Manuscript accepted on :04-12-2025
Published online on: 15-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Ramdas Bhat
Second Review by: Dr. Masumeh Saeedi
Final Approval by: Dr. Eman Refaat Youness
Nyein Nyein Aye1 , Aung Myo Oo2
, Aung Myo Oo2 , Ohnmar Lwin3
, Ohnmar Lwin3 , Ma Saung Oo4
, Ma Saung Oo4 , Kay Thi Myint5
, Kay Thi Myint5 , Khin Than Yee6
, Khin Than Yee6 , Myint Myint Maw7
, Myint Myint Maw7 , Minn Han8
, Minn Han8 , Thin Thin Aung9
, Thin Thin Aung9 , Phyu Phyu Khin10
, Phyu Phyu Khin10 and Mya Mya Thwin11*
and Mya Mya Thwin11*
1Department of Physiology, University of Medicine 2, North Okkalapa, Yangon, Myanmar.
2Biochemistry Unit, International Medical School, Management and Science University, Shah Alam, Selengor, Malaysia.
3Physiology Department, Faculty of Medicine, Manipal University College Malaysia, Melaka, Malaysia.
4Obstetrics and Gynecology Unit, International Medical School, Management and Science University, Shah Alam, Selangor, Malaysia.
5 Department of Surgery, M Kandiah Faculty of Medicine & Health Sciences, Universiti Tunkul Abdul Rahman, Malaysia.
6Preclinical Department, Faculty of Medicine, Universiti Kuala Lumpur, Royal College of Medicine Perak, Malaysia.
7Obstetrics and Gynecology Unit, International Medical School, Management and Science University, Shah Alam, Selangor, Malaysia.
8 Central Executive Committee, Dental Association, Yangon, Myanmar.
9Anatomy Unit, International Medical School, Management and Science University, Selangor 40100, Malaysia.
10Department of Physiology, University of Medicine, Taungyi, Myanmar.
11Physiology Unit, International Medical School, Management and Science University, Shah Alam, Selengor, Malaysia.
Corresponding Author E-mail: drmyamyathwin2011@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3325
Abstract
Hypertension is closely linked to insulin resistance, hyperglycemia, and hyperinsulinemia among non-communicable diseases, and individuals with insulin resistance are at increased risk of premature cardiovascular mortality. Modifying dietary salt intake may influence insulin resistance and related metabolic outcomes. This randomized crossover study assessed the effects of low-salt and high-salt diets on insulin resistance in healthy individuals. Fifty-one apparently healthy male medical students from the University of Medicine 2 participated. Each participant completed a 7-day low-salt diet (<50 mmol/day sodium) and a 7-day high-salt diet (>165 mmol/day sodium) in random order. Dietary compliance was verified by urinary sodium excretion, which was appropriately lower during the low-salt phase and higher during the high-salt phase. On the 8th day of each dietary period, fasting blood samples were collected to measure plasma glucose and serum insulin. Fasting plasma glucose did not differ significantly between the low-salt (5.32 ± 0.50 mmol/L) and high-salt (5.20 ± 0.42 mmol/L) diets. However, fasting serum insulin levels were significantly higher during the low-salt phase compared with the high-salt phase (17.32 ± 8.78 μIU/mL vs. 12.68 ± 5.69 μIU/mL, p<0.001). HOMA-IR was also higher with the low-salt diet (4.1 ± 2.14 vs. 2.9 ± 1.32, p<0.001), whereas QUICKI values were lower (0.32 ± 0.03 vs. 0.33 ± 0.02, p<0.001). These findings indicate that a low-salt diet is associated with greater insulin resistance and reduced insulin sensitivity compared with a high-salt diet. Consequently, recommending sodium intake <50 mmol/day may not be beneficial for improving insulin sensitivity in healthy individuals.
Keywords
Healthy male; High salt; insulin resistance; Insulin sensitivity, Low salt
Download this article as:| Copy the following to cite this article: Aye N. N, Oo A. M, Lwin O, Oo M. S, Myint K. T, Yee Y. T, Maw M. M, Han M, Aung T. T, Khin P. P. Thwin M. M. The Impact of Low and High Salt Intake on Insulin Resistance in Healthy Individuals. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Aye N. N, Oo A. M, Lwin O, Oo M. S, Myint K. T, Yee Y. T, Maw M. M, Han M, Aung T. T, Khin P. P. Thwin M. M. The Impact of Low and High Salt Intake on Insulin Resistance in Healthy Individuals. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/48VhXZ8 |
Introduction
Non-communicable diseases (NCDs) are the leading global cause of mortality, with key contributors including cardiovascular diseases (such as hypertension and ischemic heart disease), diabetes, cancers, and chronic respiratory conditions. The prevalence of diabetes is highest in South-East Asia, the Eastern Mediterranean, and high-income countries in the Western Pacific. According to the World Health Organization (WHO), NCDs are responsible for 74% of all global deaths, accounting for 41 million mortality each year.¹ Among these diseases, hypertension and diabetes mellitus are strongly linked to insulin resistance, hyperglycemia, and hyperinsulinemia.²,³ Insulin resistance is characterized by a diminished sensitivity or responsiveness to the metabolic actions of insulin. Impaired glucose tolerance and fasting hyperglycemia are significant risk factors for the eventual development of diabetes and cardiovascular diseases.⁴,⁵ Epidemiological evidence has also identified hyperinsulinemia as an independent risk factor for cardiovascular disease.⁶ Moreover, individuals with insulin resistance face a heightened risk of premature death due to cardiovascular complications.⁷,⁸
Dietary salt intake is a significant determinant of blood pressure and overall cardiovascular risk.⁹,¹⁰ Research by DiNicolantonio et al¹¹ demonstrated that excessive salt consumption not only elevated blood pressure but also worsened insulin resistance. Similarly, Wu¹² reported that normal subjects on a high-salt diet had reduced insulin sensitivity compared to those on a low-salt diet, suggesting a connection between high salt intake, hypertension, and insulin resistance.
However, contrasting evidence exists. Townsend et al¹³ observed increased glucose uptake in healthy volunteers on a high-salt diet during euglycemic clamp conditions compared to those on a low-salt diet. Additionally, Ogihara et al¹⁴ reported that high-salt-fed rats exhibited enhanced insulin-induced tyrosine phosphorylation of insulin receptor substrates (IRS-1, IRS-2 in muscle and liver, and IRS-3 in the liver) despite the presence of insulin resistance. Similarly, Mitiko et al¹⁵ found that high salt intake increased GLUT4 gene expression, enhancing insulin pathways. These findings suggest a complex and inconclusive relationship between high salt intake and insulin sensitivity. Some researchers have also proposed that a low-salt diet reduced insulin-stimulated glucose uptake in animal models.¹⁶,¹⁷
High salt intake is widely regarded as a public health concern due to its role in increasing blood pressure, a known risk factor for cardiovascular disease. As a result, salt restriction is often promoted to mitigate cardiovascular risk. However, growing evidence suggests that salt restriction may have unintended adverse effects on certain individuals.
This study aimed to evaluate the impact of both low-salt and high-salt diets on insulin resistance in healthy individuals. The findings were expected to contribute to the ongoing debate on whether dietary salt modifications provide meaningful benefits for managing insulin resistance and related metabolic disorders.
Materials and Methods
This study was a quasi-experimental design involving fifty healthy male medical students aged 18–30 years. Eligible participants had a BMI ≥18.5 kg/m² (no upper BMI limit was applied, as all volunteers fell within the normal to slightly overweight range), systolic blood pressure <140 mmHg and diastolic blood pressure <90 mmHg (JNC 8, 2013), serum creatinine <1.2 mg/dL, and fasting plasma glucose <126 mg/dL. Individuals with acute illnesses (such as influenza or diarrhea), a history of diabetes mellitus, cardiovascular diseases (including valvular heart disease and arrhythmias), hypertension, or heavy smoking were excluded. Only males were included to minimize hormonal variability associated with the menstrual cycle, which can influence sodium balance, insulin sensitivity, and metabolic measurements, thereby ensuring greater internal consistency in the study outcomes.
Study design
After a 10-hour fast, individuals are given a low-salt diet (< 50mmol/day sodium). On day 5 of the intervention, dietary salt consumption was measured by collecting spot urine samples. On day 8 of the intervention, venous blood samples and fasting plasma glucose levels were taken, while serum samples were stored for insulin testing. The same method was used to intervene with a high sodium diet (> 165 mmol/day) as mentioned in Figure 1.
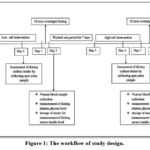 |
Figure 1: The workflow of study design. |
Measurement of Plasma Glucose
An enzymatic colorimetric test is used to measure plasma glucose level by using (Glucose oxidase, phenol, 4-aminophenazone) method. A total of 10 µL of standard or sample was drawn using a specially designed 10 µL micropipette. The absorbance of the standard and sample was measured against the reagent blank within 60 minutes (ΔA) at 500 nm wavelength.
Measurement of serum insulin level
Serum insulin levels were measured using an enzyme-linked immunosorbent assay (ELISA) kit. Serum and urinary creatinine were determined using an auto-creatinine Liquicolor method based on the Jaffe reaction and quantified by a photometric colorimetric test.
Results were presented as mean ± SD. Student’s paired t-test was used to compare data between the low-salt and high-salt intervention periods. Skewed variables were expressed as median and interquartile range and analyzed using the non-parametric Wilcoxon signed-rank test. A p-value of <0.05 was considered statistically significant.
Results
Fasting plasma glucose levels of normal healthy subjects after low salt and high salt interventions
As shown in Figure 2, fasting plasma glucose levels in the current study were 5.32 ± 0.50 mmol/l for low salt intake and 5.2 ± 0.42 mmol/l for high salt intake. It was discovered that there was no significant difference in fasting plasma glucose levels between low-salt and high-salt interventions (p>0.05).
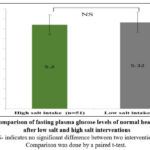 |
Figure 2: Comparison of fasting plasma glucose levels of normal healthy subjects after low salt and high salt interventions. |
Fasting serum insulin levels of normal healthy subjects after high salt and low salt interventions
In the present study, fasting serum insulin levels in low salt and high salt intakes were 17.32 ± 8.78 μIU/ml and 12.68 ± 5.69 μIU/ml, respectively, as depicted in Figure 3. It was found that fasting serum insulin levels were significantly higher in low salt intake than in high salt intake (p< 0.001).
 |
Figure 3: Comparison of fasting serum insulin levels of normal healthy subjects after low salt and high salt interventions |
Homeostasis Model Assessment for Insulin Resistance (HOMA-IR) of normal healthy subjects after low salt and high salt intervention
As shown in Figure 4, medium and interquartile range of insulin resistance (HOMA-IR) of normal healthy subjects after low salt and high salt intervention were 3.8 (2.4-5.6) and 2.7 (1.8-3.6), respectively. After statistical analysis of data from the present study, HOMA-IR of normal healthy subjects was significantly higher in low salt intake than in high salt intake (p<0.001).
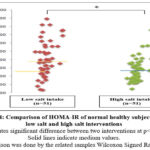 |
Figure 4: Comparison of HOMA-IR of normal healthy subjects after low salt and high salt interventions |
Quantitative Insulin Sensitivity Check Index (QUICKI) of normal healthy subjects after low salt and high salt interventions
QUICKI of normal healthy subjects after low salt and high salt interventions were 0.32 ± 0.03 and 0.33 ± 0.02, respectively, as seen in Figure 5. After statistical analysis of data from the present study, QUICKI of normal healthy subjects was significantly lower in low salt intake than in high salt intake (p <0.001).
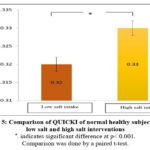 |
Figure 5: Comparison of QUICKI of normal healthy subjects after low salt and high salt interventions. |
Discussion
The present study investigated the impact of low and high salt intake on insulin resistance in healthy individuals, aiming to clarify whether sodium restriction or excess has metabolic consequences beyond blood pressure regulation. Our findings demonstrated that fasting plasma glucose levels were not significantly altered between the two dietary interventions; however, fasting serum insulin levels were markedly higher under the low-salt condition compared to the high-salt condition. This translated into significantly elevated HOMA-IR and reduced QUICKI scores during the low-salt phase, indicating greater insulin resistance.
These findings are consistent with several previous studies. Garg et al¹⁸ reported that insulin resistance, assessed by HOMA-IR, was significantly higher in healthy subjects consuming a low-salt diet compared with those on a high-salt diet. Similar results were described by Townsend et al¹³, who employed the hyperinsulinemic-euglycemic clamp and demonstrated improved glucose disposal rates under high-salt conditions. A systematic review by DiNicolantonio et al¹¹ further consolidated evidence showing that sodium restriction often leads to elevations in fasting insulin and overall worsening of insulin resistance.
However, not all studies have yielded uniform results. Patel et al¹⁹ found no significant differences in fasting insulin concentrations in their meta-analysis of nonrandomized trials investigating sodium restriction, while some smaller studies have suggested neutral or even favorable effects of low-salt diets on glucose metabolism in specific subgroups.²,⁴ These discrepancies may be explained by differences in baseline sodium intake, intervention duration, or participant characteristics.
Mechanistically, one well-documented pathway involves activation of the renin–angiotensin–aldosterone system (RAAS) during low-salt intake, which can impair insulin signaling.¹⁴ Sympathetic nervous system activation during sodium restriction may also reduce tissue perfusion and glucose uptake.¹⁸ In contrast, high-salt intake suppresses RAAS activation, potentially improving insulin sensitivity. However, chronic high-salt consumption may still induce metabolic disturbances via the aldose reductase–fructokinase pathway.²⁰
Limitations of the study
Our study has several strengths, including the use of both fasting insulin and surrogate indices of insulin resistance (HOMA-IR and QUICKI), which provided consistent evidence of impaired insulin sensitivity under low-salt conditions. The crossover design minimized inter-individual variability, and compliance with dietary manipulation was objectively verified through urinary sodium measurements. Nonetheless, several limitations should be acknowledged. The intervention period was short, only eight days, and it remains uncertain whether the observed metabolic changes would persist, diminish, or adapt with longer exposure. The sample size was modest, and we did not measure mechanistic biomarkers such as plasma renin activity, aldosterone, or catecholamines, which could have clarified the contribution of RAAS or sympathetic activation. Moreover, participants were healthy, normotensive adults, limiting generalizability to older individuals or those with obesity, insulin resistance, or hypertension. Future studies should include larger, more diverse populations and employ gold-standard assessments of insulin sensitivity, such as the hyperinsulinemic-euglycemic clamp.
Most notably, each dietary phase lasted only one week, meaning the findings reflect short-term metabolic responses and cannot be extrapolated to long-term effects or used to infer causal dietary recommendations. It also remains unclear whether the increase in insulin resistance seen during low-salt intake represents a transient adaptive response or a sustained physiological effect. Longer interventions, ideally spanning several weeks or months, are needed to determine how metabolic, hormonal, and renal adaptations evolve over time.
Conclusion
In conclusion, our study provides evidence that low-salt intake impairs insulin sensitivity in healthy individuals, as reflected by higher fasting insulin levels, elevated HOMA-IR, and reduced QUICKI compared to high-salt intake, despite no significant differences in fasting glucose. These findings add to growing evidence that sodium restriction, while effective for lowering blood pressure, may adversely affect glucose metabolism in some populations. The results underscore the importance of personalized dietary recommendations and highlight the need for longer-term, mechanistic studies to fully delineate the metabolic consequences of sodium intake. Striking an optimal balance in sodium consumption may therefore be crucial for promoting both cardiovascular and metabolic health.
Acknowledgement
We sincerely acknowledge the valuable guidance and support of our supervisors. Our heartfelt appreciation goes to all the participants. We confirm that this work was carried out solely by the authors listed in this article, and all responsibilities regarding claims related to its content rest with the authors.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
Authorizing body for ethical approval by Ethical Committee, University of Medicine 2, Yangon, Myanmar.
Informed Consent Statement
This study was conducted in accordance with the ethical standards of the institutional and national research committees. Written informed consent was obtained from all participants prior to inclusion in the study. The privacy and confidentiality of all participants were strictly maintained throughout the research process.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors contribution
- Nyein Nyein Aye: Conceptualization, Methodology, Funding Acquisition, Writing – Original Draft
- Aung Myo Oo: Writing – Review & Editing
- Ohnmar Lwin: Writing – Review & Editing
- Ma Saung Oo: Funding Acquisition
- Kay Thi Myint: Visualization, Funding Acquisition
- Khin Than Yee: Funding Acquisition
- Myint Myint Maw: Funding Acquisition
- Minn Han: Funding Acquisition
- Thin Thin Aung: Funding Acquisition
- Phyu Phyu Khin: Supervision.
- Mya Mya Thwin: Conceptualization Writing – Original Draft, Funding Acquisition, Supervision
References
- World Health Organization. Non-communicable diseases: Global Health Estimates 2022 — Life Expectancy and Leading Causes of Death and Disability. Geneva: WHO; 2022. Available from: https://www.who.int/ data/gho/data/themes/topics/topic-details/GHO/ncd-mortality
- Ohishi M. Hypertension with diabetes mellitus: physiology and pathology. Hypertens Res. 2018;41(6):389–393. https://doi.org/10.1038/s41440-018-0034-4
CrossRef - Sowers JR, Whaley-Connell A, Epstein M. The emerging clinical implications of the role of aldosterone in the metabolic syndrome and resistant hypertension: a narrative review. Ann Intern Med. 2009;150(11):776–783. https://doi.org/10.7326/0003-4819-150-11-200906020-00007
CrossRef - Huang Y, Cai X, Mai W, Li M, Hu Y. Association between prediabetes and risk of cardiovascular disease and all-cause mortality: systematic review and meta-analysis. BMJ. 2016;355:i5953. https://doi.org/10.1136/bmj.i5953
CrossRef - Levitan B, Song Y, Ford ES, Liu S. Is non-diabetic hyperglycemia a risk factor for cardiovascular disease? A meta-analysis of prospective studies. Arch Intern Med. 2004;164(19):2147–2155. https://doi.org/10.1001/archinte.164.19.2147
CrossRef - Fazio S, Affuso F, Cesaro A, Tibullo L, Fazio V, Calabrò P. Insulin resistance/hyperinsulinemia as an independent risk factor that has been overlooked for too long. Biomedicines. 2024;12(7):1417. https://doi.org/10.3390/biomedicines12071417
CrossRef - Ormazabal V, Nair S, Elfeky O, Aguayo C, Salomon C, Zuñiga FA. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc Diabetol. 2018;17(1):122. https://doi.org/10.1186/s12933-018-0762-4
CrossRef - Ford ES. The metabolic syndrome and mortality from cardiovascular disease and all causes: findings from the NHANES II Mortality Study. Atherosclerosis. 2004;173(2):309–314. https://doi.org/10.1016/j.atherosclerosis.2003.12.022
CrossRef - Jaques DA. Sodium intake as a cardiovascular risk factor. Nutrients. 2021;13(9):3177. https://doi.org/10.3390/nu13093177
CrossRef - World Health Organization. Global Status Report on Noncommunicable Diseases 2010. Geneva: WHO; 2011.
- DiNicolantonio JJ, O’Keefe JH, Lucan SC. Effects of dietary sodium on insulin resistance and glucose tolerance: a review of clinical evidence. J Metab Health. 2023;2(1):1–15. https://doi.org/10.1097/MH.0000000000000078
- Wu Q. The role of dietary salt in metabolism and energy balance: insights beyond cardiovascular disease. Diabetes Obes Metab. 2023;25(5):1147–1161. https://doi.org/10.1111/dom.14980
CrossRef - Townsend RR, Kapoor S, McFadden CB. Salt intake and insulin sensitivity in healthy human volunteers. Clin Sci (Lond). 2007;113(3):141–148. https://doi.org/10.1042/CS20070141
CrossRef - Ogihara T, Asano T, Ando K, Chiba Y, Sekine N, Sakoda H, et al. Insulin resistance with enhanced insulin signaling in high-salt diet-fed rats. Diabetes. 2001;50(3):573–583. https://doi.org/10.2337/diabetes.50.3.573
CrossRef - Mitiko MO, Hissako DS, Roberta CO. Changes in dietary sodium consumption modulate GLUT4 gene expression and early steps of insulin signaling. Am J Physiol Regul Integr Comp Physiol. 2004;286(4):R779–R785. https://doi.org/10.1152/ajpregu.00439.2003
CrossRef - Pinto PR, Yoshinaga MY, Bianco VD, Bochi AP, Ferreira GS, Pinto IF, et al. Dietary sodium restriction alters muscle lipidomics related to insulin resistance in mice. J Biol Chem. 2021;296:100344. https://doi.org/10.1016/j.jbc.2021.100344
CrossRef - Prada P, Okamoto MM, Furukawa LN, Machado UF, Heimann JC, Dolnikoff MS. High- or low-salt diet from weaning to adulthood: effect on insulin sensitivity in Wistar rats. Am J Hypertens. 2000;13(4):424–429. https://doi.org/10.1016/S0895-7061(99)00238-9
CrossRef - Garg R, Hurwitz S, Williams GH, Brown NJ, Hopkins PN, Adler GK, et al. Low-salt diet increases insulin resistance in healthy subjects. Metabolism. 2011;60(7):965–968. https://doi.org/10.1016/j.metabol.2010.09.004
CrossRef - Patel SM, Cobb JP, Cogswell ME. Effects of sodium reduction on insulin sensitivity: a systematic review and meta-analysis. J Clin Hypertens. 2015;17(10):785–791. https://doi.org/10.1111/jch.12622
CrossRef - Lima LC, Peres CM, Graça FA, et al. High-salt diet activates aldose reductase–fructokinase pathway to induce metabolic syndrome and diabetes. Proc Natl Acad Sci USA. 2018;115(9):2227–2232. https://doi.org/10.1073/pnas.1713837115
CrossRef







