Manuscript accepted on :23-10-2025
Published online on: 07-11-2025
Plagiarism Check: Yes
Reviewed by: Dr. Ramdas Bhat
Second Review by: Dr. Shabana Khatoon
Final Approval by: Dr. Patorn Piromchai
Pydikondala Nehitha Purna1 , Pydiraju Kondrapu2
, Pydiraju Kondrapu2 , Alekhya Ketha3
, Alekhya Ketha3 , Shashikala Metri4
, Shashikala Metri4 , and Nagaraju Bandaru5*
, and Nagaraju Bandaru5*
1Department of Pharmaceutical Sciences, Koringa College of Pharmacy, Korangi, India
2Department of Pharmacology, Adarsa College of Pharmacy, G. Kothapalli, Andhra Pradesh, india
3Department of Pharmaceutical Chemistry, Sree Dattha Institute of Pharmacy, Sheriguda, Ibrahimpatnam, Hyderabad, India.
4Department of Pharmacognosy, Gokaraju Rangaraju College of Pharmacy, Hyderabad, Telangana, India.
5Department of Pharmacology, School of Pharmaceutical Sciences, Sandip University, Nashik, Maharashtra, India.
Corresponding Author E-mail: bnrajupharma@gmail.com.
DOI : https://dx.doi.org/10.13005/bpj/3281
Abstract
Alzheimer’s disease (AD) is the most prevalent neurodegenerative disorder characterized by progressive cognitive decline and memory impairment. Early and accurate diagnosis remains a pivotal challenge to improve patient outcomes and guide therapeutic interventions. In recent years, cerebrospinal fluid (CSF) and blood biomarkers have emerged as critical tools for the detection and monitoring of AD pathology. CSF biomarkers such as amyloid-beta 42 (Aβ42), total tau (t-tau), and phosphorylated tau (p-tau) directly reflect hallmark pathological changes, including amyloid plaque accumulation and neurofibrillary tangles, offering high diagnostic specificity. However, lumbar puncture’s invasiveness limits routine use, prompting the development of blood-based biomarkers. Advances in ultrasensitive assay technologies have enabled reliable quantification of plasma Aβ42/40 ratios, phosphorylated tau isoforms (p-tau181, p-tau217), and neurofilament light chain (NfL), correlating strongly with AD pathology and clinical progression. Blood biomarkers provide a minimally invasive, accessible approach suitable for large-scale screening, early detection, and longitudinal monitoring. Integrating CSF and blood biomarkers with neuroimaging and cognitive assessments enhances diagnostic precision and supports personalized treatment strategies. Nonetheless, several challenges remain, including inter-assay variability, population heterogeneity, and standardization across laboratories, and limited validation in diverse clinical settings. Addressing these limitations is essential for translating biomarker discoveries into routine clinical practice. This review synthesizes current knowledge on CSF and blood biomarker advancements, evaluates their clinical applications, and discusses emerging challenges and future perspectives in biomarker-driven diagnosis and management of Alzheimer’s disease.
Keywords
Alzheimer’s disease; Amyloid-beta; Blood biomarkers; CSF biomarkers; Tau protein
Download this article as:| Copy the following to cite this article: Purna P. N, Kondrapu P, Ketha A, Metri S, Bandaru N. Review on Role of Cerebrospinal fluid and Blood biomarkers in Alzheimer’s Disease Diagnosis. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Purna P. N, Kondrapu P, Ketha A, Metri S, Bandaru N. Review on Role of Cerebrospinal fluid and Blood biomarkers in Alzheimer’s Disease Diagnosis. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4hSVosg |
Introduction
Alzheimer’s disease is a brain disorder that gradually deteriorates memory and thinking abilities, eventually leading to the failure to do even the most basic tasks. Memory loss with initial symptoms that may differ from individual to individual. This is typically one of the earliest symptoms of Alzheimer’s. There are typically four stages of Alzheimer’s disease progression preclinical, moderate, and severe also called as late stage of the disease. Alzheimer’s disease is caused by damage to nerve cells in the brain. People suffering from Alzheimer’s disease may experience changes in mood, personality, and behavior. The buildup of β-amyloid protein in brain cells may result in nerve cell injury.1 The estimated prevalence rate in India is gradually increasing with an increase in the people’s changes in lifestyle especially adults in India who are suffering from Alzheimer’s disease. According to an Alzheimer’s report in 2023 7.4% of the Indian population is suffering from Alzheimer’s disease. Alzheimer’s is mostly seen in women than men across Indian country.2 Alzheimer’s is a degenerative neurological disorder caused by the deposition of β-amyloid peptides in the brain. That tangle around the brain nerve cells which lead to damage in the brain cells. 3 Overall estimated amounts of Alzheimer’s patients worldwide wide 50 million which may increase during the period by 2050.4 The main purpose of this study is to understand Alzheimer’s disease and its mechanisms and future methods of treatment by using biomarkers.
Early diagnosis of disease is crucial for effective therapy. Patients with Alzheimer’s disease should have numerous neurological evaluations, as well as an MRI of their brain. Early detection can be accomplished by observing the major symptoms of the disease, such as memory loss, language impairment, and trouble doing daily chores. Clinical diagnosis and medical history of the patient play a key role in the diagnosis of Alzheimer’s at the first stages of the treatment. During the clinical examination of the patient, the mini-mental state examination (MMSE) and the Montreal Cognitive Assessment (MoCA) are done to examine the cognitive condition of the patient.5
Biomarkers are the indications of the biological process and are also helpful in the diagnosis of the disease. Biomarkers are not only useful in disease diagnosis but also help in the treatment of the patient at all levels of the system.6 Biomarkers are usually seen in the blood and CSF through brain imagining. Biomarkers enable us to identified targets of the disease. Biomarkers such as amyloid and tau neurofilament light chains and glial fibrillatory acidic protein and a few biomarkers are used to measure Alzheimer’s disease. Biomarkers play a vital role in the development of drug discovery for long-term diseases such as cancer, heart and Alzheimer’s, and certain neurological diseases.7
Pathophysiology of Alzheimers Disease
Alzheimer’s disease is also caused due to the genetic factors in the patient mostly with the patients above 60 plus age. Genetically they are further categorized into two familial cases and sporadic cases.8
Familial cases are generally seen in families who have been affected with Alzheimer’s disease in more than one generation, while sporadic cases are complex because they involve both genetic and environmental factors. In terms of pathological conditions, it is two different concepts; one is about the decline of acetylcholine (ACh) levels in the brain, which results from neuron loss and leads to an increase in the effects of Alzheimer’s disease. The other concept which is most widely seen is the aggregation of β-amyloid proteins which accumulates in the brain neuronees and leads to neuron loss.
Down syndrome, also known as trisomy 21, is a genetic disorder caused by the presence of an extra copy of chromosome 21. This additional genetic material affects the development and function of various organs and systems, including the brain. Among the genes located on chromosome 21 is the amyloid precursor protein (APP) gene. In individuals with Down syndrome, the presence of an extra chromosome 21 results in a duplication of the APP gene, leading to its overexpression. The APP gene plays a crucial role in the production of amyloid-β (Aβ) protein. Under normal physiological conditions, the APP protein is cleaved by enzymes in a tightly regulated process. However, in the presence of excess APP—as seen in individuals with Down syndrome—this process becomes dysregulated, leading to increased production of amyloid-β peptides. These peptides have a strong tendency to aggregate and form insoluble plaques in the brain tissue.
The accumulation of amyloid-β plaques is one of the hallmark pathological features of Alzheimer’s disease. These plaques disrupt cell-to-cell communication and activate immune responses that trigger inflammation and the eventual destruction of brain cells. As a result, individuals with Down syndrome are at a significantly higher risk of developing early-onset Alzheimer’s disease, often beginning in their 40s or 50s. The overexpression of the APP gene is therefore a key molecular link between Down syndrome and Alzheimer’s disease, highlighting the critical role of genetic factors in neurodegenerative disease progression.
Genetic Mutations and Alzheimer’s disease: Role of Aβ, PSEN1, and PSEN2 in Aβ Aggregation
In certain familial forms of Alzheimer’s disease, mutations in specific genes have been found to play a critical role in the early onset and progression of the condition. Among these, mutations in the amyloid-beta (Aβ) region of the amyloid precursor protein (APP) gene, as well as in the presenilin 1 (PSEN1) and presenilin 2 (PSEN2) genes, are of particular importance. These genes are directly involved in the formation and functioning of the gamma-secretase enzyme complex, which is responsible for cleaving the APP protein to produce amyloid-β peptides.
When mutations occur in PSEN1 or PSEN2, they alter the activity of the gamma-secretase complex. This abnormal activity results in the production of amyloid-β peptides that are longer and more prone to aggregation—especially Aβ42, a highly aggregation-prone form. Similarly, mutations in the APP gene itself can lead to increased production or altered processing of Aβ, further promoting plaque formation. These aggregated Aβ peptides clump together to form toxic amyloid plaques, a major pathological hallmark of Alzheimer’s disease.
The buildup of these plaques in the brain interferes with neuronal communication, triggers inflammation, and ultimately contributes to widespread neurodegeneration. Thus, mutations in the APP, PSEN1, and PSEN2 genes not only disrupt normal protein processing but also directly accelerate the aggregation of amyloid-β, leading to early and aggressive forms of Alzheimer’s disease, particularly in familial cases. Understanding these genetic mutations is crucial for developing targeted therapies and early diagnostic approaches. The presence of the APOE4 allele is considered one of the risk factors for Alzheimer’s disease development in terms of genetic inheritance.9
Amyloid Plaques and Tau Tangles
One of the critical events in the pathogenesis of Alzheimer’s disease is the abnormal processing of the amyloid precursor protein (APP). During its metabolism, a fragment called the APP C-terminal fragment can accumulate within neurons. This fragment serves as a precursor to amyloid-β (Aβ) peptides, which, when produced in excess or abnormally cleaved, begin to aggregate. These aggregated Aβ peptides progressively form amyloid fibrils, which are insoluble and toxic to brain tissue. As the amyloid fibrils accumulate, they lead to the formation of amyloid plaques—dense, extracellular deposits predominantly found in the grey matter of the brain. These plaques are often surrounded by dystrophic neuritis and activated glial cells, causing a chronic inflammatory response in the brain. The persistent inflammation, driven by microglial activation and cytokine release, further contributes to neuronal injury and degeneration.
Parallel to the amyloid pathway is the tauopathy pathway, which involves abnormalities in the microtubule-associated protein tau. In Alzheimer’s disease, tau becomes abnormally hyperphosphorylated and starts forming paired helical filaments (PHFs). These filaments subsequently aggregate into neurofibrillary tangles (NFTs) within neurons. The presence of NFTs disrupts the normal microtubule structure, impairing axonal transport and cellular communication.
The combined effects of amyloid plaque deposition and tau-related neurofibrillary tangles culminate in neuronal loss and synaptic dysfunction. This neuronal degradation is a key contributor to the progressive cognitive decline observed in Alzheimer’s disease. The interplay between these two pathological hallmarks—amyloid and tau—underpins the complex neurodegenerative cascade characteristic of this disorder.
APP accumulation leading to axonal traffic defects influences the formation of PHF & NFT propagation. Whereas amyloid accumulation at grey matter which causes inflammation triggers APOE4 Allee. Thus, the two pathways lead to the development of Alzheimer’s disease.10
Neuroinflammation and Oxidative Stress
Neuroinflammation is one of the risk factors that initiate the inflammatory response in the brain by activating microglia which leads to proinflammatory reactions in the brain leading to cells or neuronal loss.11 Due to the release of inflammatory agents like TNF-α, interleukins in the brain. The main reason for the release of proinflammatory agents in the brain is an accumulation of the Aβ cells and NFT leading to damage to the neuron cells and activation of the microglia cells by DAMP’s the stimulation of proinflammatory molecules such as TNFα, IL-1β, IL-6, and IL-18 cause’s Neuroinflammation in the brain.12
Oxidative stress is mainly caused by reactive oxygen species (ROS). ROS plays a key role in the phosphorylation. The accumulation of the Aβ and NFT cells in the brain leads to an imbalance in the homeostasis in the brain.13 It not only initiates the ROS generation but also obstructs the removal of the ROS the levels of Fe, Cu, and Zn are also responsible for neuron loss in the brain.14 In Alzheimer’s patients there is a significant increase in metals like Fe, Cu, and Zn these metals induce H2O2 production.15
Genetic and Environmental Factors
Frequent results in Alzheimer’s disease with early onset. Only about 5% of familial Alzheimer’s disease cases are caused by autosomal mutations in the APP, PSEN1, or PSEN2 genes of the α-secretase. SAD is characterized by reduced hippocampus volume and increased disease prevalence, including diabetes, obesity, and poor Aβ clearance. It is linked to late-onset Alzheimer’s disease. Out of 70 individuals with AD have the gene allele in their DNA which is APOE which has APOE-2, APOE3, and APOE4.APOE-e4 is the gene that increases the risk of developing AD in individuals with an increase in their age.16-17
Environmental factors such as Pollution: pollution is also considered one of the risk factors in the present generations due to the gradual increase in pollution may lead to the initiation of inflammatory biomarkers in the brain which ultimately stimulates the accumulation of Aβ concentration levels in the brain which leads to cognitive decline. Prolonged exposure to the pollution may also lead to Alzheimer’s disease.18
Bad Nutrition: It is also considered a risk factor for developing Alzheimer’s disease. Nowadays due to increasing consumption of fatty foods and high-calorie intake, alcohol consumption may lead to an increase in the risk of developing obesity, hypertension, diabetes, and neuronal diseases in the future. This can be prevented by increasing the intake of vitamin and mineral-rich foods and antioxidants, omega-3 fatty acids in their diet.19
Improper sleep: with the increase in age people also develop sleep disorders. Sleep is the only state where the brain cleanses the toxins through CSF. Insufficient sleep leads to the accumulation of Aβ cells.20 Ooms et.al found that short-term sleep deprivation significant rise in Aβ-42 levels in CSF in healthy humans.21
These pathological processes form the foundation for identifying biomarkers, which are discussed in the following section on CSF biomarkers.”
CSF Biomarkers
Overview of CSF Biomarkers
CSF is the cerebrospinal fluid that circulates the sub-arachnoid space in the brain. Biomarkers are generally used to diagnose and find a particular disease.22
There are a few biomarkers that we usually see in every case of Alzheimer such as APOE, beta-amyloid, t-Tau protein, and p-Tau, and a few novel biomarkers such as Beta-site Amyloid.
Amyloid-Beta (Aβ) In CSF
The APP protein is the precursor to beta-amyloid in the cerebrospinal fluid. One of the most difficult components of researching AD initial changes is tracking them throughout time.23 These changes in individuals with Aβ plaque and NFT neuropathy could be challenging when they don’t show symptoms over a while. A method to address these difficulties is to examine individuals who carry a genetically inherited AD mutation in the amyloid precursor protein (APP), presenilin 1 (PSEN1), or presenilin 2 (PSEN2) gene, that results to increased relative output of the Aβ42 peptide throughout life and early brain Aβ plaque deposition.24
Tau and Phosphorylated Tau (P-Tau)
Tau is not adequately phosphorylated, resulting in neuronal and synaptic loss in the brain. In Alzheimer’s disease, tau phosphorylation increases threefold. Mutations of the MAPT gene alter tau’s normal interaction to tubulin, causing a buildup of phosphorylated tau.25 Tau can be phosphorylated at over 40 different locations. Tau phosphorylation at threonine 181 (p-tau181) increases in CSF in AD but not in other neurodegenerative disorders.26
Neurofilament Lightchain (Nfl)
Neurofilament heavy chain (NfH, 200-220 kDa), medium chain (NfM, 145-160 kDa), light chain (NFL, 68-70 kDa), and α-internexin (58-66 kDa in the central nervous system) or peripherin (57-59 kDa in the peripheral nervous system).are all subunits of neurofilaments, which are intermediate filament and class IV proteins.27
Glial Fibrillatory Acidic Proteins (GFAP)
Astrocytes are considered as the biomarkers of Alzheimer’s disease. It is also considered one of the most sensitive biomarkers in Alzheimer’s disease.28
Limitations and Challenges in CSF Biomarkers The most important drawback of this invasive treatment is the necessity for a lumbar puncture to collect CSF. This can be painful for patients and may discourage some from having testing owing to perceived risk.29 CSF biomarker concentrations might vary across people and over time, posing interpretation challenges.30 Certain CSF markers are raised in certain neurodegenerative disorders like Alzheimer’s, but they may also be high in other neurological disorders, limiting their diagnostic specificity.31
Blood-Based Biomarkers
Overview of Blood Biomarkers
Among the wide array of biomarkers currently studied in Alzheimer’s disease, blood biomarkers have emerged as particularly valuable due to their accessibility, affordability, and growing diagnostic reliability. Unlike cerebrospinal fluid (CSF) biomarkers, which require lumbar puncture and are more invasive, blood-based biomarkers offer a non-invasive, cost-effective, and scalable solution for both clinical and research settings. As depicted in Figure 1, blood biomarkers are becoming indispensable tools in the early detection and monitoring of Alzheimer’s disease, offering a promising alternative for routine screening and longitudinal tracking of disease progression.
Plasma Amyloid β and Tau
Plasma levels of amyloid-β (Aβ) and tau proteins—specifically phosphorylated tau—are increasingly being used to reflect the core pathological hallmarks of Alzheimer’s disease. The cholinergic system, which plays a vital role in cognitive function and memory, relies on neurotransmitters such as acetylcholine (Ach), dopamine, adrenaline, and noradrenaline for signal transmission. Dysregulation in these systems, often mirrored by altered levels of Aβ and tau in the blood, provides important insights into the ongoing neurodegenerative processes and helps guide therapeutic interventions.32
Neurofilament Light Chain in Blood
The neurofilament light chain (NfL) is a structural protein of neurons that is released into the bloodstream during axonal damage. Elevated NfL levels in blood serve as a sensitive marker of neurodegeneration, correlating with disease severity and progression. Its growing diagnostic and prognostic utility across various neurodegenerative disorders, including Alzheimer’s disease, has made it a key focus in blood-based biomarker research.
Emerging Blood Biomarkers
Beyond Aβ, tau, and NfL, several emerging blood biomarkers are under active investigation. These include markers related to inflammation, oxidative stress, synaptic function, and glial activity. Advances in proteomic and transcriptomic technologies have allowed for the discovery of novel molecules that may enhance the specificity and sensitivity of Alzheimer’s diagnosis and contribute to a more comprehensive understanding of disease pathology.
Advantages and Challenges of Blood Biomarkers
Blood biomarkers offer several compelling advantages: they are easily accessible, minimally invasive, cost-effective, and time-efficient. These features make them particularly attractive for use in population-wide screening, repeated assessments, and early disease detection. However, challenges remain in terms of standardization, sensitivity to early-stage pathology, and distinguishing Alzheimer’s disease from other neurodegenerative conditions. Continued research and technological refinement are essential to overcome these limitations and fully harness the potential of blood biomarkers in clinical practice.33
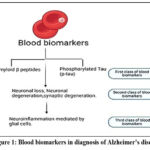 |
Figure 1: Blood biomarkers in diagnosis of Alzheimer’s disease |
Technological Advances in Bioarkers Discovery
Mass Spectroscopy and Proteomics
There are many advantages of using mass spectrometry-based proteomics to investigate the pathophysiology of Alzheimer’s disease. Technical and economic restrictions have previously hampered mass-spectrometry-based proteomic investigations; nevertheless, these variables have lately become less limiting, increasing the number of proteomics employing AD brain tissue. Proteomics is used to compare protein expression in AD and control brain homogenates, which are often confined to one susceptible brain area.34 This method’s ability to be used with formalin-fixed paraffin-embedded (FFPE) tissue is one of its main advantages. Most human tissue specimens for research are FFPE blocks from autopsies. One example protein that we discovered using proteomics and have since followed up on in a targeted study is secernin-1.Phosphorylated tau interacts with proteins involved in two major protein breakdown processes in the cell: the ubiquitin-proteasome system, TEM, and the phagosome-lysosome system.
Imaging Technologies and Biomarkers
The two main approaches for getting structure information on the brains of Alzheimer’s patients are computed tomography (CT) and magnetic resonance imaging (MRI).35 Guidelines propose utilizing one of them to assess individuals in the clinical setting who have cognitive/dementia syndrome (CDS).36 Because of its excellent spatial resolution, MRI is the most often used technology in diagnosing Alzheimer’s disease, which allows the difference between two arbitrarily similar but not identical tissues and multiparametric acquisitions to be recognized. Diffusion tensor imaging (DTI) and magnetic resonance spectroscopy (MRS) approaches have shown to define a spectrum of alterations associated with vascular diseases and white matter disorders in the brain, as well as to identify microstructure and metabolic abnormalities.37
Liquid Biopsy for Alzheimer’s Disease
A liquid biopsy is a test that is conducted on a sample of blood, urine, or other bodily fluid to check for cancer cells from a tumors or tiny pieces of RNA, DNA, or any other components. To understand the genetic and molecular changes in the DNA Blood-based liquid biopsy serves as a best alternative to CSF-based liquid biopsy due to its non-invasive nature and cost-effectiveness.38Each biological cluster’s clinical trajectory will then show, producing a novel liquid biopsy-based risk prediction matrix for the pathophysiology of AD in at-risk patients who do not exhibit symptoms. Following that, the clinical trajectories of each biological cluster will be plotted, producing a novel liquid biopsy-based risk prediction matrix for the pathophysiology of AD in at-risk individuals who do not exhibit symptoms.39 Vitamin B5 serves as a precursor for the enzyme PANK3, which is involved in the synthesis of Coenzyme A. This discovery bolsters earlier studies linking metabolic deregulation to Alzheimer’s and raises the possibility that food may contribute to the pathophysiology of the illness.40
Artificial Intelligence and Machine Learning in Alzheimers Disease:
MRI, PET, and CT scans are key methods for finding Alzheimer’s disease. MRI provides detailed pictures of the brain’s anatomy, making it possible to identify shrinkage in areas like the hippocampus, which is frequently impacted in AD. By measuring metabolic activity and amyloid beta deposition, PET scans provide information on the disease’s pathological and functional features. AI methods, particularly machine learning (ML), have been successfully employed to evaluate a wide range of data modalities for dementia-related disorders and to investigate various biomarkers. An open-source cohort called PREVENT-AD (Presymptomatic Evaluation of Experimental or Novel Treatments for Alzheimer’s disease) was created to study AD before symptoms appear. The Deep and Frequent Phenotyping cohort gathers information from imaging, digital wearable’s, fluid biomarkers, and clinical assessments that regularly measure cognition across time.41
When it comes to dementia, “early diagnosis” means detecting the illness early on, usually when cognitive decline is mild, enabling earlier intervention and management. “Difference from another dementia” means differentiating between dementia types such as Alzheimer’s, vascular dementia, or Lewy body dementia, which can show distinct symptoms and patterns of cognitive decline even in their initial stages.42 Are no major contributing factors that could be caused by anything could be delirium or neurological or systemic conditions apart from AD.There are several recognized phenotypic types of AD dementia, including atypical non-amnestic (mostly difficulties in word-finding, spatial cognition, or executive abilities) and typical amnestic (mainly impairments in learning and recall of newly learnt formation).43
Lewy body dementia range of conditions involving alterations in mobility,cognition, and autonomic functions. Dementia with Lewy bodies and dementia caused by Parkinson’s illness. Sleep disruption, muscle stiffness, and early visual hallucinations. Neurons have deposits of α-synuclein.44
Lobar degeneration in the front temporal region Frontal and temporal lobe focal atrophy; MRI shows knife-edge atrophy. Early onset, behavioral and personality abnormalities, linguistic difficulty, and a significant familial par.45
Vascular dementia focuses on neurologic symptoms and a stepwise progression. Moderate motor symptoms in sub cortical vascular dementia, depression, retrieval issues, dysexecutive syndrome, and reduced processing speed. AD symptoms are similar.46
Predicting Disease Progression and Cognitive Decline
Diagnostic and Prognostic Utility of Biomarkers have appeared as crucial tools in the early diagnosis of dementia, offering a means to detect pathophysiological changes before the onset of clinical symptoms. Anticipating cognitive symptoms can be aided by identifying biomarkers of Alzheimer’s disease (AD) pathology and implement preventative strategies. Early detection leads to personalized treatment solutions, needing continued study and cross-disciplinary collaboration to have a thorough understanding of AD pathophysiology. The integration of wearable devices and sensors for detecting digital biomarkers is rapidly advancing the field of Alzheimer’s disease (AD) research. Cognitive, behavioral, and motor changes often precede clinical symptoms, making early detection crucial for intervention. Researchers are increasingly leveraging consumer-grade mobile and wearable technologies to find digital biomarkers that can aid in large-scale screening and patient monitoring. 47 A key breakthrough in this area is the use of Smartphone and tablet-based digital biomarker tasks, such asAlodiaiADL tasks, which assess spatial memory and navigation. Additionally, episodic memory tasks like the Gallery Game have shown efficacy in finding preclinical stages of AD, improving recruitment for clinical trials, and disease tracking48. Sensor-based biomarkers are also gaining traction in early AD detection. These sensors, capable of monitoring eye movements, pupillary reflexes, and speech, offer valuable insights into disease onset. Camera and light sensors capture early-stage deficits in eye movements, a hallmark of AD.49 Similarly, speech analysis using machine learning tools such as the open SMILE v2.1 toolkit has shown promising results in diagnosing AD through acoustic featureextraction50. These approaches offer a non-invasive, cost-effective alternative to traditional biomarkers like imaging or blood tests. Blood-based biomarkers, although present in lower concentrations due to the blood-brain barrier, hold enormous potential for AD screening when paired with advanced analytical methods. Plasma-based screening of biomarkers such as amyloid-beta (Aβ1-42), tau proteins, and neurofilament light chain (NFL) has proved high diagnostic accuracy.51The use of mass spectrometry and immunoassays to measure these markers significantly enhances early diagnosis. Notably, longitudinal studies have confirmed that plasma biomarkers like p-tau181 and NFL can effectively track disease progression and cognitive decline, making them powerful tools for monitoringAD52. Biosensors are another promising frontier in AD research. Recent advancements include a label-free electrochemical biosensor for detecting amyloid-beta oligomers and a graphene oxide-based sensor for miRNA-34a, both of which could play a significant role in early AD diagnosis 53. The diagnosis of AD still heavily relies on medical imaging methods, especially positron emission tomography (PET) and magnetic resonance imaging (MRI).These methods enable non-invasive visualization of brain changes associated with AD. Deep learning models, trained on PET and MRI scans, have shown superior performance in finding AD-related abnormalities compared to traditional radiology assessments. AI-driven models, such as InceptionV3 and PETNet, offer highly correct and computationally efficient approaches for early AD diagnosis 54. The autonomic nervous system (ANS) also undergoes pathological changes in AD, with heart rate variability (HRV) serving as a potential biomarker for cognitive impairment 55. Studies suggest that reduced parasympathetic activity correlates with AD progression. Photoplethysmography (PPG) technology integrated into smartwatches and pulse oximeters has been used to measure HRV, offering an accessible and non-invasive method for AD detection 56 multi-sensor approaches combining HRV monitoring with gait analysis have deproteinising results in distinguishing mild cognitive impairment from normal aging. The integration of digital biomarkers, wearable technologies, biosensors, blood-based markers, and advanced imaging techniques is revolutionizing AD research. These innovative approaches not only fasciately detection but also enhance disease monitoring, treatment response assessment, and clinical trial recruitment. As technological advancements continue, the potential for non-invasive, cost-effective, and scalable solutions in AD diagnostics becomes increasingly practical, laying the path for more focused treatment approaches and better patient outcomes.
Biomarkers Used in Clinical Trials for Alzheimer’s disease
In clinical trials, biomarkers play a crucial role in assisting with early diagnosis and monitoring the course of disease, and evaluating therapeutic responses. These biomarkers are derived from cerebrospinal fluid (CSF), blood, imaging modalities, and genetic or epigenetic markers. In AD clinical trials, biomarkers are categorized based on their involvement in key pathological processes: amyloid pathology, tau pathology, neurodegeneration, neuroinflammation, synaptic dysfunction, and genetic predisposition. While CSF and imaging biomarkers stay the gold standard, emerging blood-based biomarkers are becoming increasingly relevant for early and non-invasive diagnosis.57 The following table presents a comprehensive overview of validated and emerging biomarkers used in Alzheimer’s disease clinical trials, detailing their category, sample type, clinical utility, and supporting references.
Table 1: Overview of Biomarkers Used in Clinical Trials for Alzheimer’s disease
| Biomarker | Type | Clinical Utility | Sample Type | Reference |
| Aβ42/Aβ40 ratio | Fluid | Diagnosis, disease staging | CSF, Plasma | 58 |
| PET Amyloid Imaging | Imaging | Early detection, clinical trial inclusion criteria | PET Scan | 59 |
| Amyloid PET (Florbetapir, Flutemetamol, Florbetaben) | Imaging | Outcome measure, disease progression tracking | PET Scan | 60 |
| CSF Total Tau (t-tau) | Fluid | Disease progression, neuronal damage indicator | CSF | 61 |
| CSF Phosphorylated Tau (p-tau181, p-tau217) | Fluid | Diagnosis, response to anti-tau therapy | CSF | 62 |
| Neurofilament Light Chain (NfL) | Fluid | Synaptic damage and neurodegeneration marker | CSF, Blood | 63 |
| Glial Fibrillary Acidic Protein (GFAP) | Fluid | Astrocytic activation, disease progression | Plasma | 64 |
| MRI-based Hippocampal Atrophy | Imaging | Structural biomarker for disease staging | MRI | 65 |
| Synaptosome-Associated Protein 25 (SNAP-25) | Fluid | Synaptic degeneration marker | CSF | 66 |
| SV2A PET Imaging | Imaging | Synaptic density marker | PET Scan | 67 |
| Plasma p-tau217 | Fluid | Non-invasive diagnostic biomarker | Plasma | 68 |
| Plasma Aβ42/Aβ40 ratio | Fluid | Screening and disease progression | Plasma | 69 |
| Apolipoprotein (APOE ε4) | Genetic | Risk stratification, clinical trial inclusion | Blood (DNA) | 70 |
| Polygenic Risk Scores (PRS) | Genetic | Risk prediction model | Blood (DNA) | 71 |
| DNA Methylation (e.g., 5-hmC in APP, PSEN1) | Epigenetic | Early diagnosis | Blood, CSF | 72 |
Standardization and Validation of Biomarkers
For Alzheimer’s disease (AD) biomarkers to be successfully incorporated into clinical practice and research, they must be standardized and validated. Standardization ensures consistency and reliability across different laboratories and studies, while validation confirms that these biomarkers accurately reflect the disease processes, they are intended to measure.73
Standardization Efforts
Several initiatives have been set up to promote the standardization of AD biomarkers
Global Biomarker Standardization Consortium (GBSC): Founded by the Alzheimer’s Association, the GBSC brings together experts from academia, industry, and regulatory agencies to develop consensus on standardizing biomarker tests for global clinical application.
Standardization of Alzheimer’s Blood Biomarkers (SABB) Workgroup: This collaborative project aims to create standardized operating procedures (SOPs) for blood-based biomarker measurements, helping their best use in clinical studies and practice. 74
Validation Methodologies
The validation of AD biomarkers involves several key steps:
Analytical Validation: Assessing the precision, accuracy, and reproducibility of biomarker assays across different platforms and laboratories.
Clinical Validation:Setting up the biomarker’s ability to detect or predict clinical outcomes associated with AD, such as cognitive decline or disease progression.
Cutoff Determination: Defining threshold values that distinguish between normal and pathological states, which is crucial for diagnostic decision-making.
Regulatory Considerations
For biomarkers to transition from research settings to routine clinical use, they must undergo rigorous evaluation by regulatory bodies to ensure their safety and efficacy. This process includes Compliance with Guidelines: Adhering to set up protocols for biomarker development and validation. DemonstratingClinical Utility: Providing evidence that the biomarker improves patient outcomes or aids in clinical decision-making 75
Clinical Guidelines and Biomarker Integration
The integration of biomarkers into clinical guidelines for Alzheimer’s disease (AD) has significantly enhanced diagnostic precision and patient management. Traditionally, AD diagnosis relied heavily on clinical assessments and cognitive evaluations. However, the advent of biomarkers has introduced objective measures that reflect underlying neuropathological changes, enabling earlier and more correct detection.76
Incorporation into Diagnostic Criteria
Recent revisions to AD diagnostic criteria emphasize a biological definition of the disease, incorporating biomarkers as central components.According to the 2024 criteria, a Core 1 biomarker test that is abnormal is enough to establish an AD diagnosis and guide clinical judgment across the disease spectrum. Amyloid positron emission tomography (PET), authorized cerebrospinal fluid (CSF) biomarkers, and accurate plasma biomarkers—particularly phosphorylated tau 217—are examples of core 1 biomarkers. These biomarkers indicate the existence of AD neuropathological alterations by mapping onto either the amyloid beta or AD tauopathy pathway.77
Biomarkers derived from blood in clinical practice:
The development of blood-based biomarkers (BBMs) provides a more accessible and less intrusive substitute for conventional diagnostic techniques such as CSF analysis and amyloid PET. BBMs are increasingly used in clinical practice to support AD diagnosis, offering a cost-effective choice for biomarker testing. Their integration into clinical guidelines is underway, with recommendations focusing on their proper use and interpretation in various clinical contexts.78
Guideline Development and Standardization Efforts
Organizations such as the Alzheimer’s Association are at the forefront of developing clinical practice guidelines that incorporate biomarkers. Utilizing rigorous methodologies like the GRADE framework, these guidelines distil the best available evidence to support decision-making in patient care. Efforts are also directed toward standardizing biomarker assays and setting up clear protocols for their clinical application, ensuring consistency and reliability across different healthcare settings.79
Challenges and Future Directions
While the integration of biomarkers into clinical guidelines marks considerable progress, challenges stay. Ensuring the widespread availability and affordability of biomarker testing, particularly in resource-limited settings, is crucial. Additionally, continuous research is needed to confirm emerging biomarkers and refine their application in diverse populations. Future guidelines will likely evolve to incorporate novel biomarkers and advanced diagnostic technologies, further enhancing the precision of AD diagnosis and treatment.80
Ethical and Practical Considerations
Numerous moral and practical difficulties arise while providing care for people with Alzheimer’s disease (AD) that healthcare providers, caregivers, and families must navigate to ensure compassionate and effective care.
Ethical Considerations
Respecting Patient Autonomy
As AD progresses, patients experience diminishing decisional ability. It’s imperative to honour their autonomy by involving them in care decisions as much as possible and respecting their previously expressed wishes.
Informed Consent
Obtaining informed consent for treatments and participation in research becomes complex as cognitive functions decline. Ensuring that patients understand the implications of medical decisions is essential, and when they cannot, involving legally authorized representatives is necessary.
Early Diagnosis Implications
While early diagnosis can ease planning and intervention, it also raises concerns about the patient’s psychological well-being, potential stigma, and the impact on personal identity.
Use of Emerging Therapies
The advent of treatments like anti-amyloid immunotherapy introduces ethical dilemmas related to beneficence and nonmaleficence. In order to ensure that patients and their families are informed, clinicians must balance the possible advantages against risks and adverse consequences. 81
Practical Considerations
Caregiver Support
Caregivers often face emotional and physical burdens, including depression and anxiety. Providing them with resources, respite care, and support networks is crucial to keeping their well-being and sustaining the quality of care.
Behavioural Management
Individuals with AD may show challenging yours. Implementing implement routines, creating safe environments, and addressing underlying causes like pain or overstimulation can mitigate these issues.
Access to Care
Barriers such as excessive costs, limited availability of specialized providers, and logistical challenges can impede access to quality care. Addressing these issues requires systemic changes and advocacy for better resources and support.
Care Planning
Developing comprehensive care plans that adapt to the evolving needs of the patient ensures continuity and quality of care. This includes medical management, daily living aid, and end-of-life considerations. 82
Future Directions in Alzheimer’s disease biomarker research (AD)
Challenges in Translating Biomarkers into Clinical Use
challenges stay one of the most complex and challenging neurodegenerative disorders, with early and correct diagnosis being a critical yet unresolved issue. Biomarker research has made significant strides in recent years, with cerebrospinal fluid (CSF) and imaging-based markers offering valuable insights into disease pathology.However, translating these biomarkers into routine clinical practice faces multiple challenges, needing further advancements to refine their utility, improve diagnostic accuracy, and enable personalized treatment strategies. One of the foremost challenges in translating AD biomarkers into clinical use is the variability in biomarker performance across different patient populations. While biomarkers in CSF and positron emission tomography (PET) imaging, such tau protein and amyloid-β (Aβ), provide robust disease indicators, their accessibility and cost remain barriers to widespreadadoption. Additionally, inter-individual variability in biomarker expression complicates standardization, making it difficult to define universally applicable diagnostic thresholds.83
Biomarker Combinations for Improved Diagnostics
Combining blood-based biomarkers like neurofilament light chain (NfL) and phosphorylated tau (p-tau), offers a promising alternative, but their validation across diverse cohorts is essential before they can replace invasive and expensive techniques.Given the limitations of single biomarkers, a shift toward biomarker combinations is appearing as a key strategy to improve diagnostic accuracy. Multi-modal approaches that integrate fluid biomarkers, neuroimaging, and digital cognitive assessments are being explored to enhance sensitivity and specificity. For instance, combining plasma p-tau with amyloid PET scans may allow for earlier detection of AD pathology, even before the onset of clinical symptoms. Similarly, machine learning algorithms applied to multi-omics data, including proteomics, transcriptomics, and metabolomics, can help uncover novel biomarker panels that offer a more holistic view of disease progression. Such combinations not only improve diagnostic precision but also ease the staging of AD, enabling early intervention strategies. Another crucial avenue in AD biomarker research is the incorporation of personalized medicine approaches.84
Methods of Personalized Medicine in Alzheimer’s disease:
Given heterogeneity AD, biomarkers must be tailored to individual patient profiles to predict disease trajectory and treatment response more effectively. Genetic factors, such as APOE ε4 status, influence biomarker expression, and disease progression, underscoring the need for stratified diagnostic models. Furthermore, emerging technologies like Spatial transcriptomics and single-cell RNA sequencing are opening the door to a better comprehension of specific disease pathways. This precision-medicine paradigm could ultimately lead to targeted therapeutic interventions, improving patient outcomes by addressing specific molecular pathways involved in disease pathology. As the field advances, to close the gap between biomarker discovery and clinical use, these issues must be resolved. Biomarker assay standardization, cross-population validation, and incorporation of AI-powered analytical instruments will be a key in shaping the future of AD diagnostics and treatment. With ongoing progress in biomarker research, the vision of early, precise, and personalized Alzheimer’s disease management is becoming an attainable reality.85
Discussion
Although cerebrospinal fluid (CSF) biomarkers such as Aβ42, t-tau, and p-tau remain the gold standard for Alzheimer’s disease (AD) diagnosis, their invasive collection limits routine application. Blood-based biomarkers, particularly plasma p-tau isoforms and the Aβ42/40 ratio, have demonstrated strong correlations with AD pathology and clinical progression, yet variability across studies raises questions about their reliability in diverse populations. For example, while p-tau217 is reported to outperform p-tau181 in distinguishing AD from other dementias, conflicting evidence suggests assay- and cohort-specific differences, indicating that no single biomarker can be universally relied upon. Similarly, neurofilament light chain (NfL) is a sensitive marker of neuronal damage but lacks specificity, as it is elevated in other neurodegenerative disorders. These inconsistencies highlight the need to interpret biomarker data in context and consider alternative explanations for abnormal values.
Another important consideration is whether biomarkers merely reflect ongoing pathology or can truly predict disease onset before symptoms appear. This debate underscores the importance of integrating biomarkers with imaging modalities, cognitive testing, and emerging digital biomarkers to improve diagnostic accuracy. Furthermore, environmental and lifestyle factors such as sleep deprivation, pollution, and nutrition may influence biomarker levels, suggesting that interpretation should extend beyond neuropathological mechanisms alone.
Overall, while significant progress has been made, translation into clinical practice requires overcoming challenges in assay standardization, validation across populations, and accessibility in resource-limited settings. Biomarker-driven diagnosis must therefore be viewed as part of a multi-modal strategy rather than a standalone solution.
Conclusion
Alzheimer’s disease remains one of the most challenging neurodegenerative disorders, where early and accurate diagnosis is crucial for effective intervention. Cerebrospinal fluid (CSF) biomarkers, particularly Aβ42, total tau, and phosphorylated tau, provide robust insights into underlying pathology and remain the current gold standard. However, their invasive collection limits widespread use. Recent advances in blood-based biomarkers, especially plasma p-tau isoforms, Aβ42/40 ratios, and neurofilament light chain (NfL), offer promising non-invasive alternatives that strongly correlate with disease progression. Despite these advancements, significant challenges remain. Variability across assays, lack of standardization, and limited validation in diverse populations restrict their translation into routine clinical settings. Moreover, some biomarkers, such as NfL, reflect general neurodegeneration rather than AD-specific pathology, underscoring the importance of multimodal approaches. Integrating fluid biomarkers with neuroimaging, genetic profiling, and digital health technologies may provide the most reliable diagnostic framework.
Overall, the evidence supports that biomarkers have the potential to shift AD diagnosis from symptom-based recognition to biologically informed, earlier detection. Yet, careful standardization, large-scale validation, and cost-effective implementation strategies are essential before these tools can be fully embedded in clinical practice. Future research should focus on combining complementary biomarkers into diagnostic panels and aligning discovery with patient-centered applications to achieve precision medicine in Alzheimer’s disease.
Acknowledgment
All authors are thankful to Management of Sandip University, Nasik, for providing facilities.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author’s contribution:
- Nagaraju Bandaru – Contributed to conceptualization and methodology,
- Pydiraju Kondrapu, Pydikondala Nehitha Purna, Shashikala Metri: Performed the literature research, analyzed the data and drafted the manuscript
- Alekya Ketha: critically revised the work
- All authors are reviewed and approved the manuscript
References
- Alzheimer’s disease facts and figures. Alzheimers Dement. 2024;20(5):3708-3821. doi:10.1002/alz.13809
CrossRef - Alvarez XA, Winston CN, Barlow JW, et al. Modulation of Amyloid-β and Tau in Alzheimer’s Disease Plasma Neuronal-Derived Extracellular Vesicles by Cerebrolysin® and Donepezil. J Alzheimers Dis. 2022;90(2):705-717. doi:10.3233/JAD-220575
CrossRef - Bartolotti N, Lazarov O. Lifestyle and Alzheimer’s Disease: The Role of Environmental Factors in Disease Development. In: Genes, Environment and Alzheimer’s Disease. Elsevier; 2016:197-237. doi:10.1016/B978-0-12-802851-3.00007-3
CrossRef - Battie C, Verschoore M. Cutaneous solar ultraviolet exposure and clinical aspects of photodamage. Indian J Dermatol Venereol Leprol. 2012;78(Suppl 1):9-14. doi:10.4103/0378-6323.97350
CrossRef - Battle MR, Vandenberghe R, Koole M, et al. Centiloid scaling for quantification of brain amyloid with [18F]flutemetamol using multiple processing methods. EJNMMI Res. 2018;8:1-11.
CrossRef - Bavato F, Barro C, Schnider LK, et al. Introducing neurofilament light chain measure in psychiatry: Current evidence, opportunities, and pitfalls. Mol Psychiatry. 2024. doi:10.1038/s41380-024-02524-6
CrossRef - Brudek T, Winge K, Folke J, et al. Autoimmune antibody decline in Parkinson’s disease and multiple system atrophy; a step towards immunotherapeutic strategies. Mol Neurodegener. 2017;12(1):1-11. doi:10.1186/s13024-017-0187-7
CrossRef - Cefali LC, Ataide JA, Moriel P, Foglio MA, Mazzola PG. Plant-based active photoprotectants for sunscreens. Int J Cosmet Sci. 2016;38(4):346-353. doi:10.1111/ics.12316
CrossRef - Chae MS, Yoon Y, Kim J, Kim T, Ha KS. Graphene-based enzyme-modified field-effect transistor biosensor for monitoring drug effects in Alzheimer’s disease treatment. Sens Actuators B Chem. 2018;272:128-135.
CrossRef - Chekuri S, Vankudothu N, Panjala S, Rao NB, Anupalli RR. Phytochemical analysis, anti-oxidant and anti-microbial activity of “Acalypha indica” leaf extracts in different organic solvents. Int J Phytomedicine. 2016;8(3):364-370. doi:10.5138/09750185.1882
CrossRef - Chen ZR, Huang JB, Yang SL, Hong FF. Role of cholinergic signaling in Alzheimer’s disease. Molecules. 2022;27(6):1816. doi:10.3390/molecules27061816
CrossRef - Chiong W, Tsou AY, Simmons Z, Bonnie RJ, Russell JA. Ethical considerations in dementia diagnosis and care: AAN position statement. Neurology. 2021;97(2):80-89. doi:10.1212/WNL.0000000000012079
CrossRef - Cicognola C, Janelidze S, Hertze J, et al. Plasma glial fibrillary acidic protein detects Alzheimer pathology and predicts future conversion to Alzheimer dementia in patients with mild cognitive impairment. Alzheimers Res Ther. 2021;13:68.
CrossRef - Corder EH, Saunders AM, Strittmatter WJ, et al. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. 1993;261(5123):921-923.
CrossRef - Cummings J. The role of biomarkers in Alzheimer’s disease drug development. Adv Exp Med Biol. 2019;1118:29-61. doi:10.1007/978-3-030-05542-4_2
CrossRef - Ding Y, Sohn JH, Kawczynski M, et al. A deep learning model to predict a diagnosis of Alzheimer disease by using 18F-FDG PET of the brain. Radiology. 2019;290(2):456-464.
CrossRef - Doecke JD, Pérez-Grijalba V, Fandos N, et al. Total Aβ42/Aβ40 ratio in plasma predicts amyloid-PET status, independent of clinical AD diagnosis. 2020;94(17):e1580-e1591.
CrossRef - Duck KA, Connor JR. Iron uptake and transport across physiological barriers. 2016;29(4):573-591. doi:10.1007/s10534-016-9952-2
CrossRef - Dunn AR, O’Connell KMS, Kaczorowski CC. Gene-by-environment interactions in Alzheimer’s disease and Parkinson’s disease. Neurosci Biobehav Rev. 2019;103:73-80. doi:10.1016/j.neubiorev.2019.06.018
CrossRef - Duthie MS, Kimber I, Norval M. The effects of ultraviolet radiation on the human immune system. Br J Dermatol. 1999;140(6):995-1009. doi:10.1046/j.1365-2133.1999.02898.x
CrossRef - Elkazzaz SK, Khodeer DM, el Fayoumi HM, Moustafa YM. Role of sodium glucose cotransporter type 2 inhibitors dapagliflozin on diabetic nephropathy in rats; Inflammation, angiogenesis and apoptosis. Life Sci. 2021;280:119018. doi:10.1016/j.lfs.2021.119018
CrossRef - Gong X, Zhang H, Liu X, et al. Is liquid biopsy mature enough for the diagnosis of Alzheimer’s disease? Front Aging Neurosci. 2022;14:977999. doi:10.3389/fnagi.2022.977999
CrossRef - Govindarajan M, Jebanesan A, Reetha D, et al. Antibacterial activity of Acalypha indica L. Eur Rev Med Pharmacol Sci. 2008;12(5):347-351. doi:10.17485/ijst/2008/v1i6.2
CrossRef - Growdon Mattsson N, Murphy E. Virtual Mentor. AMA J Ethics. 2011;13(5):298-304.
CrossRef - Nasreen S, Rohanian M, Hough J and Purver M (2021) Alzheimer’s Dementia Recognition From Spontaneous Speech Using Disfluency and Interactional Features. Comput. Sci. 3:640669. doi: 10.3389/fcomp.2021.640669
CrossRef - Hakim RW, Fadilah F, Tarigan TJE, Jusman SWA, Purwaningsih EH. Molecular study of Acalypha indica to leptin, alpha glucosidase, and its antihyperglycemic effect on alpha glucosidase. Pharmacogn J. 2021;13(6):1492-1499. doi:10.5530/pj.2021.13.211
CrossRef - Hampel H, Goetzl EJ, Kapogiannis D, Lista S, Vergallo A. Biomarker-drug and liquid biopsy co-development for disease staging and targeted therapy: Cornerstones for Alzheimer’s precision medicine and pharmacology. Front Pharmacol. 2019;10:310. doi:10.3389/fphar.2019.00310
CrossRef - Hansson O, Blennow K, Zetterberg H, Dage J. Blood biomarkers for Alzheimer’s disease in clinical practice and trials. Nat Aging. 2023;3(5):506-519. doi:10.1038/s43587-023-00403-3
CrossRef - Hansson O, Edelmayer RM, Boxer AL, et al. The Alzheimer’s Association appropriate use recommendations for blood biomarkers in Alzheimer’s disease. Alzheimers Dement. 2022;18(12):2669-2686. doi:10.1002/alz.12756
CrossRef - Hansson O, Lehmann S, Otto M, Zetterberg H, Lewczuk P. Advantages and disadvantages of the use of the CSF amyloid β (Aβ) 42/40 ratio in the diagnosis of Alzheimer’s disease. Alzheimers Res Ther. 2019;11(1):34. doi:10.1186/s13195-019-0485-0
CrossRef - Harper L, Barkhof F, Scheltens P, Schott JM, Fox NC. An algorithmic approach to structural imaging in dementia. J Neurol Neurosurg Psychiatry. 2014;85(6):692-698. doi:10.1136/jnnp-2013-306285
CrossRef - Hickey JP, Collins AE, Nelson ML, Chen H, Kalisch BE. Modulation of oxidative stress and neuroinflammation by cannabidiol (CBD): Promising targets for the treatment of Alzheimer’s disease. Curr Issues Mol Biol. 2024;46(5):4379-4402. doi:10.3390/cimb46050266
CrossRef - Hoogmartens J, Cacace R, van Broeckhoven C. Insight into the genetic etiology of Alzheimer’s disease: A comprehensive review of the role of rare variants. Alzheimers Dement Diagn Assess Dis Monit. 2021;13(1):e12155. doi:10.1002/dad2.12155
CrossRef - Hu N, Yu JT, Tan L, Wang YL, Sun L, Tan L. Nutrition and the risk of Alzheimer’s disease. Biomed Res Int. 2013;2013:524820. doi:10.1155/2013/524820
CrossRef - Hu N, Yu JT, Tan L, et al. Elevated plasma neurofilament light was associated with multi-modal neuroimaging features in Alzheimer’s disease signature regions and predicted future tau deposition. BMC Neurol. 2024;24(1):1-19.
CrossRef - Huang WJ, Zhang X, Chen WW. Role of oxidative stress in Alzheimer’s disease (review). Biomed Rep. 2016;4(5):519-522. doi:10.3892/br.2016.630
CrossRef - Iliyasu MO, Musa SA, Oladele SB, Iliya AI. Amyloid-beta aggregation implicates multiple pathways in Alzheimer’s disease: Understanding the mechanisms. Front Neurosci. 2023;17:1081938. doi:10.3389/fnins.2023.1081938
CrossRef - Jack CR, Andrews JS, Beach TG, et al. Revised criteria for diagnosis and staging of Alzheimer’s disease: Alzheimer’s Association Workgroup. Alzheimers Dement. 2024;20(8):5143-5169. doi:10.1002/alz.13859
CrossRef - Jack CR Jr, Bennett DA, Blennow K, et al. NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimers Dement. 2018;14(4):535-562.
CrossRef - Janelidze S, Stomrud E, Smith R, et al. Cerebrospinal fluid p-tau217 performs better than p-tau181 as a biomarker of Alzheimer’s disease. Nat Commun. 2020;11(1):1683. doi:10.1038/s41467-020-15436-0
CrossRef - Juganavar A, Joshi A, Shegekar T. Navigating early Alzheimer’s diagnosis: A comprehensive review of diagnostic innovations. Cureus. 2023;15(11):e44937. doi:10.7759/cureus.44937
CrossRef - Kaimal S, Abraham A. Sunscreens. Indian J Dermatol Venereol Leprol. 2011;77(3):300-307. doi:10.4103/0378-6323.82403
CrossRef - Karantzoulis S, Galvin JE. Distinguishing Alzheimer’s disease from other major forms of dementia. Expert Rev Neurother. 2011;11(11):1579-1591. doi:10.1586/ern.11.155
CrossRef - Kaya D, Isik AT. Cerebrospinal fluid biomarkers for normal pressure hydrocephalus. Biomark Neuropsychiatry. 2023;9:100071. doi:10.1016/j.bionps.2023.100071
CrossRef - Yiannopoulou KG, Papageorgiou SG. Current and future treatments in Alzheimer disease: An update. Saga J. 2020;2:1-10.
CrossRef - Lancaster C, Koychev I, Bartlett J, et al. Gallery Game: Smartphone-based assessment of long-term memory in adults at risk of Alzheimer’s disease. J Clin Exp Neuropsychol. 2020;42(4):358-369. doi:10.1080/13803395.2019.1702205
CrossRef - Lee J, Meijer E, Langa KM, et al. Prevalence of dementia in India: National and state estimates from a nationwide study. Alzheimers Dement. 2023;19(7):2898-2912. doi:10.1002/alz.12928
CrossRef - Leipp F, Vialaret J, Mohaupt P, et al. Glial fibrillary acidic protein in Alzheimer’s disease: A narrative review. Brain Commun. 2024;6(6):fcae396. doi:10.1093/braincomms/fcae396
CrossRef - Bandaru N, Bonthu MG, Gayatri AP, et al. Review on Exploring Role of Vitamin D on Alzheimer’s Disease: Mechanistic Insights and Implications. Journal of Pharmacology and Pharmacotherapeutics. 2025;16(2):164-171. doi:10.1177/0976500X241312726
CrossRef - Maji SK, Anoop A, Singh PK, Jacob RS. CSF biomarkers for Alzheimer’s disease diagnosis. Int J Alzheimers Dis. 2010;2010:606802. doi:10.4061/2010/606802
CrossRef - Bandaru N, Shamim N, Nagalakshmi S. B, Sunanda T, Hanisha C, Gambhire M. S, Dudhe P. B, Kranthi Y, Kumar P. K, Gowravi P. N. S. Preparation of Platinum Nanoparticles of Biophytum reinwardtii and Evaluation of Neuroprotective Activity of MPTP-induced Parkinson’s Disease in Zebra Fish. Biomed Pharmacol J 2024;17(3).
CrossRef - Prakash, K. A., Nagmoti, D. S., Borkar, M. S., Pannalal, H. K., Dhokchawle, B. V., & Bandaru, N. (2025). Integrating AYUSH Therapies for the Management of Neurodegenerative Disorders. Journal of Natural Remedies, 25(5), 995–1021. https://doi.org/10.18311/jnr/2025/48564
CrossRef - Kale M, Wankhede N, Pawar R, Ballal S, Kumawat R, Goswami M, Khalid M, Taksande B, Upaganlawar A, Umekar M, Kopalli SR, Koppula S. AI-driven innovations in Alzheimer’s disease: Integrating early diagnosis, personalized treatment, and prognostic modelling. Ageing Res Rev. 2024 Nov;101:102497. doi: 10.1016/j.arr.2024.102497.
CrossRef - Mielke MM, Anderson M, Ashford JW, et al. Recommendations for clinical implementation of blood-based biomarkers for Alzheimer’s disease. Alzheimers Dement. 2024;20(4):1110-1125. doi:10.1002/alz.14184
CrossRef - Morgan AE, Mc Auley MT. Vascular dementia: From pathobiology to emerging perspectives. Ageing Res Rev. 2024;96:102278. doi:10.1016/j.arr.2024.102278
CrossRef - Nilsson J, Portelius E, Ashton NJ, et al. Cerebrospinal fluid biomarker panel for synaptic dysfunction in a broad spectrum of neurodegenerative diseases. Brain. 2024;147(8):2414-2427. doi:10.1093/brain/awae118
CrossRef - Ooms S, Overeem S, Besse K, et al. Effect of 1 night of total sleep deprivation on cerebrospinal fluid β-amyloid 42 in healthy middle-aged men: A randomized clinical trial. JAMA Neurol. 2014;71(8):971-977. doi:10.1001/jamaneurol.2014.1173
CrossRef - Opwonya J, Dong D, Koychev I, et al. Saccadic eye movement in mild cognitive impairment and Alzheimer’s disease: A systematic review and meta-analysis. Neuropsychol Rev. 2022;32(4):972-991. doi:10.1007/s11065-022-09536-3
CrossRef - Pandey N, Yu Z, Chen B, et al. Plasma phospho-tau217 as a predictive biomarker for Alzheimer’s disease in a large South American cohort. Alzheimers Res Ther. 2025;17(1):1-14. doi:10.1186/s13195-025-01430-y
CrossRef - Papaliagkas V, Kalinderi K, Vareltzis P, et al. CSF biomarkers in the early diagnosis of mild cognitive impairment and Alzheimer’s disease. Int J Mol Sci. 2023;24(10):8976. doi:10.3390/ijms24108976
CrossRef - Park JE, GTCYCSMCSet al. Diagnostic Blood Biomarkers in Alzheimer’s Disease. Biomedicines. 2022;10(12):3153. doi:10.3390/biomedicines10123153
CrossRef - Pasquier F. Early diagnosis of dementia: neuropsychology. J Neurol. 1999;246(1):6-9. doi:10.1007/s004150050299
CrossRef - Perpetuini D, Cardone D, Filippini C, Chiarelli AM, Merla A. Modelling impulse response function of functional infrared imaging for general linear model analysis of autonomic activity. 2018 International
Symposium on Fundamentals of Electrical Engineering (ISFEE). 2018:1-4. doi:10.1109/ISFEE.2018.8659443
CrossRef - Perrin RJ, Fagan AM, Holtzman DM. Multimodal techniques for diagnosis and prognosis of Alzheimer’s disease. Nature. 2009;461(7266):916-922. doi:10.1038/nature08538
CrossRef - Piaceri I, Nacmias B, Sorbi S. Genetics of familial and sporadic Alzheimer’s disease. Front Biosci (Elite Ed). 2013;5:167-177. doi:10.2741/E702
CrossRef - Pillai JA, Bonner-Jackson A, Bena J, et al. Highly Elevated Cerebrospinal Fluid Total Tau Level Reflects Higher Likelihood of Non-Amnestic Subtype of Alzheimer’s Disease. J Alzheimers Dis. 2019;70(4):1051-1059. doi:10.3233/JAD-190191
CrossRef - Prasad S, Katta MR, Abhishek S, et al. Recent advances in Lewy body dementia: A comprehensive review. Dis Mon. 2023;69(5):101441. doi:10.1016/j.disamonth.2022.101441
CrossRef - Priyanka S, Inala MSR, Nandini H, Kutty A, Kiranmayee P. A pilot study on sun protection factor of plant extracts: An observational study. Asian J Pharm Clin Res. 2018;11(4):67-71. doi:10.22159/ajpcr.2018.v11i4. 23671
CrossRef - Rabinovici GD, Miller BL. Frontotemporal lobar degeneration: Epidemiology, pathophysiology, diagnosis and management. CNS Drugs. 2010;24(5):375-398. doi:10.2165/11533100-000000000-00000
CrossRef - Rai L, Bleichner R, Hilfiker R, et al. Digital Biomarkers Based Individualized Prognosis for People at Risk of Dementia: The AltoidaML Multi-site External Validation Study. Adv Exp Med Biol. 2020;1194:29-34. doi:10.1007/978-3-030-32633-4_5
CrossRef - Rao G, Gong H, Wang X, et al. MRI measurements of brain hippocampus volume in relation to mild cognitive impairment and Alzheimer disease: A systematic review and meta-analysis. Medicine (Baltimore). 2023;102(21):e34997. doi:10.1097/MD.0000000000034997
CrossRef - Rawat P, Sehar U, Bisht J, et al. Phosphorylated Tau in Alzheimer’s Disease and Other Tauopathies. Int J Mol Sci. 2022;23(21):12841. doi:10.3390/ijms232112841
CrossRef - Sahukari R, Punabaka J, Bhasha S, et al. Phytochemical profile, free radical scavenging and anti-inflammatory properties of Acalypha indica root extract: Evidence from in vitro and in vivo studies. Molecules. 2021;26(20):6251. doi:10.3390/molecules26206251
CrossRef - Salvadó G, Horie K, Barthélemy NR, et al. Disease staging of Alzheimer’s disease using a CSF-based biomarker model. Nat Aging. 2024;4(5):694-708. doi:10.1038/s43587-024-00599-y
CrossRef - Schindler SE, Galasko D, Pereira AC, et al. Acceptable performance of blood biomarker tests of amyloid pathology — recommendations from the Global CEO Initiative on Alzheimer’s Disease. Nat Rev Neurol. 2024;20(7):426-439. doi:10.1038/s41582-024-00977-5
CrossRef - Schöll M, Verberk IMW, del Campo M, et al. Challenges in the practical implementation of blood biomarkers for Alzheimer’s disease. Lancet Healthy Longev. 2024;5(10):e637-e647. doi:10.1016/j.lanhl.2024.07.013
CrossRef - Strimbu K, Tavel JA. What are biomarkers? Curr Opin HIV AIDS. 2010;5(6):463-466. doi:10.1097/COH.0b013e32833ed177
CrossRef - Suresh S, Singh SA, Rushendran R, et al. Alzheimer’s disease: The role of extrinsic factors in its development, an investigation of the environmental enigma. Front Neurol. 2023;14:1303111. doi:10.3389/fneur.2023. 1303111
CrossRef - Tahami Monfared AA, Stern Y, Doogan S, Irizarry M, Zhang Q. Understanding Barriers Along the Patient Journey in Alzheimer’s Disease Using Social Media Data. Neurol Ther. 2023;12(3):899-918. doi:10.1007/s40120-023-00472-x
CrossRef - Tamagno E, Guglielmotto M, Vasciaveo V, Tabaton M. Oxidative stress and beta amyloid in Alzheimer’s disease. Which comes first: The chicken or the egg? Antioxidants (Basel). 2021;10(9):1479. doi:10.3390/antiox10091479
CrossRef - Windfeld ES, Brooks MS-L. Medical waste management – A review. J Environ Manage. 2015;163:98-108. doi:10.1016/j.jenvman.2015.08.013
CrossRef - Pathak VK, Navneet. Hospital waste management in India: A critical review. Environ Sci Pollut Res Int. 2017;24(1):192-204. doi:10.1007/s11356-016-8102-9
- Chartier Y, Emmanuel J, Pieper U, et al. Safe Management of Wastes from Health-Care Activities. 2nd ed. World Health Organization; 2014.
- Kargar M, Dobaradaran S, Hosseini SMS. A Review on Hospital Waste Management Strategies and Their Environmental Impacts. Iran J Public Health. 2020;49(2):15-24.
- Mmereki BT, Baldwin A, Li B. Waste management in hospitals: A case study of Gaborone private hospitals. Waste Manag Res. 2019;37(1):27-37. doi:10.1177/0734242X18811704.







