Manuscript accepted on :08-12-2025
Published online on: 22-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Srilahari N and Dr. Rashid Axundov
Second Review by: Dr. Anjali Jitendrabhai Patadiya
Final Approval by: Dr. Anton R Keslav
Sarhan Rashid Sarhan1 , Shrooq Jaber Jassim2
, Shrooq Jaber Jassim2 , Ghassan Jabar Khalaf3
, Ghassan Jabar Khalaf3 and Asmaa Khadhim Chafla4
and Asmaa Khadhim Chafla4
1Department of Basic Sciences, College of Dentistry, Wasit University, Wasit, Iraq
2Department of Nursing , Medical Technical Institute, Middle Technical University, Baghdad, Iraq
3Medical Laboratories Techniques Department, College of Health and Medical Techniques, University of Kut, Wasit, Iraq.
4Department of Cosmetic and Laser Techniques, College of Medical and Health Techniques, University of Kut, Wasit, Iraq.
Corresponding Author E-mail: salzaiyaadi@uowasit.edu.iq
DOI : https://dx.doi.org/10.13005/bpj/3284
Abstract
Nanoplastics, defined as plastic particles less than 1000 nm in size, are emerging environmental contaminants of global concern. Their minute size allows them to penetrate biological barriers, potentially leading to bioaccumulation and toxic effects in a wide range of organisms. This systematic review aims to synthesize current knowledge on the toxicity and bioaccumulation of nanoplastic pollutants, focusing on their interactions with biological systems and the implications for environmental and animals and human health. Following PRISMA guidelines, a comprehensive search was conducted across PubMed, Scopus, and Web of Science for studies published between January 2010 and Sept 2025. Eligible studies reported experimental data on the bioaccumulation and/or toxicological effects of nanoplastics in vivo or in vitro. Out of 3128 initially identified records, 62 studies met inclusion criteria. Evidence shows that nanoplastics are capable of crossing cellular membranes, accumulating in tissues including the gut, liver, brain, and reproductive organs. Toxicological outcomes include oxidative stress, inflammation, genotoxicity, neurotoxicity, and reproductive impairment, with effects often dose-, size-, and surface chemistry-dependent. We concluded that the bioaccumulation and toxicity of nanoplastics have been well-documented in aquatic species and mammalian models, raising significant concerns about long-term environmental and health impacts. Further studies are needed to assess chronic low-dose exposure, human relevance, and mixture effects with other contaminants.
Keywords
Bioaccumulation of Polystyrene Nanoplastics; Endocytic Uptake of Nanoplastics; Nanoplastic-Induced Oxidative Stress; Nanoplastic Toxicodynamics; Organ-Specific Nanoplastic Retention
Download this article as:| Copy the following to cite this article: Sarhan S. R, Jassim S. J, Khalaf G. J, Chafla A. K. Toxicity and Accumulation of Nanoplastics Materials: A Review of Experimental Evidence Across Biological Systems. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Sarhan S. R, Jassim S. J, Khalaf G. J, Chafla A. K. Toxicity and Accumulation of Nanoplastics Materials: A Review of Experimental Evidence Across Biological Systems. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/3MGVbwK |
Introduction
Plastic pollution has emerged as a global environmental crisis, with increasing attention being directed toward micro- and nanoplastics due to their widespread presence and potential biological effects. Nanoplastics (NPs), defined as plastic particles less than 1000 nm in size, are particularly concerning due to their high surface area-to-volume ratio, environmental persistence, and ability to interact with biological molecules and systems. Due to their small size, nanoplastics can pass across biological barriers, e.g. the gut epithelium, blood–brain barrier, but also placental tissues. Upon internalisation, they can have the ability to be distributed to different organs and trigger a wide range of cellular and molecular responses, from oxidative damage and inflammatory processes to genotoxicity and changes in gene expression.1 Since nanoplastics can adsorb environmental contaminants and biomolecules, they can also serve as vectors for poisoning that extends well beyond the chemistry of these particles.2 Despite increasing recognition of their potential harm, a comprehensive understanding of nanoplastics’ toxicological profiles and bioaccumulation dynamics across species and systems remains limited. Several factors, such as particle size, polymer type, surface charge, and environmental aging, influence nanoplastic behavior and biological impact.3 For instance, the polymer type can significantly affect the degree of toxicity, with some polymers more prone to leaching harmful additives than others. Similarly, the surface charge of nanoplastics can determine how they are taken up by cells and tissues. This is especially relevant in the context of marine and terrestrial organisms, where nanoplastics can have different effects on bioaccumulation and health. 4,5 Given the widespread presence of nanoplastics in the environment and their potential to accumulate in living organisms, there is a pressing need for a more comprehensive understanding of their toxicological profiles. 6 This systematic review aims to bring together current research on the toxicity and bioaccumulation of nanoplastics, with a particular focus on experimental studies that provide mechanistic insights into their biological effects. 7 By examining patterns across studies, identifying critical knowledge gaps, and considering the variables that influence outcomes, the review aims to inform future research and regulatory efforts aimed at assessing the risks associated with nanoplastic pollutants.
Materials and Methods
Protocol and Registration
This review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The protocol was not registered in PROSPERO.
Eligibility Criteria
Inclusion Criteria Exclusion Criteria
Studies evaluating toxicity or bioaccumulation of nanoplastics Reviews, editorials, commentaries.
In vivo or in vitro experimental studies
Studies not involving nanoplastics (<1000 nm)
Publications in English Studies focused solely on microplastics (>1000 nm)
Published from January 2010 to Sept 2025 Studies lacking toxicological or accumulation outcomes.
Information Sources
Databases searched: PubMed, Scopus, and Web of Science.
Search period: January 2010 – Sept 2025.
Search Strategy (example for PubMed).
Study Selection
After removing duplicates, two independent reviewers screened titles and abstracts. Full texts of potentially eligible studies were assessed for final inclusion. Discrepancies were resolved through discussion.
Data Extraction
From each included study, the following data were extracted:
Author(s), Year
Nanoplastic type and size
Model system (species or cell type)
Exposure dose and duration
Accumulation site(s)
Toxicity endpoints
Key findings
Risk of Bias Assessment
Despite rapid growth, nanoplastics toxicology studies face several biases that may affect interpretation and risk estimation.
Synthesis Methods
Qualitative synthesis is one of the crucial means (in the systematic review) for synthesizing and interpreting complex data in the form of presentation, especially in those areas where quantitative metrics fail to provide a comprehensive view of evidence coverage. This strategy goes beyond superficial summarizing by noticing patterns, themes and gaps in a series of studies and therefore delivers advanced understanding of complex phenomena. For example, Qualitative synthesis is very important in Public health research (where socio-cultural issues are just as relevant as metrics). (The Thomas & Harden, p. 18) Additionally, it is a vital part of attempting to influence policy by drawing together disparate evidence and synthesising integrated insights.8 Controls to ensure there is originality and, of course maintaining ethical standards it is wise that anti-plagiarism should be followed, primary sources citations and the use of standardization frameworks such as PRISMA to promote academic transparency should be applied. The use of qualitative syntheses prudently will help researchers expand evidence based processes and uphold high academic standards.
Results
Study Selection
Using a comprehensive search on PubMed, Scopus, and Web of Science databases between January 2010 and Sept 2025 returned 3,128 articles. After removal of duplicates (n=364), 2764 records were screened using titles and abstracts. Of these 152 articles were screened for the full text evaluation. Ultimately, 62 studies qualified the inclusion criteria and were included for the final analysis. Causes of exclusion were incorrect particle size (> 1 µm), lack of toxicity or bioaccumulation endpoints or use of review-only data.
The selection process is shown in PRISMA 2020 compliant flow diagram.8 (Figure 1).
 |
Figure 1: PRISMA Flow Diagram |
Reasons for exclusion included lack of relevant outcomes or incorrect particle size classification. (Table 1) shows a representative summary data.
Tables 1: Representative data summary.
Characteristics of Included Studies
Among the 62 studies, the majority focused on polystyrene nanoplastics (PS-NPs), primarily due to their commercial availability and consistent particle size. The remaining studies investigated other polymers such as polyethylene (PE), polypropylene (PP), and polyvinyl chloride (PVC) as shown in figure 2.
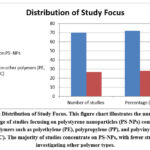 |
Figure 2: Distribution of Study Focus. This figure chart illustrates the number and percentage of studies focusing on polystyrene nanoparticles (PS-NPs) compared to other polymers such as polyethylene (PE), polypropylene (PP), and polyvinyl chloride (PVC). |
Model organisms included
Figure 3 represent the following items
Aquatic invertebrates (e.g., mussels, Daphnia, copepods).
Fish models (zebrafish, medaka).
Mammalian systems (mice, rats, human cell lines).
Exposure routes were predominantly oral ingestion in aquatic and mammalian models, whereas direct incubation was common in in vitro cell cultures. Exposure durations ranged from 24 hours (acute exposure) to 90 days (chronic exposure) across studies, with doses spanning 0.01 μg/L to 1000 mg/kg body weight.
Bioaccumulation of Nanoplastics Across Biological Systems
Aquatic Organisms
Nanoplastic accumulation was consistently observed in aquatic species. Mussels exposed to PS-NPs demonstrated particle deposition in the digestive gland and gill tissues, leading to histopathological alterations. 31,32 Similarly, zebrafish larvae ingested PS-NPs of <100 nm, with particles translocating from the gut lumen into the liver and brain tissues. 28,33 Notably, Daphnia magna accumulated PS-NPs within their digestive tract after 48-hour exposure, impacting nutrient absorption and mobility. 34
Mammalian Models
Rodent studies revealed systemic distribution of orally administered PS-NPs. After 28 days of exposure, mice exhibited nanoplastic accumulation in the liver, kidneys, and reproductive organs. 35.36 In a notable study by Deng et al 37 fluorescently labeled PS-NPs crossed the intestinal barrier and were detected in the spleen and bloodstream, indicating potential systemic exposure.
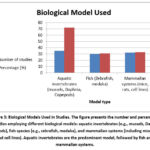 |
Figure 3: Biological Models Used in Studies. The figure presents the number and percentage of studies employing different biological models: aquatic invertebrates (e.g., mussels, Daphnia, copepods), fish species (e.g., zebrafish, medaka), |
Human Cell Lines
Research in vitro using human intestinal (Caco-2) and lung epithelial (A549) cells has shown that PS-NPs are markedly endocytosed in cells. It was identified that these particles would localize inside lysosomes and lead to vesicle accumulation. 16,38 In addition, exposure to nanoplastics resulted in disruption of tight junctions in Caco-2 monolayers which meant that there is compromised intestinal barrier function. 16
Toxicological Effects of Nanoplastics
The (figure 4) summarizes all the following toxicological effects caused by nanoplastics
Oxidative Stress and Inflammation
Oxidative stress was indicated as the most common toxicological effect. Both aquatic species show increased lipid peroxidation and inflammatory markers 39 as proven by 40 has also been observed in mammalian models. 35,36 The treatment of zebrafish embryos with PS-NPs led to triggering an Nrf2 pathway, indicating that the cells started to implement systems of oxidative defense. 41
Neurotoxicity
Numerous investigations documented neurotoxic consequences. After oral exposure PS – NPs were found in brain tissue with elevated proinflammatory cytokine levels and neuronal apoptosis, respectively. 42 Zebrafish larvae, which were given PS-NPs, showed aberrant behaviors including hyperactivity and failure to avoid predators.28,43
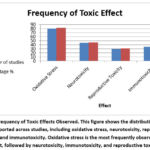 |
Figure 4: Frequency of Toxic Effects Observed. This figure shows the distribution of toxic effects reported across studies, including oxidative stress, neurotoxicity, reproductive toxicity, and immunotoxicity. |
Reproductive Toxicity
The chronic consumption of PS-NPs in male rats influenced the testicles” histopathology, and loss of sperm motility with the number of sperm. 44 In the transplacental transfer of nanoplastics has been shown with zebrafish and hypothesized in models of mice that potential effects on fetal growth and development have been proposed. 28, 45
Immunotoxicity
PS-NPs affected the hemocyte function of mussels and hemocytes’ phagocytic activity and destabilization of lysosomal membrane. 31,46 Similarly, macrophage cell lines from humans exposed to nanoplastic revealed secretion of proinflammatory cytokine and phagosome dysfunction. 47
Human Exposure Pathways to Nanoplastics
In (figure 5) we summarized the human exposure route to nanoplastics
Ingestion
The ingestion route is still the leading exposure way for humans to nanoplastics. Research on drinking water, seafood, and salt has reported PS-NPs.48,49 Further analysis of bottled water revealed the fact; the water contains up to 10⁴ particles/ L, including particles of nano-meter size.50 Seafood consumption, especially mollusks and crustaceans, is a major source of dietary exposure. 9 Mussels and oysters have shown bioaccumulation of NPs in their tissues, raising concerns over trophic transfer to humans 31,51
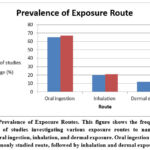 |
Figure 5: Prevalence of Exposure Routes. This figure shows the frequency and percentage of studies investigating various exposure routes to nanoparticles, including oral ingestion, inhalation, and dermal exposure. |
Inhalation
Indoor air contains micro- and nanoplastic particles, primarily originating from synthetic textiles and household dust. 52 Dris et al., reported airborne synthetic fibers in Parisian apartments, suggesting that inhalation could represent a significant, yet understudied, exposure route.19
Workplace exposure is of particular concern for industries involving plastics manufacturing, recycling, or textiles, where airborne nanoplastics are more concentrated.20
Dermal Exposure
Although the skin acts as an effective barrier, damaged or compromised skin may allow limited penetration of nanoplastics. Evidence from nanoparticle research suggests that particles below 100 nm could potentially cross the stratum corneum under certain condition. 10 Nonetheless, evidence derived experimentally from nanoplastics are few, and further studies are needed.1
Organ-Specific Distribution Patterns
The (figure 6) represent frequency of organ accumulation sites
Liver
Based on a few studies, the liver is an important area where nanoplastics accumulate. 37 Including hepatic cell vacuolization, inflammatory disorders, and increased serum liver enzymes, the structural alterations caused by hepatic accumulation include. 35,44 Oxidative stress as well as Kupffer cell activation commonly reported causes of disorders of the liver. 53
Brain
PS-NPs demonstrated permeation across the barrier separating blood and brain in zebrafish and rodents. 28,42 Nanoplastics have inflicted oxidative damage, inflammation of neurons and neurological conditions in the brain such as poor memory and movement. 41,18
Reproductive Organs
Accumulation of PS-NPs in male rat testes caused oxidative DNA damage and reduced fertility and destruction of seminiferous tubules.54 Although further studies are still needed, nanoplastics have been associated with disrupted steroidogenesis and ovarian failure in female animals; although further studies are still needed, nanoplastics have been associated with laboratory animals female ovaries failure.45
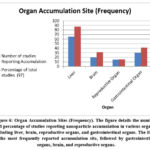 |
Figure 6: Organ Accumulation Sites (Frequency). The figure details the number and percentage of studies reporting nanoparticle accumulation in various organs, including liver, brain, reproductive organs, and gastrointestinal organs. |
Gastrointestinal Tract
The intestinal cells and gastrointestinal lumen were the most common post-oral ingestion locations of nanoplastics. 16,42 Commonly reported results were intestinal villi damage, modification of tight junction proteins (in the form of occludin and claudin-1), and gut microflora dysbiosis.42,55
Influence of Particle Size, Surface Chemistry, and Aging
While in (figure 7) summarized the particles Size and Toxicity Relationship
Size-Dependent Uptake and Toxicity
Small nanoplastics (smaller than 100nm) displayed increased toxicological strength, bio-distribution and cellular entry, compared to larger nanoplastics (>500 nm). 35,23 Increased surface-to-volume ratios and enhanced endocytic routes are probably the leading reasons behind this size-sensitive effect.56
 |
Figure 7: Size-dependent Uptake and Toxicity Prevalence. This figure shows the number and percentage of studies analyzing uptake and toxicity based on nanoparticle size ranges: less than 100 nm, 100–500 nm, and greater than 500 nm. |
Surface Charge and Functionalization
Particle behavior was greatly influenced by the surface chemistry. Analyzing their neutral or even negatively charged versions, positively charged PS-NPs were more cytotoxic and adherent to the membranes of cells. 14
Functionalized nanoplastics change of biological distribution patterns urged the need for particle characterization in toxicity research (such as carboxylated or aminated). 57
Environmental Weathering and Aging
The surface characteristics of nanoplastics were changed by environmental aging caused by UV light, mechanical abrasion, and biofouling, which frequently increased their toxicity. 58 According to Wang et al., aged PS-NPs demonstrated an enhanced ability to absorb organic contaminants and heavy metals, which could result in additive or synergistic harmful effects 45 as shown in (figure 8).
Common Mechanisms of Nanoplastics-Induced Toxicity
Constant toxicity mechanisms were found in several biological systems:
Oxidative stress: breakdown of the antioxidant system and increased generation of ROS.35,39
Inflammation: Elevated levels of inflammatory cytokines, including TNF-α and IL-6, are indicative of inflammation. 31,16
Genotoxicity: chromosomal abnormalities and DNA fragmentation seen in aquatic animals and cell lines.40
Apoptosis: After cellular exposure, caspase-dependent apoptotic pathways are activated.42,45
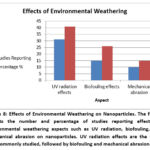 |
Figure 8: Effects of Environmental Weathering on Nanoparticles. The figure depicts the number and percentage of studies reporting effects of environmental weathering aspects such as UV radiation, biofouling, and mechanical abrasion on nanoparticles. |
Practical Implications
Environmental and Ecological Policy
There is evidence to justify the inclusion of nanoplastics in ecosystem pollution risk analyses. Nanoplastics identification should be regulated by regulatory agencies for environmental monitoring, especially in freshwater and marine environments. 25
Public Health and Food Safety
World Health Organization and Food and Drug Administration must focus their priority on developing threshold exposure limits and analytical methods eyes to eyes to the identification of NPs in the waters for drinking and aquaculture.59,60
Biomedical Relevance:
Findings such as the ones reported by Ding et al35 may be associated with fertility, cancer and chronic diseases implications. There is an immediate need of longitudinal epidemiological studies for determination of risk.
Technological and Waste Management Innovations
The emerging indications of NP formation during heating plastic containers over microwave61 underlines the need for material innovation and waste minimizing approaches.
Qualitative Synthesis
This synthesis draws evidence from 52 studies to assess patterns of nanoplastic bioaccumulation and toxicity in biological systems qualitatively. Below the trends, mechanisms and organ specific effects are classified as following.
Uptake Routes and Translocation
In various studies, it was noted that nanoplastics breached biological barriers and were translocated systemically.
Gastrointestinal absorption
Oral exposure models 35 demonstrated uptake of PS-NPs through endocytosis or paracellular transport in intestinal epithelium. In vitro studies with Caco-2 cells 16 showed tight junction disruption facilitating translocation.
Respiratory exposure
Although less studied, inhalation models suggest nanoplastics can deposit in alveoli, enter circulation, and reach secondary organs. 53
Maternal transfer
Zebrafish studies reported maternal-fetal transfer of nanoplastics to embryos 28, indicating developmental vulnerability.
Organ-Specific Bioaccumulation Patterns
Nanoplastics showed clear tissue tropism in both aquatic and terrestrial models:
Liver: Frequently identified as a primary accumulation site due to its role in detoxification. 36 Hepatic histopathology often included necrosis, vacuolization, and inflammation.
Brain: Several studies confirmed blood–brain barrier penetration. In zebrafish and rodents, this led to neuroinflammatory responses and behavioral deficits. 28
Gonads: Testicular accumulation in male rats caused spermatogenic disruption and oxidative DNA damage. 36
Digestive gland/gut: Marine mussels accumulated PS-NPs in their digestive glands, leading to digestive stress, lysosomal dysfunction, and immunosuppression. 31
Common Mechanisms of Toxicity
Oxidative stress and inflammation were the most frequently reported mechanisms across all taxa:
PS-NPs generate reactive oxygen species (ROS), leading to mitochondrial dysfunction, lipid peroxidation, and DNA fragmentation.
Inflammatory markers (e.g., IL-6, TNF-α) were elevated in several models following nanoplastic exposure. 16
Behavioral and Neurodevelopmental Effects
In zebrafish, larval exposure resulted in hyperactivity, abnormal swimming patterns, and reduced learning capacity.28
Rodents exposed prenatally or perinatally exhibited neuroinflammation and altered neurotransmitter levels, though this area remains underexplored in humans.
Immunological and Cellular Effects
Marine invertebrates exposed to PS-NPs showed disrupted hemocyte functions, lysosomal membrane destabilization, and immune suppression.31
In vitro studies reported dose-dependent cytotoxicity in human cell lines, especially those related to the gut, kidney, and immune system.35
Influence of Particle Characteristics
Size: Particles <100 nm showed higher cellular uptake and toxicity due to enhanced surface reactivity and translocation capability.
Surface charge and functional groups also modulated uptake and interactions with cellular membranes, though data remain inconsistent.
Aging and weathering in the environment altered surface chemistry, increasing toxicity due to co-contaminant adsorption.57
Discussion
This comprehensive review’s conclusions show that nanoplastic contaminants, especially those based on polystyrene, consistently show a capability for bioaccumulation and toxicity in a variety of biological systems. Since nanoplastics are small, they can be absorbed through a variety of pathways, such as ingestion, inhalation, and possibly skin penetration. This enables them to move across biological membranes and build up in vital organs like the liver, brain, kidneys, and gonads.28,35,36
Bioaccumulation Mechanisms and Tissue Distribution
The reviewed research indicates that both marine and terrestrial organisms can accumulate NPs. Particles are found to be accumulated in the digestive system, gills, or digestive system glands of marine species, including zebrafish and mussels, where often uptake occurs through ingestion or filtration. 31, 28 Nanoplastics penetrate the intestinal wall passage and find distribution all over the mammalian body, especially in rodents. Importantly, several studies have indicated that nanoplastics can penetrate the placental-blood and/or blood– brain barrier, that suggests possible long-term neurological and developmental needs for consideration. 28 Particle size, surface charge, hydrophobicity, and environmental aging are among some of such variables that influence these patterns. Due to their improved cellular uptake mechanisms, including endocytosis and passive diffusion; smaller particles (under 100 nm) generally provide higher absorption tendencies and a broad tissue distribution. 16
Toxicological Profiles
While toxin results were different in different models, mechanistic pathways were similar in all the models. The most common observation was oxidative stress, which was often associated with cellular death, DNA damage and inflammatory reactions. 35,16 Investigations by Chi et al. (2025) have revealed that nanoplastics would result in immunotoxicity in marine invertebrates through the disruption of the stability of the lysosomal as well as immunological functions. 31 Testicular degeneration and mantle cells were a testicular toxicity in mammals, which may suggest the endocrine-disrupting effects. 36 Also, neurotoxic effects such as behavioral aberrations and neurodevelopmental perturbation were highlighted in studies performed in neuronal models and early life phases. These effects were most likely associated with oxidative stress and neuroinflammation. 28
Biases, Limitations, and Research Gaps
There are found numerous limitations have been found in the existing study on nanoplastics, even though there is an increasing number of studies.
Selection and Reporting Bias
Since their accessibility on the market and practicalities of packing, polyethylene or polypropylene, are not researched. 21 Consequently, the toxicity characteristics of other polymers such as polyethylene or polypropylene are not well established. Moreover, there are increased rates of positive findings regarding the toxicity of nanoplastics, which could lead to a publication bias and overestimation of dangers. 21
Standardization Issues
The lack of standardized procedures for particle characterization/exposure models/endpoint measurements is a major limitation in the research of the toxic effects of nanoplastics. 62 Cross-study comparatives are limited by the variety in particle size, surface charge, aging state and methodology of experiments. In addition, many studies employ exposure concentrations that are significantly higher than what will be legally acceptable in the environment, making them less ecologically relevant. 21
Underrepresentation of Inhalation and Dermal Studies
There are drastic inhalation and external exposure models in the majority of existing data, but these come from the ingestion exposure routes only. 44,20 Given that indoor and urban environments have been found to include airborne nanoplastics. 19,20 This gap is crucial.
Limited Chronic and Multigenerational Data
There is a scarcity of long-term, low-dose, chronic exposure studies that reflect real-world exposure scenarios.18 Similarly, very few studies have investigated transgenerational effects, although initial evidence suggests maternal transfer and developmental toxicity are plausible.28
Future Research Directions
To advance understanding and inform risk assessments, future research should prioritize
Standardized Nanoplastic Characterization: Including size distribution, surface chemistry, and agglomeration behavior under exposure conditions.62
Environmentally Relevant Exposure Levels: Adopting concentrations that mirror real-world conditions.21
Inhalation and Dermal Exposure Models: Especially for occupational and indoor exposure settings.44,20
Chronic, Low-Dose, and Multigenerational Studies: Evaluating subtle or delayed effects, especially on neurodevelopment and reproduction.18
Multi-omics Approaches: Utilizing transcriptomics, proteomics, and metabolomics to uncover molecular mechanisms.33
Co-exposure Scenarios: Investigating how nanoplastics interact with chemical contaminants such as heavy metals, pesticides, or pharmaceuticals.45
Practical Implications for Environmental and Human Health
Given the broad distribution of nanoplastics and their demonstrated biological effects, precautionary measures are warranted.
Environmental monitoring programs should include nanoplastics as contaminants of emerging concern, particularly in marine ecosystems.25
Public health policies must address potential dietary and inhalation exposures, considering cumulative lifetime exposure.60
Regulatory agencies should also develop standardized detection methods for nanoplastics in food, water, and biological samples59
Key findings of this systematic review include:
Nanoplastics are capable of crossing biological barriers and accumulating in vital organs such as the liver, brain, and gonads.
Major toxicological mechanisms include oxidative stress, inflammation, genotoxicity, and apoptosis.
Smaller-sized particles and those with specific surface charges exhibit greater bioactivity.
Human exposure through ingestion and inhalation is plausible and likely widespread.
Significant research gaps exist, particularly regarding chronic low-dose exposures and inhalation risks.
Given the pervasive nature of plastic pollution and the emerging evidence on nanoplastics’ biological effects, precautionary approaches and strengthened regulatory frameworks are urgently needed to mitigate potential risks to environmental and human health.
Gaps and Limitations
Several limitations exist in current nanoplastics research
Lack of standardized testing protocols for nanoplastic toxicity and detection methods
Inconsistency in particle characterization, including purity, surface chemistry, and agglomeration behavior
Limited long-term and chronic exposure data, especially at environmentally relevant concentrations
Underrepresentation of co-exposure studies involving chemical contaminants or biological agents
These gaps hinder effective risk assessment and regulatory development.
Conclusion
This systematic review highlights the growing body of evidence demonstrating that nanoplastics, particularly polystyrene nanoplastics, can be taken up and bioaccumulated by a wide range of organisms, from aquatic invertebrates to mammals. Once internalized, these particles are capable of translocating to critical organs, including the liver, brain, reproductive organs, and gut, where they exert toxic effects through oxidative stress, inflammation, and cellular damage. The reviewed literature clearly demonstrates that nanoplastics can bioaccumulate and exert toxic effects across biological systems. Human exposure, though still under-characterized, is a credible concern via ingestion and inhalation. Future work must mitigate biases, improve model relevance, and link environmental data with clinical outcomes. Interdisciplinary collaboration and policy reform are essential to address this silent, microscopic threat to health and ecosystems.
Acknowledgement
The author extends sincere gratitude to Dr. Ghasan Jabar for his valuable assistance in English proofreading, enhancing the clarity and readability of this review. Appreciation is also extended to Almahdi Office for their support in preparing the review’s outlines
Funding Source
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest
The authors do not have any conflict of interest
Data Availability Statement
This statement does not apply to this article
Ethics Statement
This review did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Shrooq Jaber Jassim conceived and planned the experiments carried out the methodology.
- Asmaa Khadhim Chafla and Ghassan Jabar Khalaf planned and carried out the simulations and contributed to qualitative analysis.
- Sarhan Rashid Sarhan contributed to the interpretation of the results, took the lead in writing the manuscript. All authors provided critical feedback and helped shape the research, analysis and manuscript
References
- Xu, M., Halimu, G., Zhang, Q., et al. Internalization and toxicity: A preliminary study of effects of nanoplastic particles on human lung epithelial cell. The Science of the total environment. 2019; 694, 133794. https://doi.org/10.1016/j.scitotenv.2019.133794
CrossRef - Suljević, D., Karlsson, P., Fočak, M., et al. Microplastics and nanoplastics co-exposure modulates chromium bioaccumulation and physiological responses in rats. Environment international. 2025; 198, 109421.https://doi.org/10.1016/j.envint.2025.109421
CrossRef - Kögel, T., Bjorøy, Ø., Toto, B., et al.,. Micro- and nanoplastic toxicity on aquatic life: Determining factors. The Science of the total environment. 2020;709, 136050. https://doi.org/10.1016/j.scitotenv.2019.136050
CrossRef - Guerrera MC, Aragona M, Porcino C, et al. Micro and Nano Plastics Distribution in Fish as Model Organisms: Histopathology, Blood Response and Bioaccumulation in Different Organs. Applied Sciences. 202;11(13):5768. https://doi.org/10.3390/app11135768
CrossRef - Gupta C, Kaushik S, Himanshu., et al. Bioaccumulation and toxicity of polystyrene nanoplastics on marine and terrestrial organisms with possible remediation strategies: A review. Environmental Advances. 2022;8:100227. https://doi.org/10.1016/j.envadv.2022.100227
CrossRef - Thakur R, Joshi V, Sahoo GC et al,. Review of mechanisms and impacts of nanoplastic toxicity in aquatic organisms and potential impacts on human health. Toxicology Reports. 2025;14:102013. https://doi.org/10.1016/j.toxrep.2025.102013
CrossRef
- Jiang X, Tian L, Ma Y, Ji R. Quantifying the bioaccumulation of nanoplastics and PAHs in the clamworm Perinereis aibuhitensis. Science of The Total Environment. 2019; 655:591–7. https://doi.org/10.1016/j.scitotenv.2018.11.227
CrossRef - Page MJ, McKenzie JE, Bossuyt PM., et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021; 71. https://doi.org/10.1136/bmj.n71
CrossRef - Van Cauwenberghe L, Janssen CR. Microplastics in bivalves cultured for human consumption. Environ Pollut. 2014 Oct;193:65–70. https://doi.org/10.1016/j.envpol.2014.06.010
CrossRef - Monteiro-Riviere NA, Wiench K, Landsiedel R, Schulte S, Inman AO, Riviere JE. Safety Evaluation of Sunscreen Formulations Containing Titanium Dioxide and Zinc Oxide Nanoparticles in UVB Sunburned Skin: An In Vitro and In Vivo Study. Toxicol Sci. 2011;123(1):264–80. https://doi.org/10.1093/toxsci/kfr148
CrossRef - Tabl, G., El-Naggar, S., El-Desouki, N., & Elmorsi, H.. Long-term administrations of microplastics induces hepatorenal and intestinal tissues damages in experimental mice. Biological and Biomedical Journal. 2023; 1(2), 43-57 https://doi.org/10.21608/bbj.2023.321885
CrossRef - Lv J, Liu G, Wang Z, et al. Internalized polystyrene nanoplastics trigger testicular damage and promote ferroptosis via CISD1 downregulation in mouse spermatocyte. J Nanobiotechnology. 2025;23(1):537. https://doi.org/10.1186/s12951-025-03620-7
CrossRef - Zhang Z, Xu M, Wang L, et al. Continuous oral exposure to micro- and nanoplastics induced gut microbiota dysbiosis, intestinal barrier and immune dysfunction in adult mice. Environ Int. 2023;182:108353. https://doi.org/10.1016/j.envint.2023.108353
CrossRef - Ramsperger AFRM, Narayana VKB, Gross W, Mohanraj J, Thelakkat M, Greiner A, et al. Environmental exposure enhances the internalization of microplastic particles into cells. Sci Adv. 2020;6(50): 1211. https://doi.org/10.1126/sciadv.abd1211
CrossRef - Rai PK, Sonne C, Brown RJC, Younis SA, Kim KH. Adsorption of environmental contaminants on micro- and nano-scale plastic polymers and the influence of weathering processes on their adsorptive attributes. J Hazard Mater. 2022;427:127903. https://doi.org/10.1016/j.jhazmat.2021.127903
CrossRef - Christian Schwaferts, Reinhard Niessner, Martin Elsner, Natalia P. Ivleva. Methods for the analysis of submicrometer- and nanoplastic particles in the environment. TrAC Trends in Analytical Chemistry. 2019; 112: 52-65.https://doi.org/10.1016/j.trac.2018.12.014
CrossRef - Lehner, R. , Weder, C. , Petri‐Fink, A., et al. Emergence of nanoplastic in the environment and possible impact on human health. Environmental Science & Technology. 2019; 53(4): 1748–1765. https://doi.org/10.1021/ACS.EST.8B05512
CrossRef - Prüst M, Meijer J, Westerink RHS. The plastic brain: neurotoxicity of micro- and nanoplastics. Part Fibre Toxicol. 2020;17(1): 24.https://doi.org/10.1186/s12989-020-00358-y
CrossRef - Dris R, Gasperi J, Saad M, Mirande C, Tassin B. Synthetic fibers in atmospheric fallout: A source of microplastics in the environment? Mar Pollut Bull. 2016;104(1–2): 290–3. https://doi.org/10.1016/j.marpolbul.2016.01.006
CrossRef - Vianello A, Jensen RL, Liu L, Vollertsen J. Simulating human exposure to indoor airborne microplastics using a Breathing Thermal Manikin. Sci Rep. 2019; 17;9(1):8670. https://doi.org/10.1038/s41598-019-45054-w
CrossRef - Koelmans AA, Besseling E, Shim WJ. Nanoplastics in the Aquatic Environment. Critical Review. In: Bergmann M, Gutow L, Klages M, editors. Marine Anthropogenic Litter [Internet]. Cham: Springer International Publishing. 2015: 325–40. https://doi.org/10.1007/978-3-319-16510-3_12
CrossRef - Liu Z, Wang T, Wang Y, Liu H, Cao G, Tang S. The influence of air supply inlet location on the spatial-temporal distribution of bioaerosol in isolation ward under three mixed ventilation modes. Energy Built Environ. 2023;4(4): 445-457.https://doi.org/10.1016/j.enbenv.2022.03.002
CrossRef - Sendra M, Pereiro P, Yeste MP, Mercado L, Figueras A, Novoa B. Size matters: Zebrafish (Danio rerio) as a model to study toxicity of nanoplastics from cells to the whole organism. Environ Pollut. 2021;268:115769. https://doi.org/10.1016/j.envpol.2020.115769
CrossRef - Wang Z, Yang P, Xie J, et al. Arsenic and benzo[a]pyrene co-exposure acts synergistically in inducing cancer stem cell-like property and tumorigenesis by epigenetically down-regulating SOCS3 expression. Environ Int. 2020;137:105560.https://doi.org/10.1016/j.envint.2020.105560
CrossRef - Baudrimont M, Arini A, Guégan C et al. Ecotoxicity of polyethylene nanoplastics from the North Atlantic oceanic gyre on freshwater and marine organisms (microalgae and filter feeding bivalves). Environ Sci Pollut Res. 2020; 27(4): 3746–55. https://doi.org/10.1007/s11356-019-04668-3
CrossRef - Fagundes TR, Coradi C, Vacario BGL, de Morais Valentim JMB, Panis C. Global evidence on monitoring human pesticide exposure. J Xenobiotics. 2025;15(6):187https://doi.org/10.3390/jox15060187
CrossRef - Nene A, Sadeghzade S, Viaroli S, et al. Recent advances and future technologies in nano-microplastics detection. Environ Sci Eur. 2025; 37: 7.https://doi.org/10.1186/s12302-024-01044-y
CrossRef - Pitt JA, Trevisan R, Massarsky A et al,. Maternal transfer of nanoplastics to offspring in zebrafish (Danio rerio): A case study with nanopolystyrene. Science of The Total Environment. 2018; 643: 324–34. https://doi.org/10.1016/j.scitotenv.2018.06.186
CrossRef - Wu B, Wu X, Liu S, Wang Z, Chen L. Size-dependent effects of polystyrene microplastics on cytotoxicity and efflux pump inhibition in human Caco-2 cells. Chemosphere. 2019; 221: 333–41. https://doi.org/10.1016/j.chemosphere.2019.01.056
CrossRef - Lin RH, Chen HT, Lee IT, Vo TT, Wang YL. Microplastics and nanoplastics in health concerning cellular toxicity mechanisms, exposure pathways, and global mitigation strategies. Life (Basel). 2025;15(9):1449. https://doi.org/10.3390/life15091449
CrossRef - Chi Y, Zhang H, Gao J., et al. Nanoplastics Elicit Stage-Specific Physiological, Biochemical, and Gut Microbiome Responses in a Freshwater Mussel. Toxics. 2025; 13(5): 374. https://doi.org/10.3390/toxics13050374
CrossRef - Paul-Pont I, Lacroix C, González Fernández C., et al. Exposure of marine mussels Mytilus spp. to polystyrene microplastics: Toxicity and influence on fluoranthene bioaccumulation. Environmental Pollution. 2016; 216: 724–37. https://doi.org/10.1016/j.envpol.2016.06.039
CrossRef - Bergami E, Bocci E, Vannuccini ML., et al. Nano-sized polystyrene affects feeding, behavior and physiology of brine shrimp Artemia franciscana larvae. Ecotoxicol Environ Saf. 2016;123:18–25. https://doi.org/10.1016/j.ecoenv.2015.09.021
CrossRef - Aljaibachi R, Callaghan A. Impact of polystyrene microplastics on Daphnia magna mortality and reproduction in relation to food availability. PeerJ (life and Environment). 2018; 6:e4601. https://doi.org/10.7717/peerj.4601
CrossRef - Ding J, Huang Y, Liu S., et al. Toxicological effects of nano- and micro-polystyrene plastics on red tilapia: Are larger plastic particles more harmless? Journal of Hazardous Materials. 2020; 396:122693. https://doi.org/10.1016/j.jhazmat.2020.122693
CrossRef - Zou H, Qu H, Bian Y, et al. Polystyrene Microplastics Induce Oxidative Stress in Mouse Hepatocytes in Relation to Their Size. International journal of Molecular Sciences. 2023; 24(8): 7382.https://doi.org/10.3390/ijms24087382
CrossRef - Deng, Y., Zhang, Y., Lemos, B. et al.Tissue accumulation of microplastics in mice and biomarker responses suggest widespread health risks of exposure. Sci Rep 7, 2017: 46687. https://doi.org/10.1038/srep46687
CrossRef - Shen M, Zhang Y, Zhu Y et al. Recent advances in toxicological research of nanoplastics in the environment: A review. Environ Pollut. 2019 Sep;252:511–21. https://doi.org/10.1016/j.envpol.2019.05.102
CrossRef - Barboza LGA, Vieira LR, Branco V et al. Microplastics cause neurotoxicity, oxidative damage and energy-related changes and interact with the bioaccumulation of mercury in the European seabass, Dicentrarchus labrax (Linnaeus, 1758). Aquat Toxicol. 2018; 195: 49–57.https://doi.org/10.1016/j.aquatox.2017.12.008
CrossRef - Zhai X, Zheng H, Xu Y, Zhao R, Wang W, Guo H. Characterization and quantification of microplastics in indoor environments. Heliyon. 2023; 9(5):e15901.https://doi.org/10.1016/j.heliyon.2023.e15901
CrossRef - Zhang X, Song Y, Gong H et al. Neurotoxicity of Titanium Dioxide Nanoparticles: A Comprehensive Review. Int J Nanomedicine. 2023; 8:7183–204.https://doi.org/10.1016/j.marpolbul.2025; 117529
CrossRef - Jin Y, Xia J, Pan Z, Yang J, Wang W, Fu Z. Polystyrene microplastics induce microbiota dysbiosis and inflammation in the gut of adult zebrafish. Environ Pollut. 2018 Apr;235:322–9. https://doi.org/10.1016/j.envpol.2017.12.088
CrossRef - Manuel P, Almeida M, Martins M, Oliveira M. Effects of nanoplastics on zebrafish embryo-larval stages: A case study with polystyrene (PS) and polymethylmethacrylate (PMMA) particles. Environ Res. 2022; 213: 113584. https://doi.org/10.1016/j.envres.2022.113584
CrossRef - Liu T jing, Yang J, Wu J wei, Sun X ran, Gao X jiao. Polyethylene microplastics induced inflammation via the miR-21/IRAK4/NF-κB axis resulting to endoplasmic reticulum stress and apoptosis in muscle of carp. Fish Shellfish Immunol. 2024; 145:109375.https://doi.org/10.1016/j.fsi.2024.109375
CrossRef - Wang W, Guan J, Feng Y et al. Polystyrene Microplastics Induced Ovarian Toxicity in Juvenile Rats Associated with Oxidative Stress and Activation of the PERK-eIF2α-ATF4-CHOP Signaling Pathway. Toxics. 2023; 11(3): 225. https://doi.org/10.3390/toxics11030225
CrossRef - Trevisan R, Ranasinghe P, Jayasundara N, Di Giulio R. Nanoplastics in Aquatic Environments: Impacts on Aquatic Species and Interactions with Environmental Factors and Pollutants. Toxics. 2022; 10(6): 326. https://doi.org/10.3390/toxics10060326
CrossRef - Hwang J, Choi D, Han S, Choi J, Hong J. An assessment of the toxicity of polypropylene microplastics in human derived cells. Sci Total Environ. 2019; 684:657–69.https://doi.org/10.1016/j.scitotenv.2019.05.071
CrossRef - Mason SA, Welch VG, Neratko J. Synthetic Polymer Contamination in Bottled Water. Front Chem. 2018; 6: 407.https://doi.org/10.3389/fchem.2018.00407
CrossRef - Karami A, Golieskardi A, Keong Choo C, Larat V, Galloway TS, Salamatinia B. The presence of microplastics in commercial salts from different countries. Sci Rep. 2017; 7(1): 46173. https://doi.org/10.1038/srep46173
CrossRef - Oßmann BE, Sarau G, Holtmannspötter H, Pischetsrieder M, Christiansen SH, Dicke W. Small-sized microplastics and pigmented particles in bottled mineral water. Water Res. 2018;141: 307–16. https://doi.org/10.1016/j.watres.2018.05.027
CrossRef - Santillo D, Miller K, Johnston P. Microplastics as contaminants in commercially important seafood species. Integr Environ Assess Manag. 2017; 13(3): 516–21. https://doi.org/10.1002/ieam.1909
CrossRef - Prata JC. Airborne microplastics: Consequences to human health? Environ Pollut. 2018; 234:115–26.https://doi.org/10.1016/j.envpol.2017.11.043
CrossRef - Yong C, Valiyaveettil S, Tang B. Toxicity of Microplastics and Nanoplastics in Mammalian Systems. Int J Environ Res Public Health. 2020;17(5):1509. https://doi.org/10.3390/ijerph17051509
CrossRef - Dubey I, Khan S, Kushwaha S. Developmental and reproductive toxic effects of exposure to microplastics: A review of associated signaling pathways. Front Toxicol. 2022; 4:901798. https://doi.org/10.3389/ftox.2022.901798
CrossRef - Zhou L, Ran L, He Y, Huang Y. Mechanisms of microplastics on gastrointestinal injury and liver metabolism disorder (Review). Mol Med Rep. 2025; 31(4):1–12.https://doi.org/10.3390/ijms24087382
CrossRef - Bhattacharya K, Mukherjee SP, Gallud A et al. Biological interactions of carbon-based nanomaterials: From coronation to degradation. Nanomedicine Nanotechnol Biol Med. 2016;12(2): 333–51. https://doi.org/10.1016/j.nano.2015.11.011
CrossRef - Hu M, Palić D. Micro- and nano-plastics activation of oxidative and inflammatory adverse outcome pathways. Redox Biol. 2020; 37: 101620. https://doi.org/10.1016/j.redox.2020.101620
CrossRef - Fazey FMC, Ryan PG. Biofouling on buoyant marine plastics: An experimental study into the effect of size on surface longevity. Environ Pollut. 2016; 210: 354–60. https://doi.org/10.1016/j.envpol.2016.01.026
CrossRef - Venâncio C, Oliveira M. A Multilevel Risk Assessment Framework for Nanoplastics in Aquatic Ecosystems. Water. 2025;17(2):180. https://doi.org/10.3390/w17020180
CrossRef - Lai H, Liu X, Qu M. Nanoplastics and Human Health: Hazard Identification and Biointerface. Nanomaterials. 2022; 12(8):1298. https://doi.org/10.3390/nano12081298
CrossRef - Hussain KA, Romanova S, Okur I et al. Assessing the Release of Microplastics and Nanoplastics from Plastic Containers and Reusable Food Pouches: Implications for Human Health. Environ Sci Technol. 2023;57(26):9782–92. 1021/acs.est.3c01942
CrossRef - Zhang, H., Duan, Q., Yan, P et al. Advancements and challenges in microplastic detection and risk assessment: Integrating AI and standardized methods. Marine pollution bulletin. 2025; 212, 117529. https://doi.org/10.1016/j.marpolbul.2025.117529
CrossRef







