Manuscript accepted on :02-09-2025
Published online on: 23-10-2025
Plagiarism Check: Yes
Reviewed by: Dr. Satya Namani
Second Review by: Dr. Thirunahari Sravanthy
Final Approval by: Dr. Anton R Keslav
Asha Khanna1,2 , Ayushi Verma1,3
, Ayushi Verma1,3 , Daya Shankar Gautam1,4*
, Daya Shankar Gautam1,4* , Pradeep Singour5
, Pradeep Singour5 and Ameesha Nigam6
and Ameesha Nigam6
1 AVIKA Biological research Foundation Lab, Jabalpur, India
2 Department of Zoology and Biotechnology, Govt. Science College, Jabalpur, India
3 Department of Biological Sciences, Rani Durgawati Vishwavidyalaya, Jabalpur, India
4 Department of Zoology, St. Aloysius’ College (Autonomous), Jabalpur, India
5 Department of Biotechnology, St. Aloysius’ College (Autonomous), Jabalpur, India
6Department of Biological Sciences and Engineering (BSBE), IIT Kanpur, India
Corresponding Author E.mail : dygautam@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3301
Abstract
Methotrexate (MTX) is frequently used in the treatment of leukemia and several other types of cancer. Its effects are antiproliferative on malignant cells. Withania sominifera (Withania leaf extract: WE) is widely used for its antibacterial, anti-inflammatory & anticancer effects in traditional medicine systems. In this investigation the effects of MTX alone & MTX + WE on the proliferation of normal human peripheral blood mononuclear cells have been studied. The antiproliferative effects were studied by MTT Assay and DNA breakage studies due to MTX were done by florescent Comet Assay. MTX was used in doses of 20µl per well. The concentrations of MTX used in this study were 75, 125 & 250 µg/ml. In some reaction wells 20 µl of WE were also added. The results were recorded after incubation for 4 & 18 hrs. A dose dependent decrease in viability % was found when the cells were treated with MTX alone both at 4 & 18 hrs of incubation but a greater fall in viability at every stage was noted after addition of WE. At 4hrs of incubation the drop-in viability was maximum at 250 µg/ml. The average viability% at this concentration with only MTX was 82.62±4.15 (significant p˂ 0.05). This dropped to 58.13± 4.45 when WE was also added. At 18hrs the average viability was 66.98± 12.35 with only MTX (250 µg/ml), but when WE was also added the viability dropped to 40.96±6.94. The study shows that the antiproliferative effect of MTX was enhanced by Withania sominifera leaf extract.
Keywords
Antiproliferative effect; Comet assay; Methotrexate; MTT assay; Withania somnifera
Download this article as:| Copy the following to cite this article: Khanna A, Verma A, Gautam D. S, Singour P, Nigam A. In Vitro Evaluation of Methotrexate Cytotoxicity and Antiproliferative Effects of Withania somnifera on Human Peripheral Blood Mononuclear Cells Using MTT and Comet Assay. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Khanna A, Verma A, Gautam D. S, Singour P, Nigam A. In Vitro Evaluation of Methotrexate Cytotoxicity and Antiproliferative Effects of Withania somnifera on Human Peripheral Blood Mononuclear Cells Using MTT and Comet Assay. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/3Jq4rE0 |
Introduction
Methotrexate (MTX) earlier known as Amethopterin is a metabolic inhibitor commonly used as an anti-cancer or antineoplastic drug. It acts as an immunosuppressant for various autoimmune diseases. MTX mainly consists of Aminopterin as its parent component which was first drug that was used for the successful treatment of leukaemia. It is used as a treatment for many types of cancer – lung cancer, breast cancer, myeloma, lymphoma etc. In the treatment of cancer Methotrexate acts as the antifolate metabolic inhibitor.
Withania somnifera (Ashwagandha) is an evergreen shrub belonging to the family Solanaceae that grows in India, the middle east and parts of Africa. It is a short shrub which grows to a height of 35-75 cm. The leaves are dark green & flowers are small, not brightly coloured and the ripe fruit is small & red. The name Somnifera indicates that it is sleep inducing and in fact calms the mind. It is used in herbal medicine and dietary supplements to treat various diseases and health conditions.1 The word ‘Ashwagandha’ refers to a horse like smell the source of which is the root. The main phytochemical constituents of the plant extract are withanolides which are a group of triterpene lactones. Its extracts have been found to have anti-inflammatory, anti-bacterial, and anti-tumor activities also. Role of Withania somnifera in the management of male infertility was investigated by Pallav Sengupta.2 Withanolidses isolated from the plant also inhibited the growth of cancerous cells, breast and colon cell lines.3
Hence it was thought that the study of the effect of Withania sominifera leaf extract on the human mononuclear cells from peripheral blood which were pre-treated with various concentrations of methotrexate at 4 and 18 hrs incubation would yield interesting results.
Materials and Methods
The research work involves the use of mononuclear cells isolated from Human peripheral blood of a pre-informed healthy normal blood donor. The present investigation has been approved by the institutional ethical committee of AVIKA biological research foundation lab, Jabalpur, Madhya Pradesh (where research work was carried out). For the research work Methotrexate injection IP 25 mg/ml (trade name- Folitrax-25), Ipca laboratories ltd., Mumbai was purchased from a local pharmacy shop. This was the drug used to treat the cells. The injection was treated as the stock solution and dilutions were made in sterile distilled water to yield solutions containing 75, 125 & 250 µg/ml of the drug respectively.
Preparation of Withania sominifera Leaf extract
Plant leaves were cleaned, dried and ground to a fine powder. Then 2.9 g of dried leaf powder along with ethanol and water in 1:1 ratio was subjected to extraction process in Soxhlet for 10hrs at 80ᴼC. This extract was filtered with Whatman filter paper and the filtrate was evaporated in hot air oven at 55ᴼC. Then stock solution of this was prepared @ 25mg/ml.4
Isolation of Lymphocytes from whole blood
In a sterile heparinised vial, 3.5ml of blood was collected from a healthy male young volunteer donor after informed consent. This was diluted to 7ml with PBS (1X). 3.5ml HisepTM Lymphocyte separation medium (HiMedia, LSM) was transferred aseptically into a centrifuge tube. This was then gently layered with 7ml of the diluted blood. It was centrifuged for 30 minutes at 400g at room temperature (RT). The erythrocytes were sedimented at the bottom and lymphocytes formed a layer above the Hisep layer. The lymphocyte layer along with half of Hisep layer was carefully pipetted out into a separate centrifuge tube after the supernatant was removed. After that, it was washed twice with isotonic PBS by adding 5ml PBS 1X to the WBC thus separated & centrifuging at 2300 RPM for 10 min. One more similar wash was given & 500µl of the suspension was used to count the cells in Hemocytometer in which 28.5 × 104 cells/ml or 2.85 × 105 cells/ml of cells were observed. The cells were appropriately diluted with TC 199 Medium (HiMedia) supplemented with FBS 1% (Fetal Bovine serum ) and PHA (Phytohemaglutinin) 15µl to achieve the final concentration of 5698 cells/well if 180 µl of cells+medium suspension was used to fill each well.
MTT Assay
Two 96 well plates of ELISA microplate reader (Lisaquant-TS, Tulip Diagnostics) were loaded (one for 4 hrs incubation and the other for 18 hrs of Incubation). First row was kept empty and the loading started from row B which contained only 180 µl cells + medium in 9 wells (forming 3 sets of triplicates of the control cells). The 3 subsequent rows were similarly loaded with 180 µl cells + medium + 20 µl of the 3 drug concentrations taken up for the experiment (D1, D2, D3 representing 75, 125 & 250 µg/ml of MTX). The next 3 rows, the cells + medium +10 µl of the drug conentration + 10µl of WE was loaded in the manner described above. 3 replicates of 20µl of each of the 3 concentrations of the drug and WE were loaded in separate cells .5,6
Prior to loading, the reference reading of the plates was taken at 630nm. The OD reading of the drug only wells was taken the momemt they were loaded. The plate was then incubated for 4hrs at 37ᴼC. Similar pattern of loading was followed for 18hrs of incubation. After 4 hrs 20µl of MTT (0.25 mg/ml) was added to the first plate & put for incubation for further 2hrs. After that 100µl of DMSO was added to each well and again incubated for 1 hour. OD readings were taken at 550nm. Similar procedure was followed for plate for 18hrs of incubation.
Calculations were done after making appropriate deductions of the readings of the empty plate at 630 nm, the drug and WE. Viability and inhibition was calculated according to the following formulas
Viability% = OD of the sample / OD of the control × 100
Inhibition% = 1- (OD of the sample / OD of the control) × 100
COMET Assay
Comet Assay is a method used to detect DNA damage at the level of individual cells and it is valuable to assess the damage to the genetic material due to toxic chemicals and pharmaceuticals. This test was optimised for alkaline conditions7 and it helps to assess single or double stranded DNA breaks at alkali labile cells.
Precoating of slides: Slides were thoroughly clean and dried. They were then uniformly coated with a 1% solution of NMPA (Normal melting point agarose) in PBS, and dried at 37ᴼC overnight. The slide should be perfectly dried and stored in a slide box whose lid is sealed with parafilm.8
Solution of 0.5% LMPA (low melting point agarose) in PBS was also prepared and kept at 4ᴼC.
Before performing the experiment, the blood was subjected to 3 concentrations os MTX in eppendorf tubes each containing 100µl of blood and 50 µl each of 75,125 & 250 µg/ml concentrations of MTX for 1hrs 30mins.
This treated blood + LMPA (maintained at 37ᴼC) was loaded on the NMPA coated slides (30 µl of blood + 90 µl of LMPA), out of which 50 µl was added on slide. Coverslip was placed and the slides were put in the freezer for 5minutes. Coverslip was then removed gently and the slide was then put in a Petri dish in cold lysis (working solution) for 90 minutes.
They were then put in cold electrophoresis buffer and the slides were made to lie in the buffer for 20 minutes in the electrophoresis tank.
After this the power was run for 20-25 minutes.
Neutralisation: The slides were gently lifted out of the tank and flooded with neutralisation buffer for 5 minutes. This was repeated 3 times. The slides were then dried and stored.
Staining: The slides were hydrated in cold distilled water for 7 minutes & stained in 1µM Ethidium bromide for 15 minutes.
They were observed in the florescence microscope in green excitation filter and red emission filter where the cells appeared red on a black background.
Results
MTT Assay
After 4hrs of incubation with MTX only
If the OD (readings after appropriate deductions and calculations) of the control were taken to be 100%, the addition of 20µl of each of the 3 concentrations of MTX only taken for study (75, 125 & 250 µg/ml) resulted in a dose dependent decrease in viability because at 250µg/ml it dropped to 82.6% which was statistically significant (p˂ 0.05).
Fig 1 shows that there was dose dependent decrease in viability with MTX only at 4hrs. If 20µl of Withania somnifera (WE) leaf extract is also added to the reaction mixture in the wells, the viability drops till further. In this case the controls showed a viability of 82.38% and there was a drop in viability at every concentration of MTX if tested with WE, reaching the lowest level of 58.13% at 250 µg/ml but the decrease was non-significant. These viability values were lower than those found with only MTX. Hence it maybe said that this was due to the antiproliferative effect of WE.
At 18 hrs incubation with only MTX, more dose dependent reduction in viability was observed at every concentration. The addition of WE in this case resulted in a further decrease in viability values. The drops with 125 and 250µg/ml were found to be statistically significant. Thus, the antiproliferative effect of WE was more evident at 18hrs of incubation.
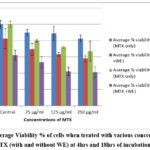 |
Figure 1: Average Viability % of cells when treated with various concentrations of MTX (with and without WE) at 4 hrs and 18hrs of incubation. |
Antiproliferative/inhibitory effect of MTX and MTX+WE
At 4 hrs incubation the results showed that 75 µg/ml of MTX if used alone gave inhibition of 10.15%, but if used with WE it showed 13.17%. Similarly, at 125 µg/ml MTX alone depressed cell viability by 11.28% but the inhibitory effect was found to be 19.24% with WE. At 250 µg/ml of MTX alone the inhibition was only 17.38% but with WE it was 24.15%.
At 18hrs of incubation it was found that MTX only caused more inhibition to cell viability than observed at 4 hrs and with the addition of WE still more inhibition of viability was evident.
COMET Assay
The comets were scored in more than 150 cells/ treatment and they were classified into 5 categories9 – 0, 1, 2, 3 & 4. According to this system TCS (total comet score ) is calculated as-
TCS= 0(n)+ 1(n)+ 2(n)+ 3(n)+ 4(n)
where n indicates the number of cells in each class. 10
The TCS of control cells thus calculated was found to be 69.3% and the TCS values showed a dose dependent increase with the treatment of the cells with 75, 125 & 250 µg/ml of MTX. At 75 µg/ml the TCS value was found to be 120.66% which is considerably higher than the control value and indicates the DNA damage caused by the drug. At 125 µg/ml, the TCS value obtained was 213.66% and 250 µg/ml it raised to 259.43%.
At 125 µg/ml comets of the category 2 & 3 showed the maximum occurrence and only 9 cells were in the category 4. At 250 µg/ml 89 cells showed category 4 comets which was the maximum score of this class. Comets of class 1 showed the maximum occurrence at 75 µg/ml and at 125 µg/ml maximum number of comets were of category 2 & 3 as shown in Table 1 and Fig 2.
Table 1: Mean frequency of each comet class per 150 cells (± Standard deviation) & overall total cell count % of lymphocytes exposed to different concentrations of MTX .
| Comet class | ||||||
| Concentration of MTX | 0 | 1 | 2 | 3 | 4 | TCS (in %) |
| Control | 87 ± 5.81 | 36 ± 4.12 | 16 ± 1.63 | 12 ± 2.25 | 0 | 69.3 |
| 75 µg/ml | 90 ± 5.88 | 82 ± 3.70 | 61 ± 2.39 | 29 ± 3.10 | 9 ± 1.25 | 120.66 |
| 125 µg/ml | 6 ± 1.0 | 37 ± 6.11 | 56 ± 6.48 | 53 ± 3.01 | 9 ± 0.83 | 213.66 |
| 250 µg/ml | 2 ± 0 | 64 ± 2.72 | 69 ± 3.87 | 57 ± 2.09 | 89 ± 3.09 | 259.43 |
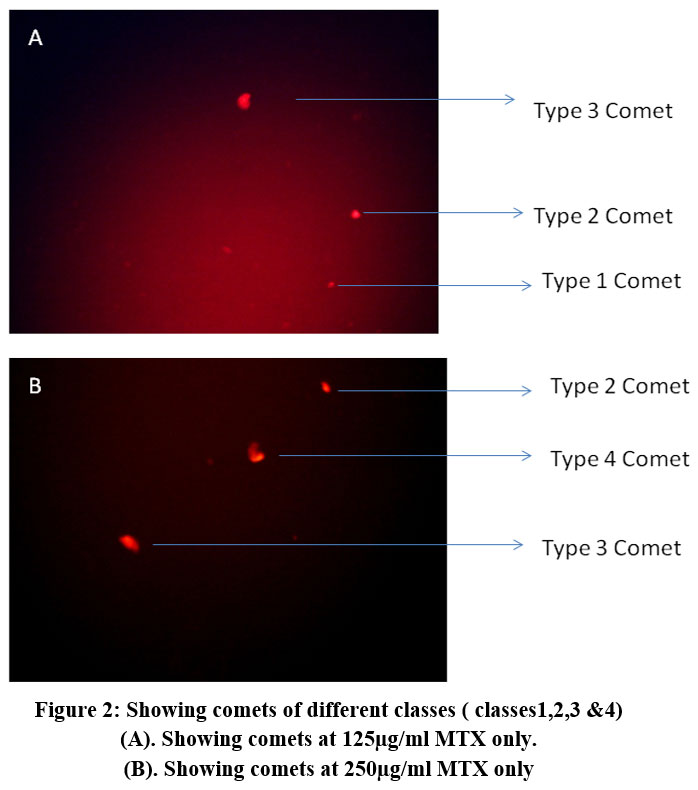 |
Figure 2: Showing comets of different classes ( classes1,2,3 &4). |
Discussion
Barani M.11 et al, 2021 worked on the effect of MTX on normal and malignant cells and found that it causes dose dependent reduction in viability of cancerous and non-cancerous cells, and the toxic doses were found to be from 0-320 µg/ml (in-vivo and in vitro in rats).
Barbisan F.12 et al, (2014) worked on the possibility that the MTX related response on human peripheral blood mononuclear cells may be modulated by Ala16 Val-SOD2 gene polymorphism. They found no appreciable decrease in viability at 10 & 100 µM of MTX treated cells showing a SOD frequency of 27.6%. Some reduction in viability was found at both concentration in AV cells (SOD frequency 48.7%).
Invitro cytotoxic studies on antineoplastic drugs on human lymphocytes was carried out by Vanshi Krishna et al (2009). 13 100µl of lymphocytes containing approximately 50000 cells were incubated with different concentrations of various anticancer drugs (50µl) for 2 hrs and the effects were judged by MTT Assay. The lowest viability in MTX was found to be 32.01% with 400µM of MTX.
Probably the reason for reduction in cell viability caused by MTX treatment is that it is a competitive inhibitor of dihydrofolate reductase. As tetrahydrofolate is necessary for de novo synthesis of DNA, it hinders the proliferation of lymphocytes used in the experiment.
The comet assay findings also show a dose dependent increase in DNA damage as evident by class 3 & class 4 comets when the cells are treated with MTX.
Conclusion
In the present investigation it was found that MTX with WE caused a dose dependent decrease in viability of the lymphocytes which was found to be more at 18 hrs of incubation. This confirmed the antiproliferative effect of Withania on normal human lymphocytes. The DNA damage caused by MTX was evidenced by the findings of the Comet assay studies.
Acknowledgment
The work was done in AVIKA Biological Research Foundation Lab, Jabalpur, (M.P.). The authors are grateful to the AVIKA Society for providing research facilities.
Funding Source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Asha Khanna: Conceptualization, Methodology, Performing the experiments and writing.
- Ayushi Verma: Donation of the blood sample, performing the experiments and calculations.
- Daya Shankar Gautam: Conceptualization, Correspondence, editing.
- Pradeep Singour: Donation of the blood sample, performing the experiments and
- Ameesha Nigam: Performing the experiments and calculations.
References
- Mikulska P, Malinowska M, Ignacyk M, et al. Ashwagandha (Withania somnifera)—Current research on the health-promoting activities: A narrative review. 2023;15(4):1057. doi:10.3390/pharmaceutics15041057
CrossRef - Sengupta P, Agarwal A, Pogrebetskaya M, Roychoudhury S, Durairajanayagam D, Henkel R. Role of Withania somnifera (Ashwagandha) in the management of male infertility. Reprod Biomed Online. 2018;36(3):311-326. doi:10.1016/j.rbmo.2017.11.007
CrossRef - Davis L, Kuttan G. Effect of Withania somnifera on cell mediated immune responses in mice. J Exp Clin Cancer Res. 2002;21(4):585-590.
- Chinembiri TN, Gerber M, du Plessis LH, du Preez JL, Hamman JH, du Plessis J. Topical delivery of Withania somnifera crude extracts in niosomes and solid lipid nanoparticles. Pharmacogn Mag. 2017;13(Suppl 3):S663-S671. doi:10.4103/pm.pm_489_16
CrossRef - Khanna A, Patel J, Tiwari B, Pagare S, Gautam DS, Ratoniya V. Cytotoxic effect of cypermethrin and neem extract on human lymphocytes. Biomed Pharmacol J. 2022;15(1). doi:10.13005/bpj/2393
CrossRef - Mosmann T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65(1-2):55-63. doi:10.1016/0022-1759(83)90303-4
CrossRef - Singh NP, McCoy MT, Tice RR, Schneider EL. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res. 1988;175(1):184-191. doi:10.1016/0014-4827(88)90265-0
CrossRef - Nandhakumar S, Parasuraman S, Shanmugam MM, Rao KR, Chand P, Bhat BV. Evaluation of DNA damage using single-cell gel electrophoresis (Comet Assay). J Pharmacol Pharmacother. 2011;2(2):107-111. doi:10.4103/0976-500X.81903
CrossRef - Khisroon M, Gul A, Khan A, et al. Comet assay based DNA evaluation of fuel filling stations and automobile workshops workers from Khyber Pakhtunkhwa province, Pakistan. J Occup Med Toxicol. 2015;10:27. doi:10.1186/s12995-015-0069-2
CrossRef - Collins AR. The comet assay for DNA damage and repair: Principles, applications, and limitations. Mol Biotechnol. 2004;26(3):249-261. doi:10.1385/MB:26:3:249
CrossRef - Barani M, Hajinezhad MR, Sargazi S, et al. Simulation, in vitro, and in vivo cytotoxicity assessments of methotrexate-loaded pH-responsive nanocarriers. Polymers (Basel). 2021;13(18):3153. doi:10.3390/polym13183153
CrossRef - Barbisan F, Motta JdeR, Trott A, et al. Methotrexate-related response on human peripheral blood mononuclear cells may be modulated by the Ala16Val-SOD2 gene polymorphism. PLoS One. 2014;9(10):e107299. doi:10.1371/journal.pone.0107299
CrossRef - Krishna IV, Vanaja GR, Kumar NS, Suman G. Cytotoxic studies of anti-neoplastic drugs on human lymphocytes—in vitro studies. Cancer Biomark. 2009;5(6):261-272. doi:10.3233/CBM-2009-0111
CrossRef
Abbreviation List:
MTX – Methotrexate,
WE – Withania sominifera leaf extract.







