Manuscript accepted on :08-10-2025
Published online on: 22-10-2025
Plagiarism Check: Yes
Reviewed by: Dr. Gowri Burle and Dr. Praveen Kumar S E
Second Review by: Dr. Tagreed Altaei
Final Approval by: Dr. Mariia Shanaida
Rania Indu* , Hirak Bhowmik
, Hirak Bhowmik , Pritha Sarkar
, Pritha Sarkar , Trika Chatterjee
, Trika Chatterjee , Moumita Ray
, Moumita Ray and Dibya Das
and Dibya Das
Department of Pharmacy, JIS University, Nilgunj Rd, Jagarata Pally, Deshpriya Nagar, Agarpara, Kolkata, West Bengal, India
Corresponding Author E-mail:drraniaindu@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3307
Abstract
Dyslipidemia is associated with an increased risk of atherosclerotic cardiovascular disease, while conventional lipid-lowering drugs may be inadequate or poorly tolerated. Banana (Musa paradisiaca) peel, an abundant agri-food waste rich in antioxidant phytochemicals, represents a sustainable and low-cost candidate for managing lipid disorders. Previous studies in other parts of the world highlighted the antidiabetic and hypolipidemic potential of kepok banana peel extract. Therefore, the present study aimed to investigate the antioxidant, antimicrobial, and antihyperlipidemic activities of hydroalcoholic extract prepared from banana peels (BPE) collected from West Bengal, India. Phytochemical screening was performed, and antioxidant capacity was evaluated using in vitro DPPH and reducing power assays. Antibacterial activity against E. coli was assessed by agar well diffusion. In vivo antihyperlipidemic efficacy was screened in rats subjected to a high-fat diet and treated with BPE (200 or 400 mg/Kg), Atorvastatin (10 mg/Kg) for 28 days prior to biochemical assessment, and compared with the untreated control group. Phytochemical screening suggested the presence of several biologically active phytoconstituents, such as saponins, phytosterols, flavonoids, carbohydrates, glycosides, alkaloids, phenols, and terpenoids. BPE scavenged DPPH radicals with an IC₅₀ of 88.11 ± 6.23 µg/mL and showed concentration-dependent reducing power. The prominent zone of inhibition (2.45cm at 1000µg/mL) against E. coli confirmed its antimicrobial properties. In hyperlipidemic rats, BPE significantly limited weight gain and normalized cholesterol, triglycerides, and LDL, while restoring HDL (p<0.01). Overall, BPE demonstrated strong antioxidant, antibacterial, and lipid-lowering effects, supporting its potential as a cost-effective, eco-friendly nutraceutical for managing oxidative stress and dyslipidemia, warranting further clinical investigation.
Keywords
Antihyperlipidemic Effect; Antimicrobial Activity; Antioxidant Activity; Banana Peel Extract; High-Fat Diet; In vitro; In vivo; Musa paradisiaca
Download this article as:| Copy the following to cite this article: Indu R, Bhowmik H, Sarkar P, Chatterjee T, Ray M, Das D. Phytochemical Screening and Evaluation of the Antioxidant, Antimicrobial and Antihyperlipidemic Activities of the Hydroalcoholic Extract from the Peel of Musa paradisiaca. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Indu R, Bhowmik H, Sarkar P, Chatterjee T, Ray M, Das D. Phytochemical Screening and Evaluation of the Antioxidant, Antimicrobial and Antihyperlipidemic Activities of the Hydroalcoholic Extract from the Peel of Musa paradisiaca. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/3JkRpaV |
Introduction
Dyslipidemia is a multifactorial condition that not only contributes to but also exacerbates the risk of cardiovascular disease. Epidemiological and clinical research have consistently demonstrated that increased levels of total cholesterol, low-density lipoprotein (LDL), and triglycerides are significant risk factors for the onset of atherosclerotic cardiovascular disease.1,2 Lipid profiles specifically the cholesterol levels are significantly rising in India, with high rates seen in 25–30% of urban and 15–20% of rural populations. Though lower than in high-income countries, individuals with familial hypercholesterolemia face a much higher risk of early cardiovascular disease, highlighting the need for early diagnosis and treatment.3,4 While several lipid-lowering drugs exist, they may be ineffective for some and can cause side effects. Traditional herbal medicine offers a complementary approach with lesser side-effects and better patient compliance including its anti-inflammatory and antioxidant benefits.5
Agri-food waste is a major contributor to environmental stress, but its reduction can lessen this impact on environmental pollution while revealing valuable resources. Increasing interest in its therapeutic potential has spurred efforts to explore innovative and sustainable applications.6 Banana, widely cultivated in tropical regions, is valued for its nutritional and therapeutic benefits. Traditionally, banana peel has been used possess a wide range of therapeutic properties, including antimicrobial, anti-ulcer, anti-hyperglycemic, antihypertensive, wound healing, and various other activities.7 Previous studies report that alcoholic extracts of M. paradisiaca peels are rich in antioxidant phenolics and flavonoids. Fresh yellow banana peel extract also shows strong antibacterial and anti-allergic properties, effective against both Gram-positive and Gram-negative bacteria.8 Another study demonstrated that hydroethanolic extracts of Musa paradisiaca leaf and peel possess antihyperlipidemic and cardioprotective effects in streptozotocin–nicotinamide-induced diabetic rats.9
Literature studies have emphasized the significant therapeutic impact of phytochemicals, bioactive compounds in plants. Antioxidant activity of phytochemicals is primarily attributed to polyphenols, flavonoids, and carotenoids, which neutralize reactive oxygen species (ROS) by donating electrons or hydrogen atoms, thereby preventing oxidative damage to lipids, proteins, and DNA. Moreover, many phytochemicals exhibit antimicrobial properties by disrupting microbial cell walls and membranes, inhibiting essential enzymes, or interfering with nucleic acid synthesis, ultimately suppressing the growth of pathogenic bacteria and fungi.10 Furthermore, phytochemicals exert regulatory effects on cholesterol metabolism by modulating pathways involved in synthesis, transport, and bile acid metabolism, and they may also beneficially influence gut microbiota composition to support cholesterol homeostasis.11 Previous studies have reported the presence of a number of phytochemicals in banana peel extract. Therefore, it is essential to explore the biological activities of Musa paradisiaca peel from the West Bengal region, which remain underexplored to date. This study thus aims to assess its in vitro antioxidant and antimicrobial properties, along with its in vivo antihyperlipidemic potential in high-fat diet-induced models, to support its therapeutic application against oxidative stress, infections, and lipid disorders.
Materials and Methods
Materials
All chemicals and reagents used in the study, including ethanol, Molisch’s reagent, Wagner’s reagent, Dragendorff’s reagent, ferric chloride, lead acetate, glacial acetic acid, sulphuric acid, Folin-Ciocalteu reagent, sodium carbonate, gallic acid, aluminium chloride, sodium hydroxide, quercetin, DPPH, sodium phosphate buffer, potassium ferricyanide, phosphate-buffered saline, sodium bicarbonate, were of analytical grade and were procured from Lobachemie and Merck Pvt. Ltd., Mumbai. Atorvastatin was obtained from Cipla Ltd., India. The biochemical diagnostic kits used for estimation of serum lipid profile parameters, including Total Cholesterol, Triglycerides, HDL, and LDL, were purchased from ARKRAY Healthcare Pvt. Ltd., India.
Collection and Identification
Ripe bananas were collected from the local markets of Kolkata and peels were obtained. The peels were verified by the Botanical Survey of India, Howrah, West Bengal (JISU/M/01&02).
Extraction and Preparation of Banana Peel Extract (BPE)
Ripe banana peels were separated, dried, and pulverized into a coarse powder. After macerating 50 grams of the powder in 70% ethanol for 72 hours, the mixture was filtered using Whatman filter paper. A rotary evaporator was used for solvent evaporation to concentrate the filtrate. The resulting residue was collected as Banana Peel Extract (BPE) and stored at 4°C for further use.
Phytochemical screening:
The preliminary phytochemical screening of the BPE (1mg/mL) was conducted to identify the primary phytoconstituents of the extract following standard protocols.
Test for tannins
Ferric chloride test- Five mL of BPE extract solution was mixed with 1 mL of 0.1% ferric chloride solution and the bluish-black or greenish-black coloration of the solution indicated a positive test for tannin.12
Test for saponins
Foam test- About 0.5 mL of the extract was mixed with 5 mL of distilled water and agitated. Thereafter, the presence of saponins was confirmed by the formation of foam.12
Test for flavonoids
Alkaline reagent test- One milliliter of the stock solution was transferred into a test tube, followed by the addition of a few drops of 1% dilute sodium hydroxide solution. The development of a yellow coloration confirmed the presence of flavonoids.12
Test for carbohydrates
Molisch’s test- Two mL of extract solution was mixed with 2 mL of concentrated sulfuric acid and treated with Molisch’s reagent. The formation of a reddish-violet ring at the interface of the two layers indicated a positive result for non-reducing sugars.13
Fehling’s test- Five milliliter of the BPE solution was mixed with an equal volume of Fehling’s solution (A and B) and boiled for 2–5 minutes. The formation of a brick-red precipitate indicated the presence of reducing sugars.13
Test for glycosides
Keller Kiliani test- The presence of glycosides was confirmed by dissolving the plant extract in acetic acid with a trace of ferric chloride and layering it over concentrated sulfuric acid. A reddish-brown color at the junction, gradually turning blue, indicated a positive result.14
Test for alkaloids:
Mayer’s test- Two milliliter of the ripe extract was mixed with 2 mL of Mayer’s reagent. The formation of a cream-colored precipitate confirmed the presence of alkaloids.15
Dragendorff’s test- Two milliliters of BPE was mixed with 2 mL of Dragendorff’s reagent. The presence of alkaloids was affirmed by the formation of an orange-red precipitate.15
Wagner’s test- Two milliliter of BPE was taken in a test tube, followed by the addition of a few drops of Wagner’s reagent. The presence of alkaloids could be confirmed by the formation of a brown or reddish precipitate.16
Hager’s test- Two milliliter of the extract was transferred into a test tube, followed by the addition of a few drops of Hager’s reagent. The formation of a yellowish or white precipitate indicated the presence of alkaloids.17
Test for phytosterols
Salkowaski test- Two milliliter of the extract was mixed with 5 mL of chloroform in a test tube and then filtered. To the resulting filtrate, a few drops of concentrated sulfuric acid were carefully added. After shaking and allowing the mixture to stand, the appearance of a golden-yellow color signified the presence of phytosterols.13
Test for phenol
Ferric chloride test- The presence of phenol in the extracts was confirmed with the formation of bluish-black color upon adding a few drops of ferric chloride (1.0 %) to 2 mL of the extract.16
Test for terpenoids
Salkowski test- Two milliliter of BPE was combined with 2 mL of chloroform in a test tube, after which 3 mL of sulfuric acid was carefully added. The appearance of a reddish-brown layer at the interface indicated the presence of terpenoids.14
Total Flavonoid content
The total flavonoid content of BPE was determined by the aluminum chloride colorimetric method. A 3 mL BPE solution (1 mg/mL) was mixed with 0.15 mL of 5% sodium nitrite. After 5 minutes, 0.3 mL of 10% aluminum chloride was added, followed 6 minutes later by 1 mL of 1 M sodium hydroxide. The mixture was diluted with 1.5 mL distilled water, kept in the dark for 30 minutes, and the absorbance was recorded at 510 nm. Quercetin served as the standard for calibration and quantification.18
Total phenolic content
The total phenolic content of BPE was assessed using the Folin–Ciocalteu colorimetric method with minor modifications. A 0.5 mL BPE aliquot (1 mg/mL) was mixed with 0.75 mL of Folin–Ciocalteu reagent. After 5 minutes, 0.4 mL of saturated sodium carbonate (75 g/L) was added, and the mixture was incubated in the dark at room temperature for 2 hours. Absorbance was recorded at 760 nm using a UV–visible spectrophotometer. Gallic acid was used to generate the standard calibration curve, and the results were expressed as mg gallic acid equivalent per gram of extract (mg GAE/g), reported as mean ± SEM from triplicates.18
In vitro Antioxidant activity
DPPH radical scavenging activity
The antioxidant activity of BPE was evaluated using the 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical scavenging assay. Different concentrations of the extract were prepared in distilled water, and a 0.002% w/v DPPH solution was made in methanol. For the assay, 150 μL of each extract concentration was mixed with 150 μL of DPPH solution, while control samples (without DPPH) were used for color correction. The mixtures were incubated in the dark at room temperature for 30 minutes, and absorbance was recorded at 518 nm. All measurements were performed in triplicate. The percentage inhibition was calculated relative to the control, and the IC₅₀ value was determined.19
The percentage inhibition was calculated using the following formula:
Percentage inhibition =[{absorbance of control – (absorbance of sample – colour factor)}/ (absorbance of control)]*100
Reducing power assay
The reducing power of the extract was assessed using the Fe³⁺–Fe²⁺ transformation method. Different concentrations of the sample (1 mL) were mixed with 2.5 mL of 0.2 M phosphate buffer (pH 6.6) and 2.5 mL of 1% potassium ferricyanide, then incubated at 50 °C for 30 minutes. After incubation, 2.5 mL of 10% trichloroacetic acid was added, and the mixture was centrifuged at 3000 g for 10 minutes. The resulting supernatant (2.5 mL) was combined with distilled water and ferric chloride, and the absorbance was recorded at 700 nm.19
Antimicrobial property
Agar well diffusion method
The antimicrobial activity of BPE was screened by the agar well diffusion method. Nutrient agar plates were prepared and inoculated with Escherichia coli suspensions. Wells were then cut by a sterile cork bore and 200 µL of BPE were placed into the wells. Following a 24-hour incubation at 37°C, the zone of inhibition was recorded. Ceftriaxone (125 µg/mL) was used as the standard.20
In Vivo Antihyperlipidemic Study
Animal
The present study was conducted and reported in compliance with the ARRIVE (Animal Research: Reporting of In Vivo Experiments) 2.0 guidelines.21 All experimental procedures were approved by the Institutional Animal Ethics Committee of TAAB Biostudy Services (1938/PO/Rc/S/17/CPCSEA). Healthy Wistar albino rats of either sex, weighing 150–200 g, were housed and cared for in compliance with the guidelines of the Committee for Control and Supervision of Experiments on Animals (CCSEA). The rats were housed under 25 ± 2 °C temperature, 12 h light/dark cycle, and 45 to 55% relative humidity with feed and water ad libitum. The animals were allocated randomly to the different study groups, and outcome assessments were conducted in a blinded manner where feasible. Prior in vivo antihyperlipidemic studies guided the number of animals per group, optimized to ensure statistical validity. All measures were followed to minimize animal suffering.
Acute toxicity study
Acute oral toxicity of BPE was conducted following OECD 423 guidelines. The animals were observed closely for the first 30 minutes, periodically during the first 24 hours, and daily for 14 days for mortality and clinical signs, thereafter up to one month.22
Induction of hyperlipidemia
Rats in the normal group received a standard diet providing 3.8 kcal/g, while hyperlipidemia was induced in the experimental group by feeding a high-fat diet (HFD) with an energy value of 5.24 kcal/g for 28 days.23,24
Study design and group division
Thirty rats were selected randomly and divided into 5 groups, as follows:
| Group details | No. of Animals | Induction | Treatment |
| Group I(Normal Control) | 6 | NA* | NA |
| Group II(HFD Control) | 6 | HFD | NA |
| Group III (Standard) | 6 | HFD | AtorvastatinDose- 10mg/Kg bodyweight (bw) orally for consecutive 28 days |
| Group IV(BPE200) | 6 | HFD | BPEDose- 200mg/Kg bw orally for consecutive 28 days |
| Group V(BPE400) | 6 | HFD | BPEDose- 400mg/Kg bw orally for consecutive 28 days |
*NA- not applicable
Atorvastatin is one of the most commonly prescribed statins that effectively lowers cholesterol and improves cardiovascular outcomes. Clinical studies, such as PROVE-IT TIMI 22, demonstrate its superior efficacy over other statins at achieving lipid targets and improving patient outcomes.25 Furthermore, it is commonly used as the standard drug in in vivo antihyperlipidemic studies.26
Animal body weights were recorded on days 1, 28, and 56 of the study. After the treatment period, rats were euthanized in accordance with CCSEA guidelines, and blood samples were collected from the retro-orbital plexus for biochemical analysis, including cholesterol, triglycerides, high-density lipoprotein (HDL), and LDL levels.
Statistical analysis
All data were expressed as mean ± standard error of mean (SEM). Statistical analysis was conducted with ANOVA followed by Tukey’s post-hoc test in SPSS v20.0, with a significance level of p< 0.05.
Results
Phytochemical screening:
The qualitative screening of BPE revealed a rich phytochemical profile that included saponins, flavonoids, carbohydrates, glycosides, alkaloids, phytosterols, phenols, and terpenoids (Table 1). The presence of both non-reducing and reducing sugars were also confirmed by the Molisch’s test and Fehling’s test, respectively. However, tannins were absent as indicated by Ferric chloride test. Overall, this finding signified the presence of several pharmacologically-relevant phytochemicals in BPE, making it a potential candidate for further therapeutic and nutraceutical exploration.
Table 1: Qualitative phytochemical screening of BPE
| Phytochemicals | Screening method | Test result |
| Tannins | Ferric chloride test | Negative |
| Saponins | Foam test | Positive |
| Flavonoids | Alkaline reagent test | Positive |
| Carbohydrates | Molisch’s test | Positive |
| Fehling’s test | Positive | |
| Glycosides | Keller Kiliani test | Positive |
| Alkaloids | Mayer’s test | Positive |
| Dragendorff’s test | Positive | |
| Wagner’s test | Positive | |
| Hager’s test | Positive | |
| Phytosterols | Salkowaski test | Positive |
| Phenols | Ferric chloride test | Positive |
| Terpenoids | Salkowaski test | Positive |
The flavonoid and phenolic content of BPE as indicated by the qualitative analysis were further quantified with Quercetin and Gallic acid standards, respectively. From the standard curve of Quercetin [y = 0.0058x, correlation coefficient (R2) of 0.9981], the flavonoid content was quantified as 160.34 ± 6.67 mg QE (Quercetin equivalents)/ g of extract. Similarly, the phenolic content was represented in terms of GAE with the standard curve equation (y = 0.0096x, R2= 0.9963). The phenolic content was quantified as 63.85 ± 5.41 mg GAE/ g of extract.
In vitro Antioxidant activity
DPPH radical scavenging activity
DPPH radical scavenging assay revealed a significant dose-dependent antioxidant property of BPE. The extract achieved 50% inhibition of DPPH radicals at a concentration of 88.11 ± 6.23 μg/mL.
Reducing power assay
As illustrated in figure 1, BPE also revealed a dose-dependent reducing power activity. Higher absorbance reflects greater electron-donating ability and stronger reducing capacity of the extract.
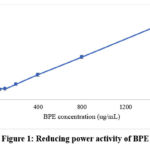 |
Figure 1: Reducing power activity of BPE |
Antimicrobial property
The antimicrobial activity of BPE was assessed against E. coli with the help of the agar well diffusion method. The extract demonstrated a concentration-dependent antibacterial effect, with inhibition zones measuring 1.98 cm, 2.25 cm, and 2.45 cm at 250, 500, and 1000 µg/mL, respectively (Figure 2). Although the activity was lower as compared to the standard antibiotic Ceftriaxone (3.0 cm at 125 µg/mL), the results indicate the presence of potent bioactive compounds in BPE.
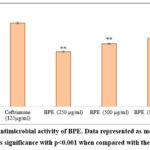 |
Figure 2: Antimicrobial activity of BPE. Data represented as mean ± SEM. ** indicates significance with p<0.001 when compared with the standard. |
In Vivo Antihyperlipidemic Study
The test extracts reported no mortality even at a dose of 2000 mg/Kg, suggesting that the median lethal dose (LD₅₀) exceeds 2000 mg/Kg. The present study further estimated the antihyperlipidemic potential of BPE in HFD-induced rats. The body weight data show significant weight gain in the HFD control group, confirming successful hyperlipidemia induction. BPE-treated groups (200 and 400 mg/Kg) showed controlled weight gain compared to the HFD group, indicating potential antihyperlipidemic effects. BPE400 was more effective, approaching the standard drug (Atorvastatin, 10 mg/Kg) in limiting weight increase by day 56 (Figure 3).
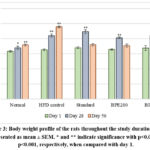 |
Figure 3: Body weight profile of the rats throughout the study duration. Data represented as mean ± SEM. * and ** indicate significance with p<0.05 and p<0.001, respectively, when compared with day 1. |
Table 2: Effect of BPE on Total cholesterol, Triglycerides, HDL, LDL in rats.
| Group | Cholesterol | Triglycerides | HDL | LDL |
| Normal | 70.23 ± 2.02** | 71.03 ± 4.22** | 44.20 ± 0.72** | 11.82 ± 1.44** |
| HFD control | 149.80 ± 2.92## | 208.93 ± 18.11## | 24.22 ± 0.45## | 83.79 ± 6.31## |
| Standard (Atorvastatin, 10mg/Kg) | 78.37 ± 5.35** | 115.52 ± 2.57** | 39.32 ± 2.23** | 15.94 ± 6.27** |
| BPE200 | 121.05 ± 7.75*## | 203.33 ± 2.00## | 31.05 ± 2.26 | 49.33 ± 10.40*# |
| BPE400 | 100.18 ± 5.15** | 151.79 ± 11.45* | 37.68 ± 2.65** | 32.14 ± 4.79** |
Values expressed as mean ± SEM (n=6). * and ** indicate significance at p<0.05 and p<0.01, respectively, when compared to the disease control group, whereas # and ## indicate significance at p<0.05 and p<0.01, respectively, when compared to the standard group.
The HFD control group demonstrated a significant increase in total cholesterol levels compared to the normal group (p < 0.01), confirming successful induction of hyperlipidemia. The standard group treated with Atorvastatin significantly reduced total cholesterol levels (p < 0.01) in comparison to the HFD control, demonstrating its efficacy. The BPE200 group revealed a moderate but significant reduction (p < 0.05), though levels remained higher than in the standard group (p < 0.01), indicating partial lipid improvement. The BPE400 group showed a greater reduction (p < 0.01), nearly comparable to the standard, and not significantly different from normal, suggesting dose-dependent antihyperlipidemic effects (Table 2).
BPE significantly lowered triglyceride levels in hyperlipidemic rats. Both doses showed improvement over the HFD control, with BPE400 showing reductions comparable to the standard (Table 2). HDL levels were significantly reduced in the HFD group (p < 0.01), indicating dyslipidemia. BPE400 restored HDL levels to near-standard values. Both Atorvastatin and BPE reduced elevated LDL levels in the HFD group, with BPE400 demonstrating greater efficacy than BPE200, highlighting its dose-responsive lipid-lowering potential.
Discussion
Banana is a widely used fruit that is consumed for its nutritious value. Banana peel is often discarded as waste and dumped in the environment without proper treatment. However, research has highlighted several beneficial aspects of this waste product. The traditional application of peel has been reported for several ailments, including cough, burns, inflammation, snakebite, excessive menstruation, anemia, diabetes, diarrhea, and ulcers.27 These studies reflected the significant potential of this waste item in pharmaceutical industry. The present study thus aimed to explore the antioxidant, antimicrobial, and antihyperlipidemic activities of hydroalcoholic extract of banana peel.
The phytochemical screening of BPE claimed the presence of several biologically active phytoconstituents such as saponins, flavonoids, carbohydrates, glycosides, alkaloids, phytosterols, phenols, and terpenoids. These phytoconstituents are well-documented for their lipid-lowering and antioxidant effects. The flavonoid and phenolic content of BPE, quantified using quercetin and gallic acid standard curves, were found to be 160.34 ± 6.67 mg QE/g and 63.85 ± 5.41 mg GAE/g of extract, indicating a substantial presence of these phytochemicals. Phenolic and flavonoid compounds are plant-based antioxidants that stabilize reactive oxygen species, thereby preventing oxidative damage. Their presence helps interrupt harmful cellular chain reactions and offers various health benefits, including anti-diabetic, anti-cancer, cardio-protective, anti-aging, and neuroprotective effects, enhancing the body’s overall antioxidant defense.18 These findings were in line with the phyto-composition of kapok banana peel.28
Overproduction of reactive oxygen species (ROS) leads to oxidative stress, which is a key contributor to the pathophysiology and progression of chronic diseases, such as hyperlipidemia, diabetes, cardiovascular diseases, and metabolic syndromes. Research has emphasized the role of dietary antioxidants in combating this ROS production, thereby minimizing oxidative stress, and ultimately preventing these chronic health problems. Secondary metabolites in plants are known for their antioxidant potential. The present study also revealed significant dose-dependent antioxidant properties of BPE via DPPH scavenging and reducing power assays.27 The IC50 value for DPPH scavenging was found to be 88.11 ± 6.23 μg/mL, whereas literature shows, the value is 243.45 μg/mL for Musa sapientum.29 Similar antioxidant activity was also reported in peel extracts of Musa acuminata.30 The antioxidant potential of BPE was further compared to that of the standard antioxidants like ascorbic acid and quercetin. As reported in literature, the DPPH scavenging potential of ascorbic acid was 41.25 μg/mL31, whereas that of quercetin was 1.84 μg/mL, indicating better antioxidant potential as that of the extract.32 Dose-dependent increase in the reducing power activity of BPE was comparable to that observed in case of both ascorbic acid and quercetin.33
Antimicrobial property against E. coli was also found to be promising for BPE which was in accordance with the study by Kapadia et. al.34 Antimicrobial property was evident against Gram-negative bacteria- P. gingivalis and A. actinomycetemcomitans. Antimicrobial property was also reported in other Gram-positive and Gram-negative species.35 The presence of several phytochemicals is presumed to be responsible for this antimicrobial activity of BPE. These findings suggest that BPE possesses promising antibacterial properties and could serve as a natural alternative or complementary agent in antimicrobial therapies.
Hyperlipidemia— characterized by increased triglycerides and LDL— is associated with obesity, playing a critical role in the pathogenesis of atherosclerosis and cardiovascular disease risk. High triglyceride and LDL in blood promotes the development of arterial plaque formation that aggravates the risk of myocardial infarction. Elevated HDL level impairs cholesterol clearance and further accelerates cardiovascular progression. These lipid abnormalities are central to metabolic dysfunction and atherosclerotic disease in obesity. Various studies have indicated the antioxidant property of phytochemicals are effective in the management of hyperlipidemia.36,37 BPE was therefore explored to estimate its antihyperlipidemic activity. BPE demonstrated a dose-dependent hypocholesterolemic effect in HFD-induced hyperlipidemic rats. BPE could also effectively reduce the triglyceride and LDL levels and restore the HDL levels. The 400 mg/Kg dose of BPE (BPE400) was particularly effective, approaching the lipid-lowering efficacy of atorvastatin. These results indicate that BPE may serve as a promising natural therapeutic option for the management of hyperlipidemia. Literature studies have reported the effect of kapok banana peel in successfully restoring dyslipidemia in diabetic animals.38 Furthermore, studies with Musa acuminata peel extract also reported effective reduction of total cholesterol in obese mice.39 However, in contradiction to this finding, treatment with 50% sonicated ethanolic banana peel extracts revealed no significant improvement in the lipid profile of diabetic rabbits, although a reduction in the total cholesterol level was noted.40 This antihyperlipidemic activity of banana peel could be attributed to the presence of secondary metabolites. Research establishes the role of flavonoids in reducing LDL levels by modulating LDL receptor expression.41 Further, isoflavones, flavones, and flavanones help lower blood cholesterol levels by inhibiting its production in the body.42
The pharmacological activity of BPE elucidated in this study could be mechanistically attributed to its rich phytoconstituent profile. High phenolic and flavonoid content prevented lipid peroxidation and oxidative damage to biomolecules, thereby contributing to the antioxidant activity of BPE. The antimicrobial property of BPE may result from phytochemicals such as alkaloids, flavonoids, and saponins either by disrupting microbial cell wall integrity, altering membrane permeability, or inhibiting key microbial enzymes, ultimately leading to growth inhibition of bacteria. The antihyperlipidemic effect of BPE, as observed in HFD-induced rats, could be associated to the ability of flavonoids and sterols to modulate lipid metabolism by enhancing LDL receptor expression, inhibiting cholesterol biosynthesis, or promoting bile acid excretion, thereby lowering circulating LDL and triglyceride levels while restoring HDL levels.10,11 Together, these mechanisms provide a plausible explanation for the antioxidant, antimicrobial, and antihyperlipidemic activities demonstrated by BPE in the present study.
There were certain limitations of the present study. Antimicrobial activity was screened only against Escherichia coli, and therefore the spectrum of activity against other Gram-positive and Gram-negative pathogens remains unexplored. Moreover, the antihyperlipidemic study focused on biochemical endpoints without detailed mechanistic elucidation of lipid metabolism pathways, which limits deeper insights into the mode of action.
Future studies should aim to investigate the underlying molecular mechanisms involved in its antihyperlipidemic and antioxidant effects, and expand antimicrobial testing against a broader range of clinically relevant microorganisms. Furthermore, well-designed preclinical and clinical trials are warranted to validate its safety, efficacy, and therapeutic potential in human populations.
Conclusion
Both in vitro and in vivo studies highlight that banana peel extract exhibits notable antioxidant, antimicrobial, and antihyperlipidemic activities, likely due to its abundance of bioactive compounds, such as flavonoids and phenolics. Nonetheless, further investigations, including mechanistic studies and well-designed clinical trials, are required to validate these findings and establish the therapeutic potential of banana peel as a cost-effective, eco-friendly alternative.
Acknowledgment
The authors would like to acknowledge JIS University for giving the platform to carry on this survey-based research work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants or any material that requires ethical approval. The animal experiments were conducted after getting clearance from the Institutional Animal Ethics Committee of TAAB Biostudy Services (1938/PO/Rc/S/17/CPCSEA).
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Rania Indu was involved in the conceptualization of the project, supervision, data analysis, interpretation, manuscript review, and final approval of the manuscript.
- Hirak Bhowmik, Pritha Sarkar, and Trika Chatterjee performed the experiments in the laboratory and recorded the observations. They also drafted the manuscript.
- Moumita Ray was involved in the conceptualization of the project, supervision, manuscript review, and final approval of the manuscript.
- Dibya Das assisted in the laboratory experiments and drafted the manuscript.
References
- Libby P, Buring JE, Badimon L, et al. Atherosclerosis. Nat Rev Dis Primers. 2019;5:56.
CrossRef - Mach F, Baigent C, Catapano AL, et al. 2019 ESC/EAS guidelines for the management of dyslipidemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2020;41:111-188.
CrossRef - Gupta R, Rao RS, Misra A, Sharma SK. Recent trends in epidemiology of dyslipidemias in India. Indian Heart J. 2017;69:382-392.
CrossRef - Karr S. Epidemiology and management of hyperlipidemia. Am J Manag Care. 2017;23(Suppl 9):S139-S148. PMID: 28978219
- Kang YM, Kim YJ, Kim K. Significance of traditional herbal medicine for dyslipidemia. Am J Transl Res. 2023;15:5373-5388.
- Yadav S, Malik K, Moore JM, et al. Valorisation of Agri-Food Waste for Bioactive Compounds: Recent Trends and Future Sustainable Challenges. Molecules. 2024;29(9):2055.
CrossRef - Pereira A, Maraschin M. Banana (Musa spp) from peel to pulp: ethnopharmacology, source of bioactive compounds and its relevance for human health. J Ethnopharmacol. 2015;160:149-163.
CrossRef - Murata K, Hayashi H, Matsumura S, Matsuda H. Suppression of benign prostate hyperplasia by Kaempferia parviflora rhizome. Pharmacogn Res. 2013;5:309.
CrossRef - Ahmed OM, Abd El-Twab SM, Al-Muzafar HM, et al. Musa paradisiaca L. leaf and fruit peel hydroethanolic extracts improved the lipid profile, glycemic index and oxidative stress in nicotinamide/streptozotocin-induced diabetic rats. Vet Med Sci. 2021;7:500-511.
CrossRef - Prakash B, Kumar A, Singh P, Songachan, L. Antimicrobial and antioxidant properties of phytochemicals. In Elsevier eBooks. 2020;1–45.
CrossRef - Weerawatanakorn M, Kamchonemenukool S, Koh Y, Pan M. Exploring phytochemical Mechanisms in the Prevention of cholesterol Dysregulation: A review. Journal of Agricultural and Food Chemistry. 2024;72(13), 6833–6849.
CrossRef - Dubale S, Kebebe D, Zeynudin A, et al. Phytochemical screening and antimicrobial activity evaluation of selected medicinal plants in Ethiopia. J Exp Pharmacol. 2023;15:51-62.
CrossRef - Asole AG, Sahu MS. Phytochemical screening of crude drugs of Zingiberaceae family. IJPS J. 2024.
- Mujeeb F, Bajpai P, Pathak N. Phytochemical evaluation, antimicrobial activity, and determination of bioactive components from leaves of Aegle marmelos. Biomed Res Int. 2014;2014:497606.
CrossRef - Auwal MS, Saka S, Mairiga IA, Sanda KA, Shuaibu A, Ibrahim A. Preliminary phytochemical and elemental analysis of aqueous and fractionated pod extracts of Acacia nilotica (Thorn mimosa). Vet Res Forum. 2014;5:95-100.
- Farag RS, Abdel-Latif MS, Abd El Baky HH, Tawfeek LS. Phytochemical screening and antioxidant activity of some medicinal plants’ crude juices. Biotechnol Rep. 2020;28:e00536.
CrossRef - Kancherla N, Dhakshinamoothi A, Chitra K, Komaram RB. Preliminary analysis of phytoconstituents and evaluation of anthelminthic property of Cayratia auriculata (in vitro). Maedica (Bucur). 2019;14:350-356.
CrossRef - Singh PK, Singh J, Medhi T, Kumar A. Phytochemical screening, quantification, FT-IR analysis, and in silico characterization of potential bio-active compounds identified in HR-LC/MS analysis of the polyherbal formulation from Northeast India. ACS Omega. 2022;7:33067-33078.
CrossRef - Olugbami JO, Gbadegesin MA, Odunola OA. In vitro free radical scavenging and antioxidant properties of ethanol extract of Terminalia glaucescens. Pharmacogn Res. 2015;7:49-56.
CrossRef - Steglińska A, Bekhter A, Wawrzyniak P, et al. Antimicrobial activities of plant extracts against Solanum tuberosum L. phytopathogens. Molecules. 2022;27:1579.
CrossRef - ARRIVE guidelines (Animal Research: Reporting of In Vivo Experiments). About. Published 2023. Accessed August 31, 2025. http://arriveguidelines.org/about
- OECD. OECD guideline for testing of chemicals: Acute oral toxicity – acute toxic class method. 2001.https://ntp.niehs.nih.gov/sites/default/files/iccvam/ suppdocs/feddocs/oecd/oecd_gl423.pdf
- Venkateshan S, Subramaniyan V, Chinnasamy V, Chandiran S. Antioxidant and anti-hyperlipidemic activity of Hemidesmus indicus in rats fed with high-fat diet. Avicenna J Phytomed. 2016;6:516-525.
- Indu R, Adhikari A, Basak P, Sur TK. Effect of concomitant therapy of anti-diabetics and hypolipidemics on biochemical and histological parameters in animal models. Asian J Pharm Pharmacol. 2019;5:771-778.
CrossRef - Basha A, Ramakrishnan S. Lipid clinical trials with special reference to Indian population. Indian Heart Journal. 2024;76, S130–S137.
CrossRef - Reddy GD, Reddy AG, Rao GS, Kumar MV. Pharmacokinetic interaction of garlic and atorvastatin in dyslipidemic rats. Indian J Pharmacol. 2012;44(2):246-252. doi:10.4103/0253-7613.93860
CrossRef - Zaini HM, Roslan J, Saallah S, et al. Banana peels as a bioactive ingredient and its potential application in the food industry. J Funct Foods. 2022;92:105054.
CrossRef - Savitri D, Djawad K, Hatta M, Wahyuni S, Bukhari A. Active compounds in kepok banana peel as anti-inflammatory in acne vulgaris: Review article. Ann Med Surg. 2022;84:104868.
CrossRef - Mahora M, Kigundu A, Waihenya R. Screening of phytochemicals, antioxidant activity, and in vivo safety profile of the hydroethanolic peel extract of Musa sapientum. Plant Trends. 2024;2:1.
CrossRef - V B, S LK, S RK. Antioxidant and anti-inflammatory properties of the two varieties of Musa acuminata: an in vitro study. Cureus. 2023;15:e51260.
- Matuszewska A, Jaszek M, Stefaniuk D, Ciszewski T, Matuszewski Ł. Anticancer, antioxidant, and antibacterial activities of low molecular weight bioactive subfractions isolated from cultures of wood degrading fungus Cerrena unicolor. PLoS ONE. 2018;13(6): e0197044.
CrossRef - Tian C, Liu X, Chang Y, et al. Investigation of the anti-inflammatory and antioxidant activities of luteolin, kaempferol, apigenin and quercetin. South African Journal of Botany, 2020;137, 257–264.
CrossRef - Priyanga K, Srimathi, Vijayalakshmi K. Investigation of Antioxidant Potential of Quercetin and Hesperidin: An In Vitro Approach. Asian Journal of Pharmaceutical and Clinical Research. 2017; 10(11):83-86.
CrossRef - Kapadia SP, Pudakalkatti PS, Shivanaikar S. Detection of antimicrobial activity of banana peel (Musa paradisiaca L.) on Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans: an in vitro study. Contemp Clin Dent. 2015;6:496-499.
CrossRef - Hikal WM, Said-Al Ahl HAH, Bratovcic A, et al. Banana peels: a waste treasure for human being. Evid Based Complement Alternat Med. 2022;2022:7616452.
CrossRef - Song JH, Kim H, Jeong M, et al. In vivo evaluation of Dendropanax morbifera leaf extract for anti-obesity and cholesterol-lowering activity in mice. Nutrients. 2021;13:1424.
CrossRef - Ebrahimi Y, Hasanvand A, Valibeik A, Ebrahimi F, Abbaszadeh S. Natural antioxidants and medicinal plants effective on hyperlipidemia. Res J Pharm Tech. 2019;12:1457-1462.
CrossRef - Indriawati R, Atiyah F. Antihyperglycemic and hypolipidemic potential of kepok banana peel in diabetic rats. IOP Conf Ser Earth Environ Sci. 2022;985:012040.
CrossRef - Berawi K. N, Bimandama M. A. The Effect of Giving Extract Etanol of Kepok Banana Peel (Musa Acuminata) Toward Total Cholesterol Level on Male Mice (Mus Musculus L.) Strain Deutschland-Denken-Yoken (Ddy) Obese. Biomed Pharmacol J. 2018;11(2).
CrossRef - Chaudhry F, Ahmad ML, Hayat Z, Sajid MW, Qamar MM, Basharat Z, Tariq MR, Bibi A, Alsulami T, Athawab SA, Mugabi R, Alohali BM, Nayik GA. Exploring the anti-diabetic potential of banana peel extracts: Impact of maceration and ultrasonication on bioactive compounds and glycemic control in diabetic rabbits. Ultrason Sonochem. 2025 Jun 11;120:107426..
CrossRef - Bjune K, Halvorsen PS, Wangensteen H, et al. Flavonoids regulate LDLR through different mechanisms tied to their specific structures. J Lipid Res. 2024;65:100539.
CrossRef - Zeka K, Ruparelia K, Arroo RRJ, Budriesi R, Micucci M. Flavonoids and their metabolites: prevention in cardiovascular diseases and diabetes. Diseases. 2017;5:19.
CrossRef
Abbreviations List
ANOVA – Analysis of variance
BPE- Banana peel extract
CCSEA- Committee for Control and Supervision of Experiments on Animals
DPPH- 1,1-diphenyl-2-picrylhydrazyl
GAE- Gallic acid equivalent
HDL- High-density lipoprotein
HFD- High-fat-diet
LD₅₀ – Median lethal dose
LDL- Low-density lipoprotein
OECD- Organisation for Economic Co-operation and Development
QE- Quercetin equivalents
ROS- Reactive oxygen species
SEM- Standard error of mean
SPSS- Statistical Package for the Social Sciences







