Manuscript accepted on :25-11-2025
Published online on: 10-12-2025
Plagiarism Check: Yes
Reviewed by: Dr Mohammad Haneef
Second Review by: Dr. Priya Bhardwaj
Final Approval by: Dr. Eman Refaat Youness
Anak Agung Gede Indraningrat1* , Pande Putu Christine Putri Purnami1
, Pande Putu Christine Putri Purnami1 , Ketut Agus Yogi Indrawan2
, Ketut Agus Yogi Indrawan2 , Ni Made Ayu Suardani Singapurwa2
, Ni Made Ayu Suardani Singapurwa2 , Made Dharmesti Wijaya3
, Made Dharmesti Wijaya3 , Desak Putu Oki Lestari4
, Desak Putu Oki Lestari4 and Geok Yuan Annie Tan5
and Geok Yuan Annie Tan5
1Department of Microbiology and Parasitology, Faculty of Medicine and Health Sciences, Warmadewa University, Denpasar, Indonesia.
2Department of Food Science and Technology, Faculty of Agriculture, Science and Technology, Warmadewa University, Denpasar, Indonesia.
3Department of Pharmacology, Faculty of Medicine and Health Sciences, Warmadewa University, Denpasar, Indonesia.
4Department of Pathology Anatomy, Faculty of Medicine and Health Sciences, Warmadewa University, Denpasar, Indonesia.
5Institute of Biological Sciences, Faculty of Science, University of Malaya, Kuala Lumpur, Malaysia.
Corresponding Author E-mail: indraningrat@warmadewa.ac.id
DOI : https://dx.doi.org/10.13005/bpj/3299
Abstract
Muntingia calabura has long been used in traditional medicine for its antioxidant and antimicrobial benefits, yet scientific validation of its ethyl acetate leaf extract, particularly against multidrug-resistant (MDR) bacteria, remains limited. This study assessed the phytochemical profile, antioxidant potential, and antibacterial efficacy of the ethyl acetate leaf extract of M. calabura. Qualitative phytochemical screening revealed the presence of flavonoids, tannins, saponins, and steroids. Antioxidant evaluation using the DPPH assay demonstrated very strong radical-scavenging activity, with an IC₅₀ value of 37.32 ppm. Antibacterial testing showed clear inhibitory effects against standard pathogenic strains, with the strongest activity observed against Streptococcus mutans (13.04±2.03 mm), followed by Escherichia coli and Staphylococcus aureus. The extract also exhibited measurable activity against clinically relevant MDR isolates, including ESBL-producing E. coli (8.01±0.10 mm), Klebsiella pneumoniae (7.8±0.5 mm), and Acinetobacter baumannii (8.3±0.2 mm). GC/MS profiling identified 64 compounds, with δ-tocopherol, a chromenone derivative, and a benzopyran analog as the most abundant constituents known for antioxidant and antibacterial properties. Collectively, these findings provide scientific support for the ethnomedicinal use of M. calabura and highlight its potential as a natural source of bioactive compounds active against both standard and MDR bacteria.
Keywords
Antibacterial; Antioxidant; Bioprospecting; Herbal plants; Muntingia calabura
Download this article as:| Copy the following to cite this article: Indraningrat A. A. G, Purnami P. P. C. P, Indrawan K. A. Y, Singapurwa N. M. A. S, Wijaya M. D, Lestari D. P. O, Tan G. Y. A. Potent Antioxidant and Antibacterial Activities of Muntingia calabura Leaf Extract: A Natural Defence Against MDR Bacteria. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Indraningrat A. A. G, Purnami P. P. C. P, Indrawan K. A. Y, Singapurwa N. M. A. S, Wijaya M. D, Lestari D. P. O, Tan G. Y. A. Potent Antioxidant and Antibacterial Activities of Muntingia calabura Leaf Extract: A Natural Defence Against MDR Bacteria. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/3KNspcW |
Introduction
Natural plants and herbal medicines have served as a significant source of bioactive compounds for centuries. The traditional use of plant-based remedies across diverse cultures reflects an empirical recognition of their therapeutic properties.1-5 These plants synthesize a vast array of compounds like alkaloids, flavonoids, tannins, and terpenoids, which function in their defence against environmental pressures and pathogens.6-8 Many of these compounds have since been scientifically validated for their therapeutic potential, which is attributed to their capacity to reduce oxidation, combat inflammation, and inhibit microbial growth.9-14
Over the past few decades, one of the growing threats to global health is antimicrobial resistance (AMR). The misuse and overuse of conventional antibiotics have accelerated the evolution of multidrug-resistant (MDR) bacteria, rendering common infections increasingly difficult to treat.15 A major clinical concern is the increasing prevalence of extended-spectrum β-lactamase (ESBL)–producing bacteria, which exhibit resistance to many frontline antibiotics.16 This worsening crisis highlights the urgent need for new and potent antimicrobial drugs, and natural sources, particularly medicinal plants, could be a promising avenue for this crucial research.1,4
Among the potential medicinal plant candidates is Muntingia calabura, a fast-growing, evergreen tree native to tropical America and widely distributed in many parts of the world, including Indonesia.17 Commonly known as the kersen tree or Jamaican cherry, its leaves, fruit, and bark have been used for centuries in traditional healing practices for managing numerous illnesses.18,19 The plant has been empirically applied to treat wounds, and it is locally recognized for its antioxidant, anti-inflammatory, and antihypertensive properties.20-22 These traditional applications suggest that M. calabura possesses a rich array of bioactive compounds that warrant further scientific exploration.19
While previous studies have highlighted the general bioactivities of M. calabura, there remains a need to specifically identify its phytochemical composition and validate its antibacterial potential against clinically relevant multidrug-resistant (MDR) strains.1 Therefore, this study was designed to conduct a comprehensive characterization of the ethyl acetate leaf extract of M. calabura. The investigation aimed to perform a qualitative phytochemical screening to identify major compound classes, to quantify its antioxidant capacity using the DPPH assay, and to determine its inhibitory effects on both pathogenic reference strains and their multidrug-resistant counterparts. Subsequently, gas chromatography coupled to mass spectrometry (GC/MS) was employed to analyse the individual phytochemical constituents within the extract that may contribute to its observed bioactivities. This research provides a detailed chemical and biological profile to scientifically validate the plant’s traditional uses and highlight its potential as a source for novel antimicrobial agents.
Materials and Methods
Sample collection and plant identification
Fresh, mature leaves of Muntingia calabura were collected in June 2024 from Pulau Singkep Street, Denpasar, Bali, Indonesia (8°41′22.1″S; 115°12′34.8″E) (Figure 1). Only healthy, intact, uniformly green leaves from fruiting trees were selected.
 |
Figure 1: M. calabura plant species (left) and M. calabura leaves (right).
|
Plant identification was performed by comparing morphological characteristics with established taxonomic references at the Characterization Laboratory of the National Research and Innovation Agency, “Eka Karya” Botanical Garden, Bedugul, Tabanan, Bali. A voucher specimen was deposited under identification number 1617-86680-1.
Sample preparation and extraction
Fresh leaves (2 kg) of M. calabura were cleaned, oven-dried at 45 °C to ≤10% moisture, ground, and sieved (40-mesh) to obtain uniform powder. A total of 200 g of the powder was macerated with ethyl acetate (SMART-Lab, Indonesia) at a 1:5 (w/v) ratio for 3 × 24 h under light-protected conditions with continuous shaking (OHAUS, USA). Filtrates were collected through Whatman No. 1 filter paper (Cytiva, China), and the residue was re-extracted under identical conditions. Combined extracts were concentrated using a rotary evaporator (Cole-Palmer, USA) at 40 °C and oven-dried to obtain the crude ethyl acetate extract.
Phytochemical screening
Qualitative phytochemical profiling of the ethyl acetate extract was performed following established protocols for preliminary plant metabolite screening.23 Alkaloids were detected using Mayer’s and Dragendorff’s reagents; tannins with FeCl₃; flavonoids with concentrated H₂SO₄; saponins using the froth test; and steroids/terpenoids via the Liebermann–Burchard reaction. Metabolite classes were confirmed based on characteristic colour changes or precipitate formation.
Antioxidant assay
The antioxidant activity of the ethyl acetate extract of M. calabura was evaluated using a DPPH radical scavenging test. The extract was serially diluted to obtain final concentrations of 5, 10, 15, 20, and 25 ppm. Each 1 mL aliquot of the diluted samples was mixed with 1 mL of a 0.1 mM DPPH solution. The mixtures were gently vortexed and then incubated in the dark for 45 minutes to allow the reaction to proceed. A reduction in absorbance at 517 nm, measured with a UV–Vis spectrophotometer, reflected the scavenging of DPPH radicals. The IC₅₀ value was determined from the inhibition percentage to indicate the antioxidant strength of the extract.
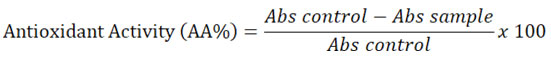
Antibacterial activity screening
The antibacterial potential of the M. calabura extract was examined using the Kirby–Bauer disk diffusion technique. Sterile 6 mm paper discs (Macherey-Nagel, Germany) were loaded with 20 µL of the extract and left to air-dry for 15 minutes to ensure complete absorption. The discs were then placed onto Luria–Bertani agar plates that had been pre-inoculated with the test organisms. The bacterial panel comprised standard strains (Staphylococcus aureus ATCC 25923, Streptococcus mutans FNCC 0405, Escherichia coli ATCC 25922, and Klebsiella pneumoniae ATCC 700603) as well as multidrug-resistant isolates, including ESBL-producing E. coli, K. pneumoniae, and Acinetobacter baumannii. Each test was carried out in triplicate. Levofloxacin (5 µg/disc; Oxoid, UK) served as the positive control, while discs containing only ethyl acetate were used as the negative control. Antibacterial efficacy was expressed as the mean diameter of inhibition zones obtained from three independent measurements for each strain.
Gas chromatography-mass spectrometry analysis
Chemical profiling of the ethyl acetate extract was conducted at the Forensic Laboratory of Polda Bali using a GC–MS system (Agilent 7890B GC coupled with a 5977B MSD; Agilent Technologies, USA). Separation was achieved on an HP-5ms Ultra Inert capillary column (30 m × 250 µm × 0.25 µm) with helium as the carrier gas (2.9 mL/min). Samples (1 µL) were injected in splitless mode at 290 °C. The oven was programmed from 70 °C (5 min) to 290 °C at 10 °C/min, with a final hold of 3 min. Mass spectra were acquired under electron ionization (70 eV) in scan mode (m/z 50–550). Compound identification was performed by comparing spectra against the NIST 17 and Wiley 11 mass spectral libraries, supported by published literature.
Statistical analysis
All quantitative data were analysed using Microsoft Excel 2023 (Microsoft Corp., USA). Antibacterial activity was expressed as mean ± standard deviation (SD) from three independent replicates. IC₅₀ values for the DPPH assay were calculated using linear regression of the concentration–inhibition curve.
Results
Analysis of phytochemicals in the ethyl acetate extract
Qualitative phytochemical tests were carried out on the ethyl acetate extract of M. calabura leaves to identify the presence of key secondary metabolite classes. The screening involved a series of chemical tests designed to produce observable reactions, such as colour changes or precipitates, in the presence of specific compounds. The overall findings are summarized in Table 1.
Table 1: Phytochemical profile of the M. calabura extract. A plus sign (+) denotes the detection of a compound class, while a minus sign (-) denotes its absence.
|
Phytochemical compounds |
Result |
|
Alkaloids |
– |
|
Tannins |
+ |
|
Flavonoids |
+ |
|
Saponins |
+ |
|
Steroids |
+ |
|
Terpenoids |
– |
In the alkaloid test, no precipitate was observed following the addition of Mayer’s or Dragendorff’s reagents, indicating a negative result. A positive reaction would typically be marked by the formation of a white precipitate with Mayer’s reagent or a yellow precipitate with Dragendorff’s reagent.
The test for tannins was conducted using an iron (III) chloride (FeCl₃) solution. When the FeCl₃ reagent was added to the ethyl acetate extract, the solution immediately changed to a dark green colour. This colour change is a characteristic positive reaction, confirming the presence of tannins.
Two separate methods were employed to detect flavonoids. In the first test, the addition of concentrated H₂SO₄ to the extract caused a colour change to slightly yellowish, which is considered a positive result. In the second test, which used a mixture of hot ethanol, concentrated hydrochloric acid (HCl), and magnesium powder, the extract did not produce the expected red or orange colour. Despite the negative result from the second test, the positive reaction with sulfuric acid was considered sufficient evidence for the detection of flavonoids.
To evaluate the presence of saponins, an aliquot of the extract was vigorously agitated in hot distilled water, which resulted in a stable foam layer measuring between 1 and 10 cm. The persistence of this foam was verified when it did not dissipate after a few drops of 1N HCl were added. This formation of a stable froth is a classic indication that saponins are present.
The Liebermann-Burchard test was used to screen for both steroids and terpenoids. Upon addition of the reagent to the extract, the solution turned a distinct blue colour. This blue coloration is a positive result for steroids. However, the test was negative for terpenoids, as the solution did not develop the reddish-orange colour that would indicate their presence.
Antioxidant screening
The extract demonstrated notable, dose-dependent antioxidant activity. Across the tested concentration range, DPPH radical scavenging by the ethyl acetate extract ranged from 12.85% at the lowest concentration to 35.97% at the highest concentration. Based on the inhibition data, the half-maximal inhibitory concentration (IC₅₀) for the extract was determined to be 37.32 ppm (Table 2). Based on standard classifications for the DPPH assay24, this value indicates that the extract possesses very strong antioxidant activity.
Table 2: Dose-response analysis and IC₅₀ value of M. calabura leaf extracts.
|
Concentration (ppm) |
Average Absorbance (Abs) |
% Inhibition |
IC50 (ppm) |
Category |
|
0 |
0.519 |
0.00 |
37.32 |
Very strong |
|
5 |
0.452 |
12.85 |
||
|
10 |
0.430 |
17.08 |
||
|
15 |
0.391 |
24.66 |
||
|
20 |
0.364 |
29.87 |
||
|
25 |
0.332 |
35.97 |
Antibacterial screening
The ethyl acetate extract of M. calabura leaves demonstrated antibacterial activity against all tested strains (Figure 2 and Table 3). The largest zone of inhibition was observed against S. mutans FNCC 0405 (13.04 ± 2.03 mm), followed by E. coli ATCC 25922 (10.93 ± 0.67 mm), S. aureus ATCC 25923 (9.87 ± 0.28 mm), and K. pneumoniae ATCC 700603 (8.52 ± 0.81 mm). In addition, the ethyl acetate extract showed antibacterial activity against MDR bacteria, with zones of inhibition of 8.0 ± 0.1 mm for E. coli ESBL, 7.8± 0.5 mm for K. pneumoniae ESBL () and 8.3± 0.2 mm for A. baumannii ESBL .
As expected, the positive control levofloxacin (Table 3) produced markedly larger inhibition zones across all strains (13.80–24.87 mm), confirming assay validity. In contrast, the negative control (ethyl acetate) showed no inhibition (0 mm) for all bacteria tested, indicating that the observed activity originated exclusively from the plant extract. Antibacterial potency was categorized using a four-level scale based on the inhibition zone diameter. Activity levels were interpreted as weak (0-5 mm), moderate (5-10 mm), strong (10-20 mm), and very strong (>20 mm).25
Table 3: Inhibition of test bacteria by the M. calabura extract. The presented inhibition zone diameters are the average of three replicates.
|
No |
Bacterial target strains |
M. calabura crude extracts |
Positive control (levofloxacin) |
Negative control (ethyl acetate) |
|
1 |
S. aureus ATCC 25923 |
9.87±0.28 |
24.87 ± 1.14 |
0 ± 0 |
|
2 |
S. mutans FNCC 0405 |
13.04±2.03 |
18.30 ± 0.94 |
0 ± 0 |
|
3 |
E. coli ATCC 25922 |
10.93±0.67 |
22.31 ± 0.49 |
0 ± 0 |
|
4 |
K. pneumoniae ATCC 700603 |
8.52±0.81 |
22.75 ± 0.56 |
0 ± 0 |
|
5 |
E. coli ESBL |
8.00±0.10 |
25.0 ± 0.30 |
0 ± 0 |
|
6 |
K. pneumoniae ESBL |
7.80±0.50 |
13.7± 0.20 |
0 ± 0 |
|
7 |
A. baumannii ESBL |
8.30±0.20 |
22.70 ± 0.60 |
0 ± 0 |
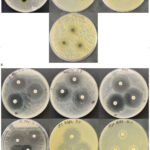 |
Figure 2. Disc diffusion assay showing the antibacterial activity of the ethyl acetate leaf extract of M. calabura. Panels arranged from left to right correspond to S. aureus ATCC 25923, S. mutans FNCC 0405, E. coli ATCC 25922, K. pneumoniae ATCC 700603, E. coli ESBL, K. pneumoniae ESBL, and A. baumannii ESBL.
|
GC/MS analysis
The GC/MS analysis (Figure 3) detected 64 different compounds from ethyl acetate extract of M. calabura. Among these 64 compounds, nine compounds have been associated with antibacterial activities (Table 4). Of these 9 potential antibacterial compounds that were detected, three compounds displayed percentage above 4 percent namely δ-tocopherol, 7-Hydroxy-3-methoxy-2-p-methoxyphenyl-4H-chromen-4-one, and 4H-1-Benzopyran-4-one,5-hydroxy-6,7-dimethoxy-2-phenyl which can be seen in Figure 3.
 |
Figure 3: Mass spectra of three of the most abundant compounds detected by GC–MS analysis of the ethyl acetate extract of M. calabura. Panels show: (A) δ-tocopherol (RT 26.875 min; 14.57%), (B) 7-Hydroxy-3-methoxy-2-p-methoxyphenyl-4H-chromen-4-one (RT 24.695 min; 6.38%), and (C) 4H-1-Benzopyran-4-one, 5-hydroxy-6,7-methoxy-2-phenyl (RT 25.824 min; 4.03%).
|
Table 4: Potential antibacterial constituents identified in the ethyl acetate crude extract of M. calabura through GC/MS analysis.
|
Compound Name |
Retention Time (RT) (min) |
Relative Area (%) |
Reference |
|
δ-tocopherol |
26.875 |
14.57 |
26 |
|
7-Hydroxy-3-methoxy-2-p-methoxyphenyl-4H-chromen-4-one |
24.695 |
6.38 |
27 |
|
4H-1-Benzopyran-4-one, 5-hydroxy-6,7-methoxy-2-phenyl- (Sloflavone) |
25.824 |
4.03 |
28 |
|
dl-α-Tocopherol (Vitamin E) |
28.166 |
3.25 |
29 |
|
Neophytadiene |
17.502 |
2.75 |
30 |
|
γ-Tocopherol |
27.406 |
2.10 |
31 |
|
Phytol |
20.181 |
1.54 |
32 |
|
n-Hexadecanoic acid (palmitic acid) |
18.829 |
1.30 |
33 |
|
Stigmasterol |
29.322 |
1.07 |
34 |
Discussion
This study aimed to provide a scientific characterization of the ethyl acetate leaf extract of M. calabura, with emphasis on its phytochemical composition, antioxidant potential, and antibacterial activity. The results demonstrate that the extract contains diverse bioactive constituents with notable therapeutic relevance. Collectively, the findings offer a solid scientific basis supporting the plant’s traditional use and highlight its promise as a candidate for modern drug discovery.18 In addition, the consistency of the quantitative data (as reflected by relatively low standard deviations across replicates) strengthens the reliability of these findings and supports the biological relevance of the observed activities.
The qualitative phytochemical screening successfully identified several major classes of bioactive compounds, including tannins, flavonoids, saponins, and steroids, within the extract. The confirmed presence of polyphenolic compounds such as flavonoids and tannins is particularly noteworthy, as it directly correlates with the extract’s potent antioxidant capacity, which was quantified as very strong with IC₅₀ value of 37.32 ppm in the DPPH assay. This exceptional radical-scavenging activity provides a clear biochemical basis for the traditional use of M. calabura in managing ailments associated with oxidative stress.18 The low IC₅₀ value also indicates that the extract exhibits antioxidant efficiency comparable to many plant species previously reported as strong natural antioxidants, further supporting its potential biomedical applications.
In the antibacterial evaluation, the extract demonstrated remarkable broad-spectrum activity, effectively inhibiting both Gram-negative (E. coli, K. pneumoniae) and Gram-positive (S. aureus, S. mutans) bacterial targets. The efficacy was most pronounced against S. mutans, which yielded the largest zone of inhibition at 13.04 ± 2.03 mm, suggesting a targeted potential for applications in oral healthcare. This finding is supported statistically by the magnitude of the inhibition zone, which falls within the strong activity category, indicating that the detected compounds exert meaningful biological effects rather than incidental or weak activity. This wide-ranging antimicrobial effect is logically attributed to the synergistic actions of the detected phytochemicals, which are well-documented to compromise bacterial cell integrity and disrupt essential metabolic pathways.19
A pivotal finding of this investigation is the extract’s definitive efficacy against clinically significant MDR bacteria. The extract produced clear zones of inhibition against ESBL-producing E. coli (8.0 ± 0.1 mm) and K. pneumoniae (7.8 ± 0.5 mm), as well as A. baumannii ESBL (8.3 ± 0.2 mm). Although these values fall within the moderate activity range, the remarkably low standard deviations as observed against E. coli ESBL indicated highly reproducible antibacterial effects against this resistant bacterial target. This capacity to inhibit pathogens that have evolved resistance to conventional antibiotics directly addresses the global AMR crisis and positions M. calabura as a promising source of novel antimicrobial leads. The ability of a crude plant extract to suppress MDR strains also suggests that its mechanisms of action may bypass conventional resistance pathways, an important implication for future drug development. 35,36
The GC/MS analysis provided a detailed chemical blueprint of the extract, linking the observed bioactivities to specific molecules among the 64 compounds detected. Three compounds with known antibacterial properties were present in particularly high concentrations: δ-tocopherol (14.57%), the flavonoid 7-Hydroxy-3-methoxy-2-p-methoxyphenyl-4H-chromen-4-one (6.38%), and 4H-1-Benzopyran-4-one, 5-hydroxy-6,7-dimethoxy-2-phenyl (4.03%). The substantial abundance of these molecules strongly indicates that they are the primary drivers of the extract’s powerful antioxidant and antibacterial performance. Furthermore, the presence of other bioactive compounds like n-hexadecanoic acid (1.30%) and phytol (1.54%) likely contributes to a synergistic effect, enhancing the overall therapeutic potential of the crude extract.37 The alignment between the relative abundance of these metabolites and the strength of the biological effects observed (e.g., higher inhibition zones in Gram-positive strains) supports their functional significance and strengthens the mechanistic interpretation of the results.
While this study provides a robust validation of the extract’s potential, its scope also defines clear imperatives for future research. The current antibacterial assessment, based on the disc diffusion method, must be advanced by quantitative analyses to establish the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) values. Moreover, it is imperative to pursue the bioassay-guided isolation of the most abundant compounds, specifically δ-tocopherol and the identified flavonoids, to confirm their individual bioactivities and elucidate their precise mechanisms of action against MDR pathogens. Such studies will also help determine whether the observed antibacterial effects arise predominantly from single active molecules or from synergistic interactions, an important distinction for future pharmaceutical development.38
Conclusion
The ethyl acetate leaf extract of M. calabura is a potent reservoir of bioactive phytochemicals, including flavonoids, tannins, and steroids. This study conclusively demonstrates that the extract possesses very strong antioxidant activity (IC₅₀ of 37.32 ppm) and exhibits significant, broad-spectrum antibacterial efficacy against both standard pathogenic and challenging multidrug-resistant strains. The inhibition zones obtained across replicates were consistent, indicating reliable antibacterial performance and supporting the validity of the findings. The high relative abundance of specific compounds, namely δ-tocopherol, 7-Hydroxy-3-methoxy-2-p-methoxyphenyl-4H-chromen-4-one, and 4H-1-Benzopyran-4-one, 5-hydroxy-6,7-dimethoxy-2-phenyl, provides a definitive chemical basis for these observed biological activities. Together, these results present a concise summary of the extract’s antioxidant strength, antibacterial potential, and phytochemical profile. Ultimately, this research provides robust scientific validation for the ethnobotanical use of M. calabura and establishes it as a high-priority candidate for the discovery and development of novel natural products to combat antimicrobial resistance.
Acknowledgment
We would like to thank the Faculty of Medicine and Health Sciences for providing lab access to perform all experiments presented in this study.
Funding Sources
This research was supported financially by the International Collaboration Research Grant fiscal year 2024 provided by Faculty of Medicine and Health Sciences Warmadewa University, grant no 102/Unwar/FKIK/UP2M/PD-13/IX/2024 awarded to Anak Agung Gede Indraningrat.
Conflict of Interest
The author(s) do not have any conflicts of interest.
Data Availability Statement
Data related to antibacterial screening and GC/MS can be accessed via the Figshare online repository https://figshare.com/s/866b07164624e1da7d6f.
Ethics Statement
This study received ethical approval from the Ethics Commission of the Faculty of Medicine and Health Sciences, Warmadewa University, Denpasar-Bali (Approval No. 545/Unwar/FKIK/EC-KEPK/XII/2024).
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not applicable.
Author contributions
- Anak Agung Gede Indraningrat: Conceptualization, Methodology, Supervision, Writing – Original Draft, Funding Acquisition.
- Pande Putu Christine Putri Purnami: Investigation, Data Collection, Antioxidant and Antibacterial Assays.
- Ketut Agus Yogi Indrawan: Sample Collection, Preparation, Extraction.
- Ni Made Ayu Suardani Singapurwa: Sample Collection, Preparation, Extraction.
- Made Dharmesti Wijaya: GC/MS Analysis, Data Interpretation, Visualization.
- Desak Putu Oki Lestari: Culture and Maintenance of Multidrug-Resistant Strains.
- Geok Yuan Annie Tan: Data Interpretation, Supervision.
References
- Vaou N, Stavropoulou E, Voidarou C, Tsigalou C, Bezirtzoglou E. Towards Advances in Medicinal Plant Antimicrobial Activity: A Review Study on Challenges and Future Perspectives. Microorganisms. 2021;9(10).
CrossRef - Ikrar Musyaffa MS, Yudistira N, Rahman MA, Basori AH, Firdausiah Mansur AB, Batoro J. IndoHerb: Indonesia medicinal plants recognition using transfer learning and deep learning. Heliyon. 2024;10(23):e40606.
CrossRef - Ralte L, Sailo H, Singh YT. Ethnobotanical study of medicinal plants used by the indigenous community of the western region of Mizoram, India. Journal of Ethnobiology and Ethnomedicine. 2024;20(1):2.
CrossRef - Zouine N, Ghachtouli NE, Abed SE, Koraichi SI. A comprehensive review on medicinal plant extracts as antibacterial agents: Factors, mechanism insights and future prospects. Sci Afr. 2024;26:e02395.
CrossRef - Gao P, Nasution AK, Ono N, Kanaya S, Altaf-Ul-Amin M. Investigating Potential Anti-Bacterial Natural Products Based on Ayurvedic Formulae Using Supervised Network Analysis and Machine Learning Approaches. Pharmaceuticals (Basel). 2025;18(2):192.
CrossRef - Idrees M, Javaid S, Nadeem S, et al. Antimicrobial and Hepatoprotective Properties of Pods of Acacia nilotica (L.) Willd. Ex Delile: In Vivo and In Silico Approaches. Dose-Response. 2024;22(4).
CrossRef - Ndhlovu PT, Asong JA, Omotayo AO, Otang-Mbeng W, Aremu AO. Ethnobotanical Survey of Medicinal Plants Used by Indigenous Knowledge Holders to Manage Healthcare Needs in Children. Plos One. 2023;18(3):e0282113.
CrossRef - Tanya M, Priyadarsini D, Nayak RK. Screening of Bioactive Compounds From Madagascar Periwinkle and Their Importance in Drug Discovery. Int J Multidiscip Res. 2022;4(6).
CrossRef - Aman Z, Singh S, Kumar P, et al. Current scenario of geriatric nutrition and health correlates: The silver age review. Annals of Phytomedicine An International Journal. 2024;13:146-157.
CrossRef - Elhag MMA, Yassin AKA, Mustafa NEM. Herbal Medicines as Antimicrobial Agents. In: Plants as Medicine and Aromatics: Uses of Botanicals.2024:261-291.
CrossRef - Pérez-Flores JG, García-Curiel L, Pérez-Escalante E, et al. Plant Antimicrobial Compounds and Their Mechanisms of Action on Spoilage and Pathogenic Bacteria: A Bibliometric Study and Literature Review. Applied Sciences (Switzerland). 2025;15(7).
CrossRef - Shahrajabian MH. Medicinal Herbs with Anti-Inflammatory Activities for Natural and Organic Healing. Current Organic Chemistry. 2021;25(23):2885-2901.
CrossRef - Srivastava S, Khan MS, Ahmad S, Dubey A, Saxena VL, Haneef M. Identification of putative antiviral bioactive compounds derived from family Asteraceae: An in silico approach. J Cell Biochem. 2024;125(4):e30538.
CrossRef - Thakur P, George N, Chakraborty M. Synergistic effect of fruit, seeds and leaves as antioxidants: A review. Plant Cell Biotechnology and Molecular Biology. 2020;21(19-20):9-14.
- Tahmasebi H, Arjmand N, Monemi M, et al. From Cure to Crisis: Understanding the Evolution of Antibiotic-Resistant Bacteria in Human Microbiota. Biomolecules. 2025;15(1).
CrossRef - Sunarno S, Puspandari N, Fitriana F, Nikmah UA, Idrus HH, Panjaitan NSD. Extended spectrum beta lactamase (ESBL)-producing Escherichia coli and Klebsiella pneumoniae in Indonesia and South East Asian countries: GLASS Data 2018. AIMS Microbiol. 2023;9(2):218-227.
CrossRef - Hasan M. An extended distribution of Non-Native Tree species Muntingia calabura L. in Rajshahi, Bangladesh. JPRB. 2021;5:12-16.
- Ansori A, Kharisma V, Solikhah T. Medicinal properties of Muntingia calabura L.: A Review. RJPT. 2021:4509-4512.
CrossRef - Kuchekar M, Upadhye M, Pujari R, Kadam S, Gunjal P. Muntingia calabura: A comprehensive review. J Pharm Biol Sci. 2021;9:81-87.
CrossRef - Hanum TI, Maha HL, Wahyuni HS, Fadli R, Saragih IY. Nanoparticles of Muntingia calabura L. Ethanol extract: Preparation and Anti-inflammatory activity. Research Journal of Pharmacy and Technology. 2024;17(10):4677-4684.
CrossRef - Kehinde BA, Nayik GA, Rafiq S. Muntingia calabura. In: Antioxidants in Fruits: Properties and Health Benefits.2020:251-270.
CrossRef - Muniyappan G, Adenam NM, Muhamad Yuzaini Azrai MY, Nurul Hijanah MH, Kernain D, Adli HK. Phytochemical Screening of Muntingia calabura Fruit for Antioxidant and Cytotoxic Activities. Journal of Advanced Research in Applied Sciences and Engineering Technology. 2022;28(1):116-125.
CrossRef - Maheshwaran L, Nadarajah L, Senadeera SPNN, Ranaweera CB, Chandana AK, Pathirana RN. Phytochemical Testing Methodologies and Principles for Preliminary Screening/ Qualitative Testing. Asian Plant Research Journal. 2024;12(5):11–38.
CrossRef - Martinez-Morales F, Alonso-Castro AJ, Zapata-Morales JR, Carranza-Álvarez C, Aragon-Martinez OH. Use of standardized units for a correct interpretation of IC50 values obtained from the inhibition of the DPPH radical by natural antioxidants. Chemical Papers. 2020;74(10):3325-3334.
CrossRef - Fachriyah E, Wibawa PJ, Awaliyah A. Antibacterial activity of basil oil (Ocimum basilicum L) and basil oil nanoemulsion 2020;1524. doi:10.1088/1742-6596/1524/1/012060.
CrossRef - Chen PJ, Lin ES, Su HH, Huang CY. Correction: Chen et al. Cytotoxic, Antibacterial, and Antioxidant Activities of the Leaf Extract of Sinningia bullata. Plants 2023, 12, 859. Plants (Basel). 2023;12(20).
CrossRef - Shamsudin NF, Ahmed QU, Mahmood S, et al. Antibacterial Effects of Flavonoids and Their Structure-Activity Relationship Study: A Comparative Interpretation. Molecules. 2022;27(4).
CrossRef - Tracanna M, Amani S, Romano E, et al. Crystal structure, spectroscopic properties and antimicrobial activity of 4H-1-Benzopyran-4-one, 5-hydroxy-2-(4-hydroxyphenyl)-7-methoxy-6,8-dimethyl from Miconia ioneura Griseb. Melastomataceae. Molecular Medicinal Chemistry. 2010;21:94-104.
- Ijoma I, Ishmael V, Ajiwe V, Ijoma K. Jatropha tanjorensis a Flora of Southeast Nigeria: Isolation and Characterization of Naringenin and Validation of Bio-enhanced Synergistical Activity of α-Tocopherol Toward Clinical Isolates of Resistant Bacterial. Makara J Sci. 2022;26:120-127.
CrossRef - Rajeswaran S, Rajan DK. Neophytadiene: Biological activities and drug development prospects. Phytomedicine. 2025;143:156872.
CrossRef - Ghimire BK, Yu CY, Chung I-M. Assessment of the phenolic profile, antimicrobial activity and oxidative stability of transgenic Perilla frutescens L. overexpressing tocopherol methyltransferase (γ-tmt) gene. PPB. 2017;118:77-87.
CrossRef - Saha M, Bandyopadhyay PK. In vivo and in vitro antimicrobial activity of phytol, a diterpene molecule, isolated and characterized from Adhatoda vasica Nees. (Acanthaceae), to control severe bacterial disease of ornamental fish, Carassius auratus, caused by Bacillus licheniformis PKBMS16. Microb Pathog. 2020;141:103977.
CrossRef - Ganesan T, Subban M, Britto D, Bharathi S, Seedevi P. Structural characterization of n-hexadecanoic acid from the leaves of Ipomoea eriocarpa and its antioxidant and antibacterial activities. Biomass Convers Biorefin. 2022;14.
CrossRef - Lestari S, Kurnia D, Mayanti T, Heliawati L. Antimicrobial Activities of Stigmasterol from Piper crocatum In Vitro and In Silico. J Chem. 2024;2024(1):2935516.
CrossRef - Ali SS, El-Zawawy NA, Al-Tohamy R, El-Sapagh S, Mustafa AM, Sun J. Lycium shawii Roem. & Schult.: A new bioactive antimicrobial and antioxidant agent to combat multi-drug/pan-drug resistant pathogens of wound burn infections. Journal of Traditional and Complementary Medicine. 2020;10(1):13-25.
CrossRef - Chowdhury SK, Misra D, Mandal V. Medicinal plant-derived antimicrobials’ fight against multidrug-resistant pathogens. In: Medicinal and Aromatic Plants: Healthcare and Industrial Applications.2021:391-427.
CrossRef - Hasan MM, Ahmed M, Karmaker M, Khan MSH, Begum R, Asna SMZH. Microbiological Analysis in Different Brands of Hepato-Protective Liquid Herbal Medicines Available in the Dhaka City. IJFCPR. 2023;3(1):053-057.
CrossRef - Isaei E, Mansouri S, Rahmani M, Sharififar F, Salary A. Novel synergistic activity of quercus infectoria gall extract with ceftazidime against standard and multiple drug resistant pseudomonas aeruginosa and Escherichia coli isolates. Archives of Iranian Medicine. 2021;24(9):684-688.
CrossRef







