Manuscript accepted on :17-10-2025
Published online on: 10-11-2025
Plagiarism Check: Yes
Reviewed by: Dr. EXBRAYAT Jean-Marie and Dr. Sathyanarayana Namani
Second Review by: Dr. Samara Sameer
Final Approval by: Dr. Mariia Shanaida
Binoy Varghese Cheriyan1 , Kaniga Pandi*
, Kaniga Pandi* , Dharshan Murali
, Dharshan Murali , Nesayan Boobalan
, Nesayan Boobalan , Jeevitha Harikrishnan
, Jeevitha Harikrishnan , Deepshikaa Kannan
, Deepshikaa Kannan , Lini priyadharshini christopher jebaraj
, Lini priyadharshini christopher jebaraj , Shamyuktha Munuswamy
, Shamyuktha Munuswamy and Shagul Hameed Manaksha
and Shagul Hameed Manaksha
Department of Pharmaceutical Chemistry, Saveetha College of Pharmacy, Saveetha Institute of Medical and Technical Sciences, Chennai, Tamil Nadu, India.
Corresponding Author E-mail: kanigapharma@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3274
Abstract
Cancer remains a major global health concern, driving the ongoing search for safer and more effective therapeutic agents. Naturally occurring polyphenolic compounds, including flavonoids, have gained considerable attention for their diverse pharmacological activities, particularly their ability to modulate cancer cell proliferation and apoptosis. This review summarizes current evidence on the anticancer potential of polyphenols and their subclasses across various human cancer cell lines, emphasizing their mechanisms in inducing programmed cell death and regulating cell cycle progression. Key flavonoids such as apigenin, quercetin, and kaempferol, along with other polyphenols like resveratrol and curcumin, exhibit significant anticancer effects through the activation of intrinsic and extrinsic apoptotic pathways and modulation of key cell cycle checkpoints. In vitro studies consistently demonstrate dose-dependent cytotoxicity, mitochondrial membrane disruption, caspase activation, and regulation of p53, Bcl-2, cyclins, and CDKs in breast, colon, lung, prostate, and cervical cancer models. Despite encouraging preclinical outcomes, limitations such as poor bioavailability, inter-model variability, and lack of standardized protocols persist. Furthermore, synergistic interactions with conventional chemotherapeutics and advancements in delivery systems and molecular profiling tools may enhance their clinical relevance. Overall, polyphenols, particularly flavonoids, represent a promising class of anticancer agents warranting further translational and clinical exploration.
Keywords
Anticancer Therapy; Apoptosis; Cancer Cell Lines; Cell Cycle Arrest; Flavonoids; Natural Compounds; Polyphenols
Download this article as:| Copy the following to cite this article: Cheriyan B. V, Pandi K, Murali D, Boobalan N, Harikrishnan J, Kannan D, Jebaraj L. P. C, Munuswamy S, Manaksha S. H. Polyphenols Target Apoptosis and Cell Cycle Regulation in Various Cancer Models: Emphasis on Flavonoid Subclasses. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Cheriyan B. V, Pandi K, Murali D, Boobalan N, Harikrishnan J, Kannan D, Jebaraj L. P. C, Munuswamy S, Manaksha S. H. Polyphenols Target Apoptosis and Cell Cycle Regulation in Various Cancer Models: Emphasis on Flavonoid Subclasses. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4oqBFSY |
Introduction
Cancer is a worldwide health issue that transcends national borders. In addition to environmental and medical causes, it occurs as a result of widespread DNA damage caused by ionising and UV radiation. Overall, the most often diagnosed cancers are stomach (7.81%), colorectal (9.7%), lung (12.7%), and breast (10.9%). Over the next 20 years, there will likely be a 50% increase in new cancer cases worldwide.1 Numerous obstacles confront global cancer care, particularly in low- and middle-income (LMIC) nations. Access to quality, affordable cancer care including screening, skilled staff, standard treatments, and supportive services is often limited in many regions.2 In 2020, over 19 million people were diagnosed with cancer, resulting in nearly 10 million deaths globally.3 By 2040, an estimated 28 million additional cases and 16 million fatalities are predicted.4,5 In 2019, the cost of cancer treatment alone was estimated to be US$1.2 trillion per year, or about 2% of the world’s total domestic product.6 Although they can be somewhat effective, traditional cancer treatments like chemotherapy and radiation therapy are frequently linked to serious side effects, poor specificity, and the development of resistance. The pressing need for safer and more efficient therapeutic alternatives is highlighted by this increasing burden. Natural products’ structural variety, bioactivity, and comparatively low toxicity have made them valuable sources of new anticancer medicines in recent years. Because they are widely accessible, efficient, and possess anti-cancer qualities, natural compounds are the most effective against cancer. Natural components have been a defining feature of the history of anticancer medication development. Numerous widely used anticancer drugs are derived from natural sources, such as paclitaxel from plants, irinotecan, vincristine, etoposide, marine-derived bleomycin, and actinomycin D and mitomycin C from bacteria. For the foreseeable future, some of these substances will remain essential components of cancer treatment.7 Flavonoids and polyphenols secondary metabolites found in fruits, vegetables, and medicinal plants show strong potential in cancer prevention and treatment, including compounds like catechin, quercetin, ellagic acid, and stilbenes. Phyllanthus emblica, Flemingia vestita, Apium graveolens, Petroselinum crispum, and other medicinal plants are among the many plant sources from which natural flavonoids are extracted. Polyphenols are a broad category of compounds that include lignans, stilbenes, phenolic acids, and flavonoids. Flavonoids, a key subclass of polyphenols, are classified into flavonols, flavones, isoflavones, flavanones, anthocyanidins, and chalcones based on structure.8 They exhibit anti-inflammatory, antioxidant, and anticancer properties by inducing apoptosis in breast, colorectal, and prostate cancers, inhibiting nucleoside diphosphate kinase-B in lung, bladder, and colon cancers, and blocking the NF-kB pathway to halt cell proliferation and induce cell cycle arrest.9 These compounds modulate key signaling pathways linked to tumor progression, particularly apoptosis and cell cycle regulation, which are pivotal targets in cancer therapy screening.10 This review evaluates flavonoids and polyphenols studied in cancer cell lines, emphasizing their role in inducing apoptosis and cell cycle arrest, while highlighting their potential in anticancer drug development.
Materials and Methods
A comprehensive and systematic literature search was conducted using major scientific databases, including PubMed, Scopus, Web of Science, and Google Scholar, focusing primarily on original research articles, review papers, and meta-analyses published using keywords such as “flavonoids,” “polyphenols,” “apoptosis,” “cell cycle arrest,” “cancer,” “molecular targets,” and specific compound names like quercetin, genistein, curcumin, and resveratrol. Only English-language publications were considered. To give a comprehensive picture of the molecular functions and therapeutic implications of these phytochemicals, included were studies that combined clinical research with in vitro and in vivo cancer models. Emphasis was placed on research identifying molecular targets, dose-dependent responses, synergistic effects, and selectivity toward cancer cells. Articles that lacked mechanistic insights or reproducible data were excluded. Information was extracted and synthesized based on compound type, cancer model, apoptotic/cell cycle effects, and therapeutic potential. References were managed and organized using Mendeley, and PRISMA guidelines were loosely followed to ensure the rigor and transparency of the review process.
Classification and Sources
Approximately 8000 polyphenols have been identified, categorized into flavonoids (flavones, flavonols, isoflavones, flavanones, anthocyanins), phenolic acids (hydroxybenzoic and hydroxycinnamic acids), stilbenes (resveratrol, piceatannol), lignans (sesamol, pinoresinol, sinol, enterodiol), and tannins (hydrolysable, non-hydrolyzable, and condensed) (The structural classification of flavonoids is depicted in Fig.1).11-13 Dietary polyphenols mainly include flavones, isoflavones, catechins, and phenolic acids.11Phenolic acids, with aromatic rings and carboxylic acid groups, are abundant in plants and known for their antioxidant properties that help prevent cardiovascular diseases.11,14,15 Their aromatic rings act as reducing agents by donating electrons and neutralizing free radicals.
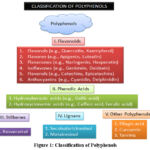 |
Figure 1: Classification of Polyphenols
|
In plant-based diets, the primary sources of phenolic acid are leafy vegetables, fruit skins, and seeds. There are several applications for these phenolic acids in cosmetics, medicine, and food.16 These compounds vary in structure from simple molecules to complex polymers, yet all possess hydroxylated aromatic rings, which contribute to their antioxidant and bioactive properties. Understanding the structural diversity of flavonoids and polyphenols is essential, as their anticancer mechanisms including apoptosis induction and cell cycle arrest are often closely linked to their chemical nature and cellular interactions. Flavonoids and polyphenols are widely distributed in various dietary and medicinal plant sources, making them readily accessible through everyday nutrition and traditional herbal remedies (Table 1).
Table 1: Dietary and Medicinal Plant Sources of Flavonoids and Polyphenols
|
Source Type |
Examples |
Key Compounds |
|
Fruits |
Apples, berries, grapes, oranges, pomegranates |
Quercetin, anthocyanins, catechins |
|
Vegetables |
Onions, kale, spinach, broccoli |
Flavonols, flavones |
|
Beverages |
Green tea, black tea, red wine, coffee |
EGCG, resveratrol, chlorogenic acid |
|
Whole Grains |
Oats, barley, brown rice |
Phenolic acids, lignans |
|
Nuts & Seeds |
Flaxseeds, walnuts, sesame seeds |
Lignans, tannins |
|
Medicinal Plants |
Curcuma longa (turmeric), Camellia sinensis (green tea), Ginkgo biloba, Scutellaria baicalensis, Glycine max (soy) |
Curcumin, EGCG, baicalin, genistein, daidzein |
|
Herbal Remedies |
Plant extracts, powders, capsules |
Mixed polyphenols and flavonoid-rich fractions |
Flavonoids
Flavonoids, a major class of polyphenolic plant metabolites, possess a 15-carbon skeleton with two aromatic rings linked by a three-carbon chain.17 Based on oxidation, conformation, and B-ring attachment, they are classified into flavones (chrysin, apigenin, baicalein), flavonols (quercetin, kaempferol), isoflavones (daidzein, glycitein), flavan-3-ols (gallocatechin, catechin, epicatechin), flavanones (hesperetin, naringenin), and anthocyanidins (delphinidin, peonidin, cyanidin, pelargonidin).11 Found in berries, onions, tea, grapes, apples, and cocoa, flavonoids offer cell-signaling, anti-thrombogenic, and neuroprotective effects.18
Flavonols
A subclass of the flavonoid family, flavonols are made up of carbonyl C4 and double bonds between C2 and C3. Plant flavonols like myricetin, kaempferol, and quercetin are commonly found in fruits and vegetables, including tomatoes, lettuce, onions, and kale.19Clinical trials have demonstrated that these flavonols may be used to prevent and treat heart disease, blood coating, cell regeneration, human gingival disease, and other heart-related conditions.12
Isoflavones
One class of phytoestrogens that are nonsteroidal substances obtained from plants are isoflavones.20The Fabaceae family is the source of isoflavones. In place of the phenyl group, isoflavones have a structure consisting of two phenyl-4H-1benzopyr-4-one.12 Isoflavones are produced by the phenylpropanoid pathway, which helps higher plants produce flavone families. Isoflavones can be found in soybeans and their byproducts. Soybeans have a high isoflavone concentration because they include two important components, genistein and daidzein.21 Isoflavones are believed to have chemo-protective qualities and may be used to treat menopause-related symptoms, according to research.11 According to studies, it also help treat osteoporosis, heart-related conditions, cancer (prostate and breast), and a number of hormonal diseases.
Flavanones
Flavanones, which make up a minor portion of flavonoids, are found in citrus fruits, tomatoes, and certain aromatic plants. In addition to its flavouring properties, they are thought to have a significant role in human health. Flavanones include naringenin from grapefruit, hesperidin from oranges, and eriodyctiol from lemons.22 In addition, flavanones go through a number of chemical reactions, including hydroxylation, glycosylation, and o-methylation.12 Flavonoids have been shown to be abundant in bergamot juice, also known as citrus bergamia.23 This indicates that bergamot extract lowers serum cholesterol, offering cardiovascular benefits by improving lipid profiles, lipoproteins, and reducing atherosclerosis risk. Prolonged use over six months has shown cholesterol-lowering effects, with LDL monitoring aiding in detecting cardiovascular events.14
Anthocyanidins
The primary pigments that give fruits and vegetables their red, pink, and purple colours are called anthocyanidins.24 Anthocyanidins present in the epidermis give fruits, vegetables, and flowers their color. They are abundant in berries, cherries, beets, radish, and strawberries,25 with aronia berries being most consumed for their strong antioxidant activity.26 Chocolate berries contain flavanols, kaempferol, cyanidin glycosides, and other beneficial compounds.12Over time, berries, honey, and olive oil have all demonstrated significant health benefits.
Flavones
Broccoli, celery, parsley, and grains are common sources of flavones. The outer coating of citrus fruits also contains significant amounts of them. Flavones were found to have an inverse effect on CAD.27 One of the flavones, tangerine, inhibits platelet synthesis, which encourages the body’s smooth muscles to multiply, resulting in atherogenesis and restenosis. Consuming citrus fruits that contain nobiletin can help reduce inflammation and cancer. Numerous studies have demonstrated that nobiletin promotes platelet activation. It also inhibits the phosphorylation of MAPKs and protein kinases.28
Other polyphenols
Polyphenols like tannins, lignans (e.g., sesamol, enterodiol), and stilbenes (e.g., resveratrol) have diverse medicinal and commercial applications based on their modes of action (Fig.1).11
Stilbenes
Stilbenes (C₁₄H₁₂), phenol-derived metabolites, are widely studied for their biological activity and health benefits.29 Stilbenes are compact chemical compounds containing one phenyl group and a core ethylene portion. At the extremities of the carbon double bonds lies the phenyl group.30Tran’s stilbenes is the colloquial term for stilbenes. Auguste Laurent, a French scientist, made the discovery of stilbenes in 1843. Their overall health advantages are currently being investigated, as are their bioavailability, metabolic rates, and absorption in the body.29
Lignin
Numerous plant tissues include the complicated chemical compound family known as lignin. Because it encourages the formation of the cell wall, lignin is especially important in plants and trees.31 They are crucial because they give trees stiffness and strength. A P Candolle, a Swiss botanist, made the initial identification of lignin in 1813. A few numbers of signal precursors are used to create the heterogeneous polymers known as lignin, which then go through various cross-linking processes.12 Coniferyl alcohol, sinapyl alcohol, and paracoumaryl alcohol are the three forms of cross-links that are generated from phenylpropane.31 Foods high in lignin include apples, peaches, tomatoes, flaxseeds, and some berries.
Bioavailability and metabolism considerations
Despite their promising anticancer properties, the bioavailability and metabolism of flavonoids and polyphenols present significant challenges for their clinical application. These compounds often exhibit poor absorption, rapid metabolism, and limited systemic availability when consumed orally.32 Among the factors influencing their absorption in the gastrointestinal tract include molecular size, glycosylation, solubility, and interactions with gut flora. After absorption, flavonoids and polyphenols undergo extensive phase I and II metabolism in the intestinal wall and liver, potentially reducing their biological activity. This metabolism includes oxidation, methylation, sulfation, and glucuronidation.33 Furthermore, the effects of conjugated metabolites, which are produced during metabolism, may differ or be less pronounced than those of the parent substances. Because it has the ability to either activate or degrade polyphenols into bioactive or inactive metabolites, the gut microbiota plays a crucial role.34 Furthermore, many flavonoids and polyphenols have a short plasma half-life, requiring high or repeated dosing to maintain therapeutic levels, which is often impractical. Many approaches, such as the utilisation of phospholipid complexes, liposomes, nanoformulations, and co-administration with bioenhancers like piperine, have been investigated to get around these restrictions. Enhancing solubility, stability, absorption, and targeted delivery are the goals of these strategies.35 Maximising these medications’ therapeutic potential and ensuring a smooth transition from in vitro research to in vivo models and clinical settings requires an understanding of their pharmacokinetic behaviour.
Mechanisms of Cancer Cell Cycle Regulation and Apoptosis
Overview of apoptosis pathways
Apoptosis is a regulated mechanism essential for eliminating damaged or malignant cells and maintaining tissue homeostasis.36 It proceeds via intrinsic and extrinsic pathways. The intrinsic pathway is triggered by internal stress like DNA damage or oxidative stress, leading to mitochondrial outer membrane permeabilization (MOMP) and release of cytochrome c, which forms the apoptosome with Apaf-1 and pro-caspase-9, activating caspase-3. The Bcl-2 family regulates this balance. The extrinsic pathway is initiated by death ligands (e.g., FasL, TNF-α, TRAIL) binding to death receptors (Fas, TNFR1, DR4/DR5), forming the DISC complex that activates caspase-8 or -10. The apoptotic process is then triggered by these initiator caspases activating effector caspases, including caspase-3. Through mitochondrial participation, the apoptotic signal may be increased by the interaction of the intrinsic and extrinsic pathways. Determining how flavonoids and polyphenols can specifically cause apoptosis in cancer cells requires an understanding of these pathways.37
Role of key regulators: Bcl-2, Bax, caspases, cytochrome c
Key regulators of apoptosis include Bcl-2, Bax, caspases, and cytochrome c. In the intrinsic pathway, the balance between pro- and anti-apoptotic Bcl-2 family proteins is crucial. Bcl-2 maintains mitochondrial membrane integrity by inhibiting apoptogen release, while Bax promotes MOMP, leading to cytochrome c release into the cytosol. The apoptosome is formed when cytochrome c enters the cytoplasm and binds to pro-caspase-9 and Apaf-1 to activate initiator caspase-9. The breakdown of cellular components is then regulated by effector caspases, primarily caspase-3, which cleave structural and repair proteins, finally resulting in apoptosis. Caspases are cysteine proteases that are produced as dormant zymogens and undergo cascading activation. Cancer cells frequently exhibit altered Bcl-2 and Bax functional status and relative expression levels, which contribute to apoptotic resistance. Therefore, a variety of anticancer drugs, including flavonoids and polyphenols, work by altering these regulators, which tilt the scales in favour of apoptosis by downregulating Bcl-2, upregulating Bax, and activating caspases. Clarifying how phytochemicals can specifically cause cancer cells to die without harming healthy tissues requires an understanding of the functions of these compounds.38
Cell cycle phases and checkpoints
The highly regulated cell cycle allows for precise cell division and replication. The cell cycle consists of G₁, S, G₂, and M phases. G₁ involves cell growth and preparation for DNA replication, while the S phase focuses on DNA synthesis. The G₂ phase, which gets the cell ready for mitosis, and the M phase, which is when cell division happens, come next. A number of cell cycle checkpoints, which keep an eye on cellular circumstances and guarantee the integrity of the genome, strictly regulate the movement through these stages. The G₁/S checkpoint detects DNA damage before replication, while the G₂/M checkpoint ensures DNA is fully and accurately replicated before mitosis. Moreover, proper chromosomal alignment and attachment to spindle fibres are maintained throughout mitosis by the spindle assembly checkpoint (SAC). Cyclins, CDKs, and their inhibitors regulate these checkpoints. For example, the tumor suppressor p53 halts the cell cycle upon DNA damage by inducing p21, a CDK inhibitor that blocks cyclin-CDK complexes. In cancer, these checkpoints are often dysregulated, allowing uncontrolled proliferation. Natural compounds like flavonoids and polyphenols have been shown to interfere with important phases of the cell cycle, particularly by causing arrest at the G₀/G₁ or G₂/M phases, which inhibits cancer growth and encourages apoptosis.39
Key regulators – p53, p21, CDKs, cyclins
Cell cycle progression is controlled by a network of important regulatory proteins, including as cyclins, p53, p21, and cyclin-dependent kinases (CDKs). These proteins work together to preserve proper cell division and genomic integrity. The tumor suppressor p53, known as the “guardian of the genome,” maintains cellular homeostasis by activating genes like p21 in response to DNA damage or stress. The potent CDK inhibitor p21 binds to cyclin-CDK complexes and prevents them from functioning, particularly those crucial for the G₁/S and G₂/M transitions. By essentially stopping the cell cycle, this inhibition enables the cell to either repair damaged DNA or, in the event that repair is not possible, to undergo apoptosis. Cyclins bind to CDKs at specific cell cycle phases to activate them: cyclin D-CDK4/6 drives G₁ progression, cyclin E-CDK2 facilitates the G₁/S transition, and cyclin A-CDK2 promotes S-phase progression. In a similar manner, cyclin B-CDK1 regulates the M phase of mitosis. A common feature of many malignancies is the dysregulation of cyclins or the overactivation of CDKs, which results in unchecked cell division. Flavonoids and polyphenols have been shown to modulate the expression or activity of these key regulators upregulating p53 and p21, and downregulating cyclins and CDKs thereby inducing cell cycle arrest and exerting anticancer effects.40
Screening Methods in Cancer Cell Lines
In vitro studies on anticancer agents, including flavonoids and polyphenols, commonly employ a range of established human cancer cell lines (Table 2) that represent different tissue origins and tumor types. These cell lines serve as reliable models for understanding the cellular and molecular mechanisms underlying cancer progression and treatment responses
Table 2: Commonly Used Human Cancer Cell Lines in Screening Flavonoids and Polyphenols
|
Cell Line |
Tissue Origin |
Key Characteristics |
Research Relevance |
|
HeLa |
Cervix (Cervical cancer) |
Highly proliferative, HPV-positive, immortalized line |
Used for general cytotoxicity, apoptosis, and DNA damage response studies.41 |
|
MCF-7 |
Breast (Adenocarcinoma) |
Estrogen receptor-positive (ER+), hormone responsive |
Ideal for studying hormonal effects, breast cancer therapies, and apoptosis pathways.42 |
|
A549 |
Lung (Adenocarcinoma) |
Non-small cell lung cancer (NSCLC) with wild-type p53 shows heightened sensitivity to oxidative stress. |
Frequently used in apoptosis, antioxidant, and anti-metastatic evaluations.43 |
|
HT-29 |
Colon (Adenocarcinoma) |
Moderately differentiated, p53 mutant, COX-2 overexpressing |
Useful for colorectal cancer, inflammation, and differentiation studies.44 |
|
HCT116 |
Colon (Carcinoma) |
p53 wild type, microsatellite instability, highly proliferative |
Model for cell cycle, DNA repair, and apoptosis-related pathways.45 |
|
PC3 |
Prostate (Carcinoma) |
Androgen receptor-negative, highly metastatic |
Suitable for hormone-independent prostate cancer and anti-migratory studies.46 |
|
DU145 |
Prostate (Carcinoma) |
Moderate metastatic potential, p53 mutant |
Used for comparison with PC3, studies on prostate cancer resistance.47 |
|
SKOV-3 |
Ovary (Ovarian carcinoma) |
Estrogen receptor-positive, multidrug resistance potential |
Used in drug resistance, apoptosis, and polyphenol sensitivity screening.48 |
|
U87 |
Brain (Glioblastoma) |
High-grade glioma, aggressive, adherent morphology |
Model for brain tumors, blood-brain barrier, and cytotoxicity of natural compounds.49 |
|
HepG2 |
Liver (Hepatocellular carcinoma) |
p53 wild type, metabolically active hepatocyte-like cells |
Common for hepatotoxicity, antioxidant, and metabolic studies.50 |
|
T47D |
Breast (Ductal carcinoma) |
ER+, PR+, p53 mutant, epithelial morphology |
Used alongside MCF-7 for hormone-responsive cancer screening.51 |
|
Caco-2 |
Colon (Adenocarcinoma) |
Differentiates into enterocyte-like cells, forms tight junctions |
Used in drug absorption, intestinal permeability, and cytotoxicity assessments.52,53 |
In vitro assays for apoptosis and cell cycle analysis
A variety of in vitro assays are employed to evaluate the apoptotic and cell cycle-modulating effects of flavonoids and polyphenols in cancer cell lines (Table.3).
Table 3: In Vitro Assays for Apoptosis and Cell Cycle Analysis
|
Assay |
Principle |
Purpose |
Output/Readout |
|
MTT/XTT Assay |
based on how mitochondrial enzymes in living cells convert tetrazolium salts (MTT or XTT) to coloured formazan |
Evaluates the proliferation and vitality of cells after treatment. |
Absorbance values indicating % of viable cells (colorimetric).53 |
|
Annexin V-FITC/PI Staining |
Phosphatidylserine (early apoptosis) is bound by Annexin V, and PI stains the DNA of late apoptotic and necrotic cells. |
Detects and distinguishes early and late apoptotic cells |
Flow cytometry or fluorescence microscopy quantification.54 |
|
Caspase Activity Assay |
Measures enzymatic cleavage of specific caspase substrates (e.g., caspase-3, -8, -9) to generate fluorescent or colorimetric signals |
Assesses activation of intrinsic and extrinsic apoptotic pathways |
Relative fluorescence or absorbance indicating caspase activation.55 |
|
Flow Cytometry for Cell Cycle |
DNA-binding dyes (e.g., PI) used to quantify total DNA content per cell |
Analyzes cell cycle phases (G₀/G₁, S, G₂/M) to detect arrest. |
Percentage of cells in each phase (G₁, S, G₂/M).56 |
|
Western Blotting |
Protein separation and detection using antibodies against target proteins |
Detects apoptosis/cell cycle regulatory proteins (e.g., Bcl-2, Bax, p53, caspases, cyclins) |
Protein expression levels (band intensity).57 |
|
RT-PCR/qPCR |
Amplifies specific mRNA transcripts using reverse transcription and real-time detection |
Measures gene expression changes related to apoptosis or cell cycle |
Fold change in mRNA expression of genes like p21, Bcl-2, CDK1, etc.58 |
Results
Cancer models – Flavonoids cause cell cycle arrest and death
Flavonoids are well documented for their ability to alter important signaling pathways that contribute to cancer growth, specifically by inducing cell cycle arrest and death. Their structural subclasses, each with distinct bioactivities in cancer cell lines, have a considerable influence on these effects.
Flavones
In a variety of models, flavones including luteolin and apigenin have shown strong anticancer effects. Apigenin induces apoptosis in breast and prostate cancer by activating the intrinsic pathway, inhibiting Bcl-2, and upregulating Bax and caspase-3. Additionally, it inhibits cyclin B1 and CDK1, which causes cell cycle arrest at the G₂/M phase. By inhibiting PI3K/Akt signaling and triggering caspases, lutein has comparable effects on lung and colon cancer cells. G₁ phase arrest results from changes to cyclin D1 and p21. The effects of luteolin, a dietary flavonoid, on HT 29 human colon cancer cells, with an emphasis on cell cycle regulation and death. In a dose-dependent fashion, they reduced the number of viable cells and inhibited DNA synthesis. G phase arrest was caused by the rapid suppression of CDK4 and CDK2 activity by about 38% at 40 μM in 2 hours. This was accompanied by a drop in cyclin D levels and phosphorylation of the retinoblastoma (Rb) protein. The 24-hour therapy substantially accelerated G₂/M arrest by suppressing cyclin B and CDC2 activity. Luteolin enhanced PARP cleavage and activated caspases 3, 7, and 9, while downregulating anti-apoptotic proteins such as p21^CIP1/WAF1, survivin, Mcl-1, Bcl-x_L, and Mdm2, promoting p53-independent apoptosis and cell cycle arrest.59 In SW480 and Caco-2 colon cancer cells, seven apigenin analogues acacetin, chrysin, kaempferol, luteolin, myricetin, naringenin, and quercetin were found to induce G₂/M cell-cycle arrest. They employed DNA flow cytometry to determine that quercetin, kampherol, and luteolin likewise caused a significant G₂/M arrest at 5–30 µM, In contrast, acacetin and chrysin boosted the G₂/M population in SW480 cells in a dose-dependent manner (0–80 µM over 48 hours). The impact diminished when the concentration exceeded 40 µM. Conversely, the Caco‑2 line did not exhibit notable arrest under similar conditions . With the exception of myricetin and naringenin, the majority of analogues significantly reduced cell numbers over time and at varying doses. Furthermore, when these analogs (5–30 µM) were combined with 20 µM apigenin, there was an additive effect a further ~22 % increase in G₂/M arrest compared to apigenin alone, though this synergy became antagonistic at higher concentrations (> 40 µM) High doses of individual analogs or their combinations also decreased cell viability, as measured by Trypan blue exclusion. Interestingly, cellular uptake of some analogs increased at ≥40 µM, but this did not correlate with their cell-cycle effects. Collectively, these findings suggest that apigenin-related flavonoids can cooperatively induce G₂/M arrest and reduce proliferation in colorectal cancer cells, highlighting their potential for combinatorial chemoprevention strategies.60
Flavonols
Flavonols like quercetin and kaempferol are well-studied for their anticancer effects, notably inducing cell cycle arrest and apoptosis. Quercetin significantly inhibits MCF-7 breast cancer cell growth by triggering apoptosis and synchronized cell cycle arrest. Treatment with quercetin (IC₅₀ ≈ 92 µM at 48 h) initially induced a transient M‑phase accumulation, evidenced by increased mitotic index and heightened cyclin B1/Cdc2 kinase activity, followed by sustained G₂ arrest characterized by reduced cyclin B1 levels and diminished Cdc2 activity. Simultaneously, quercetin upregulated the CDK inhibitor p21^CIP1/WAF1, which bound to and suppressed the cyclin B1–Cdc2 complex, while notable p53-independent growth suppression was observed. The flavonoid also induced significant apoptosis, demonstrated by increased sub‑G₁ DNA content, chromatin condensation, and DNA fragmentation, all of which were markedly reduced upon antisense-mediated p21 inhibition. Thus, quercetin was shown to execute a dual anticancer mechanism in MCF‑7 cells: halting proliferation via reversible mitotic arrest and G₂ checkpoint activation, and triggering apoptosis that is tightly linked to p21, but independent of p53 status.61Kaempferol significantly affects DNA integrity, apoptosis, cell cycle regulation, and proliferation in MDA-MB-231 triple-negative breast cancer (TNBC) cells through its anticancer mechanisms. The flavonol was more successful in inhibiting cell proliferation in TNBC MDA MB 231 cells than in ER-positive BT474 cells. Kaempferol significantly changed the distribution of cell cycles after 48 hours of treatment, boosting G₂-phase cells from 9.3% to 37.5% and decreasing G₁-phase cells from 85.5% to 51.4%, indicating a robust induction of G₂/M arrest. At the same time, apoptosis and DNA damage indicators were significantly increased: phosphorylated ATM, cleaved versions of caspase-9 and caspase-3, and γH2AX, a DNA double-strand break indicator, were all upregulated. By stopping the cell cycle at the G₂/M checkpoint, triggering DNA damage reactions, and triggering mitochondrial-mediated apoptosis, kaempferol prevents the growth of TNBC cells. Given its selective potency in aggressive breast cancer subtypes, kaempferol shows notable potential as a therapeutic agent against TNBC.62In HL 60 human acute leukemia cells, structurally identical flavonoids with antioxidant qualities influence apoptosis and cell cycle progression. The study examined six flavonoids: quercetin, myricetin, kaempferol, isorhamnetin, 3′ methylquercetagetin, and taxifolin. The results showed that while 3′ methylquercetagetin and taxifolin had negligible effects, quercetin, myricetin, kaempferol, and isorhamnetin significantly decreased cell growth. the role of specific structural components, including the 6 hydroxyl group and the C2-C3 double bond, in mediating antiproliferative activity. Flow cytometry data showed that myricetin and quercetin were the most effective apoptosis inducers. . Myricetin, isorhamnetin, and kaempferol induced concentration- and time-dependent increases in G₀/G₁ and S phase cells, while quercetin alone triggered G₂/M arrest. Notably, antioxidant assays revealed that myricetin showed the greatest capacity to scavenge DPPH radicals, though it was less effective at reducing intracellular ROS in response to menadione stress. On the other hand, kaempferol was very effective at scavenging ROS when cells were under stress. Apart from their antioxidant capacity, small structural differences may affect flavonoids’ capacity to stop the cell cycle’s advancement and cause leukemia cells to undergo apoptosis.63
Flavanones
Naringenin, a citrus flavonoid, shows anticancer effects by inducing apoptosis and cell cycle arrest across various cancer cell lines. In HepG2 cells, it inhibits proliferation, increases p53, and induces biphasic arrest at G₀/G₁ and G₂/M phases. Apoptosis is evidenced by nuclear condensation, sub-G₁ population increase, cytochrome c release, elevated Bax/Bcl-2 ratio, and caspase-3 activation, suggesting its potential as a liver cancer therapeutic.64 Naringenin’s anticancer actions in A431 human epidermoid carcinoma cells revealed a ROS-mediated apoptotic mechanism in conjunction with G₀/G₁ death reduction in cell viability that is dose-dependent (p < 0.01) and accompanied by apoptotic hallmarks such as DNA fragmentation and nuclear condensation. Intracellular ROS generation increased markedly (p < 0.001), leading to mitochondrial membrane depolarization. Flow cytometry revealed caspase-3 activation and G₀/G₁ cell cycle arrest, indicating that naringenin inhibits epidermoid carcinoma cell growth through oxidative stress, mitochondrial dysfunction, caspase activation, and cell cycle blockade.65 Citrus-derived flavanones naringin and naringenin have several anticancer properties, including the capacity to regulate key cellular functions that inhibit cancer, including invasion, metastasis, angiogenesis, proliferation, apoptosis, and cell cycle arrest. By disrupting multiple signaling pathways, these flavanones produce a variety of pleiotropic effects, including regulating inflammation and cytokine production, inhibiting growth factor receptors (EGFR, VEGF), and suppressing downstream kinases (PI3K/Akt, NF-κB, MAPKs), which results in cell cycle blockade and intrinsic and extrinsic apoptosis. In particular, they cause cytochrome c release, activate caspases-8 and -9, and upregulate pro-apoptotic proteins (Bax, Bak); these actions result in effector caspase-3, which initiates apoptosis. Additionally, via altering cell cycle regulators including p53, p21, CDKs, and cyclins, naringin and naringenin cause arrest at the G₀/G₁ and G₂/M phases. Their capacity to stop angiogenesis, invasion, and metastasis further increases their therapeutic potential.66
Isoflavones
Isoflavones, particularly genistein, are plant-derived polyphenolic compounds predominantly found in soybeans and legumes, and are well-known for their estrogen-like activity and strong anticancer potential. Genistein induced apoptosis, G₂/M arrest, and reduced proliferation in HCT 116 (p53 wild-type) and SW 480 (p53 mutant) colon cancer cells. In HCT 116, it dose- and time-dependently increased G₂/M-phase cells, downregulated CDC2 and CDC25A, and activated ATM, p53, p21^WAF1/CIP1, and GADD45α. Using flow cytometry with Annexin V/PI labelling to validate enhanced apoptosis, especially in p53-competent cells, the p53-dependency of this response was shown. Furthermore, real-time PCR and Western blot investigations revealed that genistein markedly downregulated anti-apoptotic factors like Bcl-2 while upregulating cell cycle checkpoint genes including CDKN1B and BRCA1/2. This implies that a crucial regulatory network mediating the chemopreventive effect of genistein is the ATM/p53–p21 axis. These findings illustrate that genistein can exert dual anticancer effects in colon cancer halting proliferation through G₂/M arrest and triggering apoptosis via the DNA damage response pathway, with efficacy influenced by p53 status.67 In human ovarian cancer cells, genistein induces DNA damage, G₂/M cell cycle arrest, and death via triggering DNA damage checkpoint mechanisms (HO 8910). G₂/M phase arrest was enforced by phosphorylating and activating ATM and ATR, which in turn activated checkpoint kinases Chk2 and Chk1. This, in turn, phosphorylated and inactivated Cdc25C/A phosphatases and inhibited Cdc2. Genistein promoted apoptosis by downregulating phosphorylated Akt, enhancing p53 activation, and reducing Bcl-2/Bax and Bcl-xL/Bax ratios. According to these results, genistein uses DNA damage response systems, particularly the ATM–Chk2–Cdc25 and ATR–Chk1–Cdc25 pathways, to halt the advancement of the cell cycle during G₂/M. When the damage is irreversible, it triggers mitochondrial-mediated death. This dual action reinforces genistein’s antiproliferative and pro-apoptotic potential in ovarian cancer via genotoxic stress.68 Numerous cell cycle modulation and cell death pathways were revealed by the biological activity of novel polysaccharide complexes and genistein derivatives in MCF 7 breast cancer cells and HL 60 leukemia cells. While XG G shown no antiproliferative activity, two analogues, IFG 027 and IFG 043, as well as the complex SPG G, demonstrated antiproliferative effects that were on par with or even stronger than genistein. Interestingly, the analogues IFG 027 and IFG 043 actually reduced cell accumulation in G₂/M, indicating different effects on cell cycle regulation from genistein and the SPG G complex, which caused G₂/M phase arrest. Both genistein and its derivatives triggered caspase-3, but SPG G encouraged apoptosis that was independent of caspase 3. Apoptotic studies revealed that SPG G, IFG 027, and IFG 043 produced phosphatidylserine externalisation and decreased mitochondrial membrane potential, indicating induction of apoptosis. However, the absence of typical apoptotic markers and the production of acidic vesicular organelles by XG G suggested either autophagic or non-apoptotic cell death. These findings illustrate that structural modifications of genistein can lead to distinct anticancer activities, from conventional apoptosis to alternative death pathways, and emphasize the potential of tailored derivatives for targeted cancer therapeutics.69 Several flavonoids, including quercetin, kaempferol, apigenin, luteolin, naringenin, and genistein, exhibit strong anticancer effects by inducing cell cycle arrest and apoptosis in various in vitro cancer models such as MCF-7, MDA-MB-231, HT-29, HCT116, A549, PC3, DU145, HepG2, HeLa, and HL-60. These effects are dose-dependent, typically observed between 10–100 µM. For example, quercetin causes G2/M arrest and apoptosis in MCF-7 cells at ~90 µM, while naringenin induces G0/G1 arrest and mitochondrial death in HepG2 cells at 50 µM. Genistein (30–60 µM) leads to G2/M arrest and caspase-mediated apoptosis in colon and ovarian cancer cells.70 Flavonoids modulate multiple apoptotic proteins, activating initiator (caspase-8, -9) and executioner (caspase-3) caspases. They downregulate anti-apoptotic proteins (Bcl-2, Bcl-xL) and upregulate pro-apoptotic factors (Bax, Bak, cytochrome c). Additionally, flavonoids influence p53 directly or via ATM/ATR, increasing p21^WAF1/CIP1 and p27^KIP1 transcription, thereby inhibiting cyclin–CDK complexes and inducing cell cycle arrest at G0/G1 or G2/M depending on cell type and flavonoid. For example, apigenin and luteolin suppress cyclin D1/CDK4/6 to enforce G1 arrest, while kaempferol and genistein downregulate cyclin B1/CDK1 to cause G2/M arrest.71 Flavonoids disrupt survival signaling cascades such PI3K/Akt, NF-κB, and MAPK/ERK pathways in addition to these traditional apoptotic and cell cycle routes. Cell survival is decreased when Akt phosphorylation is suppressed, while pro-inflammatory and anti-apoptotic gene expression is decreased when NF-κB transcriptional activity is inhibited. The regulation of MAPK by flavonoids influences the ratio of apoptosis to proliferation. These pleiotropic molecular effects not only result in cytotoxicity and growth arrest, but also sensitize cancer cells to chemotherapeutic agents, making flavonoids potential candidates for combination therapies.72 Taken together, the evidence underscores that flavonoids act as multifunctional anticancer agents, capable of targeting key hallmarks of cancer such as uncontrolled proliferation, evasion of apoptosis, and dysregulated cell cycle progression. Their attractiveness as natural substances for cancer chemoprevention and adjuvant therapy approaches is increased by their selective toxicity to cancer cells and minimal toxicity to healthy cells.
Polyphenols’ Role in Cell Cycle Arrest and Apoptosis
Resveratrol, curcumin, gallic acid, and ellagic acid are polyphenols with strong anticancer properties, primarily through apoptosis induction and cell cycle regulation. Found in fruits, vegetables, spices, and medicinal plants, these compounds target key survival pathways in cancer cells.
Resveratrol, a stilbene in berries and grapes, induces apoptosis via mitochondrial and death receptor pathways by activating caspases-3, -8, and -9, increasing Bax, and releasing cytochrome c. It also modulates cyclin A, cyclin B1, CDK1, p21, and enhances p53 signaling, leading to S or G2/M phase arrest in prostate cell lines like PZ-HPV-7, LNCaP, and PC-3. Resveratrol significantly increased apoptosis and decreased proliferation in cancer cell lines, which led to a buildup of cells in the G₀/G₁ phase over time. A higher Bax/Bcl-2 ratio suggested the presence of the intrinsic apoptotic pathway, and both LNCaP and PC-3 cells experienced apoptosis as a result of caspase-9 and -3 activation. Resveratrol has been demonstrated to decrease CDK4 expression, cyclin D1/CDK4 kinase activity, and cyclins D1 and E in LNCaP cells. In contrast, both PC-3 and LNCaP cells displayed decreased levels of cyclin B and Cdk1, as well as decreased cyclin B/Cdk1 kinase activity. Additionally, resveratrol increased levels of p53, p21, and p27 but only in LNCaP cells suggesting a more robust G₀/G₁ checkpoint control in androgen-sensitive cells. Together, these molecular changes explain the observed cell cycle arrest and apoptosis induction, demonstrating that resveratrol’s anticancer actions entail unique pathways impacted by p53-related pathway activation and androgen sensitivity.73HCT 116 and Caco 2 colorectal cancer cells experienced considerable G₁/S phase cell cycle arrest and caspase-dependent death as a result of resveratrol’s antiproliferative and pro-apoptotic actions. Significant cell accumulation at the G₁/S checkpoint and a marked decrease in cell viability were the outcomes of treatment with increasing resveratrol concentrations (10-100 µM for 24 hours). Additionally, there was a dose-dependent decrease in important cell cycle regulators like cyclin D1, CDK2, CDK4, PCNA, and an increase in p21 expression. Annexin V/PI staining and Western blot confirmed apoptosis via increased cleaved caspase-9, caspase-7, and PARP, indicating activation of the intrinsic pathway. Notably, Caco‑2 cells exhibited greater sensitivity to resveratrol compared to HCT‑116 cells, demonstrating more pronounced effects on both proliferation inhibition and cell cycle arrest. These findings demonstrate how strongly resveratrol inhibits colorectal cancer cell growth by concurrently targeting the caspase and cyclin CDK pathways, confirming its potential as a chemopreventive medication against colon neoplasia.74 Resveratrol’s effects on human neuroblastoma in a mouse xenograft model include both in vitro cytotoxicity and tumour growth suppression. Resveratrol induced G₀/G₁ arrest in neuroblastoma cells by upregulating p21 and downregulating CDK2, CDK4, and cyclin D1. Apoptosis was marked by sub-G₁ accumulation, chromatin condensation, DNA fragmentation, caspase-9/3 activation, and PARP cleavage. These cellular findings were supported by in vivo studies showing that resveratrol administration significantly suppressed tumor growth in immunodeficient mice bearing neuroblastoma xenografts, without inducing major systemic toxicity . Together, the data highlight resveratrol’s dual action hindering cell cycle progression and inducing apoptosis as key contributors to its antitumor efficacy against neuroblastoma both in cell culture and animal models.75 Resveratrol efficiently and dose-dependently suppressed cell proliferation (50-250 µM), and its cytotoxic effects on 4T1 murine breast cancer cells demonstrated that it was more harmful to cancer cells than to normal cells. Following resveratrol administration, transcriptome profiling identified 330 differentially expressed genes related to apoptosis and cell cycle regulation, of which 103 were upregulated and 227 were downregulated. Gene expression and pathway analysis revealed that resveratrol induced S-phase arrest, which led to a rise in S-phase cells and a fall in G₀/G₁ phase cells. Resveratrol induced strong anti-proliferative effects in breast and bladder cancer cells by causing S and G₁ phase arrest, respectively, and promoting apoptosis. It increased early and late apoptotic populations, activated caspases 9 and 3, cleaved PARP, downregulated p-Rb, cyclin D1, CDK4, and upregulated p21.76 At a dose of 20 mg/kg/day, resveratrol also significantly suppressed tumour growth in T24 xenografts by reducing production of VEGF and FGF 2, decreasing Akt phosphorylation while increasing p38 MAPK activity, and modulating important signaling pathways. According to these findings, resveratrol is a strong contender for bladder cancer chemoprevention since it exhibits anticancer properties through a complex mechanism that includes cell cycle arrest, mitochondrial-mediated apoptosis, and inhibition of survival and angiogenic pathways.77
Curcumin, derived from turmeric (Curcuma longa), exerts pleiotropic effects by inhibiting the NF-κB, PI3K/Akt, and STAT3 pathways, leading to apoptosis and G2/M arrest. It suppresses cyclin D1, Bcl-2, and survivin, while enhancing cleaved PARP and caspase activation. Curcumin’s anticancer actions on BxPC 3 human pancreatic cancer cells at a physiologically appropriate dose of 2.5 μM showed a DNA damage-mediated mechanism that causes apoptosis and G₂/M cell cycle arrest. After 24 hours of treatment, increased phosphorylation of ATM, Chk1, and H2A.X indicated DNA damage response activation, while reduced DNA polymerase-β suggested impaired DNA repair. This signaling cascade resulted in Cdc25C phosphorylation (Ser‑216) and subsequent downregulation of cyclin B1/CDK1 complexes, effectively enforcing G₂/M arrest; these effects were confirmed by flow cytometry. Concomitant apoptotic induction was evidenced by caspase-3 activation and PARP cleavage, selectively affecting cancerous cells but sparing normal HPDE‑6 pancreatic ductal epithelial cells. Importantly, siRNA reduction of ATM or Chk1 abolished curcumin’s effects by preventing apoptosis, G₂/M arrest, and phosphorylation of ATM/Chk1/Cdc25C. The importance of the ATM–Chk1–Cdc25C axis in curcumin’s anticancer properties is shown by this.78 Curcumin inhibited proliferation of PANC-1 and BxPC-3 cells in a dose- and time-dependent manner, induced apoptosis, and caused G₂/M phase arrest, as shown by CCK-8 and flow cytometry analyses. Immunofluorescence and Western blot investigations showed autophagosome production and elevation of LC3-II and Bax, while Bcl-2 was downregulated, indicating a link between autophagy and apoptosis. A strong inverse correlation between the LC3-II/Bax ratio and cell viability implied that both autophagy and apoptosis contribute cooperatively to curcumin’s cytotoxic effects. These findings position curcumin as a multitargeted anticancer agent that simultaneously activates autophagic pathways, mitochondrial apoptosis, and G₂/M cell cycle arrest, particularly at higher doses underscoring its potential for therapeutic development in pancreatic cancer.79 Curcumin’s antiproliferative actions in human osteosarcoma (HOS) cells show that, at an IC₅₀ of roughly 4 μg/mL, it causes sequential cell cycle arrest and death. Flow cytometry analysis revealed that curcumin treatment results in biphasic arrest at both G₁/S and G₂/M phases, followed by extensive apoptotic cell death, characterized by nuclear fragmentation and DNA degradation. Mechanistically, cyclin B1 (G₂/M) and cyclin D1 (G₁/S) and CDC2 (CDK1) were downregulated in conjunction with these cell cycle blocks. Apoptosis was facilitated by caspase 3 activation and PARP cleavage.80
Ellagic acid, found in pomegranates and berries, induces G₀/G₁ arrest by inhibiting cyclin D1 and upregulating p21 and p27. It generates ROS, disrupts mitochondrial potential, and triggers apoptosis in MCF-7 cells via the TGF-β/Smad3 pathway. EA reduces cell proliferation by downregulating cyclins A2 and E2 and overexpressing CDK inhibitors such p21, CIP1, p15, and p19. Together, these inhibitors were able to eliminate cyclin CDK complexes and halt the cell cycle’s progression. Using cDNA microarray analysis after 6, 12, and 24-hour EA exposure, the authors observed major transcriptional shifts with over 4,700 genes altered and subsequently validated 16 genes within the TGF‑β/Smad signaling axis via RT‑PCR and Western blotting. Activation of this pathway through Smad3 phosphorylation emerged as the core mechanism: EA enhanced TGF‑β signaling, which in turn increased expression of CDK inhibitors while suppressing cyclins, effectively enforcing G₀/G₁ arrest. Additionally, this cell cycle blockade correlated with induction of apoptosis, positioning TGF‑β/Smad3 as a critical effector of EA’s anticancer activity in breast cancer cells.81The combined effects of ellagic acid, quercetin, and resveratrol polyphenols commonly found in muscadine grapes on MOLT‑4 human leukemia cells and discovered notable synergistic pro-apoptotic and antiproliferative activities. While combinations greatly increased caspase 3 activation and decreased cell proliferation more than the sum of their individual effects, each drug alone caused transitory cell cycle arrest. Isobolographic analysis confirmed synergy with combination indices of 0.64 (ellagic acid + resveratrol) and 0.68 (quercetin + resveratrol).82Gallic acid, present in tea, grapes, and gallnuts, induces G₂/M arrest and mitochondrial apoptosis via ROS, Bax upregulation, and Bcl-2 downregulation. In DU145 prostate cancer cells, it reduced proliferation dose- and time-dependently by activating ATM, H2A.X, and Chk2, indicating a DNA damage response. G₂/M phase arrest resulted from this activation’s inhibition of Cdc25A/C phosphatases’ and Cdc2 kinase’s phosphorylation. The upstream ATM-Chk2 signaling pathway was validated when caffeine, an ATM inhibitor, abolished these effects. Additionally, gallic acid cleaved PARP, caspase-9, and caspase-3, suggesting that it employed mitochondrial pathways to trigger apoptosis. It’s interesting to note that apoptosis continued even after pan-caspase inhibition, which raises the prospect of caspase-independent processes.83
Gallic acid’s antiproliferative and pro-apoptotic effects on HL 60 human promyelocytic leukemia cells led to G₀/G₁ cell cycle arrest and a dose- and time-dependent reduction in cell viability. Mechanistically, gallic acid elevated levels of CDK inhibitors p21 and p27 while concurrently suppressing cyclins D and E, effectively halting cell cycle progression. Additionally, the compound triggered DNA damage, evidenced by DAPI staining and comet assay, and induced mitochondrial dysfunction with reduced membrane potential and increased intracellular Ca²⁺ . The process of apoptosis included both mitochondria-dependent mechanisms such as the release of cytochrome c, AIF, and endonuclease G, as well as death receptor pathways such as Fas/FasL, caspase 8, and Bid. These changes were verified at the protein and mRNA levels using real-time PCR and Western blotting.84 Together, these polyphenols exert multi-targeted anticancer effects, modulating redox balance, gene expression, and cell signaling to inhibit cancer cell proliferation and enhance apoptotic responses. Their natural origin, low toxicity, and broad spectrum of activity position them as attractive candidates for both preventive and therapeutic strategies in oncology. According to mechanistic study, these drugs work by blocking cyclins and cyclin-dependent kinases, activating intrinsic mitochondrial apoptotic pathways, and changing key regulators like p53, Bax, Bcl-2, and caspases. Depending on the kind of cancer cell, resveratrol, for example, induces caspase-mediated apoptosis through the ATM/Chk2 or PI3K/Akt suppression pathways, leading to either G1/S or G2/M phase arrest. Curcumin exhibits similar effects, downregulating cyclin B1 and CDK1 and activating the ATM/Chk1 axis to cause cell cycle arrest and death.85 Moreover, polyphenols can cooperate with conventional chemotherapy medications such as doxorubicin, cisplatin, and 5-fluorouracil. These combinations often enhance drug efficacy, reduce resistance, and lower the effective therapeutic dose, thereby minimizing systemic toxicity. Studies have demonstrated that co-treatment with polyphenols not only augments cancer cell death but also modulates drug transporters and survival signaling pathways, improving overall therapeutic outcomes.86 Notably, polyphenols exhibit a degree of selectivity, exerting cytotoxic effects preferentially on malignant cells while sparing normal cells. This differential action is attributed to altered redox status, mitochondrial integrity, and proliferation rates in cancer cells. Such selectivity makes polyphenols attractive candidates for integrative cancer therapy, supporting their continued investigation in preclinical and clinical settings.87
Discussion
Synergistic and Combinatorial Effects
The combination of flavonoids and polyphenols with conventional cancer therapies has gained significant attention for enhancing therapeutic efficacy and minimizing side effects. Combining flavonoids or polyphenols with chemotherapeutic drugs like doxorubicin, 5-fluorouracil, and cisplatin has been shown in numerous trials to provide synergistic effects, such as increased apoptosis, decreased drug resistance, and greater susceptibility of cancer cells.88 Quercetin and curcumin sensitize cancer cells to cisplatin by downregulating Bcl-2 and upregulating Bax and caspases.89 Similarly, by regulating oxidative stress and blocking survival signaling pathways including NF-κB and PI3K/Akt, resveratrol increases the cytotoxic potential of doxorubicin.90 Flavonoids such as genistein and luteolin have been shown to function as radiosensitizers in the setting of radiation therapy by increasing DNA damage, blocking DNA repair processes, and encouraging cell cycle arrest. This not only boosts the radiation response in tumor cells but also protects normal cells through their antioxidant properties, thereby improving the therapeutic window.91 Furthermore, combining multiple phytochemicals such as ellagic acid, gallic acid, and resveratrol has demonstrated additive or even synergistic effects in various cancer models. These combinations enhance cellular stress, disrupt multiple signaling pathways, and offer a broader spectrum of anticancer action. The combinatorial use of polyphenols with existing therapies thus holds promise for overcoming resistance, reducing toxicity, and achieving more effective cancer management, paving the way for integrative and personalized oncology strategies. Numerous studies have documented the enhanced efficacy and reduced toxicity of combining flavonoids and polyphenols with conventional cancer therapies. These organic chemicals not only improve the therapeutic efficacy, but they also reduce the unfavourable side effects typically associated with radiation and chemotherapy. (Table.4)
Table 4: Enhanced Efficacy and Reduced Toxicity of Flavonoid/Polyphenol-Based Combination Therapies in Cancer
|
Flavonoid/ Polyphenol |
Combined With |
Cancer Type |
Enhanced Efficacy |
Reduced Toxicity |
Ref |
|
Quercetin |
Cisplatin |
Ovarian, Lung Cancer |
↑ Apoptosis via mitochondrial pathway↓ MDR protein activity |
↓ Nephrotoxicity by antioxidant protection |
89 |
|
Curcumin |
Doxorubicin |
Breast Cancer |
Inhibited NF-κB and PI3K/Akt↑ Apoptotic signaling |
↓ Cardiotoxicity |
88 |
|
Resveratrol |
5-FU |
Colorectal Cancer |
↑ S-phase arrest and caspase activation |
↓ Gastrointestinal mucosal damage |
92 |
|
Genistein |
Radiotherapy |
Prostate, Pancreatic Cancer |
↑ Radiosensitization by cell cycle arrest and DNA damage |
↓ Inflammation and fibrosis in normal tissues |
91 |
Limitations and Challenges
Despite the promising anticancer properties of polyphenols and flavonoids demonstrated in numerous preclinical studies, several limitations and challenges hinder their full therapeutic translation. One of the primary concerns is their poor bioavailability and metabolic stability. Many polyphenols, including curcumin and quercetin, undergo rapid metabolism and systemic elimination, resulting in low plasma concentrations that may not replicate the effects observed in vitro. Additionally, the discrepancy between in vitro, in vivo, and clinical outcomes poses a major challenge. While cell culture models allow for mechanistic insights, they fail to fully mimic the complexity of human tumors, such as tumor microenvironment interactions, immune responses, and pharmacokinetics. Another significant hurdle is the lack of standardized screening protocols. Different studies often use varying cell lines, dosages, time points, and assay methods, making it difficult to compare or replicate findings across research groups. Furthermore, the absence of large-scale, head-to-head comparative studies on flavonoids and polyphenols limits the ability to rank their efficacy, safety profiles, and mechanisms systematically. The use of uncharacterized plant extracts in some studies also complicates the identification of active constituents and their individual roles. To overcome these obstacles, standardized in vitro and in vivo models, sophisticated delivery methods to boost bioavailability, and well-planned clinical trials are needed. These problems need to be fixed if polyphenols and flavonoids are to reach their full therapeutic potential in the treatment of cancer.
Future Perspectives
Future research into the anticancer potential of flavonoids and polyphenols is poised to benefit greatly from advances in nanotechnology and targeted delivery systems. Nanocarriers like as liposomes, solid lipid nanoparticles, and polymeric micelles can assist overcome one of the main barriers to the widespread use of these treatments by dramatically increasing their bioavailability, stability, and tumor-specific accumulation. Furthermore, a strong platform for deciphering the intricate molecular mechanisms impacted by polyphenols and locating biomarkers of response is provided by the combination of omics technologies like as transcriptomics, proteomics, and metabolomics. These tools can facilitate the development of precision oncology strategies, where patient-specific flavonoid or polyphenol responsiveness is predicted based on molecular signatures. There is also increasing interest in tailoring polyphenol-based interventions as part of personalized cancer therapy, leveraging individual metabolic profiles, genetic predispositions, and tumor characteristics. Such approaches could allow for optimized combination therapies, lower toxicity, and improved patient outcomes. However, the realization of these strategies requires robust in vivo validation in animal models and, more importantly, well-structured clinical trials to establish safety, efficacy, and dosage guidelines. Presently, a limited number of clinical studies have explored these compounds in isolation or adjunctively, and their outcomes underscore the need for larger, controlled trials with standardized endpoints. As interdisciplinary collaboration grows between pharmacologists, oncologists, and systems biologists, flavonoid and polyphenol research holds great promise for next-generation, personalized anticancer therapies.
Conclusion
Many flavonoids and polyphenols found in dietary and medicinal plants influence essential biological processes such cell cycle arrest and apoptosis induction, which has potent anticancer effects. These natural chemicals have shown strong dose-dependent antiproliferative effects on a variety of human cancer cell lines, including those from the liver, ovary, breast, colon, lung, and leukemia. According to their mechanism of action, they interfere with important pathways like PI3K/Akt, NF-κB, and MAPKs, control important signaling molecules like Bcl-2 family proteins, caspases, cyclins, CDKs, and tumour suppressors like p53 and p21, and work through both intrinsic and extrinsic apoptotic pathways. Their capacity to halt tumor cell progression and promote programmed cell death underscores their potential as promising agents for cancer chemoprevention and therapy. Reaffirming their significance, flavonoids and polyphenols represent a non-toxic, readily available, and multi-targeted approach to combating cancer. Their selective action on malignant cells and synergistic effects with conventional therapies highlight their utility as adjunct treatments in modern oncology. However, despite encouraging in vitro findings, the translation of these compounds into clinical applications remains limited due to challenges related to bioavailability, metabolism, and mechanistic complexity. Therefore, further in-depth mechanistic studies, pharmacokinetic evaluations, and clinical investigations are essential to validate their therapeutic efficacy and optimize their use in evidence-based integrative cancer care and treatment strategies.
Acknowledgement
The authors express their thanks to Saveetha College of Pharmacy – SIMATS for providing the necessary facilities to carry out this research work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author’s Contribution
- Binoy Varghese Cheriyan: Visualization, Supervision, Project Admininstration.
- KanigaPandi: Writing – Original Draft.
- Dharshan murali, Nesayan Boobalan, Lini priyadharshini christopher jebaraj : Funding Acquisition, Resources.
- Jeevitha Harikrishnan , Deepshikaa Kannan: Data Collection, Analysis.
- Shamyuktha Munuswamy , Shagul Hameed Manaksha: Analysis.
References
- Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71(3):209-249. doi:10.3322/caac.21660
CrossRef - Gelband H, Sankaranarayanan R, Gauvreau CL, et al. Costs, affordability, and feasibility of an essential package of cancer control interventions in low-income and middle-income countries: key messages from Disease Control Priorities, 3rd edition. Lancet. 2016;387(10033):2133-2144. doi:10.1016/S0140-6736(15)00755-2
CrossRef - Ferlay J, Ervik M, Lam F, et al. Global cancer observatory: cancer today. Lyon: International agency for research on cancer. 2020;20182020.
- Inada H, Li Q, Bachani A, et al. Forecasting global road traffic injury mortality for 2030. Injury prevention. 2020;26(4):339-43.
CrossRef - Vicini A, Landrigan P, Straif K. The Rising Global Cancer Pandemic–Complete Book. Journal of Moral Theology. 2022;2(CTEWC Book Series 2):i-221.
CrossRef - Wild CP, Weiderpass E, Stewart BW, eds. World Cancer Report: Cancer research for cancer prevention. Lyon (FR): International Agency for Research on Cancer. 2020.
- Kalyani K, Nagaraj M. Comparing the phytochemical analysis of amaranthus and novel pineapple peel to determine the flavonoids concentration. Interactions. 2024;245(1):247.
CrossRef - Srimathi B, Roy A, Elumalai P. Chemotherapeutic Effect of Rutin on TGF-β/SMAD2 Signalling Molecules Gene Expression in Oral Cancer Cells. Oral & Maxillofacial Pathology Journal. 2024;15(2).
- Javith I, Kumar D RS, Parthasarathy PR, et al. Anti-proliferative and apoptotic effect of papaya leaf and green tea mediated silver nanoparticles using HCT-116 colon cancer cells. Frontiers in Health Informatics. 2024;13(3).
- Bharadwaj N, Manimuthu MS, Vimal S, et al. Evaluation of In vitroAnti-Cancer Activity of Methanolic Leaf Extract of Phoenix pusillaon Colon Cancer Cell Line. J Pharm Bioallied Sci. 2024;16(Suppl 2):S1181-S1185. doi:10.4103/jpbs.jpbs_522_23
CrossRef - Rasouli H, Farzaei MH, Khodarahmi R. Polyphenols and their benefits: A review. International journal of food properties. 2017;20(sup2):1700-41.
- Singla RK, Dubey AK, Garg A, et al. Natural Polyphenols: Chemical Classification, Definition of Classes, Subcategories, and Structures. J AOAC Int. 2019;102(5):1397-1400. doi:10.5740/jaoacint.19-0133
CrossRef - Cheynier V. Phenolic compounds: from plants to foods. Phytochemistry reviews. 2012;11(2):153-77.
CrossRef - Palanivelu P, Jagadesan V, Vijayaraghavan R, Rajkumar D, Krishnamoorthy M, Rajakumar S. Terminalia Arjuna bark extract reduces high-fat diet induced cardiac damage in Wistar rats by altering biochemical and histological parameters. Indian J Pharm Educ Res. 2023;57(1):83-93.
CrossRef - Shahidi F, Yeo J. Bioactivities of Phenolics by Focusing on Suppression of Chronic Diseases: A Review. Int J Mol Sci. 2018;19(6):1573. Published 2018 May 25. doi:10.3390/ijms19061573
CrossRef - Rashmi HB, Negi PS. Phenolic acids from vegetables: A review on processing stability and health benefits. Food Res Int. 2020;136:109298. doi:10.1016/j.foodres.2020.109298
CrossRef - Harborne JB. The flavonoids: advances in research since.1980.
- Kumar D, Roshini LT, Sindya J, Perumal E. Assessment of Rutin’s Anti-Metastatic Potential: Targeting the CXCL8/CXCR2 Chemokine Signaling Pathway in Oral Cancer Cell Line.
- Sandu M, Bîrsă LM, Bahrin LG. Flavonoids–small molecules, high hopes. Acta Chemica Iasi. 2017;25(1):6-23.
CrossRef - Nikolić IL, Savić-Gajić IM, Tačić AD, et al. Classification and biological activity of phytoestrogens: A review. Adv. Technol. 2017;6:96-106.
CrossRef - Zaheer K, Humayoun Akhtar M. An updated review of dietary isoflavones: Nutrition, processing, bioavailability and impacts on human health. Crit Rev Food Sci Nutr. 2017;57(6):1280-1293. doi:10.1080/10408398.2014.989958
CrossRef - Calderón-Oliver M, Ponce-Alquicira E. Fruits: A source of polyphenols and health benefits. InNatural and artificial flavoring agents and food dyes.2018; 189-228. Academic Press.
CrossRef - Mandalari G, Bennett RN, Bisignano G, et al. Characterization of flavonoids and pectins from bergamot (Citrus bergamia Risso) peel, a major byproduct of essential oil extraction. J Agric Food Chem. 2006;54(1):197-203. doi:10.1021/jf051847n
CrossRef - Wrolstad RE. Anthocyanin pigments—Bioactivity and coloring properties. Journal of Food Science. 2004;69(5):C419-25.
CrossRef - Welch CR, Wu Q, Simon JE. Recent Advances in Anthocyanin Analysis and Characterization. Curr Anal Chem. 2008;4(2):75-101. doi:10.2174/157341108784587795
CrossRef - Krga I, Milenkovic D. Anthocyanins: From Sources and Bioavailability to Cardiovascular-Health Benefits and Molecular Mechanisms of Action. J Agric Food Chem. 2019;67(7):1771-1783. doi:10.1021/acs.jafc.8b06737
CrossRef - Cutrim CS, Cortez MA. A review on polyphenols: Classification, beneficial effects and their application in dairy products. International Journal of Dairy Technology. 2018;71(3):564-78.
CrossRef - Jiang N, Doseff AI, Grotewold E. Flavones: From Biosynthesis to Health Benefits. Plants (Basel). 2016;5(2):27. Published 2016 Jun 21. doi:10.3390/plants5020027
CrossRef - Shen T, Wang XN, Lou HX. Natural stilbenes: an overview. Nat Prod Rep. 2009;26(7):916-935. doi:10.1039/b905960a
CrossRef - Chou YC, Ho CT, Pan MH. Stilbenes: chemistry and molecular mechanisms of anti-obesity. Current Pharmacology Reports. 2018;4(3):202-9.
CrossRef - Popa VI, Dumitru M, Volf I, et al. Lignin and polyphenols as allelochemicals. Industrial crops and products. 2008;27(2):144-9.
CrossRef - Hollman PC. Absorption, bioavailability, and metabolism of flavonoids. Pharmaceutical biology. 2004;42(sup1):74-83.
CrossRef - Domínguez-Avila JA, Wall-Medrano A, Velderrain-Rodríguez GR, et al. Gastrointestinal interactions, absorption, splanchnic metabolism and pharmacokinetics of orally ingested phenolic compounds. Food Funct. 2017;8(1):15-38. doi:10.1039/c6fo01475e
CrossRef - Kawabata K, Yoshioka Y, Terao J. Role of Intestinal Microbiota in the Bioavailability and Physiological Functions of Dietary Polyphenols. Molecules. 2019;24(2):370. Published 2019 Jan 21. doi:10.3390/molecules24020370
CrossRef - Kumar M, Kumar D, Kumar S, et al. A Recent Review on Bio-availability Enhancement of Poorly Water-soluble Drugs by using Bioenhancer and Nanoparticulate Drug Delivery System. Curr Pharm Des. 2022;28(39):3212-3224. doi:10.2174/1381612829666221021152354
CrossRef - Khosravi-Far R, Esposti MD. Death receptor signals to mitochondria. Cancer Biol Ther. 2004;3(11):1051-1057. doi:10.4161/cbt.3.11.1173
CrossRef - Lossi L. The concept of intrinsic versus extrinsic apoptosis. Biochem J. 2022;479(3):357-384. doi:10.1042/BCJ20210854
CrossRef - Ola MS, Nawaz M, Ahsan H. Role of Bcl-2 family proteins and caspases in the regulation of apoptosis. Mol Cell Biochem. 2011;351(1-2):41-58. doi:10.1007/s11010-010-0709-x
CrossRef - Barnum KJ, O’Connell MJ. Cell cycle regulation by checkpoints. Methods Mol Biol. 2014;1170:29-40. doi:10.1007/978-1-4939-0888-2_2
CrossRef - Engeland K. Cell cycle regulation: p53-p21-RB signaling. Cell Death Differ. 2022;29(5):946-960. doi:10.1038/s41418-022-00988-z
CrossRef - Ralić V, Nešić MD, Dučić T, et al. Analysis of Biomolecular Changes in HeLa Cervical Cancer Cell Line Induced by Interaction with [Pd (dach) Cl2]. Inorganics. 2025;13(1):20.
CrossRef - Kodous AS, Fathy RM, Mazhar A, et al. Green synthesis of β-carotene-loaded liposome as antibacterial, antibiofilm and anti-inflammatory modulator: targeting NO/iNOS/NF-κB signaling pathway in MCF-7 cancer cell line. Process Biochemistry. 2025;153:135-53.
CrossRef - Al-Ghanimi B, Abd Nusaif KI, Al-Baiati MN. Studying of Using a Novel Nano Chitosan Loaded with Amoxicillin in the Treatment of Lung Cancer Cell Line (A549). Moroccan Journal of Chemistry. 2025;13(2):480-94.
- Jesenko T, Kranjc Brezar S, Pisljar Z, et al. Effective targeting of E2F1 transcription factor via siRNA gene electrotransfer in HT-29 colorectal carcinoma xenografts. Bioelectrochemistry. 2025;165:108994. doi:10.1016/j.bioelechem.2025.108994
CrossRef - Wadoo R, Ali T, Jan I, et al. In-vitro antioxidant and anti-proliferative activity of aerial parts of Senecio Laetus Edgew on breast cancer (MCF-7) and colon carcinoma (HCT116) cell lines. BMC Complement Med Ther. 2025;25(1):45. Published 2025 Feb 11. doi:10.1186/s12906-025-04789-y
CrossRef - Józefczyk A, Adamczuk G, Humeniuk E, et al. The extracts from centaurea species abolished the cytotoxic effects of doxorubicin on breast cancer cell line – MCF-7 and bortezomib on prostate cancer cell line- PC3. Food Chem Toxicol. 2025;197:115271. doi:10.1016/j.fct.2025.115271
CrossRef - Machado RS, Tristão DC, Araújo NM, et al. Dicentrine and its N-oxide derivatives induces apoptotic and necrotic cell death in prostate cancer cell lines. European Journal of Medicinal Chemistry Reports. 2025;13:100257.
CrossRef - Onal S, Alkaisi MM, Nock V. On-chip non-contact mechanical cell stimulation – quantification of SKOV-3 alignment to suspended microstructures. Heliyon. 2024;11(1):e41433. Published 2024 Dec 30. doi:10.1016/j.heliyon.2024.e41433
CrossRef - Yu SC, Ping YF, Yi L, et al. Isolation and characterization of cancer stem cells from a human glioblastoma cell line U87. Cancer Lett. 2008;265(1):124-134. doi:10.1016/j.canlet.2008.02.010
CrossRef - Dalkılıç S, Kadıoğlu Dalkılıç L, İsbenov E, et al. Investigation of Cytotoxic, Antioxidant, Apoptotic/Necrotic Activity of Aquilaria agallochaRoot Extract and Determination of Gene Expression Levels in HepG2, MCF-7 Cancer Cell Lines. Life (Basel). 2025;15(4):651. Published 2025 Apr 16. doi:10.3390/life15040651
CrossRef - Sanji AS, J M, Kennedy JF, et al. Purification and characterization of lectin from Phyllanthus reticulatus (PRL) plant fruit inducing cytotoxic effect on T47D and SKBR3 breast cancer cell lines. Int J Biol Macromol. 2025;307(Pt 2):142006. doi:10.1016/j.ijbiomac.2025.142006
CrossRef - Mustafa NN, El-Desouky MA, Shawush NA, et al. Apoptosis induction in ascorbic acid treated human colorectal cancer cell lines (Caco-2). Journal of Biologically Active Products from Nature. 2025;15(1):56-71.
CrossRef - Mani S, Swargiary G. In vitro cytotoxicity analysis: MTT/XTT, trypan blue exclusion. InAnimal cell culture: principles and practice. Springer International Publishing .2023; 267-284.
CrossRef - Liu YL, Guo YH, Song XQ, et al. A method for analyzing programmed cell death in xylem development by flow cytometry. Front Plant Sci. 2023;14:1196618. Published 2023 Jun 9. doi:10.3389/fpls.2023.1196618
CrossRef - Tong J, Rufli S, Wong WW. Measuring Caspase Activity Using a Fluorometric Assay or Flow Cytometry. J Vis Exp. 2023;(193):10.3791/64745. Published 2023 Mar 24. doi:10.3791/64745
CrossRef - Lonati L, Barbieri S, Guardamagna I, et al. Radiation-induced cell cycle perturbations: a computational tool validated with flow-cytometry data. Sci Rep. 2021;11(1):925. Published 2021 Jan 13. doi:10.1038/s41598-020-79934-3
CrossRef - Sule R, Rivera G, Gomes AV. Western blotting (immunoblotting): history, theory, uses, protocol and problems. Biotechniques. 2023;75(3):99-114. doi:10.2144/btn-2022-0034
CrossRef - Bong D, Sohn J, Lee SV. Brief guide to RT-qPCR. Mol Cells. 2024;47(12):100141. doi:10.1016/j.mocell.2024.100141
CrossRef - Lim DY, Jeong Y, Tyner AL, et al. Induction of cell cycle arrest and apoptosis in HT-29 human colon cancer cells by the dietary compound luteolin. Am J Physiol Gastrointest Liver Physiol. 2007;292(1):G66-G75. doi:10.1152/ajpgi.00248.2006
CrossRef - Wang W, VanAlstyne PC, Irons KA, et al. Individual and interactive effects of apigenin analogs on G2/M cell-cycle arrest in human colon carcinoma cell lines. Nutr Cancer. 2004;48(1):106-114. doi:10.1207/s15327914nc4801_14
CrossRef - Choi JA, Kim JY, Lee JY, et al. Induction of cell cycle arrest and apoptosis in human breast cancer cells by quercetin. Int J Oncol. 2001;19(4):837-844. doi:10.3892/ijo.19.4.837
CrossRef - Zhu L, Xue L. Kaempferol Suppresses Proliferation and Induces Cell Cycle Arrest, Apoptosis, and DNA Damage in Breast Cancer Cells. Oncol Res. 2019;27(6):629-634. doi:10.3727/096504018X15228018559434
CrossRef - Rusak G, Gutzeit HO, Müller JL. Structurally related flavonoids with antioxidative properties differentially affect cell cycle progression and apoptosis of human acute leukemia cells. Nutrition Research. 2005;25(2):143-55.
CrossRef - Arul D, Subramanian P. Naringenin (citrus flavonone) induces growth inhibition, cell cycle arrest and apoptosis in human hepatocellular carcinoma cells. Pathol Oncol Res. 2013;19(4):763-770. doi:10.1007/s12253-013-9641-1
CrossRef - Ahamad MS, Siddiqui S, Jafri A, et al. Induction of apoptosis and antiproliferative activity of naringenin in human epidermoid carcinoma cell through ROS generation and cell cycle arrest. PLoS One. 2014;9(10):e110003. Published 2014 Oct 16. doi:10.1371/journal.pone.0110003
CrossRef - Stabrauskiene J, Kopustinskiene DM, Lazauskas R, et al. Naringin and Naringenin: Their Mechanisms of Action and the Potential Anticancer Activities. Biomedicines. 2022;10(7):1686. Published 2022 Jul 13. doi:10.3390/biomedicines10071686
CrossRef - Zhang Z, Wang CZ, Du GJ, et al. Genistein induces G2/M cell cycle arrest and apoptosis via ATM/p53-dependent pathway in human colon cancer cells. Int J Oncol. 2013;43(1):289-296. doi:10.3892/ijo.2013.1946
CrossRef - Ouyang G, Yao L, Ruan K, et al. Genistein induces G2/M cell cycle arrest and apoptosis of human ovarian cancer cells via activation of DNA damage checkpoint pathways. Cell Biol Int. 2009;33(12):1237-1244. doi:10.1016/j.cellbi.2009.08.011
CrossRef - Switalska M, Grynkiewicz G, Strzadala L, et al. Novel genistein derivatives induce cell death and cell cycle arrest through different mechanisms. Nutr Cancer. 2013;65(6):874-884. doi:10.1080/01635581.2013.804938
CrossRef - Kawaii S, Tomono Y, Katase E, et al. Antiproliferative activity of flavonoids on several cancer cell lines. Biosci Biotechnol Biochem. 1999;63(5):896-899. doi:10.1271/bbb.63.896
CrossRef - Jodynis-Liebert J, Kujawska M. Biphasic Dose-Response Induced by Phytochemicals: Experimental Evidence. J Clin Med. 2020;9(3):718. Published 2020 Mar 6. doi:10.3390/jcm9030718
CrossRef - Chae HS, Xu R, Won JY, et al. Molecular Targets of Genistein and Its Related Flavonoids to Exert Anticancer Effects. Int J Mol Sci. 2019;20(10):2420. Published 2019 May 16. doi:10.3390/ijms20102420
CrossRef - Benitez DA, Pozo-Guisado E, Alvarez-Barrientos A, Fernandez-Salguero PM, Castellón EA. Mechanisms involved in resveratrol-induced apoptosis and cell cycle arrest in prostate cancer-derived cell lines. J Androl. 2007;28(2):282-293. doi:10.2164/jandrol.106.000968
CrossRef - Liu B, Zhou Z, Zhou W, et al. Resveratrol inhibits proliferation in human colorectal carcinoma cells by inducing G1/S‑phase cell cycle arrest and apoptosis through caspase/cyclin‑CDK pathways. Mol Med Rep. 2014;10(4):1697-1702. doi:10.3892/mmr.2014.2406
CrossRef - Chen Y, Tseng SH, Lai HS, et al. Resveratrol-induced cellular apoptosis and cell cycle arrest in neuroblastoma cells and antitumor effects on neuroblastoma in mice. Surgery. 2004;136(1):57-66. doi:10.1016/j.surg.2004.01.017
CrossRef - Wu H, Chen L, Zhu F, et al. The Cytotoxicity Effect of Resveratrol: Cell Cycle Arrest and Induced Apoptosis of Breast Cancer 4T1 Cells. Toxins (Basel). 2019;11(12):731. Published 2019 Dec 13. doi:10.3390/toxins11120731
CrossRef - Bai Y, Mao QQ, Qin J, et al. Resveratrol induces apoptosis and cell cycle arrest of human T24 bladder cancer cells in vitro and inhibits tumor growth in vivo. Cancer Sci. 2010;101(2):488-493. doi:10.1111/j.1349-7006.2009.01415.x
CrossRef - Sahu RP, Batra S, Srivastava SK. Activation of ATM/Chk1 by curcumin causes cell cycle arrest and apoptosis in human pancreatic cancer cells. Br J Cancer. 2009;100(9):1425-1433. doi:10.1038/sj.bjc.6605039
CrossRef - Zhu Y, Bu S. Curcumin Induces Autophagy, Apoptosis, and Cell Cycle Arrest in Human Pancreatic Cancer Cells. Evid Based Complement Alternat Med. 2017;2017:5787218. doi:10.1155/ 2017/5787218
CrossRef - Lee DS, Lee MK, Kim JH. Curcumin induces cell cycle arrest and apoptosis in human osteosarcoma (HOS) cells [published correction appears in Anticancer Res. 2010;30(10):4389]. Anticancer Res. 2009;29(12):5039-5044.
- Chen HS, Bai MH, Zhang T, Li GD, et al. Ellagic acid induces cell cycle arrest and apoptosis through TGF-β/Smad3 signaling pathway in human breast cancer MCF-7 cells. Int J Oncol. 2015;46(4):1730-1738. doi:10.3892/ijo.2015.2870
CrossRef - Mertens-Talcott SU, Percival SS. Ellagic acid and quercetin interact synergistically with resveratrol in the induction of apoptosis and cause transient cell cycle arrest in human leukemia cells. Cancer Lett. 2005;218(2):141-151. doi:10.1016/j.canlet.2004.06.007
CrossRef - Agarwal C, Tyagi A, Agarwal R. Gallic acid causes inactivating phosphorylation of cdc25A/cdc25C-cdc2 via ATM-Chk2 activation, leading to cell cycle arrest, and induces apoptosis in human prostate carcinoma DU145 cells. Mol Cancer Ther. 2006;5(12):3294-3302. doi:10.1158/1535-7163.MCT-06-0483
CrossRef - Yeh RD, Chen JC, Lai TY, et al. Gallic acid induces G₀/G₁ phase arrest and apoptosis in human leukemia HL-60 cells through inhibiting cyclin D and E, and activating mitochondria-dependent pathway. Anticancer Res. 2011;31(9):2821-2832.
- Hefer M, Huskic IM, Petrovic A, et al. A mechanistic insight into Beneficial effects of polyphenols in the Prevention and Treatment of Nephrolithiasis: evidence from recent in Vitro studies. Crystals. 2023;13(7):1070.
CrossRef - Lewandowska U, Gorlach S, Owczarek K, et al. Synergistic interactions between anticancer chemotherapeutics and phenolic compounds and anticancer synergy between polyphenols. Postepy Hig Med Dosw (Online). 2014;68:528-540. Published 2014 Jan 2. doi:10.5604/17322693.1102278
CrossRef - Kampa M, Nifli AP, Notas G, et al. Polyphenols and cancer cell growth. Rev Physiol Biochem Pharmacol. 2007;159:79-113. doi:10.1007/112_2006_0702
CrossRef - Montané X, Kowalczyk O, Reig-Vano B, et al. Current Perspectives of the Applications of Polyphenols and Flavonoids in Cancer Therapy. Molecules. 2020;25(15):3342. Published 2020 Jul 23. doi:10.3390/molecules25153342
CrossRef - Arzuman L, Beale P, Chan C, et al. Synergism from combinations of tris(benzimidazole) monochloroplatinum(II) chloride with capsaicin, quercetin, curcumin and cisplatin in human ovarian cancer cell lines. Anticancer Res. 2014;34(10):5453-5464.
- Al-Abd AM, Mahmoud AM, El-Sherbiny GA, et al. Resveratrol enhances the cytotoxic profile of docetaxel and doxorubicin in solid tumour cell lines in vitro. Cell Prolif. 2011;44(6):591-601. doi:10.1111/j.1365-2184.2011.00783.x
CrossRef - Tiwari P, Mishra KP. Flavonoids sensitize tumor cells to radiation: molecular mechanisms and relevance to cancer radiotherapy. Int J Radiat Biol. 2020;96(3):360-369. doi:10.1080/09553002.2020.1694193
CrossRef - Moutabian H, Majdaeen M, Ghahramani-Asl R, et al. A systematic review of the therapeutic effects of resveratrol in combination with 5-fluorouracil during colorectal cancer treatment: with a special focus on the oxidant, apoptotic, and anti-inflammatory activities. Cancer Cell Int. 2022;22(1):142. Published 2022 Apr 2. doi:10.1186/s12935-022-02561-7
CrossRef
Abbreviations List
ATM – Ataxia-Telangiectasia mutated
ATR – stands for Ataxia Telangiectasia and Rad3-related.
CDKs – Cyclin-Dependent Kinases.
DISC – Death-Inducing Signaling Complex
DPPH – 2,2-Diphenyl-1-picrylhydrazyl antioxidant activity
HL – Hodgkin Lymphoma
HT – Hormone Therapy
LMIC – Low- and Middle-Income Countries
LNCaP – stands for Lymph Node Carcinoma of the Prostate.
MAPKS – mitogen-activated protein kinases
MCF – Michigan Cancer Foundation
MDA-MB cells – MD Anderson – Metastatic Breast cancer cell lines
MOMP – mitochondrial outer membrane permeabilization
MTI – Microtubule Inhibition Assay
NF-κB – Nuclear Factor kappa-light-chain-enhancer of activated B cells.
PARP – Poly (ADP-ribose) Polymerase
PCR/qPCR– Quantitative Polymerase Chain Reaction
ROS – Reactive Oxygen Species response
SAC – Spindle Assembly Checkpoint
siRNA – Small interfering RNA
SPGG – Sulfated Pentagalloyl Glucoside
STAT3 – Signal Transducer and Activator of Transcription 3
TGF-β – Transforming Growth Factor-beta
TNBC – Triple-Negative Breast Cancer
TNF-alpha and TRAIL – Tumor Necrosis Factor-alpha /-Related Apoptosis-Inducing Ligand
XTI – Possibly Xenograft Tumor Inhibition assay







