Manuscript accepted on :09-09-2025
Published online on: 09-10-2025
Plagiarism Check: Yes
Reviewed by: Dr. Shiv Gunjegaonkar
Second Review by: Dr. Sonam Bhutia
Final Approval by: Dr. Prabhishek Singh
Umamaheswari Duraisamy* , Kumar Mohan
, Kumar Mohan , Anand Sanjay Kamaraj
, Anand Sanjay Kamaraj , Sudharsan Jayasankar
, Sudharsan Jayasankar , Thirumoorthy Poomalai
, Thirumoorthy Poomalai and Vignesh Ezhilarasan
and Vignesh Ezhilarasan
Department of Pharmaceutical Analysis, Vinayaka Mission’s College of Pharmacy, VMRF (DU), Salem, India Corresponding Author E-mail:
DOI : https://dx.doi.org/10.13005/bpj/3316
Abstract
The goal of this study was to create and evaluate dispersible aspirin tablets in order to increase patient compliance, especially for those who have difficulty swallowing regular pills. Aspirin, a widely used analgesic and anti-inflammatory medicine, frequently causes stomach discomfort in its standard form. Dispersible pills have the potential to improve stomach tolerance and have a quicker onset of action. To achieve rapid dispersion in water, relevant excipients such as Disintegrants, binders, and diluents were added to the direct compression method utilised in the tablet and wet granulation procedures. The physicochemical characteristics of the produced tablets, such as weight variation, hardness, friability, disintegration time, and homogeneous drug content, were assessed. After optimization, the tablet characteristics exhibited promising results, releasing more than 85% of the drug after 30 minutes and dispersing in less than 3 minutes. There may be quicker absorption and therapeutic benefits as a result of this rapid disintegration and dissolving profile. Expedited stability testing found no noticeable modifications in the tablet’s chemical and physical features throughout a three-month period. Dispersible aspirin tablets are a potential dose form that provide a practical and efficient substitute for conventional aspirin tablets, according to the study’s findings. This formulation strategy may help increase patient compliance, especially in the case of elderly and paediatric patients, as well as those who have swallowing issues.
Keywords
Aspirin; Disintegration time; Fast dissolving; Oral dispersible tablet; Pre-formulation; Swallowing
Download this article as:| Copy the following to cite this article: Duraisamy U, Mohan K, Kamaraj A. S, Jayasankar S, Poomalai T, Ezhilarasan V. Formulation and Quality Control Test for Fast- Dispersible Aspirin Tablets. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Duraisamy U, Mohan K, Kamaraj A. S, Jayasankar S, Poomalai T, Ezhilarasan V. Formulation and Quality Control Test for Fast- Dispersible Aspirin Tablets. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/48m1AWX |
Introduction
Capsules and tablets are the most used solid dosage forms. Yet, many individuals have serious swallowing problems. Drinking water helps facilitate the swallowing of oral dosage forms. Traditional dosage forms, tablets are often difficult for people to swallow when they are dehydrated, experiencing motion sickness, or suddenly developing a cold, allergies, or bronchitis. Due to these characteristics, tablets that dissolve or disintegrate rapidly in the oral cavity have attracted a lot of interest.1-3 A solid dosage form that dissolves fast on the tongue usually in a few seconds is called an oral dispersible tablet. The term “oral dispersible tablet” was introduced by the European pharmacopoeia to describe these dosage forms and highlight their significance. The term describes a tablet that can be taken orally and quickly dissolves before being swallowed.4
A highly specialised method called drug delivery system (DDS) is used to deliver pharmaceutical chemicals throughout the body in order to provide specific therapeutic effects. In order to ensure that the right amount of medication is delivered in the appropriate location and time within the body, these systems are essential to modern medicine. This approach is crucial for minimizing side effects, maximizing drug efficacy, and improving patient outcomes.5 DDS is essential for improving a medication’s therapeutic index. By regulating the medication release’s rate, time, and place, these systems can significantly improve both pharmacokinetics (the movement of drugs within the body) and pharmacodynamics (the effects of drugs on the body). For instance, traditional oral or injectable formulations may lead to fluctuating drug levels in the bloodstream, which can reduce efficacy and increase side effects. However, advanced DDS can provide sustained and controlled release of the drug, maintaining optimal therapeutic levels over extended periods and minimizing the need for frequent dosing. 6
Drug and Excipients Profile
Aspirin
 |
Scheme 1: AspirinClick here to view Scheme |
Non-proprietary name
Ph Eur, BP, USPNF: Acetylsalicylic acid (ASA)
Synonyms: Acetylsalicylic acid (ASA)Acetyl salicylate, 2-Acetoxybenzic acid, O-Acetylsalicylic acid, Salicylic acid acetate
Chemical name and CAS registry number
Acetylsalicylic acid and 50-78-2
Molecular weight of aspirin: 180.15 g/mol
Function categories: Analgesic, Anti-inflammatory, Antipyretic, Antiplatelet.
Applications in Pharmaceutical Formulation and Technology
Aspirin is commonly formulated as tablets for oral administration, often combined with other ingredients like buffers, fillers, and binders. there can be filled into capsules, providing a convenient and easy-to-swallow dosage form.
Patients who struggle to swallow tablets or capsules, whether young or old, can utilise the aspirin liquids.
This is used in injectable formulations for acute pain management or anti-inflammatory treatment and it is incorporated into creams, gels, or patches for localized pain relief and anti-inflammatory effects and formulated as sustained-release or extended-release tablets to provide prolonged pain relief.7-9
Materials and Methods
For the formulation and assessment experiments, the following chemicals and tools were employed.
Table 1: List of Materials
| Materials used | Category | Manufacture/supplier |
| Aspirin | Active pharmaceutical ingredient | Zhenjiang Gaopeng Pharmaceutical |
| Sodium Starch Glycolate, Croscarmellose sodium, Crospovidone | Super disintegrant | JRS Pharma |
| Mannitol | Sweetener | Tate & Lyle |
| Microcrystalline cellulose | Diluents | Lactose India Limited |
| D. Sorbitol | Sweetener & stabilizer | Tate & Lyle |
| Talc | Glidant | Mondo Minerals |
| Magnesium Stearate | Lubricant | Perrigo |
Tablet 2: List of formulation related Equipment
| S. No | List of equipment | Manufacturer/ supplier |
| 1 | Tablet compression machine | Cadmach, Ahmedabad, India. |
| 2 | Digital weight Balance | Radwag |
| 3 | Hot air Oven | Thermo Scientific |
| 4 | Rapid Mixer Granulator | Medipharma |
| 5 | Disintegration tester | Electrolab |
| 6 | Friability test apparatus | Campbell electronics, Mumbai |
Preparation of standard stock solution
A 50 ml volumetric flask was taken with 50 mg of aspirin. Distilled water was used to dissolve up to the required level. This yields a stock solution with a concentration of 100 mg/ml; 5 ml was taken out and diluted to obtain 50ml of concentration (10 mg/ml).10,11
Standard curve preparation of Aspirin
A Concentration of 1 to 10 mg/ml was obtained by withdrawing aliquots ranging from 1 to 10 ml from the standard solution stock solution and adding distilled water up to 10 ml. We measured the absorbance of these solutions at 275 nm.
Preformulation Studies
Pre- formulation analysis refers to pharmaceutical and analytical study conducted with the intention of supporting efforts to improve the drug substance’s dosage form through formula improvement. Pre- formulation produces the basic knowledge needed to expand on the right formula for toxicological use. In addition to providing body paints for the medication mixture with the pharmaceutical excipients inside the dosage form, it supplies the statistics required to characterise the medicinal ingredient. Therefore, using the drug sample that was collected, the following pre-formulation investigations were carried out. 12
Organoleptic characters
This includes analysing the drug’s colour, flavour, and odour. A record of the drug’s colour is particularly helpful in choosing the right batches.
Bulk Density (Db)
It is ratio of the bulk quantity of powder to the total mass of powder. It was measured by adding the load powder, which had been passed through a general sieve #20- to a measuring cylinder and recording the starting weight. It is provided by and expressed as g/ml.
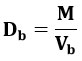
The results were mentioned in the table: 09
Tapped Density (Dt)
It represents the ratio of the powder’s total mass to its tapped quantity. The volume of the powder was determined by tapping it 750 times, and if the volumes differed by less than 2%, the tapped quantity was recorded. If the proportion exceeds 2%, tapping is repeated 1250 times, and the volume tapped is recorded. Tapping was continued until there was less than 2% difference between each volume (in a bulk density apparatus).
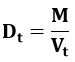
The results were mentioned in the table: 09
Angle of Repose (θ)
The angle of repose can be used to determine the friction forces in a loose powder. It signifies the powder’s waft residences. It is said to be the highest possible angle between the horizontal plane and the powder pile floor.
tan θ= h / r
tan θ= h / r
θ = tan-1 (h / r)
The powder aggregate was able to consistently float through the funnel and drop at a precise height on a platform (h). The angle of repose was then established by estimating the radius and peak of the created powder heap.13
The results were mentioned in the table: 9
Table 3: Angle of Repose of Powder of Flow Properties.
| Flow | Angle of repose |
| Excellent | 25-30 |
| Good | 30-35 |
| Fair | 35-40 |
| Poor | 40-45 |
| Very poor | 45-50 |
Carr’s index (or) % compressibility
It displays the features of powder flow. It is given and stated as a percentage.
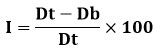
Table 4: Relationship between % compressibility and flow ability
| Flow | Carr’s index |
| Excellent | 5-15 |
| Good | 12-16 |
| Fair | 18-21 |
| Poor | 23-35 |
| Very poor | 35-38 |
| Extremely poor | More than 40 |
The results were mentioned in the table: 9
Hausner ratio:
The Hausner ratio is a crude measure of powder flow easiness. The following formula is used to calculate it.
Hausner ratio =Dt/Db
Where,
Dt is the tapped density.
Db is the bulk density.
Better flow characteristics are indicated by a lower Hausner ratio (<1.25) than by a higher one (>1.25).
The results were mentioned in the table: 9
Table 5: MANUFACTURING METHOD – formulation data for aspirin oral dispersible tablet with different Super Disintegrants
| Ingredients | F1(mg/tab) | F2(mg/tab) | F3(mg/tab) | F4(mg/tab) | F5(mg/tab) | F6(mg/tab) |
| Aspirin(mg) | 150 | 150 | 150 | 150 | 150 | 150 |
| Micro crystalline cellulose(mg) | 40 | 40 | 40 | 30 | 30 | 30 |
| Sodium starch glycolate(mg) | 30 | – | – | 40 | – | – |
| Crospovidone(mg) | – | 30 | – | – | 40 | – |
| Croscarmellose sodium(mg) | – | – | 30 | – | – | 40 |
| D-sorbitol(mg) | 20 | 20 | 20 | 20 | 20 | 20 |
| Mannitol(mg) | 4 | 4 | 4 | 4 | 4 | 4 |
| Talc(mg) | 3 | 3 | 3 | 3 | 3 | 3 |
| Magnesium stearate(mg) | 3 | 3 | 3 | 3 | 3 | 3 |
| Total | 250 | 250 | 250 | 250 | 250 | 250 |
* Mean ± SD (n=6)
Direct compression technique
As specified in the table, precisely weigh the materials.
Microcrystalline cellulose, sodium croscarmellose, crospovidone, mannitol, D-sorbitol, and sodium starch glycolate should be passed through sieve number 60 and combined for five minutes in a plastic bag.
Talc and magnesium stearate are used to lubricate the powder mixture, which is mixed for 20 minutes. It’s prepared for compression.
The mixture is compressed on a lab press machine using a multiple tolling twenty station single rotary with a single pouch (8mm) to make round-shaped tablets. Every tablet has a weight of 250 mg. In order to obtain tablets with a hardness of 3-5 kg/cm2, the compression force is adjusted.14,15
Evaluation of Oral Dispersible Tablets 16
In process parameters evaluation
Weight variation test
20 tablets were chosen at random from the batch and each one was weighed separately to look for variations in weight. Specification for weight variation according to IP.
Table 6: Weight Variation Specification as per IP
| S. No | % Deviation | Average weight of tablet |
| 1 | ±10 | 80 mg or less |
| 2 | ±7.5 | More than 80 mg but less than 250mg |
| 3 | ±5 | 250 mg or more |
Hardness test
The hardness or tablet crushing strength (fc), or the force required to fracture a tablet in a diametric compression, was measured with a Monsanto tablet hardness tester. It is stated as kg/cm2.
The results were mentioned in the table: 12
Thickness
A Digimatic vernier calliper was utilised to ascertain the thickness of the tablets (Mitutoya, Japan). For every batch, three pills were utilised, and average results were determined.
The results were mentioned in the table: 12
Friability (F)
Using the Roche friabilator, the tablet’s friability was assessed. In a plastic cylinder that rotates at 25 rpm and drops a tablet six inches in height with each rotation, this gadget combines the forces of shock and abrasion on the tablet. 100 revolutions were applied to a pre-weighted sample of tablets in the friabilator. After being reweighed, the tablets were dusted with a gentle muslin cloth. With this formula, we may find the friability (F).

Percentage friability and weight loss must both meet acceptance standards of less than 1%.
The results were mentioned in the table: 12
Finished product parameter
Disintegration time testing
With 900ml of pure water without a disc at room temperature, the USP tablet disintegration test instrument was used to determine it. Six tablets were used for the test. The maximum disintegration time of 30 seconds is the limit specified.
Wetting time of tablet
The porosity, compressibility, and absorption capacity of the tablets are all determined by the wetting time test. Wetting times can be measured as an additional confirmatory test for tablet evaluation, Since the dissolution process of a tablet is dependent on tablet wetting followed by tablet disintegration.
The results were mentioned in the table: 12
Water absorption Ratio
A piece of tissue paper folded twice was placed in a tiny Petri dish containing 6 mL of water. After placing a tablet on the paper, the time it took for it to get entirely wet was recorded. We then weighed the wetted pill. Equation (a) was used to get the water absorption ratio (R).
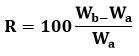
The results were mentioned in the table: 12
Stability Studies
A drug is deemed stable if its physical properties have not changed considerably or negatively, and its chemical or biological activity has not fallen below a predetermined level of marked potency during a time frame beginning with the date of production and formulation packaging. The stability of the active component must be a major factor in the design and evaluation of drug dosage forms before deciding whether to approve or reject them. A drug is deemed stable if its physical properties have not changed considerably or negatively, and its chemical or biological activity has not fallen below a predetermined level of marked potency during a time frame beginning with the date of production and formulation packaging. The stability of the active component must be a major factor in the design and evaluation of drug dosage forms before deciding whether to approve or reject them.
Objective of the study
Stability testing is done to forecast how a drug’s ingredient or product will behave over time in response to various environmental factors like temperature, humidity, and light. This allows for suggested storage conditions, intervals between tests, and shelf lives. Generally speaking, it takes a while to observe how quickly a product deteriorates at room temperature. The expedited stability study principles are used in order to prevent this unfavourable delay.
Table 7: ICH guidelines for stability study
| Time period | Study | Storage condition |
| 12 month | Long term | 250C ± 20C/60% RH ± 5RHOR
300C ± 20C/65% RH ± 5%RH |
| 6 month | Intermediate | 300C ± 20C/65% RH ± 5%RH |
| 3 month | Accelerated | 400C ± 20C/75% RH ± 5%RH |
The stability investigation for the optimised formulation was conducted at 400C±20C/75% RH±5 % RH for one month.
The results were mentioned in the table: 13
Results
Pre-formulation Studies of pure drug and excipients
Pre-formulation analysis refers to pharmaceutical and analytical study conducted with the intention of supporting efforts to improve the drug substance’s dosage form system. It provides the documentation required to describe the nature of the drug ingredient and provides body paintings for the drug mixture with pharmaceutical users inside the dosage form. Therefore, using the drug sample that was collected, the following pre-formulation investigations were carried out. Pre-formulation study parameters were computed using the method described in Section 5.5 of the experimental work.
Table 8: Pre-formulation Study of Pure Drug (Aspirin).
| Parameters | Result | Conclusion |
| Bulk Density (gm/cm3) | 0.255 | —— |
| Tapped Density (gm/cm3) | 0.334 | —– |
| Angle of Repose (θ) | 300 40’ | Excellent |
| Carr’s Index (%) | 23.6 | Excellent Flow |
| Hausner Ratio | 1.11 | Better Flow |
| Melting Point | 119-120oC | —- |
| Solubility | Freely soluble in methanol, ethanol, chloroform, very slightly soluble in water. | |
To guarantee efficient formulation, aspirin’s physicochemical characteristics, stability, and compatibility with excipients and packaging materials are assessed in pre-formulation research. Solubility, melting point, polymorphism, particle size, and chemical stability are important factors.
Table 9: Pre-formulation Study of the blend
| BatchCode | F1 | F2 | F3 | F4 | F5 | F6 |
| BulkDensity (gm/cm3) | 0.252 | 0.223 | 0.232 | 0.233 | 0.242 | 0.241 |
| Tapped Density (gm/cm3) | 0.335 | 0.344 | 0.333 | 0.333 | 0.354 | 0.334 |
| Angle of repose (θ) | 310 14’ | 300 44’ | 290 34’ | 290 24’ | 320 33’ | 310 19’ |
| %Compressibility | 24.77 | 26.20 | 26.22 | 26.40 | 26.80 | 25.20 |
| Hausner’sRatio | 1.32 | 1.36 | 1.35 | 1.30 | 1.29 | 1.28 |
Mean ±SD (n=6)
Discussion
Based on the flow ability scale, the medication has good flow characteristics. The excipients had no impact whatsoever on the blend’s flow. It chose to use the direct compression method as a result.
Determination of UV absorption maxima of Aspirin
Using a double beam UV/VIS spectrophotometer, UV scanning was performed for a 10 mg/ml drug solution from 200–400 nm in methanol as a blank. The wavelength at which the maximum was discovered was 275 nm. (Figure 1).
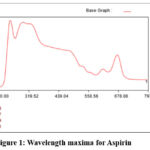 |
Figure 1: Wavelength maxima for AspirinClick here to view Figure |
Calibration curve
Aspirin at varying concentrations (1 to 10 µg/ml) was produced using distil water and subjected to UV analysis at 25 nm, with matching media serving as a blank. In the 1 to 10 µg/ml range, the absorbance complies with the Beers-Lamberts law. The data are shown in Table 10 and Figure 2.
Table 10: Standard calibration curve of Aspirin
| Concentration(µg/ml) | Absorbance |
| 1 | 0.0090 |
| 2 | 0.032 |
| 3 | 0.0545 |
| 4 | 0.0843 |
| 5 | 0.1052 |
| 6 | 0.1244 |
| 7 | 0.1486 |
| 8 | 0.1666 |
| 9 | 0.1836 |
| 10 | 0.1997 |
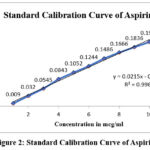 |
Figure 2: Standard Calibration Curve of Aspirin |
Table.11: Regression Analysis
| Parameters | Value |
| R2 | 0.996 |
| Slope | 0.021 |
| Intercept | 0.075 |
Considering the concept of initial identity, it can be inferred that the substance adhered to the original identity. It was also determined by scanning the drug in methanol dissolving fluid that the drug’s maximal wavelength was 275 nm.
Evaluation studies of OD Tablets
Table 12: Evaluation of Post compression parameters of Oral Dissolving tablets of Aspirin
| Test | F1 | F2 | F3 | F4 | F5 | F6 |
| Weight Variation (mg)** | 251±1.15 | 253±1.28 | 254±1.25 | 251±1.35 | 252±1.48 | 253 ± 1.45 |
| Hardness(kg/cm2 )* | 4.96 ±0.136 | 5.02 ±0.125 | 4.89 ±0.154 | 4.69 ± 0.226 | 4.78 ± 0.188 | 4.39 ± 0.12 |
| Thickness(mm)* | 4.51±0.0142 | 4.60±0.0112 | 4.50±0.0112 | 4.33±0.0132 | 4.41±0.0132 | 4.49 ± 0.0133 |
| Friability(%)* | 0.542 | 0.547 | 0.545 | 0.49 | 0.44 | 0.42 |
| Dis-integrationTime
(sec)* |
13 | 17 | 12 | 15 | 14 | 10 |
| WettingTime
(sec)* |
53.2±1.527 | 54.5±1.577 | 51.1±0.577 | 51.1±1.527 | 45.6±1.577 | 15.3 ± 1.527 |
| WaterAbsorption
Ratio* |
64.03±1.577 | 89.06±0.577 | 69.33±0.577 | 75.66±1.527 | 89.01 ± 1.01 | 57.2 ± 1.527 |
| Drug-Content
(%)* |
95.84 | 95.76 | 96.57 | 97.30 | 98.76 | 99.89 |
*Mean±SD (n=6) **Mean±SD (n=20)
Disintegration Time of Formulations F1 to F6
When evaluating oral dissolving tablets, the primary metric to be considered is the disintegration time. The image below visually depicts the effect of four distinct super-Disintegrants and the variances in their functionalities. The combination of two super disintegrates at moderate concentration was found to have the shortest disintegration time among all tablet formulations. It is caused by the water being absorbed quickly—in less than a second—without forming lumps, which results in a burst effect. It was discovered that as the concentration of super-disintergrant increased, disintegration time decreased. According to the type of super-disintergrant, the disintegration time follows the order.
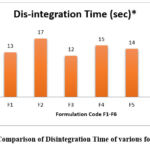 |
Figure 3: Comparison of Disintegration Time of various formulations |
Wetting Time of Formulations F1 to F6
The most crucial factor in oral dissolving tablet formulations is the wetting time. The time it takes for water to reach the tablet’s surface indicates how long it will take for the tablet to become wet. This signifies both the drug’s hydrophilicity and the impact of super-Disintegrants. According to the current study, crospovidone had the shortest wetting time among the several super-Disintegrants. Additionally, a graphic comparison of all the formulations from F1 through F6 is shown in the picture below. In the present investigation, wetting times are lowered for all batches as the super-disintergrant ratio rises. Each formulation has a different wetting time, which is listed in this sequence.
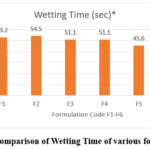 |
Figure 4: Comparison of Wetting Time of various formulations. |
Stability Studies of Oral Dissolving Tablets
The manufacturing company bears the task of ensuring that products arrive to customers in an active state. Pharmaceutical stability is therefore a crucial factor. The capacity of a certain formulation in a given container to maintain its physical, chemical, microbiological, therapeutic, and toxicological specifications is known as drug stability; in other words, a medication’s stability is its resistance to deterioration. The approved potency label requires a minimum potency of 90%. Stability testing is therefore required for pharmaceutical items. The table was below the Table 13.
Table 13: Stability studies of optimized formulation F6 at room temperature 250C.
| Parameters | Drug content (%)* | Disintegration Time (sec.)* | Wetting Time (sec.)* | Hardness (kg/cm2)* | Friability %* |
| Controlled | 99.89 | 10 | 15.3 ± 1.527 | 4.39 ± 0.12 | 0.42 |
| After 15 days | 99.09 | 11 | 15.2 ± 1.517 | 4.28 ± 0.11 | 0.41 |
| After 1 month | 98.79 | 11 | 15.01 ± 1.501 | 4.21 ± 0.01 | 0.37 |
| Time | Cumulative % Drug Release | ||||
| Controlled | After 15 days | After 1 month | |||
| 0 | 0 | 0 | 0 | ||
| 1 | 50.31 | 50.01 | 49.81 | ||
| 3 | 55.01 | 54.81 | 54.21 | ||
| 5 | 77.92 | 77.02 | 76.82 | ||
| 7 | 86.43 | 85.03 | 84.83 | ||
| 10 | 100.05 | 100.00 | 99.70 | ||
| 15 | 100.04 | 100.00 | 99.75 | ||
| 20 | 100.04 | 100.00 | 99.75 | ||
*Mean±SD (n=6)
Conclusion
The dispersible aspirin tablets were successfully formulated with the desired characteristics, including appropriate hardness, disintegration time, and dispersion quality. The use of suitable excipients, such as Disintegrants and binders, was crucial in achieving these properties. Stability studies indicated that the dispersible aspirin tablets maintained their efficacy and quality over the proposed shelf life. The formulation was stable under the tested storage conditions, which supports its potential for long-term use.
Acknowledgement
The author would like to Vinayaka mission’s research foundation for granting the research work. The department of pharmaceutical chemistry, Vinayaka mission’s college of pharmacy, VMRF (DU) for supporting and guiding us. Thank everyone who assisted and encouraged the research, particularly for the authorship.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement-
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author’s Contribution
- Umamaheswari D: wrote the protocol and first draft of the manuscript.
- Kumar Mohan: The study’s analyses.
- Anand sanjay kamaraj and Sudharsan Jayasankar: The literature searches.
- Thirumoorthy Poomalai: Data collection, writing – review and editing.
- Vignesh Ezhilarasan: Data collection, writing – review and editing.
References
- Yoshida T, Kojima H. Oral drug delivery systems applied to launched products: value for the patients and industrial considerations. Molecular Pharmaceutics. 2023;20(11):5312-31.
CrossRef - Kumar ML, Shree KJ, Mounika N, et al. Formulation and quality control of orally disintegrating tablets (ODTs): recent advances and perspectives. World Journal of Pharmaceutical Research. 2024;13(17):288-308.
- Ur Rahman A, Nasir F, Ali Khattak M, et al. Comparative pharmacokinetic evaluation of glimepiride orodispersible and conventional tablets in rabbits. Drug Development and Industrial Pharmacy. 2024;50(2):173-80.
CrossRef - Zawada K, Czarnecka K, Girek M, et al. New hybrids of tacrine and indomethacin as multifunctional acetylcholinesterase inhibitors. Chemical Papers. 2021;75:249-64.
CrossRef - Belayneh A, Tessema Z. A systematic review of the stability of extemporaneous pediatric oral formulations. The Scientific World Journal. 2021;2021(1):8523091.
CrossRef - Li X, Shen M, Yang J, et al. Pillararene-based stimuli-responsive supramolecular delivery systems for cancer therapy. Advanced Materials. 2024;36(16):2313317.
CrossRef - Khope RG, Gotmare SC, Dhoble NN, et al. Intranasal drug delivery: a non-invasive method for improving neurotherapeutic delivery. Asian Journal of Pharmaceutical Research. 2024;14(1):53-61.
CrossRef - Petrovic S, Bita B, Barbinta-Patrascu ME. Nanoformulations in pharmaceutical and biomedical applications: green perspectives. International Journal of Molecular Sciences. 2024;25(11):5842.
CrossRef - Sun Y, Zhang W, Gu J, et al. Magnetically driven capsules with multimodal response and multifunctionality for biomedical applications. Nature Communications. 2024;15(1):1839.
CrossRef - Nagai N, Ogata F, Kadowaki R, et al. Design of an oral tablet containing furosemide nanoparticles with elevated bioavailability. Journal of Oleo Science. 2024;73(4):563-71.
CrossRef - Lee JS, Lee HG. Nano-encapsulation of a combination of clove oil and thymol and their application in fresh-cut apples and raw minced beef. Food Control. 2023;148:109683.
CrossRef - Zhang Z, Wuqiong WA, Zhang Y, et al. Research progress in theoretical calculation of pharmaceutical formulation design. CIESC Journal. 2024;75(4):1429.
- Sharma A, Bhatia D. Programmable bionanomaterials for revolutionizing cancer immunotherapy. Biomaterials Science. 2024;12(21):5415-5432.
CrossRef - Wu S, Lin L, Shi L, et al. An overview of lipid constituents in lipid nanoparticle mRNA delivery systems. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology. 2024;16(4):e1978.
CrossRef - Zhang Y, Zhang X, Gao Y, et al. Principles of lipid nanoparticle design for mRNA delivery. BMEMat. 2024;:e12116.
CrossRef - Shelke MV, Khade MP, Salve MT. Review on formulation and evaluation of bromocriptine mesylate as oral dispersible tablet. International Journal of Sciences and Innovation Engineering. 2025;2(5):884-99.







