomedical Research Ethics Committee (BREC) of the University of KwaZulu-Natal (UKZN) (BE 012/18), South Africa.
Design
Anthropometric characteristics measurements
Standardized anthropometric techniques were used to complete height (meters) and weight (kilograms) measurements. Full attention was given to ensure that the participant’s body was fully upright, and the participant was barefoot with minimal clothing. The subjects’ backs were in contact with the wall, with both heels placed side by side and touching the base of the wall for the height measurement. Weight was measured using a scale, and height was measured using a stadiometer. BMI calculations were completed (kg/m²) using an average of three measurements.22 BMI was categorized as <18.5 (underweight), 18.5–24.9 (normal), 25.0–29.9 (overweight), and ≥30 (obese) kg/m2.23
Lung function measurements
The spirometry measurements were performed by a trained physiotherapist using the MIR SPIROLAB II spirometer (Mediotronics Physical Medicine, Johannesburg, South Africa). The American Thoracic Society (ATS) recommendations were followed.24 The test was performed in a seated position with the use of a nose clip. Subjects were required to perform a maximal inhalation, with sealed lips over the mouthpiece, followed by a rapid maximal exhalation. Subjects continued exhaling for a minimum of 6 seconds to facilitate Forced Expiratory Volume in one second (FEV1) and Forced Vital Capacity (FVC) measurements. Tests were performed in triplicate until the two highest recorded values varied by less than 3%. Measurements were completed under standard environmental conditions based on temperature (18–22°C), atmospheric pressure (760 mmHg), and a relative atmospheric humidity of 30%–60%.25 Measurements were taken in the morning, between 8 AM and 12 PM, to minimize diurnal variations in lung function.
Cytokine measurement
Groups of 10 participants were taken to the NSN LAB (Verulam, KwaZulu-Natal, South Africa) over a period of 8 sessions to collect the resting blood samples. To control circadian rhythm, all testing was performed in the morning hours at the NSN LAB using the Beckman Coulter Access Immuno-Assay System. ELISA kits were used for the determination of TNF-α (Immunotech, Beckman Coulter Company, Marseille, France, Cat no: IM1121), IL-6 (Immunotech, Beckman Coulter Company, Marseille, France, Cat no: A30945), and IL-10 (Invitrogen, USA, Cat no: BMS215INST) levels. Blood samples (6 mL) were obtained from the antecubital vein in the recreational athletic and sedentary groups by a medical doctor (registered with the Health Professional Council of South Africa) to ensure uniformity in blood collection. The samples were collected, and anonymous labels (e.g., SW1 for the first swimming participant, S1 for the first soccer participant, and V1 for the first volleyball participant) were placed on each tube. The samples were then analyzed immediately. The cytokine levels in the samples were determined according to the manufacturer’s instructions for the Beckman Coulter South Africa kit.
Statistics
IBM Statistical Package for Social Sciences (IBM-SPSS v.27, Chicago, IL, USA) was utilized to analyze the data. Mean and standard deviations were used to present continuous data. The Kolmogorov-Smirnov test was used to assess the normality of the data. Pearson correlation analysis was used to assess the strength of the relationship between cytokine and respiratory profiles. An ANOVA analysis was conducted to compare spirometry values among the swim, soccer, and volleyball groups. Independent t-tests were conducted to compare each sport discipline with the sedentary group. The value of p<0.05 was considered statistically significant.
Results
Anthropometric characteristics
Online Resource 1 presents the anthropometric characteristics of swimmers, soccer players, volleyball players, and the sedentary group. In the swim group, the findings were as follows: a mean height of 1.72 ± 0.05 m, a weight of 53.72 ± 3.97 kg, and a BMI of 18.23 ± 0.86 kg/m². The soccer group had a mean height of 1.82 ± 0.01 m, a weight of 68.52 ± 2.96 kg, and a BMI of 20.73 ± 0.85 kg/m². The volleyball group measures reflect a mean height of 1.75 ± 0.05 m, a weight of 62.32 ± 2.25 kg, and a BMI of 20.37 ± 0.85 kg/m². The sedentary group had a mean height of 1.68 ± 0.03 m, a weight of 82.20 ± 2.53 kg, and a BMI of 28.89 ± 1.35 kg/m².
Respiratory patterns in the swim, soccer, volleyball, and sedentary groups
The data is presented as mean spirometry values across the swim, soccer, volleyball, and sedentary groups (Figure 1). The mean Forced Vital Capacity (FVC) for the swim group was 5.51 L, Forced Expiratory Volume in one second (FEV1) was 5.21 L, Peak Expiratory Flow Rate (PEFR) was 10.51 L/min, VC was 5.23 L, FEV1/FVC ratio was 94.75%, and Maximal Voluntary Ventilation.
 |
Figure 1: Spirometry values (FVC-Forced Vital Capacity, FEV1-Forced Expiratory Volume (FEV1), PEFR-Peak Expiratory Flow Rate, VC-Vital Capacity, FEV1/FVC ratio-Forced Expiratory Volume-Forced Vital Capacity Ratio, and MVV- Maximal Voluntary Ventilation) among sedentary, swim, soccer, and volleyball groups.
|
(MVV) was 170.65 L/min. For the soccer group, the mean FVC was 4.99 L, FEV1 was 4.59 L, PEFR was 9.85 L/min, Vital Capacity (VC) was 4.95 L, FEV1/FVC ratio was 91.56%, and MVV was 164.52 L/min. The volleyball group showed mean values of 5.10 L for FVC, 4.82 L for FEV1, 9.29 L/min for PEFR, 5.11 L for VC, 91.24% for FEV1/FVC ratio, and 160.12 L/min for MVV. In contrast, the sedentary group presented with a mean FVC of 3.73 L, FEV1 of 3.23 L, PEFR of 9.13 L/min, VC of 4.11 L, FEV1/FVC ratio of 86.79%, and MVV of 115.81 L/min. The analysis of variance (ANOVA) revealed that the respiratory patterns among the three recreational sport groups (swimming, soccer, volleyball) were significantly different (p < 0.05). Specifically, there was a statistically significant difference between groups for FVC (F(2,57) = 10.02, p<0.05), FEV1 (F(2,57)=11.55, p<0.05), PEFR (F(2,57)=20.33, p<0.05), VC (F(2,57)=14.84, p<0.05), FEV1/FVC ratio (F(2,57)=4.29, p<0.05), and MVV (F(2,57)=113.18, p<0.05) (Online Resource 2). Comparisons between individual recreational sport groups and the sedentary group revealed further significant differences (Online Resource 3-5). The swim group showed significantly higher FEV1/FVC ratio (t29.767=6.94, p<0.05) and MVV (t22.026=25.82, p<0.05) compared to the sedentary group. The soccer group also exhibited significantly greater PEFR (t21.131=3.27, p<0.05), FEV1/FVC ratio (t28.525=4.742, p<0.05), and MVV (t21.148=23.19, p<0.05) than the sedentary group. For the volleyball group, FEV1 (t23.76=10.70, p<0.05) and MVV (t20.55=21.25, p<0.05) were significantly higher than in the sedentary group.
Cytokine Profile of the Groups
The results show that the swim group recorded mean cytokine levels of IL-6 (2.16 pg/mL), IL-10 (2.07 pg/mL), and TNF-α (2.41 pg/mL). For the soccer group, the mean levels were IL-6 (2.25 pg/mL), IL-10 (3.01 pg/mL), and TNF-α (1.87 pg/mL). The volleyball group exhibited mean levels of IL-6 (2.00 pg/mL), IL-10 (2.31 pg/mL), and TNF-α (2.11 pg/mL). In contrast, the sedentary group demonstrated substantially higher mean cytokine levels: IL-6 (10.72 pg/mL), IL-10 (9.91 pg/mL), and TNF-α (11.04 pg/mL) (Figure 2). The correlation between IL-6, IL-10, and TNF-α and spirometry values among the athlete groups shows that the correlations between IL-6 and pulmonary values were not significant (p > 0.05) for all three recreational sport groups (swimming, soccer, volleyball) and the sedentary group (Online Resource 6). The data show that no significant correlations were reported between IL-10 and spirometry values in recreational athletes (p > 0.05). However, a significant correlation (p < 0.05) was found for FVC, PEFR, and the FEV1/FVC ratio in the sedentary group (Online Resource 7). Additionally, no correlation was found between TNF-α and the spirometry values (p > 0.05) in the swim, soccer, and sedentary groups. However, TNF-α correlated negatively with FEV1 (r=−0.47, p<0.05), VC (r=−0.54, p<0.05), and the FEV1/FVC ratio (r=−0.58, p<0.01) in the volleyball players (Online Resource 8).
Further, when cytokine values between the swim and sedentary groups were compared, significant differences (p<0.05) were observed for IL-6 (t22.65 = -49.35), 8.55 lower; IL-10 (t20.03 =−30.34), 7.83 lower; and TNF-α (t19.42 =−24.31), 8.62 lower values were observed (Online Resource 9). The cytokine values comparison (p<0.05) between the soccer and sedentary groups shows that IL-6 (t22.67=−48.85), IL-10 (t19.74=−27.04), and TNF-α (t19.05=−25.98), values were significantly lower (8.47, 6.96 and 9.17, respectively) in the soccer group compared to the sedentary group (Online Resource 10). The cytokine values between the volleyball and sedentary groups were found to be significantly different (p<0.05), with IL-6 (t37.81=−39.69), IL-10 (t19.65=−29.73), and TNF-α (t19.86=−25.00) values, all being lower (8.75, 7.64 and 8.92, respectively) in the volleyball group (Online Resource 11). The ANOVA results for the three recreational sport groups regarding cytokine levels showed that there was no statistically significant difference in IL-6 values among the swim, soccer, and volleyball groups (F(2,57) = 2.176, p = 0.123). However, there was a statistically significant difference between groups for IL-10 values (F(2,57)=154.017, p<0.05) and for TNF-α values (F(2,57)=51.466, p<0.05) (Online Resource 12).
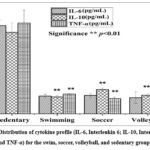 |
Figure 2: Distribution of cytokine profile (IL-6, Interleukin 6; IL-10, Interleukin 10, and TNF-α) for the swim, soccer, volleyball, and sedentary groups.
|
Discussion
This study aimed to determine the respiratory and cytokine profiles in three recreational sport groups (swimming, soccer, and volleyball) and a sedentary group of adult males in South Africa. Significant inter-group variations were observed in height, weight, and Body Mass Index (BMI). All athletic groups maintained healthy mean BMI values, with swimmers exhibiting notably lower weight and BMI compared to other athletic cohorts. The soccer group displayed higher weights and BMIs than the swim and volleyball groups, while volleyball players showed increased heights relative to soccer players. The sedentary group presented with the highest weight and BMI, falling into the overweight category (Online Resource 1).
Furthermore, it examined the correlation between individual cytokine levels (IL-6, IL-10, and TNF-α) and respiratory patterns (FVC, FEV1, PEFR, VC, FEV1/FVC, and MVV) within these groups. The findings offer valuable insights into the physiological adaptations associated with various types of regular recreational physical activity and highlight the detrimental effects of a sedentary lifestyle on these parameters.
The results clearly indicate that recreational athletes, regardless of their specific sport, demonstrate superior lung function compared to their sedentary counterparts (Figure 1). This aligns with a vast body of literature asserting the positive impact of physical activity on respiratory health.1, 6 The significant differences observed in FEV1, FVC, and MVV between the active and sedentary groups underscore the importance of regular exercise for maintaining and improving pulmonary capacity. Recent studies continue to reinforce this, showing that consistent physical activity is associated with better lung function and reduced risk of respiratory impairments.1 The high MVV values observed in all three recreational sport groups, particularly when compared to the sedentary group, are consistent with the understanding that athletes develop greater respiratory muscle endurance and efficiency through consistent training, allowing them to operate at higher proportions of their maximal ventilatory capacity.16 This enhanced ventilatory capacity is crucial, especially in light of the global challenges posed by respiratory illnesses like COVID-19, where robust lung function can serve as a protective factor against severe outcomes.17, 19
Beyond the general benefit of activity, our study found significant differences in respiratory patterns among the recreational sport groups themselves. Swimmers, in particular, consistently exhibited superior lung volumes and respiratory capacities compared to both recreational soccer and volleyball players. This finding is in strong agreement with numerous previously published studies.5 The unique aquatic environment plays a significant role in these adaptations. The hydrostatic pressure of water provides external resistance to breathing, particularly during exhalation, which serves as a natural training mechanism for the respiratory muscles, leading to increased strength and endurance. This continuous resistance training can enhance lung elasticity and promote more effective breathing patterns.26 Furthermore, the horizontal supine position often adopted during swimming promotes thoracic breathing and can reduce “physiological dead space,” thereby improving lung diffusion capacity.27 The cooler water temperatures (21–28°C) also contribute by improving respiratory patterns, reducing breaths per minute, and lowering pulse rates, optimizing cardiorespiratory efficiency.28 This specialized training environment explains why swimmers, despite not always having the highest height or overall body mass, consistently exhibit superior pulmonary function.
While soccer players in our study also showed better lung function than the sedentary group, their spirometry values were generally lower than those of swimmers. This is consistent with findings that highlight the specific adaptations unique to water-based activities.29 Soccer involves intermittent, high-intensity movements that primarily challenge the aerobic and anaerobic systems. Regular running and sprinting in soccer training have a positive impact on lung function by improving pulmonary capacity.30 Similarly, volleyball players demonstrated improved respiratory function compared to sedentary individuals, attributed to the frequent short bursts of high-intensity exercise followed by low-intensity periods, which demand significant aerobic and anaerobic energy systems.31 However, the cumulative and constant resistance provided by water in swimming appears to confer a distinct advantage in developing maximal lung volumes and capacities. This sport-specific adaptation of the respiratory system underscores that while any physical activity is beneficial, certain activities, such as swimming, may offer more pronounced pulmonary benefits —a crucial consideration for public health recommendations aimed at optimizing lung health.
A significant finding of this study is that all three recreational sport groups exhibited substantially lower resting levels of IL-6, IL-10, and TNF-α compared to the sedentary group (Figure 2). This supports the well-established notion that long-term physical activity, even at a recreational level, promotes a lower systemic inflammatory state.32 Regular exercise induces an anti-inflammatory environment by reducing pro-inflammatory cytokine production and enhancing anti-inflammatory responses.33 This is critically important, as chronic low-grade systemic inflammation is a hallmark of many non-communicable diseases (NCDs) and can exacerbate outcomes in infectious diseases, as seen with COVID-19.34, 35 The high levels of IL-6, IL-10, and TNF-α observed in our sedentary group (Figure 2), which fall above normal ranges,36 further reinforce the link between sedentary behavior and elevated systemic inflammation.37, 38 This chronic inflammatory state in sedentary individuals can contribute to an increased risk of NCDs and potentially poorer immune responses.39, 40 Interestingly, while all active groups exhibited lower inflammatory markers than the sedentary group, notable differences were observed in cytokine profiles among the various recreational sports disciplines. Specifically, IL-10 and TNF-α levels were statistically significantly lower among the swim, soccer, and volleyball groups (Online Resource 8). The volleyball group showed the lowest IL-6 level, followed by the swim and soccer groups. Our finding that the swim group did not have the lowest absolute IL-6 levels contrasts with some previous studies, where water-based activities were associated with minimal inflammatory responses.41 This discrepancy might be attributed to various factors, including differences in exercise intensity, duration, training protocols, or measurement timing relative to the last training session. Swimming, being predominantly concentric and low-impact, generally induces less muscle damage and, consequently, a lower inflammatory response compared to eccentric activities common in land-based sports.41 However, IL-6 responses can also be influenced by the number of muscle groups activated during exercise.42 Soccer and volleyball, involving both upper and lower limb movements, might theoretically elicit a broader IL-6 response due to greater muscle activation, which aligns with our soccer findings but not entirely with our volleyball results. The complex interplay of exercise mode, intensity, and duration on cytokine kinetics requires further nuanced investigation.43
The lower pro-inflammatory IL-10 expressions in the recreational sport groups compared to the sedentary group in our study is a counter-intuitive finding initially, as IL-10 is generally considered an anti-inflammatory cytokine, and exercise is expected to increase its production.11 However, the highly elevated IL-10 in the sedentary group warrants closer examination. This could be a compensatory response in the sedentary group to chronically elevated pro-inflammatory cytokines, such as IL-6 and TNF-α (Figure 2). In a state of chronic low-grade inflammation, the body may upregulate anti-inflammatory cytokines, such as IL-10, to try to dampen the inflammatory processes. Previous research on older sedentary men, for instance, has shown higher IL-10 levels, which may be linked to age-related inflammaging or a compensatory mechanism.32 The self-recruitment of participants in our sedentary group could have introduced a selection bias, potentially including individuals with underlying low-grade inflammatory conditions that manifest as higher baseline cytokine levels, including a compensatory rise in IL-10. Dietary habits also significantly influence inflammatory profiles, and differences in dietary intake could confound the interpretation of cytokine levels across cohorts.44 This complex interaction between pro- and anti-inflammatory cytokines in chronic sedentary states requires further detailed investigation, perhaps through longitudinal studies that track dietary intake and inflammatory markers over time.
The lack of a statistically significant association between IL-6 expression and respiratory patterns across all groups (Online Resource 5) is a notable finding. While some studies report an inverse relationship between higher IL-6 levels and reduced FEV1 and FVC in respiratory inflammation,45 other studies, consistent with ours, have found no such correlation in healthy populations or specific disease states.46 This suggests that while IL-6 is a key inflammatory marker, its direct correlation with baseline spirometry values in generally healthy (even if sedentary) young adult males might be limited, or its influence becomes more pronounced in overt respiratory diseases or acute inflammatory conditions.
Interestingly, while IL-10 showed no significant correlations with spirometry in the recreational athletes, it did correlate significantly with FVC, PEFR, and FEV1/FVC ratio in the sedentary group (Online Resource 6). This might suggest that in a sedentary population experiencing chronic low-grade inflammation, the body’s compensatory anti-inflammatory response (reflected by IL-10) might play a more discernible role in modulating lung function, potentially attempting to mitigate the negative effects of inflammation on respiratory parameters. Studies have indeed highlighted a potential role for IL-10 in modulating inflammatory responses and lung function, particularly in contexts of disease or chronic stress.47 For active individuals with already low baseline inflammation, the subtle variations in IL-10 might not translate into significant correlations with robust lung function parameters.
Conversely, TNF-α showed negative correlations with FEV1, VC, and FEV1/FVC ratio in the volleyball group (Online Resource 4), indicating that higher TNF-α levels were associated with lower lung function in this specific sport group. This finding aligns with previously published work linking elevated pro-inflammatory TNF-α with reduced spirometric values.48 While the exact mechanism specific to volleyball players requires further investigation, this could imply that the intermittent high-intensity nature of volleyball might, in some cases, lead to transient inflammatory responses involving TNF-α that could subtly impact lung mechanics. However, the lack of a similar correlation in the swim and soccer groups, or the sedentary group, suggests that this relationship might be context-dependent, possibly influenced by factors such as muscle damage patterns, recovery kinetics, or individual variability within the volleyball cohort. Prior studies on TNF-α and FEV1 have also yielded inconsistent results, underscoring the complexity of these relationships.49, 50
Conclusion
This study provides compelling evidence that consistent recreational physical activity positively influences both respiratory patterns and cytokine profiles in young adult males. The findings reinforce that anthropometric characteristics and respiratory patterns are largely sport-specific, with swimming demonstrating superior pulmonary adaptations compared to land-based sports like soccer and volleyball. More broadly, all recreational sport groups exhibited significantly healthier respiratory parameters and markedly lower resting inflammatory markers (IL-6, IL-10, and TNF-α) compared to the sedentary group. This underscores the profound benefit of even recreational-level physical activity in maintaining optimal lung health and fostering an anti-inflammatory state, both of which are crucial for overall well-being and resilience against disease.
While direct correlations between specific cytokines and spirometry values were not always consistent across all active groups, the significant inverse relationship between TNF-α and lung function in the volleyball group, and the positive correlations of IL-10 with spirometry in the sedentary group, warrant further exploration. These findings suggest nuanced interactions between inflammation and respiratory mechanics that may differ based on activity level and specific sport demands. From a public health perspective, the message remains clear: a physically active lifestyle, even though accessible recreational sports, is a powerful intervention against the rising burden of non-communicable diseases and contributes to enhanced respiratory and immune health, which has become even more pertinent in the era of widespread respiratory infections like COVID-19. Continued research with larger, more diverse samples and longitudinal designs will further elucidate these complex physiological relationships.
Acknowledgment
The author would like to thank Late Dr K. Moodley for her supervision and assistance in the project.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data availability Statement
This statement does not apply to this article.
Ethics statement
This article contains studies with human participants. All persons gave their informed consent prior to their inclusion in the study and that details that might disclose the identity of the subjects under study have been omitted. Regulatory, ethical, and institutional approval was obtained from the Biomedical Research Ethics Committee (BREC) of the University of KwaZulu-Natal (UKZN) (BE 012/18), South Africa.
Informed Consent Statement
The informed consent was obtained for experimentation and that it conforms to the standards currently applied in the country of origin. The privacy rights of human subjects must always be observed.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not applicable
Author contributions
- Joel Padayachee: Data Collection, Methodology, and Writing – Original Draft.
- Ajit Kumar: Visualization, review editing, and figure formatting.
- Takshita Sooknan: Review editing.
- Irene Mackraj: Conceptualization, Analysis, Supervision, and Editing.
References
- Ogonowska-Slodownik A, Labecka MK, Maciejewska-Skrendo A, et al. Effect of water- and land-based exercise on lung function in children with post-COVID-19 condition: secondary results from a randomised controlled trial. ERJ Open Res. 2025;11(3):00838–02024. https://doi.org/10.1183/23120541.00838-2024
CrossRef - Chen X, Ma J, Dong L, Chen Y, Chen K. The association between physical activity and lung function in adolescents: a cross-sectional NHANES study. Front Med. 2025;12:1538221. https://doi.org/10.3389/fmed.2025.1538221
CrossRef - Rajaure YS, Thapa B, Budhathoki L, Rana SR, Khadka M. Comparison of spirometric parameters in athletes engaged in aerobic and anaerobic sports: a cross-sectional study. Ann Med Surg. 2023;85(6):2502–2505. https://doi.org/10.1097/ms9.0000000000000729
CrossRef - El Nady HG, El Refay AS, Salah DA, Fahmy RF, Mansour SA, Sherif LS. Impact of sport specific type on pulmonary function and anthropometric measures of adolescents. Biomed Pharmacol J. 2023;16(2):763–771. https://doi.org/10.13005/bpj/2658
CrossRef - Rodrigues J, Jesus B, Caseiro P, Ferreira AJ, Rama L. Lung function changes with swim training in healthy and allergic endurance athletes. J Funct Morphol Kinesiol. 2025;10(2). https://doi.org/10.3390/jfmk10020231
CrossRef - Navya JJ, Krishna KRC. Comparative study of lung function between athletes and non-athletes. Int J Life Sci Biotechnol Pharm Res. 2025;14(5):930–940. https://doi.org/10.69605/ijlbpr_14.5.2025.159
- Chiwaridzo M, von der Heiden C, Kamba FE, Mkumbuzi NS. Differences in playing experience, anthropometry and performance measures between Under 16 schoolboy rugby players classified as starters or non-starters: A comparative cross-sectional study. S Afr J Sports Med. 2025;37(1). https://doi.org/10.17159/2078-516X/2025/v37i1a19947
CrossRef - Hackett DA. Lung function and respiratory muscle adaptations of endurance- and strength-trained males. Sports. 2020;8(12):160. https://doi.org/10.3390/sports8120160
CrossRef - Bamne SN. Comparative study of pulmonary functions in swimmers and badminton players of Indore city. Natl J Physiol Pharm Pharmacol. 2017;7(9):999–1002. https://doi.org/10.5455/njppp.2017.7.0518230052017
CrossRef - Malkowska P, Sawczuk M. Cytokines as biomarkers for evaluating physical exercise in trained and non-trained individuals: A narrative review. Int J Mol Sci. 2023;24(13):11156. https://doi.org/10.3390/ijms241311156
CrossRef - Docherty S, Harley R, McAuley JJ, et al. The effect of exercise on cytokines: implications for musculoskeletal health: a narrative review. BMC Sports Sci Med Rehabil. 2022;14(1):5. https://doi.org/10.1186/s13102-022-00397-2
CrossRef - Reis A, Furtado GE, Menuchi M, Borges GF. The Impact of Exercise on Interleukin-6 to Counteract Immunosenescence: Methodological Quality and Overview of Systematic Reviews. Healthcare. 2024;12(10):954. https://doi.org/10.3390/healthcare12100954
CrossRef - Hennigar SR, McClung JP, Pasiakos SM. Nutritional interventions and the IL-6 response to exercise. FASEB J. 2017;31(9):3719–3728. https://doi.org/10.1096/fj.201700080R
CrossRef - Gómez-Rubio P, Trapero I. The effects of exercise on IL-6 levels and cognitive performance in patients with schizophrenia. Diseases. 2019;7(1):11. https://doi.org/10.3390/diseases7010011
CrossRef - Sohail MU, Al-Mansoori L, Al-Jaber H, et al. Assessment of serum cytokines and oxidative stress markers in elite athletes reveals unique profiles associated with different sport disciplines. Front Physiol. 2020;11:600888. https://doi.org/10.3389/fphys.2020.600888
CrossRef - Thirupathi A, Yong W, Oflaz O, Agascioglu E, Gu Y. Exercise and COVID-19: Exercise intensity reassures immunological benefits of post-COVID-19 condition. Front Physiol. 2023;14:1036925. https://doi.org/10.3389/fphys.2023.1036925
CrossRef - Nahvi R, Shakibaee A, Salesi M, Esmaeili H, Bazgir B. The role of physical activity in COVID-19 mortality rate: A cross-sectional study. Iran J Nurs Midwifery Res. 2024;29(6):726–730. https://doi.org/10.4103/ijnmr.ijnmr_109_23
CrossRef - Balbinot F, Gerbase MW. Physical activity predicts better lung function in children and adolescents. Pediatr Exerc Sci. 2024:1–8. https://doi.org/10.1123/pes.2024-0034
CrossRef - Myers J, Jones AW. Editorial: The relationship of physical activity and cardiorespiratory fitness with acute COVID-19 infection and post COVID-19 conditions. Front Sports Act Living. 2024;6:1444947. https://doi.org/10.3389/fspor.2024.1444947
CrossRef - Silva-Santos S, Monteiro AM, Barbosa TM, et al. Associations between coronavirus and immune response, cardiorespiratory fitness rehabilitation and physical activity: A brief report. Int J Environ Res Public Health. 2023;20(5): 4651. https://doi.org/10.3390/ijerph20054651
CrossRef - McKay AKA, Stellingwerff T, Smith ES, et al. Defining training and performance caliber: A participant classification framework. Int J Sports Physiol Perform. 2022;17(2):317–331. https://doi.org/10.1123/ijspp.2021-0451
CrossRef - Ettarh R, Van de Vijver S, Oti S, Kyobutungi C. Overweight, obesity, and perception of body image among slum residents in Nairobi, Kenya, 2008–2009. Prev Chronic Dis. 2013;10:E212. https://doi.org/10.5888/pcd10.130198
CrossRef - Koyanagi A, Stubbs B, Vancampfort D. Correlates of sedentary behavior in the general population: A cross-sectional study using nationally representative data from six low- and middle-income countries. PLoSOne. 2018;13(8). https://doi.org/10.1371/journal.pone.0202222
CrossRef - Graham BL, Steenbruggen I, Miller MR, et al. Standardization of spirometry 2019 update. An official american thoracic society and european respiratory society technical statement. Am J Respir Crit Care Med. 2019;200(8):e70–e88. https://doi.org/10.1164/rccm.201908-1590ST
CrossRef - Durmic T, Lazovic Popovic B, Zlatkovic Svenda M, et al. The training type influence on male elite athletes’ ventilatory function. BMJ Open Sport Exerc Med. 2015;3(1):e000240. https://doi.org/10.1136/bmjsem-2017-000240
CrossRef - Päivinen M, Keskinen K, Tikkanen H. Swimming-induced changes in pulmonary function: special observations for clinical testing. BMC Sports Sci Med Rehabil. 2021;13:55. https://doi.org/10.1186/s13102-021-00277-1
CrossRef - Martín-Valero R, Cuesta-Vargas AI, Labajos-Manzanares MT. Evidence-based review of hydrotherapy studies on chronic obstructive pulmonary disease patients. Int J Aquatic Res Educ. 2012;6(3):1–14. https://doi.org/0.25035/ijare.06.03.08
CrossRef - Michalak K, Poziomska-Piątkowska E. The effect of swimming activity on lung function parameters among smoking and non-smoking youth – research extended. Hum Mov. 2015;16:229–233. https://doi.org/10.1515/humo-2015-0045
CrossRef - Tareq Z, Al-Madfai Z, Saeed Al-Ani G. The effect of training and sport type on pulmonary function parameters among Iraqi soccer and futsal players. J Sports Phys Educ. 2018;3(5):27–30. https://doi.org/10.9790/6737-03052730
- Maćkała K, Fostiak M, Schweyen B, Osik T, Coch M. Acute effects of a speed training program on sprinting step kinematics and performance. Int J Environ Res Public Health. 2019;16(17):3138. https://doi.org/10.3390/ijerph16173138
CrossRef - Gaurav V, Singh A. Anthropometric characteristics of Indian volleyball players in relation to their performance level. Turk J Sport Exercise. 2013;16(1):87–89. https://doi.org/10.15314/tjse.53123
CrossRef - Perez-Castillo IM, Rueda R, Bouzamondo H, et al. Does lifelong exercise counteract low-grade inflammation associated with aging? A systematic review and meta-analysis. Sports Med. 2025;55(3):675–696. https://doi.org/10.1007/s40279-024-02152-8
CrossRef - Ayari S, Abellard A, Carayol M, Guedj E, Gavarry O. A systematic review of exercise modalities that reduce pro-inflammatory cytokines in humans and animals’ models with mild cognitive impairment or dementia. Exp Gerontol. 2023;175:112141. https://doi.org/10.1016/j.exger.2023.112141
CrossRef - Dean E, Soderlund A, Gosselink R, Jones AYM, Yu HP, Skinner M. Immuno-modulation with lifestyle behaviour change to reduce SARS-CoV-2 susceptibility and COVID-19 severity: goals consistent with contemporary physiotherapy practice. Physiotherapy. 2022;114:63–67. https://doi.org/10.1016/j.physio.2021.08.006
CrossRef - Hajifathalian K, Kumar S, Newberry C, et al. Obesity is associated with worse outcomes in covid-19: Analysis of early data from new york city. Obesity. 2020;28:1606–1612. https://doi.org/doi.org/10.1002/oby.22923
CrossRef - Kim HO, Kim HS, Youn JC, Shin EC, Park S. Serum cytokine profiles in healthy young and elderly population assessed using multiplexed bead-based immunoassays. J Transl Med. 2011;9:113. https://doi.org/10.1186/1479-5876-9-113
CrossRef - Rodas L, Riera-Sampol A, Aguilo A, Martinez S, Tauler P. Effects of habitual caffeine intake, physical activity levels, and sedentary behavior on the inflammatory status in a healthy population. Nutrients. 2020;12(8):2325. https://doi.org/10.3390/nu12082325
CrossRef - Friedenreich CM, Ryder-Burbidge C, McNeil J. Physical activity, obesity and sedentary behavior in cancer etiology: epidemiologic evidence and biologic mechanisms. Mol Oncol. 2021;15(3):790–800. https://doi.org/10.1002/1878-0261.12772
CrossRef - Bhuiyan MA, Galdes N, Cuschieri S, Hu P. A comparative systematic review of risk factors, prevalence, and challenges contributing to non-communicable diseases in South Asia, Africa, and Caribbeans. J Health Popul Nutr. 2024;43(1):140. https://doi.org/10.1186/s41043-024-00607-2
CrossRef - Zaid NSN, Muhamad AS, Jawis MN, et al. The Effect of Exercise on Immune Response in Population with Increased Risk Factors for Cardiovascular Disease: A Systematic Review. Malays J Med Sci. 2024;31(5):83–108. https://doi.org/10.21315/mjms2024.31.5.6
CrossRef - Pawluk H, Wozniak A, Tafelska-Kaczmarek A, et al. The Role of IL-6 in ischemic stroke. Biomolecules. 2025;15(4). https://doi.org/10.3390/biom15040470
CrossRef - Mendham AE, Duffield R, Marino F, Coutts AJ. Differences in the acute inflammatory and glucose regulatory responses between small-sided games and cycling in sedentary, middle-aged men. J Sci Med Sport. 2015;18(6):714–719. https://doi.org/10.1016/j.jsams.2014.09.008
CrossRef - Gerosa-Neto J, Monteiro PA, Inoue DS, et al. High- and moderate-intensity training modify LPS-induced ex-vivo interleukin-10 production in obese men in response to an acute exercise bout. Cytokine. 2020;136:155249. https://doi.org/10.1016/j.cyto.2020.155249
CrossRef - Nilsson MI, Bourgeois JM, Nederveen JP, et al. Lifelong aerobic exercise protects against inflammaging and cancer. PLoSOne. 2019;14(1):e0210863. https://doi.org/10.1371/journal.pone.0210863
CrossRef - Adair D, Bagheri A, Yosef M, et al. High interleukin (IL)-6 is associated with lower lung function and increased likelihood of metabolic dysfunction in asthma. Pulm Ther. 2025;11(1):41–54. https://doi.org/10.1007/s41030-024-00281-z
CrossRef - El-Shimy WS, El-Dib AS, Nagy HM, Sabry W. A study of IL-6, IL-8, and TNF-α as inflammatory markers in COPD patients. Egypt J Bronchol. 2014;8:91–99. https://doi.org/10.4103/1687-8426.145698
CrossRef - Chavda VP, Feehan J, Apostolopoulos V. Inflammation: The cause of all diseases. Cells. 2024;13(22):1906. https://doi.org/10.3390/cells13221906
CrossRef - Jiang E, Fu Y, Wang Y, Ying L, Li W. The role and clinical significance of myeloperoxidase (MPO) and TNF-alpha in prognostic evaluation of T-COPD. BMC Pulm Med. 2025;25(1):192. https://doi.org/10.1186/s12890-025-03655-4
CrossRef - Phillips KM, Lavere PF, Hanania NA, Adrish M. The emerging biomarkers in chronic obstructive pulmonary disease: A narrative review. Diagnostics. 2025;15(10):1245. https://doi.org/10.3390/diagnostics15101245
CrossRef - Quarta S, Massaro M, Carluccio MA, et al. An exploratory critical review on TNF-α as a potential inflammatory biomarker responsive to dietary intervention with bioactive foods and derived products. Foods. 2022;11(16). https://doi.org/10.3390/foods11162524
CrossRef







