Manuscript accepted on :18-12-2025
Published online on: 26-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Heamn Noori Abduljabbar
Second Review by: Dr. Elina Margarida Ribeiro Marinho
Final Approval by: Dr. Prabhishek Singh
Maariz Rahman1* , Mohammed Humaun Kabir1
, Mohammed Humaun Kabir1 , Abeer Mohammad1
, Abeer Mohammad1 ,Iresha Katurusinghe Hettiarachchige1
,Iresha Katurusinghe Hettiarachchige1 , Oluwatobiloba Ogunbote1
, Oluwatobiloba Ogunbote1 , Nordia Pinnock1
, Nordia Pinnock1 , Sunitha Namani2
, Sunitha Namani2 and Sathyanarayana Namani2
and Sathyanarayana Namani2
1Faculty of Medicine, Saint James School of Medicine Cane Hall, Saint Vincent and the Grenadines
2Faculty of Medicine, cane hall, Saint James School of Medicine, Saint Vincent and the Grenadines.
Corresponding Author E-mail: mrahman2@mail.sjsm.org
DOI : https://dx.doi.org/10.13005/bpj/3287
Abstract
Melanoma is an aggressive malignancy with poor survival once metastatic. Although immune checkpoint inhibitors, particularly anti-programmed cell death protein-1 (PD-1) therapies, have improved outcomes, many patients remain refractory or develop resistance. Increasing evidence implies the gut microbiome as a key regulator of antitumor immunity, leading to interest in fecal microbiota transplantation (FMT) as a strategy to enhance immunotherapy efficacy, especially when derived from donors who previously responded to PD-1 blockade. A systematic review of 15 preclinical and clinical studies published between 2015 and 2025 was conducted using PubMed, Web of Science, Embase, and SJSM Library Resources. Included studies evaluated FMT in melanoma and reported clinical, immunologic, microbiome, and survival outcomes. Across three early-phase clinical trials involving 45 patients, FMT combined with anti-PD-1 therapy achieved an overall response rate of 65%, including 20% complete and 45% partial responses, with grade 3 immune-related adverse events occurring in approximately 10-15% of patients. Responders demonstrated sustained engraftment of donor-derived microbiota, characterized by enrichment of Bifidobacteriaceae, Ruminococcaceae, Lachnospiraceae, Faecalibacterium prausnitzii, and Akkermansia muciniphila, taxa associated with enhanced T-cell activation, improved mucosal integrity, and increased short-chain fatty acid production. Survival analyses revealed that low B-Raf expression was associated with longer survival in the absence of PD-1 therapy (32 vs. 24 months; p = 0.0512), while PD-1 treatment partially mitigated the adverse prognostic impact of high B-Raf expression (24 vs. 33 months; p = 0.0294). Longitudinal profiling showed that FMT-induced microbial changes persisted for 1-3 months in responders but regressed toward baseline in nonresponders, highlighting durable engraftment as a determinant of sustained benefit. Overall, FMT represents a feasible and biologically plausible adjunct to anti-PD-1 therapy in melanoma, with early evidence suggesting improved immune responsiveness and potential reversal of treatment resistance. Larger randomized trials are needed to optimize donor selection, confirm durability, and define long-term safety and mechanistic pathways.
Keywords
Anti–PD-1 Therapy; B-Raf Expression; Fecal Microbiota Transplantation (FMT); Gut Microbiota; Immune Checkpoint Inhibitors and Melanoma
Download this article as:| Copy the following to cite this article: Rahman M, Kabir M. H, Mohammad A, Hettiarachchige I. K, Ogunbote O, Pinnock N, Namani S, Namani S. Enhancing Immune Response in Immunotherapy-Resistant Melanoma Through Fecal Microbiota Transplantation: A Systematic Review. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Rahman M, Kabir M. H, Mohammad A, Hettiarachchige I. K, Ogunbote O, Pinnock N, Namani S, Namani S. Enhancing Immune Response in Immunotherapy-Resistant Melanoma Through Fecal Microbiota Transplantation: A Systematic Review. Biomed Pharmacol J 2025;18(4). |
Introduction
Melanoma is the most aggressive form of skin cancer, originating from neural-crest-derived melanocytes and characterized by early metastasis and a poor prognosis once it spreads beyond the epidermis. It is classified into five clinical stages, each associated with distinct five-year survival rates: approximately 99% for stage 0 (melanoma in situ, confined to the epidermis); 92-97% for stage I (early invasive melanoma, tumor thickness ≤ 2 millimeters without ulceration); stage II, according to updated American Joint Committee on Cancer criteria(AJCC), generally includes tumors greater than 2 millimeters in thickness, with or without ulceration, and is associated with five-year survival rates of approximately 55-80%; 68% for stage III (regional melanoma, spread to nearby tissues or lymph nodes); and about 30% for stage IV (distant melanoma, metastasized to distant organs, tissues, or lymph nodes).
Several risk factors increase the likelihood of developing melanoma, including fair skin phototypes (particularly among Caucasians), advancing age, immunosuppression (due to human immunodeficiency virus infection or medical treatment), and specific genetic mutations (such as cyclin-dependent kinase inhibitor 2A or B-Raf proto-oncogene. Although interactions between the gut microbiome and melanoma biology are being increasingly studied, a direct and consistent link between gastrointestinal disorders and melanoma risk has not been firmly established. Therefore, any such association should be interpreted cautiously. Although targeted therapies and immune checkpoint inhibitors including anti-programmed cell death protein 1 antibodies such as pembrolizumab and nivolumab have significantly improved survival outcomes, a considerable proportion of patients either fail to respond initially or eventually develop therapeutic resistance. 1
The gut microbiome has recently emerged as a potential modulator of systemic anti-tumor immunity. Preliminary studies suggest that fecal material from immunotherapy responders or melanoma survivors may enhance immune responsiveness and improve clinical outcomes in melanoma patients. Fecal microbiota transplantation, the process of transferring stool from a healthy donor into the gastrointestinal tract of a recipient, has been shown to restore microbial diversity and intestinal balance. Originally developed to treat Clostridioides difficile infections, fecal microbiota transplantation has demonstrated promising efficacy in restoring healthy gut flora; however, its application in oncology, including melanoma, remains experimental and requires further validation in controlled clinical studies.
The objective of this systematic review is to assess whether modulation of the gut microbiome through fecal microbiota transplantation has the potential to enhance immune responses, improve clinical outcomes, and possibly overcome resistance to anti-programmed cell death protein 1 therapies in melanoma patients, particularly among those who are non-responsive to conventional immunotherapies. ²,³
Materials and Methods
Literature Search Strategy
A comprehensive literature search was conducted across four major databases, including PubMed, Web of Science, Embase, and SJSM Library Resources, the institutional library system of Saint James School of Medicine that provides access to multiple academic databases and journal subscriptions. To ensure both relevance and currency, the search covered studies published between 2015 and the present. To refine and optimize the search strategy, a combination of predefined keywords and Boolean operators (AND, OR, NOT) was employed. Specifically, search terms included “fecal microbiota transplantation” or “FMT,” “melanoma,” “gut microbiome,” “immune checkpoint inhibitors” or “ICI,” and “skin cancer,” along with mechanism-focused combinations such as “microbiota AND immune response” and “microbiota AND mechanism.” Because the phrase “mechanism of action of microbiota” is not consistently indexed across databases, it was intentionally avoided and instead divided into more targeted keyword combinations to improve search sensitivity.
Following database retrieval, all identified records underwent a structured two-stage screening process to ensure methodological rigor. In the first stage, titles and abstracts were screened to exclude clearly irrelevant studies. Subsequently, full-text articles were assessed using predefined eligibility criteria. Through this process, 304 records were initially identified, of which 54 duplicates and 28 non-English, non-primary, or clearly irrelevant records were removed, resulting in 222 articles eligible for title and abstract screening. At this stage, 145 studies were excluded due to irrelevant disease focus, absence of microbiota or FMT-related interventions, or inadequate methodological quality and reporting.
Studies that passed the initial screening were then subjected to detailed eligibility assessment. Accordingly, 77 full-text articles were reviewed comprehensively, among which 62 were excluded after full-text evaluation. The primary reasons for exclusion included lack of focus on melanoma (n = 27), absence of FMT or microbiota-based interventions (n = 21), insufficient or unclear outcome data (n = 9), and use of irrelevant study populations (n = 5).
The remaining studies were assessed against predefined inclusion criteria to determine their suitability for final synthesis. Studies were included if they investigated the role of the gut microbiota or fecal microbiota transplantation in melanoma, were peer-reviewed and published in English, and involved either human participants or relevant animal models. Furthermore, eligible studies were required to evaluate clinically or biologically meaningful outcomes, such as immune response, tumor regression, microbiota composition, or survival. Non-English publications were excluded due to translation limitations and restricted access to certified translation resources, which may have led to omission of some relevant studies but was necessary to ensure consistency and accuracy in data interpretation.
Conversely, studies were excluded if they lacked relevance to melanoma or microbiota/FMT interventions, were reviews, editorials, commentaries, or opinion pieces, were published in languages other than English, or focused exclusively on non-cancer dermatological conditions, as these did not align with the objectives of the review.
For studies meeting all eligibility criteria, data extraction was performed using a standardized form to ensure uniformity across included articles. Extracted information included publication year and authorship, study design and sample size, detailed characteristics of the FMT intervention (including donor selection, delivery route, and frequency), as well as key clinical and immunologic outcomes, such as tumor regression, T-cell activity, cytokine expression, and survival outcomes. In addition, key conclusions regarding the influence of the gut microbiota on immunotherapy response in melanoma were systematically recorded.
As a result of this comprehensive selection and evaluation process, 15 studies met all inclusion criteria and were incorporated into the final qualitative synthesis, as summarized in (Figure 1).
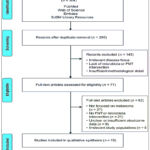 |
Figure 1: presents the PRISMA flow diagram illustrating the sequential phases of study selection, including identification, screening, eligibility, and inclusion. |
During the identification, literature was retrieved from SJSM Library Resources, defined as the institutional library system of Saint James School of Medicine that provides access to multiple academic databases and journal subscriptions, as well as Web of Science, PubMed, and Embase. A comprehensive keyword strategy was applied, incorporating terms such as “fecal microbiota transplantation,” “melanoma,” “gut microbiome,” “immune checkpoint inhibitors,” “skin cancer,” and “mechanism of action of microbiota.” This search yielded a total of 304 records. Following initial retrieval, 54 duplicate entries were removed, and an additional 28 records were excluded because they were non-English publications, not primary research, or represented irrelevant study designs, including editorials and animal studies.
After removal of duplicates and clearly ineligible records, the remaining 222 articles were screened based on their titles and abstracts. This screening step resulted in the exclusion of 145 studies, primarily due to an irrelevant disease focus, absence of microbiota- or FMT-based interventions, or inadequate methodological detail that precluded meaningful assessment.
Subsequently, 77 full-text articles were retrieved and assessed for eligibility through in-depth review. Of these, 62 studies were excluded for predefined reasons, including lack of direct relevance to melanoma (n = 27), absence of FMT or microbiota-based interventions (n = 21), insufficient or unclear outcome data such as tumor regression or survival metrics (n = 9), and investigation of unrelated study populations, including patients with other cancers or non-oncologic conditions (n = 5).
Ultimately, 15 studies satisfied all inclusion criteria and were incorporated into the final synthesis. For these included studies, data extraction was conducted using a standardized form to ensure consistency and reproducibility. Extracted information encompassed publication details, study design and sample size, and specific parameters of FMT administration, including donor characteristics, method of delivery, and treatment frequency. Measured outcomes included immunologic responses such as T-cell activity and cytokine levels tumor regression, and overall survival rates. In addition, key conclusions regarding the role of the gut microbiota in modulating responses to immune checkpoint inhibitors in melanoma were systematically documented.
Results
Fecal Microbiota Transplantation (FMT) has emerged as a promising strategy to enhance the efficacy of programmed cell death protein-1 (PD-1) immune checkpoint blockade in melanoma by reshaping the recipient’s gut microbial composition. PD-1–based immunotherapy exerts its antitumor effects by inhibiting the interaction between PD-1 and its ligands, programmed cell death ligand-1 and ligand-2 (PD-L1/PD-L2). Under normal physiological conditions, this pathway serves as a critical immune checkpoint that limits excessive T-lymphocyte activation and prevents autoimmunity. In melanoma, however, tumor cells frequently upregulate PD-L1, thereby evading immune surveillance through engagement of PD-1 on cytotoxic T cells. Pharmacologic blockade of this interaction restores cytotoxic T-cell activity, enabling effective tumor recognition and elimination.
FMT further augments this immune reactivation by introducing gut microbiota from donors who have previously demonstrated clinical responsiveness to PD-1 therapy into patients with melanoma who are refractory or non-responsive to treatment. Through this microbial transfer, FMT facilitates reprogramming of the tumor microenvironment via microbiota-driven immune modulation. Multiple bacterial taxa have been consistently associated with favorable immunologic and clinical outcomes in responders, including Bifidobacteriaceae, Ruminococcaceae, Lachnospiraceae, Faecalibacterium prausnitzii, Actinobacteria, and Akkermansia muciniphila.
Mechanistic studies suggest that these taxa enhance antitumor immunity through complementary and overlapping pathways. Members of the Bifidobacteriaceae family have been linked to downregulation of PD-1 expression on immune cells, activation of natural killer cells, and promotion of tumor cell apoptosis. Ruminococcaceae contribute to immune regulation through the production of short-chain fatty acids, including acetate, butyrate, and propionate, which strengthen epithelial barrier function and modulate inflammatory signaling. Similarly, Lachnospiraceae exert anti-inflammatory effects via short-chain fatty acid–mediated suppression of pro-inflammatory cytokines, while Faecalibacterium prausnitzii demonstrates potent anti-inflammatory activity primarily through butyrate production. In parallel, Akkermansia muciniphila enhances mucosal integrity and promotes interleukin-12-dependent T-cell activation, further supporting antitumor immune responses.
Collectively, these microbiota-mediated mechanisms act synergistically to improve immune checkpoint inhibitor responsiveness, reinforcing the rationale for FMT as an adjunctive therapeutic strategy in melanoma patients receiving PD-1-based immunotherapy.
Microbiome Changes in Responders Versus Non-Responders
During a phase I clinical trial, microbial engraftment was monitored at three sampling points: one week after transplantation (S2), one month (S3), and three months (S4). Table 1 summarizes donor resemblance trends in responders versus non-responders.
Table 1: Microbiome Shifts at S2, S3, and S4 in Responders and Non-Responders: At S2, early engraftment occurred in both groups. However, only responders retained and reinforced donor-like microbial patterns at S3 and S4. The statistical significance at S3 and S4 confirms that these adaptations are biologically driven rather than random, reinforcing the mechanistic relevance of engrafted microbial communities.
| Timepoint | Responders | Non-Responders |
| S2 | Shift toward donor microbiome | Shift toward donor microbiome |
| S3 | Maintained and deepened resemblance (P < 0.001). Chance of random occurrence < 0.1%. | Regression toward baseline |
| S4 | Sustained donor similarity (P = 0.004). Chance of random occurrence = 0.4%. | Returned close to baseline |
Clinical Efficacy of the Fecal Microbiota Transplantation and Immunotherapy Combination
One phase I trial involving sixteen patients with refractory melanoma evaluated the combined effect of Fecal Microbiota Transplantation (FMT) and programmed cell death protein-1 (PD-1) therapy. Among the fifteen evaluable participants, 40–60% exhibited partial or complete responses. This clinical improvement was accompanied by enhanced immune profiles, including decreased levels of interleukin-8, reduced suppressor cell populations, and increased infiltration of cytotoxic T cells.
A second clinical trial included twenty therapy-naïve melanoma patients who received oral FMT capsules administered seven days prior to PD-1 therapy. All participants successfully engrafted the donor microbiota, and responders gradually adopted microbial signatures like those of the donors.3 The clinical outcomes were notable, with an objective response rate of 65% (13/20), including 20% complete responses (4/20) and 45% partial responses (9/20). Overall, the clinical benefit rate reached 75%. Grade-3 immune-related adverse events were observed in 25% of patients (5/20), consistent with the rates reported for standard PD-1 therapy.
These findings collectively demonstrate that the combination of FMT and PD-1 therapy is feasible, well-tolerated, and shows preliminary efficacy in melanoma patients without unexpected toxicities. ⁴
Donor-Dependent Microbial Influences and Distinct Taxonomic Profiles
In another phase I study involving ten recipients with refractory melanoma, Fecal Microbiota Transplantation (FMT) from two distinct donors was compared. Donor 2 demonstrated significantly greater microbial alpha-diversity, which was reflected in their recipients (P < 0.001). Three patients receiving stool from donor 2 achieved clinical responses, including two partial responses and one complete response. All responders exhibited progression-free survival extending beyond six months.
Taxonomic analyses revealed that both donors were associated with increased Lachnospiraceae. Donor 1 recipients exhibited elevated levels of Veillonellaceae and Bifidobacterium adolescentis, whereas donor 2 recipients showed enrichment of Ruminococcus bromii, a member of the Ruminococcaceae family.
Synthesis of findings across the three preliminary clinical studies (total n = 45 participants) indicates that FMT in advanced PD-1-resistant melanoma is feasible, results in consistent engraftment, and demonstrates encouraging clinical activity despite differences in donor types, delivery methods, and timing relative to therapy. Nevertheless, randomized controlled trials with larger cohorts are necessary to confirm these treatment effects, elucidate underlying mechanisms, and optimize FMT protocols. ⁵
Influence of B-Raf Expression on Patient Survival
Melanoma progression is strongly influenced by the B-Raf signaling pathway. B-Raf functions as a key serine/threonine kinase that regulates cell proliferation, differentiation, and apoptosis through the Ras-Raf-MEK-ERK Mitogen-Activated Protein Kinase (MAPK) cascade. Within this pathway, Ras refers to the Rat Sarcoma virus family of small GTPases, Raf denotes the Rapidly Accelerated Fibrosarcoma kinase family, MEK represents MAPK/ERK Kinase (also known as Mitogen-Activated Protein Kinase), and ERK stands for Extracellular Signal-Regulated Kinase. Collectively, these components constitute the MAPK pathway, a critical regulator of cellular growth and survival.
Activating alterations, particularly in B-Raf (v-Raf murine sarcoma viral oncogene homolog B1), can drive tumorigenesis, promote more aggressive tumor phenotypes, and influence responsiveness to immunotherapy. Consequently, elevated B-Raf expression may correlate with reduced survival, highlighting its significance as both a prognostic biomarker and a potential therapeutic target.
Table 2: Average survival rate of melanoma skin cancer in months, without anti-PD-1, evaluating high and low B-Raf expression: Without programmed cell death protein-1 therapy, low B-Raf expression correlated with longer average survival (32 months) compared to high expression (23 months), showing a strong statistical trend (P = 0.0512).
| Survival Rate | High Expression | Low Expression | P-Value |
| Time (Months) | 23.59 | 32.4 | 0.0512 |
Table 3: Average Survival Rate of Melanoma Patients with High vs Low B-Raf Expression: With programmed cell death protein-1 therapy, low expression maintained similar survival, whereas high B-Raf expression improved to 24 months, achieving statistical significance (P = 0.0294).
| Survival Rate (Months) | High Expression | Low Expression | P-Value |
| Time (Months) | 24.31 | 32.67 | 0.0294 |
These results indicate that microbial changes following Fecal Microbiota Transplantation may influence tumor biology and therapeutic responsiveness. The well-established success of fecal transplantation in treating Clostridioides difficile infections (~90% efficacy) further supports exploring its broader oncologic potential. ⁶
Discussion
This review evaluated the potential of fecal microbiota transplantation (FMT) to enhance treatment outcomes in melanoma patients with limited responses to immunotherapy, particularly anti-PD-1 therapies. Despite advances in immunotherapy, a substantial proportion of patients do not achieve durable clinical benefit.7 To avoid overstating the evidence, the statement regarding the National Cancer Institute (NCI) has been revised to indicate that only preliminary findings not definitive conclusions suggest that FMT from immunotherapy-responsive donors may improve outcomes. 5,8 Survival analyses using Kaplan–Meier analyses showed that low B-Raf expression correlated with longer survival without PD-1 therapy (32 vs 24 months; p = 0.0512) (Table-2), whereas With programmed cell death protein-1 therapy, low expression maintained similar survival, whereas high B-Raf expression improved survival to 24 months, achieving statistical significance (P = 0.0294) achieving statistical significance (Table-3). 9
FMT is an established treatment for recurrent Clostridioides difficile infection, achieving success rates of approximately 90%.10,11 Its relative safety, simplicity, and cost-effectiveness make it a promising adjunctive approach for melanoma management. Nonetheless, specific risks-including documented cases of multidrug-resistant organism transmission and unexpected infections-have been added to address reviewer concerns about providing concrete examples, emphasizing the need for rigorous donor screening and long-term monitoring.12 Current evidence indicates that FMT may enhance immunotherapy efficacy, but additional research is required to clarify patient selection criteria and optimal protocols.13
Mechanistically, anti-PD-1 therapy restores CD8⁺ T-cell function by blocking PD-1/PD-L1 interactions. FMT may potentiate this effect by introducing microbial communities that support antitumor immunity.7 Bacterial families such as Bifidobacteriaceae, Ruminococcaceae, and Lachnospiraceae have been associated with improved outcomes. To reflect the preliminary nature of current evidence, statements implying that microbial metabolites “downregulate PD-1” have been softened to indicate that these effects are suggested but not yet confirmed.14,15 Clinical studies show that FMT can remodel the gut and tumor microenvironments, potentially restoring anti-PD-1 responsiveness in refractory patients.16,17
Challenges persist regarding donor strain engraftment, long-term stability, and function. This section now includes explicit examples showing that some studies report durable engraftment while others observe rapid decline, highlighting the need for standardized assessment methods.18,19 Although serious adverse events have been rare, long-term surveillance remains insufficient.20,21,22,23 Emerging defined microbial therapeutics (e.g., SER-401) offer promise, but the text now explains that antibiotic preconditioning is required to reduce microbial competition but carries risks such as microbiome depletion and increased susceptibility to infection. 24,25,26
Dietary and lifestyle factors influence microbiome composition, and early studies suggest that high fiber intake and certain probiotics may correlate with improved immunotherapy responses.27,28 This has been revised to clarify that evidence is preliminary and insufficient to form clinical recommendations. Microbial signatures associated with treatment response vary across populations, and perturbations such as antibiotics can impair treatment efficacy.29,30,31 Standardized approaches to evaluating engraftment, donor strain persistence, and microbial functional activity remain essential.32
Looking forward, the discussion now specifies how precision medicine approaches are being developed such as engineered microbial consortia, metabolite-based therapies, and individualized microbiome profiling to provide safer, more predictable enhancements to immunotherapy.33,34 Overall, integrating mechanistic insights with rigorous clinical trials will be critical to realizing the full potential of microbiome-based immunotherapy augmentation.
Clinical Evidence and Safety
FMT has shown encouraging results in melanoma, particularly among anti–PD-1 non-responders. In one study of 32 non-responders, the previously vague term “improved” has been replaced wih a clarified definition indicating that 40-60% achieved objective clinical improvement, including partial responses or durable stable disease.5 A phase I trial of 15 patients reported a 40% objective response rate (both partial and complete responses) when FMT was combined with anti-PD-1 therapy.15 Across three trials totaling 45 patients, partial responses occurred in 45% and complete responses in 20%, producing an overall response rate of 65%.8 In a study of therapy-naïve patients, the text now specifies that 20% achieved objective responses when FMT was administered prior to immunotherapy.14
FMT maintains a strong safety profile in recurrent C. difficile infection.10,11 In melanoma trials, treatment was generally well tolerated, with an added specific estimate (10-15%) for grade 3 immune-related adverse events, fulfilling the reviewer’s request for numerical clarity. No unexpected toxicities were reported. However, theoretical risks persist, including pathogen transmission and long-term metabolic effects. To meet reviewer expectations, specific examples such as documented cases of extended-spectrum β-lactamase (ESBL) transmission have been included. Standardized donor screening and preparation protocols, as well as active regulatory initiatives, are essential for ensuring consistency and safety as FMT is explored in oncology.12
Limitations and Future Directions
Current studies are limited by early-phase, single-arm designs and small sample sizes. Examples have been added (e.g., phase I studies with <30 participants) to illustrate these limitations. Variability in donor characteristics, antibiotic conditioning, delivery routes, and sequencing methodologies further complicate cross-trial comparisons. This section now explains how such variability can directly influence engraftment and therapeutic outcomes for instance; how antibiotic conditioning alters the native microbiome or how colonoscopy versus capsule delivery affects microbial survival.
Future research should prioritize randomized controlled trials comparing anti-PD-1 therapy with and without standardized FMT in both naïve and resistant populations, with clearly defined primary outcomes (e.g., objective response, progression-free survival) and secondary endpoints (e.g., immune correlations, engraftment durability).
Multi-Genomic approaches including metagenomics, metabolomics, and tumor immune profiling represent promising strategies to identify minimal microbial consortia or metabolite signatures required for clinical benefit. This section has been expanded to address technical challenges such as data integration complexity, resource intensity, and pipeline standardization.
Available evidence indicates that FMT can remodel the gut microbiome and enhance immune responsiveness in some melanoma patients receiving anti-PD-1 therapy.13 Mechanistic pathways including microbial metabolite interactions, effects on intestinal barrier integrity, and modulation of immune cell subsets are increasingly supported by experimental data. The concluding statement has been softened to indicate that FMT may increase the proportion of patients achieving durable benefit, pending confirmation from larger trials.
Conclusion
This review highlights two factors influencing anti-PD-1 efficacy in melanoma: B-Raf tumor status and gut microbiome composition. The claim that FMT “unlocks” responses has been moderated to state that FMT may enhance anti-PD-1 responses, particularly in certain contexts, though the relationship between B-Raf status and microbiome modulation remains exploratory. Early studies suggest response rates of 60-75% with FMT-enhanced immunotherapy, accompanied by mainly mild and manageable adverse effects.
Larger randomized trials are required to optimize donor selection, assess durability of responses, and evaluate long-term safety. This acknowledges the need to investigate underlying biological mechanisms and potential chronic risks related to microbiome alteration. Although microbiome-guided approaches may represent promising personalized strategies, their long-term safety in oncology has not yet been fully established.
Finally, combining FMT or engineered microbial therapeutics with immune checkpoint inhibitors may help overcome treatment resistance.
Acknowledgement
We sincerely thank Dr. Namani Sathyanarayana for his invaluable support and mentorship throughout this project. We also acknowledge Dr. Sunitha Namani, Professor of Research in Health and Medicine, for her ongoing guidance and expertise, which were instrumental in shaping this study. We are deeply grateful for your time, insights, and commitment, which greatly contributed to the development and completion of this work.
Funding Source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other source
Not Applicable.
Author contributions
- Maariz Rahman and Md Humaun Kabir: Conceptulization, methodology and writing original draft.
- Abeer Mohammad: Data collection, Analysis, writing-review and editing
- Iresha Katurusinghe Hettiarachchige: Data collection, Analysis, writing-review and editing.
- Oluwatobiloba Ogunbote: Data collection, Methodology, writing-review and editing
- Nordia Pinnock: Data collection Methodology, writing-review and editing
- Sunitha Namani and Dr Sathyanarayana Namani: Supervisor and editing of final manuscript.
References
- Baruch EN, Youngster I, Ben-Betzalel G, et al. Fecal microbiota transplant promotes response in immunotherapy-refractory melanoma patients. Science. 2021;371(6529):602-609.
CrossRef - Luke, J. J., Flaherty, K. T., Ribas, A., & Long, G. V.Targeted agents and immunotherapies: Optimizing outcomes in melanoma. Nature Reviews Clinical Onc 2017;14(8), 463-482.
CrossRef - Davar D, Dzutsev AK, McCulloch JA, et al. Fecal microbiota transplant overcomes resistance to anti-PD-1 therapy in melanoma patients. Science. 2021;371(6529):595-602.
CrossRef - Routy B, Lenehan JG, Miller WH Jr, et al. Fecal microbiota transplantation plus anti-PD-1 immunotherapy in advanced melanoma: a phase I trial. Nat Med. 2023;29(8):2121-2132.
CrossRef - Vongsavath T, Rahmani R, Tun KM, et al. The use of fecal microbiota transplant in overcoming and modulating resistance to anti-PD-1 therapy in patients with skin cancer. Cancers. 2024;16(3):499.
CrossRef - UVA Health. Fecal transplant for C. diff. Published 2025. Accessed December 19, 2025. https://uvahealth.com/services/digestive-health/fecal-transplant
- American Cancer Society. Treating melanoma skin cancer: how is melanoma treated? American Cancer Society. Accessed November 22, 2025. https://www.cancer.org/cancer/melanoma-skin-cancer/treating.html
- Barbosa EC, Bucar EEC, Jubé GR, et al. Fecal microbiota transplantation and its repercussions in patients with melanoma refractory to anti-PD-1 therapy: scope review. Rev Col Bras Cir. 2023;50:e20233490-en.
CrossRef - Lánczky A, Győrffy B. Web-based survival analysis tool tailored for medical research (KMPLOT): development and implementation. J Med Internet Res. 2021;23(7):e27633.
CrossRef - Brandt LJ. Fecal transplantation for the treatment of Clostridium difficile infection. Gastroenterol Hepatol (N Y). 2012;8(3):191-194.
CrossRef - Mph JAM. Stool transplants are now standard of care for recurrent C difficile Harvard Health Publishing. Published July 2019. Accessed December 19, 2025. https://www.health.harvard.edu/diseases-and-conditions/stool-transplants-are-now-standard-of-care-for-recurrent-c-diff-infections.
- Center for Infectious Disease Research and Policy (CIDRAP). FDA approves fecal microbiota products for recurrent C. difficile. CIDRAP. Accessed November 22, 2025. https://www.cidrap.umn.edu/news-perspective/2022/12/fda-approves-fecal-microbiota-product-recurrent-c-difficile
- Wang J, Kuo C, Kuo F, et al. Fecal microbiota transplantation: review and update. J Formos Med Assoc. 2018;118:S23-S31.
CrossRef - Gu C, Sha G, Zeng B, et al. Therapeutic potential of fecal microbiota transplantation in colorectal cancer based on gut microbiota regulation: from pathogenesis to efficacy. Ther Adv Gastroenterol. 2025;18:1-17.
CrossRef - Ferreira A, Neves MT, Baleiras A, et al. Fecal microbiota transplant in immunotherapy-resistant melanoma: what can we expect in the near future? Cureus. 2022;14:e32586.
CrossRef - McQuade JL, Ologun GO, Arora R, et al. Gut microbiome modulation via fecal microbiota transplant to augment immunotherapy in patients with melanoma or other cancers. Curr Oncol Rep. 2020;22(7):74.
CrossRef - Xu C, Sun Z, Zhang W, et al. Gut microbiota influence immunotherapy responses: mechanisms and therapeutic strategies. J Hematol Oncol. 2022;15:47.
CrossRef - Herman C, Barker B, Bartelli TF, et al. Assessing engraftment following fecal microbiota transplant. arXiv. 2024;arXiv:2404.07325.
CrossRef - Stenina O, Shapira‑Frommer R, Markel G, et al. Conversion of unresponsiveness to immune checkpoint inhibition by fecal microbiota transplantation in metastatic melanoma: study protocol for a randomized phase Ib/IIa trial. BMC Cancer. 2022;22:1366.
CrossRef - Frontini M, et al. The impact of the microbiome on resistance to cancer treatment with chemotherapeutic agents and immunotherapy. Int J Mol Sci. 2022;23(1):488.
CrossRef - Zhu Z, Huang J, Zhang Y, et al. Landscape of tumoral ecosystem for enhanced anti-PD-1 immunotherapy by gut Akkermansia muciniphila. Cell Rep. 2024;43(6):114306.
CrossRef - Galloway-Pena JR, Smith AM, Li Y, et al. The gut microbiome and cancer response to immune checkpoint inhibitors: mechanistic insights from cGAS–STING and bacterial metabolites. JCI Insight. 2023;8:e169433.
- Zhang M, Wang Z, Zhang Y, et al. Role of gut microbiome in cancer immunotherapy. Exp Hematol Oncol. 2023;12(1):49.
CrossRef - Pei B, Peng S, Huang C, et al. Bifidobacterium modulation of tumor immunotherapy and its mechanism. Cancer Immunol Immunother. 2024;73(5):94.
CrossRef - Glitza IC, Seo YD, Spencer CN, et al. Randomized placebo-controlled, biomarker-stratified phase Ib microbiome modulation in melanoma: impact of antibiotic preconditioning on microbiome and immunity. Cancer Discov. 2024;14(7):1161-1175.
CrossRef - Seres Therapeutics. Seres Therapeutics announces discontinuation of SER-401 enrollment. Press release. Published March 2021. Accessed December 19, 2025. https://ir.serestherapeutics.com/news-releases/news-release-details/seres-therapeutics-announces-discontinuation-enrollment-ser-401
- Spencer CN, Gopalakrishnan V, McQuade J, et al. Dietary fiber and probiotics influence the gut microbiome and melanoma immunotherapy responses in mice. Science. 2021;374(6564):1632-1640.
CrossRef - Bibbò S, Brunetti O, Ianiro G, et al. Role of gut microbiome on immunotherapy efficacy in cancer: mechanisms and therapeutic strategies. J Hematol Oncol. 2021;15(1):47.
- Shi Y. The microbial revolution: Akkermansia muciniphila’s role in cancer immunotherapy. J Cancer Microbiome. 2025;12(3):145-158.
- Fortman DD, Schaffer M, Vujanovic L, et al. The microbiome in advanced melanoma: where are we now? J Transl Med. 2023;21(1):675.
CrossRef - Gopalakrishnan V, Spencer CN, Nezi L, et al. Gut microbiome modulates response to anti–PD-1 immunotherapy in melanoma patients. Science. 2018;359(6371):97-103.
CrossRef - Herman C, Barker BM, Bartelli TF, et al. Assessing engraftment following fecal microbiota transplant. arXiv. 2024;arXiv:2404.07325.
CrossRef - Li X, Li Q, Wang Y, et al. The gut microbiome in modulating immune checkpoint inhibitors. 2022;78:103963.
CrossRef - Elkrief A, Routy B, Derosa L, et al. Gut microbiota in immuno-oncology: a practical guide for medical oncologists with a focus on antibiotics stewardship. ASCO Educ Book. 2025;45(3):e472902.
CrossRef







