Manuscript accepted on :11-11-2025
Published online on: 15-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Adinarayana Andy
Second Review by: Dr. Vikrant Dandekar
Final Approval by: Dr. Prabhishek Singh
Guli Raimova 1* , Siroj Khodjiyev 1
, Siroj Khodjiyev 1 , Kabil Nasirov1
, Kabil Nasirov1 , Nozim Khoshimov1
, Nozim Khoshimov1 , Mukhamatkodir Ortikov 1
, Mukhamatkodir Ortikov 1 , Alisher Mukhtorov3
, Alisher Mukhtorov3 , Rustam Makhmudov2
, Rustam Makhmudov2 , Gaуbullo Lutpillaеv2, Zamira Teshabayeva4
, Gaуbullo Lutpillaеv2, Zamira Teshabayeva4 , Rano Nosirova4
, Rano Nosirova4 , Zulfizar Tuychiyeva4, Sabina Normurodova4 and Akbarova Gulbakhor5
, Zulfizar Tuychiyeva4, Sabina Normurodova4 and Akbarova Gulbakhor5
1Department of Electrophysiology, Institute of Biophysics and Biochemistry, National University of Uzbekistan, Tashkent, Uzbekistan
2Department of Protein and Peptide Chemistry, Institute of Bioorganic Chemistry, Academy of Sciences of the Republic of Uzbekistan, Tashkent, Uzbekistan
3Department of Human and Animal Physiology, Nаtiоnаl Univеrsitу оf Uzbеkistаn named after M. Ulugbek,Tashkent, Uzbekistan
4Department of Pedagogical of preschool education of Chirchik State Pedagogical University, Chirchik, Uzbekistan
5Department of Pedagogical University named after Nizam, Tashkent State, Uzbekistan
Corresponding Author E-mail: guliraimova111@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3310
Abstract
In type 2 diabetes mellitus, all hemostatic systems involved in blood clotting, regulation of vascular tone, the condition of blood cells, and plasma enzyme systems are activated. In this disease, there is a clear correlation between hemocoagulation disorders and the degree of glycemia. In this regard, an important and urgent task is to study these mechanisms and to search for new drugs that can prevent further progression. In this study, experiments on model rats were conducted to test the effects of polyphenols as antidiabetic agents. Polyphenol-containing foods and beverages such as coffee, guava tea, blueberries, olive oil, propolis, chocolate, red wine, grape seeds, and cocoa have been shown to exert beneficial effects in individuals with type 2 diabetes mellitus. These compounds contribute to better glucose regulation, enhance vascular health, reduce insulin resistance, and help lower HbA1c levels, thereby potentially mitigating both the progression and complications of diabetes, including alterations in the hemostatic system. This paper presents a study of polyphenol preparations isolated from the Pistacia vera plant (compound ANK-1), the Karelinia caspia plant (compound KCL-1), and an extract from the Isatis tinctoria L. plant (compound ITL-2), and their effects on biochemical parameters and the blood-clotting system in rats with experimentally induced type 2 diabetes caused by streptozotocin.
Keywords
Anticoagulants; Fibrinogen; Hemostatic system; Polyphenols; Streptozotocin; Type 2 diabetes mellitus
Download this article as:| Copy the following to cite this article: Raimova G, Khodjiyev S, Nasirov K, Khoshimov N, Ortikov M, Mukhtorov A, Makhmudov R, Lutpillaеv G, Teshabayeva Z, Nosirova R, Tuychiyeva Z, Normurodova S, Gulbakhor A. Effect of Polyphenols on the Hemostatic System in Rats with Streptozotocin-induced Type II Diabetes. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Raimova G, Khodjiyev S, Nasirov K, Khoshimov N, Ortikov M, Mukhtorov A, Makhmudov R, Lutpillaеv G, Teshabayeva Z, Nosirova R, Tuychiyeva Z, Normurodova S, Gulbakhor A. Effect of Polyphenols on the Hemostatic System in Rats with Streptozotocin-induced Type II Diabetes. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4rYq2Vq |
Introduction
The International Diabetes Federation reports that both obesity and type 2 diabetes mellitus have reached epidemic proportions globally.1 Dangerous complications resulting from chronic hyperglycemia lead to disruptions of the hemostatic system, vascular wall damage, and the development of atherosclerosis.2 Numerous preclinical investigations are currently underway to identify effective strategies for the prevention and management of type 2 diabetes mellitus.
Two major pathological mechanisms play a central role in the development of this disease: insulin resistance and dysfunction of pancreatic beta cells. These disturbances impair insulin’s ability to regulate glucose, lipid, and protein metabolism effectively.3-6 Type 2 diabetes mellitus typically progresses through multiple stages, with the transition from prediabetes to clinically manifest diabetes often occurring over several years.7,8
Among experimental models, those induced by dietary factors most accurately reflect the etiology and pathogenic mechanisms of type 2 diabetes in humans.9 To develop effective therapeutic agents against type 2 diabetes and obesity, it is essential to establish experimental models that closely mimic the clinical features of these conditions and enable rapid reproduction of the key pathogenetic stages involved in their progression.10 One commonly used approach involves administering the diabetogenic agent streptozotocin in combination with a high-fat or high-carbohydrate diet.¹¹ Streptozotocin is selectively taken up by pancreatic beta cells via the glucose transporter GLUT2, leading to their partial destruction. Consumption of a diet rich in fats and carbohydrates contributes to obesity, elevated insulin levels, insulin resistance, and impaired glucose tolerance.12
The proportion of fats, proteins, and carbohydrates in the diet of laboratory animals, along with the duration of feeding, influences body weight as well as baseline levels of glucose, insulin, triglycerides, cholesterol, and free fatty acids in plasma.13 Considerable epidemiological evidence indicates that polyphenols can modulate and prevent metabolic disturbances associated with type 2 diabetes.14
The aim of the present study was to evaluate the effects of polyphenolic compounds derived from Pistacia vera (compound ANK-1), Karelinia caspia (compound KCL-1), and an extract from Isatis tinctoria L. (compound ITL-2) on type 2 diabetes mellitus in a rat model induced by a high-fat diet and a single dose of streptozotocin.
Materials and Methods
Experiments were performed on 36 white mongrel male rats weighing 280–340 g (n = 6 per group; total n = 36). The animals were maintained under standard vivarium conditions with natural light and free access to water and food.
All experimental procedures were conducted on outbred white male rats maintained on a standard vivarium diet, in full compliance with the requirements of the World Society for the Protection of Animals and the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (1986), as well as the American Psychological Association (2017) Ethical Principles of Psychologists and Code of Conduct (2002; amendments effective June 2010 and January 2017).
A model of type 2 diabetes mellitus was induced by feeding animals a high-fat diet in combination with a single intraperitoneal injection of streptozotocin. On the 61st day of the experiment, after a 12-hour fasting period, a freshly prepared streptozotocin solution (35 mg/kg, 0.1 M citrate buffer, pH 4.5) was administered intraperitoneally. Following injection, animals were provided with unrestricted access to food and water to prevent hypoglycemic coma. Under the influence of streptozotocin, rats developed acute necrosis of pancreatic β-cells, depletion of insulin, and severe hyperglycemia within 24–72 hours, which persisted for an extended period.12
The studied compounds were administered orally three times before and five times after the injection of streptozotocin at a dose of 10 mg/kg. The control group of animals (n = 6) received 0.3 ml of physiological saline.
On days 1 and 5 after priming, blood was collected dropwise from the conjunctival plexus into Eppendorf tubes. For coagulological assays, 50 µl of non-stabilized blood was used for antithrombotic testing, and 0.5 ml was collected with sodium citrate (1:9), mixed thoroughly, and centrifuged at 3,000 × g for 10 minutes (rotor OPN-8, centrifuge RU180 L2000 gy).
The following coagulogram parameters were evaluated: prothrombin time (PT, s), activated partial thromboplastin time (APTT, s), plasma recalcification time (PR, s), and fibrinogen concentration (F, mg/dL). Measurements were performed using a CYANCoag coagulometer (Belgium, model CY003, SN 5400439)15 and Cypress Diagnostics kits (Germany).
To assess antithrombotic activity, fresh blood clots (50 µl) were incubated with 0.5 ml of physiological saline at 37 °C for 1 hour (UF-View spectrophotometr UV-5100). The supernatant was analyzed spectrophotometrically at 410 nm (22–24 °C). Clot inhibition or induction (SIS, %) was calculated using the following formula:

Biochemical parameters were determined by measuring glucose (mmol/L), total protein (g/dL), alanine aminotransferase (ALT, U/L), aspartate aminotransferase (AST, U/L), total cholesterol (mmol/L), and triglycerides (mmol/L) in serum using a CYANSmart semi-automatic biochemical analyzer (Belgium) with diagnostic test kits from Cypress Diagnostics.
Polyphenolic compounds used in this study included ANK-1 isolated from Pistacia vera (C₄₈H₃₆O₃₀, M = 1092), KCL-1 isolated from Karelinia caspia (C₁₉H₂₀O₉, M = 392), and ITL-2, an extract from Isatis tinctoria L. The reference drug used was Diaglizide® MR (30 mg tablets; Farmak, Ukraine).
ANK-1 is a pale-yellow amorphous powder (Rf 0.4242, 1-system) with melting point 298–301 °C (decomposition). UV spectrum (MeOH, λmax nm): 230, 279. ESI-MS (m/z 1092 [M–H]⁻; fragments 939, 787, 769, 635, 617, 483, 465, 447, 431, 331, 295, 169). ¹H NMR (CD₃OD): δ 6.28 (d, 8.3 Hz, H-1), 5.58 (d, 8.3 Hz, H-2), 5.98 (m, 9.6 Hz, H-3), 5.62 (m, 9.6 Hz, H-4), 5.58 (dd, 9.6, 8.3 Hz, H-2), 4.54 (m, H-5), 4.45 (d, 12.2 Hz, H-6), 4.25 (dd, 12.2, 4.28 Hz, H-6). ¹³C NMR (CD₃OD): 93.9 (C-1), 72.5 (C-2), 74.5 (C-3), 70.1 (C-4), 74.5 (C-5), 63.8 (C-6); galloyl carbons 166.3–167.3 (C-7), 120.3–121.1 (C-8), 110.4–111.0 (C-9), 146.4–147.5 (C-10), 140.3–140.8 (C-11).
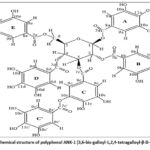 |
Figure 1: Chemical structure of polyphenol ANK-1 (3,6-bis-galloyl-1,2,4-tetragalloyl-β-D-glucose).17 |
Hydrolysis with 5 % HCl produced glucose and gallic acid. Comparison with literature data confirmed the structure as 6-O-bisgalloyl-1,2,3,4-tetra-O-galloyl-β-D-glucose.17
KCL-1: Dried leaves of Karelinia caspia (1 kg) were crushed and extracted with EtOH–H₂O (7:3; 3 L × 3 times, 2 h each). The 9 L extract was evaporated, yielding 2.6 L of aqueous fraction, which was partitioned with EtOAc. The EtOAc fraction was chromatographed on 70 g of silica gel using a CHCl₃–MeOH gradient of increasing polarity. Seven fractions (1.1, 2.9, 1.7, 3.3, 4.5, 6.1, and 45 g) were collected and pooled based on TLC analysis. Fraction 7 (45 g) was evaporated in vacuo and further purified on silica gel (CH₂Cl₂–EtOAc 7:3).
Iriflophenone 2-O-rhamnoside: white-yellow amorphous powder; m/z 391.1019 [M–H]⁻ (C₁₉H₁₉O₉). ¹H NMR (600 MHz, DMSO-d₆): δ 9.80 (4H, OH), 5.81 (2H, H-3, H-5), 7.53 (2H, J = 8.7 Hz, H-2′, H-6′), 6.76 (2H, J = 8.7 Hz, H-3′, H-5′); ¹³C NMR (MeOD): 17.9 (rha-6), 70.7 (rha-5), 71.4 (rha-2), 71.7 (rha-3), 73.5 (rha-4), 100.2 (rha-1), 95.5 (C-3), 97.9 (C-5), 109.2 (C-1), 115.9 (C-3′, C-5′), 132.6 (C-1′), 132.8 (C-2′), 158.3 (C-6), 158.4 (C-2), 163.0 (C-4, C-4′), 197.6 (C-7).
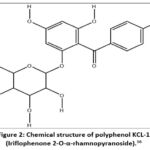 |
Figure 2: Chemical structure of polyphenol KCL-1 (Iriflophenone 2-O-α-rhamnopyranoside).16 |
Statistical analysis
Experimental data were processed using Origin 7.1 (Microsoft, USA). For independent samples following a normal distribution, Student’s t-test was applied, and differences were considered significant at p < 0.05. Histograms and tables present arithmetic means ± standard errors from 3–5 independent experiments.
Results
It has been demonstrated that in rodents, a single high dose of streptozotocin, in addition to the destruction of pancreatic β-cells, can also cause non-specific alterations in α-cells responsible for glucagon synthesis. In terms of hormonal and metabolic changes, low-dose streptozotocin-induced diabetes closely resembles type 2 diabetes in humans.18
Biochemical parameters, particularly glucose levels in the blood of rats on the first day after a single intraperitoneal administration of streptozotocin, confirm the successful establishment of a type 2 diabetes model (Graph 1).
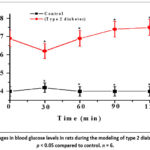 |
Figure 3: Changes in blood glucose levels in rats during the modeling of type 2 diabetes mellitus. p < 0.05 compared to control. n = 6. |
As shown in Table 1, following intraperitoneal administration of streptozotocin in the control group of animals at 1, 2, 3, 24, and 72 hours, and after 5 days of observation, there was a consistent increase in serum glucose levels by 1.72-, 2.48-, 2.25-, 2.97-, 2.72-, 2.2-, and 1.9-fold (p ≤ 0.01) relative to baseline values (4.0 ± 0.27 mmol/L). (Fig. 3).
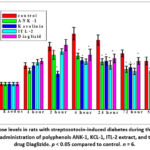 |
Figure 4: Glucose levels in rats with streptozotocin-induced diabetes during therapeutic and prophylactic administration of polyphenols ANK-1, KCL-1, ITL-2 extract, and the reference drug Diaglizide. p < 0.05 compared to control. n = 6. |
Preliminary (three-fold) administration of the studied polyphenolic compounds significantly attenuated the post-streptozotocin increase in blood glucose compared with the control group. The maximum rise in serum glucose (by 66% and 63%) in control animals was observed 4–24 hours after streptozotocin administration (11.9 ± 1.0 and 10.9 ± 0.8 mmol/L). In contrast, the rise in glucose levels was markedly lower in treated groups:
ANK-1: decreased by 29.4% and 49.5%,
KCL-1: decreased by 35% and 13%,
ITL-2: decreased by 36% and 37%,
Diaglizide (reference): decreased by 28% and 18% relative to control.
By the 5th day of the experiment, serum glucose levels in animals treated with ITL-2 were comparable to baseline values, while those in the ANK-1, KCL-1, and Diaglizide groups were significantly lower than in the control group.
Analysis of biochemical parameters in the control group (Table 1) revealed that aspartate aminotransferase (AST) and alanine aminotransferase (ALT) increased by 1.6-fold (p ≤ 0.01) and 3.1-fold (p ≤ 0.01), respectively, compared with intact animals (31.5 ± 4.2 and 39.2 ± 2.1 U/L). Other biochemical indices showed no significant changes.
After 5 days, the same parameters in the control group remained elevated by 1.3-fold (p < 0.01) and 2.1-fold (p < 0.01) relative to the intact group.
Therapeutic and prophylactic administration of ANK-1, KCL-1, and ITL-2 effectively neutralized the toxic influence of streptozotocin on AST and ALT activity. Moreover, glucose concentrations in rats treated with ANK-1 and ITL-2 did not differ significantly from those in intact controls.
Table 1: Biochemical parameters of blood in rats with diabetes under therapeutic and prophylactic use of ANK-1, KCL-1 polyphenols, ITL-2 extract, and Diaglizide (M ± m, n = 6).
| Preparations | Total proteing/l | ALT,U/l | AST,U/l | Glucose,mmol/ l | Totalcholesterol, mmol/l | Triglyceridesmmol/ l |
| Control | 65.4±2.8 | 31.5±4.2 | 39.2±2,1 | 3.1±0.12 | 1.8±0.08 | 0.87±0.02 |
| T2DM days | 64±1.3 | 97.3±6.8* | 64.1±5.2* | 10.9±0.8* | 2.1±0.12* | 1.11±0.1* |
| ANK-1 | 65.0±2.1 | 41.5±4.2** | 48.2±2.8** | 6.4±0.4** | 1.9±0.11 | 0.89±0.07 |
| KCL-1 | 65.4±2.8 | 31.5±4.2** | 49.2±2.1** | 7.6±0.67** | 1.7±0.10 | 0.88±0.01 |
| ITL-2 | 65.4±2.8 | 46.7±3.2** | 40.8±3,2** | 7.0±0.57** | 1.8±0.08 | 0.97±0.03 |
| Diaglizid | 64.1±1.1 | 45.2±3.4** | 41.6±2.8** | 8.0±0.67** | 1.7±0.07 | 0.98±0.02 |
| After 5 days of treatment | ||||||
| T2DM | 64.4±2.0 | 66.8±2.2* | 52.5±4.1* | 7.6±0.63* | 2.0±0.10 | 1.12±0.05 |
| ANK-1 | 65.0±2.1 | 41.5±4.2** | 48.2±2.8* | 4.0±0.41** | 1.9±0.11 | 0.89±0.07 |
| KCL-1 | 65.1±2,1 | 40.8±4.2** | 28.3±2.8** | 6.0±0.47** | 1.9±0.11 | 0.87±0.07 |
| ITL-2 | 24.7±2.2 | 28.3±3.2** | 40.8±2.4** | 4.0±0.32** | 1.68±0.12 | 0.86±0.06 |
| Diaglizide | 24.3±2,6 | 48.1±3.5** | 36.2±2.1** | 7.0±0.47** | 1.85±0.14 | 0.88±0.05 |
Notes: p < 0.01 relative to intact animals; p < 0.01 relative to control animals.
As shown by coagulogram data in Table 2, streptozotocin in the control group induced hypercoagulation, expressed as a significant reduction in prothrombin time (PT) by 30% (p = 0.0001) one day after administration, relative to baseline (22.0 ± 1.4 s). Activated partial thromboplastin time (APTT) did not differ significantly from the initial values.
Diabetes-associated hemostatic changes include elevated coagulation proteins — factors VIII/vWF, VII, X, and fibrinogen — which contribute to prothrombotic states. Concurrently, the inhibition system (protein C, antithrombin III) and fibrinolytic activity are suppressed, resulting in elevated plasminogen activator inhibitor (PAI-1).19
Plasma recalcification time (PR), which reflects internal prothrombinase formation and platelet activity, decreased by 35.5% (p = 0.0001), while fibrinogen concentration increased by 96.7% (p = 0.00001) (325.8 ± 24 mg/dL).
Administration of ANK-1 caused a statistically significant prolongation in coagulation parameters:
PT increased by 52% (p = 0.00941) vs. control (12.9 ± 1.15 s);
APTT extended by 84% (p = 0.00103) vs. control (21.7 ± 1.9 s);
PR increased by 237% (p = 0.00016) vs. control (27.6 ± 2.3 s);
Fibrinogen decreased by 70% (p = 0.000511) vs. control (625.0 ± 35 mg/dL).
Treatment with KCL-1 also significantly improved coagulation parameters: PT increased by 196.4% (p = 0.01), APTT by 33.8% (p = 0.001), and PR by 58.3% (p = 0.0001) compared with control. Fibrinogen decreased by 16% (p = 0.05).
Administration of ITL-2 led to an increase in PT by 35.5% (p = 0.01), a decrease in APTT by 38.8% (p = 0.001), and an increase in PR by 242.7% (p = 0.0001). Fibrinogen decreased slightly by 5% (p > 0.05).
The reference drug Diaglizide resulted in increased PT by 51.0% (p = 0.01) and PR by 126.6% (p = 0.0001), while APTT showed no significant difference from control; fibrinogen decreased by 20.0% (p < 0.05).
After five days, PT, APTT, PR, and fibrinogen levels in control animals remained unchanged relative to day 1. In treated groups, PT increased 2.4-fold with ANK-1 and KCL-1 (p = 0.01), 2.1-fold with ITL-2, and 2.8-fold with Diaglizide. APTT increased 1.6-fold only in the KCL-1 group.
As known, APTT reflects deficiencies in intrinsic pathway factors (XII, XI, IX, VIII, X, V, II), while PT evaluates the extrinsic pathway (VII, X, V, II).12-15 Changes in Ca2+ concentration significantly affect the activity of ionotropic receptors, presynaptic Ca²⁺ channels, platelet α₂-adrenergic receptors, P2Y12 receptors, and GPIIb/IIIa (αIIbβ3) integrins — all of which influence coagulation and platelet aggregation.18
The maximum PR increase relative to baseline and control was 3.5-2.1-fold. Fibrinogen content in experimental groups remained close to initial levels, except in the Diaglizide group, where it was 51% (p = 0.01) higher than baseline.
Table 2: Coagulogram parameters in rats with streptozotocin-induced diabetes under therapeutic and prophylactic use of ANK-1, KCL-1 polyphenols, ITL-2 extract, and Diaglizide (M ± m, n = 6).
| Preparations | Coagulogram | |||
| parameters Prothrombin time (PT), sec. | (APTT) sec. | Secrecalcification Plasma recalcification, sec. | Fibrinogen, mg/dl | |
| Control | 22.0±1.4 | 40.6±3.0 | 56.4±2.5 | 325.8±24 |
| T2DM | 15.5±1.2* | 39.3±2.4 | 37.5±2.5* | 641.0±39* |
| ANK-1 | 1 23.7±2.1 | 34.5±2.8 | 64.3±5.6 | 525.0±35 |
| KCL-1 | 65.2±5.4* ** | 52.6±2.5* | 89.3±6.6* ** | 537.3±24* |
| ITL-2 | 36.5±1.3* ** | 24.4±2.0* ** | 128.5±10.5* ** | 608.0±34 * |
| Diaglizid | 33.2±2.4* ** | 41.3±3.0 | 127.8±10.2 | 511.0±24 |
| After 5-fold treatment | ||||
| T2DM | 12.1±1.1* | 38.7±2.9* | 33.0±2.3 * | 512.5±35* |
| ANK-1 | 19.6±1.5 | 39.9±2.8 | 101.4±9.5 | 367.8±24 |
| KCL-1 | 29.4±2.0* ** | 60.3±4.9* ** | 76.5±2.3* ** | 319.5±25** |
| ITL-2 | 26.0±1.7* ** | 38.2±2.5 | 80.5±2.6* ** | 411.5±34** |
| Diaglizid | 34.0±2.5* ** | 35.5±1.4 | 117.5±3.3* ** | 492.0±24* ** |
Note: APTT — activated partial thromboplastin time; *p < 0.001 in relation to the intact group of animals; *p < 0.005 in relation to the control group of animals.
The antithrombotic activity of the investigated compounds was evaluated by measuring degradation of 50 µl of clotted blood (Table 3). One day after streptozotocin administration, the control group showed a 44% reduction in color intensity (p < 0.001) compared to intact animals (0.640 ± 0.05). Conversely, all treated groups exhibited a 117.5% increase (p < 0.001) relative to the control (0.360 ± 0.03).
In control rats, clot mass increased by 74% (p < 0.001) compared with intact animals (16.2 ± 1.0 mg). Treatment with ANK-1, KCL-1, ITL-2, and Diaglizide inhibited clot formation by 38–55% (p < 0.001) relative to control (28.2 ± 1.8 mg).
Table 3: Antithrombotic activity after therapeutic and prophylactic administration of ANK-1, Karelinia (KCL-1), ITL-2 and the reference drug Diaglizide in rats with diabetes (M ± m, n = 6)
| Tests | Control | T2DM | ANK-1 | KCL-110 mg/kg | ITL-210 mg/kg | Diaglizide10 mg/kg |
| TimeStudy time, | After 1 day | |||||
| ExtinctionPlasma extinction 410 nm OE | 0.640±0,05 | 0.360±0,03 | 0.770±0.05** | 0.783±0.03** | 0.747±0.04** | |
| AA, O.E. | *-1.46 | +2.1.1 | +2.1 | 2.1 | ||
| Weight of the clot. mg | 16.2±1.0 | 28.2±1.8 | 14.1±0.7** | 17.4±1.0** | 12.8±0.6** | |
| SIS, % | +74.1 | -50.0 | -38.3 | -54.6 | ||
| TimeStudy time, | 5 days | |||||
| ExtinctionAfter Plasma extinction 410nm | 0.64±0.05 | 0.46±0.04 | 0.67±0.05· ** | 0.67±0.05** | 0.683±0.03* * | 0.647±0.04** |
| AA, O. E. | *-1.39 | +31.3** | +1.45 | +1.48 | 1.4 | |
| Weight of the clot. mg | 16.2±1.0 | 20.2±1.5 | 14.1±0.7** | 16.1±0.7** | 17.4±1.0** | 16.8±0.6** |
| SIS, % | +24.7 | **-22.5 | -20.3 | -13.9 | -16.8 | |
Notes: *p < 0.05 relative to the intact group; *p < 0.05 relative to the control group.
Discussion
The results obtained convincingly confirm the hypoglycemic and antithrombotic effects of the investigated polyphenolic agents in the streptozotocin-induced hyperglycemia model. Streptozotocin administration is known to cause selective β-cell destruction and a decline in insulin secretion, resulting in a rapid increase in blood glucose concentration. In the control group of animals, glucose levels increased by 63–66% within 4–24 hours after administration, demonstrating the activation of these pathological mechanisms. Against the background of treatment with the studied agents, the hyperglycemic response was markedly attenuated.
Significant reductions in serum glucose levels were observed in the ANK-1 and ITL-2 groups compared with the control. A similar but less pronounced effect was recorded for KCL-1 and the reference drug Diaglizide, indicating differences in hypoglycemic efficacy. Of particular interest are the findings in the ITL-2 group: by day 5 of the experiment, blood glucose concentrations did not differ from baseline values, indicating that ITL-2 not only suppresses hyperglycemia but also contributes to stabilization of carbohydrate metabolism under diabetic conditions. The remaining agents also significantly lowered glucose levels, although complete normoglycemia was not achieved.
These findings confirm the antidiabetic potential of the investigated polyphenolic compounds. Their administration prevents pronounced hyperglycemia, mitigates β-cell injury, and supports the partial normalization of metabolic disturbances associated with diabetes mellitus.
The observed improvements in carbohydrate metabolism under ANK-1, KCL-1, and ITL-2 treatment were accompanied by changes in coagulation parameters. In particular, an increase in activated partial thromboplastin time (APTT) indicated modulation of the intrinsic coagulation pathway factors (XII, XI, IX, VIII, X, V, and II). Similarly, alterations in prothrombin time (PT) reflected effects on extrinsic pathway factors (VII, X, V, and II).
Elevation of PT by 2.1–3.5-fold indicated normalization of coagulation dynamics, while fibrinogen concentration remained close to initial levels, suggesting prevention of hyperfibrinogenemia. Evaluation of antithrombotic activity further supported these observations. In the control group, a 44% reduction in blood staining and a 74% increase in thrombus weight were recorded, demonstrating enhanced thrombogenic potential and hemostatic imbalance typical of diabetic conditions.
In contrast, rats treated with the polyphenolic compounds exhibited a 117.5% increase in blood staining and a 38–55% reduction in thrombus weight relative to control animals. These findings indicate a pronounced antithrombotic effect, reflected by suppression of hypercoagulability, inhibition of thrombus formation, and partial restoration of the hemostatic system balance.
Overall, the results suggest that ANK-1, KCL-1, and ITL-2 stabilize hemostatic function and significantly reduce thrombogenic risk in experimental diabetes. This highlights their therapeutic promise and potential utility in preventing diabetes-associated thromboembolic complications.
Study Limitations
The present study was conducted on a limited number of experimental animals (n = 6 per group), which may restrict the statistical power to detect minor intergroup differences. Furthermore, only a single experimental model—streptozotocin-induced type 2 diabetes—was utilized. Thus, the findings may not capture the complete pathophysiological complexity of human diabetes.
Future studies should include larger sample sizes, multiple diabetes models, and extended observation periods to evaluate long-term metabolic and hemostatic outcomes. Molecular and histological investigations are also necessary to elucidate the intracellular mechanisms underlying β-cell protection and coagulation regulation by polyphenolic compounds.
Future Scope
Further studies should explore the molecular mechanisms and pharmacokinetics of these polyphenols and assess their potential application in clinical models of diabetes-associated vascular and thrombotic complications.
Conclusion
In rats with type II diabetes mellitus, streptozotocin administration induced hypercoagulation via the external (prothrombin time) pathway of blood clotting, resulting in a 30–40% reduction in prothrombin time and a 60–97% increase in fibrinogen concentration. In the control group, biochemical parameters such as AST, ALT, and glucose were significantly elevated throughout the experiment.
All investigated agents, administered orally at doses of 10–15 mg/kg, effectively normalized coagulation activity under both therapeutic and prophylactic conditions. The most pronounced antithrombotic effect was observed with the polyphenols ANK-1, KCL-1, and ITL-2, where antithrombotic activity increased 2.1-fold and clot inhibition reached 50.5% and 38.7%, respectively.
Therapeutic and prophylactic use of polyphenols ANK-1, KCL-1, and ITL-2 effectively prevented the toxic influence of streptozotocin on hepatic enzymes (AST and ALT), while glucose concentrations in the ANK-1 and ITL-2 groups did not differ from intact controls.
Overall, the studied polyphenols demonstrated comparable or superior efficacy to the reference drug Diaglizide, confirming their potential as effective antidiabetic and antithrombotic agents in experimental type II diabetes mellitus.
Acknowledgment
This work was supported by the Science and Technology Development Coordination Committee.
Funding Sources
This work was financed by the grant АL-27-4722022401 of the Science and Technology Development Coordination Committee under the Cabinet of Ministers of the Republic of Uzbekistan.
Conflict of Interest
The author(s) do not have any conflict of interest
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The experimental protocols complied with the standards and requirements for the humane treatment of animals and the provisions of the Ethical Commission of the IBB at the National University of Uzbekistan. (Protocol No. 7 BEC/IBB-NUU of 04/07/2022) on the use of laboratory animals. Preparations of isolated aortic segments were obtained using a known method. This study did not involve human participants, and therefore, informed consent was not required.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical trial registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable.
Authors’ Contribution
- Guli Raimova: Conceptualization, Methodology, Writing – review & editing, Writing – Original Draft, Supervision, Methodology, Analysis, Writing – review & editing, Writing – original draft, Validation, Formal analysis
- Kabil Nasirov: Conceptualization, Methodology, Writing – review & editing, Supervision, Resources, Funding acquisition, Project Administration.
- Siroj Khodzhiev; Formal analysis, Data Collection, Project Administration.
- Alisher Mukhtorov: Visualization, Resources
- Nozim Hoshimov: Visualization, Resources, Writing – Review & Editing
- Mukhamatkodir Ortikov: Data Collection, Analysis
- Rustam Maxmudov and Gaуbullo Lutpillaеv: Isolation of polyphenol ANK-1
- Zamira Teshabayeva Resources; Investigation; Formal analysis
- Rano Nosirova Resources and Zulfizar Tuychiyeva: Writing – Review & Editing
- Sabina Normurodova and Akbarova Gulbakhor: Data curation
References
- Trubitsyna N.P., Zaitseva N.V., Severina A.S., et al. Chronic kidney disease in patients with type 2 diabetes mellitus: new drug targets. Diabetes Mellitus. 2022; 25: 492–498.
CrossRef - Ametov A.S., Solov’eva O.L. Disorders in the hemostatic system in diabetes mellitus and ways to correct them when prescribing combined therapy with Diabeton MV and metformin. Diabetes Mellitus. 2007; 10: 33–39.
CrossRef - Zhao S., Chu Y., Zhang C., et al. Diet-induced central obesity and insulin resistance in rabbits. J Anim Physiol Anim Nutr (Berl). 2008; 92: 105–111.
CrossRef - Liu Y., Wang Z.B., Yin W.D., et al. Preventive effect of Ibrolipim on suppressing lipid accumulation and increasing lipoprotein lipase in the kidneys of diet-induced diabetic minipigs. Methods Mol Biol. 2011; 10: 117.
CrossRef - Arias-Mutis O.J., Marrachelli V.G., Ruiz-Saurí A., et al. Development and characterization of an experimental model of diet-induced metabolic syndrome in rabbits. PLoS One. 2017; 18.
CrossRef - Avramoglu K.R., Laplante M.A., Le Q.K., et al. The genetic and metabolic determinants of cardiovascular complications in type 2 diabetes: recent insights from animal models and clinical investigations. Can J Diabetes. 2013; 37: 351–358.
CrossRef - Shestakova M.V., Sukhareva O.Yu. Type 2 diabetes mellitus: ease of diagnosis and choice of treatment. Endocrinological. 2012;12: 5–9.
- Poudyal H., Panchal S., Brown L. Comparison of purple carrot juice and β-carotene in a high-carbohydrate, high-fat diet-fed rat model of the metabolic syndrome. Br J Nutr. 2010; 104(9): 1322–1332.
CrossRef - Tominaga A., Ishizaki N., Naruse Y., et al. Repeated application of low-frequency electroacupuncture improves high-fructose diet-induced insulin resistance in rats. Acupunct Med. 2011; 29: 276–283.
CrossRef - Kaidash O.A., Ivanov V.V., Vengerovsky A.I., et al. An experimental model of type 2 diabetes mellitus in rats caused by a high-fat diet and low-dose streptozotocin. Bull Siberian Med. 2020; 19: 41–47.
CrossRef - Raimova G., Nasirov K., Khodjiyev S., et al. ANK-1, ANK-2, ITL-2 polyphenols in a dexamethasone-induced rat model of type 2 diabetes mellitus. Treatment J Angiother. 2024; 8: 1–11.
CrossRef - Cao H., Ou J., Chen L., et al. Dietary polyphenols and type 2 diabetes: human studies and clinical trials. Crit Rev Food Sci Nutr. 2018; 21: 356–363.
- Khoshimov N., Raimova G., Nasirov K., et al. The effect of sulphated cellulose on the system of haemostasis. Res J Pharm Technol. 2021; 14: 3283–3289.
CrossRef - Raimova G., Khoshimov N., Nasirov K., et al. Anti-thrombotic action of sulfated polysaccharides on thrombosis caused by thromboplastin. Res J Pharm Technol. 2021; 14: 6085–6088.
CrossRef - Lutpillayev G’, Rahimov R. Flavonoids of the Karelinia caspia plant. Acad Res Educ Sci. 2024; 2181–1385.
- Djuraev I.U., Karimov I.B., Nasirov K.N., Nurmatov K.E., et al. The influence of polyphenols on calcium dynamics in synaptosomes of model rats with attention-deficit hyperactivity disorder of varying ages. Trends Sci. 2025; 22: 10434.
CrossRef - Khoshimov N., Mukhtorov A., Nasirov K., Rakhimov R., Mamadaminov R. Effects of polyphenols on changes in the transport of Ca²⁺ NMDA receptors under the influence of L-glutamate. Res J Pharm Technol. 2023; 16: 1205–1209.
CrossRef - Nasirov K.N., Karimov I.B., Djuraev I.U., et al. Effect of polyphenols on changes in the hemostatic system of blood plasma in healthy and model rats with Alzheimer’s disease. Trends Sci. 2024; 21: 8081.
- Ugli, D. J. I., Bakhtiyarovich, K. I., et al. The Influence of Polyphenols on Calcium Dynamics in Synaptosomes of Model Rats with Attention Deficit Hyperactivity Disorder of Varying Ages. Trends in Sciences, 2025; 22:10434.
CrossRef







