Manuscript accepted on :27-10-2025
Published online on: 03-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Md. Sarwar Hossain
Second Review by: Dr. Samara Sameer
Final Approval by: Dr. Mariia Shanaida
Iroda Ganieva* , Ilyos Xudoynazarov, Alisher Khaitbaev
, Ilyos Xudoynazarov, Alisher Khaitbaev , Muzaffar Shokirov
, Muzaffar Shokirov , Mirjalol Jalilov
, Mirjalol Jalilov , Dilshod Mansurov
, Dilshod Mansurov , Mukhammadjon Mustafakulov and Iqboljon Yuldashev
, Mukhammadjon Mustafakulov and Iqboljon Yuldashev
Department of Natural compounds and applied Chemistry, National University of Uzbekistan, Tashkent, Uzbekistan.
Corresponding author’s e-mail: irodaganieva.123.a@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3327
Abstract
Rosmarinus officinalis L. (rosemary) is a medicinal and aromatic plant traditionally valued for its antioxidant, antimicrobial, and anti-inflammatory activities, largely due to its terpenoid-rich essential oils. In this study, 1,8-cineole was successfully isolated in pure form from rosemary cultivated in Uzbekistan using solvent extraction followed by vacuum fractional distillation, and its identity was confirmed by gas chromatography–mass spectrometry (GC–MS). Toxicological investigations demonstrated that oral administration of 1,8-cineole produced no adverse effects at doses of 200–400 mg/kg, while higher concentrations caused dose-dependent toxicity, with an LD₅₀ value of 680 mg/kg, classifying the compound as a low-toxic substance. Sub-chronic studies revealed no significant alterations in body weight, behavior, renal function, or hematological parameters, indicating good tolerance at repeated low doses. Complementary in silico ADMET predictions further confirmed favorable pharmacokinetic properties, including high gastrointestinal absorption, strong blood–brain barrier permeability, moderate clearance, and compliance with major drug-likeness rules, with minimal predicted toxicity. Additionally, molecular docking analyses demonstrated that 1,8-cineole interacts with key inflammatory mediators—COX-2 (−6.2 kcal/mol), IL-6 (−5.2 kcal/mol), MAPK (−5.3 kcal/mol), NF-κB (−5.2 kcal/mol), and TNF-α (−6.8 kcal/mol)—with the strongest affinities observed for COX-2 and TNF-α. These multitarget interactions suggest potential modulation of prostaglandin synthesis and cytokine signaling. Taken together, these findings highlight 1,8-cineole as a pharmacologically safe and promising bioactive compound, underscoring its potential for therapeutic and industrial applications.
Keywords
ADMET; GC–MS; pharmacokinetics; Rosmarinus officinalis; 1,8-cineole
Download this article as:| Copy the following to cite this article: Ganieva I, Xudoynazarov I, Khaitbaev A, Shokirov M, Jalilov M, Mansurov D, Mustafakulov M, Yuldashev I. In silico Anti-Inflammatory Potential, ADMET Pharmacokinetics and Isolation of 1,8-Cineole from Rosmarinus officinalis. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Ganieva I, Xudoynazarov I, Khaitbaev A, Shokirov M, Jalilov M, Mansurov D, Mustafakulov M, Yuldashev I. In silico Anti-Inflammatory Potential, ADMET Pharmacokinetics and Isolation of 1,8-Cineole from Rosmarinus officinalis. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4pEKYPo |
Introduction
Rosmarinus officinalis L. (rosemary) is a perennial aromatic and medicinal herb belonging to the family Lamiaceae. For centuries, it has been widely used in folk medicine, culinary traditions, and as a natural preservative due to its remarkable pharmacological potential. Traditionally, rosemary has been employed as a remedy for ailments such as respiratory disorders, digestive problems, headaches, and inflammation. Its therapeutic importance is largely associated with its rich phytochemical composition, particularly essential oils, phenolic diterpenes, flavonoids, and terpenoids, which collectively contribute to its strong antioxidant, antimicrobial, anti-inflammatory, and neuroprotective properties.1,2
The essential oil of R. officinalis has been extensively studied across different regions of the world, and its major bioactive constituents are consistently reported to include eucalyptol (1,8-cineole), camphor, α-pinene, and borneol. Among these, 1,8-cineole is often found as the predominant compound, responsible for many of rosemary’s characteristic aroma and pharmacological effects. Despite numerous investigations globally, the phytochemical profile of rosemary cultivated in Uzbekistan remains underexplored. To date, there is no scientific evidence regarding the isolation of pure terpenoid compounds such as 1,8-cineole from rosemary grown under the specific agro-climatic conditions of Uzbekistan.3,4
The qualitative and quantitative composition of secondary metabolites in medicinal plants can vary significantly depending on geographical origin, climate, soil properties, altitude, and other ecological factors. Such variations not only influence the concentration of major constituents but may also lead to the presence of unique minor compounds with distinct biological activities. Therefore, phytochemical investigations of plants grown in diverse regions are essential, as they contribute both to the understanding of plant metabolic diversity and to the discovery of novel bioactive molecules with potential applications in medicine, food, and cosmetics.5,6
1,8-Cineole (also known as eucalyptol) is a naturally occurring monoterpenoid and one of the most important constituents of essential oils from various aromatic plants. Pharmacologically, it exhibits a broad spectrum of biological activities, including antimicrobial, expectorant, anti-inflammatory, bronchodilatory, and antioxidant effects. It is widely used as an active ingredient in pharmaceutical formulations for the treatment of respiratory tract diseases, such as asthma, bronchitis, and sinusitis. Furthermore, 1,8-cineole has demonstrated significant potential as a natural preservative in the food industry, as well as an aroma compound in perfumery and cosmetics. Its industrial importance is further highlighted by its high abundance in Eucalyptus globulus leaves (70–90%), as well as in Rosmarinus officinalis, Laurus nobilis, and Salvia officinalis.7,8
Given its broad pharmacological profile, 1,8-cineole has been increasingly investigated for new therapeutic applications, including neuroprotection, cardiovascular support, and synergistic antimicrobial effects in combination with conventional antibiotics. At the same time, studies on its absorption, distribution, metabolism, excretion, and toxicity (ADMET) properties have underscored its favorable pharmacokinetic profile, making it a promising lead compound for drug development.9,10
In this context, the present study was designed to isolate 1,8-cineole from R. officinalis cultivated in Uzbekistan and to characterize it using gas chromatography–mass spectrometry (GC–MS). In addition, the biological potential of the isolated compound was evaluated through acute toxicity studies in vivo, as well as ADMET and pharmacokinetic analyses in silico. To the best of our knowledge, this is the first report on the is olation and biological characterization of pure 1,8-cineole from rosemary grown in Uzbekistan. The results of this work not only expand our understanding of the phytochemical composition of R. officinalis under local environmental conditions but also highlight the potential of this compound as a bioactive agent with diverse pharmacological applications.11,12
Materials and Methods
Plant Material Preparation
Fresh aerial parts of cultivated plant Rosmarinus officinalis L. were collected in May 2025 from the Bostonliq district of Tashkent region, Republic of Uzbekistan (Figure 1). The plant material was authenticated by a botanist, and voucher specimens were deposited for reference. The collected aerial parts were air-dried under shade at a stable temperature not exceeding 30 °C for 10 days. After drying, the leaves were carefully separated from the stems and directly used for extraction.13,14 The average natural leaf length was approximately 2–4 cm.
 |
Figure 1: Rosmarinus officinalis L. (rosemary) plant. |
Extraction Procedure
For the extraction, 20 g of air-dried rosemary leaves were accurately weighed using an analytical balance and transferred into a 500 ml round-bottom flask. The plant material was macerated in acetone at a ratio of 1:10 (w/v), corresponding to 200 ml of solvent. The flask was fitted with a reflux condenser and placed in a water bath at 70–80 °C for 3.5 h.
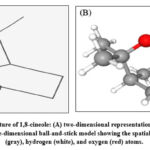 |
Figure 2: Chemical structure of 1,8-cineole: (A) two-dimensional representation highlighting the oxygen atom (red), and (B) three-dimensional ball-and-stick model showing the spatial arrangement of carbon (gray), hydrogen (white), and oxygen (red) atoms. |
After extraction, the resulting solution was concentrated using a rotary evaporator (Rotavapor) under reduced pressure at 60 °C and 100 rpm for 45 min. The concentrated extract separated into two phases: a colorless liquid fraction and a viscous dark-colored fraction. The colorless liquid fraction was transferred into a 200 ml distillation flask connected to a fractional distillation column equipped with a thermometer, condenser, and receiving flask. The system was operated under vacuum conditions.15,16
Vacuum distillation was initiated once the pressure reached 30–50 mbar, and fractions of 1–3 ml were collected separately into pre-weighed flasks. Based on the known boiling point of 1,8-cineole under reduced pressure (50–60 °C), fractions collected within this temperature range were subjected to further analysis. The crude extract (primary extract) obtained from distillation was stored in airtight containers at 4 °C until subsequent purification and characterization steps.17,18
GC–MS Analysis
The fractions obtained within the boiling range of 50–60 °C were analyzed by gas chromatography–mass spectrometry (GC–MS) to confirm the presence and purity of 1,8-cineole. GC–MS analysis was performed using a Agilent 7890A GC system coupled with a 5975C inert XL MSD detector. Separation was achieved on a capillary column [HP-5MS, 30 m × 0.25 mm × 0.25 µm]. The oven temperature was initially set at [e.g., 50 °C for 2 min], then programmed to increase at a rate of 5 °C/min up to 250 °C, and held for 10 min. The carrier gas was helium at a constant flow rate of 1 ml/min (Figure 2A,B). The injection volume was 1 µl, with a split ratio of 1:20. The mass spectra were recorded in the range of 40–500 m/z. Identification of 1,8-cineole was confirmed by comparison of its retention time and fragmentation pattern with reference standards and the NIST mass spectral library.19,20,21
Acute Toxicity Study
Acute toxicity of the isolated 1,8-cineole was evaluated in vivo following OECD guidelines. Healthy laboratory mice/rats (specify species, strain, sex, and weight) were randomly divided into experimental groups (n = 6 per group). Animals were administered increasing doses of 1,8-cineole orally, and observed for 14 days for behavioral, physiological, and mortality changes. Clinical signs such as changes in respiration, locomotor activity, grooming, and food intake were monitored. The LD₅₀ value was estimated based on the mortality rate and dose–response relationship.22,23,24 Ethical approval for animal experiments was obtained from the Institutional Animal Care and Use Committee (IACUC).
Animal Ethics
All experimental procedures and preoperative care protocols were approved by the Institutional Animal Use Committee. Animals were maintained under standard vivarium conditions (humidity 55–65%, temperature 22 ± 2 °C) with free access to food and water. All experiments were conducted in compliance with the European Convention for the Protection of Animals Used for Scientific Purposes (1998) and the International Bioethical Guidelines of the Institute of Biophysics and Biochemistry, National University of Uzbekistan (approval code: BRC/IBB-N44/2024/75-1). Surgical interventions were performed under sodium pentobarbital anesthesia, and every effort was made to minimize animal suffering. Male albino Wistar rats weighing 180–220 g were used in the study.25
Statistical Analysis
Statistical data were obtained from 3–6 independent experiments (n = 3–6). A paired t-test was applied for the analysis of combined datasets, while unpaired t-tests were used for comparisons between independent groups. Statistical significance was determined at two levels: p < 0.05 and p < 0.01.
In Silico ADMET and Pharmacokinetic Analysis
The pharmacokinetic and ADMET (Absorption, Distribution, Metabolism, Excretion, and Toxicity) properties of 1,8-cineole were predicted using the ADMETlab 2.0 online platform. Parameters analyzed included water solubility, lipophilicity (logP), human intestinal absorption (HIA), blood–brain barrier (BBB) permeability, cytochrome P450 enzyme interactions, plasma protein binding (PPB), and drug-likeness indices (Lipinski’s rule of five). Toxicological endpoints such as hepatotoxicity, carcinogenicity, hERG inhibition, and Ames mutagenicity were also evaluated (Table 1). These computational analyses provided insights into the drug-likeness and safety profile of 1,8-cineole, complementing the experimental findings.26,27
Results
GC–MS Analysis
The chemical composition of the fractions obtained from Rosmarinus officinalis leaves was analyzed using gas chromatography–mass spectrometry (GC–MS). The total ion chromatogram (TIC) revealed a sharp and dominant peak at a retention time of 7.78 min (Figure 4). This peak indicated the presence of a single major constituent in the distilled fraction, suggesting a high degree of purity in the isolated compound.28,29
 |
Figure 3: Mass spectrum of the isolated fraction obtained from Rosmarinus officinalis showing the characteristic fragmentation pattern of 1,8-cineole. |
The corresponding mass spectrum (Figure 2A) showed several characteristic fragment ions at m/z 53.5, 67.4, 81.8, 93.7, 108.7, 124.1, 137.8, 152.8, and 164.5, which are consistent with the fragmentation pattern of 1,8-cineole (eucalyptol) as reported in the NIST mass spectral library. The molecular ion peak (m/z = 154) confirmed the molecular weight of C₁₀H₁₈O, further supporting the identification (Figure 3).
The fragmentation profile can be explained by typical bond cleavages of monoterpenoids:
The base peak at m/z4 is attributed to the formation of a stable allylic cation.
Peaks at m/z8 and 93.7 represent common fragments of cyclic monoterpenes.
Higher mass fragments such as m/z8 and 152.8 correspond to rearranged oxygenated ions, which are typical for 1,8-cineole.
Comparison with standard reference spectra demonstrated a >95% similarity index, confirming that the isolated compound was indeed 1,8-cineole. This finding is in agreement with previous reports describing 1,8-cineole as the predominant volatile compound in rosemary essential oil, along with camphor and α-pinene. However, in this study, the applied vacuum fractional distillation at 50–60 °C (30–50 mbar) selectively enriched the 1,8-cineole fraction, enabling its isolation in a pure form.30,31
 |
Figure 4: Total ion chromatogram (TIC) of the distilled fraction obtained from Rosmarinus officinalis leaves showing a single dominant peak at a retention time of 7.78 min, corresponding to 1,8-cineole. |
Notably, the GC–MS analysis of R. officinalis cultivated in Uzbekistan revealed a chemical profile comparable to rosemary grown in Mediterranean regions, where 1,8-cineole content typically ranges between 30–50% of the total essential oil. The isolation of pure 1,8-cineole in our study emphasizes the efficiency of the extraction and fractionation protocol used.32,33
Table 1: 1,8-Cineole (Eucalyptol) was identified as the main component, accounting for 100% of the total mass.
| Peak | RT (min) | Area% | Identification (Library ID) | Quality |
| 1 | 7.781 | 100.00 | 1,8-Cineole (Eucalyptol) | 83 |
Overall, the GC–MS results demonstrate that R. officinalis grown under the agro-climatic conditions of Uzbekistan is a rich source of 1,8-cineole, which was successfully isolated and confirmed by its chromatographic behavior and mass spectral fragmentation pattern.
Acute Toxicity Studies
Experimental Setup
The acute toxicity of 1,8-cineole isolated from Rosmarinus officinalis was investigated in male Swiss albino mice weighing 25 ± 2.0 g. A total of 150 animals were used for the experiments, and all procedures were carried out in accordance with international ethical standards, including the Helsinki Declaration (2000), the guidelines of the Council for International Organizations of Medical Sciences (CIOMS), and the institutional bioethics regulations of the Institute of Biophysics and Biochemistry, National University of Uzbekistan (2019). Experimental protocols were approved by the institutional Animal Use Committee, and all efforts were made to minimize animal suffering. Animals were maintained under standard vivarium conditions with free access to food and water, and quarantined for 10–14 days prior to experimentation.34,35
The acute oral toxicity was assessed following the Litchfield and Wilcoxon method. Animals received single doses of 1,8-cineole by oral gavage at concentrations of 200, 400, 800, 900, and 1000 mg/kg body weight. Control groups received the vehicle only. The animals were carefully monitored during the first 24 h after administration, with hourly observations for behavioral and functional changes such as locomotor activity, grooming, tremors, respiration rate, posture, and mortality. Subsequently, animals were observed daily for 14 days under standard vivarium conditions.
Observations During the Acute Toxicity Test
At 200 and 400 mg/kg, no mortality or abnormal behavioral changes were observed. The animals maintained normal activity, feeding behavior, and weight gain.
At 800 mg/kg, moderate toxic effects were recorded, including lethargy, decreased locomotor activity, piloerection, and tremors. Approximately 50% of animals in this group died within the observation period.36
At 900 mg/kg, severe toxic manifestations such as respiratory distress, tremors, and hypoactivity were observed, with a mortality rate of 66.6%.
At 1000 mg/kg, pronounced signs of toxicity were recorded, including convulsions, loss of motor coordination, and high mortality (83.3%).
The overall median lethal dose (LD₅₀) was calculated to be 680 mg/kg, with confidence intervals LD₁₀ ≥ 580 mg/kg and LD₈₄ ≥ 730 mg/kg (Table 2). According to OECD and WHO classification guidelines, these results place 1,8-cineole in Toxicity Class IV (low-toxic compounds) when administered orally.
Table 2: Acute oral toxicity of 1,8-cineole in male Swiss albino mice. Mortality was recorded following single-dose administration at 200–1000 mg/kg. The median lethal dose (LD₅₀) was calculated as 680 mg/kg, with confidence limits LD₁₀ ≥ 580 mg/kg and LD₈₄ ≥ 730 mg/kg.
| Animal type | Sex | Doses mg/kg | Number of animal deaths in the group | LD10 | LD16 | LD50 | LD84 |
| Mouse | Male | 200 | 6/0 | ≥580 mg/kg | ≥680 mg/kg | ≥730 mg/kg | |
| 400 | 6/0 | ||||||
| 800 | 6/3 | ||||||
| 900 | 6/4 | ||||||
| 1000 | 6/5 |
Body Weight Dynamics
Body weight was monitored before dosing and throughout the 14-day observation period. In the groups receiving 200 and 400 mg/kg, no significant deviations from control animals were recorded. At 800 and 900 mg/kg, body weight gain was reduced compared to control animals, which correlated with decreased feeding activity and overall weakness. At the lethal dose of 1000 mg/kg, most animals exhibited rapid weight loss prior to death. Statistical analysis using Student’s t-test confirmed significant differences (p < 0.05) between control and high-dose groups.
Chronic Toxicity Studies
To complement acute toxicity testing, the cumulative and sub-chronic effects of 1,8-cineole were evaluated in Wistar albino rats of both sexes weighing 210 ± 14.1 g. Animals were administered repeated oral doses of 1,8-cineole for 21 days. For cumulative toxicity testing, the animals received fractions of the calculated LD₅₀ (0.1–1.12 × LD₅₀) at 4-day intervals. The cumulative coefficient (Kc) was calculated according to the formula:
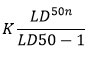
where LD₅₀n is the mean lethal dose upon repeated administration, and LD₅₀-1 is the mean lethal dose upon single administration. For 1,8-cineole, the cumulative coefficient indicated weak cumulative potential, consistent with previously reported safety profiles of monoterpenes.
Sub-chronic Toxicity and Urine Analysis
Sub-chronic toxicity studies were conducted over a 14-day period at oral doses of 5, 10, and 20 mg/kg in rats (n=6 per group). Urine samples were collected on days 7 and 14 for biochemical and cytological analysis. The following parameters were evaluated: specific gravity, pH, presence of leukocytes, nitrites, protein, glucose, ketones, bilirubin, urobilin, and hemoglobin.
Results indicated that urine parameters in experimental animals did not differ significantly from the control group. The urine pH remained within physiological ranges (6.2–7.8), and no pathological components (proteinuria, hematuria, glycosuria, bilirubinuria) were detected. Four-hour urine output was consistent across all groups, with no significant differences in diuresis between control and treated animals (Table 3). These results suggest that sub-chronic administration of 1,8-cineole at therapeutic doses does not impair renal function or cause significant metabolic disturbances.
Hematological and Coagulation Parameters
Peripheral blood samples collected on days 7 and 14 were analyzed using light microscopy and thromboelastography (TEG). No significant changes were observed in leukocyte, erythrocyte, platelet counts, or reticulocyte indices in treated groups compared to controls. TEG analysis confirmed that 1,8-cineole did not induce abnormalities in clotting time (R), clot formation time (K), or maximum amplitude (MA). This indicates that the compound does not significantly affect the hemostatic system under the tested conditions.
Discussion of Toxicological Findings
The results demonstrate that 1,8-cineole possesses a relatively low acute oral toxicity profile, with an LD₅₀ of 680 mg/kg in mice. These findings align with published data on the safety of 1,8-cineole and other monoterpenes, which are generally regarded as safe when used in moderate concentrations. Importantly, no significant sub-chronic or cumulative toxic effects were observed in rats at doses up to 20 mg/kg over 14 days.
Table 3: Effect of sub-chronic administration of 1,8-cineole (5, 10, and 20 mg/kg) on urinary parameters of Mice on days 7 and 14. No significant deviations from control values were observed in urine specific gravity, pH, biochemical indices, or diuresis.
| Tests | Control | Day 7; doses mg/kg | Day 14; doses mg/kg | ||||
| 5,0 | 10,0 | 20,0 | 5,0 | 10,0 | 20,0 | ||
| Relative density of urine | 1,010 | 1,010 | 1,010 | 1,005 | 1,030 | 1,020 | 1,015 |
| Leukocytes | neg | neg | neg | neg | neg | neg | neg |
| Nitrites | neg | neg | neg | neg | neg | neg | neg |
| pH | 6,2 | 6,6 | 7,1 | 7.6 | 7.7 | 7.8 | 7.8 |
| Protein | neg | neg | neg | neg | neg | neg | neg |
| Glucose | neg | neg | neg | neg | neg | neg | neg |
| Ketone | neg | neg | neg | neg | neg | neg | neg |
| Urobilin | Norm | Norm | Norm | Norm | Norm | Norm | Norm |
| Biluribine | neg | neg | neg | neg | neg | neg | Neg |
| Blood | neg | neg | neg | neg | neg | neg | Neg |
| Hemoglobin | neg | neg | neg | neg | neg | neg | Neg |
| 4-hour urine,% | 80 | 80 | 68 | 70 | 56 | 66 | 84 |
Collectively, the toxicological evaluation suggests that 1,8-cineole isolated from Rosmarinus officinalis cultivated in Uzbekistan is a safe bioactive compound with low acute toxicity and negligible chronic effects at therapeutic doses. These results provide a strong basis for its potential application in pharmacology, cosmetics, and the food industry.37
In silico ADMET and Pharmacokinetic Analysis
The pharmacokinetic and toxicity profiles of 1,8-cineole were predicted using the ADMETlab 2.0 platform. The results revealed a set of favorable absorption, distribution, metabolism, excretion, and toxicity (ADMET) parameters, indicating its potential as a drug-like molecule (Figure 5).
Absorption
1,8-Cineole demonstrated good gastrointestinal absorption potential. The compound was predicted to be permeable across Caco-2 cells, suggesting efficient intestinal absorption. Human intestinal absorption (HIA) probability was high, indicating suitability for oral delivery. Although it was identified as a substrate of P-glycoprotein (P-gp), which may influence efflux and bioavailability, the predicted oral bioavailability indices (F20% and F30%) were acceptable (Table 4).
Table 4: In silico ADMET prediction of 1,8-cineole.
| Compounds | Water solubility | Pharmacokinetics | Druglikeness | Medicinal chemistry | |||||||
| Log S(ESOL) | Log S (Ali) | Giabsorption | BBBpermeant | Log Kp | Lipinski | Ghose | Muegge | BioavailabilityScore | PAINS | Leadikenes | |
| 1,8-Cineole | -2.52 | -2.59 | High | Yes | -5.30 cm/s | Yes | No | No | 0.55 | 0 | No |
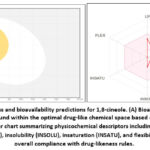 |
Figure 5: Drug-likeness and bioavailability predictions for 1,8-cineole. (A) Bioavailability radar plot showing the compound within the optimal drug-like chemical space based on WLOGP vs. TPSA parameters. |
Distribution
The compound exhibited 92.7% plasma protein binding (PPB), suggesting that most of the drug would circulate bound to serum proteins, which can prolong systemic exposure. The predicted volume of distribution (Vdss = 0.44 L/kg) indicated a moderate tissue distribution profile. Importantly, 1,8-cineole demonstrated strong blood–brain barrier (BBB) permeability, suggesting its ability to cross into the central nervous system, which may explain its reported neuroprotective and aromatherapeutic effects.
Metabolism
The metabolism profile predicted that 1,8-cineole interacts with several cytochrome P450 (CYP) isoenzymes. It was identified as a substrate of CYP2C19, CYP2D6, and CYP2B6, indicating hepatic metabolism through multiple pathways. Inhibition potential was low for most CYPs, although weak inhibitory activity was predicted for CYP2C19 and CYP2C8, suggesting minimal risk of drug–drug interactions. Hepatic microsomal stability was moderate, implying that the compound undergoes partial first-pass metabolism but retains sufficient systemic bioavailability.
Excretion
The predicted plasma clearance (CL = 9.89 mL/min/kg) and half-life (t½ = 1.14 h) suggest that 1,8-cineole is moderately eliminated from the body, primarily via hepatic metabolism and renal excretion. This pharmacokinetic profile supports its use in formulations requiring rapid onset but relatively short systemic persistence.
Toxicity
The toxicity prediction demonstrated a favorable safety profile. 1,8-Cineole was non-mutagenic in Ames test prediction, non-carcinogenic, and showed no significant risk of hepatotoxicity, nephrotoxicity, or cardiotoxicity (hERG channel inhibition was low). Skin sensitization, eye irritation, and respiratory toxicity potentials were negligible. Importantly, sub-chronic and developmental toxicity indices were within acceptable ranges.
Medicinal Chemistry Assessment
Drug-likeness evaluations confirmed compliance with Lipinski’s rule of five, Pfizer rule, and GSK rule, supporting its suitability as an orally bioavailable compound. The compound demonstrated a quantitative estimate of drug-likeness (QED = 0.52), which is considered acceptable. Structural alerts for pan-assay interference (PAINS), reactive compounds, and aggregators were absent, further supporting its drug-like characteristics.
Summary of ADMET Findings
Taken together, the ADMETlab predictions suggest that 1,8-cineole is a drug-like monoterpenoid with good intestinal absorption, ability to cross the BBB, moderate distribution and clearance, and minimal predicted toxicity. Its favorable pharmacokinetic profile aligns with its widespread use in pharmaceuticals, cosmetics, and food industries. These computational results complement the experimental toxicity studies and provide a strong rationale for further pharmacological development of 1,8-cineole (Figure 6).
 |
Figure 6: Network diagram of in silico ADMET properties of 1,8-cineole. The compound is represented at the center, with major pharmacokinetic categories (Absorption, Distribution, Metabolism, Excretion, and Medicinal Chemistry) as intermediate nodes. |
Molecular Docking Studies of 1,8-Cineole with Key Inflammatory Protein Targets
Rationale for Protein Selection
Inflammation is a complex biological response involving multiple signaling pathways and effector proteins. To elucidate the potential anti-inflammatory activity of 1,8-cineole, a natural monoterpene oxide commonly found in essential oils of Eucalyptus species, molecular docking studies were performed against a panel of inflammation-related proteins. The rationale for selecting these targets is as follows:
Cyclooxygenase-2 (COX-2, PDB ID: 5KIR)
COX-2 is an inducible isoform of cyclooxygenase responsible for the conversion of arachidonic acid to prostaglandins, central mediators of pain, fever, and inflammation. Inhibition of COX-2 is a classical mechanism for many nonsteroidal anti-inflammatory drugs (NSAIDs).
Interleukin-6 (IL-6, PDB ID: 1P9M)
IL-6 is a pleiotropic pro-inflammatory cytokine that drives chronic inflammation and autoimmune responses. It contributes to the acute phase reaction, T-cell activation, and vascular inflammation. Blocking IL-6 signaling is a validated therapeutic strategy in rheumatoid arthritis and other inflammatory diseases.
Mitogen-Activated Protein Kinase (MAPK, PDB ID: 5UOJ)
MAPKs regulate intracellular signaling cascades linked to cytokine production, immune cell activation, and stress responses. Their abnormal activation perpetuates inflammation and tissue injury. Targeting MAPKs is therefore relevant for assessing anti-inflammatory potential.
Nuclear Factor kappa-B (NF-κB, PDB ID: 2I9T)
NF-κB is a transcription factor that regulates the expression of numerous pro-inflammatory cytokines, chemokines, adhesion molecules, and enzymes (such as COX-2 and iNOS). Inhibition of NF-κB activation represents a key anti-inflammatory mechanism.
Tumor Necrosis Factor alpha (TNF-α, PDB ID: 2AZ5*)
TNF-α is a master regulator of inflammation and immune responses. It binds TNFR1/TNFR2 receptors and triggers NF-κB and MAPK pathways. Therapeutic neutralization of TNF-α is widely used in autoimmune disorders such as rheumatoid arthritis and Crohn’s disease. By targeting this set of proteins, docking studies provide a comprehensive picture of how 1,8-cineole may exert anti-inflammatory actions at the molecular level.
Docking with COX-2 (5KIR)
Docking of 1,8-cineole with the COX-2 enzyme revealed a binding affinity of −6.2 kcal/mol, indicative of a moderate but biologically relevant interaction. The ligand engaged primarily through alkyl interactions with key hydrophobic residues: LEU B:392, VAL B:349, VAL B:523, and ALA B:327 (Figure 7).
 |
Figure 7: Molecular docking interaction of 1,8-cineole with the COX-2 enzyme (PDB ID: 5KIR). The docking results show that 1,8-cineole binds within the COX-2 active site with a binding energy of −6.2 kcal/mol. |
Significance of these interactions
Hydrophobic contacts are crucial in stabilizing ligands within the COX-2 active pocket, which is predominantly nonpolar in nature. The alkyl interactions with LEU and VAL residues mimic the binding pattern of known NSAIDs, suggesting that 1,8-cineole may partially occupy the arachidonic acid binding site, thereby interfering with prostaglandin biosynthesis.
Docking with IL-6 (1P9M)
Interaction with the IL-6 protein yielded a binding energy of −5.2 kcal/mol. The binding involved alkyl and π-alkyl interactions with residues PHE A:147, MET A:117, HIS A:145, and VAL A:121. π-alkyl interactions with aromatic residues such as PHE and HIS are important for stabilizing small hydrophobic ligands like 1,8-cineole within the cytokine’s binding region. Although the energy score is lower than COX-2, the ability of 1,8-cineole to associate with IL-6 suggests a potential modulatory effect on cytokine signaling, consistent with reported immunomodulatory properties of cineole (Figure 8).
 |
Figure 8: Molecular docking interaction of 1,8-cineole with the IL-6 protein (PDB ID: 1P9M). The docking analysis revealed a binding affinity of −5.2 kcal/mol. |
Docking with MAPK (5UOJ)
Docking against MAPK produced a binding energy of −5.3 kcal/mol. The ligand established alkyl and π-alkyl interactions with ARG A:5, PHE A:8, and ILE A:346. These interactions indicate positioning of 1,8-cineole near residues critical for MAPK conformational stability. The involvement of ARG and ILE residues highlights the importance of hydrophobic as well as polar environments in accommodating cineole. This binding could plausibly attenuate MAPK activation, thereby reducing downstream inflammatory signaling (Figure 9).
 |
Figure 9: Molecular docking interaction of 1,8-cineole with the MAPK protein (PDB ID: 5UOJ). The docking study demonstrated a binding affinity of −5.3 kcal/mol. |
Docking with NF-κB (2I9T)
Docking with NF-κB resulted in a binding energy of −5.2 kcal/mol. The interactions included alkyl, π-alkyl, and nucleic acid contacts involving ARG A:605 and DA D:724. NF-κB is a DNA-binding transcription factor. The observed contacts with nucleotide residues (DA D:724) suggest that 1,8-cineole may influence the DNA-binding capacity of NF-κB. By interfering with the protein–DNA interaction interface, cineole could reduce NF-κB–mediated transcription of pro-inflammatory genes (Figure 10).
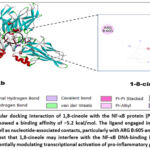 |
Figure 10: Molecular docking interaction of 1,8-cineole with the NF-κB protein (PDB ID: 2I9T). The docking results showed a binding affinity of −5.2 kcal/mol. |
Docking with TNF-α
The most favorable binding energy was observed with TNF-α, reaching −6.8 kcal/mol. The docking revealed π-alkyl interactions with TYR C:119. The stronger binding affinity indicates a relatively stable association between cineole and TNF-α. The involvement of TYR suggests that π-electron interactions stabilize the ligand at a functionally relevant site. Since TNF-α is a trimeric cytokine, disruption of its active binding conformation by small molecules could attenuate receptor activation and dampen the entire inflammatory cascade (Figure 11).
 |
Figure 11: Molecular docking interaction of 1,8-cineole with the TNF-α protein. Docking analysis revealed a binding affinity of −6.8 kcal/mol, the strongest interaction among the studied targets. |
Comparative Analysis
Among all targets, TNF-α (−6.8 kcal/mol) and COX-2 (−6.2 kcal/mol) showed the strongest binding, suggesting that 1,8-cineole may exert dual inhibition at both upstream (cytokine level) and downstream (prostaglandin synthesis) inflammatory pathways. IL-6, MAPK, and NF-κB interactions, although weaker (−5.2 to −5.3 kcal/mol), support the idea of a multitarget modulation, a characteristic often attributed to phytochemicals. Hydrophobic interactions (alkyl and π-alkyl) dominate in all cases, consistent with the lipophilic nature of cineole. Such interactions are essential for ligand stabilization within protein pockets, even at modest binding energies.
This docking study demonstrates that 1,8-cineole exhibits moderate to strong binding affinities with key inflammatory mediators, including COX-2, IL-6, MAPK, NF-κB, and TNF-α. The observed binding energies (−5.2 to −6.8 kcal/mol) and interaction profiles suggest that cineole’s anti-inflammatory activity may result from simultaneous modulation of multiple signaling pathways, rather than selective inhibition of a single protein. This polypharmacological behavior is typical of natural compounds and supports the traditional use of cineole-rich essential oils in the management of inflammatory disorders.
Discussion
The present study investigated the isolation, characterization, and biological evaluation of 1,8-cineole from Rosmarinus officinalis cultivated in Uzbekistan. Using vacuum fractional distillation followed by GC–MS analysis, 1,8-cineole was successfully obtained in a pure form. The mass spectral data, with characteristic fragment ions at m/z 53.5, 67.4, 81.8, 93.7, 108.7, 124.1, 137.8, 152.8, and 164.5, were in complete agreement with reference spectra, confirming the identity of the compound. The retention time of 7.78 min further validated the high purity of the isolated fraction. These findings align with reports from other regions, where 1,8-cineole is recognized as a principal constituent of rosemary essential oil, typically ranging between 30–50% of the total oil fraction.
A notable contribution of this study is the demonstration that R. officinalis cultivated under the climatic and ecological conditions of Uzbekistan also produces 1,8-cineole in appreciable quantities. Since the chemical composition of essential oils is highly dependent on geographical and environmental factors, the confirmation of 1,8-cineole presence in locally cultivated rosemary provides a valuable basis for its potential exploitation in pharmaceutical, cosmetic, and food industries within the region.
Toxicological evaluation is a critical prerequisite for the development of bioactive compounds into therapeutic candidates. Acute toxicity studies revealed that single oral administration of 1,8-cineole up to 400 mg/kg produced no observable adverse effects in mice. At higher doses, dose-dependent toxicity was evident, with significant mortality occurring at 800–1000 mg/kg. The calculated LD₅₀ value of 680 mg/kg indicates that 1,8-cineole belongs to toxicity class IV (low-toxic substances) according to OECD guidelines. These findings are consistent with previous reports that describe 1,8-cineole and related monoterpenoids as compounds with relatively low acute toxicity when administered orally.
Sub-chronic toxicity tests conducted over 14 days demonstrated no significant alterations in body weight, behavior, or survival at doses of 5–20 mg/kg. Urinalysis parameters, including specific gravity, pH, protein, glucose, bilirubin, and hemoglobin, remained within physiological ranges. Importantly, no pathological changes were observed in urine sediments, and diuresis values did not differ significantly between treated and control animals. Hematological analysis further confirmed the absence of toxic effects on erythrocytes, leukocytes, and platelets, while thromboelastography data indicated that 1,8-cineole did not adversely affect coagulation parameters. These results suggest that the compound is well tolerated at sub-chronic doses and supports its potential for safe therapeutic use.
In silico ADMET predictions provided additional insights into the pharmacological potential of 1,8-cineole. The compound exhibited high gastrointestinal absorption, strong blood–brain barrier (BBB) permeability, and a bioavailability score of 0.55. These properties support its oral administration and suggest possible central nervous system effects, which correlate with its traditional use in aromatherapy and reported neuroprotective actions.
Metabolic predictions indicated that 1,8-cineole is a substrate for multiple CYP450 isoenzymes (CYP2C19, CYP2D6, CYP2B6, and CYP2C9), suggesting hepatic metabolism through diverse pathways. Although minor inhibitory effects against CYP2C19 and CYP2C8 were predicted, the overall risk of drug–drug interactions remains low. Excretion parameters, including moderate plasma clearance (9.89 mL/min/kg) and a short half-life (t½ = 1.14 h), indicate relatively rapid elimination from the body, which may be advantageous for compounds requiring short systemic persistence.
Importantly, toxicity predictions revealed no mutagenic, carcinogenic, hepatotoxic, or cardiotoxic effects, and the compound complied with Lipinski’s, Pfizer, and GSK drug-likeness rules. These findings confirm that 1,8-cineole exhibits favorable pharmacokinetic and safety characteristics, reinforcing its potential as a drug-like monoterpenoid.
Molecular docking further supported the anti-inflammatory potential of 1,8-cineole. The compound bound with moderate affinity to COX-2 (−6.2 kcal/mol), IL-6 (−5.2 kcal/mol), MAPK (−5.3 kcal/mol), and NF-κB (−5.2 kcal/mol), and with the strongest affinity to TNF-α (−6.8 kcal/mol). Hydrophobic interactions (alkyl and π-alkyl) dominated across targets, suggesting multitarget modulation of inflammatory pathways. Strong binding to COX-2 and TNF-α indicates potential dual action on prostaglandin synthesis and cytokine signaling.
When compared with other well-studied monoterpenoids such as limonene and menthol, 1,8-cineole displays a similarly low toxicity profile but offers distinctive pharmacological benefits, particularly in respiratory and inflammatory disorders. Limonene is known for its anticancer and gastroprotective activities, while menthol is widely applied for its cooling and analgesic effects. In contrast, 1,8-cineole has demonstrated strong mucolytic, bronchodilatory, and anti-inflammatory activities, making it particularly valuable for respiratory conditions such as bronchitis, asthma, and sinusitis. The ability of 1,8-cineole to cross the BBB may also provide opportunities for its application in neuroprotection and cognitive enhancement, areas that warrant further investigation.
Taken together, the findings of this study provide a comprehensive evaluation of 1,8-cineole isolated from R. officinalis cultivated in Uzbekistan. The results confirm its chemical identity, establish its relatively low acute and sub-chronic toxicity, and demonstrate its favorable pharmacokinetic and safety profile through in silico predictions. These outcomes not only reinforce the pharmacological potential of 1,8-cineole but also highlight the importance of studying medicinal plants in specific ecological regions, where unique environmental conditions may influence the yield and activity of bioactive compounds.
Future research should focus on detailed pharmacological testing, including in vivo models of inflammation, oxidative stress, and respiratory diseases, as well as clinical evaluations to further validate the therapeutic potential of 1,8-cineole.
Conclusion
In this study, 1,8-cineole was successfully isolated in pure form from Rosmarinus officinalis cultivated in Uzbekistan and identified by GC–MS. Toxicological assessment demonstrated a relatively low acute toxicity profile (LD₅₀ = 680 mg/kg) and no significant adverse effects upon sub-chronic administration. In silico ADMET predictions confirmed favorable pharmacokinetic properties, including high intestinal absorption, BBB permeability, and compliance with major drug-likeness rules, with minimal predicted toxicity. Taken together, these findings highlight 1,8-cineole as a safe and pharmacologically promising bioactive compound, supporting its potential application in pharmaceuticals, cosmetics, and food industries.
Acknowledgement
The author would like to thank Abdullaev Izzatullo for creating manuscript
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Iroda Ganieva*: Conceptualization, Methodology, Supervision, Writing – Review & Editing. Ilyos Xudoynazarov: Data Curation, Formal Analysis, Writing – Original Draft.
Alisher Khaitbaev: Investigation, Resources, Validation.
Muzaffar Shokirov: Software, Visualization, Data Analysis.
Mirjalol Jalilov: Methodology, Experimental Work, Data Collection.
Ilshod Mansurov: Formal Analysis, Literature Review, Draft Preparation.
Mukhammadjon Mustafakulov and Iqboljon Yuldashev: Investigation, Project Administration, Writing – Review & Editing
References
- De Oliveira JR, de Jesus D, Figueira LW, et al. Biological activities of Rosmarinus officinalis L. (rosemary) extract as analyzed in microorganisms and cells. Exp Biol Med (Maywood). 2017;242(6):625-634.
CrossRef - I Abdullaev, U Gayibov, S Omonturdiev, et al. Molecular pathways in cardiovascular disease under hypoxia: Mechanisms, biomarkers, and therapeutic target. The JBR 2025; 39(3): 254-269.
CrossRef - Rašković A, Milanović I, Pavlović N, et al. Antioxidant activity of rosemary (Rosmarinus officinalis L.) essential oil and its hepatoprotective potential. BMC Compl Altern Med 2014; 14: 225.
CrossRef - AA Abdullaev, DR Inamjanov, DS Abduazimova, et al. Sílybum Mariánum’s impact on physiological alterations and oxidative stress in diabetic rats. Biomedical and Pharmacology Journal 2024; 17(2): 1291-1300.
CrossRef - González-Minero FJ, Bravo-Díaz L, Ayala-Gómez A. Rosmarinus officinalis L. (Rosemary): An Ancient Plant with Uses in Personal Healthcare and Cosmetics. Cosmetics. 2020; 7(4):77.
CrossRef - O Gaibullayeva, A Islomov, D Abdugafurova, et al. Inula helenium root extract in sunflower oil: Determination of its content of water-soluble vitamins and immunity-promoting effect. Biomedical and Pharmacology Journal 2024; 17(4): 2729-2737.
CrossRef - AQQ Azimova, AX Islomov, SA Maulyanov, et al. Determination of vitamins and pharmacological properties of Vitis vinifera plant fruit part (mixed varieties) syrup-honey. Biomedical and Pharmacology Journal 2024; 17(4): 2779-2786.
CrossRef - AV Mahmudov, OS Abduraimov, SB Erdonov, et al. Bioecological features of Nigella sativa in different conditions of Uzbekistan. Plant Science Today 2022; 9(2): 421-426.
CrossRef - OS Zoirovich, AIZ Ugli, ID Raxmatillayevich, et al. The effect of Ájuga Turkestánica on the rat aortic smooth muscle ion channels. Biomedical and Pharmacology Journal 2024; 17(2): 1213-1222.
CrossRef - D Inomjonov, I Abdullaev, S Omonturdiev, et al. In vitro and In vivo studies of Crategus and Inula helenium extracts: Their effects on rat blood pressure. Trends in Sciences 2025; 22(3):
CrossRef - A Abdullaev, I Abdullaev, A Bogbekov, et al. Antioxidant potential of Rhodiola heterodonta extract: Activation of Nrf2 pathway via integrative in vivo and in silicoTrends in Sciences 2025; 22(5): 9521.
CrossRef - Andrade JM, Faustino C, Garcia C, Rosmarinus officinalis L.: an update review of its phytochemistry and biological activity. Future Sci OA. 2018;1:4(4)
CrossRef - Abdelaziz, m. E., pokluda, r., & abdelwahab, m. M. Influence of compost, microorganisms and npk fertilizer upon growth, chemical composition and essential oil production of rosmarinus officinalis l. Notulae Botanicae Horti Agrobotanici Cluj-Napoca, 2007; 35(1): 86–90.
- Bajalan, Iman & Rouzbahani, Razieh et al. Antioxidant and antibacterial activities of the essential oils obtained from seven Iranian populations of Rosmarinus officinalis. Industrial Crops and Products. 2017; 107:305-311.
CrossRef - Alexandra, Jeimmy & Cuervo Andrade, Jairo et al. Effect of organic fertilization on yield and quality of rosemary (Rosmarinus officinalis L.) essential oil. Agronomía Colombiana. 2017; 35:232-237.
CrossRef - Derwich, E. & Benziane, Zineb et al. Aromatic and medicinal plants of Morocco: chemical composition of essential oils of Rosmarinus officinalis and Juniperus phoenicea. J. Appl. Biol. Pharm. Technol. 2011;2: 145-153.
- Gachkar, Latif & Yadegari, Davood et al. Chemical and biological characteristics of Cuminum cyminum and Rosmarinus officinalis essential oils. Food Chemistry. 2007;102: 898-904.
CrossRef - UG Gayibov, SN Gayibova, DS Abduazimova, et al. Antioxidant and cardioprotective properties of polyphenolic plant extract of Rhus glabra Plant Science Today 2024; 11(3): 2348-1900.
CrossRef - TF Aripov and UG Gayibov. Antiradical and antioxidant activity of the preparation “Rutan” from Rhus coriaria Journal of Theoretical and Clinical Medicine 2023; 4: 164-170.
- AV Mahmudov, OS Abduraimov, SB Erdonov, et al. Seed productivity of Linum usitatissimum in different ecological conditions of Uzbekistan. Plant Science Today 2022; 9(4): 1090-1101.
CrossRef - Genena, Aziza & Hense, et al. Rosemary (Rosmarinus officinalis) – A study of the composition, antioxidant and antimicrobial activities of extracts obtained with supercritical carbon dioxide. Ciencia E Tecnologia De Alimentos – CIENCIA TECNOL ALIMENT. 2025; 28: 2600
CrossRef - PJ Tonge. Drug-target kinetics in drug discovery. ACS Chemical Neuroscience Journal 2018; 9(1): 29-39.
CrossRef - Sodiqova, S., Kadirova, S., Zaynabiddinov, A., et al. Channelopathy Activity Of A-41(Propyl Ester of Gallic Acid): Experimental and Computational Study of Antihypertensive Activity. Trends in Sciences, 2025; 22(9): 10496.
CrossRef - Zaripova, M., Abdullaev, I., Bogbekov, et al. In Vitro and in Silico Studies of Gnaphalium U. Extract: Inhibition of α-amylase and α-glucosidase as a Potential Strategy for Metabolic Syndrome Regulation. Trends in Sciences, 2025; 22(8): 10098.
CrossRef - UG Gayibov, EJ Komilov, RN Rakhimov, et al. Influence of new polyphenol compound from Euphorbia plant on mitochondrial function. Journal of Microbiology Biotechnology and Food Science 2019; 8(4): 1021-1025.
CrossRef - González-Trujano ME, Peña EI, Martínez AL, et al. Evaluation of the antinociceptive effect of Rosmarinus officinalis L. using three different experimental models in rodents. J Ethnopharmacol. 2007; 111(3):476-82.
CrossRef - Khaitbaev, Alisher & Aslanov, et al. Synthesis of new imino derivatives of gossypol. Chemistry of Natural Compounds – Chem Nat Compd. 1994; 30. 40-41.
CrossRef - Y Umidakhon, B Erkin, G Ulugbek, et al. Correction of the mitochondrial NADH oxidase activity, peroxidation and phospholipid metabolism by haplogenin-7-glucoside in hypoxia and ischemia. Trends in Sciences 2022; 19(21): 6260.
CrossRef - MK Pozilov, U Gayibov, MI Asrarov, et al. Physiological alterations of mitochondria under diabetes condition and its correction by polyphenol gossitan. Journal of Microbiology Biotechnology and Food Science 2022; 12(2): 2224.
CrossRef - Vakhobjonovna, A. G, Jurayevich K. E, Ogli A. I. Z, et al. Tannins as Modulators in the Prevention of Mitochondrial Dysfunction. Trends in Sciences, 2025; 22(8), 10436.
CrossRef - Z Shakiryanova, R Khegay, U Gayibov, et al. A Latif and O Smirnova. Isolation and study of a bioactive extract enriched with anthocyanin from red grape pomace (Cabernet Sauvignon). Agronomy Research 2023; 21(3): 1293-1303.
- U Gayibov, SN Gayibova, KP Ma’murjon, et al. Influence of quercetin and dihydroquercetin on some functional parameters of rat liver mitochondria. Journal of Microbiology, Biotechnology and Food Sciences 2021; 11(1): 2924.
CrossRef - Utamuradova, Sharifa & Daliev, et al. Influence of Gold on Structural Defects of Silicon. East European Journal of Physics. 2024; 327-335.
CrossRef - MR Zaripova, SN Gayibova, RR Makhmudov, et al. Characterization of Rhodiola heterodonta (Crassulaceae): Phytocomposition, antioxidant and antihyperglycemic activities. Preventitive Nutrition Food Science 2024; 29(2), 135-145.
CrossRef - Daliev, Khodjakbar & Utamuradova, et al. Defective Structure of Silicon Doped with Dysprosium. East European Journal of Physics. 2024; 283-287.
CrossRef - Xu J, Hu ZQ, Wang C, et al. Acute and subacute toxicity study of 1,8-cineole in mice. Int J Clin Exp Pathol, 2024;7(4):1495-501.
- Mansurov DA, Khaitbaev AK, Khaitbaev KK, et al. Relationship between structural properties and biological activity of (-)-menthol and some menthyl esters. Comput Biol Chem. 2025;115:108357.
CrossRef







