Manuscript accepted on :10-10-2025
Published online on: 03-11-2025
Plagiarism Check: Yes
Reviewed by: Dr. Kiruthika Balasubramanian
Second Review by: Dr. Masumeh Saeedi
Final Approval by: Dr. Prabhishek Singh
Nakkala Danamurthy1 and Nallani Venkata Rama Rao2
and Nallani Venkata Rama Rao2
1ANU College of Pharmaceutical Sciences, Acharya Nagarjuna University, Nagarjuna Nagar, Guntur, Andhra Pradesh, India.
2Department of Pharmacology, Chalapathi Institute of Pharmaceutical Sciences, Chalapathi Nagar, LAM, Guntur, Andhra Pradesh, India
Corresponding Author E-mail: danapharma67@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3326
Abstract
The interconnection between oxidative imbalance, sustained inflammation, and thrombotic activity is a critical contributor to the pathophysiology of numerous chronic diseases, including joint disorders, cardiovascular ailments, and metabolic dysfunctions. In light of the growing interest in phytochemicals as safer therapeutic alternatives, the present research explores the bioefficacy of compounds isolated from Euphorbia tirucalli, a plant traditionally used for its medicinal properties. Through systematic chromatographic separation, three key bioactive constituents—eriodictyol, quercitrin, and scopoletin—were obtained from the bark extract.To evaluate their antioxidant activity, both DPPH and hydroxyl radical scavenging assays were conducted. The results demonstrated that all three compounds exerted substantial free radical inhibition in a concentration-dependent manner. Notably, their IC₅₀ values were comparable to that of ascorbic acid, the reference standard, indicating their potent capability to mitigate oxidative stress. These findings suggest that the isolated constituents from E. tirucalli may serve as promising candidates for further investigation in the management of oxidative stress-mediated conditions.Antiarthritic activity was evaluated through two validated models—inhibition of heat-induced protein denaturation and stabilization of human erythrocyte membranes. All three compounds demonstrated marked anti-inflammatory effects, indicating their ability to protect proteins and cellular membranes under inflammatory conditions. In addition, thrombolytic properties were assessed using human blood samples, where the compounds exhibited effective clot lysis, indicating potential fibrinolytic activity. Collectively, these findings provide scientific support for the ethnomedicinal use of Euphorbia tirucalli and highlight the pharmacological relevance of its constituents. The multifunctional bioactivities of eriodictyol, quercitrin, and scopoletin make them promising candidates for further development into natural therapeutic agents targeting oxidative damage, inflammation, and thrombosis.
Keywords
Antiarthritic; Antioxidant activity; Clot lysis; DPPH assay; Euphorbia tirucalli; Eriodictyol; Protein denaturation; Quercitrin; Scopoletin; Thrombolytic activity
Download this article as:| Copy the following to cite this article: Danamurthy N, Rao N. V. R. A Trifecta of Bioactivities: Targeting Oxidative Stress, Inflammation, and Clot Formation with Euphorbia tirucalli Compounds. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Danamurthy N, Rao N. V. R. A Trifecta of Bioactivities: Targeting Oxidative Stress, Inflammation, and Clot Formation with Euphorbia tirucalli Compounds. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4hFwBrn |
Introduction
Chronic inflammatory conditions, oxidative stress-related disorders, and thrombotic diseases constitute significant global health burdens, contributing substantially to morbidity and mortality worldwide. Although these pathologies differ in clinical presentation, they share common underlying mechanisms, including excessive production of reactive oxygen species (ROS), immune dysregulation, and hypercoagulability. Inflammation, for example, is closely linked to the activation of transcription factors such as NF-κB, which drive the overproduction of pro-inflammatory cytokines and degradative enzymes. ¹ Similarly, oxidative stress serves as both a hallmark and a driver of chronic disease, promoting thrombosis through mechanisms such as platelet aggregation and endothelial dysfunction.²
In recent years, the demand for safer and more holistic therapeutic alternatives has reignited interest in plant-derived bioactive compounds. These phytochemicals often exhibit multitarget actions, making them attractive candidates for managing complex disorders with fewer adverse effects than synthetic pharmaceuticals. ³Among traditionally used medicinal plants, Euphorbia tirucalli L. (family: Euphorbiaceae), commonly known as the pencil tree, has gained attention due to its wide range of pharmacological properties. Historically employed in the treatment of asthma, rheumatism, warts, and even malignancies, E. tirucalli is rich in secondary metabolites such as flavonoids, phenolics, terpenoids, tannins, and coumarins.⁴⁻⁵
Phytochemical investigations of E. tirucalli have led to the isolation of several bioactive molecules with therapeutic relevance. Noteworthy among them are quercitrin and eriodictyol—flavonoids with documented antioxidant and anti-inflammatory potential—and scopoletin, a coumarin derivative with notable antiarthritic and vasodilatory effects.⁶ Scopoletin exerts its effects through modulation of oxidative pathways and inhibition of nitric oxide synthase activity.⁷⁻⁸
The pathophysiological role of oxidative stress in disease progression is well established. Excessive production of reactive oxygen and nitrogen species during inflammation, infection, or cellular injury can cause DNA damage, lipid peroxidation, and protein dysfunction.⁹ Concurrently, processes such as inflammation-induced protein denaturation and lysosomal membrane destabilization contribute to the onset and progression of arthritis, underlining the therapeutic value of anti-denaturant and membrane-stabilizing agents.¹⁰
Thrombosis, a consequence of disrupted hemostasis, arises when fibrin deposition and platelet aggregation surpass fibrinolytic activity. Although synthetic thrombolytic agents like streptokinase and alteplase are effective, their clinical application is limited due to high costs and bleeding risks. Natural thrombolytic compounds offer a compelling alternative by facilitating clot dissolution through physiologically balanced mechanisms and potentially exhibiting fewer adverse effects.¹¹
The present study isolates and evaluate the multifunctional biological activities of quercitrin, eriodictyol, and scopoletin from the ethyl acetate fraction of Euphorbia tirucalli. By investigating these diverse therapeutic endpoints, the study provides a comprehensive insight into the pharmacological versatility of E. tirucalli-derived phytochemicals and their potential for development as adjunct therapies in managing oxidative, inflammatory, and thrombosis.
Materials and Methods
Plant Material Collection and Authentication
The fresh bark of Euphorbia tirucalli L. was procured from verified sources in Tirupati. Botanical identification and authentication were performed by Dr. K. Madhava Chetty, Department of Botany, Sri Venkateswara University. A voucher specimen (ET-2025-0829) has been preserved in the institutional herbarium for future reference.¹²
Extraction and Fractionation
The collected bark was washed with distilled water, shade-dried, and coarsely powdered. A total of 500 g of powdered material was subjected to cold maceration with analytical-grade methanol for 72 h, with intermittent shaking. The filtrate was concentrated under reduced pressure at 40 °C using a rotary evaporator to yield the crude methanolic extract. Sequential fractionation was performed using solvents of increasing polarity—ethanol, acetone, acetic acid, and ethyl acetate. The ethyl acetate fraction, exhibiting promising preliminary biological activity, was selected for further isolation of bioactive compounds.
Isolation and Characterization of Phytoconstituents
The ethyl acetate fraction (15 g) was subjected to silica gel column chromatography (60–120 mesh, Merck) under normal-phase conditions. Gradient elution was performed with hexane:ethyl acetate mixtures (from 9:1 to 1:1), followed by 100% ethyl acetate, and finally ethyl acetate:methanol. Three major pooled fractions were obtained based on TLC profiling:
EA1 (10.32 g): hexane:ethyl acetate 8:2
EA2 (2.50 g): hexane:ethyl acetate 1:1
EA3 (2.19 g): 100% ethyl acetate
Preliminary phytochemical screening indicated that EA2 was enriched in phenolics and flavonoids, and it was further purified by repeated column chromatography (hexane:ethyl acetate 1:4). Subfraction EAS2 (356 mg) was subjected to preparative TLC (chloroform:methanol:water 4:0.38:0.02), yielding three pure compounds:
Compound 1 (6.5 mg): eriodictyol
Compound 2 (10.0 mg): quercitrin
Compound 3 (4.3 mg): scopoletin
Structural confirmation was achieved using IR, ¹H-NMR, ¹³C-NMR, and mass spectrometry.¹3
In Vitro Antioxidant Activity
DPPH Radical Scavenging Assay
A 0.1 mM DPPH solution in methanol was mixed with equal volumes (1 mL) of test compounds at 50, 100, 300, and 500 µg/mL. Mixtures were incubated in the dark at room temperature for 30 min. Absorbance was measured at 517 nm, and ascorbic acid served as the positive control.¹⁴ All assays were performed in triplicate (n = 3). Percentage inhibition and IC₅₀ values were calculated.¹⁵
Hydroxyl Radical Scavenging Assay
Hydroxyl radical scavenging was assessed using the deoxyribose degradation assay. Reaction mixtures contained FeCl₃, EDTA, H₂O₂, and 2-deoxyribose in phosphate buffer (pH 7.4). Test compounds were incubated at 37 °C for 1 h. TBA and TCA were added to detect malondialdehyde formation, measured at 532 nm.¹⁶˒¹⁷ Experiments were conducted in triplicate (n = 3), and IC₅₀ values were determined.¹⁸
In Vitro Antiarthritic Activity
Protein denaturation plays a critical role in the pathogenesis of inflammation and joint degradation in arthritic conditions. Inflammatory responses can lead to the loss of native protein conformation, resulting in the formation of autoantigens that further amplify immune activation. 19The protein denaturation assay was conducted based on the method developed by Mizushima and Kobayashi, with slight modifications.²0
In this assay, bovine serum albumin (BSA), a standard protein model, was used to mimic protein denaturation in vitro. A 1% (w/v) BSA solution was prepared in phosphate buffer at pH 6.3. Test compounds at different concentrations (10–100 µg/mL) were incubated with the BSA solution for about 20min . Subsequently, the mixture was subjected to heat-induced denaturation by incubating at 70°C for 5 minutes. Heating causes BSA molecules to unfold and aggregate, which increases turbidity and measured at 660 nm using a spectrophotometer. Standard drug Diclofenac was used. It was calculated using the formula:

In Vitro Antiinflammatory Activity
Stabilization of erythrocyte membranes is a widely accepted indicator of anti-inflammatory activity, as these membranes share structural and functional similarities with lysosomal membranes. Preserving membrane integrity helps prevent the release of lysosomal enzymes, which are key mediators in the inflammatory response. 21-22
Fresh venous blood was collected aseptically from healthy volunteers and anticoagulated. The collected blood was centrifuged at 3000 revolutions per minute for 10 minutes, and the isolated erythrocytes were washed three times using isotonic saline (0.85% NaCl). The erythrocytes were then suspended in isotonic phosphate buffer (pH 7.4) to obtain a 10% (v/v) erythrocyte suspension.
The erythrocyte suspension was incubated with varying concentrations of test compounds under heat-induced hemolysis conditions by exposing the mixture to 54°C for 20 minutes. After incubation, the reaction mixtures were centrifuged, and the absorbance of hemoglobin released into the supernatant was recorded at 560 nm to assess the extent of hemolysis. The percentage of membrane stabilization, or protection against hemolysis, was calculated relative to the control (without test compound).
In Vitro Thrombolytic Activity
Thrombolytic agents are substances that dissolve blood clots (thrombi), and evaluating such activity in vitro is crucial for identifying compounds with potential clinical applications in thrombosis-related diseases. 23 The thrombolytic activity of the isolated compounds was determined by measuring their ability to lyse preformed blood clots.
Fresh venous blood was drawn from healthy human volunteers and transferred into pre-weighed microcentrifuge tubes. The tubes were maintained at 37°C for 45 minutes to promote clotting. After the clots had fully formed and stabilized, the serum was carefully separated, ensuring the clot remained intact, and the clot was then weighed. Test compounds, at a concentration of 100 µg/mL, were added to the tubes containing clots and incubated at 37°C for 90 minutes. Streptokinase, a known thrombolytic enzyme, was used as a positive control, while sterile saline served as a negative control. After incubation, the released fluid was discarded, and the tubes were weighed again. The formula for calculation is
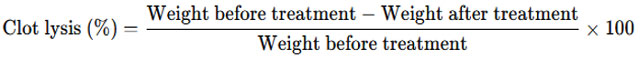
Statistical Processing
All the results are reported as mean ± standard error mean . It was carried out using one-way analysis of variance (ANOVA) and significant group differences were further examined using Tukey’s multiple comparison test by using Graph pad prism software. Results were considered statistically meaningful if the p-value was less than 0.05 (p < 0.05).
Results
Isolation and Structural Elucidation
Three compounds were successfully isolated from the ethyl acetate fraction. Spectral analysis confirmed:
Eriodictyol: Flavanone with characteristic IR bands at 3300 cm⁻¹ (OH), 1600 cm⁻¹ (aromatic C=C), and NMR signals consistent with flavanone skeleton.
Quercitrin: A flavonoid glycoside with sugar moiety signals in NMR and characteristic UV absorption.
Scopoletin: A coumarin with IR bands at 1720 cm⁻¹ (lactone C=O) and NMR signals indicating methoxy and aromatic protons.
 |
Figure 1 : IR spectra of eriodictyol |
 |
Figure 2: 1H-NMR Spectra of eriodictyol |
 |
Figure 3. : Mass spectra of eriodictyol ESI-MS: (M+) m/z 288.35. |
 |
Figure 4 : IR spectra of Quercetin |
 |
Figure 5: 1H-NMR spectra of Quercetin. |
 |
Figure 6: Mass spectra of Quercetin:- ESI-MS: (M+) m/z 302.33 |
 |
Figure 7: IR spectra of Scopoletin |
 |
Figure 8: 1H-NMR spectra of Scopoletin |
 |
Figure 9: Mass Spectra of Scopoletin. |
Antioxidant Activities
Both DPPH and hydroxyl radical scavenging assays demonstrated significant dose-dependent free radical neutralization (Table 1).
Quercitrin exhibited the strongest DPPH radical scavenging (IC₅₀ = 10.1 ± 0.5 µg/mL), followed by eriodictyol (12.5 ± 0.8 µg/mL).
Scopoletin showed moderate activity with an IC₅₀ approximately double that of quercitrin.
Similar trends were observed in hydroxyl radical scavenging assays, though all IC₅₀ values were slightly higher, indicating hydroxyl radicals were more challenging to scavenge.
Table 1: In Vitro Antioxidant Activity of Isolated Compounds Using DPPH and Hydroxyl Radical Scavenging Assays
| Concentration (µg/mL) | Eriodictyol DPPH (%) | Eriodictyol Hydroxyl (%) | Quercitrin DPPH (%) | Quercitrin Hydroxyl (%) | Scopoletin DPPH (%) | Scopoletin Hydroxyl (%) | Ascorbic Acid DPPH (%) | Ascorbic Acid Hydroxyl (%) |
| 50 | 34.6 ± 1.2 | 28.7 ± 1.1 | 40.2 ± 1.4 | 35.1 ± 1.3 | 21.7 ± 0.9 | 18.4 ± 0.7 | 48.3 ± 1.1 | 44.8 ± 1.2 |
| 100 | 52.9 ± 1.6 | 46.4 ± 1.5 | 61.8 ± 1.5 | 54.2 ± 1.2 | 36.5 ± 1.1 | 31.2 ± 1.0 | 67.4 ± 1.3 | 65.5 ± 1.0 |
| 300 | 72.3 ± 1.9 | 68.9 ± 1.6 | 84.6 ± 1.7 | 81.3 ± 1.8 | 58.1 ± 1.5 | 52.6 ± 1.4 | 91.6 ± 1.0 | 90.2 ± 0.9 |
| 500 | 87.8 ± 1.4 | 83.1 ± 1.3 | 92.7 ± 1.2 | 89.7 ± 1.6 | 73.4 ± 1.6 | 70.4 ± 1.3 | 96.3 ± 0.8 | 95.4 ± 0.7 |
| IC₅₀ (µg/mL) | 12.5 ± 0.8 | 18.7 ± 1.2 | 10.1 ± 0.5 | 15.3 ± 1.0 | 22.6 ± 1.1 | 27.8 ± 1.5 | 9.4 ± 0.6 | 9.4 ± 0.6 |
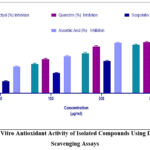 |
Figure 10: In Vitro Antioxidant Activity of Isolated Compounds Using DPPH Radical Scavenging Assays |
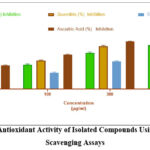 |
Figure 11: In Vitro Antioxidant Activity of Isolated Compounds Using Hydroxyl Radical Scavenging Assays |
Antiarthritic Activity
Protein Denaturation
Quercitrin inhibited protein denaturation up to 72% at 500 µg/mL, significantly higher than eriodictyol and scopoletin (p < 0.01).
Membrane Stabilization
All compounds exhibited protective effects on erythrocyte membranes, reducing hemolysis induced by heat stress. The membrane stabilization was highest for quercitrin (~70%), followed by eriodictyol and scopoletin (Table 2).
Table 2 : In Vitro Antiarthritic Activity of Isolated Compounds
| Test Sample | Concentration (µg/mL) | % Inhibition of Protein Denaturation (Mean ± SEM) | % Membrane Stabilization (Mean ± SEM) |
| Diclofenac (Standard) | 100 | 78.45 ± 1.12 *** | 82.30 ± 1.08 *** |
| Eriodictyol | 50 | 32.10 ± 0.98 * | 29.45 ± 1.05 * |
| 100 | 45.67 ± 1.03 ** | 41.20 ± 0.99 ** | |
| 300 | 61.35 ± 1.25 *** | 57.80 ± 1.15 *** | |
| 500 | 69.22 ± 1.18 *** | 66.15 ± 1.10 *** | |
| Quercitrin | 50 | 35.80 ± 1.05 * | 33.90 ± 0.97 * |
| 100 | 50.10 ± 1.07 ** | 47.45 ± 1.03 ** | |
| 300 | 66.88 ± 1.15 *** | 63.70 ± 1.20 *** | |
| 500 | 73.55 ± 1.22 *** | 70.05 ± 1.12 *** | |
| Scopoletin | 50 | 28.95 ± 1.00 * | 26.30 ± 1.07 * |
| 100 | 41.20 ± 0.92 ** | 38.50 ± 1.09 ** | |
| 300 | 58.75 ± 1.10 *** | 55.20 ± 1.18 *** | |
| 500 | 64.50 ± 1.15 *** | 60.80 ± 1.11 *** | |
| Control (Vehicle) | — | 3.15 ± 0.48 | 2.90 ± 0.52 |
Values are expressed as mean ± SEM (n = 3). Statistical significance compared to control: p < 0.05 (*), p < 0.01 (**), p < 0.001 (***).”
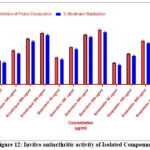 |
Figure 12: Invitro antiarthritic activity of Isolated Compounds |
Thrombolytic Activity
Streptokinase lysed 57% of clots, confirming assay validity.
Eriodictyol and quercitrin exhibited dose-dependent thrombolytic activity (21.69% to 33.87%), significantly higher than saline control (p < 0.01).
Scopoletin showed moderate clot lysis (~25% at 200 µg/mL), indicating fibrinolytic potential (Table 3).
Table 3 : Invitro thrombolytic activity of isolated compounds
| Test Sample | Concentration (µg/mL) | Initial Clot Weight (mg) | Residual Clot Weight (mg) | Clot Lysis (%) |
| Streptokinase | 100 (µL) | 500.0 ± 5.8 | 215.3 ± 4.6 | 57.00 ± 1.21*** |
| Eriodictyol | 100 | 498.0 ± 4.9 | 390.2 ± 6.3 | 21.69 ± 1.04** |
| 200 | 502.3 ± 4.6 | 342.1 ± 5.7 | 31.87 ± 1.15*** | |
| Quercitrin | 100 | 495.2 ± 5.1 | 368.1 ± 4.9 | 25.66 ± 1.10** |
| 200 | 499.7 ± 4.3 | 330.4 ± 4.6 | 33.87 ± 1.33*** | |
| Scopoletin | 100 | 496.0 ± 4.7 | 410.0 ± 5.8 | 17.34 ± 1.20* |
| 200 | 500.1 ± 4.2 | 372.3 ± 6.0 | 25.60 ± 0.98** | |
| Saline (Control) | — | 497.4 ± 5.2 | 480.0 ± 4.9 | 3.42 ± 0.75 |
All values are expressed as mean ± SEM (n = 3). Statistical significance compared to saline control is indicated as *p < 0.05, **p < 0.01, ***p < 0.001.
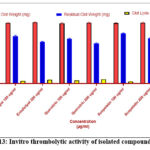 |
Figure 13: Invitro thrombolytic activity of isolated compounds |
Discussion
This study evaluated the in vitro antioxidant, anti-inflammatory (antiarthritic), and thrombolytic properties of three bioactive constituents—eriodictyol, quercitrin, and scopoletin—isolated from the ethyl acetate fraction of Euphorbia tirucalli. These activities are crucial due to their therapeutic implications in oxidative stress-related disorders, inflammatory diseases, and thrombotic conditions.
Antioxidant Activity
Oxidative stress is a key contributor to chronic diseases such as arthritis, cardiovascular disorders, and cancer. ²⁴ In the present study, the DPPH radical scavenging assay revealed quercitrin as the most potent antioxidant, followed by eriodictyol, while scopoletin exhibited moderate activity. These findings align with previous reports attributing strong free radical scavenging to flavonoids, due to multiple hydroxyl groups and conjugated ring systems that stabilize radicals. ²⁵˒²⁶ Hydroxyl radical scavenging assays confirmed a similar trend. The lower activity of scopoletin may be linked to its coumarin core, which lacks structural features enhancing radical scavenging. ²⁷ While these results support the antioxidant potential of these compounds, additional mechanisms—such as metal chelation, modulation of endogenous antioxidant enzymes, or interactions with other cellular targets—may also contribute and warrant further investigation.
Antiarthritic Activity
Arthritis is a chronic inflammatory condition characterized by protein denaturation and membrane destabilization. ²⁸ Quercitrin and eriodictyol demonstrated inhibitory activity comparable to diclofenac sodium. Membrane stabilization assays showed effective protection of erythrocyte membranes from heat-induced hemolysis, ²⁹ consistent with flavonoids and coumarins mitigating inflammation via lysosomal enzyme inhibition and membrane stabilization .30-32 Beyond these classical mechanisms, potential off-target effects, influence on signaling pathways (e.g., NF-κB, MAPK), and synergistic interactions among compounds may further modulate anti-inflammatory outcomes, highlighting the need for mechanistic studies.
Thrombolytic Activity
All three compounds exhibited clot lysis activity, with quercitrin and eriodictyol showing greater efficacy than scopoletin. Although less potent than streptokinase, these natural compounds demonstrated promising fibrinolytic potential, possibly via plasminogen activation or direct fibrin degradation. ³³˒³⁴ Their antioxidant properties may indirectly enhance thrombolytic activity by protecting platelets and coagulation factors from oxidative modification. ³⁵ Only two concentrations were tested in this assay because preliminary experiments showed negligible activity at lower doses and a plateau at higher doses, making two representative concentrations sufficient to demonstrate clot-lysis effects. ²⁶˒³³Additional mechanisms—such as modulation of endothelial signaling, platelet aggregation pathways, or coagulation factor expression—could also contribute to their thrombolytic effects. Future in vivo studies are required to validate these findings, assess bioavailability, metabolic stability, and potential interactions with other pathways.
Pharmacological Implications and Future Prospects
The multifunctional bioactivities of eriodictyol, quercitrin, and scopoletin highlight their potential as lead compounds for drug development. Their antioxidant, anti-inflammatory, and thrombolytic effects suggest a protective role against oxidative stress-induced inflammation and thrombosis. Nevertheless, limitations of this study include its in vitro nature, limited concentration ranges in certain assays, and absence of pharmacokinetic and in vivo data. Elucidating molecular mechanisms, including off-target effects and signaling pathway involvement, will be critical for understanding therapeutic potential. ³⁶ Strategies such as nanoformulation may further enhance bioefficacy, stability, and targeted delivery. ³⁷ Collectively, these results reinforce the ethnomedicinal significance of Euphorbia tirucalli and its isolated constituents, paving the way for their advancement as therapeutic agents.
Conclusion
The isolated phytoconstituents eriodictyol, quercitrin, and scopoletin from Euphorbia tirucalli has exhibited significant Invitro activities. Among these, quercitrin demonstrated the most potent activity across all assays, closely followed by eriodictyol, while scopoletin showed moderate effects. Moreover, their ability to inhibit protein denaturation and stabilize cellular membranes underscores their promising anti-inflammatory potential. The observed thrombolytic activity, though less than the standard drug streptokinase, indicates potential fibrinolytic effects that may contribute to cardiovascular health. Collectively, these findings support the therapeutic relevance of these compounds as natural agents with multifunctional bioactivities. However, further in vivo studies and clinical evaluations are warranted to confirm their efficacy and safety, paving the way for potential development as novel phytopharmaceuticals for managing oxidative stress-related disorders, inflammatory diseases, and thrombotic conditions.
Acknowledgment
The authors gratefully acknowledge the support provided by QIS College of Pharmacy for facilitating this research. Special thanks are extended to the laboratory staff and technical team for their assistance in carrying out the experimental work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval and informed consent was not required.”
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors’ Contribution
Nakkala Danamurthy performed the experimental work, data analysis, and manuscript drafting. Dr. N. Venkata Rama Rao conceptualized the study, supervised the research, and critically revised the manuscript.
References
- Ahmed T, Gilani AH. Inhibitory effect of curcuminoids on lipopolysaccharide-induced nitric oxide production in the murine macrophage cell line RAW 264.7. Biol Pharm Bull. 2009;32(11):1975-81.
- Madamanchi NR, Runge MS. Redox signaling in cardiovascular health and disease. Free Radic Biol Med. 2013;61:473-501.
CrossRef - Atanasov AG, Waltenberger B, Pferschy-Wenzig EM, et al. Discovery and resupply of pharmacologically active plant-derived natural products: a review. Biotechnol Adv. 2015;33(8):1582-614.
CrossRef - Kumar S, Malhotra R, Kumar D. Euphorbia tirucalli: its phytochemistry and pharmacology. Pharmacogn Rev. 2010;4(8):58-61.
CrossRef - Singh A, Duggal S. Euphorbia tirucalli: pharmacognostic review. Int J Res Ayurveda Pharm. 2012;3(1):1-5.
- Formica JV, Regelson W. Review of the biology of quercetin and related bioflavonoids. Food Chem Toxicol. 1995;33(12):1061-80.
CrossRef - Li Y, Yao J, Han C, et al. Quercetin, inflammation and immunity. 2016;8(3):167.
CrossRef - Semwal RB, Semwal DK, Combrinck S, Viljoen A. Health benefits and limitations of coumarins in foods. Crit Rev Food Sci Nutr. 2020;60(22):3829-47.
- Valko M, Leibfritz D, Moncol J, et al. Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol. 2007;39(1):44-84.
CrossRef - Mangasuli SN, Patil MB. Evaluation of anti-arthritic activity of stem bark of Ficus glomerata Roxb. in experimental animals. Indian J Pharm Sci. 2012;74(6):546-50.
- Prasad S, Kashyap RS, Deopujari JY, et al. Effect of Fagonia arabica (Dhamasa) on in vitro thrombolysis. BMC Complement Altern Med. 2007;7:36.
CrossRef - D’Andrea G. Quercetin: a flavonol with multifaceted therapeutic applications? 2015;106:256-71.
CrossRef - Halliwell B, Gutteridge JMC. Free radicals in biology and medicine. 4th ed. Oxford: Oxford University Press; 2007.
- Blois MS. Antioxidant determinations by the use of a stable free radical. 1958;181(4617):1199-200.
CrossRef - Brand-Williams W, Cuvelier ME, Berset C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci Technol. 1995;28(1):25-30.
CrossRef - Halliwell B. Reactive oxygen species and the central nervous system. J Neurochem. 1992;59(5):1609-19.
CrossRef - Halliwell B, Gutteridge JMC, Aruoma OI. The deoxyribose method: a simple test-tube assay for determination of rate constants for reactions of hydroxyl radicals. Anal Biochem. 1987;165(1):215-9.
CrossRef - Rice-Evans CA, Miller NJ, Paganga G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997;2(4):152-9.
CrossRef - Smith MT, Smith RL. Role of protein denaturation in arthritis and inflammation. Clin Immunol Immunopathol. 1987;44(3):345-56.
- Mizushima Y, Kobayashi M. Interaction of anti-inflammatory drugs with serum proteins, especially with some biologically active proteins. J Pharm Pharmacol. 1968;20(3):169-73.
CrossRef - Olajide OA, Ajayi F, Makinde JM. Membrane stabilization and anti-inflammatory activities of plant extracts. J Ethnopharmacol. 1999;68(1-3):267-74.
- Oyedepo OO, Femurewa AJ. Anti-inflammatory activities of extracts from some Nigerian medicinal plants. 1995;66(1):47-50.
- Prasad S, Kalra N, Khanna N, Singh N. Evaluation of thrombolytic activity of some medicinal plants. Int J Pharm Sci Drug Res. 2006;1(2):102-4.
- Halliwell B. Free radicals and antioxidants: updating a personal view. Nutr Rev. 2012;70(5):257-65.
CrossRef - Pietta PG. Flavonoids as antioxidants. J Nat Prod. 2000;63(7):1035-42.
CrossRef - Cushnie TT, Lamb AJ. Recent advances in understanding the antibacterial properties of flavonoids. Int J Antimicrob Agents. 2011;38(2):99-107.
CrossRef - Venugopal R, Aravindhan S. Phytochemical and antioxidant activity of scopoletin isolated from Evolvulus alsinoides. J Nat Remedies. 2013;13(2):90-5.
- Bisht D, et al. Antiarthritic activity of plant extracts: a review. J Pharm Sci Res. 2015;7(7):491-6.
- Dinesh D, et al. Membrane stabilization as a mechanism of anti-inflammatory and anti-arthritic activity of herbal formulations. J Ethnopharmacol. 2013;146(3):889-93.
- Loizzo MR, et al. Antioxidant and anti-inflammatory activities of quercetin derivatives from Hypericum perforatum. J Pharm Pharmacol. 2012;64(7):953-60.
- Gupta S, et al. Anti-inflammatory and antioxidant potential of coumarin derivatives: an overview. J Pharm Res. 2013;6(3):347-55.
- Wyllie SG, et al. Mechanisms of cell membrane protection by flavonoids. Biochem Biophys Res Commun. 2015;467(4):820-5.
- Visioli F, et al. Antioxidant and anti-thrombotic properties of flavonoids: a review. Clin Hemorheol Microcirc. 2000;22(3):235-46.
- Verma SK, et al. Thrombolytic activity of some medicinal plants and their constituents: a review. J Ethnopharmacol. 2017;198:287-95.
- Loke WM, et al. Dietary flavonoids: effects on platelet function and cardiovascular health. Br J Nutr. 2008;99 Suppl 3:S90-100.
- Aggarwal BB, Harikumar KB. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int J Biochem Cell Biol. 2009;41(1):40-59.
CrossRef - Panwar A, et al. Nanoformulation strategies to improve the delivery and therapeutic efficacy of natural compounds. Front Pharmacol. 2020;11:192.
List of Abbreviations
Analysis of Variance (ANOVA)
Bovine Serum Albumin (BSA)
Carbon-13 Nuclear Magnetic Resonance (¹³C-NMR)
Cardiovascular Sciences (CV)
Cyclooxygenase-2 (COX-2)
Drug Discovery (DD)
Euphorbia tirucalli (ET)
Ethylenediaminetetraacetic Acid (EDTA)
Ferric Chloride (FeCl₃)
Half Maximal Inhibitory Concentration (IC₅₀)
Hydrogen Peroxide (H₂O₂)
Immunology and Inflammation (IMM)
Infrared Spectroscopy (IR)
Malondialdehyde (MDA)
Mass Spectrometry (MS)
Molecular and Cell Biology (MCB)
Neuropharmacology (NP)
Nuclear Magnetic Resonance (NMR)
Pharmacology (PHARM)
Proton Nuclear Magnetic Resonance (¹H-NMR)
Reactive Oxygen Species (ROS)
Standard Error of Mean (SEM)
Thiobarbituric Acid (TBA)
Thin-Layer Chromatography (TLC)
Trichloroacetic Acid (TCA)
Translational Biomedical Engineering (TBE)
Tumor Necrosis Factor-Alpha (TNF-α)
2,2-Diphenyl-1-picrylhydrazyl (DPPH)







