Manuscript accepted on :23-10-2025
Published online on: 20-11-2025
Plagiarism Check: Yes
Reviewed by: Dr. Noora Thamer Abdulaziz
Second Review by: Dr. Huzefa A. Ujjainwala
Final Approval by: Dr. Prabhishek Singh
Renukadevi Jeyavelkumaran , Jayasri Bantaram
, Jayasri Bantaram , Karthikha Vijayakumar Sumathi
, Karthikha Vijayakumar Sumathi , Sam Helinto Jeya
, Sam Helinto Jeya , Sri Vaishnavi Velegatla
, Sri Vaishnavi Velegatla and Akshay Babu Sharadha
and Akshay Babu Sharadha
Department of Pharmaceutics, Saveetha College of Pharmacy, Saveetha Institute of Medical and Technical Sciences (SIMATS), Chennai, India
Corresponding Author E-mail:renukadevij.scop@saveetha.com
DOI : https://dx.doi.org/10.13005/bpj/3308
Abstract
Cancer development is inextricably associated with oxidative stress and inflammation, and tumor cells respond with increased glutathione S-transferase (GST) activity and cellular glutathione (GSH) concentrations, leading to therapy resistance. Hemidesmus indicus is a commonly employed medicinal plant in Ayurveda, with bioactive compounds that may have anticancer and anti-inflammatory activities. The aim of this investigation was to assess the therapeutic value of bioactive fractions of H. indicus by targeting the GST and GSH pathways in tumor inflammation. Bioactive fractions were isolated using bioassay-guided fractionation and identified by gas chromatography-mass spectrometry (GC-MS) as principal compounds Phthalic acid, Oleanen-3yl acetate, and Eicosane. Cytotoxicity against HeLa cells showed pronounced antiproliferative activity with an IC50 value of 37.44 µg/mL. The bioactive fractions inhibited GST activity and consumed GSH content in HeLa cells, inducing oxidative stress-mediated apoptosis. Moreover, anti-inflammatory assays such as protein denaturation and red blood cell membrane stabilization validated its anti-inflammatory potential. These data indicate that H. indicus bioactive fractions act as potent modulators of oxidative stress and inflammation, and thus potential candidates for anticancer and anti-inflammatory drug development. More “in vivo” research and clinical trials are needed to establish their therapeutic applications.
Keywords
Bioactive Fractions; HeLa Cells; Hemidesmus indicus; Phytochemical Profiling; Redox Modulation; Tumor Inflammation
Download this article as:| Copy the following to cite this article: Jeyavelkumaran R, Bantaram J, Sumathi K. V, Jeya S. H, Velegatla S. V, Sharadha A. B. Therapeutic Potential of Hemidesmus indicus Bioactive Fractions: Inhibiting GST and GSH in Tumor Inflammation. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Jeyavelkumaran R, Bantaram J, Sumathi K. V, Jeya S. H, Velegatla S. V, Sharadha A. B. Therapeutic Potential of Hemidesmus indicus Bioactive Fractions: Inhibiting GST and GSH in Tumor Inflammation. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4pkWWO2 |
Introduction
Hemidesmus indicus is an important ancient Ayurvedic medicinal plant with reported anti-inflammatory, antioxidant, and anticancer activities. These activities are attributed due to the presence of different bioactive compounds with therapeutic potential in the lead optimization process of drug discovery.1 H. indicus is one of the promising sources for the identification of lead compounds, effective against oncology- and inflammation-related diseases.2 Activity-guided fractionation approaches are usually adopted to isolate bioactive compounds and process them through drug discovery pipelines for clinical applications.3 This work followed an integrated drug discovery approach, wherein the roots of Hemidesmus indicus were extracted and fractionated using a bioassay-guided fractionation method to afford compounds with potential biological activities. Phytochemical profiling and bioassay-guided fractionation were performed to identify the most active fractions, while further analyses were conducted by means of gas chromatography-mass spectrometry with a view to pinpointing the exact bioactive compounds present.4 The fractions exhibiting some biological activity, including anticancer and anti-inflammatory potentials, are advanced for further analysis to streamline such a process of identifying promising drug leads.
The modulation of pathways of oxidative stress in cancerous cells is one of the prime study areas that create initial point of drug discovery targeted at tumor progression.5 Cancerous cells such as HeLa cells possess higher levels of antioxidant defense, such as glutathione S-transferase and reduced glutathione, which render them resistant to many conventional therapies.6The activity-guided assays screened the Hemidesmus indicus fractions so as to reduce GSH levels and inhibit GST activity, which would selectively assess the state of oxidative stress in cancer cells. A mechanism involving disruption of normal redox balance in cancer cells initiates apoptosis without harming the normal cells.7 Downregulation of GST and depletion of GSH in HeLa cells treated with bioactive fractions underline their lead compound potentiality for further drug development.
Apart from its pro-oxidative effects, H, indicus exerts potent anti-inflammatory action, an important modality in drug discovery against cancer and chronic inflammatory disorders. Inflammation is identified as one of the driving forces in cancer proliferation and metastasis; thus targeting inflammatory pathways offers twin benefits in cancer therapy.8 Activity-guided fractionation assays such as protein denaturation inhibition and HRBC membrane stabilization tests have shown significant and potent suppression of inflammation by bioactive fractions through inhibition of key pathways involved in inflammation. Thus, these findings have established H. indicus as strong lead with multi-faceted properties influencing at both the oxidative stress and inflammation pathways in cancer biology. The present study was aimed to assess the therapeutic potential of bioactive fractions of Hemidesmus indicus by targeting GST and GSH in tumor inflammation and lays a solid base for developing new anticancer and anti-inflammatory agents from H, indicus.
Materials and Methods
Materials
Hemidesmus indicus plant material utilized in the present work was obtained from a local shop and authenticated by Mrs. Vandhana, Lecturer, Department of Pharmacognosy, Saveetha College of Pharmacy. All chemicals and equipment necessary for the work were supplied by the lab facilities at Saveetha Institute of Medical and Technical Sciences (SIMATS), Chennai, India.
Methods
Extraction, fractionation, and QSAR analysis of bioactive compounds from H. indicus
The current study is a follow-up of our previously published work by (Devi JR)9
, in which the same bioactive fraction of Hemidesmus indicus has been used for the work. In the previous study, bioactive molecules were extracted from H. indicus under reduced pressure, followed by solvent fractionation at different polarity levels through n-hexane, ethyl acetate, chloroform, methanol, and water. Phytochemical analysis and antimicrobial assays of the resultant fractions had been carried out through agar diffusion methods. The bioactive fraction was also analyzed by gas chromatography–mass spectrometry (GC-MS) for the identification of their constituents, and a QSAR model was established through the Comparative Molecular Field Analysis (CoMFA) technique to relate the biological activity of the compounds to their 3D molecular features. In the current research, the same bioactive fraction and previously characterized ligands were used to carry out their therapeutic potential.9 The individual phytoconstituents were docked followed by simulation to assess their tumor inflammation properties.10
Determination of total antioxidant activity by DPPH (diphenyl picryl hydrazyl radical) method
The radical scavenging activity of the extracts was determined according to the method.11 12Vero and HeLa cells were seeded and exposed to both crude and bioactive fractions of H. indicus for 24hr.Viable cells were obtained from the culture flask by means of a cell scraper. The cells were subjected to centrifugation and homogenized in a cold tris HCl solution using an ultrasonicator. The sonicated cell suspension was centrifuged at 300 rpm for 10 min at 4˚C and the supernatant was collected and prepared for the quantification of DPPH (2, 2’ -diphenyl-β-picrylhydrazyl radical (DPPH) scavenging activity. The absorbance was measured at 517 nm using Spectrophotometer (Shimadzu UV-1900i).
Determination of Glutathione-S-transferase (GST) activity
Vero and HeLa cells were plated and treated with crude and bioactive fractions of H. indicus for 24hr. The cells were centrifuged and then sonicated at 10,000 rpm for 15 minutes at 4˚C with glutathione and CDNB (1-chloro-2, 4-dinitrobenzene). The absorbance was measured at 340 nm using Spectrophotometer (Shimadzu UV-1900i). The GST was expressed as (nmol mg-1 min-1).13
Determination of Reduced glutathione (GSH) activity
Vero and HeLa cells were seeded and exposed to both crude and bioactive fractions of H. indicus for 24 h. Cells were obtained from the culture flasks using an automated cell scraper Cells were first sonicated for 3000 rpm and then centrifuged at 4OC. The supernatant was used to quantify glutathione concentration. DTNB reagent ((5,5-dithio-bis-(2-nitrobenzoic acid)) was added.14 The absorbance was measured at a 412 nm using Spectrophotometer (Shimadzu UV-1900i).. The GSH was expressed as µg dL-1.
Determination of cytotoxicity assay of bioactive fraction of Hemidesmus indicus
The cytotoxicity of bioactive fraction of H. indicus on HeLa cells were determined by MTT assay. Cells were plated in 96-well microtiter plates (and incubated at 5 % CO2 incubator ( Thermoscientific, 2021). Then, various concentrations of bioactive fraction were added and incubated. The images were viewed in inverted microscope (Labtech, 2015) to assess the percentage viability. Viable cells were determined by measuring the absorbance at 540 nm.15
Percentage cell viability of cells was expressed using the following formula:
% cell viability = A540 of treated cells / A540 of untreated cells × 100
Inhibition of protein denaturation using bovine serum albumin
Different concentrations consisting 5% aqueous solution of BSA were treated with bioactive fraction of H. indicus. Aspirin treated solution were used as control the mixtures were incubated at 37°C for 20 min and subsequently heated. Then phosphate buffer saline was added to each test tube and absorbance measured at 660 nm. The percentage inhibition of protein denaturation.16,17 was calculated as follows

Inhibition of protein denaturation using Fresh hen’s egg albumin:
Solutions with fresh egg albumin, phosphate buffered saline (pH 6.4) and crude extract/fractions solutions of different concentrations were prepared. Doubled distilled water was taken as control. Reaction mixture was placed in an incubator followed by heating.18,19 Percentage inhibition of protein denaturation was calculated by using the following formula:
% of inhibition = 100 X [Vt /Vc-1]
Where, Vt = absorbance of test sample, Vc = absorbance of control
Inhibiton of hemolysis assay by human red blood cell (HRBC) membrane stabilization
Different concentrations HRBC stock mixed with hypotonic solution (0.9% NaCl) was added to varying concentrations of bioactive fraction of H. indicus The positive control consisted of HRBC and hypotonic solution in varying concentration of diclofenac sodium. The mixture was incubated for 10 min at room temperature and then centrifuged. The haemoglobin content of the supernatant was collected and analysed at 540 nm.20The percentage inhibition of hemolysis was calculated using the formula.
% inhibition = [Absorbance (control) –Absorbance (test) / Absorbance (control)] x 100
Values were reported as mean ± SEM and analyzed statistically using Microsoft Excel. Values of p<0.05 are regarded as significant.
Results
Determination of total antioxidant activity by DPPH (diphenyl picryl hydrazyl radical) method
The radical scavenging activity of the crude and bioactive fraction of Hemidesmus indicus was determined using the DPPH method. The antioxidant activity of crude and bioactive fractions of H. indicus was assessed on Vero and HeLa cells using DPPH method. The crude extract exhibited high quenching activity 79.33±2.88 %in HeLa cells in comparison to 55.33±0.57 % in Vero cells (Table 1)
Determination of Glutathione-S-transferase (GST) activity
GST activity of crude extract and bioactive fractions, studied on Vero and HeLa cell lines, showed that crude extract exerted a significant reduction in GST activity in HeLa cells whereas there was no marked reduction in enzyme activity in Vero cells (Table 1.Fig 1).
Table 1: Effect of crude extract, bioactive fraction of Hemidesmus indicus on glutathione S transferase (GST) activity, reduced glutathione (GSH) concentration, DPPH scavenging activity on HeLa and Vero cells.
| Fraction | GST activity(nmol mg-1 min-1) | GSHµg dL-1 | DPPH scavenging activity (%) | |||
| HeLa | Vero | HeLa | Vero | HeLa | Vero | |
| Extract | 4.5±0.1 | 3.6±0.15 | 21.6±0.57 | 64.33±2.51 | 79.33±2.88 | 55.33±0..57 |
| Bioactive fraction | 8.13±0.81 | 1.76±0.15 | 34.3±1.15 | 39.33±1.52 | 55.6±1.15 | 34.3±1.52 |
| Control | 12.9±1.0 | 4.4±1.0 | 38±0.15 | 32.0±0.12 | – | – |
The data represents average ± SE of three independent experiments
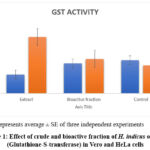 |
Figure 1: Effect of crude and bioactive fraction of H. indicus on GST (Glutathione-S-transferase) in Vero and HeLa cells |
Determination of GSH (reduced glutathione)
The elevated GSH as seen in untreated HeLa cells were decreased after treatment with crude extract of H. indicus. There was no marked decrease in GSH content in bioactive fraction treated cells (Table 1, Fig 2). In Vero cells bioactive fraction did not bring any significant change in GSH whereas extract increased the GSH when compared to control cells.
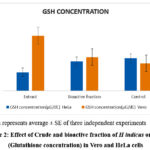 |
Figure 2: Effect of Crude and bioactive fraction of H indicus on GSH (Glutathione concentration) in Vero and HeLa cells |
Hemidesmus indicus exhibits high phenolic and flavonoid content, making it effective antioxidants to scavenge free radicals and prevent oxidative stress in cells. The plant also had an impact on the antioxidant activity to downregulate the effect of oxidative stress on cells which is one of the factors that support the growth of cancer cells.21 It is also evident that this plant might improve cellular antioxidant protection through a number of antioxidant enzymes that point to those higher intracellular concentrations of ROS in cancer cells. Similarly, it is also helpful in protection of oxidative impairment in normal cells and promotion of oxidative stress in cancer cells, which strengthen the two faceted aspect of cancer treatment.22Finally, the antioxidants capacity of Hemidesmus indicus influences the balance in oxidative stress is responsible for the revealed cancer and inflammation suppressing potentials. The results on modulation of Glutathione S-Transferase (GST) activity and Glutathione (GSH) concentration by Hemidesmus indicus are discussed in the light of recent literature demonstrated that the administration of the extracts of Hemidesmus indicus overexpressed the activity of GST and other cancer cells thereby resulting in the suppression of the detoxification process and consequently increasing susceptibility to oxidative stress. The extracts from the plant could reduce GSH levels in cancer cells; which if reduced will lead to increased oxidative stress and apoptosis. Hemidesmus indicus down regulates the GST and GSH in cancer cells but not in normal cells evidencing the selectivity of the compound for cancer cells. The bioactive compounds present in the plant were capable to inducing oxidative stress in cancer cells by reducing GSH and therefore substantiated the potentials of the plant for cancer treatment. The enzymatic activity of Hemidesmus indicus may serve as a redox focusing on cancer cells subjecting to the current investigation.
Determination of cytotoxicity assay of bioactive fraction of Hemidesmus indicus
The percentage viability of HeLa cells decreases with the decrease in the concentration of bioactive fraction extract of H. indicus. The extract from bioactive fraction showed IC50 value of 37.44 µg mL-1 on HeLa cell line. The cell viability of HeLa cell lines decreased with change in concentration of H indicus extract (Fig 3). The fraction was reported to contain phthalic acid, Oleanen 3yl acetate and Eicosane. Among these compounds Oleanane triterpenoids, including oleanolic acid and synthetic derivatives, have shown promising anticancer and anti-inflammatory properties through modulation of multiple signaling pathways.23 Oleanolic acid affects various molecular targets involved in cancer progression, including caspases, AMPK, ERK1/2, and PI3K/Akt/mTOR pathways.24 Synthetic oleanane triterpenoids demonstrate cytoprotective effects at low doses and induce apoptosis at higher concentrations, primarily through the Keap1/Nrf2/ARE pathways.25Similar research supports that the Hemidesmus indicus bioactive compounds significantly exerted cytotoxic effects against breast and cervical cancer cells and through the induction of cell-cycle arrest and apoptosis. These compounds were reported to inhibit NF-κB and STAT3, key regulators of inflammation and tumorigenesis26. Further elucidation of exact mechanism of action is done through insilico studies by molecular docking and QSAR studies, which indicated good interaction of bioactive compounds with molecular targets related to cancer. Current research work substantiates the role of xenobiotics in this ancient herbal plant which explore the novel leads for the improving metabolism and health, inhibiting the development of cancer and other diseases.
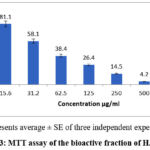 |
Figure 3: MTT assay of the bioactive fraction of H.indicus |
Inhibition of protein denaturation (bovine serum albumin assay)
The results obtained further substantiated that the effect of bioactive fraction on protein denaturation were correlated with the degree of concentration. The maximum percentage inhibition observed was 80% the highest concentration tested, indicating the potential anti-inflammatory properties of the extracts (Fig 4). The control sample, which did not contain the extract, exhibited no significant inhibition, thus confirming the activity of the H. indicus components.
Inhibition of protein denaturation (Hen’s egg albumin assay)
The results revealed a significant reduction in protein denaturation, with percentage inhibition increasing with the increase in concentration of the extract (Fig 5). The highest percentage inhibition was recorded at 78%, indicating strong anti-inflammatory activity. Diclofenac sodium, used as a standard control, exhibited comparable inhibition, thereby supporting the efficacy of the extracts. The control without the extract showed negligible inhibition.
Human red blood cell (HRBC) membrane stabilization assay
The findings showed that the bioactive fraction possessed a depolymerising ability of the HRBC membrane, by lowering haemolysis in a concentration dependent manner. The maximum inhibition was achieved for the highest extract concentration and reached 84% inhibiting hemolysis, which is not statistically different from inhibiting effect of diclofenac sodium, the reference drug. Based on these findings, it can be concluded that there are anti-inflammatory potentials of H. indicus which could have its way through the membrane stabilization.These results thus suggest the anti-inflammatory property of Hemidesmus indicus as supported by in vitro studies Hemidesmus indicus contains anti-inflammatory compound in its extract in terms of inhibition of pro-inflammatory biomarkers and cyclooxygenase enzymes and stabilizing lysosomal membrane.The extracts from the plant were capable of inhibiting the ‘key enzyme’ involved in the inflammatory process such as COX-2 and 5-LOX resulting to increased inflammation It was discovered that the extract from Hemidismus Indicus could modulate the NF-κB signaling pathway, which plays a key role in regulating inflammation. This modulation was credited with the plant’s anti-inflammatory effects, as demonstrated in both cell and animal studies.
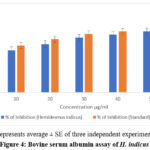 |
Figure 4: Bovine serum albumin assay of H. indicus |
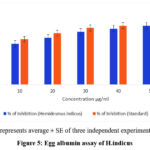 |
Figure 5: Egg albumin assay of H.indicus |
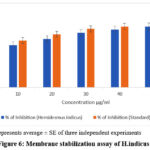 |
Figure 6: Membrane stabilization assay of H.indicus |
Discussion
Hemidesmus indicus, a commonly employed traditional Indian medicine, is found to possess anti-inflammatory, antioxidant, and anticancer activities, possibly mediated by different signalling mechanisms. The research here underscores its therapeutic value, especially in HeLa cells, wherein its bioactive fractions regulate GST and GSH levels and enhance oxidative stress-induced apoptosis. GC-MS profiling revealed some major compounds such as phthalic acid, oleanen-3yl acetate, and eicosane, which are associated with antioxidant activity and xenobiotic metabolism. Interestingly, 2-Hydroxy-4-methoxybenzoic acid (HMBA) has the ability to inhibit the biosynthesis of prostaglandins, which inhibit tumor growth. Phthalic acid derivatives inhibit GST, leading to cancer cell detoxification deficiency and an increase in ROS accumulation, and GSH depletion compromises cellular defense and increases Bax-induced apoptosis. The dual mechanism highlights Hemidesmus indicus as a potential anticancer drug, necessitating future molecular investigations and clinical establishment. Though the results are promising, it is of paramount importance to note that further “in vivo” and clinical studies are required to establish the efficacy and safety for such bioactive compounds. Formulating these phytoconstituents is essential in near time to establish them as a potent drug leads with improved biopharmaceutics and pharmacokinetics properties.
Conclusion
The bioactive fractions of Hemidesmus indicus was found to exhibit anti-inflammatory, antioxidant, and anticancer activities. Fraction was able to modulate key biochemical processes and act as inducer for phase II xenobiotic metabolism such as GST activity and levels of GSH regulators of oxidative stress in cancer cells. As a cytotoxic agent against HeLa cells, can be optimised a drug lead in cancer with proper structural optimisation. Further, Hemidesmus indicus fraction has anti-inflammatory action through cell membrane stabilization and prevention of protein denaturation, to treat inflammation. Further “in vivo” studies are essential along with clinical trials for drug lead optimisation.
Acknowledgment
The authors want to thank the management of Saveetha Institute of Medical and Technical Sciences(SIMATS), Chennai, India for providing the all necessary facilities for this study.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author contributions
Renukadevi Jeyavelkumaran contributed in drafting the manuscript’s content, articulating the research findings and discussions contributing to technical coherence and quality.
Jayasri Bantaram, Karthikha Vijayakumar Sumathi, Sam Helinto Jeya, Sri Vaishnavi Velegatla, Akshay Babu Sharadha contributed equally in formatting and editing the manuscript.
References
- Das S, Singh Bisht S. The bioactive and therapeutic potential of Hemidesmus indicus R. br. (Indian Sarsaparilla) root. Phytother Res. 2013;27(6):791-801. doi:10.1002/ptr.4788
CrossRef - Sena S, Van Staden J, Kumar V, Husen A. Hemidesmus indicus (L.) R. Br. ex Schult as natural bioactive products: An evidence-based review focused on inflammation related cancer prevention potential. Curr Res Biotechnol. 2023;6(100165):100165. doi:10.1016/j.crbiot.2023.100165
CrossRef - Ory L, Nazih EH, Daoud S, et al. Targeting bioactive compounds in natural extracts – Development of a comprehensive workflow combining chemical and biological data. Anal Chim Acta. 2019;1070:29-42. doi:10.1016/j.aca.2019.04.038
CrossRef - Adarshan S, Sree VSS, Muthuramalingam P, et al. Understanding macroalgae: A comprehensive exploration of nutraceutical, pharmaceutical, and omics dimensions. Plants. 2023;13(1):113. doi:10.3390/plants13010113
CrossRef - Das K, Bhatt N, Parambil AM, et al. Divergent Responses of Hydrophilic CdSe and CdSe@CdS Core–Shell Nanocrystals in Apoptosis and In Vitro Cancer Cell Imaging: A Comparative Analysis. JFB. 2023;14(9):448. doi:10.3390/jfb14090448
CrossRef - Prasher P, Sharma M, Singh SK, et al. Luteolin: a flavonoid with a multifaceted anticancer potential. Cancer Cell Int. 2022;22(1):386. doi:10.1186/s12935-022-02808-3
CrossRef - Godwin I, Anto NP, Bava SV, Babu MS, Jinesh GG. Targeting K-Ras and apoptosis-driven cellular transformation in cancer. Cell Death Discov. 2021;7(1):80. doi:10.1038/s41420-021-00457-5
CrossRef - Hibino S, Kawazoe T, Kasahara H, et al. Inflammation-induced tumorigenesis and metastasis. Int J Mol Sci. 2021;22(11):5421. doi:10.3390/ijms22115421
CrossRef - Devi JR, Devi GN, Bavanilatha M, et al. Pharmacophore based QSAR modelling of natural leads in antimicrobial drug design. Current Chinese Chemistry. 2021;1(1):80-84. doi:10.2174/2666001601666200206102612
CrossRef - J Renukadevi, S Karthikha V, J Sam Helinto, D. Prena, Arockiya Rabin A. Molecular dynamic simulation studies of hemidesmus indicus-derived oleanen-3-yl acetate in stat3 based tumor signaling. J Appl Pharm Res. 2024;12(5):124-132. doi:10.69857/joapr.v12i5.670
CrossRef - Laohavechvanich P, Muangnoi C, Butryee C, Kriengsinyos W. Protective effect of makrut lime leaf (Citrus hystrix) in HepG2 cells, Implications for oxidative stress”. Science Asia. 2010;36:112-117.
CrossRef - Praveen G, Krishnamoorthy K, Veeraraghavan VP, Jayaraman S. Antioxidant and anti-inflammatory activity of the ethanolic extract of Euphorbia hirta leaf extract: An in vitro and in silico study. J Pharm Bioallied Sci. 2024;16(Suppl 2):S1304-S1307. doi:10.4103/jpbs.jpbs_591_23
CrossRef - Sandbacka M, Isomaa B. Glutathione S-transferase and UDP-glucuronyltransferase activity in primary cultures of rainbow trout gill epithelial cells. Comp Biochem Physiol C Toxicol Pharmacol. 2000;127(3):307-315. doi:10.1016/s0742-8413(00)00156-0
CrossRef - Rahman I, Kode A, Biswas SK. Assay for quantitative determination of glutathione and glutathione disulfide levels using enzymatic recycling method. Nat Protoc. 2006;1(6):3159-3165. doi:10.1038/nprot.2006.378
CrossRef - Subramanian AK, Setvaji NR. Evaluation of cytotoxicity of green synthesized strontium fluorapatite nanoparticles on human gingival fibroblasts: An in vitro study. World J Dent. 2024;15(1):25-29. doi:10.5005/jp-journals-10015-2357
CrossRef - Sakat S, Juvekar AR, Gambhire MN. In vitro antioxidant and anti-inflammatory activity of methanolic extract of Oxalis corniculata Linn. International Journal of Pharmacy and Pharmaceutical Sciences. 2010;2(1):146-155.
- Varshini A, Cecil A, Kumar SR. Preparation of Camellia sinensis (Green Tea) and Acacia nilotica (Babul) Herbal Formulation and its Anti-Inflammatory Activity. Pharmacognosy Res. 2023;15(2):255-257. doi:10.5530/pres.15.2.027
CrossRef - Hasan AUH. Evaluation of in vitro and in vivo therapeutic efficacy of Ribes alpestre Decne in Rheumatoid arthritis. Braz J Pharm Sci. 2019;55. doi:10.1590/s2175-97902019000217832
CrossRef - Balasubramaniyam S, Rathinam T, Srinivasan M, Arulselvan P, Mickymaray S, Alfaiz FA. Green-engineered synthesis of zinc oxide (ZnO) nanoparticles using Musa paradisiaca: evaluation of antioxidant, antimicrobial, anti-inflammatory, and antihyperglycemic activities. 3 Biotech. 2025;15(10):358. doi:10.1007/s13205-025-04526-9
CrossRef - Rajapandi S, Nangan S, Thirumalaivasan N, et al. Synthesis and characterization of nitrogen-doped carbon dots from Carissa carandoss for environmental and in-vitro biomedical applications. Waste Biomass Valorization. Published online December 27, 2024. doi:10.1007/s12649-024-02808-6
CrossRef - Som S, Dhanabal AJ, Ponnusankar SP. Phytochemical Profiling of Hemidesmus indicus (L.) R. Br. ex Schult and its Antioxidant, Anti-Inflammatory and Phcogj.com Neuroprotection Linked Enzyme Inhibitory Properties. Pharmacog J. 2021;13(1):196-205.
CrossRef - Kurutas EB. The importance of antioxidants which play the role in cellular response against oxidative/nitrosative stress: current state. Nutr J. 2015;15(1). doi:10.1186/s12937-016-0186-5
CrossRef - Baer-Dubowska W, Narożna M, Krajka-Kuźniak V. Anti-cancer potential of synthetic oleanolic acid derivatives and their conjugates with NSAIDs. Molecules. 2021;26(16):4957. doi:10.3390/molecules26164957
CrossRef - Žiberna L, Šamec D, Mocan A, et al. Oleanolic acid alters multiple cell signaling pathways: Implication in cancer prevention and therapy. Int J Mol Sci. 2017;18(3):643. doi:10.3390/ijms18030643
CrossRef - Liby KT, Sporn MB. Synthetic oleanane triterpenoids: Multifunctional drugs with a broad range of applications for prevention and treatment of chronic disease. Pharmacol Rev. 2012;64(4):972-1003. doi:10.1124/pr.111.004846
CrossRef - Turrini E, Calcabrini C, Tacchini M, et al. In vitro study of the cytotoxic, cytostatic, and antigenotoxic profile of Hemidesmus indicus (L.) R.br. (Apocynaceae) crude drug extract on T lymphoblastic cells. Toxins (Basel). 2018;10(2):70. doi:10.3390/toxins10020070







